30 April 2021: Clinical Research
Comparison of Different Hydration Strategies in Patients with Very Low-Risk Profiles of Contrast-Induced Nephropathy
Shuai Miao1BCE, Zheng-Kai Xue1B, Ya-Ru Zhang1F, Hao Zhang1A, Jing-Jin Che1C, Tong Liu1DF, Hua-Yue Tao2C, Guangping Li1AD, Kang-Yin Chen1ADG*DOI: 10.12659/MSM.929115
Med Sci Monit 2021; 27:e929115
Abstract
BACKGROUND: Hydration remains the mainstay of contrast-induced nephropathy (CIN) prevention, and new biomarkers of cystatin C (Cys C) and neutrophil gelatinase-associated lipocalin (NGAL) have been suggested. This study aimed to explore whether hydration is essential in patients with very low-risk profiles of CIN who are undergoing coronary angiography.
MATERIAL AND METHODS: A total of 150 patients were enrolled and randomly distributed to 3 groups: the Preventive Group (n=50, saline hydration was given 6 h before the procedure until 12 h after the procedure), the Remedial Group (n=50, saline hydration was given after procedure for 12 h), and the No Hydration (NH) group (n=50, saline was only given during the procedure). Serum creatinine (Cr), Cys C, and urinary NGAL were tested 3 times at different times.
RESULTS: Six patients were excluded because of Mehran risk score >2. There was no CIN among 144 individuals. At 24 h and at 72 h after the procedure, we found no significant differences in the levels of Cr and Cys C (0.72±0.11 mg/L for the Preventive Group, 0.67±0.14 mg/L for the Remedial Group, and 0.70±0.1 6 mg/L for the NH Group) among the 3 groups. Urinary NGAL also did not differ significantly among the 3 groups at 6 h or at 48 h (6.31±6.60 ng/ml for the Preventive Group, 5.00±5.86 ng/ml for the Remedial Group, and 6.97±6.37 ng/ml for the NH Group) after the procedure. Subgroup analysis in patients who underwent percutaneous coronary intervention (PCI) showed that there was no significant difference in serum Cr, Cys C, or urinary NGAL at different time points among the 3 groups.
CONCLUSIONS: Saline hydration during the perioperative period might be unnecessary in patients with very low-risk profiles of CIN.
Keywords: cystatin C, Fluid Therapy, Lipocalins, Nephrosis, Lipoid, Contrast Media, Coronary Angiography, Creatinine, Kidney Diseases, Lipocalin-2, percutaneous coronary intervention, Prospective Studies, Risk Factors
Background
With the increasing number of patients receiving intravascular iodinated contrast agents worldwide, contrast-induced nephropathy (CIN) has become the third leading cause of acute kidney injury in hospitalized patients [1]. The injury is especially common in patients whose renal function has been already compromised [2]. Several scoring systems have been developed to identify high-risk patients with CIN. Among them, the Mehran risk score is based on 8 risk factors (hypotension, use of intra-aortic balloon pump, congestive heart failure, age, anemia, diabetes, volume of contrast medium, and estimated glomerular filtration rate) is commonly used for scoring patients undergoing percutaneous coronary intervention (PCI) [3]. Four stratifications of CIN risk have been established from the cut-off points and the intervals defined as follows: low, <5 points; moderate, 6–10 points; high, 11–15 points; and very high, >15 points [3].
According to the current guideline recommendation, saline hydration is the most effective method to prevent CIN [4, 5]. However, the necessity of saline hydration in patients with low-risk profiles of CIN has not been reported. A retrospective analysis showed that the occurrence of CIN in low-risk patients was as high as 10%, suggesting that these patients may need preventive hydration [6]. Although the diagnosis of CIN depends on the absolute level or dynamic changes of serum creatinine (Cr) after the contrast use [7], Cr is supposed to be an insensitive biomarker to detect minor renal injury. Therefore, we suppose some mild renal injuries might be ignored when Cr was used as the only biomarker to define the diagnosis of CIN. Numerous studies have suggested that cystatin C (Cys C) and neutrophil gelatinase-associated lipocalin (NGAL) are both sensitive and early biomarkers for mild renal injury [8–10]. Because the number of patients receiving contrast arteriography is greatly increasing, we investigated the safety of the contrast procedure in outpatient or day admission departments. Moreover, there has been no randomized study evaluating the necessity of intravenous saline hydration in patients with very low-risk profiles of CIN. Therefore, the present study explored whether hydration is essential in patients undergoing coronary angiography who have very low-risk profiles for CIN using the new biomarkers of Cys C and NGAL.
