10 January 2021: Clinical Research
Personality Traits, Stress, and Emotional Intelligence Associated with Central Serous Chorioretinopathy
Anita Pusic Sesar1ABCDEFG, Antonio Sesar1ACDEF, Kajo Bucan2DFG, Irena Sesar1ADG, Katarina Cvitkovic1BCFG, Ivan Cavar3ACDEF*DOI: 10.12659/MSM.928677
Med Sci Monit 2021; 27:e928677
Abstract
BACKGROUND: The aim of the present study was to investigate the relationship between personality traits, stress, emotional intelligence, and central serous chorioretinopathy (CSCR).
MATERIAL AND METHODS: This prospective case-control study included 57 patients with acute CSCR and 57 age- and sex-matched controls with refractive errors. Inclusion criteria for CSCR group were acute unilateral onset of visual disturbances within 2 weeks until the first visit to the ophthalmologist and ophthalmoscopic finding of a round or oval macular detachment confirmed by optical coherence tomography as a dome-shaped serous neuroretinal elevation.
RESULTS: Using the Sixteen Personality Factor Questionnaire (16 PF), patients with CSCR achieved slightly higher scores on primary characteristics such as warmth (P=0.612) and perfectionism (P=0.137) when compared to the control subjects. Mean scores measured with the Social Readjustment Rating Scale (SRRS) were significantly higher in patients with CSCR (P=0.004), which means that these patients had notably elevated average reactivity to stressful life events. In addition, the number of patients with a high stress level was higher in the CSCR group than in the control group. Considering the level of emotional intelligence measured with the Trait Emotional Intelligence Questionnaire-Short Form (TEIQue-SF), patients with CSCR achieved significantly lower scores on well-being (P=0.003) and sociability (P=0.011) factors, as well as on total score (P=0.014).
CONCLUSIONS: A higher level of perceived stress is the most important psychological risk factor for CSCR. According to our results, a low level of emotional intelligence may be an additional factor that contributes to the occurrence of CSCR.
Keywords: central serous chorioretinopathy, emotional intelligence, Stress, Psychological, Type A Personality
Background
Central serous chorioretinopathy (CSCR) is a disorder characterized by localized serous detachment of the neurosensory retina in the macula, and it is often associated with detachment of the retinal pigment epithelium (RPE). It is ranked as the fourth most frequent non-surgical retinopathy, with a reported annual incidence of 9.9 for men and 1.7 for women per 100 000 individuals [1,2]. CSCR typically occurs unilaterally in young and middle-aged persons, with a peak of prevalence at around 45 years [1,3]. Although the pathogenetic mechanism is still unclear, it is assumed that the hyperpermeability of choroidal blood vessels causes an increase in tissue hydrostatic pressure, which in turn results in subretinal fluid accumulation and damage to the RPE [4]. CSCR is usually classified into acute and chronic forms based on the duration of subretinal fluid and the structural macular changes detected by multimodal imaging [5,6]. Common clinical presentations of acute CSCR include blurred vision, metamorphopsia, relative central scotoma, reduced contrast sensitivity, and dome-shaped serous neuroretinal elevation seen on optical coherence tomography (OCT). In most cases, spontaneous resolution of subretinal fluid occurs within 3–4 months, with recovery of visual function, although recurrent episodes or persistent fluid can occur in some patients [3,5,6]. Chronic CSCR is characterized by serous retinal detachment for longer than 4–6 months, associated with widespread RPE atrophy, photoreceptor damage, and permanent vision loss [2,5–7]. There is no criterion standard treatment of CSCR, and numerous therapeutic options have been proposed, including standard laser photocoagulation, subthreshold micropulse laser, photodynamic therapy with verteporfin, intravitreal administration of anti-vascular endothelial growth factor drugs, oral mineralocorticoid antagonists, psychological support, and reduction and avoidance of stress [3,4,6,8].
