02 April 2021: Database Analysis
In Silico Network Analysis of Ingredients of in Osteoporosis
Feiqi Huang12AE, Huizhi Guo1BE, Yuanbiao Wei2BD, Xiao Zhao2DF, Yangsheng Chen2DF, Zhan Lin2BF, Yanhui Zhou2F, Ping Sun3AEG*DOI: 10.12659/MSM.929219
Med Sci Monit 2021; 27:e929219
Abstract
BACKGROUND: Cornus officinalis (CO), also known as ‘Shanzhuyu’, is one of the most common traditional Chinese herbs used against osteoporosis. Although previous studies have found that CO has beneficial effects in alleviating osteoporosis, its mechanisms remain unclear.
MATERIAL AND METHODS: In this study, we applied system bioinformatic approaches to investigate the possible therapeutic mechanisms of CO against osteoporosis. We collected the active ingredients of CO and their targets from the TCMSP, BATMAN-TCM, and ETCM databases. Next, we obtained the osteoporosis targets from differentially expressed mRNAs from the Gene Expression Omnibus (GEO) gene series (GSE35958). Next, the shared genes of the CO pharmacological targets and osteoporosis-related targets were selected to construct the protein-protein interaction network, based on the results from the STRING database. Subsequently, Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment analyses were carried out by using the clusterProfiler package in R software.
RESULTS: In all, there were 58 unique CO compounds and 518 therapeutic targets. Based on the GO and KEGG enrichment results of 98 common genes, we selected the top 25 terms, based on the terms’ P values. We found that the anti-osteoporotic effect of CO may mostly involve the regulation of calcium metabolism and reactive oxygen species, and the estrogen signaling pathway and osteoclast differentiation pathway.
CONCLUSIONS: We found the possible mechanisms of CO in treating osteoporosis may be based on multiple targets and pathways. We also provided a theoretical basis and promising direction for investigating the exact anti-osteoporotic mechanisms of CO.
Keywords: Computational Biology, Osteoporosis, Pharmacologic Actions, Computer Simulation, Cornus, Drugs, Chinese Herbal, Estrogens, gene ontology, Molecular Docking Simulation, Osteoclasts, Protein Interaction Maps
Background
Osteoporosis is a systemic bone disease mainly observed in postmenopausal women and the elderly. It is characterized by reduced bone mass and strength and an increased incidence of fractures [1]. Postmenopausal osteoporosis and senile osteoporosis are the 2 main subtypes of primary osteoporosis. In 2016 in China, osteoporosis affected up to 36% of people over the age of 60 years [2]. Currently, it is reported that about 13% of the Chinese population has osteoporosis [3]. Bone fracture is a common complication of osteoporosis, and it was estimated that more than 5.99 million elderly people could have osteoporotic fractures by 2050 in China, with an expected economic cost estimated at 175.4 billion yuan [4,5]. Therefore, it is necessary to take measures to maintain bone mass and lower the incidences of osteoporotic fractures in the population. Clinically, the treatment of the different types of osteoporosis mainly consists of antiresorptive agents and anabolic agents, including calcium, vitamin D, bisphosphonates, and calcitonin [6,7]. However, it is worth noting that these osteoporosis treatments have harmful adverse effects, such as bisphosphonates increasing the risk of breast cancer, jaw necrosis, or even atypical femur fractures [8,9], and excessive calcium intake possibly contributing to the increased risk of cardiovascular events [10]. Therefore, it is important to find safer treatments of osteoporosis. As an alternative to osteoporosis treatment with bisphosphonates or denosumab, an antibody to receptor activator of nuclear factor kappa-B ligand (RANKL), researchers have proposed the use of natural products, such as PMA-zeolite clinoptilolite and calcium-containing zeolite granules, antioxidant compounds such as quercetin and phytoestrogens, and antiresorptive compounds originating from olive oil [11].
In recent years, the development of novel drugs from traditional Chinese medicine (TCM) has attracted increasing attention. TCM possesses the features of having a good curative effect and few adverse effects and thus has been widely used in China for thousands of years. With its satisfactory therapeutic effect and relatively low toxicity, TCM has served as an important alternative treatment in East Asia, Europe, and North America [12,13]. Moreover, kidney tonifying drugs have been widely applied as osteoporosis treatment and have proved effective in treating osteoporosis in experiments [14–16].
Although CO has shown potential effects for treating osteoporosis, few studies have comprehensively or systematically investigated its therapeutic mechanism. This may be because CO exerts its therapeutic efficacy through multimolecular balancing in the human body, which is difficult to prove in the scientific experiments widely used in Western medicine [24]. Fortunately, systems pharmacology, an integrity and synergy analysis method, provides researchers a valid tool to explore the therapeutic mechanisms of TCM at a holistic level [25].
In the current study, we utilized an integrated systems pharmacology approach to comprehensively dissect the pharmacological mechanisms of CO in treating osteoporosis. First, we collected the potential active ingredients of CO and their targets through multiple databases. Second, the related osteoporosis targets were obtained from the Gene Expression Omnibus (GEO) data set and then matched with the above CO targets. Third, the protein-protein interaction (PPI) network was constructed on the basis of the common targets from the second step. Finally, Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment analyses were carried out using the clusterProfiler package in R software. Our study provides new insight into the analysis of the therapeutic effects of CO on osteoporosis, based on systems pharmacology and microarray data analysis.
Material and Methods
SCREENING OF POTENTIAL PHARMACOLOGICAL COMPOUNDS AND TARGETS OF CO:
We utilized 3 frequently used databases to acquire the therapeutic ingredients of CO, including the Traditional Chinese Medicine System Pharmacology Database (TCMSP, http://lsp.nwu.edu.cn/tcmsp.php) [26]; BATMAN-TCM platform (http://bionet.ncpsb.org/batman-tcm/) [27], and the Encyclopedia of Traditional Chinese Medicine platform (http://www.tcmip.cn/ETCM/index.php/Home/Index/index.html) [28]. For further compound screening, we set the oral bioavailability threshold at 30% and drug-likeness greater than or equal to 0.18. Oral bioavailability, one of the valid tools in drug screening, signifies the percent of oral administered drugs entered into the body’s systemic circulation [29]. Drug-likeness is used to screen out chemically unsuitable compounds and identifies the degree to which a compound is “druglike” [30]. For the BATMAN-TCM database, the thresholds for TAR-score and P value cutoffs were set at ≥55 and <0.05, respectively, to collect potential ingredients. Moreover, the default cutoff value ≥0.8 was used in the ETCM screening part. These ingredients exert their therapeutic functions by interacting with targets; therefore, we further collected the targets predicted by the TCMSP, BATMAN-TCM, and ETCM databases.
THE ACQUISITION OF DIFFERENTIALLY EXPRESSED GENES IN OSTEOPOROSIS:
We used the GEO database (
NETWORK CONSTRUCTION:
The powerful and open-source software Cytoscape (3.7.2, http://www.cytoscape.org/) [31] was used to construct 2 critical networks visually, including a compound-target network and PPI network. Next, degree centrality [32], a critical topological parameter, was used to sort nodes in the networks; the greater the number of connections between nodes there were in a network, the more importance a node had.
For the construction of the PPI network, we first drew a Venn diagram using R software (R 3.6.2 for Windows) to obtain the common genes between osteoporosis and potential ingredient targets, which were considered as critical targets when CO exerted its anti-osteoporotic function. Next, the interactions between these intersecting genes were analyzed in the STRING platform [33], and the threshold of the interaction score was set at the highest confidence (0.900) to obtain the most credible interaction. Finally, the exported data of the PPI were used to construct the PPI network in Cytoscape.
GO AND KEGG PATHWAY ENRICHMENT ANALYSIS:
After obtaining the common genes, the potential therapeutic targets obtained from GO enrichment (biological process and molecular function) and KEEG pathway enrichment were analyzed through the clusterProfiler package [34] in R. All of these results contributed to exploring the potential biological pathways and functions involved in the anti-osteoporotic function of CO.
Results
IDENTIFICATION OF CO TARGETS:
By screening the 3 databases, we collected 14 compounds from TCMSP, 18 from BATMAN-TCM, and 28 from ETCM, yielding 58 unique compounds. Moreover, TCMSP identified 130 bioactive targets, BATMAN-TCM identified 299 targets, and ETCM identified 582 targets. Finally, a total of 518 targets were obtained after taking the union set and deleting duplicate items (Figure 1). In this network, the top 5 compounds were cetylic acid, ursolic acid, linoleic acid, isoamyl alcohol, and alpha-corocalene.
