15 October 2020: Lab/In Vitro Research
Epigallocatechin Gallate (EGCG) Improves Anti-Angiogenic State, Cell Viability, and Hypoxia-Induced Endothelial Dysfunction by Downregulating High Mobility Group Box 1 (HMGB1) in Preeclampsia
Min Zhong1ABCDEFG, Julan Peng1ABCDEFG, Lanhua Xiang1ABCDEFG, Xinhuang Yang1ABCDEFG, Xianghua Wang1ABCDEFG, Yanbin Zhu2ABCDEFG*DOI: 10.12659/MSM.926924
Med Sci Monit 2020; 26:e926924
Abstract
BACKGROUND: Preeclampsia (PE) is a serious complication of pregnancy with no effective therapy. This study assessed whether epigallocatechin gallate (EGCG) could reduce the production of anti-angiogenic factors, improve cell viability, and suppress endothelial dysfunction in vitro via regulating high mobility group box 1 (HMGB1) in preeclampsia.
MATERIAL AND METHODS: Human umbilical vein endothelial cells (HUVECs) grown in conditioned medium from hypoxic JEG-3 cells were used to investigate the effects of EGCG on anti-angiogenic state, cell viability, and markers of endothelial dysfunction. To confirm that EGCG exerted its effects via HMGB1, we also examined the impact of EGCG on anti-angiogenic state, cell viability, and endothelial dysfunction following HMGB1 treatment in vitro.
RESULTS: EGCG inhibited HMGB1 expression in hypoxic trophoblast cells in a dose-dependent manner. In addition, EGCG relieved anti-angiogenic state and endothelial dysfunction in hypoxic trophoblast cells by downregulating HMGB1. Moreover, EGCG dose-dependently promoted cell proliferation by downregulating HMGB1.
CONCLUSIONS: Taken together, our data show the protective role of EGCG in preeclampsia and revealed EGCG-mediated effects on the production of anti-angiogenic factors, cell viability, and endothelial dysfunction through downregulating HMGB1. These observations suggest that EGCG is a novel therapeutic candidate for preeclampsia.
Keywords: endothelial cells, HMGB1 Protein, Pre-Eclampsia, Angiogenesis Inhibitors, Catechin, Cell Hypoxia, Down-Regulation, Pregnancy
Background
Preeclampsia (PE) is a pregnancy disease characterized by hypertension and proteinuria after 20 weeks of pregnancy, which can lead to adverse pregnancy outcomes [1]. Despite extensive research, the etiology and pathogenesis of PE are not completely understood, and there is no effective treatment for the pregnancy complications [2].
According to the two-stage model of PE, placental hypoxia and inadequate perfusion lead to the release of inflammatory factors and cell fragments into the blood, causing clinical symptoms characterized by systemic inflammatory response and endothelial dysfunction [3]. In addition, recent studies have shown that trophocyte hypoxia and dysfunction of vascular endothelial cells play important regulatory roles in the occurrence and development of PE [4,5].
High mobility group box 1 (HMGB1), which belongs to a high mobility group box that is known for high mobility in electrophoresis, has been researched most intensively [6]. After damage of pathogenic factors, HMGB1 interacts with receptor for advanced glycation end-products and Toll-like receptors through active and passive release, causing the release of inflammatory mediators and stimulating the immune inflammatory response of the body [7,8]. It had been reported that placental hypoxia induced the expression and release of HMGB1 from trophoblasts [9]. Accumulating reports presented high levels of placental HMGB1 in women with PE [9,10]. Moreover, increasing evidence suggests that placental hypoxia-induced HMGB1 release plays a central role in endothelial dysfunction [9,11]. Therefore, HMGB1 may be an important therapy target for PE.
Epigallocatechin gallate (EGCG), the most abundant and bioactive polyphenol in green tea, has been demonstrated to possess many biological functions [12]. A study proved that EGCG could promote the treatment efficacy of oral nifedipine against severe PE [13]. Chae et al. proved that EGCG could inhibit Ang II-induced elevation of vascular cell adhesion molecule 1 (VCAM-1) and intercellular cell adhesion molecule-1 (ICAM-1) in human umbilical vein endothelial cells (HUVECs) [14]. EGCG can enhance the endothelial nitric oxide synthase (eNOS) activation and nitric oxide (NO) formation in HUVECs, indicating that EGCG may help improve endothelial functions [15]. Several studies showed that EGCG can inhibit the expression of HMGB1 [16,17].
Accordingly, we performed this study to assess the relationship among EGCG, HMGB1, and PE and to analyze EGCG-mediated effects on angiogenesis, trophoblast cell viability, and endothelial function, aiming to provide a novel treatment for PE.