Material and Methods
STUDY POPULATION AND DESIGN:
This study was a randomized, prospective, open-label clinical trial aimed to compare different hydration strategies in patients with very low-risk profiles of CIN. From November 2017 to March 2019, a total of 150 patients who agreed to receive coronary angiography (CAG) were enrolled into the present study. We defined very low-risk profiles of CIN as Mehran risk score ≤2. In brief, patients should meet the following criteria: age <75 years, no hypotension, no use of intra-aortic balloon pump (IABP), no congestive heart failure, no diabetes mellitus, no anemia, total contrast media volume <200 mL, and eGFR >60 mL/min/1.73 m2. We calculated eGFR using the Modification of Diet in Renal Disease (MDRD) study equation [11]. Patients were excluded if they had one of the following criteria: 1) acute myocardial infarction; 2) use of a drug leading to nephrotoxicity; 3) acute or chronic infection; 4) uncontrolled hypertension; 5) obvious dysfunction of liver and/or kidney; 6) malignant tumor. The study was approved by the Medical Ethics Committees of the Second Hospital of Tianjin Medical University and the written informed consents were obtained from all the participants.
HYDRATION STRATEGIES AND RANDOMIZATION:
After the baseline measurements and assessments, eligible patients were randomly assigned (1: 1: 1) to one of the following comparing groups: 1) preventive hydration group (Preventive Group, n=50, saline hydration was given 6 h before the procedure until 12 h after the procedure); 2) remedial hydration group (Remedial Group, n=50, saline hydration was given after the procedure lasting for 12 h); 3) no hydration group (NH group, n=50, saline hydration was only given during the procedure and the total volume of normal saline was no more than 500 mL).
The randomization schedule involved sealed, sequentially numbered envelopes that contained the treatment allocated using computer-generated random numbering. Interventionists who performed the procedure and the laboratory staff were blinded to the treatment allocation to minimize selection biases.
PROCEDURES:
All the patients received optimal medical therapies according to the current guideline recommendations. Angiography was performed via trans-radial or trans-femoral artery approach. According to the current guideline recommendation, the infusion speed of intravenous normal saline was 1 mL/Kg/h. Baseline demographic and clinical data were collected. Serum levels of BUN, Cr, and Cys C were tested at 1 h before and at 24 h and 72 h after the contrast procedure in order to evaluate changes in renal function. Urine samples were collected at 1 h before and at 6 h and 48 h after the procedure. The urine samples were centrifuged and stored at −80°C to analyze the urinary NGAL by enzyme-linked immunosorbent assay (ELISA, R&D Systems). The V/CrCl ratio was calculated by dividing the volume of contrast by the CrCl, which was calculated by applying the Cockcroft-Gault formula according to the serum Cr concentration [12].
STATISTICAL ANALYSIS:
All data were analyzed using SPSS 18.0 software (SPSS-PC Inc. Chicago, IL, U.S.A). Data are reported as mean±standard deviation (SD) unless otherwise specified. Categorical variables are expressed as proportions and the chi-square (χ2) test was applied for statistical comparisons. One-way analysis of variance (ANOVA) was used to assess the difference of continuous variables among the 3 groups. Because urinary NGAL concentration remarkably deviated from normal distributions in the present study, the non-parametric Kruskal-Wallis H test was applied to evaluate the differences among groups.
Results
As shown in Figure 1, 6 patients were excluded from the present study because the total contrast volume exceeded the upper limit (200 mL). Thus, a total of 144 patient with low-risk profiles were eligible for intention-to-treat analysis.
