27 January 2021: Lab/In Vitro Research
Ginsenoside Rb1 Facilitates Browning by Repressing Wnt/β-Catenin Signaling in 3T3-L1 Adipocytes
Qingxin Fan12BCDEF, Pengjiao Xi3ACD, Derun Tian13AG, Lianqun Jia4AFG, Yuan Cao4BDF, Kaixuan Zhan4BCDF, Tianwei Sun5BCDG, Yinlong Zhang6BCD, Qiming Wang14ABCDEFG*DOI: 10.12659/MSM.928619
Med Sci Monit 2021; 27:e928619
Abstract
BACKGROUND: The discovery of browning in white adipose tissue has provided new ideas for treating obesity. Many studies have reported that ginsenoside Rb1 (G-Rb1) has activity against diabetes, inflammation, and obesity, but further investigation is needed on the effect and mechanism of G-Rb1 on browning.
MATERIAL AND METHODS: We treated 3T3-L1 adipocytes with 0–200 μM G-Rb1, and 0.5 μM Compound 3f and 30 μM SKL2001 were used to activate Wnt/b-catenin signaling. Adipocyte activity was evaluated by Cell Counting Kit-8. Oil Red O staining was used to detect the lipid droplets. Quantitative real-time polymerase chain reaction was used to measure the expression of Cd-137, Cited-1, Txb-1, Prdm-16, and Ucp-1 mRNA. Western blotting was used to measure the expression of Ucp-1, pGSK-3β (Ser 9), GSK- 3β, and β-catenin proteins. The expression of Ucp-1 was also detected with immunofluorescence.
RESULTS: Adipocyte activity was not affected by 0–100 μM G-Rb1. However, G-Rb1 dose-dependently reduced the accumulation of lipid droplets; increased the expression of Cd-137, Cited-1, Txb-1, Prdm-16, and Ucp-1 mRNA; and increased the expression of Ucp-1, pGSK-3β (Ser 9), GSK-3β, and β-catenin proteins. The accumulation of lipid droplets and the expression of Ucp-1 protein decreased as b-catenin increased.
CONCLUSIONS: G-Rb1 at various concentrations (0–100 μM) promoted the browning of adipocytes in a dose-dependent manner. Further, we confirmed that activation of Wnt/β-catenin signaling could inhibit browning. Therefore, the browning promoted by G-Rb1 may be associated with the inhibition of Wnt/β-catenin signaling.
Keywords: 3T3-L1 Cells, Ginsenosides, Maillard Reaction, Obesity, Adipocytes, Adipocytes, White, Adipogenesis, Adipose Tissue, White, Lipid Metabolism, Lipids, Transcription Factors, beta Catenin
Background
Obesity is defined as a disease, and it is a public health problem that has become an epidemic [1]. In 2015, more than 600 million adults were obese and the number is growing. Obesity is an independent risk factor for many diseases, notably type 2 diabetes mellitus, cardiovascular disease, and hypertension. Multimodal lifestyle interventions (dietary, exercise, and behaviors), pharmacotherapy, and bariatric surgery are the main source of treatment at present [2]. In recent years, following the discovery of brown fat in adults, the browning of adipose tissue has been proposed as a new strategy for the treatment of obesity [3].
Ginsenoside is a sterol compound primarily extracted from the roots, stems, and flower buds of plants belonging to the genus
Since Roel Nusse and Hans Clevers discovered the first member of the Wnt family in 1982, the important role of Wnt signaling in embryogenesis, tumorigenesis, and osteogenesis has gradually become recognized [10–12]. Wnt/β-catenin signaling is referred to as the typical Wnt signaling pathway. When Wnt/β-catenin signaling is activated, β-catenin accumulates in the cytoplasm and becomes a transcription switch that regulates cell fate by forming an active complex and recruiting co-activators [13]. A β-catenin deficiency can cause smooth muscle to be replaced by adipose in the uterus [14]. Overexpressing Wnt10b reduced the volume of interscapular brown fat in mice, and activating Wnt signaling decreased Ucp-1 by inhibiting PPAR-γ2 coactivator 1-alpha
Material and Methods
CHEMICALS:
Ginsenoside Rb1 (HPLC ≥98%, Figure 1A) was purchased from Solarbio (Beijing, China). Compound 3f (C-3f) and SKL2001 (S-2001) were purchased from MedChemExpress (Houston, TX, USA).
CELL CULTURE AND ADIPOGENIC DIFFERENTIATION:
The methods for cell culture and adipogenic differentiation followed a previous report from our group [17]. After being maintained at 10 μg/mL insulin for 8 days, the adipocytes were incubated with G-Rb1 (0, 10, 50, 100, and 200 μM) from day 0 to 7.
CELL VIABILITY:
Ninety microliters (5×104/mL) of 3T3-L1 adipocytes was seeded into 96-well plates. After 24 h, the adipocytes were treated with 10 μL of G-Rb1 (0, 100, 500, 1000, and 2000 μM). After the medium was refreshed, 10 μL of CCK-8 (Solarbio) solution was added to each well for 2 h. The absorbance was determined at 450 nm, and the adipocyte viability was calculated. Each well had 4 replicates.
OIL RED O STAINING:
After fixed, the adipocytes were immersed in filtered Oil Red O stain (ORS; Solarbio) working solution for 20 min. The dye solution was then discarded, and the adipocytes were washed twice with double-distilled H2O. The cells were observed under an inverted microscope and images were captured. To quantify the triglyceride content of each group, we used 100% isopropanol to dissolve the ORS and measured the absorbance at 490 nm. ImageJ was used to measure the diameters of lipid droplets: average diameter of lipid droplets=total area/number of droplets.
RNA EXTRACTION AND QUANTITATIVE REAL-TIME POLYMERASE CHAIN REACTION:
Total RNA was extracted by the TRIzol method according to the manufacturer’s instructions (Invitrogen, Carlsbad, CA, USA). Two micrograms of total RNA was reverse-transcribed into cDNA by RevertAid RT Reverse Transcription Kit (Thermo Fisher Scientific, Waltham, MA, USA). After reverse transcription, 1 μL of cDNA, 0.2 μL of forward primers, 0.2 μL of reverse primers, and 5 μL of FastStart Universal SYBR Green Master were added in nuclease-free water to make 10 μL, and cDNA was amplified by quantitative real-time polymerase chain reaction (qRT-PCR). The 2−ΔΔCT method was utilized to analyze the data. The primer sequences are listed in the Table 1.
