11 September 2020: Clinical Research
A Retrospective Study of the C-Reactive Protein to Lymphocyte Ratio and Disease Severity in 108 Patients with Early COVID-19 Pneumonia from January to March 2020 in Wuhan, China
Miao Yang1ACDEG, Xiaoping Chen2ABCF*, Yancheng Xu1ADFGDOI: 10.12659/MSM.926393
Med Sci Monit 2020; 26:e926393
Abstract
BACKGROUND: The aim of this study was to determine the effect of C-reactive protein (CRP), lymphocytes (LYM), and the ratio of CRP to LYM (CRP/LYM) on assessing the prognosis of COVID-19 severity at early stages of disease.
MATERIAL AND METHODS: A total of 108 hospitalized patients diagnosed with COVID-19 in Zhongnan Hospital of Wuhan University from January 17, 2020 to March 12, 2020 were enrolled. Data of demographic parameters, clinical characteristics, laboratory indicators, clinical manifestation, and outcome of disease were collected. The patients were divided into a severe group and a non-severe group according to diagnosis and classification, which followed the guidelines and management of the Chinese National Health Council COVID-19. The receiver-operating characteristic (ROC) analysis and comparison of ROC curves were used for the laboratory findings for assessment of COVID-19 severity.
RESULTS: Of the 108 patients, 42 patients (38.9%) were male and 24 patients (22.2%) were considered severe cases, with the mean age of 51.0 years old. Males and patients with comorbidities were more likely to become severe cases. CRP increased and LYM decreased in the severe group.The results for the areas under the curve (AUC) of CRP/LYM and CRP used to assess severe COVID-19 were 0.787 (95% CI 0.698–0.860, P<0.0001) and 0.781 (95% CI 0.693–0.856, P<0.0001), respectively; both results were better than that of LYM. The associated criterion value of CRP/LYM was calculated, with an excellent sensitivity of 95.83%.
CONCLUSIONS: The effect of CRP/LYM and CRP on the assessment for severe COVID-19 may be superior to LYM alone. CRP/LYM is a highly sensitive indicator to assess the severity of COVID-19 in the early stage of disease.
Keywords: Coronavirus, Lymphocytes, Risk Adjustment, Aged, 80 and over, Area Under Curve, Betacoronavirus, C-Reactive Protein, COVID-19, Coronavirus Infections, cytokine release syndrome, Length of Stay, Lymphocyte Count, Pandemics, Pneumonia, Viral, ROC Curve, SARS-CoV-2, Severity of Illness Index, young adult
Background
A series of unexplained cases of pneumonia in Wuhan were reported to the World Health Organization (WHO) on December 31, 2019, and was followed by a worldwide pandemic of infectious disease. The outbreak of this pandemic came from a new coronavirus called severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), which is the cause of the 2019 coronavirus disease (COVID-19) [1]. In the past 20 years, severe epidemics due to 2 important beta-coronaviruses have resulted in more than 10 000 infections. The coronaviruses were the acute respiratory syndrome coronavirus (SARS-CoV) and the Middle East respiratory syndrome coronavirus (MERS-CoV); the mortality rates were 10% and 42% respectively [2–5]. As of May 16, 2020, as a new member of the coronavirus family, the mortality caused by the SARS-CoV-2 had reached up to 6.8% [6]. Although the overall mortality of COVID-19 is not higher than the former 2 coronaviruses, its clinical infection range seems to be wider and more contagious, including asymptomatic infections, mild patient cases, ordinary patient cases, and severe patient cases [7,8].
Previous studies have reported common clinical features of the COVID-19. Patients may present with fever, dry cough, fatigue, headache, dyspnea, normal or decreased white blood cell count, and imaging evidence of pneumonia. Severe cases are prone to various complications and may further increase the chance of human death [9,10]. Shen et al. [11] suggested that patients with COVID-19 were more likely to progress within 1 week. The early definition of COVID-19 was defined as within 7 days of clinical symptoms, and within 7 days of positive viral nucleic acid test for asymptomatic patients [12]. Accordingly, early assessment and classification for the severity of the disease is essential to improve patients’ clinical outcomes. At the same time, classification can also help clinicians find patients who may have aggravated conditions as soon as possible; it is more beneficial to allocate limited medical resources for people who need more active treatments. Consequently, clinicians need more valuable laboratory biochemical indicators that can help to assess the severity of the disease in the early stage of the infection. In previous articles, the fluctuation of C-reactive protein (CRP) and lymphocytes (LYM) in patients with COVID-19 have gradually gained attention [12–16]. It was found that increased CRP and decreased LYM may be related to the severity of the disease. However, few articles have analyzed and compared the sensitivity and specificity of these 2 parameters for evaluating the severity of the disease.