POTENTIAL TARGETS OF OSTEOPOROSIS:
A total of 4661 differentially expressed genes were detected in the bone samples of patients with osteoporosis, including 2205 upregulated genes and 2456 downregulated genes. We then drew a volcano map to display the gene distribution of the GSE35958 data set (Figure 2). After obtaining the differentially expressed matrix of this data set, we constructed a clustering graph to map the first 50 differential genes sorted by their absolute value of fold change. As shown in Figure 3, the first 4 columns are the control samples and the last 5 are the osteoporotic samples. The changes in color depth are according to the genes’ fold changes presented inside the cells.
INTERSECTION TARGETS AND THE PPI NETWORK:
The 518 predicted targets of CO were intersected with 4661 differentially expressed genes in the osteoporotic bone samples, and 98 therapeutic targets were identified. These key targets may be the therapeutic genes for CO exerting its anti-osteoporotic effect (Figure 4). Subsequently, we determined the expression profile of these 98 genes based on the background of the original chip shown in Figure 5. Among these therapeutic targets, there were 59 overexpressed genes and 39 underexpressed genes. The top 50 common targets are shown in Table 1.
We then obtained the interaction between these common genes based on the result from the STRING database, in which their minimum required interaction score was set to the highest confidence level (0.900). Next, this PPI network, consisting of 52 nodes and 62 edges, was visualized by Cytoscape (Figure 6). Based on the nodes’ area, color depth, and location, the genes PIK3R1, PRKCD, ESR1, JUN, IDH1, and ADRA2A may play key roles in this network.
GO ENRICHMENT ANALYSIS:
We conducted the GO enrichment of these 98 targets using the clusterProfiler package. A total of 594 biological process terms and 91 molecular function terms were enriched. We sorted these terms in ascending order of P value and gene count; the top 25 biological process and molecular function terms are shown in Figures 7 and 8. These enrichment results indicated that CO may exert its anti-osteoporotic effect by regulating the transport of the calcium ion transmembrane, release of sequestered calcium ion into cytosol, negative regulation of sequestering of calcium ion, regulation of sequestering of calcium ion, sequestering of calcium ion, and response to reactive oxygen species (ROS).
KEGG PATHWAY ENRICHMENT ANALYSIS:
We then conducted the KEGG pathway enrichment of 98 overlapping genes to determine the possible therapeutic mechanism of CO in osteoporosis. With a cutoff P value of 0.05, a total of 92 pathways were enriched, and then the top 15 pathways were selected based on their P values (Figure 9). Among these pathways, the estrogen signaling pathway (hsa04915) ranked first (P<0.0001) and osteoclast differentiation (hsa04380) ranked twenty-first (P<0.001); these may be the critical signaling pathways for CO in treating osteoporosis.
Discussion
Osteoporosis is a progressive bone disease characterized by decreased bone loss and abnormal bone tissue structure [35]. With the aging of society worldwide, osteoporosis is a common disease threatening this population; osteoporosis also imposes a significant economic burden on healthcare systems [36]. Therefore, it is economical and desirable to alleviate osteoporosis and its related fractures [37]. The loss of bone mass resulting from osteoporosis is caused by the lack of balance between bone absorption, which is determined by osteoclasts, and bone formation, which is determined by osteoblasts [38]. Bone resorption inhibitors are currently the primary treatment for osteoporosis. However, the available bone resorption drugs have serious adverse effects associated with long-term use, such as cardiovascular events, atypical long bone fractures, and jaw osteonecrosis [39,40]. Therefore, there is a practical need to find alternative and safer treatments capable of protecting against osteoporosis [41].
CO helps prevent osteoporosis by prohibiting osteoclast activity and promoting osteogenic differentiation, which may be associated with its regulating effects on related targets. Therefore, we applied the network pharmacology method to explore the potential mechanism of CO at a systems level. Through the compounds-targets network (Figure 4), we found that the top 5 ingredients of CO, cetylic acid, ursolic acid, linoleic acid, isoamyl alcohol, and alpha-corocalene, may play critical roles in this network. Therefore, the promising anti-osteoporotic effects of CO may be closely related to these compounds. Lee et al found, through in vitro and in vivo experiments, that ursolic acid enhances osteoblast differentiation and promotes bone formation [42]. Also, ursolic acid suppresses osteoclast differentiation and inhibits osteoclast-induced bone loss, which may be related to the NF-κB signaling pathway [43]. Currently, there is no literature available about the effects of linoleic acid on osteoclasts or osteoblasts. However, a study showed that linoleic acid, as the agonist for peroxisome proliferator activator receptors, can significantly improve osteoblast differentiation and increase serum bone formation markers and BMD in male rats [44]. Moreover, conjugated linoleic acid, the isomer of linoleic acid, possesses the promising regulatory ability of osteoclast and osteoblast differentiation [45]. Although some compounds, such as morroniside and loganin, have been extensively studied in the field of osteoporosis [23], they were not involved in the present study owing to their low oral bioavailability or drug-likeness values, which are critical pharmacokinetic tools for drug screening. Therefore, we will prioritize compounds such as ursolic acid in further studies on the regulatory effects of CO on osteoclastic and osteoblastic differentiation.
A Venn graph between CO targets and differentially expressed genes of osteoporosis identified 98 common genes as therapeutic targets, which may play critical roles in CO exerting its anti-osteoporotic effect. Indeed, some clinical trials and animal experiments found that these genes have anti-osteoporotic effects. Transforming growth factor-β1 (TGF-β1) is a highly expressed bone matrix protein that regulates interactions between osteoclasts and osteoblasts to balance bone remodeling, thereby maintaining sufficient bone mass. It has been shown that the polymorphism of the TGF-β1 gene has an adverse effect on BMD and increases the susceptibility of osteoporosis [46]. Similarly, Zhou et al used peripheral blood monocytes for a cell model and confirmed that NFKB2, via exon arrays, was differentially expressed in high and low BMD groups [47]. Moreover, 2-sample mendelian randomization confirmed that the ESR1 gene influenced BMD and was therefore associated with fracture risk [48]. Wang et al also found that ESR1 is a key risk factor for osteoporotic fracture in Chinese postmenopausal women [49]. PIK3R1, a key regulatory factor of the PI3K/Akt signaling pathway, plays a critical role in alleviating osteoporosis [50,51]. Kim et al showed that cells laced with Bax attenuate osteoclast apoptosis [52].
According to the drug theory of “multi-ingredient, multitarget” [53], the above results showed that the multiple active ingredients of CO may interact with different genes to exert its anti-osteoporotic effect. Therefore, we further conducted GO and KEGG pathway enrichment to analyze the functions of these genes. GO is a collection of controlled vocabularies for gene functions, and many genomes have been annotated with GO terms, including biological process, cellular component, and molecular function. Therefore, GO enrichment analysis finds GO terms that are abnormally expressed for a given set of interesting genes, enabling their biological interpretation. Moreover, KEGG pathway enrichment demonstrates additional information about the patterns of genes interacting in pathways [54]. After conducting these 2 enrichments based on 98 common genes, we suggested that the therapeutic effect of CO in osteoporosis treatment may involve the regulation of calcium metabolism and ROS biological processes and the estrogen signaling and osteoclast differentiation pathways.
The differentiation of osteoclasts and osteoblasts and their proliferation are all closely related to ROS [54,55]. ROS can cause serious damage to periodontal tissue through accelerating bone resorption, which is closely related to the upregulated osteoclast differentiation by NF-κB and the calcineurin-NFAT pathway [56,57]. We found several GO biological process terms are associated with reactive ROS, including GO: 0000302 (response to ROS), GO: 0034614 (cellular response ROX), GO: 0006979 (response to oxidative stress), and GO: 0034599 (cellular response to oxidative stress). Although there are no studies on the anti-ROS effect of CO compounds on osteoblasts or osteoclasts, several studies confirmed that CO compounds, such as ursolic acid and linoleic acid, can reduce ROS production in cancer cells [55,56]. Hwang et al showed that the ethanol extract of CO can protect RAW 264.7 cells from lipopolysaccharide-induced oxidative stress [57]. Therefore, it is worth validating the anti-ROS effects of CO compounds on bone cells in the future.