WESTERN BLOTTING:
High-efficiency RIPA (Solarbio) was used to extract the total protein. After the treatment was applied in 6-well plates, the culture medium was removed and each well was washed with phosphate-buffered saline. Prechilled cell lysate (with 1 mM phenylmethylsulfonyl fluoride, 200 μL/well) was added to each well and repeatedly pipetted. The well contents were then centrifuged at 4°C, the supernatant was removed, and 4× SDS-PAGE loading buffer (Solarbio, Beijing, China) was added to it (ratio of supernatant to loading buffer=3: 1), followed by mixing and heating at 100°C. Electrophoresis was used to separate this mixture by SDS-PAGE. In an ice bath, the protein in the gel was transferred to a polyvinylidene difluoride membrane. After blocking, the membrane was incubated in Tris-buffered saline with Tween 20 with anti-Gapdh antibody (1: 1000; #bs-13282R, Bioss), anti-Ucp-1 antibody (1: 500; #bs-1925R, Bioss), anti-GSK-3β antibody (1: 1000; #bs-0023R, Bioss), anti-pGSK-3β antibody (Ser9; 1: 1000, #bs-2066R; Bioss), and anti-β-catenin antibody (Ser9; 1: 1000; #bs-1165R, Bioss), respectively, at 4°C overnight. The membrane was next incubated in Tris-buffered saline with Tween 20 with IgG-horseradish peroxidase (1: 8000; no. ZB-2306; Zsbio) at room temperature for 2 h. An enhanced chemiluminescence substrate (Cell Signaling Technology, Danvers, MA, USA) and a chemiluminescence detection system (Promega, Madison, WI, USA) were used to visualize the targeting protein. Gapdh was used as an internal reference.
IMMUNOFLUORESCENCE:
Triton X-100 (0.5%, Sigma-Aldrich, St. Louis, MO, USA) was used to increased cytomembrane permeability. After being blocked with blocking buffer (1% bovine serum albumin, 22.52 mg/mL glycine, and 0.1% Tween-20 in phosphate-buffered saline) for 30 min, the membrane was incubated with primary antibody dilution buffer, IgG-Alexa Fluor® 594 (Cell Signaling Technology, Beverly, USA), DAPI (Solarbio), and antifade mounting medium (Thermo Fisher Scientific, Waltham, MA, USA), successively. The membranes were kept in the dark throughout the process.
STATISTICAL ANALYSIS:
Data from each group are expressed as means±standard deviation. Differences between 2 groups were evaluated with
Results
EFFECT OF G-RB1 ON THE VIABILITY OF ADIPOCYTES:
We used the CCK-8 method to detect cell viability in 3T3-L1 adipocytes exposed to various concentrations of G-Rb1. At 200 μM, G-Rb1 significantly reduced the viability of adipocytes (Figure 1B).
EFFECT OF G-RB1 ON THE BROWNING OF ADIPOCYTES:
To confirm the browning effect of G-Rb1, we treated 3T3-L1 adipocytes with 0, 10, 50, and 100 μM G-Rb1. The ORS results showed that G-Rb1 could significantly reduce lipid content and droplet diameter (Figure 2A–2C). The qRT-PCR results showed that 100 μM G-Rb1 could stably promote mRNA expression of browning-related genes (B-RGs), such as Cd-137, Cited-1, Tbx-1, Prdm-16, and Ucp-1 (Figure 2D–2H). The increase of Ucp-1 protein was further verified by western blotting (Figure 2I, 2J). Taken as a whole, the data showed that G-Rb1 promoted browning in a dose-dependent manner.
G-RB1 INHIBITS THE WNT/β-CATENIN SIGNALING PATHWAY:
To confirm the effect of G-Rb1 on Wnt/β-catenin signaling, we measured the expression of pGSK-3β (Ser 9), GSK-3β, pGSK-3β/GSK-3β, and β-catenin by western blot analysis. The results showed that the expression of pGSK-3β (Ser 9), GSK-3β, pGSK-3β/GSK-3β, and β-catenin decreased with increasing G-Rb1 concentration, and these negative effects were most evident in the 100 μM group (Figure 3A–3E).
C-3F PARTLY ELIMINATED THE EFFECT OF G-RB1 ON BROWNING:
To confirm the role of Wnt/β-catenin signaling in the process of G-Rb1 promoting browning, we used 0.5 μM C-3f, a type of Wnt/β-catenin signaling activator, to determine whether G-Rb1 (100 μM) can promote browning by inhibiting Wnt/β-catenin signaling. ORS results showed that the accumulation and diameter of lipid droplets in the C-3f+ G-Rb1 group were higher than in the G-Rb1 group, but lower than in the C-3f group (Figure 4A–4C). Western blot results showed that 0.5 μM C-3f could increase the content of β-catenin by about 1.36 times and decrease the content of Ucp-1 by 0.62 times. In addition, the content of β-catenin in the C-3f+G-Rb1 group was higher than in the G-Rb1 group and lower than in the C-3f group, and the content of Ucp-1 in the C-3f+G-Rb1 group was lower than in the G-Rb1 group and higher than in the C-3f group (Figure 4D–4F). These results were confirmed by immunostaining (Figure 4G), and they proved that C-3f could partly eliminate the effect of G-Rb1 on browning by activating Wnt/β-catenin signaling.