Table 1 shows that the baseline characteristics were well balanced among the 3 groups. No CIN occurred in the present study, suggesting all enrolled patients had very low-risk profiles. The patients had a mean age of 62.85±7.25 years old, and 52.08% were male. A total of 93 (64.58%) patients had hypertension and 8 (5.56%) patients were previously diagnosed with stroke. No significant difference was found in the baseline of estimated glomerular filtration rate (eGFR) before the procedure among the 3 groups, and the total volumes of contrast media were also similar among these study arms. We also evaluated the ratio of contrast volume-to-creatinine clearance (V/CrCl), and no significant difference was observed.
The results of biomarkers are shown in Table 2. The levels of baseline Cr, eGFR, and Cys C were similar among the 3 groups. Twenty-four hours after contrast administration, the levels of Cr, Cys C, and eGFR did not differ significantly among the 3 groups. Similarly, the above biomarkers also showed no significant difference at 72 h after the procedure. The serial results of urinary NGAL before and after procedure were similar among the 3 groups.
To evaluate the impacts of PCI which usually had higher volume of contrast media on the renal biomarkers, we conducted the subgroup analysis stratified by receiving PCI or receiving coronary angiography (CAG) only (Table 3). The total contrast volumes in the PCI subgroup were significantly higher than were those in the CAG subgroup (123.66±27.96 mL vs 56.62±14.61 mL,
Discussion
This perspective randomized study revealed that preventive hydration seems to be unnecessary in very low-risk patients receiving contrast media.
CIN is defined as the increase of serum Cr by at least 44 ummol/L (0.5 mg/dL) or the increase of Cr level by 25% within 48–72 h after administration of contrast agent [13]. The pathogenesis of CIN is thought to be multifactorial. After contrast media administration, CIN was associated with altered renal hemodynamics and increased oxygen consumption, which could lead to hypoxia and increased levels of reactive oxygen species, eventually resulting in acute kidney injury [14,15]. Many clinical and experimental trials focusing on CIN prevention have been reported. Most studies have shown that preventive hydration can effectively reduce the incidence of CIN disease, especially in patients with impaired renal function [16,17]. However, the necessity of hydration in patients with very low-risk profiles of CIN remains unclear.
Previous studies have evaluated the effect of saline hydration on CIN patients with low risk. Calabro et al [6] assessed the effectiveness of normal saline hydration plus N-acetyl cysteine (NAC) in preventing CIN in low-risk patients in a retrospective study. A total of 152 consecutive patients undergoing CAG were enrolled. The study had a historical control group with 172 low-risk patients. Cr levels were measured as the principal biomarker to evaluate acute kidney injury. Eventually, the overall incidence of CIN was 2.6% (4 patients) in the treatment group and 11.2% (19 patients) in the historical control group (
However, the above study was not a randomized or prospective study [6]. The present study is the first randomized clinical trial using highly sensitive biomarkers to evaluate the effectiveness of different hydration strategies in very low-risk patients. Our study showed that there was no CIN in these very low-risk cases. The levels of Cr and eGFR also did not differ significantly among the 3 groups. Moreover, even for highly sensitive renal biomarkers such as Cys C and NGAL, no significant difference was found among the 3 groups. Therefore, based on these results, it appears that there is no need for hydration in patients with very low-risk profiles. The cause of the discrepancy between the present study and the aforementioned study by Calabro et al [6] might be as follows. First, we enrolled patients with lower Maheran risk scores (≤2) as compared with Calabro’s study [6] (2.03 in treatment group vs 2.28 in control group, respectively). Second, even in the NH group, patients received normal saline infusion during the procedure for their safety. Third, although only 6 patients received contrast media >200 mL, we excluded them from the present study. Patients in the Calabro et al study received a higher volume of contrast. We think these reasons may explain the discrepancy and no cases of CIN occurred in the present study.