In addition, the anti-osteoporotic effect of CO may be associated with the regulation of calcium ions; the top 5 related GO biological process terms were GO: 0051209 (release of sequestered calcium ion into cytosol), GO: 0051282 (regulation of sequestering of calcium ion), GO: 0051208 (sequestering of calcium ion), GO: 0097553 (calcium ion transmembrane import into cytosol), and GO: 0006816 (calcium ion transport). Calcium supplementation is a critical part of the prevention and treatment of osteoporosis. Increasing evidence shows that aging is associated with a disturbed calcium balance. Heshmati et al found that, in addition to estrogen deficiency, women with osteoporosis who are postmenopausal have a parathyroid hormone defect, which leads to a negative calcium balance [58]. Ursolic acid has been found to promote calcium balance and have an inhibitory effect on the age-induced secondary hyperparathyroidism of female rats, which may explain the anti-osteoporotic effect of ursolic acid [59]. Linoleic acid is the main source of n−6 fatty acids, which are able to regulate calcium balance, osteoblastogenesis, and osteoblast activity [60]. Gallic acid and β-sitosterol have also been shown to have osteogenic activity and a promotional effect on serum calcium and ALP in an OVX-induced rat model [61]. Moreover, our KEGG enrichment analysis revealed that the calcium signaling pathway (hsa04020) is one of the top 25 pathways. Therefore, the anti-osteoporotic ability of CO may be associated with calcium balance; therefore, the calcium regulatory effect of other compounds deserve further investigation.
In terms of KEGG enrichment, the estrogen signaling pathway (hsa04915) ranked first, which means this pathway plays a critical role in CO’s ability to treat osteoporosis (Figure 10). Deficient estrogen is a critical cause of osteoporosis, and estrogens can alleviate bone loss through decreased osteoclastogenesis [62]. Recently, Park et al showed the anti-menopausal effects of CO can promote the expression of ESR1 mRNA, one of the common genes from our results, and statistically significantly improve the BMD of OVX-induced mice [62]. Conjugated linoleic acid has been found to attenuate bone loss in OVX-induced mice resulting from estrogen deficiency [45]. Gallic acid and β-sitosterol can also ameliorate the consequences of OVX due to estrogenic modulation [61]. Retinol, a commonly used vitamin, can intensify the bone constructs and bone mass in estrogen deficient rats [64].
Moreover, based on the result of our present KEGG enrichment analysis, CO may have a regulating role in the osteoclast differentiation pathway (hsa04380). Six common genes, TGFB1, NFKB2, JUN, PPP3R1, PIK3R1, and IFNB1, were enriched in this signaling pathway (Figure 11). These genes have been previously reported to promote osteoclastogenesis [65–67]. Uroslic acid was found to suppress NF-κB signaling and c-Jun N-terminal kinase signaling [43]. A recent study also indicated that ursolic acid negatively regulates PI3K/AKT signaling, inhibiting TGF-β1-induced human endothelial cell fibrosis [68]. Uroslic acid can also reduce phosphorylation of nuclear factor κB kinase subunit β and IκBα, as well as suppress the activation of NF-κB and MAPKs of macrophages under the stimulation of lipopolysaccharide [69]. A previous study demonstrated that linoleic acid can reduce the expression levels of P-mTOR, P-Akt, and P-PI3K proteins in RAW 264.7 cells [70].
The present study has limitations and aspects that can be improved in the future. We discussed only a small proportion of potential anti-osteoporotic mechanisms for CO. Because of inadequate funding, the blood compositions of CO were not confirmed through mass spectrometry, which may have caused certain errors. Moreover, we lacked in vivo and in vitro experimental validation, thereby limiting the reliability of the current results. However, our results still provided us with promising compounds and pathways for prioritizing future research.
Conclusions
The present study illustrated that the potential targets and underlying mechanisms of CO in treating osteoporosis may occur through regulating ROS and calcium metabolism. Based on the KEGG enrichment results, we determined that these anti-osteoporotic effects may be mainly involved with the estrogen signaling pathway and osteoclast differentiation pathway. However, because the present results were generated from data mining and data analysis, in vitro and in vivo experiments should be conducted in the future.
Figures
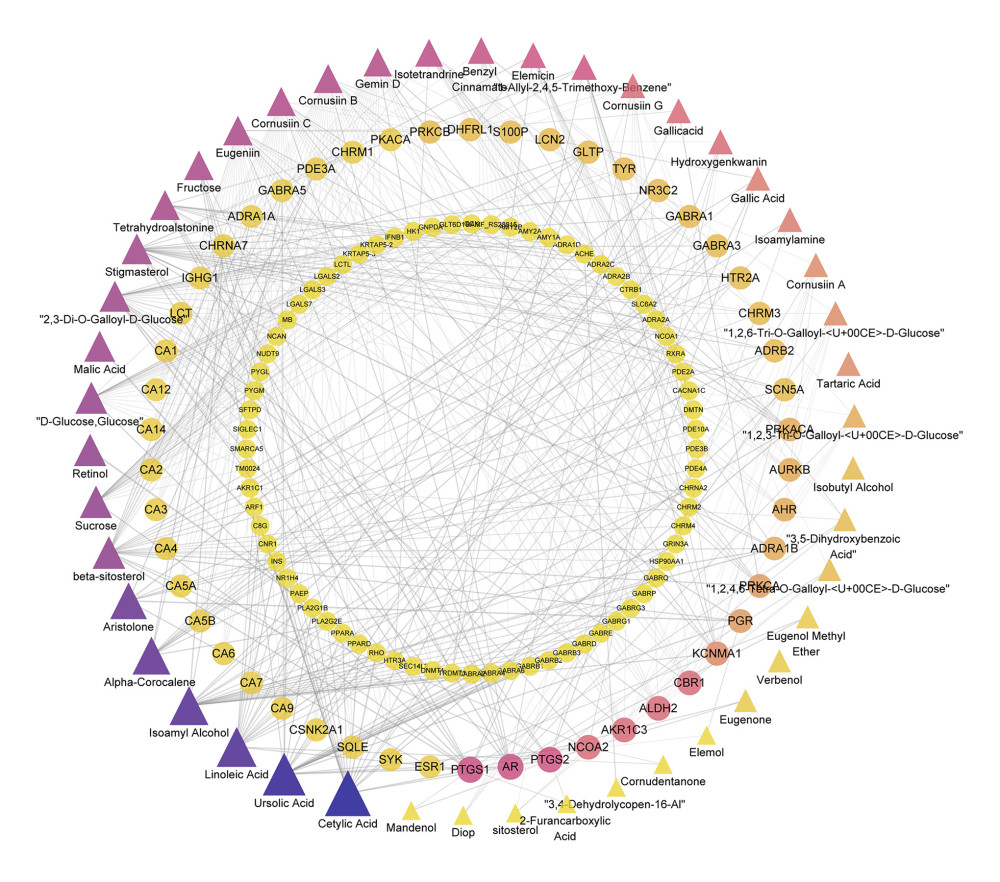 Figure 1. The network for compounds-targets connection. The triangle-shaped nodes are the compounds of Cornus officinalis and the round nodes represent their targets. The area and color of all nodes change according to their degree value. Only the nodes with degrees greater than 2 are presented.
Figure 1. The network for compounds-targets connection. The triangle-shaped nodes are the compounds of Cornus officinalis and the round nodes represent their targets. The area and color of all nodes change according to their degree value. Only the nodes with degrees greater than 2 are presented. 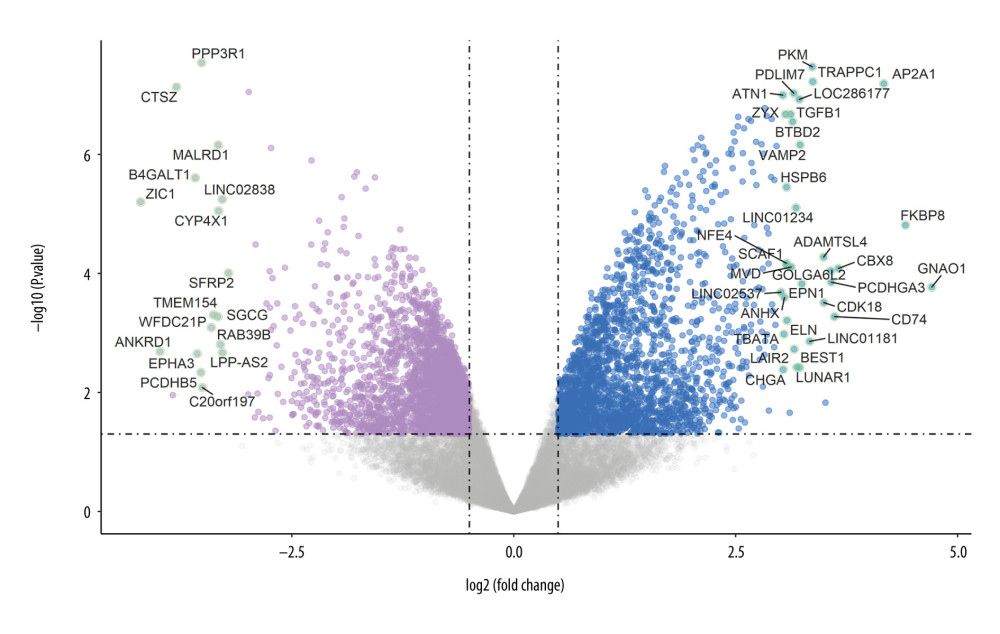 Figure 2. The volcano map to display the gene expression profile of GSE35958. The green dots are the genes with P<0.001 and |logfc| >3.