Material and Methods
CELL CULTURE AND TREATMENT:
The JEG-3 cell line and HUVECs were obtained from American Type Culture Collection (ATCC) (Rockville, MD, USA). JEG-3 was maintained in Dulbecco’s minimum essential medium (DMEM)/F12 (Invitrogen, NY, USA) supplemented with 10% fetal bovine serum (FBS; Hyclone, PA, USA), 50 IU/mL penicillin G, and 50 μg/mL streptomycin (Invitrogen, NY, USA) at 37°C in the presence of 8% O2, 5% CO2, and 87% N2. Hypoxic treatment was performed in an incubator supplied with 1% O2, 5% CO2, and 94% N2 for 24 h. The conditioned medium was collected immediately for the subsequent testing and culture of HUVECs. HUVECs were grown in DMEM/F12 containing 10% FBS or conditioned medium from hypoxic JEG-3 cells in the presence of 8% O2, 5% CO2, and 87% N2 at 37°C.
EGCG (10, 30, 50 μm) and EGCG (50 μm) [18] +HMGB1 (5 ng/ml) [19] were added into cultured JEG-3 cells or HUVECs for 24 h.
CELL PROLIFERATION ASSAY:
HUVECs were incubated with conditioned medium or conditioned medium containing EGCG (10, 30, 50 μm) or EGCG (50μm)+HMGB1 (5 ng/ml) for 24 h. Cell Counting Kit-8 (CCK-8; Beyotime, Shanghai, China) and BrdU (Beyotime, Shanghai, China) were used to determine cell proliferation according to the manufacturer’s instructions. For CCK-8 assay, HUVECs were incubated with 10 μl CCK-8 for 2 h, and absorbance at 450 nm was measured with a microplate reader (Bio-Rad, CA, USA). For BrdU cell proliferation assay, HUVECs were incubated with 5 μM BrdU for 4 h before 4% paraformaldehyde fixation. The BrdU signal was revealed by rat anti-BrdU antibody and goat anti-rat IgG. After DAPI staining, the proportion of BrdU-positive HUVECs was counted under the fluorescence microscope (Nikon, Japan).
(ENZYME-LINKED IMMUNOSORBENT ASSAY) ELISA:
The levels of vascular endothelial growth factor (VEGF) and soluble fms-like tyrosine kinase 1 (sFtl1) in JEG-3 cell culture supernatants and the levels of asymmetric dimethylarginine (ADMA), soluble endoglin (sEng), and nitric oxide (NO) in HUVECs culture supernatants were tested by use of corresponding ELISA kits according to the manufacturer’s recommendations.
WESTERN BLOTTING ASSAY:
HUVECs and JEG-3 cells were lysed in RIPA buffer containing protease and phosphatase inhibitors for 30 min. The proteins in each sample were separated on SDS-PAGE gel and transferred onto PVDF membranes. PVDF membranes were incubated with anti-HMGB1, anti-ICAM-1, anti-VCAM-1, anti-ET-1, anti-E-selectin, anti-eNOS, anti-phospho-eNOS, and anti-GAPDH antibodies. The protein bands were visualized using an enhanced chemiluminescence system (Perfect Biotech, Shanghai, China).
REAL-TIME QUANTITATIVE PCR:
Total RNA extracted by Trizol (Invitrogen, NY, USA) was reverse-transcribed to complementary DNA using the PrimeScript RT reagent kit (Takara, Japan). The cDNA was quantified with SYBR-Green Supermix (Invitrogen, NY, USA) by the ABI PRISM 7000 Sequence Detection System (ABI/Perkin Elmer, Foster City, CA, USA). Gene expression data were calculated using the 2−ΔΔCt method.
STATISTICAL ANALYSIS:
Data are presented as mean±SD and analyzed using one-way ANOVA followed by Tukey’s post-tests with SPSS software (SPSS, Armonk, NY, USA), and
Results
EGCG INHIBITED HMGB1 EXPRESSION IN HYPOXIC TROPHOBLAST CELLS:
Hypoxic treatment was performed in an incubator supplied with 1% O2, 5% CO2, and 94% N2. We then examined HMGB1 levels using RT-qPCR (Figure 1A) and western blot assay (Figure 1B). HMGB1 production significantly increased in cells under hypoxia. EGCG attenuated the hypoxia-induced increase in HMGB1 expression in a dose-dependent manner.
EGCG RELIEVED THE ANTI-ANGIOGENIC STATE IN HYPOXIC TROPHOBLAST CELLS BY DOWNREGULATING HMGB1:
To explore the molecular mechanisms associated with the influence of EGCG on preeclampsia, we next investigated whether EGCG affects the production of angiogenic factor VEGF (Figure 2A) and anti-angiogenic factor sFlt1 (Figure 2B). Compared with the control group, decreased VEGF levels and increased sFlt1 levels were observed in JEG-3 cells when exposed to hypoxic conditions. The cells treated with EGCG plus hypoxia released more VEGF and less sFlt1 than cells cultured under hypoxia alone. We also confirmed that the effects of EGCG on VEGF and sFlt1 production were reversed by HMGB1 treatment. These results suggest that EGCG can improve the anti-angiogenic state by inhibiting HMGB1.