CIN is largely associated with contrast volume. Therefore, we performed subgroup analysis stratified by PCI or CAG only. Although the contrast volumes in the PCI subgroup were significantly higher than in the CAG subgroup, there was still no significant difference in renal biomarkers among the 3 treatment strategies in patients who underwent PCI. The evidence suggests that in very low-risk patients, providing no hydration is as safe as providing preventive or remedial hydration strategies, even when patients received relatively higher volumes of contrast media.
Coronary intervention has been extensively used in the diagnosis and treatment of ischemic heart disease. Special attention has been paid to CIN due to its worse clinical outcomes compared to those without [18–20]. Although CIN is not common in patients with normal renal function, it occurs in 5% of inpatients and 2% of outpatients [21,22]. Currently, the definition of CIN is based on the absolute level or the relatively changed percentage of Cr after contrast administration. However, neither Cr nor eGFR is a sensitive or early biomarker to detect acute kidney injury. Cys C was reported to be a good biomarker for acute renal failure (ARF) and it can detect ARF 1 or 2 days earlier than Cr [23]. Cys C increased 8 h after percutaneous coronary intervention in CIN patients, and reached its peak 24 h after use of contrast medium [24]. Urinary NGAL levels increase at 4 h after PCI, which is much earlier than for serum Cr, and even Cys C. Since NGAL has small molecular size and resistance to degradation, it can predict CIN with good sensitivity (76%) and specificity (80%) [25]. According to the results of the present study, there is no need to use preventive or remedial hydration strategies for patients during the periprocedural period when their Mehran risk score is ≤2. In brief, the present study suggests that it is safe, economical, and time-saving for physicians to perform contrast procedures in outpatient or day admission departments for this particular subset of patients with Mehran risk score ≤2, regardless of CAG or PCI.
The present study has several limitations. First, it is a single-center study with relatively small sample size, which may weaken the power of results. Thus, further multicenter clinical trials with larger study populations are needed to provide stronger conclusions. In the present study, we used highly sensitive biomarkers such as Cys C and NGAL to detect early kidney injury. We did not find significant differences among the 3 different strategies, which indicated that the effects of contrast agents on renal function were very slight in patients with very low risk of CIN. Second, we did not conduct routine follow-up to monitor the mid- to long-term changes in renal biomarkers. However, CIN usually occurs within 48 to 72 h after administration of a contrast agent. We tested serum Cr and Cys C at 24 and 72 h after the procedure, suggesting that the lack of follow-up data did not have a significant effect on the conclusions. Third, the Mehran score is generated on a list of variables, among which contrast volume can only be measured after the procedure. Therefore, we could not precisely predict the Mehran score before randomization. Finally, the present study was an open-label trial. However, the interventionists and laboratory staff were blinded to the allocated treatment strategies. Therefore, the choice of treatment strategies and the results of laboratory tests would be reliable.
Conclusions
Briefly, it is unnecessary to use a preventive or remedial hydration strategy for patients with Mehran risk score ≤2 during the periprocedural period. Additionally, it is suggested that conducting a contrast procedure in outpatient or day admission departments would be safe, economical, and time-saving for patients with Mehran risk score ≤2, regardless of CAG or PCI.