The present study aimed to determine the effect of laboratory tests, especially CRP, LYM, and the ratio CRP to LYM (CRP/LYM) on assessing the prognosis of COVID-19 severity at early stages. Therefore, we hope to provide new ideas and directions for more accurate, timely, and targeted clinical personalized evaluation of COVID-19 patients.
Material and Methods
STUDY DESIGN AND PARTICIPANTS:
A retrospective study was conducted from January 17, 2020 to March 12, 2020 that included 108 hospitalized patients diagnosed with COVID-19 in Zhongnan Hospital of Wuhan University. Throat-swab specimens were obtained for SARS-CoV-2 re-examination every other day after suspicion of COVID-19. The specimen suspensions were used for real-time reverse transcription polymerase chain reaction (RT-PCR) assay of SARS-CoV-2 RNA, which was performed using ORF1ab/N gene PCR kit (Biogerm, Cat# SJ-HX-226-1,2, Shanghai, China) according to the protocol described previously [9]. Fasting whole blood samples from patients were collected in EDTA anticoagulant-treated tubes and analyzed within 30 minutes. Routine peripheral blood cells, including white blood cells, hemoglobin, platelets, lymphocytes, neutrophils, and monocytes were analyzed by Beckman Coulter DxH 800 automated blood analyzer and related reagents (Beckman, CA, USA). Routine serum biochemical parameters, including alanine aminotransferase (ALT), aspartate aminotransferase (AST), total bilirubin (TBIL), creatinine, and CRP were measured by Beckman Coulter AU automatic biochemical analyzer and related reagents (Beckman, CA, USA). Computed tomography (CT) scan (Discovery, GE Medical Systems, Milwaukee, MN, USA) was performed on patients to assess lung injury. The diagnosis and classification of COVID-19 followed the guidelines and management of the Chinese National Health Council COVID-19 [17]. Briefly, patients were categorized as severe cases according to one of these criteria: a) dyspnea, respiratory rate ≥30 breaths per minute; b) blood oxygen saturation ≤93% at rest; c) PaO2/FiO2 ≤300 mmHg; d) lung injury progresses by more than 50% within 24 to 48 hours from CT imaging.
This study was approved by the ethics board at Zhongnan Hospital of Wuhan University, Wuhan, China (No. 2020011); informed consents were obtained from all patients upon admission to Zhongnan Hospital of Wuhan University.
DATA COLLECTION:
According to the diagnosis, the patients were divided into 2 groups: a severe group and a non-severe group. The following data were collected: demographic data, clinical symptoms, chronic comorbidities, laboratory results at the beginning of admission, clinical manifestations, severity rating, hospital stays, and prognostic outcomes. At least 2 trained staff collected and reviewed data to avoid subjective biases.
STATISTICAL ANALYSIS:
Chi-square tests or Fisher’s exact tests were performed for categorical variables. The normal distribution of measurement data was statistically tested at the first time. When the data was normally distributed,
Results
DEMOGRAPHIC PARAMETERS, CLINICAL CHARACTERISTICS, AND LABORATORY FINDINGS:
A total of 108 patients with COVID-19 hospitalization were included in this study. Among them, 24 patients were severe cases (22.2%) and 42 patients were male (38.9%), with the median age of 51.0 years (IQR, 35.0–66.0; range, 20–96 years). The comorbidities, signs and symptoms, clinical manifestations, hospital stays, clinical outcome, and laboratory results were compared between the non-severe group and the severe group (Tables 1, 2). This revealed that male patients, elderly patients, and patients with comorbidities were more likely to become severe cases. In comparison with the non-severe group, the hospital stays of the severe group were extended to 20.5 days (P=0.001, Table 1). Meanwhile, the clinical outcomes were even worse in the severe group: 5 patients (20.8%) were dead in the severe group, while no patients (0.0%) were dead in non-severe group (P=0.0004, Table 1). From the laboratory results, it was shown that CRP increased and LYM decreased significantly in the severe group (P<0.0001 and P=0.033, respectively, Table 2).