SKL2001 PARTLY ELIMINATED THE EFFECT OF G-RB1 ON BROWNING:
To further confirm the role of Wnt/β-catenin signaling in the process of G-Rb1 promoting browning, we used another Wnt/β-catenin signaling activator, SKL2001(S-2001). To ensure cell viability and inhibition of Wnt/β-catenin signaling, we treated the 3T3-L1 adipocytes with 30 μM S-2001. ORS results showed that the accumulation and diameter of lipid droplets in the S-2001+ G-Rb1 group were higher than in the G-Rb1 group but lower than in the S-2001 group (Figure 5A–5C). Western blot results showed that 30 μM S-2001 could increase the content of β-catenin by about 1.74 times and decrease the content of Ucp-1 by 0.73 times. In addition, the content of β-catenin in the S-2001+G-Rb1 group was higher than in the G-Rb1 group and lower than in the S-2001 group, and the content of Ucp-1 in the S-2001+G-Rb1 group was lower than in the G-Rb1 group and higher than in the S-2001 group (Figure 5D–5F). These results were further confirmed by immunostaining (Figure 5G), and they proved that S-2001 could partly eliminate the effect of G-Rb1 on browning by activating Wnt/β-catenin signaling.
Discussion
With the confirmation of browning in white adipose tissue and the discovery of brown adipose tissue in adults, enhancing browning has become a new strategy for the treatment of obesity [3,18]. Natural extracts have gradually attracted public attention because of their benign safety profile and affordable prices. Currently, many natural extracts have been shown to modulate the browning, such as apple polyphenols [19], swertiajaponin [20], and
As an easy-to-operate procedure that produces reliable results, CCK-8 is gradually replacing MTT in cell viability testing. In our research, we found that G-Rb1 at a concentration of 0–100 μM has no obvious effect on adipocyte viability. At a concentration of 200 μM, G-Rb1 significantly inhibited cell viability, and this inhibitory effect was more obvious as the intervention time increased. This finding is supported by results from Park et al. [9], who also found that low concentrations of G-Rb1 (0.1, 1, and 10 μM) could increase the viability of 3T3-L1 adipocytes. These results suggest that G-Rb1 has good biosafety.
Ucp-1 is abundant in brown adipocytes, which can convert fat and sugar into heat instead of ATP [22]. The reduction of lipid accumulation, minimization of lipid droplets, and augmentation of B-RG expression are characteristics of brown adipocytes [23]. Initially, Mu et al. [8] found that 10 μM G-Rb1 could increase Ucp-1 mRNA by approximately 1.5 times. Subsequently, Park et al. [9] found that G-Rb1 could increase Ucp-1, Prdm-16, and Pgc-1α proteins in a dose-dependent manner up to 40 μM. In our study, we found that the dose-dependent effects extended to 100 μM. ORS results showed that G-Rb1 could reduce the accumulation and size of lipid droplets in 3T3-L1 adipocytes. qRT-PCR results showed that Cd137, Cited-1, Tbx-1, Prdm-16, and Ucp-1 mRNA expression was increased. Western blot results further verified the increased expression of Ucp-1 protein.
Wnt/β-catenin signaling plays an important role in lipid metabolism [24]. When it is in an inactive state, cytosolic β-catenin is phosphorylated by GSK-3β kinase, and phosphorylated β-catenin will be degraded by proteases. When the Wnt/β-catenin signaling pathway is activated, GSK-3β (Ser 9 or 21) is phosphorylated and inactive. Cytosolic β-catenin gradually accumulates and enters the nucleus to regulate gene expression [13]. Recently, natural extracts have attracted attention with regard to browning. The pro-browning effect of
Conclusions
In summary, our research further clarified the browning effect promoted by G-Rb1 in 3T3-L1 adipocytes. After Wnt/β-catenin signaling was activated by C-3f or SKL2001, we found that the browning effect of G-Rb1 might be related to the inhibition of Wnt/β-catenin signaling. The mechanism between G-Rb1 and Wnt/β-catenin signaling needs to be further explored.
Figures
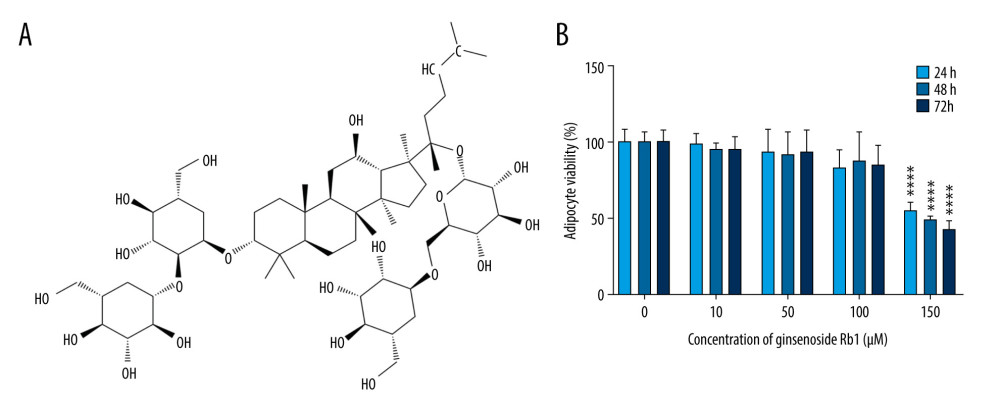 Figure 1. Effect of ginsenoside Rb1 (G-Rb1) on the adipocyte viability. (A) The structure of G-Rb1. (B) 3T3-L1 adipocytes were treated with G-Rb1 (0, 10, 50, 100, and 200 μM) for 24, 48, and 72 h. Data are expressed as mean±SD (n=10). **** P<0.0001 vs. 0 μM.