Figure 2. The volcano map to display the gene expression profile of GSE35958. The green dots are the genes with P<0.001 and |logfc| >3. 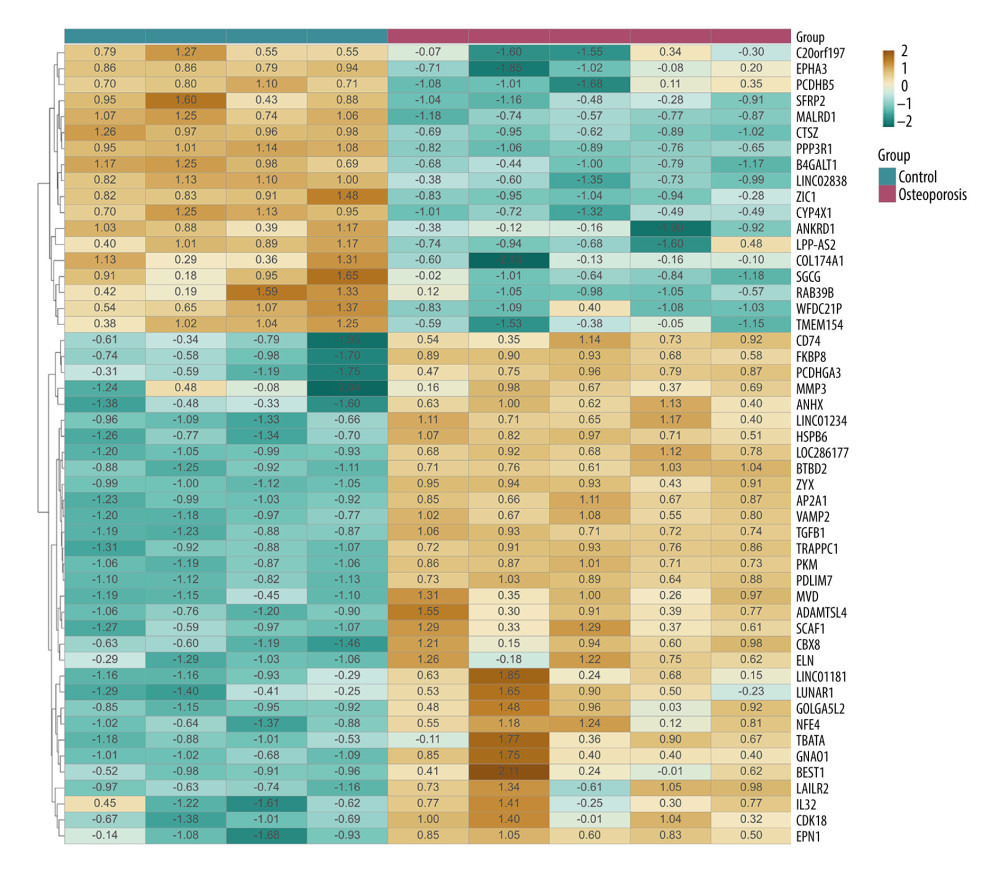 Figure 3. The top 50 differentially expressed genes in GSE35958. The color depth changes are according to their fold change values presented inside the cells
Figure 3. The top 50 differentially expressed genes in GSE35958. The color depth changes are according to their fold change values presented inside the cells 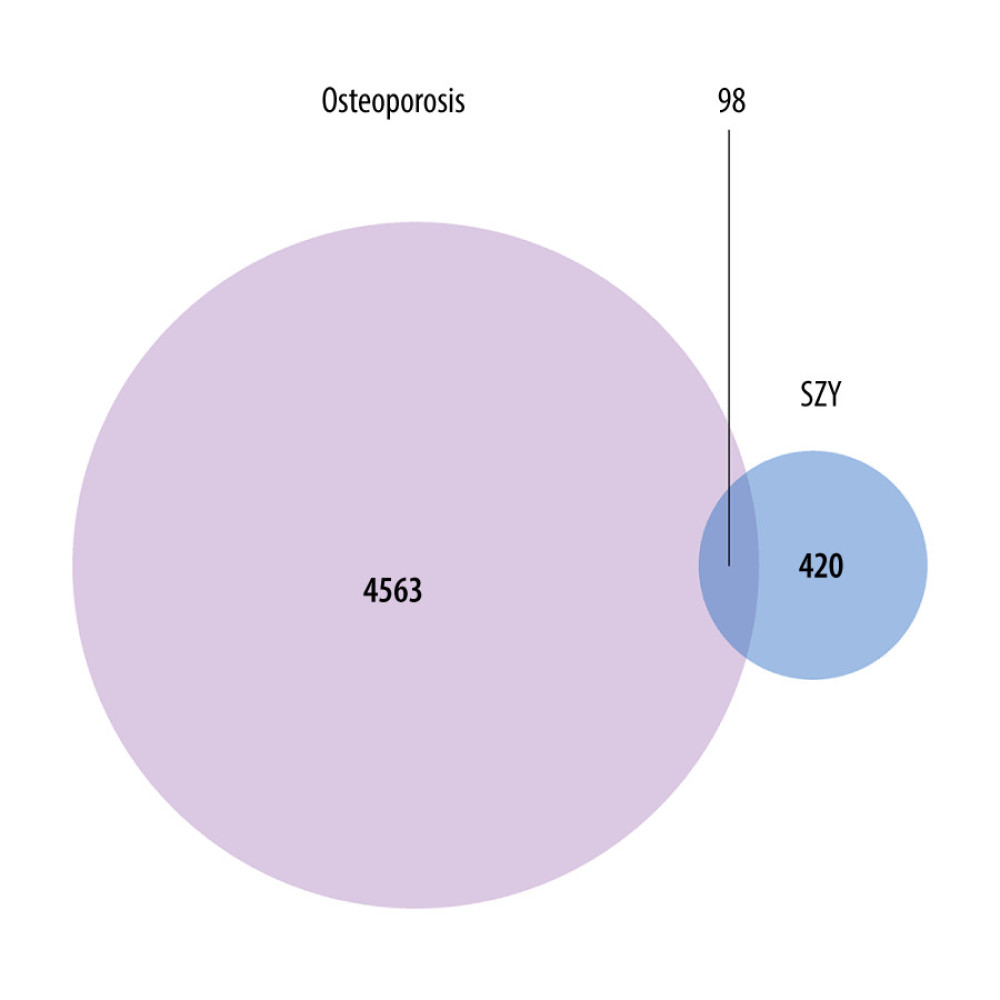 Figure 4. The Venn diagram presents the targets of Cornus officinalis and osteoporosis. The overlapping targets are the potential therapeutic genes for Cornus officinalis to exert its anti-osteoporotic effect.