Figures
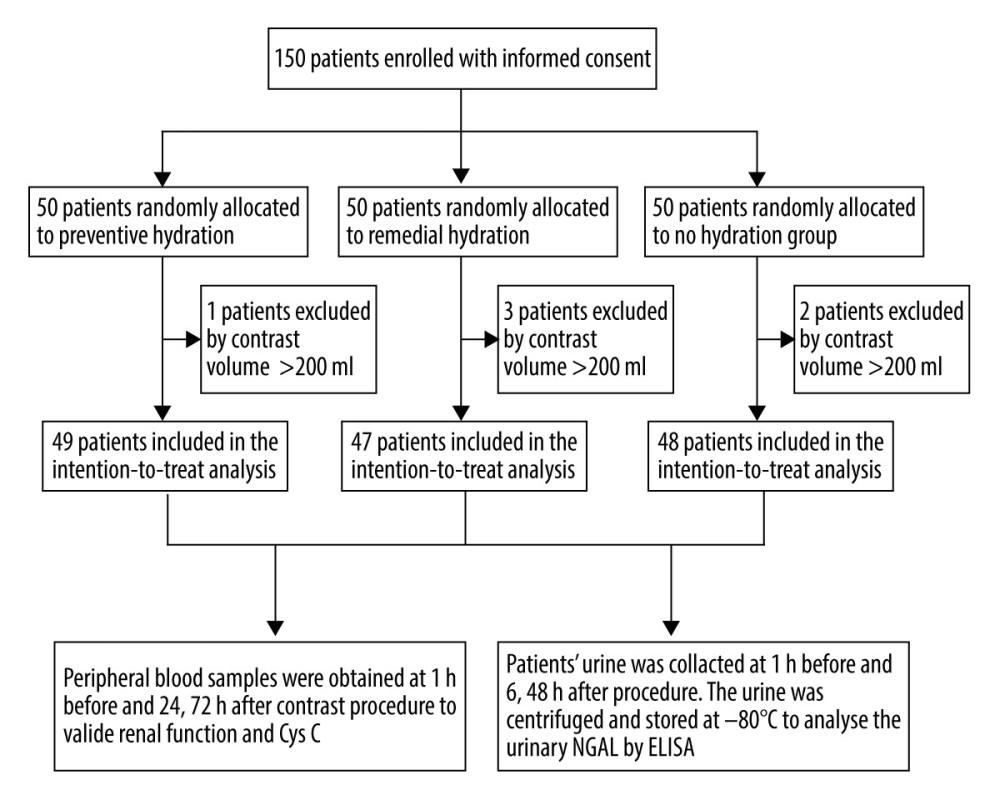 Figure 1. Study flow chart.
Figure 1. Study flow chart. References
1. McCullough PA, Sandberg KR, Epidemiology of contrast-induced nephropathy: Rev Cardiovasc Med, 2003; 4(Suppl 5); S3-9
2. McCullough PA, Contrast-induced acute kidney injury: J Am Coll Cardiol, 2008; 51; 1419-28
3. Mehran R, Aymong ED, Nikolsky E, A simple risk score for prediction of contrast-induced nephropathy after percutaneous coronary intervention: Development and initial validation: J Am Coll Cardiol, 2004; 44; 1393-99
4. Stacul F, van der Molen AJ, Reimer PContrast Media Safety Committee of European Society of Urogenital Radiology (ESUR), Contrast induced nephropathy: Updated ESUR Contrast Media Safety Committee guidelines: Eur Radiol, 2011; 21; 2527-41
5. Kodzwa R, Updates to the ACR manual on contrast media: Radiol Technol, 2017; 89; 186-89
6. Calabro P, Bianchi R, Crisci M, Use and efficacy of saline hydration and N-acetyl cysteine to prevent contrast-induced nephropathy in low-risk populations undergoing coronary artery angiography: Intern Emerg Med, 2011; 6; 503-7
7. Mehran R, Nikolsky E, Contrast-induced nephropathy: Definition, epidemiology, and patients at risk: Kidney Int Suppl, 2006(100); S11-15
8. Padhy M, Kaushik S, Girish MP, Serum neutrophil gelatinase associated lipocalin (NGAL) and cystatin C as early predictors of contrast-induced acute kidney injury in patients undergoing percutaneous coronary intervention: Clin Chim Acta, 2014; 435; 48-52