Figure 1. Effect of ginsenoside Rb1 (G-Rb1) on the adipocyte viability. (A) The structure of G-Rb1. (B) 3T3-L1 adipocytes were treated with G-Rb1 (0, 10, 50, 100, and 200 μM) for 24, 48, and 72 h. Data are expressed as mean±SD (n=10). **** P<0.0001 vs. 0 μM. 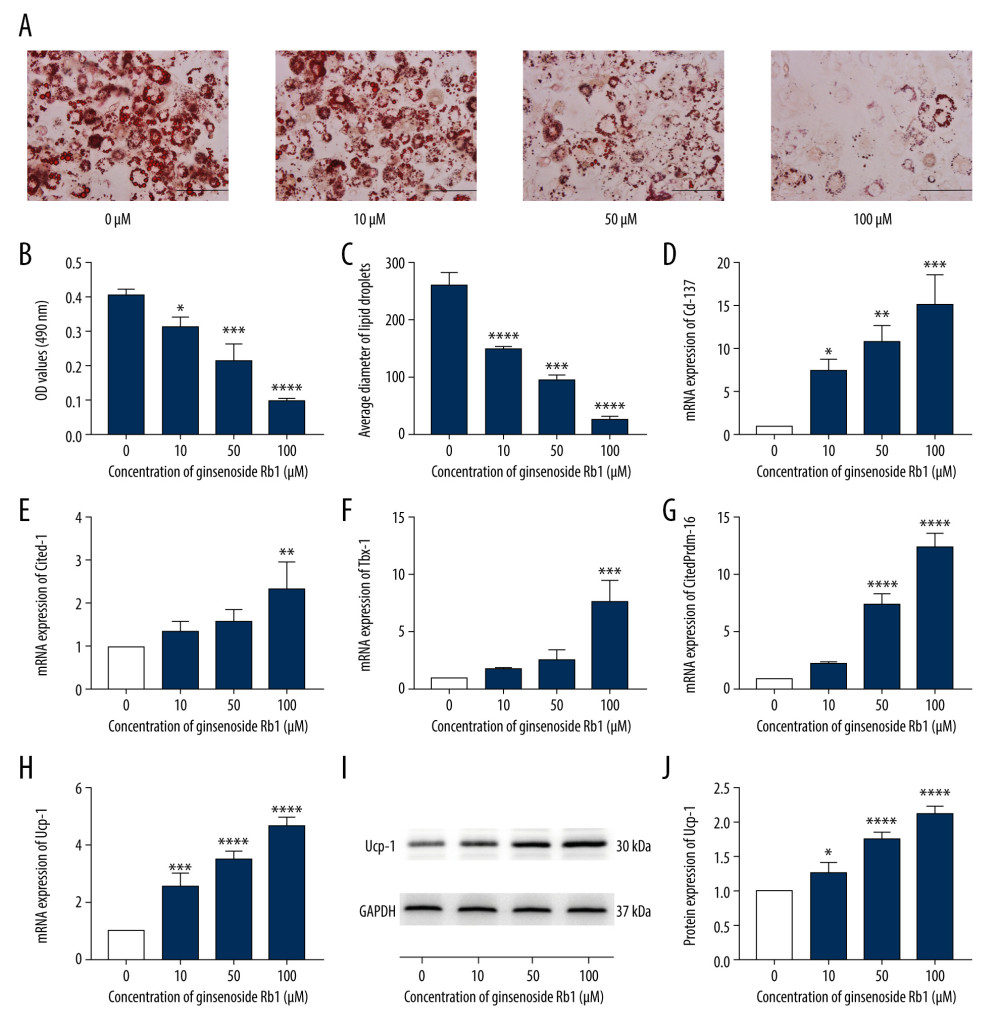 Figure 2. Effect of ginsenoside Rb1 (G-Rb1; 0, 10, 50, and 100 μM) on the browning of adipocytes. (A) Lipid droplets were measured by Oil Red O staining. Scale bar=100 μm. (B) Lipid content assessing according to the absorbance at 490 nm. (C) ImageJ measured the average diameter of lipid droplets. (D–H) Quantitative real-time polymerase chain reaction measured the mRNA expression of browning-related genes (Cd-137, Cited-1, Tbx-1, Prdm-16, and Ucp-1). (I, J) Western blotting measured the protein expression of Ucp-1. Data are expressed as mean±SD (n=3). * P<0.05, ** P<0.01, *** P<0.001, **** P<0.0001 vs. 0 μM.
Figure 2. Effect of ginsenoside Rb1 (G-Rb1; 0, 10, 50, and 100 μM) on the browning of adipocytes. (A) Lipid droplets were measured by Oil Red O staining. Scale bar=100 μm. (B) Lipid content assessing according to the absorbance at 490 nm. (C) ImageJ measured the average diameter of lipid droplets. (D–H) Quantitative real-time polymerase chain reaction measured the mRNA expression of browning-related genes (Cd-137, Cited-1, Tbx-1, Prdm-16, and Ucp-1). (I, J) Western blotting measured the protein expression of Ucp-1. Data are expressed as mean±SD (n=3). * P<0.05, ** P<0.01, *** P<0.001, **** P<0.0001 vs. 0 μM. 