EGCG DOSE-DEPENDENTLY PROMOTED CELL PROLIFERATION BY DOWNREGULATING HMGB1:
To analyze the role of EGCG in the proliferation of hypoxic trophoblast cells, CCK-8 assay and BrdU cell proliferation assay were used to assess cell viability. The results showed that EGCG dramatically enhanced the cell viability in a dose-dependent manner under hypoxic conditions. HMGB1 treatment reversed the proliferative effect of EGCG hypoxia-induced trophoblast cells (Figure 3A). Using BrdU as a marker for cell proliferation, newly generated surviving cells were detected by immunohistochemistry staining. Quantitative analysis of BrdU-positive cells also indicated that HMGB1 treatment suppressed the proliferative-promoting effect of EGCG in hypoxic trophoblast cells (Figure 3B).
EGCG SUPPRESSED ENDOTHELIAL DYSFUNCTION BY DOWNREGULATING HMGB1:
To evaluate the effects of EGCG on endothelial dysfunction in HUVECs, we assessed the main markers of endothelial dysfunction. Levels of the cell adhesion molecules ICAM-1, VCAM-1, ET-1, and E-selectin were determined using western blot assay (Figure 4A). EGCG successfully rescued the hypoxia-induced increase of ICAM-1, VCAM-1, ET-1, and E-selectin levels in HUVECs cultured in conditioned medium from hypoxic JEG-3 cells. Similarly, the decreased production of ICAM-1, VCAM-1, ET-1, and E-selectin following EGCG treatment was markedly rescued by HMGB1. Furthermore, sEng and ADMA expression were detected using ELISA assay (Figure 4B). sEng and ADMA values were higher in HUVECs cultured in conditioned medium from hypoxic JEG-3 cells than that in those cultured in normal medium, which were dose-dependently rescued by EGCG. With the addition of HMGB1, there was a significant increase in sEng and ADMA expression. These results suggest that EGCG can improve endothelial dysfunction by downregulating HMGB1.
To further assess the potential role of EGCG in endothelial dysfunction in PE, we investigated NO levels (Figure 5A) and activation of eNOS (Figure 5B). Compared with the control group, decreased NO level and p-eNOS expression were observed in HUVECs grown in conditioned medium from hypoxic JEG-3 cells. EGCG treatment induced more release of NO and p-eNOS in HUVECs than in cells cultured with conditioned medium alone. Moreover, the regulatory effects of EGCG on NO and p-eNOS production were inhibited by HMGB1 protein. All these results consistently show that EGCG suppressed endothelial dysfunction by downregulating HMGB1.
Discussion
PE is a type of pregnancy-specific disease with unknown clinical pathogenesis and complex and diverse risk factors. After onset, it poses a serious threat to the life of the mother and fetus [1,20]. Therefore, many preeclampsia studies have explored early prediction and intervention, aiming to decrease the incidence and mortality rates in mothers and infants [21]. Although the etiology of PE remains unclear, some studies proved that hypoxia plays an important role in the pathophysiological process of PE [22]. Long-term hypoxia causes an increase in the apoptosis of trophoblasts and endothelial dysfunction, which can result in the development of PE [4,5].
HMGB1 is a non-histone DNA-binding protein that is ubiquitously expressed in mammals [7]. As a late inflammatory factor, HMGB1 is considered to be closely related to a variety of inflammatory diseases [7,8]. Recently, HMGB1 was found to be highly expressed in the placenta of PE women [9,10]. Placental hypoxia can promote the expression and release of HMGB1 from trophoblasts [9]. In the present study, we found upregulation of HMGB1 in JEG-3 cells exposed to anoxia.
EGCG is the most abundant catechin in green tea and has a variety of biological activities [12]. A study suggested that EGCG can improve the treatment efficacy of oral nifedipine against severe PE [13], and EGCG treatment can markedly inhibit the expression of HMGB1 induced by balloon injury [16]. EGCG can alleviate lung injury by inhibiting the expression of HMGB1 and RAGE [17]. In the present study, we demonstrated that EGCG can inhibit the expression of HMGB1 in JEG-3 cells induced by hypoxia. In addition, EGCG treatment can promote HUVECs proliferation, and HMGB1 protein can inhibit the proliferative effect of EGCG on HUVECs induced by culture in medium of hypoxic JEG-3 cells. Thus, we speculated that HMGB1 is involved in the therapeutic effect of EGCG on PE.
Recently, angiogenic or anti-angiogenic factors such as VEGF and sFlt-1 have been found to be associated with the onset and endothelial dysfunction of PE [23,24]. Our data confirmed that EGCG treatment could enhance the level of VEGF and inhibit the level of sFlt-1 in JEG-3 cells induced by hypoxia. And as expected, the effects of HMGB1 protein on VEGF and sFlt-1 were contrary to that of EGCG.