Figure 4. The Venn diagram presents the targets of Cornus officinalis and osteoporosis. The overlapping targets are the potential therapeutic genes for Cornus officinalis to exert its anti-osteoporotic effect. 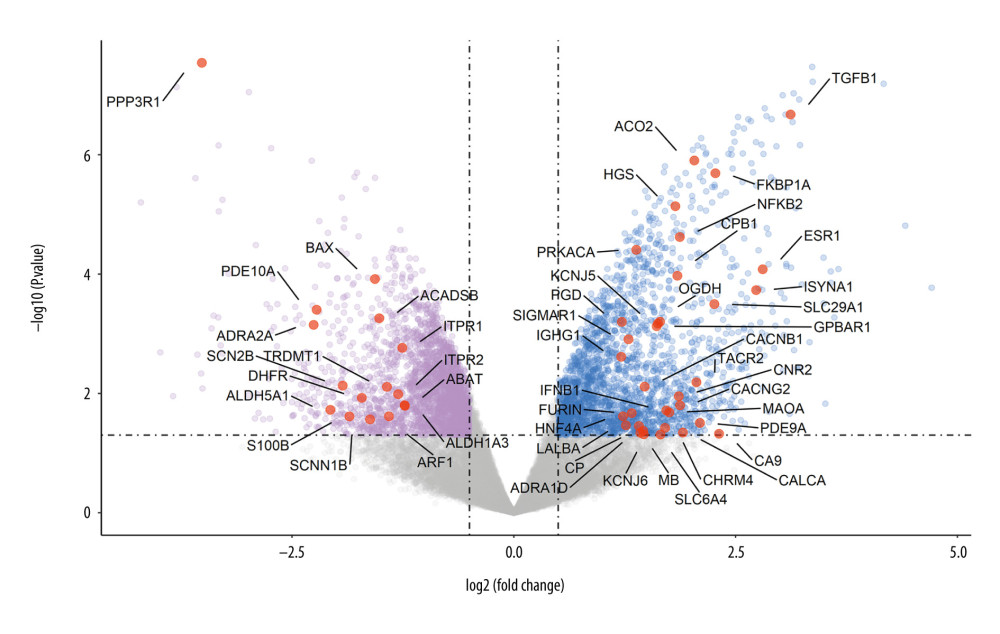 Figure 5. The volcano map displays the gene expression profile of common genes between Cornus officinalis and osteoporosis. The red dots are the top 50 genes within 98 therapeutic targets, according to their fold change values.
Figure 5. The volcano map displays the gene expression profile of common genes between Cornus officinalis and osteoporosis. The red dots are the top 50 genes within 98 therapeutic targets, according to their fold change values. 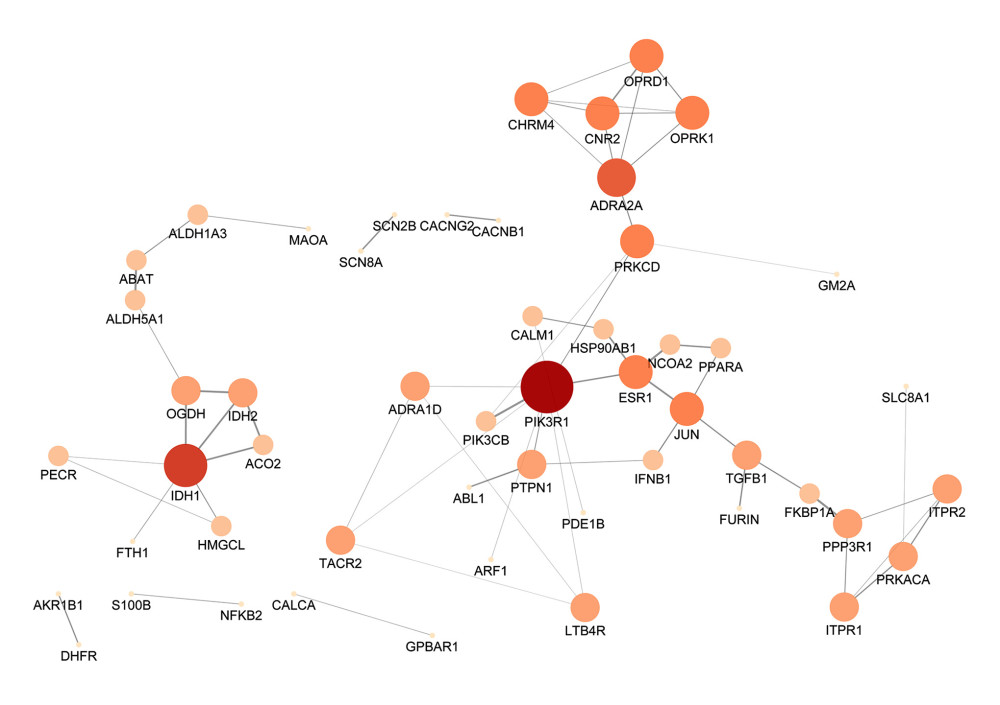 Figure 6. The protein-protein interaction network of 98 candidate targets of Cornus officinalis in treating osteoporosis. The area and color of all nodes change according to their degree value.
Figure 6. The protein-protein interaction network of 98 candidate targets of Cornus officinalis in treating osteoporosis. The area and color of all nodes change according to their degree value. 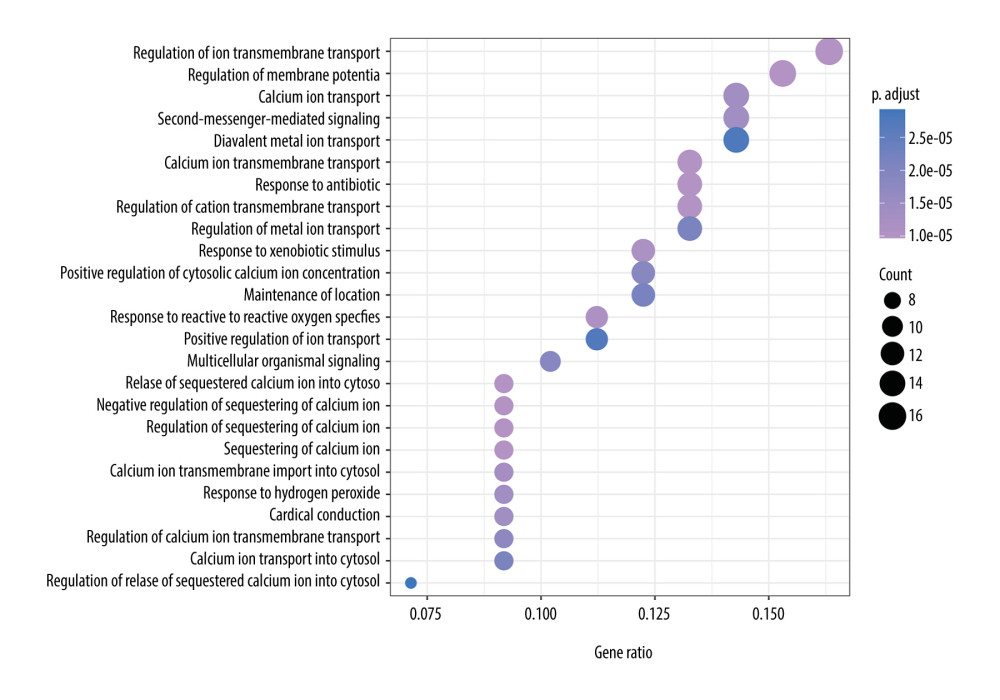 Figure 7. The top 25 processes of the biological process enrichment.
Figure 7. The top 25 processes of the biological process enrichment. 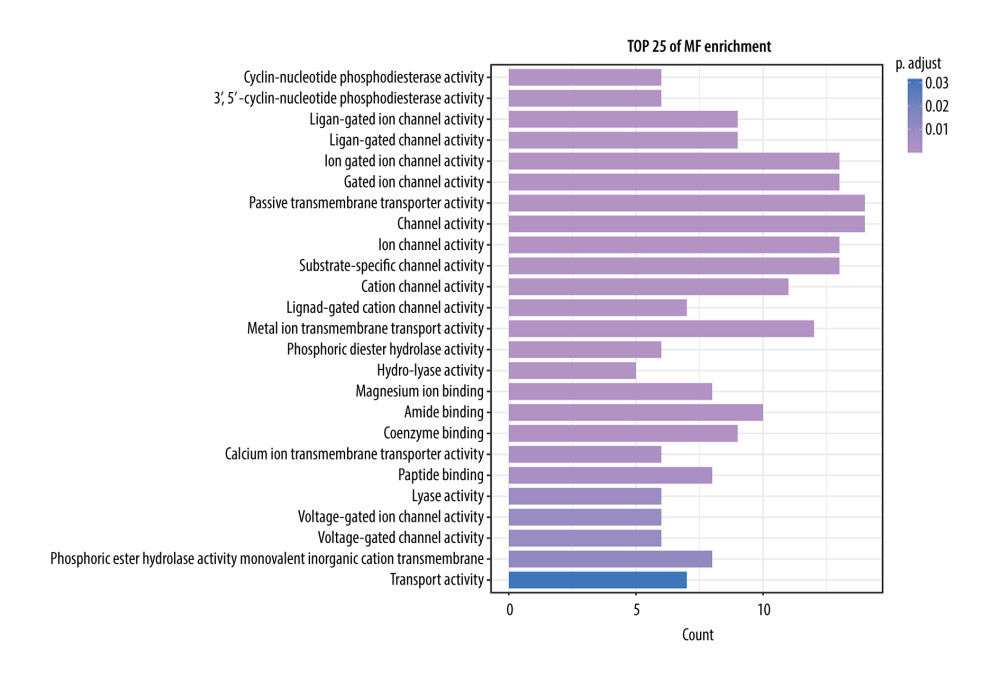 Figure 8. The top 25 processes of the molecular function enrichment.