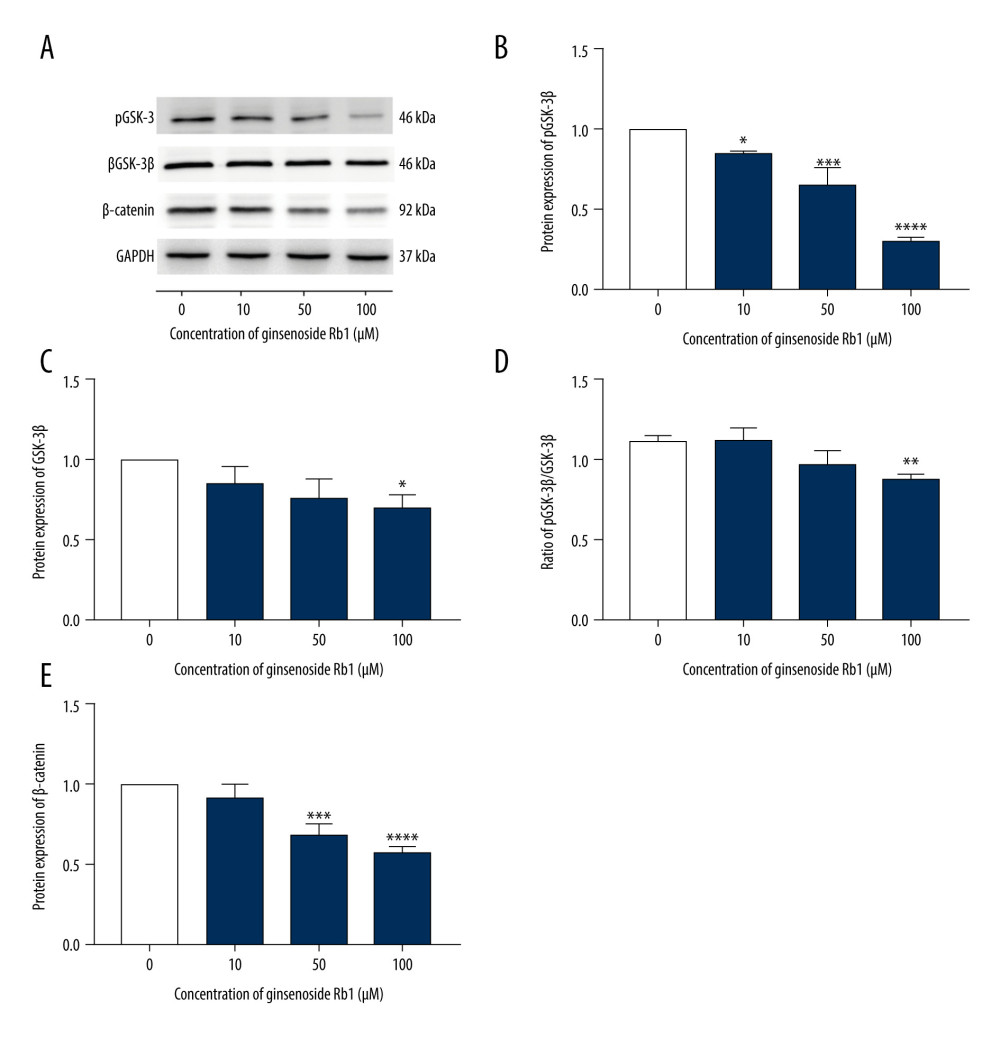 Figure 3. Effect of ginsenoside Rb1 (G-Rb1; 0, 10, 50, and 100 μM) on Wnt/β-catenin signaling. (A) Western blotting results of browning-related genes in 3T3-L1 adipocytes. (B–E) ImageJ quantized the gray value of each protein band. Data are expressed as mean±SD (n=3). * P<0.05, ** P<0.01, *** P<0.001, **** P<0.0001 vs. 0 μM.
Figure 3. Effect of ginsenoside Rb1 (G-Rb1; 0, 10, 50, and 100 μM) on Wnt/β-catenin signaling. (A) Western blotting results of browning-related genes in 3T3-L1 adipocytes. (B–E) ImageJ quantized the gray value of each protein band. Data are expressed as mean±SD (n=3). * P<0.05, ** P<0.01, *** P<0.001, **** P<0.0001 vs. 0 μM. 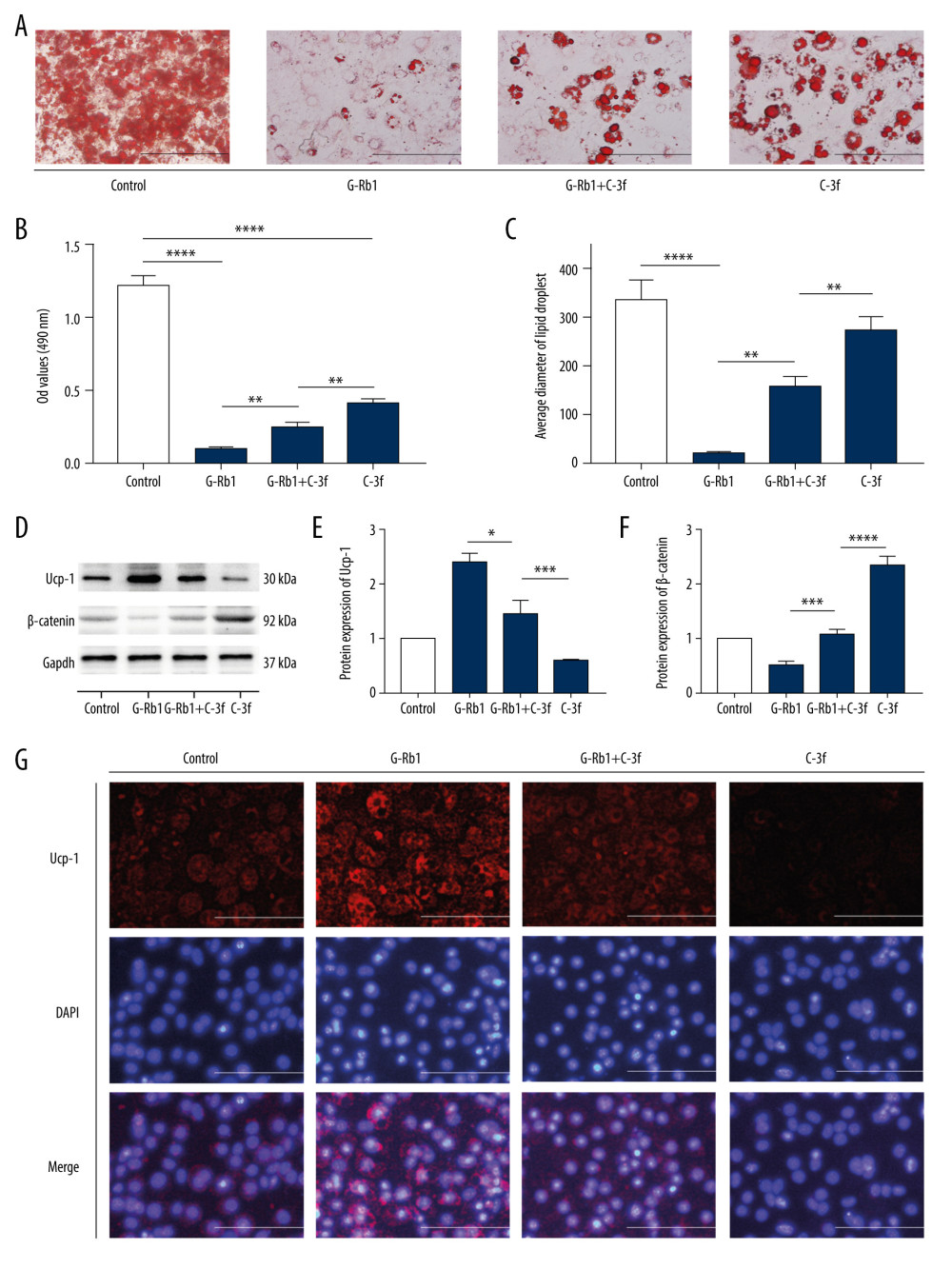 Figure 4. Effect of ginsenoside Rb1 (G-Rb1) on browning treated with Compound 3f. (A) Oil Red O staining detected the lipid droplets. Scale bar=100 μm. (B) Optical density values at 490 nm. (C) Lipid droplets’ diameter of ORS was measured by ImageJ. (D) Western blotting results for Ucp-1 and β-catenin, with Gapdh as the internal reference. (E, F) ImageJ quantized the gray value of each protein band. (G) Immunofluorescence was performed to detect the expression of Ucp-1. Scale bar=50 μm. Data are expressed as mean±SD (n=3). * P<0.05, ** P<0.01, *** P<0.001, **** P<0.0001.