Over time, many risk factors have been described as possible causes of CSCR, such as hypertension, autoimmune diseases, pregnancy, gastroesophageal reflux, use of steroids, sleep disturbances, and use of psycho-pharmacological drugs [2,9,10]. Additionally, the impact of psychological risk factors, such as personality type A and stress, is well documented [9,11,12]. Besides the well-described characteristics of personality type A, such as a competitive drive, a sense of urgency, an aggressive nature, and a hostile temperament, other personality and temperament characteristics, including emotional dissociation, a lower level of cooperativeness, reduced tolerance to frustration, emotional instability, and insecurity, have all been related to the development of CSCR [13,14]. Likewise, it has been demonstrated that CSCR patients reported more critical life events, unfavorable stress coping strategies, and psychosomatic problems [15].
Despite numerous studies, a clear personality profile and reactivity to stress in CSCR patients are yet to be fully established. Furthermore, it has been suggested that emotional intelligence could have a “stress-buffering” effect and favor faster recovery after an acute stress event [16]. To the best of our knowledge, no study has evaluated the association between levels of emotional intelligence and CSCR.
Based on previous research, the aim of this study was to clarify association of personality traits, stress, and emotional intelligence with acute CSCR.
Material and Methods
PATIENTS:
A prospective case-control study was conducted at the Department of Ophthalmology, University Clinical Hospital Mostar, Mostar, Bosnia and Herzegovina, between November 2017 and June 2020. A total of 114 subjects were included in the study, 57 with acute CSCR (cases) and 57 with refractive errors between −3.00 diopters (D) and +3.00 D without other associated ocular abnormalities (controls). The control subjects were matched by sex and age (±2 years) to the case group. Inclusion criteria for CSCR group were acute unilateral onset of visual disturbances (vision loss, metamorphopsia, micropsia, dyschromatopsia or central scotoma) within 2 weeks until the first visit to the ophthalmologist and ophthalmoscopic finding of a round or oval macular detachment confirmed by OCT as dome-shaped serous neuroretinal elevation. Patients were excluded from the study if they presented any of the following: structural damages in the macula suggestive of previous CSCR episodes (e.g., atrophic changes to the outer retina and RPE), other maculopathies capable of causing macular exudation (e.g., choroidal neovascularization, age-related macular degeneration, pathologic myopia), surgical and/or laser procedures on an eye, and other ocular pathology that significantly reduced visual acuity.
Patients who were selected for the study were interviewed and gave written informed consent prior to participation in the study. All procedures and examinations were approved by the Ethics Committee of the University Clinical Hospital Mostar. The study was in compliance with the Declaration of Helsinki.
METHODS:
All participants underwent a standard ophthalmological examination that included best-corrected visual acuity (BCVA), applanation tonometry, slit lamp biomicroscopy, dilated fundus examination, and OCT (Optopol Technology Sp. z o.o., Zawiercie, PL). Patients with CSCR were observed for 4 months, and within this period they received no treatment. All respondents filled out 4 questionnaires.
STATISTICAL ANALYSIS:
Statistical analysis was performed with IBM SPSS Statistics for Windows, Version 25.0. (IBM Corp., Armonk, NY, USA). Categorical variables are presented as the numbers with proportions. The differences between groups for categorical variables were compared using the chi-square test. Normality of numerical variables was tested using the Shapiro-Wilks test. Numerical variables are presented as mean and standard deviation (M±SD) or median and interquartile range (C [IQR]). The differences between groups for numerical variables were compared using the
Results
Baseline characteristics of CSCR and control subjects are shown in Table 1. There were 37 (64.9%) males and 20 (35.1%) females in both groups of examinees. Males were significantly more represented than females (χ2=5.070; p=0.024).
Systemic risk factors for CSCR are presented in Table 2. No statistically significant difference was found between the CSCR and control group for any of the analyzed variables. However, univariate logistic regression analysis showed that patients with autoimmune diseases (OR, 95% CI: 5.385 [0.609–47.632]), gastroesophageal reflux (OR, 95% CI: 2.036 [0.179–23.110]), sleep disturbances (OR, 95% CI: 1.264 [0.582–2.745]), and hypertension (OR, 95% CI: 1.115 [0.446–2.786]) were more likely to have CSCR.