NO is mainly catalyzed and produced by eNOS, which has the function of vasodilatation and protecting vascular endothelium under physiological contents [25,26]. Accumulating evidence has suggested that intercellular adhesion molecule-1 (ICAM-1), vascular cell adhesion molecule 1 (VCAM-1), endothelin 1 (ET-1), and E-selectin play pivotal roles in the mechanisms of endothelial dysfunction [27,28]. In the present study, the expression of ICAM-1, VCAM-1, ET-1, and E-selectin was upregulated and the levels of NO and eNOS were downregulated in HUVECs cultured with culture medium of hypoxic JEG-3 cells. In contrast, downregulation of ICAM-1, VCAM-1, ET-1, and E-selectin and upregulation of NO and eNOS were observed in HUVECs treated with EGCG. Moreover, we proved that the therapeutic effects of EGCG on endothelial dysfunction were reversed by HMGB1.
Conclusions
The present results suggest that HMGB1 contributes to trophoblastic injury and endothelial dysfunction. The protective effects of EGCG on angiogenesis, cell viability, and endothelial function are involved in the modulation of HMGB1 expression. These findings may help in discovery of a novel medicine for treatment of PE.
Figures
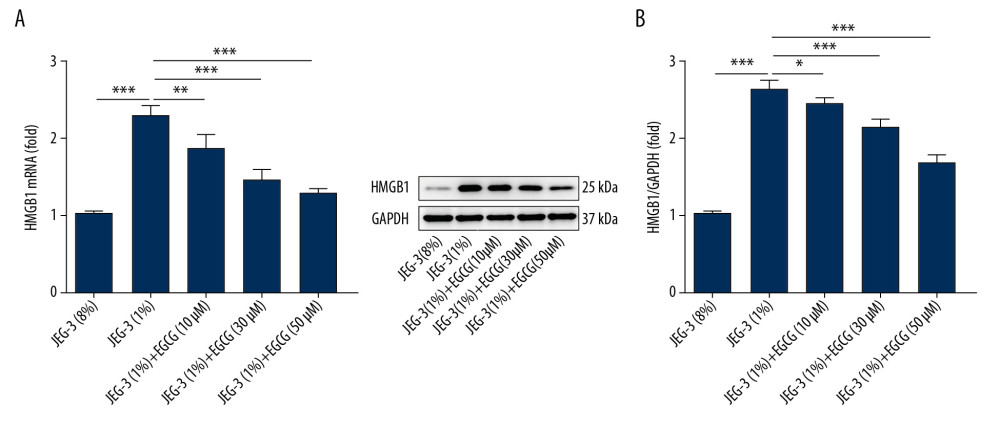 Figure 1. EGCG inhibited HMGB1 expression in hypoxic trophoblast cells. (A) HMGB1 mRNA levels determined by RT-qPCR; (B) HMGB1 protein levels determined by western blot assay. * p<0.05, ** p<0.01, *** p<0.001.
Figure 1. EGCG inhibited HMGB1 expression in hypoxic trophoblast cells. (A) HMGB1 mRNA levels determined by RT-qPCR; (B) HMGB1 protein levels determined by western blot assay. * p<0.05, ** p<0.01, *** p<0.001. 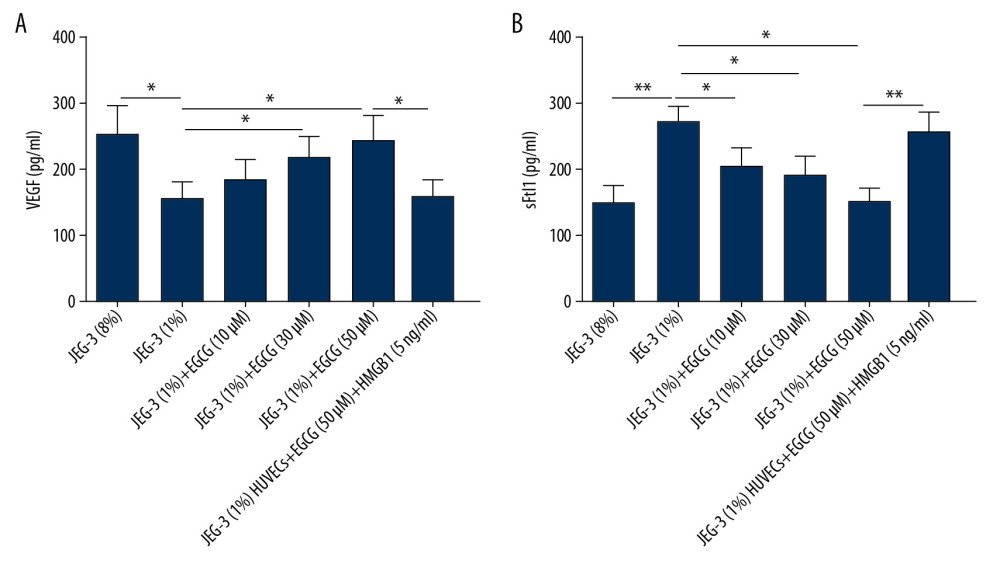 Figure 2. EGCG relieved anti-angiogenic state in hypoxic trophoblast cells by downregulating HMGB1. (A) VEGF production detected by ELISA assay; (B) sFlt1 production detected by ELISA assay. * p<0.05, ** p<0.01.