ANALYSIS OF ROC CURVES BY LABORATORY FINDINGS:
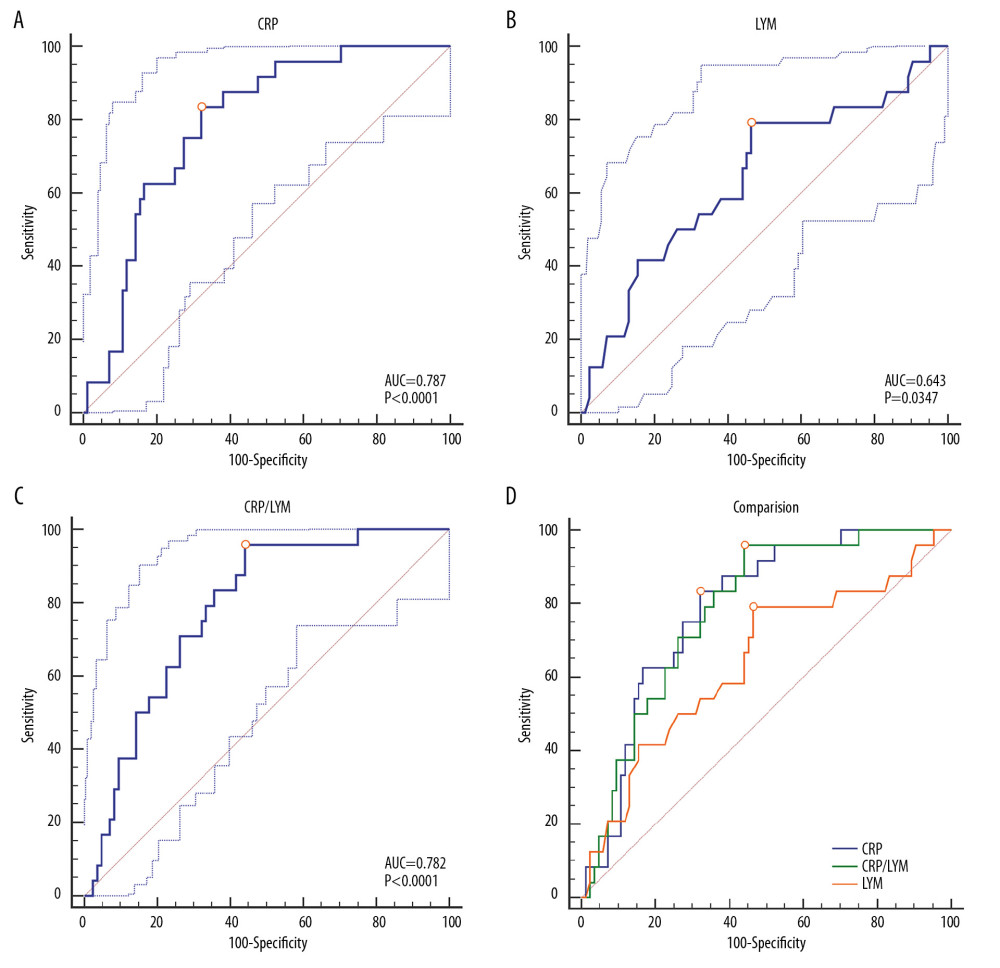
ROC analysis was performed on the laboratory findings of the blood samples collected at the first admission of the patient. All the results were used to evaluate the ability of various biochemical indicators to assess the severity of COVID-19. The areas under the curve (AUC) of different laboratory biomarkers are shown in Figure 1 and Table 3. One of the most interesting findings was that the AUC of CRP was 0.787 (95% CI 0.698–0.860, P<0.0001). Correspondingly, the associated criterion value of CRP in ROC was 26.3 mg/L, with the sensitivity and specificity of 83.33% and 67.86% respectively (Figure 1A, Table 3). Another important finding was that the AUC of LYM was only 0.643 (95% CI 0.545–0.733, P=0.0347), with the sensitivity and specificity of 79.17% and 53.57% respectively, which was not as good as expected (Figure 1B and Table 3). Next, the ratio of CRP to LYM (CRP/LYM) was analyzed, and it was found that its AUC value reached surprisingly to 0.781 (95% CI 0.693–0.856, P<0.0001, Table 3). Especially worth mentioning was that its associated criterion value was 16.62 mg·10−9, with the excellent sensitivity of 95.83% (Figure 1C, Table 3).