Expression of primary personality traits using 16 PF is presented in Table 3. Patients with CSCR achieved higher scores on primary characteristics such as warmth (
Measurement of stress levels using SRRS in CSCR patients and the controls is shown in Table 4. The comparison results clearly show that the number of patients with a high stress level, with an 80% chance of a major health breakdown in the next 2 years, was 3 times higher in the CSCR group than in the control group. Furthermore, mean scores of the SRRS were significantly higher in patients with CSCR compared to the controls (
Table 5 reveals the comparison of the emotional intelligence levels between the patients with CSCR and the controls. Differences in the expression of emotional intelligence between the groups of examinees were significant. Patients with CSCR achieved significantly lower scores on the well-being (
Discussion
This research shows that systemic risk factors, including hypertension, autoimmune disease, use of steroids, gastroesophageal reflux, pregnancy, sleep disturbances, and use of psycho-pharmacologic drugs, were slightly more prevalent in CSCR patients compared to the matched controls. Tittl et al. reported that individuals with CSCR were more likely to use psycho-pharmacologic medications, suggesting higher levels of psychological stress and increased sympathetic nervous system activity in these subjects [12]. Ji et al. found that prevalence of poor sleep quality in CSCR patients was significantly higher than in healthy controls [22]. However, no significant difference was noticed in obstructive sleep apnea tendency after adjusting CSCR patients with controls based on body mass index, which is a significant risk factor for sleep apnea [22,23]. Furthermore, considering other systemic risk factors, our results are comparable to the results of other studies, which revealed that these factors predispose individuals to the development of CSCR [2,9,24,25].
In this study, we found that patients with acute CSCR had slightly more pronounced primary characteristics, such as warmth, a basic personality type A feature, and perfectionism in comparison to the control group. Yannuzzi suggested that personal style of behavior, personality traits, and values may be risk factors for the development of CSCR in individuals with personality type A, characterized by a competitive drive, a sense of urgency, an aggressive nature, and a hostile temperament [11]. A prospective study conducted in Greece confirmed the association between personality type A and CSCR [24]. Furthermore, Spahn et al. assessed the personality profiles of 24 CSCR patients, 12 of whom had relapsed CSCR. The authors reported that patients with CSCR were more emotionally unstable and insecure, as well as flexible and spontaneous [14]. By contrast, van Haalen et al. showed that personality type A is not more prevalent in patients with chronic CSCR, which may be the result of more successful use of certain coping strategies to improve self-management [26]. Although many authors suggest an association between CSCR and some personality type features, it is still not possible to assemble a specific personality profile in these patients.
In our study, a higher level of perceived stress was detected in the CSCR group compared to the controls. Likewise, the CSCR subjects reported 3 times more severe stress episodes in the past year than reported by healthy subjects. In 1927, Horniker proposed an angiospastic theory of CSCR development, assuming that psychological disorders could cause or contribute to the onset of the disease [27]. A higher incidence of CSCR has been described in military personnel, which is attributed to psychological trauma and greater exposure to stressful situations during war [28]. Gelber and Schatz reported that very disturbing psychological events preceded vision loss in 91% of CSCR cases. They noted that acute stress disorders preceded the first visual symptoms of CSCR by an average of 7 days [29]. Various stressful situations that can affect the individual or be related to the general population, have been considered in the context of a higher incidence of CSCR, including divorce, bankruptcy, shift work, or periods of political and economic crisis in the affected countries [1,30–32]. Conrad et al. reported that CSCR patients are more vulnerable to stress due to inappropriate coping strategies [33]. Furthermore, CSCR patients reported having more critical life events, poor stress coping strategies, and noticeable psychosomatic problems, which were more pronounced in patients with acute rather than chronic CSCR [15]. In addition, an elevated stress score in CSCR patients has been reported to be highly correlated with serum cortisol levels [34,35]. Numerous studies have documented the association between therapeutic use of corticosteroids and the development of CSCR [36–38]. However, elevated levels of sympathetic activity and elevated plasma concentrations of epinephrine and norepinephrine have been found in patients with CSCR [39,40]. Catecholamines and glucocorticoids are mediators released into the body in response to psychological stress [41], and some authors speculate that they may be responsible for changes in autoregulation of choroidal blood flow and, consequently, the development of CSCR [12,42].