Figure 2. EGCG relieved anti-angiogenic state in hypoxic trophoblast cells by downregulating HMGB1. (A) VEGF production detected by ELISA assay; (B) sFlt1 production detected by ELISA assay. * p<0.05, ** p<0.01. 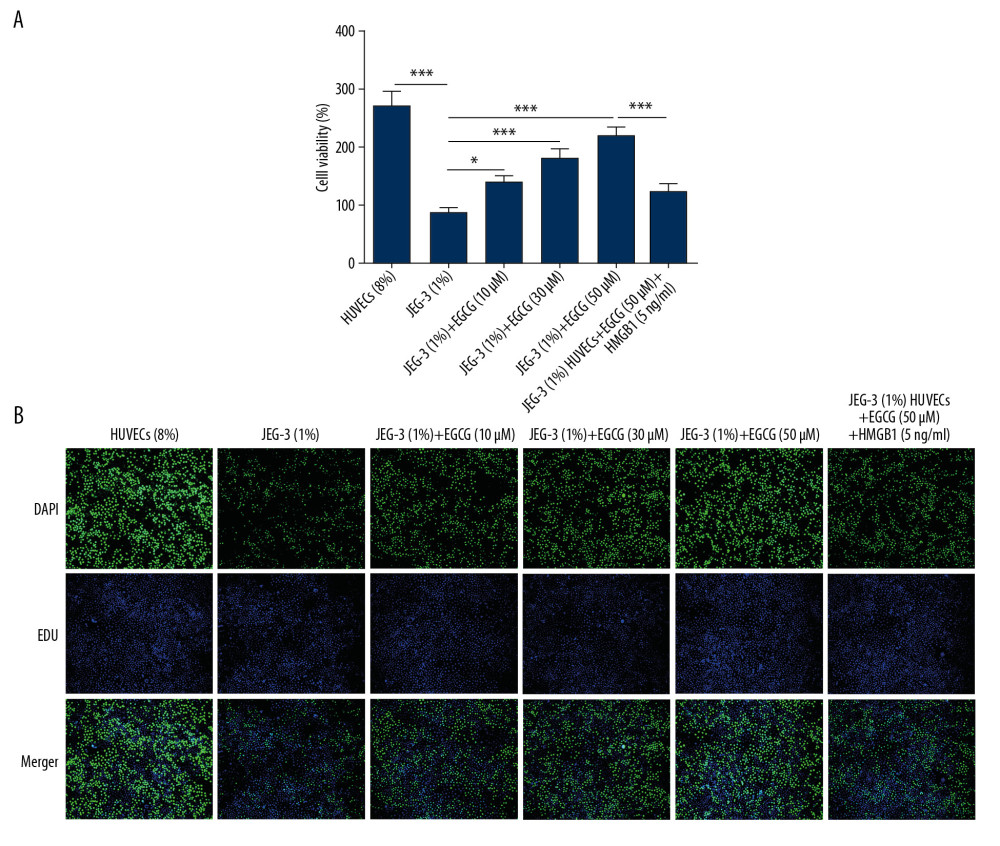 Figure 3. EGCG dose-dependently promoted cell proliferation by downregulating HMGB1. (A) Cell proliferation assessed using CCK-8 assay. (B) Cell proliferation assessed using BrdU incorporation assay. * p<0.05, *** p<0.001.