COMPARISON OF ROC CURVES FOR ASSESSING SEVERE COVID-19:
As shown in Figure 1D and Table 4, there was no difference between AUC of CRP and CRP/LYM (95% CI 0.039–0.049, P=0.8254). On the other hand, no matter the difference between AUC of CRP and LYM (95% CI 0.009–0.280, P=0.0372) or CRP/LYM and LYM (95% CI 0.04–0.238, P=0.0061) both made sense statistically. It can thus be inferred that the AUC of CRP and CRP/LYM to assess the severity of disease were higher than the LYM score, which proved the outstanding ability of CRP to assess the severity of COVID-19 patients in the early stage.
Discussion
The current study found that males and patients with comorbidities were more likely to become severe cases. Compare to the non-severe group, the hospital stays were extended and the clinical outcomes were even worse in the severe group, which was similar to previous research studies [18]. The outbreak of the COVID-19 pandemic has caused a major impact and a heavy burden on the global health care system [19,20]. Therefore, in the rational use of limited medical resources, it is important to evaluate COVID-19 patients who may further aggravate the disease at an early stage, and this strategy will help to improve the prognosis of patients and control the spread of epidemics. Our results indicated that a significant increase of CRP and a corresponding decrease of LYM were observed in severe patients in the early stage of COVID-19. It was found that CRP/LYM and CRP may be more meaningful for early detection of severe COVID-19 patients than LYM changes alone. Interestingly, CRP/LYM has shown a more prominent advantage, which associated criterion value was 16.62 mg·10−9, with the excellent sensitivity of 95.83%. In other words, CRP/LYM, as a classic but innovative parameter, can assess the severity of COVID-19 in the early stage so as to gain more opportunities for clinical diagnosis and treatment.
In order to assess the effect of different laboratory indicators on the estimation of disease severity, we analyzed the laboratory data of COVID-19 patients at the beginning of admission to hospital. The results of the ROC analysis showed that the AUC of CRP used to assess severe COVID-19 was 0.787 (95% CI 0.698–0.860,
A considerable amount of literature have shown that the distinguished decrease in the number of lymphocytes in patients with COVID-19 may be related to the SARS-CoV-2, which depleted many immune cells and inhibited the immune function of human cells [26–28]. Therefore, lymphocytes were considered to be one of the signs that the course of disease may have worsen recently [29,30]. In our results, similar conclusions were observed. But interestingly, the ROC analysis showed that the AUC of LYM used to assess severe COVID-19 was only 0.643 (95% CI 0.545–0.733,
From this result, it seems like that the increase of CRP is more advantageous than the decrease changes in LYM alone to assessing the degree of disease progression in the early course of COVID-19. It is speculated that the reason for this phenomenon may be related to the significant increase in IL-6. Some scholars pointed out that COVID-19 patients had abnormal immune functions, which was characterized by normal or high cytokine production capacity and increased circulating cytokines, especially elevated IL-6 [31]. As aforementioned, CRP is directly induced by the action of IL-6 in the acute phase of inflammation [22]. Therefore, in the early stage of the disease, the change of CRP may be more rapid and obvious than other biochemical markers. On the other hand, the possible underlying mechanism of lymphocyte deficiency was related to the SARS-CoV-2, which may directly infect lymphocytes, leading to their apoptosis [32]. Early in the course of COVID-19, when the viral load in the body has not reached its peak stage, the changes in lymphopenia may not be obvious as soon as possible. In the data of this study, the LYM value resulted from the first blood collection during hospitalization. Therefore, it may not yet have been reduced more significantly at this stage. Meanwhile, CRP may have shown more obvious changes to help clinicians to evaluate the severity of COVID-19.
Just as with CRP, CRP/LYM showed more advantages in assessing the severity of COVID-19, and was better than LYM changes alone. Especially, the associated criterion value of CRP/LYM was 16.62 mg·10−9, with the excellent sensitivity of 95.83%, which brought greater significance to clinical diagnosis and treatment. We have reason to believe that CRP/LYM can be used as a classic but innovative parameter to evaluate the severity of COVID-19.
To our knowledge, this is the first time that statistical comparisons of these ROC curves were performed to explore the role of CRP/LYM, CRP, and LYM for assessing the severity of COVID-19 early in the disease. Particularly, the effect of CRP/LYM and CRP on the assessment of severe disease transformation may be superior to LYM alone. In addition, we have proposed the associated criterion value of CRP/LYM with an excellent sensitivity. This research examines the emerging position of CRP/LYM in the context for assessing disease progression. This investigation had some limitations. The sample size of the research was limited (the number of patients included was only 108), and the patients were all from the same hospital, thus the data may be biased. Furthermore, due to the limited sample size, we did not conduct relevant subgroup analysis, which may limit the interpretation of the results.