Analysis of the emotional intelligence level showed that CSCR subjects had significantly lower scores on the well-being and sociability factors, as well as on the total score of TEIQue-SF. Recent studies suggest that emotional intelligence can act as a “stress buffer”. The proposed theory assumes that individuals with a higher level of emotional intelligence, when confronted with a stressful situation, show more adaptive responses such as decreased reactivity to stress and faster recovery after a stressful event [16]. Singh and Sharma reported that a high level of emotional intelligence was inversely related to the levels of acute and chronic perceived stress, whereas general intelligence showed no association with either perceived stress or reactivity to stress [43]. Furthermore, higher emotional intelligence scores were reported to be associated with notably reduced stress reactivity at the psychological level, as well as at the biological level as measured by salivary cortisol levels [44]. Within certain populations, the association of emotional intelligence with personality dimensions such as neuroticism or a specific personality type (such as personality type A) has also been described. Neuroticism was very negatively associated with emotional intelligence, while in type A subjects, stress symptoms were notably predicted by the insufficient use of effective coping strategies and weaknesses in the general mood component of emotional intelligence [45,46]. Our results suggest that certain components of emotional intelligence may play a role in the occurrence of acute CSCR. Lower levels of emotional intelligence need to be considered primarily in the context of a higher reactivity to stressful events, which may require use of appropriate coping strategies. Therefore, an integrated approach that, in addition to medical treatment, includes adequate psychosocial support could be beneficial in patients with CSCR.
There are some limitations of this study. First, we did not include all the possible risk factors for CSCR, such as anxiety, depression, and obsessive-compulsive behavior, which would have made the study more comprehensive. Second, a multicenter study is needed to make the results more thorough and conclusive.
Conclusions
A higher level of perceived stress is the most important psychological risk factor for CSCR. Although there is still no consensus, it seems reasonable to suspect that stress is a major factor that promotes biological processes, including hormonal, neural, and inflammatory mediators, which could be responsible for the development of CSCR. According to our results, a low level of emotional intelligence may be an additional factor contributing to the occurrence of CSCR. Despite the existing knowledge, it remains unclear how a variety of psychological factors can influence the pathological changes in the choroid and the RPE. Further longitudinal studies with a larger sample of subjects need to be conducted to more exactly identify the relationship between certain personality characteristics, emotional intelligence levels, and susceptibility to stress in patients with CSCR.
Tables
Table 1. Baseline characteristics of patients with acute CSCR and controls. Table 2. Presence of systemic risk factors in the CSCR and control group.
Table 2. Presence of systemic risk factors in the CSCR and control group.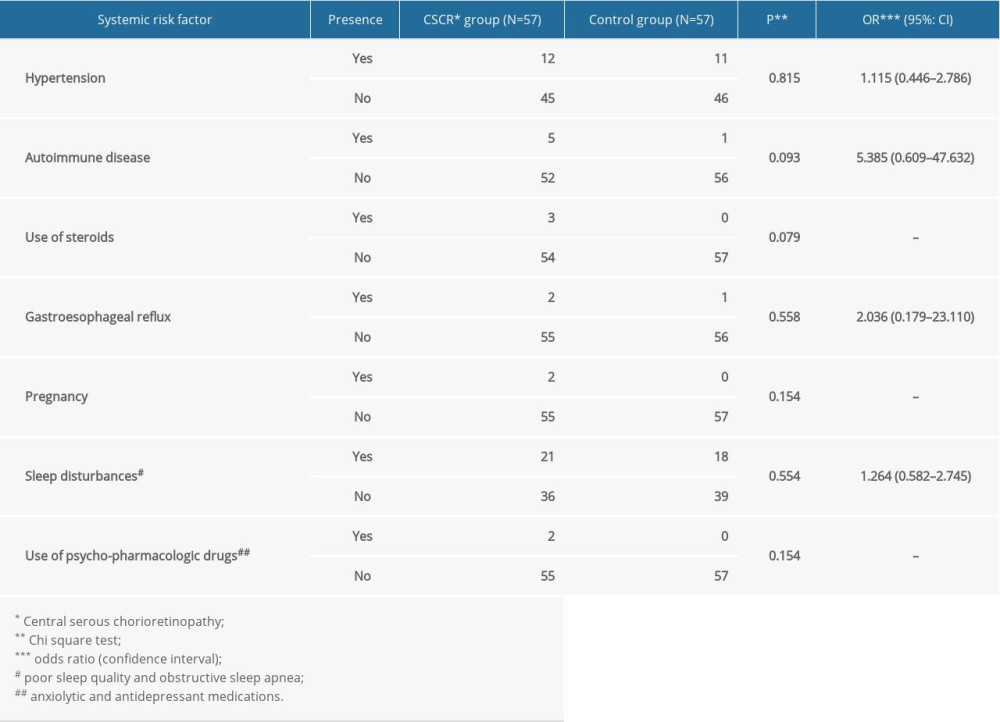 Table 3. Personality traits of the CSCR patients and controls according to the 16 PF.