Figure 8. The top 25 processes of the molecular function enrichment. 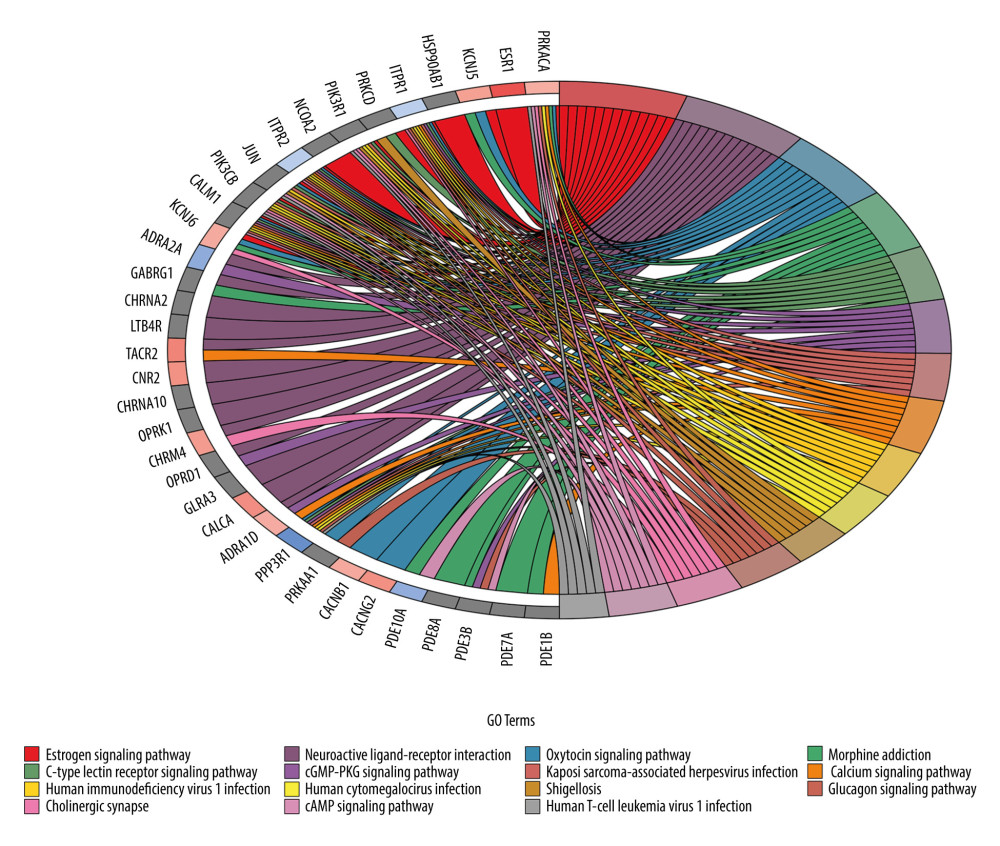 Figure 9. Chord diagram for the top 15 Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment terms.
Figure 9. Chord diagram for the top 15 Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment terms. 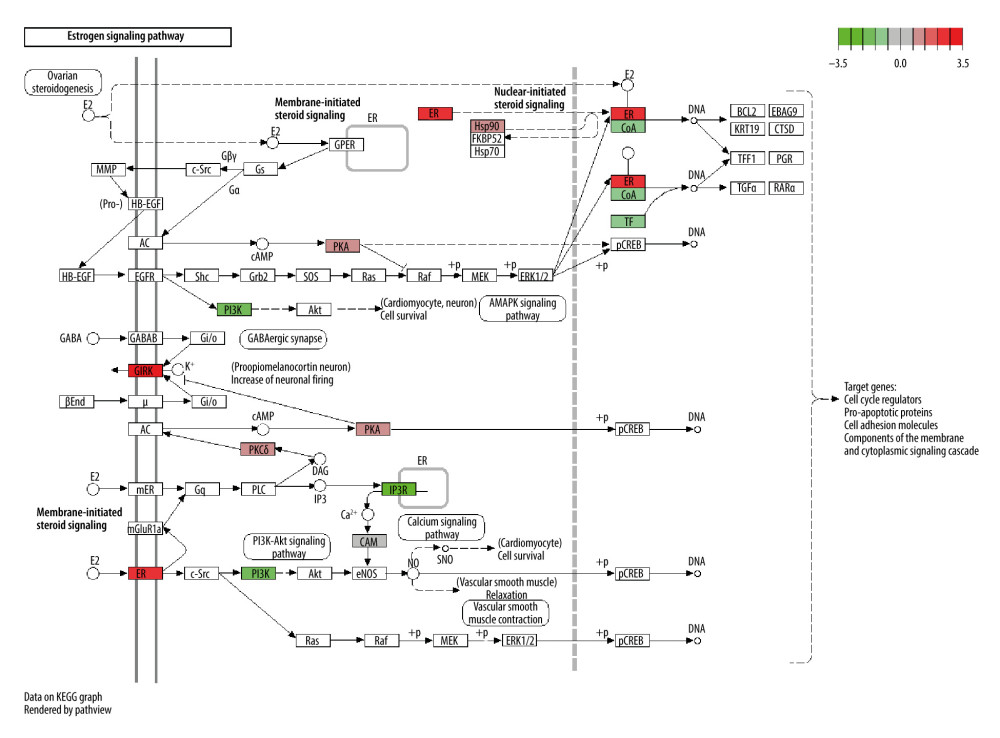 Figure 10. The estrogen signaling pathway ranked first in Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment terms.
Figure 10. The estrogen signaling pathway ranked first in Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment terms. 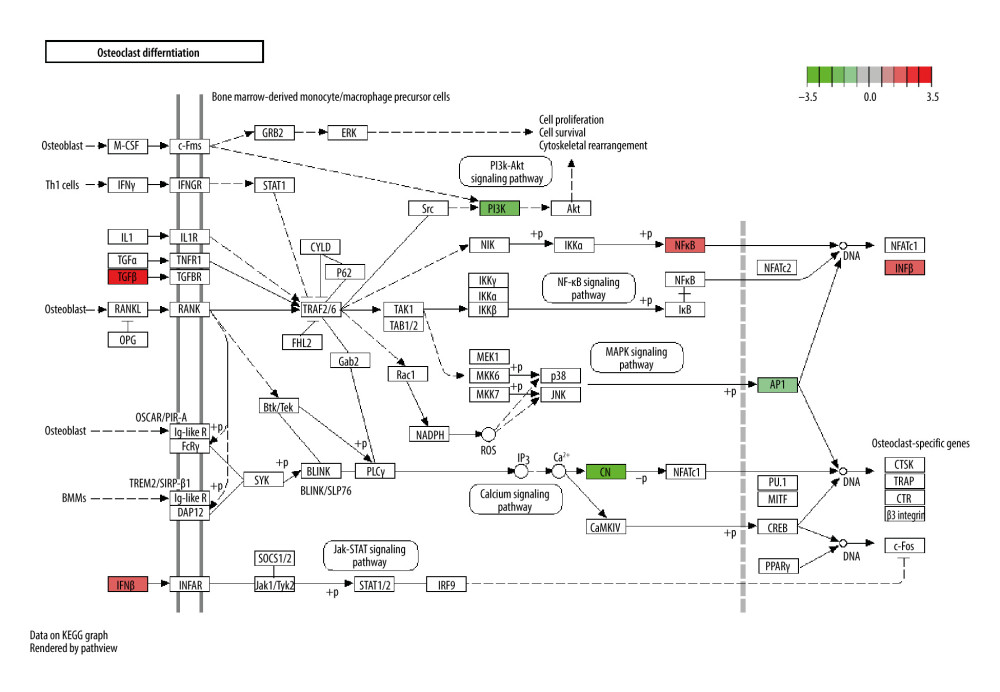 Figure 11. The osteoclast differentiation signaling pathway ranked twenty-first in Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment terms.