Conclusions
In conclusion, our study found that CRP elevated and lymphocytes decreased significantly in the most severe COVID-19 patients. In the early stage of the disease, the effect of CRP/LYM and CRP on the severity assessment may be superior to lymphocytes alone. Last but not least, CRP/LYM is a classic but innovative parameter. It is recommended that all COVID-19 patients should complete this test at their first assessment.
References
1. Zhu N, Zhang D, Wang W, China novel coronavirus investigating and research team. A novel coronavirus from patients with pneumonia in China, 2019: N Engl J Med, 2020; 382(8); 727-33
2. Kuiken T, Fouchier RA, Schutten M, Newly discovered coronavirus as the primary cause of severe acute respiratory syndrome: Lancet, 2003; 362(9380); 263-70
3. Drosten C, Günther S, Preiser W, Identification of a novel coronavirus in patients with severe acute respiratory syndrome: N Engl J Med, 2003; 348(20); 1967-76
4. de Groot RJ, Baker SC, Baric RS, Middle East respiratory syndrome coronavirus (MERS-CoV): Announcement of the Coronavirus Study Group: J Virol, 2013; 87(14); 7790-92
5. White J, Middle Eastern Respiratory Syndrome Coronavirus (MERS-CoV): Clin Microbiol Newsl, 2014; 36(15); 115-22
6. WHO: Coronavirus disease (COVID-19) outbreak situation Available at https://www.who.int/emergencies/diseases/novel-coronavirus-2019
7. Zhu J, Ji P, Pang J, Clinical characteristics of 3,062 COVID-19 patients: A meta-analysis: J Med Virol, 2020; 1-13
8. Wang R, Pan M, Zhang X, Epidemiological and clinical features of 125 Hospitalized Patients with COVID-19 in Fuyang, Anhui, China: Int J Infect Dis, 2020; 95; 421-28
9. Wang D, Hu B, Hu C, Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China: JAMA, 2020; 323(11); 1061-69
10. Chen N, Zhou M, Dong X, Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: A descriptive study: Lancet, 2020; 395(10223); 507-13