Figure 3. EGCG dose-dependently promoted cell proliferation by downregulating HMGB1. (A) Cell proliferation assessed using CCK-8 assay. (B) Cell proliferation assessed using BrdU incorporation assay. * p<0.05, *** p<0.001. 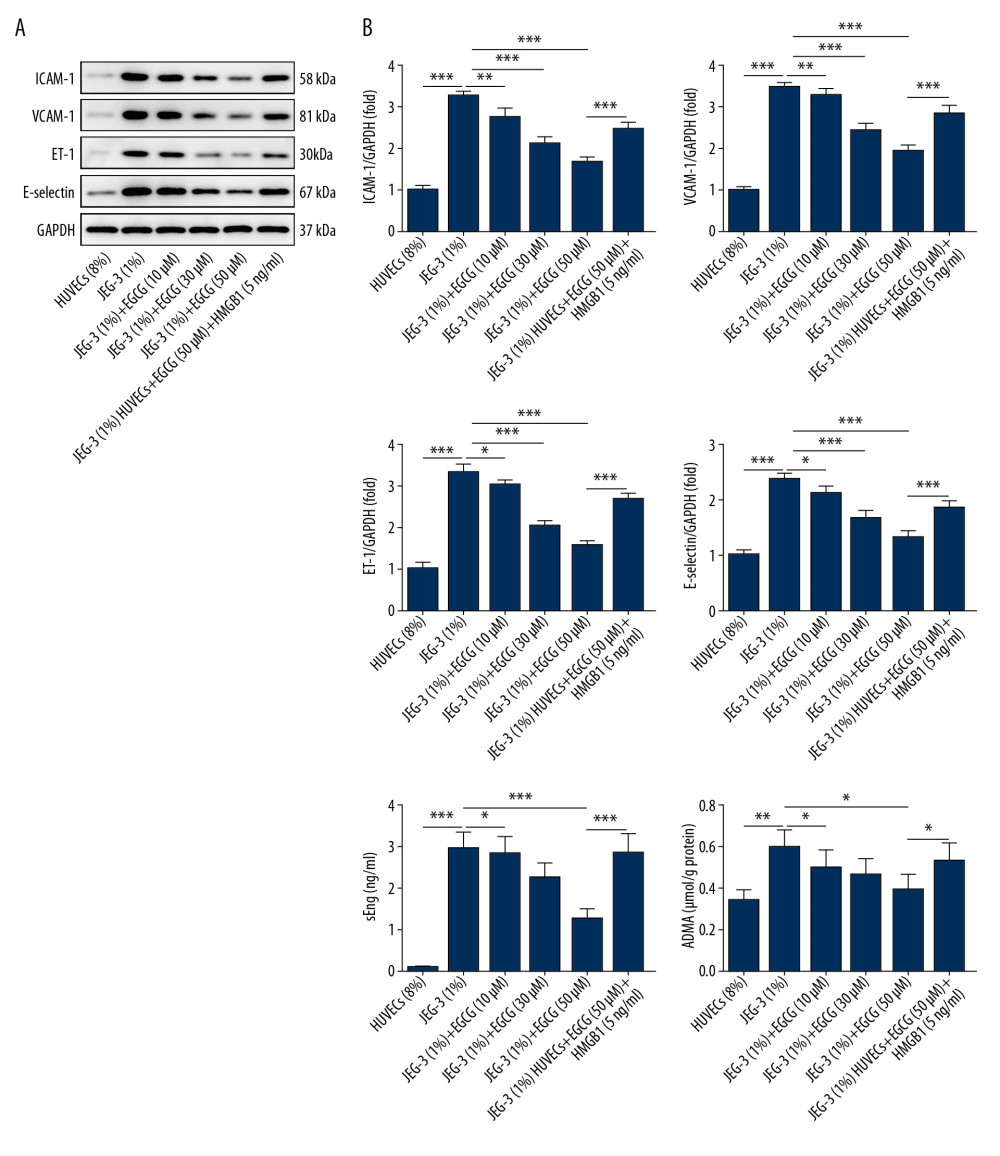 Figure 4. EGCG suppressed endothelial dysfunction by downregulating HMGB1. (A) Cell adhesion molecules levels such as ICAM-1, VCAM-1, ET-1, and E-selectin detected using western blot assay. (B) sEng and ADMA expression detected using ELISA assay. * p<0.05, ** p<0.01, *** p<0.001.
Figure 4. EGCG suppressed endothelial dysfunction by downregulating HMGB1. (A) Cell adhesion molecules levels such as ICAM-1, VCAM-1, ET-1, and E-selectin detected using western blot assay. (B) sEng and ADMA expression detected using ELISA assay. * p<0.05, ** p<0.01, *** p<0.001. 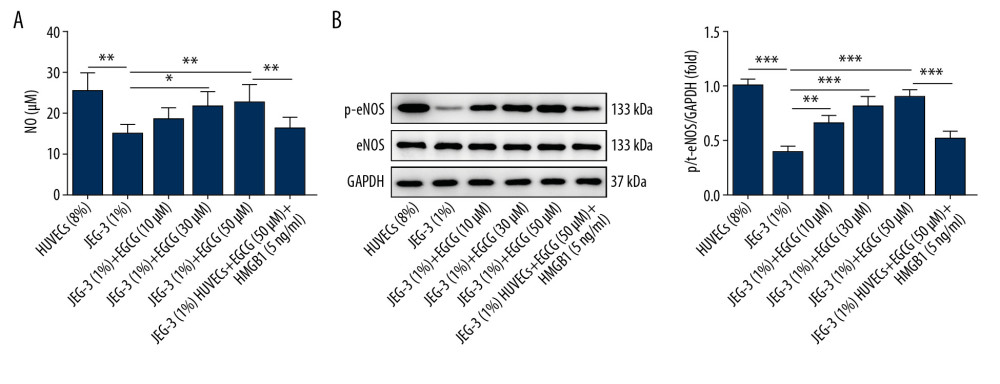 Figure 5. EGCG suppressed endothelial dysfunction by downregulating HMGB1. (A) NO levels determined by ELISA assay. (B) p-eNOS and eNOS expression detected by western blot assay. * p<0.05, ** p<0.01, *** p<0.001.