Figure 4. Effect of ginsenoside Rb1 (G-Rb1) on browning treated with Compound 3f. (A) Oil Red O staining detected the lipid droplets. Scale bar=100 μm. (B) Optical density values at 490 nm. (C) Lipid droplets’ diameter of ORS was measured by ImageJ. (D) Western blotting results for Ucp-1 and β-catenin, with Gapdh as the internal reference. (E, F) ImageJ quantized the gray value of each protein band. (G) Immunofluorescence was performed to detect the expression of Ucp-1. Scale bar=50 μm. Data are expressed as mean±SD (n=3). * P<0.05, ** P<0.01, *** P<0.001, **** P<0.0001. 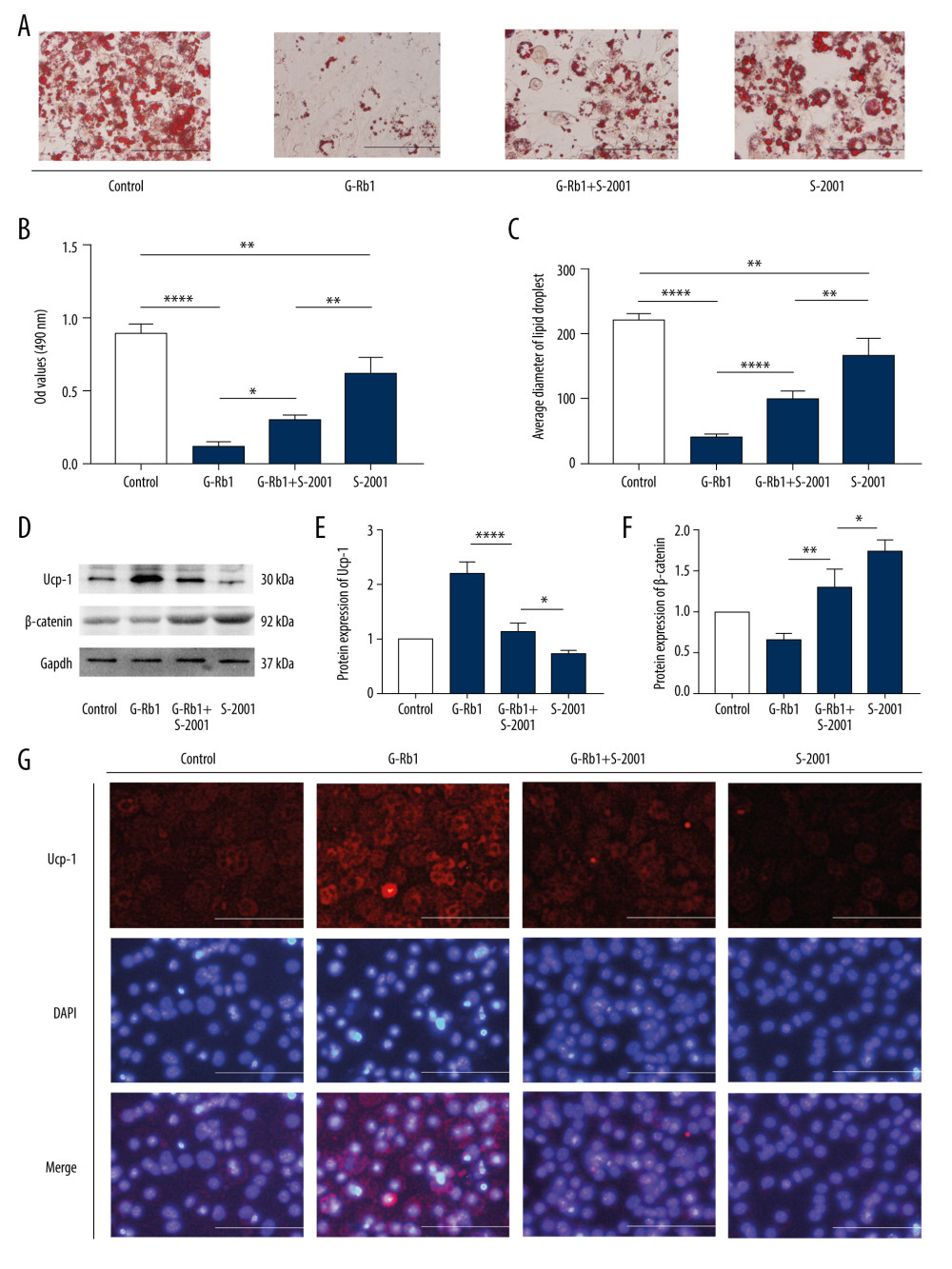 Figure 5. Effect of ginsenoside Rb1 (G-Rb1) on browning treated with SLK2001. (A) Oil Red O staining (ORS) detected the lipid droplets. Scale bar=100 μm. (B) Optical density values at 490 nm. (C) Lipid droplets’ diameter of ORS was measured by ImageJ. (D) Western blotting results of Ucp-1 and β-catenin, with Gapdh as the internal reference. (E, F) ImageJ quantized the gray value of each protein band. (G) Immunofluorescence was performed to detect the expression of Ucp-1. Scale bar=50 μm. Data are expressed as mean±SD (n=3). * P<0.05, ** P<0.01, **** P<0.0001.