9. Kjeldsen L, Johnsen AH, Sengelov H, Isolation and primary structure of NGAL, a novel protein associated with human neutrophil gelatinase: J Biol Chem, 1993; 268; 10425-32
10. Paragas N, Qiu A, Zhang Q, The Ngal reporter mouse detects the response of the kidney to injury in real time: Nat Med, 2011; 17; 216-22
11. Chudek J, Kolonko A, Owczarek AJ, Clinical factors increasing discrepancies of renal function assessment with MDRD and Cockcroft-Gault equations in old individuals: Eur Geriatr Med, 2018; 9(5); 713-20
12. Cockcroft DW, Gault MH, Prediction of creatinine clearance from serum creatinine: Nephron, 1976; 16; 31-41
13. Shaker OG, El-Shehaby A, El-Khatib M, Early diagnostic markers for contrast nephropathy in patients undergoing coronary angiography: Angiology, 2010; 61; 731-36
14. Heyman SN, Rosen S, Rosenberger C, Renal parenchymal hypoxia, hypoxia adaptation, and the pathogenesis of radiocontrast nephropathy: Clin J Am Soc Nephrol, 2008; 3; 288-96
15. Tumlin J, Stacul F, Adam A, Pathophysiology of contrast-induced nephropathy: Am J Cardiol, 2006; 98; 14K-20K
16. Jurado-Roman A, Hernandez-Hernandez F, Garcia-Tejada J, Role of hydration in contrast-induced nephropathy in patients who underwent primary percutaneous coronary intervention: Am J Cardiol, 2015; 115; 1174-78
17. Mueller C, Prevention of contrast-induced nephropathy with volume supplementation: Kidney Int Suppl, 2006(100); S16-19