Figure 11. The osteoclast differentiation signaling pathway ranked twenty-first in Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment terms. References
1. Zhou L, Hong G, Li S, Fangchinoline protects against bone loss in OVX mice via inhibiting osteoclast formation, bone resorption and RANKL-induced signaling: Int J Biol Sci, 2020; 16(2); 309-19
2. Li JY, Jia YS, Chai LM, Effects of Chinese herbal formula Erxian decoction for treating osteoporosis: A systematic review: Clin Interv Aging, 2017; 12; 45
3. Zeng Q, Li N, Wang Q, The prevalence of osteoporosis in China, a nationwide, multicenter DXA survey: J Bone Miner Res, 2019; 34(10); 1789-97
4. Zhang W, Xue K, Gao Y, Systems pharmacology dissection of action mechanisms of Dipsaci Radix for osteoporosis: Life Sci, 2019; 235; 116820
5. Song M, Zhou L, Liu X, The relationship between digestive system diseases complicated with osteoporosis and intestinal flora: Chin J Osteoporos, 2018; 25(1); 130-34
6. Cosman F, de Beur SJ, LeBof MS, Clinician’s guide to prevention and treatment of osteoporosis: Osteoporos Int, 2014; 25(10); 2359-81
7. Tamechika SY, Sasaki K, Hayami Y, Patient satisfaction and efficacy of switching from weekly bisphosphonates to monthly minodronate for treatment and prevention of glucocorticoid-induced osteoporosis in Japanese patients with systemic rheumatic diseases: A randomized, clinical trial: Arch Osteoporosis, 2018; 13(1); 67
8. Kharazmi M, Schilcher J, Hallberg P, Michaëlsson KJLBisphosphonate-associated atypical fractures of the femur: An update of the current evidence: Lakartidningen, 2019; 116 FMUU [in Swedish]
9. Tabatabaei-Malazy O, Salari P, Khashayar P, Larijani B, New horizons in treatment of osteoporosis: Daru, 2017; 25(1); 2
10. Cano A, Chedraui P, Goulis DG, Calcium in the prevention of postmenopausal osteoporosis: EMAS clinical guide: Maturitas, 2018; 107; 7-12
11. Kraljević , Pavelić S, Micek V, Bobinac D, Treatment of osteoporosis with a modified zeolite shows beneficial effects in an osteoporotic rat model and a human clinical trial: Exp Biol Med (Maywood), 2020 [Online ahead of print]
12. Sham TT, Chan C-O, Wang YH, A review on the traditional Chinese medicinal herbs and formulae with hypolipidemic effect: BioMed Res Int, 2014; 2014; 925302
13. Xiao LJ, Tao R, Traditional Chinese medicine (TCM) therapy: Substance and Non-substance Addiction, 2017; 261-80, Springer
14. Zhou LP, Wong KY, Yeung HT, Bone protective effects of danggui buxue tang alone and in combination with tamoxifen or raloxifene in vivo and in vitro: Front Pharmacol, 2018; 9; 779
15. Gu F, Jiang J, Wang S, An experimental research into the potential therapeutic effects of anti-osteoporosis decoction and Yougui pill on ovariectomy-induced osteoporosis: Am J Transl Res, 2019; 11(9); 6032-39
16. Xiao YP, Zeng J, Jiao LN, Xu XYReview for treatment effect and signaling pathway regulation of kidney – tonifying traditional Chinese medicine on osteoporosis: Zhongguo Zhong Yao Za Zhi, 2018; 43(1); 21-30
17. Kim JY, Kim YK, Choi MK, Effect of Cornus officinalis on receptor activator of nuclear factor-kappaB ligand (RANKL)-induced osteoclast differentiation: J Bone Metab, 2012; 19(2); 121-27
18. Park E, Lee CG, Kim J: Plants, 2020; 9(9); 1114
19. Park E, Lim E, Yeo S: Nutrients, 2020; 12(2); 369
20. Bellavia D, Dimarco E, Costa V, Flavonoids in bone erosive diseases: Perspectives in osteoporosis treatment: Trends Endocrinol Metab, 2020; 32(2); 76-94
21. Wong SK, Chin KY, Ima-Nirwana S, Quercetin as an agent for protecting the bone: A review of the current evidence: Int J Mol Sci, 2020; 21(17); 6448
22. Huang J, Zhang Y, Dong L: J Ethnopharmacol, 2018; 213; 280-301
23. Li M, Wang W, Wang P, The pharmacological effects of morroniside and loganin isolated from Liuweidihuang Wan, on MC3T3-E1 cells: Molecules, 2010; 15(10); 7403-14
24. Zhang Y, Bai M, Zhang B, Uncovering pharmacological mechanisms of Wu-tou decoction acting on rheumatoid arthritis through systems approaches: Drug-target prediction, network analysis and experimental validation: Sci Rep, 2015; 5; 9463
25. Zhang W, Huai Y, Miao Z, Systems pharmacology for investigation of the mechanisms of action of traditional Chinese medicine in drug discovery: Front Pharmacol, 2019; 10; 743
26. Ru J, Li P, Wang J, TCMSP: A database of systems pharmacology for drug discovery from herbal medicines: J Cheminform, 2014; 6(1); 13
27. Liu Z, Guo F, Wang Y, BATMAN-TCM: A bioinformatics analysis tool for molecular mechanism of traditional Chinese medicine: Sci Rep, 2016; 6; 21146
28. Xu HY, Zhang YQ, Liu ZM, ETCM: An encyclopaedia of traditional Chinese medicine: Nucleic Acids Res, 2019; 47(D1); D976-82
29. Xu X, Zhang W, Huang C, A novel chemometric method for the prediction of human oral bioavailability: Int J Mol Sci, 2012; 13(6); 6964-82
30. Gan D, Xu X, Chen D: Med Sci Monit, 2019; 25; 5700-16
31. Shannon P, Markiel A, Ozier O, Cytoscape: A software environment for integrated models of biomolecular interaction networks: Genome Res, 2003; 13(11); 2498-504
32. Missiuro PV, Liu K, Zou L, Information flow analysis of interactome networks: PLoS Comp Biol, 2009; 5(4); e1000350
33. Szklarczyk D, Morris JH, Cook H, The STRING database in 2017: Quality-controlled protein–protein association networks, made broadly accessible: Nucleic Acids Res, 2017; 45(D1); 362-68
34. Yu G, Wang LG, Han Y, He QY, clusterProfiler: An R package for comparing biological themes among gene clusters: OMICS, 2012; 16(5); 284-87
35. Tatangelo G, Watts J, Lim K, The cost of osteoporosis, osteopenia, and associated fractures in Australia in 2017: J Bone Miner Res, 2019; 34(4); 616-25
36. Luo D, Ren H, Li T, Rapamycin reduces severity of senile osteoporosis by activating osteocyte autophagy: Osteoporos Int, 2016; 27(3); 1093-101
37. Mitchell P, Åkesson K, Chandran M, Implementation of Models of Care for secondary osteoporotic fracture prevention and orthogeriatric Models of Care for osteoporotic hip fracture: Best Pract Res Clin Rheumatol, 2016; 30(3); 536-58
38. Fukumoto S, Matsumoto T, Recent advances in the management of osteoporosis: F1000Research, 2017; 6; 625
39. Rossini M, Adami G, Adami S, Safety issues and adverse reactions with osteoporosis management: Expert Opin Drug Saf, 2016; 15(3); 321-32
40. Kharazmi M, Hallberg P, Warfvinge G, Michaëlsson K, Risk of atypical femoral fractures and osteonecrosis of the jaw associated with alendronate use compared with other oral bisphosphonates: Rheumatology (Oxford), 2014; 53(10); 1911-13
41. Wang X, Yamauchi K, Mitsunaga T, A review on osteoclast diseases and osteoclastogenesis inhibitors recently developed from natural resources: Fitoterapia, 2020; 142; 104482
42. Lee SU, Park SJ, Kwak HB, Anabolic activity of ursolic acid in bone: Stimulating osteoblast differentiation in vitro and inducing new bone formation in vivo: Pharmacol Res, 2008; 58(5–6); 290-96
43. Jiang C, Xiao F, Gu X, Inhibitory effects of ursolic acid on osteoclastogenesis and titanium particle-induced osteolysis are mediated primarily via suppression of NF-κB signaling: Biochimie, 2015; 111; 107-18
44. Still K, Grabowski P, Mackie I, The peroxisome proliferator activator receptor alpha/delta agonists linoleic acid and bezafibrate upregulate osteoblast differentiation and induce periosteal bone formation in vivo: Calcif Tissue Int, 2008; 83(4); 285-92
45. Rahman MM, Fernandes G, Williams PJL, Conjugated linoleic acid prevents ovariectomy-induced bone loss in mice by modulating both osteoclastogenesis and osteoblastogenesis: Lipids, 2014; 49(3); 211-24