Table 3. Personality traits of the CSCR patients and controls according to the 16 PF.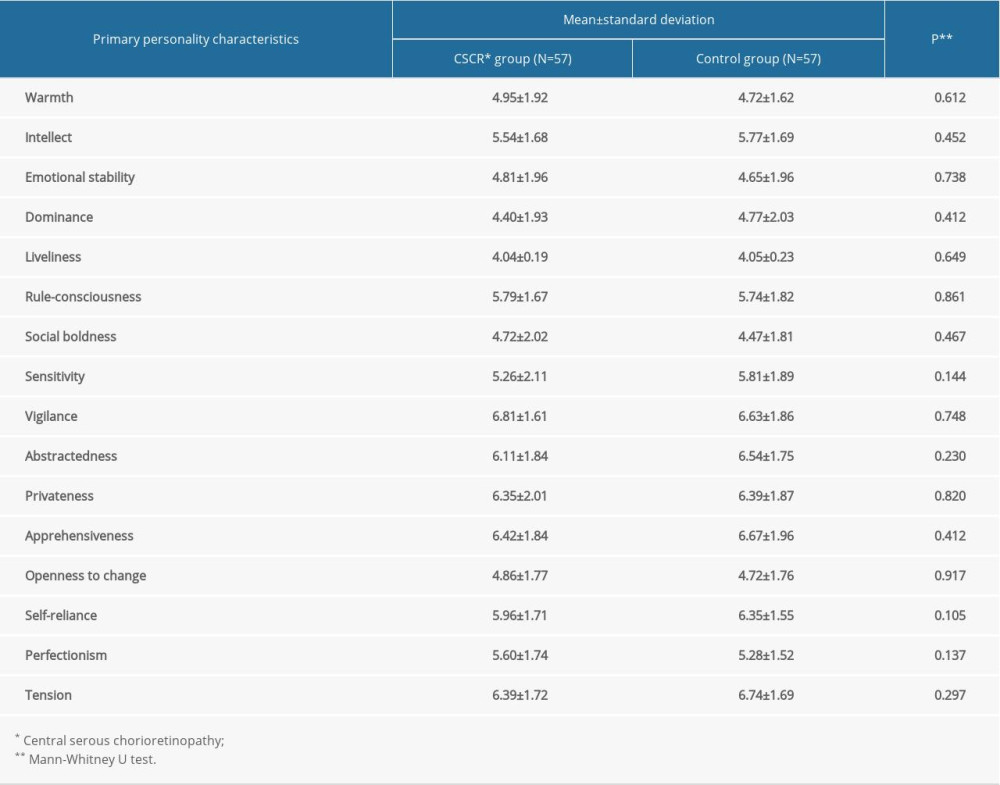 Table 4. The level of stress in the CSCR patients and controls measured by SRRS.