Figure 5. Effect of ginsenoside Rb1 (G-Rb1) on browning treated with SLK2001. (A) Oil Red O staining (ORS) detected the lipid droplets. Scale bar=100 μm. (B) Optical density values at 490 nm. (C) Lipid droplets’ diameter of ORS was measured by ImageJ. (D) Western blotting results of Ucp-1 and β-catenin, with Gapdh as the internal reference. (E, F) ImageJ quantized the gray value of each protein band. (G) Immunofluorescence was performed to detect the expression of Ucp-1. Scale bar=50 μm. Data are expressed as mean±SD (n=3). * P<0.05, ** P<0.01, **** P<0.0001. References
1. Zhang Q, Xia X, Fang S, Yuan X, Relationship between fat mass and obesity-associated (FTO) gene polymorphisms with obesity and metabolic syndrome in ethnic Mongolians: Med Sci Monit, 2018; 24; 8232-38
2. Cuthbert K, Hardin S, Zelkowitz R, Mitchell K, Eating disorders and overweight/obesity in veterans: Prevalence, risk factors, and treatment considerations: Curr Obes Rep, 2020; 9(2); 98-108
3. Finlin BS, Zhu B, Confides AL, Mast cells promote seasonal white adipose beiging in humans: Diabetes, 2017; 66(5); 1237-46
4. Yang G, Zhuo J, Lin Y, Ginsenoside Rb1 prevents dysfunction of endothelial cells by suppressing inflammatory response and apoptosis in the high-fat diet plus balloon catheter-injured rabbit model via the G protein-coupled estrogen receptor-mediated phosphatidylinositol 3-kinases (PI3K)/Akt pathway: Med Sci Monit, 2019; 25; 7407-17
5. Hong Y, Lin Y, Si Q, Ginsenoside Rb2 alleviates obesity by activation of brown fat and induction of browning of white fat: Front Endocrinol (Lausanne), 2019; 10; 153
6. Lee K, Seo YJ, Song JH, Ginsenoside Rg1 promotes browning by inducing UCP1 expression and mitochondrial activity in 3T3-L1 and subcutaneous white adipocytes: J Ginseng Res, 2019; 43(4); 589-99
7. Kim K, Nam KH, Yi SA, Ginsenoside Rg3 induces browning of 3T3-L1 adipocytes by activating AMPK signaling: Nutrients, 2020; 12(2); 427
8. Mu Q, Fang X, Li X, Ginsenoside Rb1 promotes browning through regulation of PPARγ in 3T3-L1 adipocytes: Biochem Biophys Res Commun, 2015; 466(3); 530-35
9. Park SJ, Park M, Sharma A, Black ginseng and ginsenoside Rb1 promote browning by inducing UCP1 expression in 3T3-L1 and primary white adipocytes: Nutrients, 2019; 11(11); 2747
10. Li X, Yang J, Bao M, Wnt signaling in bone metastasis: Mechanisms and therapeutic opportunities: Life Sci, 2018; 208; 33-45
11. El-Sahli S, Xie Y, Wang L, Liu S, Wnt signaling in cancer metabolism and immunity: Cancers (Basel), 2019; 11(7); 904
12. Steinhart Z, Angers S, Wnt signaling in development and tissue homeostasis: Development, 2018; 145(11); dev146589
13. MacDonald BT, Tamai K, He X, Wnt/beta-catenin signaling: Components, mechanisms, and diseases: Dev Cell, 2009; 17(1); 9-26
14. Harada H, Tsuda Y, Yabuki K, Inhibition of WNT/β-catenin signaling under serum starvation and hypoxia induces adipocytic transdifferentiation in human leiomyoma cells: Lab Invest, 2018; 98(4); 439-48
15. Kang S, Bajnok L, Longo KA, Effects of Wnt signaling on brown adipocyte differentiation and metabolism mediated by PGC-1alpha: Mol Cell Biol, 2005; 25(4); 1272-82
16. Lo KA, Ng PY, Kabiri Z, Wnt inhibition enhances browning of mouse primary white adipocytes: Adipocyte, 2016; 5(2); 224-31
17. Xi P, Xue J, Wu Z, Liver kinase B1 induces browning phenotype in 3 T3-L1 adipocytes: Gene, 2019; 682; 33-41
18. Gerngroß C, Schretter J, Klingenspor M, Active brown fat during 18F-FDG PET/CT imaging defines a patient group with characteristic traits and an increased probability of brown fat redetection: J Nucl Med, 2017; 58(7); 1104-10
19. Tamura Y, Tomiya S, Takegaki J, Apple polyphenols induce browning of white adipose tissue: J Nutr Biochem, 2020; 77; 108299
20. Moon KM, Lee B, Cho WK, Swertiajaponin as an anti-browning and antioxidant flavonoid: Food Chem, 2018; 252; 207-14
21. Kang NH, Mukherjee S, Yun JW: Nutrients, 2019; 11(3); 577
22. Yau WW, Yen PM, Thermogenesis in adipose tissue activated by thyroid hormone: Int J Mol Sci, 2020; 21(8); 3020
23. Rodrigues KCDC, Pereira RM, de Campos TDP, The role of physical exercise to improve the browning of white adipose tissue via POMC neurons: Front Cell Neurosci, 2018; 12; 88
24. Chen N, Wang J, Wnt/β-catenin signaling and obesity: Front Physiol, 2018; 9; 792
25. Seo YJ, Kim KJ, Choi J: Nutrients, 2018; 10(6); 712
26. Qi G, Zhou Y, Zhang X, Cordycepin promotes browning of white adipose tissue through an AMP-activated protein kinase (AMPK)-dependent pathway: Acta Pharm Sin B, 2019; 9(1); 135-43
27. Han Y, Wu JZ, Shen JZ, Pentamethylquercetin induces adipose browning and exerts beneficial effects in 3T3-L1 adipocytes and high-fat diet-fed mice: Sci Rep, 2017; 7(1); 1123
28. Ross SE, Hemati N, Longo KA, Inhibition of adipogenesis by Wnt signaling: Science, 2000; 289(5481); 950-53
29. Longo KA, Wright WS, Kang S, Wnt10b inhibits development of white and brown adipose tissues: J Biol Chem, 2004; 279(34); 35503-9
Figures
 Figure 1. Effect of ginsenoside Rb1 (G-Rb1) on the adipocyte viability. (A) The structure of G-Rb1. (B) 3T3-L1 adipocytes were treated with G-Rb1 (0, 10, 50, 100, and 200 μM) for 24, 48, and 72 h. Data are expressed as mean±SD (n=10). **** P<0.0001 vs. 0 μM.