18. Rihal CS, Textor SC, Grill DE, Incidence and prognostic importance of acute renal failure after percutaneous coronary intervention: Circulation, 2002; 105; 2259-64
19. James MT, Ghali WA, Tonelli M, Acute kidney injury following coronary angiography is associated with a long-term decline in kidney function: Kidney Int, 2010; 78; 803-9
20. Mehran R, Dangas GD, Weisbord SD, Contrast-associated acute kidney injury: N Engl J Med, 2019; 380; 2146-55
21. Curtis LM, Agarwal A, HOpe for contrast-induced acute kidney injury: Kidney Int, 2007; 72; 907-9
22. Weisbord SD, Palevsky PM, Prevention of contrast-induced nephropathy with volume expansion: Clin J Am Soc Nephrol, 2008; 3; 273-80
23. Herget-Rosenthal S, Marggraf G, Husing J, Early detection of acute renal failure by serum cystatin C: Kidney Int, 2004; 66; 1115-22
24. Rickli H, Benou K, Ammann P, Time course of serial cystatin C levels in comparison with serum creatinine after application of radiocontrast media: Clin Nephrol, 2004; 61; 98-102
25. Bachorzewska-Gajewska H, Malyszko J, Sitniewska E, NGAL (neutrophil gelatinase-associated lipocalin) and cystatin C: Are they good predictors of contrast nephropathy after percutaneous coronary interventions in patients with stable angina and normal serum creatinine?: Int J Cardiol, 2008; 127; 290-91
Figures
Tables
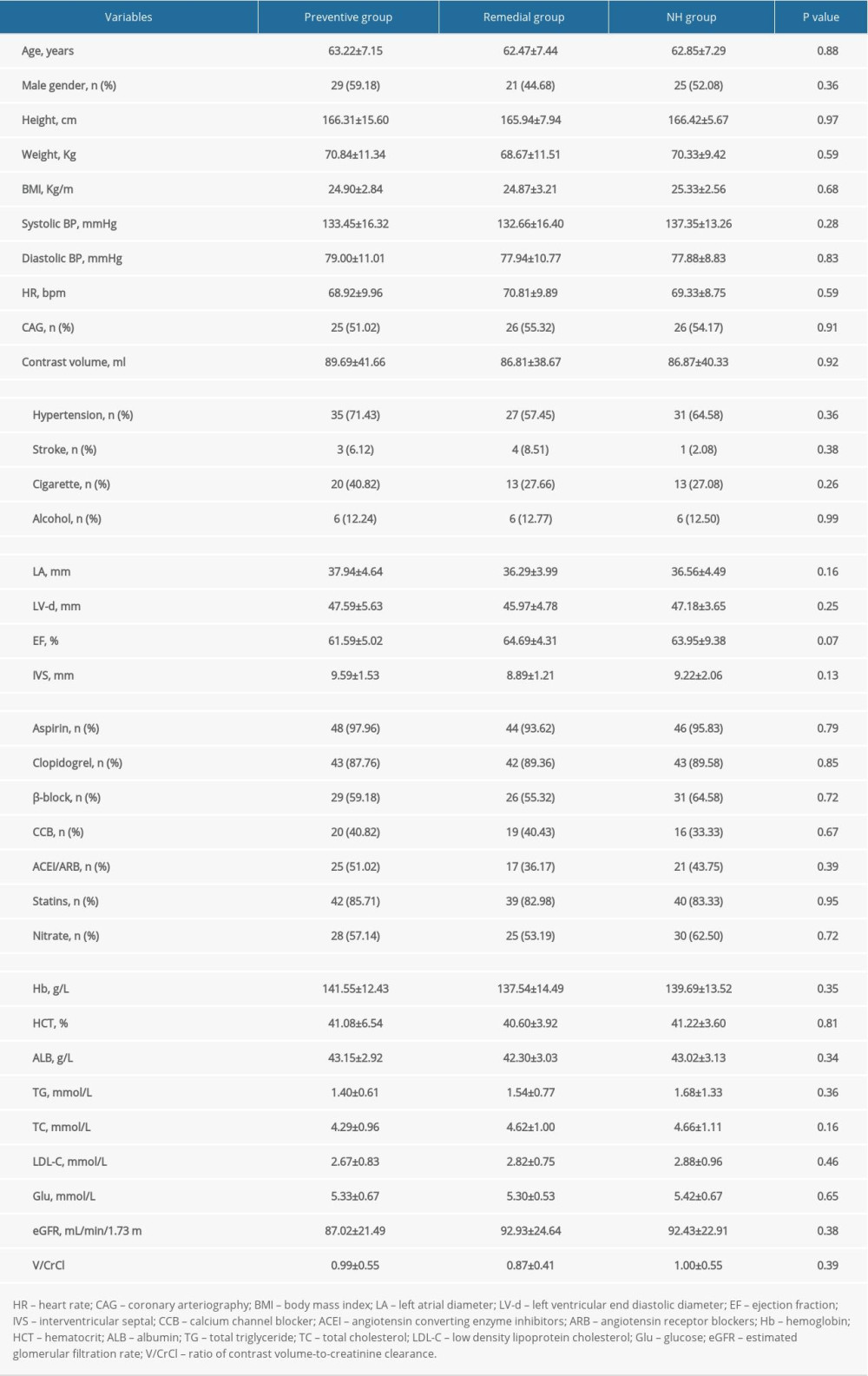 Table 1. Baseline characteristics of the 3 groups.
Table 1. Baseline characteristics of the 3 groups.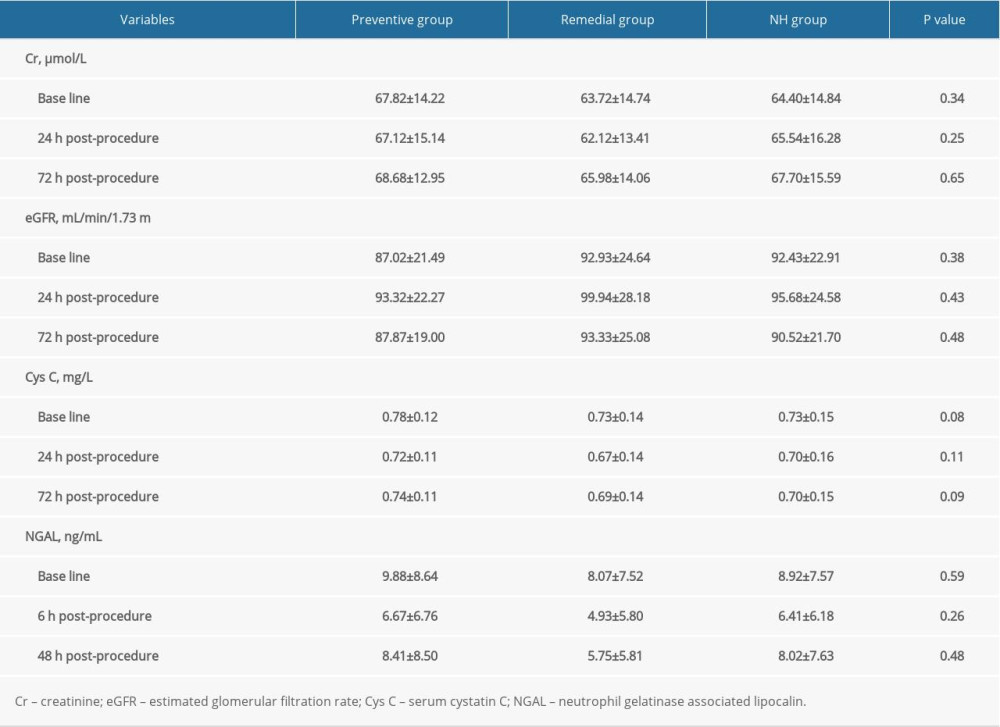 Table 2. Comparisons of renal biomarkers among the 3 groups.