Figure 5. EGCG suppressed endothelial dysfunction by downregulating HMGB1. (A) NO levels determined by ELISA assay. (B) p-eNOS and eNOS expression detected by western blot assay. * p<0.05, ** p<0.01, *** p<0.001. References
1. ACOG Practice Bulletin No. 202, Gestational Hypertension and Preeclampsia: Obstet Gynecol, 2019; 133(1); e1-e25
2. Li Q, Han Y, Xu P, Elevated microRNA-125b inhibits cytotrophoblast invasion and impairs endothelial cell function in preeclampsia: Cell Death Discov, 2020; 6; 35
3. Alasztics B, Kukor Z, Pánczél Z, Valent SThe pathophysiology of preeclampsia in view of the two-stage model: Orv Hetil, 2012; 153(30); 1167-76 [in Hungarian]
4. Bellos I, Karageorgiou V, Kapnias D, The role of interleukins in preeclampsia: A comprehensive review: Am J Reprod Immunol, 2018; 80(6); e13055
5. Sava RI, March KL, Pepine CJ, Hypertension in pregnancy: Taking cues from pathophysiology for clinical practice: Clin Cardiol, 2018; 41(2); 220-27
6. Bellussi LM, Cocca S, Chen L, Rhinosinusal inflammation and high mobility group box 1 protein: A new target for therapy: J Otorhinolaryngol Relat Spec, 2016; 78(2); 77-85
7. Khambu B, Yan S, Huda N, Yin XM, Role of high-mobility group box-1 in liver pathogenesis: Int J Mol Sci, 2019; 20(21); 5314
8. Paudel YN, Angelopoulou E, Piperi C, Enlightening the role of high mobility group box 1 (HMGB1) in inflammation: Updates on receptor signalling: Eur J Pharmacol, 2019; 858; 172487
9. Hu Y, Yan R, Zhang C, High-mobility group box 1 from hypoxic trophoblasts promotes endothelial microparticle production and thrombophilia in preeclampsia: Arterioscler Thromb Vasc Biol, 2018; 38(6); 1381-91
10. Chen Q, Yin YX, Wei J, Increased expression of high mobility group box 1 (HMGB1) in the cytoplasm of placental syncytiotrophoblast from preeclamptic placentae: Cytokine, 2016; 85; 30-36
11. Jiang R, Cai J, Zhu Z, Hypoxic trophoblast HMGB1 induces endothelial cell hyperpermeability via the TRL-4/caveolin-1 pathway: J Immunol, 2014; 193(10); 5000-12
12. Sharifi-Rad M, Pezzani R, Redaelli M, Preclinical pharmacological activities of epigallocatechin-3-gallate in signaling pathways: An update on cancer: Molecules, 2020; 25(3); 467
13. Shi DD, Guo JJ, Zhou L, Wang N, Epigallocatechin gallate enhances treatment efficacy of oral nifedipine against pregnancy-induced severe pre-eclampsia: A double-blind, randomized and placebo-controlled clinical study: J Clin Pharm Ther, 2018; 43(1); 21-25
14. Chae YJ, Kim CH, Ha TS, Epigallocatechin-3-O-gallate inhibits the angiotensin II-induced adhesion molecule expression in human umbilical vein endothelial cell via inhibition of MAPK pathways: Cell Physiol Biochem, 2007; 20(6); 859-66
15. Liu S, Sun Z, Chu P, EGCG protects against homocysteine-induced human umbilical vein endothelial cells apoptosis by modulating mitochondrial-dependent apoptotic signaling and PI3K/Akt/eNOS signaling pathways: Apoptosis, 2017; 22(5); 672-80
16. Yang B, Gao P, Wu X, Epigallocatechin-3-gallate attenuates neointimal hyperplasia in a rat model of carotid artery injury by inhibition of high mobility group box 1 expression: Exp Ther Med, 2017; 14(3); 1975-82
17. Li Y, Chen L, Guo F, Effects of epigallocatechin-3-gallate on the HMGB1/RAGE pathway in PM2.5-exposed asthmatic rats: Biochem Biophys Res Commun, 2019; 513(4); 898-903
18. Yang GZ, Wang ZJ, Bai F, Epigallocatechin-3-gallate protects HUVECs from PM2.5-induced oxidative stress injury by activating critical antioxidant pathways: Molecules, 2015; 20(4); 6626-39
19. Zhao M, Feng Y, Xiao J, Sodium tanshinone IIA sulfonate prevents hypoxic trophoblast-induced endothelial cell dysfunction via targeting HMGB1 release: J Biochem Mol Toxicol, 2017; 31(7); jbt.2.1903
20. Rahnemaei FA, Fashami MA, Abdi F, Abbasi M, Factors effective in the prevention of preeclampsia: A systematic review: Taiwan J Obstet Gynecol, 2020; 59(2); 173-82