Table 4. The level of stress in the CSCR patients and controls measured by SRRS.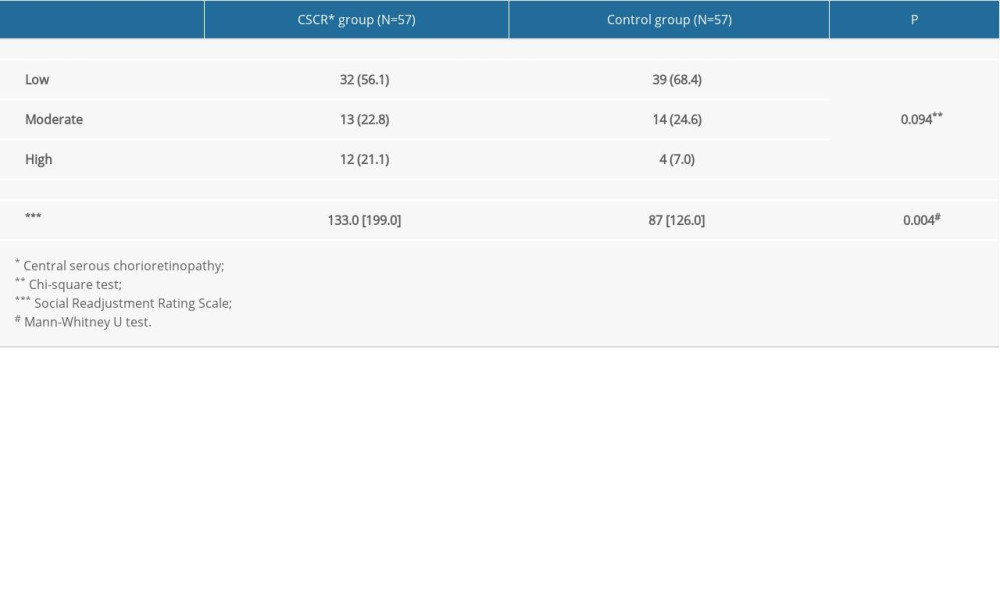 Table 5. The level of emotional intelligence in the CSCR patients and controls measured by TEIQue-SF.
Table 5. The level of emotional intelligence in the CSCR patients and controls measured by TEIQue-SF.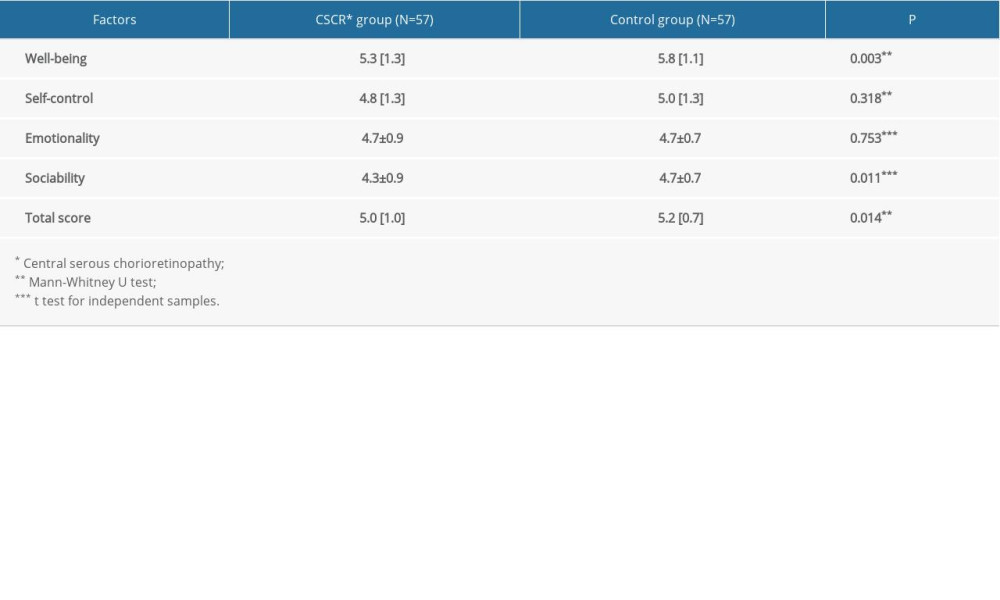
References
1. Wang M, Munch IC, Hasler PW, Central serous chorioretinopathy: Acta Ophthalmol, 2008; 86; 126-45
2. Kitzmann AS, Pulido JS, Diehl NN, The incidence of central serous chorioretinopathy in Olmsted County, Minnesota, 1980–2002: Ophthalmology, 2008; 115; 169-73
3. Liegl R, Ulbig MW, Central serous chorioretinopathy: Ophthalmologica, 2014; 232; 65-76
4. Nicholson B, Noble J, Forooghian F, Meyerle C, Central serous chorioretinopathy: Update on pathophysiology and treatment: Surv Ophthalmol, 2013; 58; 103-26
5. Daruich A, Matet A, Dirani A, Central serous chorioretinopathy: Recent findings and new physiopathology hypothesis: Prog Retin Eye Res, 2015; 48; 82-118
6. van Rijssen TJ, van Dijk EHC, Yzer S, Central serous chorioretinopathy: Towards an evidence-based treatment guideline: Prog Retin Eye Res, 2019; 73; 100770
7. Fok ACT, Chan PPM, Lam DSC, Lai TYY, Risk factors for recurrence of serous macular detachment in untreated patients with central serous chorioretinopathy: Ophthalmic Res, 2011; 46; 160
8. Iaccono P, Battaglia Parodi M, Central serous chorioretinopathy treatments: A mini review: Ophthalmic Res, 2016; 55; 76-83
9. Liu B, Deng T, Zhang J, Risk factors for central serous chorioretinopathy. A systematic review and meta-analysis: Retina, 2016; 36; 9-19
10. Haimovici R, Koh S, Gagnon DR, Risk factors for central serous chorioretinopathy: A case-control study: Ophthalmology, 2004; 111; 244-49
11. Yannuzzi LA, Type A behavior and central serous chorioretinopathy: Trans Am Ophthalmol Soc, 1986; 84; 799-845
12. Tittl MK, Spaide RF, Wong D, Systemic findings associated with central serous chorioretinopathy: Am J Ophthalmol, 1999; 128; 63-68
13. Conrad R, Geiser F, Kleiman A, Temperament and character personality profile and illness-related stress in central serous chorioretinopathy: ScientificWorldJournal, 2014; 16(2014); 631687
14. Spahn C, Wiek J, Burger T, Hansen L, Psychosomatic aspects in patients with central serous chorioretinopathy: Br J Ophthalmol, 2003; 87; 704-8
15. Lahousen T, Painold A, Luxenberger W, Psychological factors associated with acute and chronic central serous chorioretinopathy: Nord J Psychiatry, 2016; 70; 24-30
16. Lea RG, Davis SK, Mahoney B, Qualter P, Does emotional intelligence buffer the effects of acute stress? A systematic review: Front Psychol, 2019; 10; 810