Figure 1. Effect of ginsenoside Rb1 (G-Rb1) on the adipocyte viability. (A) The structure of G-Rb1. (B) 3T3-L1 adipocytes were treated with G-Rb1 (0, 10, 50, 100, and 200 μM) for 24, 48, and 72 h. Data are expressed as mean±SD (n=10). **** P<0.0001 vs. 0 μM. Figure 2. Effect of ginsenoside Rb1 (G-Rb1; 0, 10, 50, and 100 μM) on the browning of adipocytes. (A) Lipid droplets were measured by Oil Red O staining. Scale bar=100 μm. (B) Lipid content assessing according to the absorbance at 490 nm. (C) ImageJ measured the average diameter of lipid droplets. (D–H) Quantitative real-time polymerase chain reaction measured the mRNA expression of browning-related genes (Cd-137, Cited-1, Tbx-1, Prdm-16, and Ucp-1). (I, J) Western blotting measured the protein expression of Ucp-1. Data are expressed as mean±SD (n=3). * P<0.05, ** P<0.01, *** P<0.001, **** P<0.0001 vs. 0 μM.
Figure 2. Effect of ginsenoside Rb1 (G-Rb1; 0, 10, 50, and 100 μM) on the browning of adipocytes. (A) Lipid droplets were measured by Oil Red O staining. Scale bar=100 μm. (B) Lipid content assessing according to the absorbance at 490 nm. (C) ImageJ measured the average diameter of lipid droplets. (D–H) Quantitative real-time polymerase chain reaction measured the mRNA expression of browning-related genes (Cd-137, Cited-1, Tbx-1, Prdm-16, and Ucp-1). (I, J) Western blotting measured the protein expression of Ucp-1. Data are expressed as mean±SD (n=3). * P<0.05, ** P<0.01, *** P<0.001, **** P<0.0001 vs. 0 μM. Figure 3. Effect of ginsenoside Rb1 (G-Rb1; 0, 10, 50, and 100 μM) on Wnt/β-catenin signaling. (A) Western blotting results of browning-related genes in 3T3-L1 adipocytes. (B–E) ImageJ quantized the gray value of each protein band. Data are expressed as mean±SD (n=3). * P<0.05, ** P<0.01, *** P<0.001, **** P<0.0001 vs. 0 μM.
Figure 3. Effect of ginsenoside Rb1 (G-Rb1; 0, 10, 50, and 100 μM) on Wnt/β-catenin signaling. (A) Western blotting results of browning-related genes in 3T3-L1 adipocytes. (B–E) ImageJ quantized the gray value of each protein band. Data are expressed as mean±SD (n=3). * P<0.05, ** P<0.01, *** P<0.001, **** P<0.0001 vs. 0 μM. Figure 4. Effect of ginsenoside Rb1 (G-Rb1) on browning treated with Compound 3f. (A) Oil Red O staining detected the lipid droplets. Scale bar=100 μm. (B) Optical density values at 490 nm. (C) Lipid droplets’ diameter of ORS was measured by ImageJ. (D) Western blotting results for Ucp-1 and β-catenin, with Gapdh as the internal reference. (E, F) ImageJ quantized the gray value of each protein band. (G) Immunofluorescence was performed to detect the expression of Ucp-1. Scale bar=50 μm. Data are expressed as mean±SD (n=3). * P<0.05, ** P<0.01, *** P<0.001, **** P<0.0001.
Figure 4. Effect of ginsenoside Rb1 (G-Rb1) on browning treated with Compound 3f. (A) Oil Red O staining detected the lipid droplets. Scale bar=100 μm. (B) Optical density values at 490 nm. (C) Lipid droplets’ diameter of ORS was measured by ImageJ. (D) Western blotting results for Ucp-1 and β-catenin, with Gapdh as the internal reference. (E, F) ImageJ quantized the gray value of each protein band. (G) Immunofluorescence was performed to detect the expression of Ucp-1. Scale bar=50 μm. Data are expressed as mean±SD (n=3). * P<0.05, ** P<0.01, *** P<0.001, **** P<0.0001. Figure 5. Effect of ginsenoside Rb1 (G-Rb1) on browning treated with SLK2001. (A) Oil Red O staining (ORS) detected the lipid droplets. Scale bar=100 μm. (B) Optical density values at 490 nm. (C) Lipid droplets’ diameter of ORS was measured by ImageJ. (D) Western blotting results of Ucp-1 and β-catenin, with Gapdh as the internal reference. (E, F) ImageJ quantized the gray value of each protein band. (G) Immunofluorescence was performed to detect the expression of Ucp-1. Scale bar=50 μm. Data are expressed as mean±SD (n=3). * P<0.05, ** P<0.01, **** P<0.0001.
Figure 5. Effect of ginsenoside Rb1 (G-Rb1) on browning treated with SLK2001. (A) Oil Red O staining (ORS) detected the lipid droplets. Scale bar=100 μm. (B) Optical density values at 490 nm. (C) Lipid droplets’ diameter of ORS was measured by ImageJ. (D) Western blotting results of Ucp-1 and β-catenin, with Gapdh as the internal reference. (E, F) ImageJ quantized the gray value of each protein band. (G) Immunofluorescence was performed to detect the expression of Ucp-1. Scale bar=50 μm. Data are expressed as mean±SD (n=3). * P<0.05, ** P<0.01, **** P<0.0001. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387