11. Shen C, Tan M, Song X, Comparative analysis of early-stage clinical features between COVID-19 and influenza A H1N1 virus pneumonia: Front Public Health, 2020; 8; 206
12. Wang L, C-reactive protein levels in the early stage of COVID-19: Med Mal Infect, 2020; 50(4); 332-34
13. Li R, Tian J, Yang F, Clinical characteristics of 225 patients with COVID-19 in a tertiary Hospital near Wuhan, China: J Clin Virol, 2020; 127; 104363
14. Chen W, Zheng KI, Liu S, Plasma CRP level is positively associated with the severity of COVID-19: Ann Clin Microbiol Antimicrob, 2020; 19(1); 18
15. Tan C, Huang Y, Shi F, C-reactive protein correlates with computed tomographic findings and predicts severe COVID-19 early: J Med Virol, 2020; 92(7); 856-62
16. Ponti G, Maccaferri M, Ruini C, Biomarkers associated with COVID-19 disease progression: Crit Rev Clin Lab Sci, 2020 [Online ahead of print]
17. : Diagnosis and treatment protocols of pneumonia caused by new coronavirus (trial version 6), revised edition, National Health Commission of the People’s Republic of China Available at http://www.nhc.gov.cn/yzygj/s7652m/202002/54e1ad5c2aac45c19eb541799bf637e9.shtml
18. Zhou F, Yu T, Du R, Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study: Lancet, 2020; 395(10229); 1054-62
19. Ripp J, Peccoralo L, Charney D, Attending to the emotional well-being of the health care workforce in a New York City health system during the COVID-19 pandemic: Acad Med, 2020 [Online ahead of print]
20. Colenda CC, Applegate WB, Reifler BV, Blazer DG: COVID-19: Financial stress test for academic medical centers, 2020 [Online ahead of print]
21. Wang G, Wu C, Zhang Q, C reactive protein level may predict the risk of COVID-19 aggravation: Open Forum Infect Dis, 2020; 7(5); ofaa153
22. Nehring SM, Goyal A, Bansal P, Patel BC: C-reactive protein (CRP), 2020, Treasure Island (FL), StatPearls Publishing