17. Cattell RB, Eber HW, Tatsuoka MM: The handbook for the Sixteen Personality Factor Questionnaire, 1970, Champaign, Institute for Personality and Ability Testing IL, U.S.A
18. Matešić K, Pinjušić K: Hrvatske norme za 16PF upitnik, Zagreb, Croatia, 2007
19. Holmes TH, Rahe RH, The Social Readjustment Rating Scale: J Psychosom Res, 1967; 11; 213-18
20. Petrides KV: Technical manual for the Trait Emotional Intelligence Questionnaires (TEIQue), 2009, London, London Psychometric Laboratory, U.K
21. Letina S: Family study of emotional intelligence, 2007, Zagreb, Croatia, Faculty of Philosophy
22. Ji Y, Li M, Zhang X, Poor sleep quality is the risk factor for central serous chorioretinopathy: J Ophthalmol, 2018; 2018 9450297
23. Brodie F, Charlson ES, Aleman T, Obstructive sleep apnea and central serous chorioretinopathy: Retina, 2015; 35; 238-43
24. Chatziralli I, Kabanarou SA, Parikakis E, Risk factors for central serous chorioretinopathy: Multivariate approach in a case-control study: Curr Eye Res, 2017; 42; 1069-73
25. Tsai DC, Chen SJ, Huang CC, Epidemiology of idiopathic central serous chorioretinopathy in Taiwan, 2001–2006: A population-based study: PLoS One, 2013; 8; e66858
26. van Haalen FM, van Dijk EHC, Andela CD, Maladaptive personality traits, psychological morbidity and coping strategies in chronic central serous chorioretinopathy: Acta Ophthalmol, 2019; 97; e572-79
27. Horniker E, Su di una forma retinite centrale di origine vasoneurotica (retinite central capillaro spastica): Ann Otal, 1927; 55; 578-600 [in Italian]
28. Zeligs M, Central angiospastic retinopathy. A psychosomatic study of the occurrence in military personnel: Psychosom Med, 1947; 9; 110-17
29. Gelber GS, Schatz H, Loss of vision due to central serous chorioretinopathy following psychological stress: Am J Psychiatry, 1987; 144; 46-50
30. Bousquet E, Dhundass M, Lehmann M, Shift work: A risk factor for central serous chorioretinopathy: Am J Ophthalmol, 2016; 165; 23-28
31. Fanny A, Gbé K, Coulibaly F, Central serous chorioretinopathy: A study of six cases observed in Abidjan between 2003 and 2005, suggesting a role played by the Ivorian political-military crisis: J Fr Ophthalmol, 2008; 31; 1018-24
32. Rouvas AA, Chatziralli IP, Ladas ID, The impact of financial crisis on central serous chorioretinopathy in Greece: Is there any correlation?: Eur J Ophthalmol, 2014; 24; 559-65
33. Conrad R, Bodeewes I, Schiling G, Central serous chorioretinopathy and psychological stress: Ophthalmologe, 2000; 97; 527-31
34. Agarwal A, Garg M, Dixit N, Godara R, Evaluation and correlation of stress scores with blood pressure, endogenous cortisol levels, and homocysteine levels in patients with central serous chorioretinopathy and comparison with age-matched controls: Indian J Ophthalmol, 2016; 64; 803-5