21. Churchill D, Duley L, Thornton JG, Interventionist versus expectant care for severe pre-eclampsia between 24 and 34 weeks’ gestation: Cochrane Database Syst Rev, 2018; 10; CD003106
22. Mori A, Nishi H, Sasaki T, HLA-G expression is regulated by miR-365 in trophoblasts under hypoxic conditions: Placenta, 2016; 45; 37-41
23. Trapiella-Alfonso L, Alexandre L, Fraichard C, VEGF (vascular endothelial growth factor) functionalized magnetic beads in a microfluidic device to improve the angiogenic balance in preeclampsia: Hypertension, 2019; 74(1); 145-53
24. Ali Z, Khaliq S, Zaki S, Altered expression of vascular endothelial growth factor, vascular endothelial growth factor receptor-1, vascular endothelial growth factor receptor-2, and Soluble Fms-like Tyrosine Kinase-1 in peripheral blood mononuclear cells from normal and preeclamptic pregnancies: Chin J Physiol, 2019; 62(3); 117-22
25. Katusic ZS, Austin SA, Neurovascular protective function of endothelial nitric oxide – recent advances: Circ J, 2016; 80(7); 1499-503
26. Zhao Y, Vanhoutte PM, Leung SW, Vascular nitric oxide: Beyond eNOS: J Pharmacol Sci, 2015; 129(2); 83-94
27. Koleva DI, Orbetzova MM, Nikolova JG, Deneva TI, Pathophysiological role of adiponectin, leptin and asymmetric dimethylarginine in the process of atherosclerosis: Folia Med (Plovdiv), 2016; 58(4); 234-40
28. Wiseman S, Marlborough F, Doubal F, Blood markers of coagulation, fibrinolysis, endothelial dysfunction and inflammation in lacunar stroke versus non-lacunar stroke and non-stroke: Systematic review and meta-analysis: Cerebrovasc Dis, 2014; 37(1); 64-75
Figures
 Figure 1. EGCG inhibited HMGB1 expression in hypoxic trophoblast cells. (A) HMGB1 mRNA levels determined by RT-qPCR; (B) HMGB1 protein levels determined by western blot assay. * p<0.05, ** p<0.01, *** p<0.001.
Figure 1. EGCG inhibited HMGB1 expression in hypoxic trophoblast cells. (A) HMGB1 mRNA levels determined by RT-qPCR; (B) HMGB1 protein levels determined by western blot assay. * p<0.05, ** p<0.01, *** p<0.001. Figure 2. EGCG relieved anti-angiogenic state in hypoxic trophoblast cells by downregulating HMGB1. (A) VEGF production detected by ELISA assay; (B) sFlt1 production detected by ELISA assay. * p<0.05, ** p<0.01.
Figure 2. EGCG relieved anti-angiogenic state in hypoxic trophoblast cells by downregulating HMGB1. (A) VEGF production detected by ELISA assay; (B) sFlt1 production detected by ELISA assay. * p<0.05, ** p<0.01. Figure 3. EGCG dose-dependently promoted cell proliferation by downregulating HMGB1. (A) Cell proliferation assessed using CCK-8 assay. (B) Cell proliferation assessed using BrdU incorporation assay. * p<0.05, *** p<0.001.
Figure 3. EGCG dose-dependently promoted cell proliferation by downregulating HMGB1. (A) Cell proliferation assessed using CCK-8 assay. (B) Cell proliferation assessed using BrdU incorporation assay. * p<0.05, *** p<0.001. Figure 4. EGCG suppressed endothelial dysfunction by downregulating HMGB1. (A) Cell adhesion molecules levels such as ICAM-1, VCAM-1, ET-1, and E-selectin detected using western blot assay. (B) sEng and ADMA expression detected using ELISA assay. * p<0.05, ** p<0.01, *** p<0.001.
Figure 4. EGCG suppressed endothelial dysfunction by downregulating HMGB1. (A) Cell adhesion molecules levels such as ICAM-1, VCAM-1, ET-1, and E-selectin detected using western blot assay. (B) sEng and ADMA expression detected using ELISA assay. * p<0.05, ** p<0.01, *** p<0.001. Figure 5. EGCG suppressed endothelial dysfunction by downregulating HMGB1. (A) NO levels determined by ELISA assay. (B) p-eNOS and eNOS expression detected by western blot assay. * p<0.05, ** p<0.01, *** p<0.001.
Figure 5. EGCG suppressed endothelial dysfunction by downregulating HMGB1. (A) NO levels determined by ELISA assay. (B) p-eNOS and eNOS expression detected by western blot assay. * p<0.05, ** p<0.01, *** p<0.001. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