23. Xu B, Fan CY, Wang AL, Suppressed T cell-mediated immunity in patients with COVID-19: A clinical retrospective study in Wuhan, China: J Infect, 2020; 81(1); e51-60
24. Chau VQ, Oliveros E, Mahmood K, The imperfect cytokine storm: Severe COVID-19 with ARDS in patient on durable LVAD support: JACC Case Rep, 2020 [Online ahead of print]
25. Cossarizza A, Gibellini L, De Biasi S, Handling and processing of blood specimens from patients with COVID-19 for safe studies on cell phenotype and cytokine storm: Cytometry A, 2020 [Online ahead of print]
26. Huang C, Wang Y, Li X, Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China: Lancet, 2020; 395(10223); 497-506
27. Yun H, Sun Z, Wu J, Laboratory data analysis of novel coronavirus (COVID-19) screening in 2510 patients: Clin Chim Acta, 2020; 507; 94-97
28. Qin C, Zhou L, Hu Z, Dysregulation of immune response in patients with COVID-19 in Wuhan, China: Clin Infect Dis, 2020 [Online ahead of print]
29. Zhang C, Zhang L, Chen X, Decreased “WBC*LYM” was observed in SARS-CoV-2-infected patients from a fever clinic in Wuhan: Clin Chem Lab Med, 2020; 58(7); 1152-55
30. Lovato A, de Filippis C, Clinical presentation of COVID-19: a systematic review focusing on upper airway symptoms: Ear Nose Throat J, 2020 [Online ahead of print]
31. Giamarellos-Bourboulis EJ, Netea MG, Rovina N, Complex immune dysregulation in COVID-19 patients with severe respiratory failure: Cell Host Microbe, 2020; 27(6); 992-1000.e3
32. Tan L, Wang Q, Zhang D, Lymphopenia predicts disease severity of COVID-19: A descriptive and predictive study: Signal Transduct Target Ther, 2020; 5(1); 33
Tables
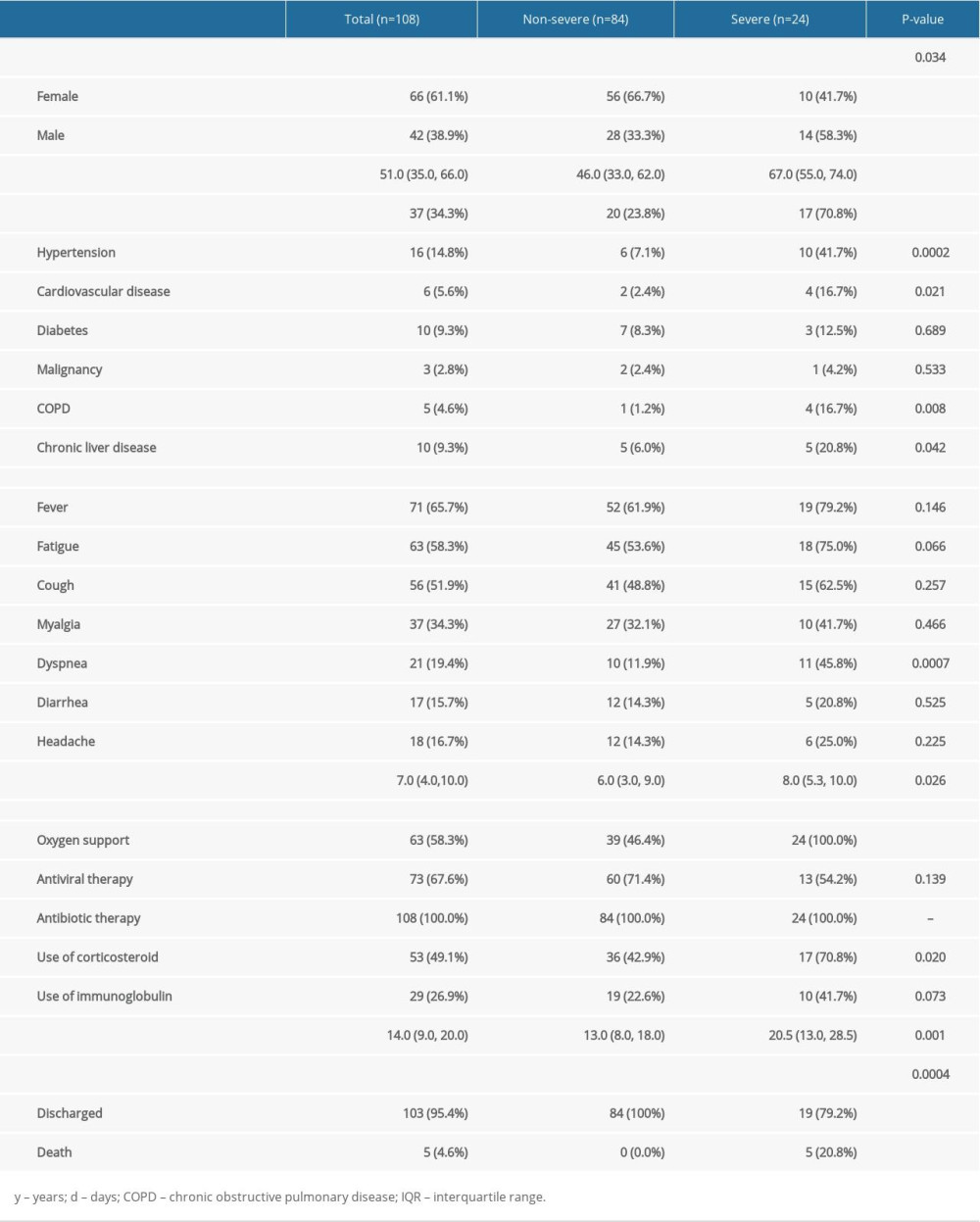 Table 1. Demographics, baseline characteristics, treatment and clinical outcomes of 108 COVID-19 patients.
Table 1. Demographics, baseline characteristics, treatment and clinical outcomes of 108 COVID-19 patients.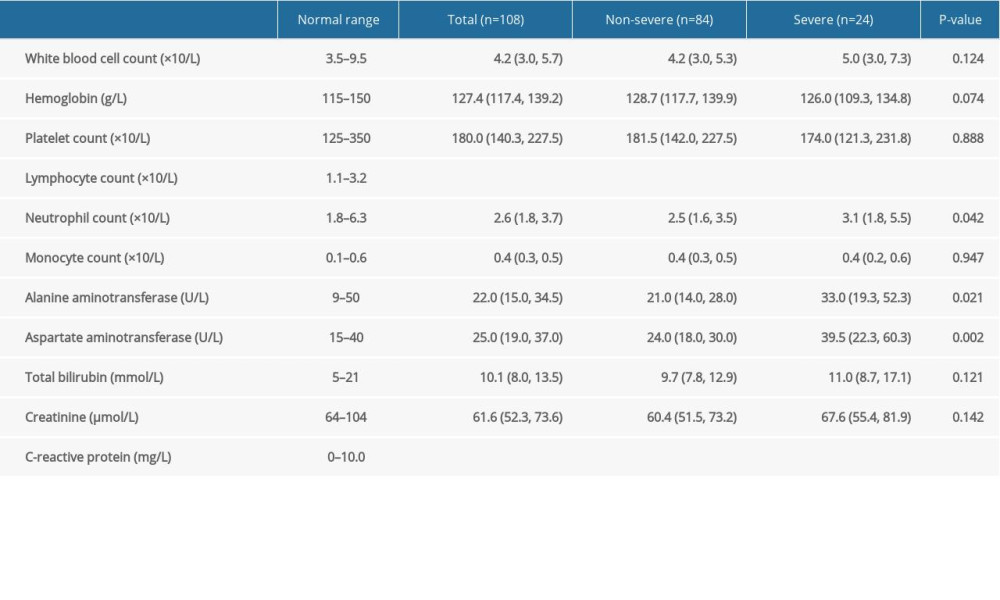 Table 2. Laboratory results of 108 COVID-19 patients.