Table 2. Comparisons of renal biomarkers among the 3 groups.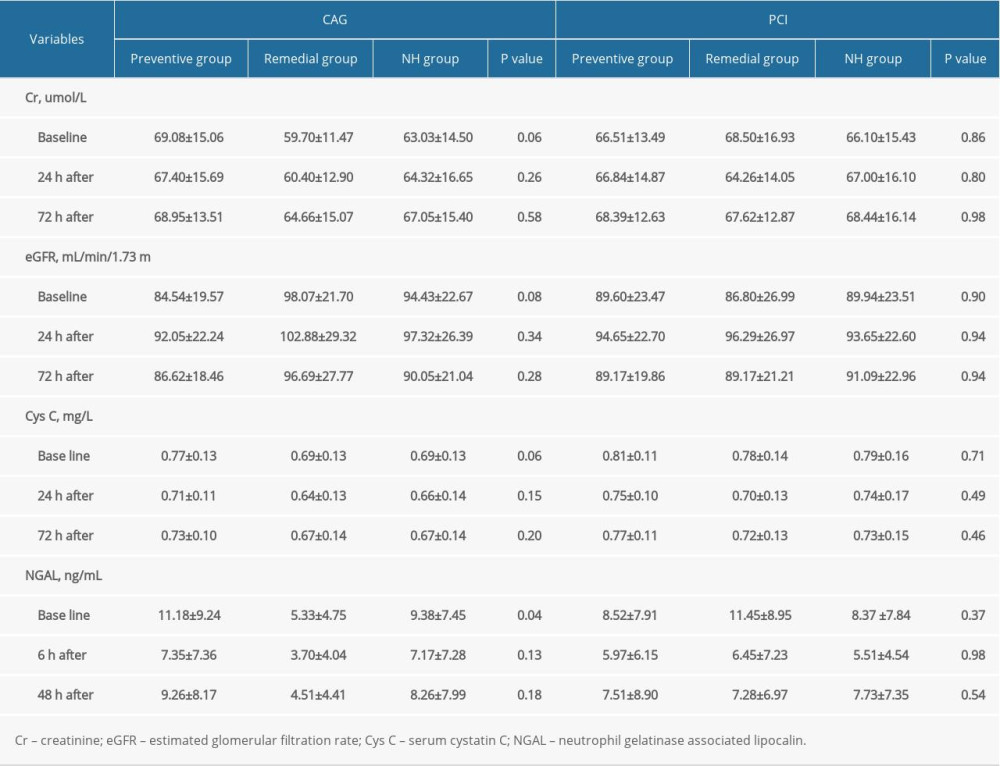 Table 3. Subgroup analyses in patients receiving CAG versus PCI.
Table 3. Subgroup analyses in patients receiving CAG versus PCI. Table 1. Baseline characteristics of the 3 groups.
Table 1. Baseline characteristics of the 3 groups. Table 2. Comparisons of renal biomarkers among the 3 groups.
Table 2. Comparisons of renal biomarkers among the 3 groups. Table 3. Subgroup analyses in patients receiving CAG versus PCI.
Table 3. Subgroup analyses in patients receiving CAG versus PCI. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