35. Garg SP, Dada T, Talward D, Psychological factors in the etiopathogenesis of central serous chorioretinopathy: Ann Ophthalmol, 2005; 37; 201-5
36. Carvalho-Recchia CA, Yannuzzi LA, Negrão S, Corticosteroids and central serous chorioretinopathy: Ophthalmology, 2002; 109; 1834-37
37. Bouzas EA, Karadimas P, Pournaras CJ, Central serous chorioretinopathy and glucocorticoids: Surv Ophthalmol, 2002; 47; 431-48
38. Nicholson BP, Atchison E, Idris AA, Bakri SJ, Central serous chorioretinopathy and glucocorticoids: An update on evidence for association: Surv Ophthalmol, 2018; 63; 1-8
39. Tewari HK, Gadia R, Kumar D, Sympathetic-parasympathetic activity and reactivity in central serous chorioretinopathy: A case-control study: Invest Ophthalmol Vis Sci, 2006; 47; 3474-78
40. Sun J, Tan J, Wang Z, Effect of catecholamine on central serous chorioretinopathy: J Huazhong Univ Sci Technolog Med Sci, 2003; 23; 313-16
41. Lucassen PJ, Pruessner J, Sousa N, Neuropathology of stress: Acta Neuropathol, 2014; 127; 109-35
42. Kim HM, Ahn J, Kim TW, Psychological factors associated with central serous chorioretinopathy: J Psychol Psychother, 2016; 6; 250
43. Singh Y, Sharma R, Relationship between general intelligence, emotional intelligence, stress levels and stress reactivity: Ann Neurosci, 2012; 19; 107-11
44. Mikolajczak M, Roy E, Luminet O, The moderating impact of emotional intelligence on free cortisol responses to stress: Psychoneuroendocrinology, 2007; 32; 1000-12
45. Abe K, Niwa M, Fujisaki K, Suzuki Y, Associations between emotional intelligence, empathy and personality in Japanese medical students: BMC Med Educ, 2018; 18; 47
46. Hisli Sahin N, Güler M, Basim HN, The relationship between cognitive intelligence, emotional intelligence, coping and stress symptoms in the context of type A personality pattern: Turk Psikiyatri Derg, 2009; 20; 243-54
Tables
 Table 1. Baseline characteristics of patients with acute CSCR and controls.
Table 1. Baseline characteristics of patients with acute CSCR and controls. Table 2. Presence of systemic risk factors in the CSCR and control group.
Table 2. Presence of systemic risk factors in the CSCR and control group. Table 3. Personality traits of the CSCR patients and controls according to the 16 PF.
Table 3. Personality traits of the CSCR patients and controls according to the 16 PF. Table 4. The level of stress in the CSCR patients and controls measured by SRRS.
Table 4. The level of stress in the CSCR patients and controls measured by SRRS. Table 5. The level of emotional intelligence in the CSCR patients and controls measured by TEIQue-SF.
Table 5. The level of emotional intelligence in the CSCR patients and controls measured by TEIQue-SF. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