Table 2. Laboratory results of 108 COVID-19 patients.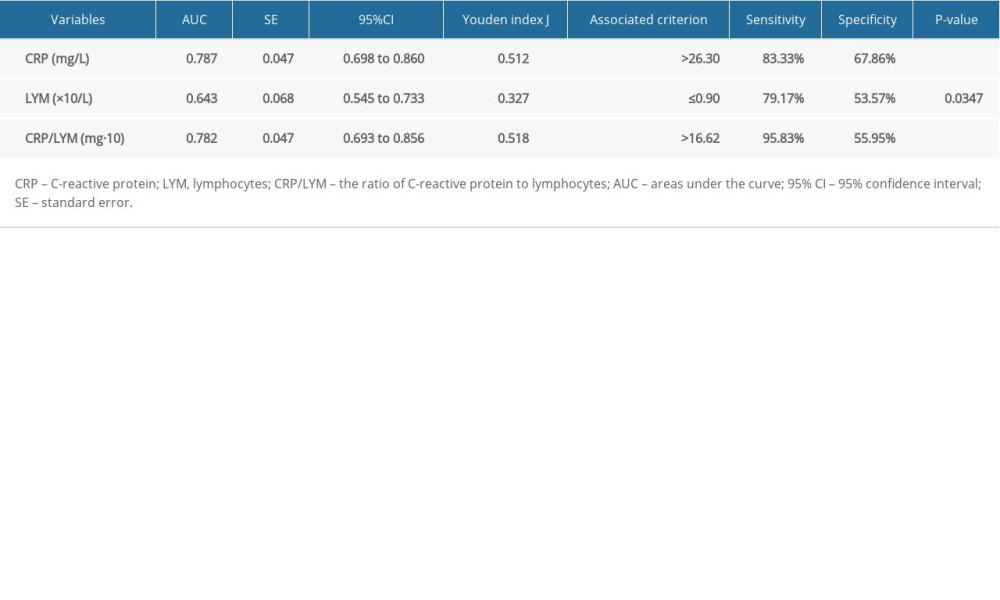 Table 3. Characteristics of ROC curves in severe COVID-19 patients.
Table 3. Characteristics of ROC curves in severe COVID-19 patients.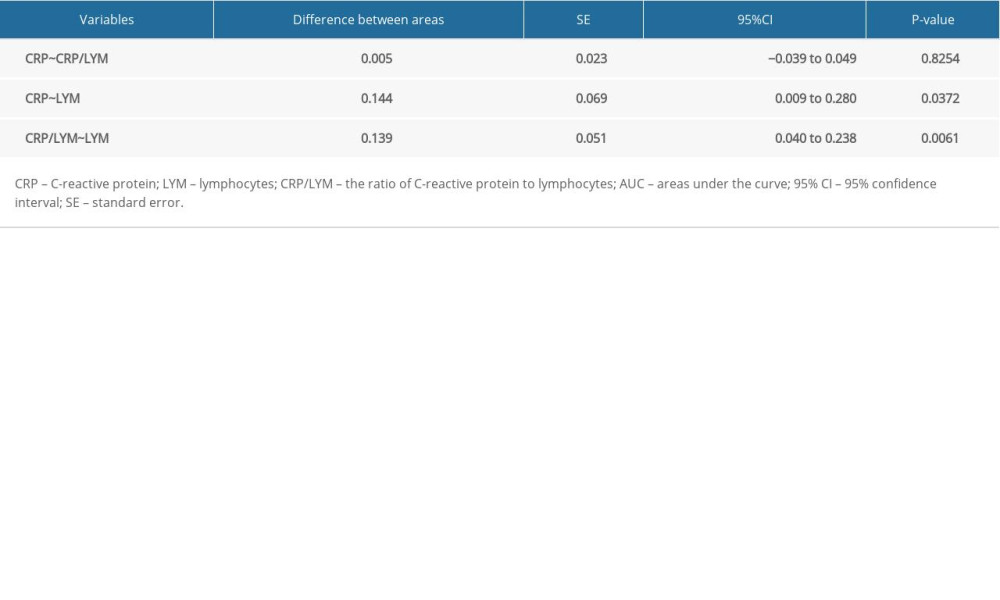 Table 4. Comparison of ROC curves in severe COVID-19 patients.
Table 4. Comparison of ROC curves in severe COVID-19 patients. Table 1. Demographics, baseline characteristics, treatment and clinical outcomes of 108 COVID-19 patients.
Table 1. Demographics, baseline characteristics, treatment and clinical outcomes of 108 COVID-19 patients. Table 2. Laboratory results of 108 COVID-19 patients.
Table 2. Laboratory results of 108 COVID-19 patients. Table 3. Characteristics of ROC curves in severe COVID-19 patients.
Table 3. Characteristics of ROC curves in severe COVID-19 patients. Table 4. Comparison of ROC curves in severe COVID-19 patients.
Table 4. Comparison of ROC curves in severe COVID-19 patients. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387