46. Taha IM, Allah AMA, Hamoudah MA, Carlus SJ, Transforming growth factor-β1 gene polymorphism and osteoporosis in postmenopausal egyptian women: Cell Mol Biol (Noisy-le-grand), 2017; 63(11); 53-57
47. Zhou Y, Zhu W, Zhang L, Transcriptomic data identified key transcription factors for osteoporosis in caucasian women: Calcif Tissue Int, 2018; 103(6); 581-88
48. Trajanoska K, Morris JA, Oei L, Assessment of the genetic and clinical determinants of fracture risk: genome wide association and mendelian randomisation study: BMJ, 2018; 362; k3225
49. Wang C, Zhang Z, Zhang H, Susceptibility genes for osteoporotic fracture in postmenopausal Chinese women: J Bone Miner Res, 2012; 27(12); 2582-91
50. Ye C, Zhang W, Hang K, Extracellular IL-37 promotes osteogenic differentiation of human bone marrow mesenchymal stem cells via activation of the PI3K/AKT signaling pathway: Cell Death Dis, 2019; 10(10); 1-12
51. Ma Y, Ran D, Zhao H, Cadmium exposure triggers osteoporosis in duck via P2X7/PI3K/AKT-mediated osteoblast and osteoclast differentiation: Sci Total Environ, 2021; 750; 141638
52. Kim H-N, Ponte F, Nookaew I, Estrogens decrease osteoclast number by attenuating mitochondria oxidative phosphorylation and ATP production in early osteoclast precursors: Sci Rep, 2020; 10(1); 1-17
53. Sheng S, Wang J, Wang L, Network pharmacology analyses of the antithrombotic pharmacological mechanism of Fufang Xueshuantong Capsule with experimental support using disseminated intravascular coagulation rats: J Ethnopharmacol, 2014; 154(3); 735-44
54. Kanehisa M, Sato YJPS, KEGG Mapper for inferring cellular functions from protein sequences: Protein Sci, 2020; 29(1); 28-35
55. Lin W, Ye H, Anticancer activity of ursolic acid on human ovarian cancer cells via ROS and MMP mediated apoptosis, cell cycle arrest and downregulation of PI3K/AKT pathway: J BUON, 2020; 25(2); 750-56
56. Li M, Zhao L, Zhang T, Redox-sensitive prodrug nanoassemblies based on linoleic acid-modified docetaxel to resist breast cancers: Acta Pharm Sin B, 2019; 9(2); 421-32
57. Hwang KA, Hwang YJ, Song J, Antioxidant activities and oxidative stress inhibitory effects of ethanol extracts from Cornus officinalis on raw 264.7 cells: BMC Complement Altern Med, 2016; 16(1); 196
58. Heshmati HM, Khosla S, Burritt MF, Metabolism. A defect in renal calcium conservation may contribute to the pathogenesis of postmenopausal osteoporosis: J Clin Endocrinol Metab, 1998; 83(6); 1916-20
59. Cao S, Tian XL, Yu WX, Oleanolic acid and ursolic acid improve bone properties and calcium balance and modulate vitamin D metabolism in aged female rats: Front Pharmacol, 2018; 9; 1435
60. Maggio M, Artoni A, Lauretani F, The impact of omega-3 fatty acids on osteoporosis: Curr Pharm Des, 2009; 15(36); 4157-64
61. Chauhan S, Sharma A, Upadhyay NK: BMC Complement Altern Med, 2018; 18(1); 233
62. Rooney AM, van der Meulen MC, Mouse models to evaluate the role of estrogen receptor α in skeletal maintenance and adaptation: Ann NY Acad Sci, 2017; 1410(1); 85-92
63. Park E, Lim E, Yeo S: Nutrients, 2020; 12(2); 369
64. Pytlik M, Kaczmarczyk-Sedlak I, Sliwinski L, Effect of concurrent administration of alendronate sodium and retinol on development of changes in histomorphometric parameters of bones induced by ovariectomy in rats: Pol J Pharmacol, 2004; 56(5); 571-80
65. Takayanagi H, Kim S, Matsuo K, RANKL maintains bone homeostasis through c-Fos-dependent induction of interferon-β: Nature, 2002; 416(6882); 744-49
66. Liu LL, Cao ZH, He CL, Ferric Ion Induction of triggering receptor expressed in myeloid cells-2 expression and PI3K/Akt signaling pathway in preosteoclast cells to promote osteoclast differentiation: Orthop Surg, 2020; 12(4); 1304-12
67. Kim JH, Kim N, Signaling pathways in osteoclast differentiation: Chonnam Med J, 2016; 52(1); 12-17
68. Zhu K, Cao C, Huang J, Inhibitory effects of ursolic acid from Bushen Yijing Formula on TGF-β1-induced human umbilical vein endothelial cell fibrosis via AKT/mTOR signaling and Snail gene: J Pharmacol Sci, 2019; 140(1); 33-42
69. Jang SE, Jeong JJ, Hyam SR, Ursolic acid isolated from the seed of Cornus officinalis ameliorates colitis in mice by inhibiting the binding of lipopolysaccharide to Toll-like receptor 4 on macrophages: J Agric Food Chem, 2014; 62(40); 9711-21
70. Platt I, El-Sohemy A, Effects of 9cis,11trans and 10trans,12cis CLA on osteoclast formation and activity from human CD14+ monocytes: Lipids Health Dis, 2009; 8; 15
Figures
 Figure 1. The network for compounds-targets connection. The triangle-shaped nodes are the compounds of Cornus officinalis and the round nodes represent their targets. The area and color of all nodes change according to their degree value. Only the nodes with degrees greater than 2 are presented.
Figure 1. The network for compounds-targets connection. The triangle-shaped nodes are the compounds of Cornus officinalis and the round nodes represent their targets. The area and color of all nodes change according to their degree value. Only the nodes with degrees greater than 2 are presented. Figure 2. The volcano map to display the gene expression profile of GSE35958. The green dots are the genes with P<0.001 and |logfc| >3.
Figure 2. The volcano map to display the gene expression profile of GSE35958. The green dots are the genes with P<0.001 and |logfc| >3. Figure 3. The top 50 differentially expressed genes in GSE35958. The color depth changes are according to their fold change values presented inside the cells
Figure 3. The top 50 differentially expressed genes in GSE35958. The color depth changes are according to their fold change values presented inside the cells Figure 4. The Venn diagram presents the targets of Cornus officinalis and osteoporosis. The overlapping targets are the potential therapeutic genes for Cornus officinalis to exert its anti-osteoporotic effect.
Figure 4. The Venn diagram presents the targets of Cornus officinalis and osteoporosis. The overlapping targets are the potential therapeutic genes for Cornus officinalis to exert its anti-osteoporotic effect. Figure 5. The volcano map displays the gene expression profile of common genes between Cornus officinalis and osteoporosis. The red dots are the top 50 genes within 98 therapeutic targets, according to their fold change values.
Figure 5. The volcano map displays the gene expression profile of common genes between Cornus officinalis and osteoporosis. The red dots are the top 50 genes within 98 therapeutic targets, according to their fold change values. Figure 6. The protein-protein interaction network of 98 candidate targets of Cornus officinalis in treating osteoporosis. The area and color of all nodes change according to their degree value.
Figure 6. The protein-protein interaction network of 98 candidate targets of Cornus officinalis in treating osteoporosis. The area and color of all nodes change according to their degree value. Figure 7. The top 25 processes of the biological process enrichment.
Figure 7. The top 25 processes of the biological process enrichment. Figure 8. The top 25 processes of the molecular function enrichment.
Figure 8. The top 25 processes of the molecular function enrichment. Figure 9. Chord diagram for the top 15 Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment terms.
Figure 9. Chord diagram for the top 15 Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment terms. Figure 10. The estrogen signaling pathway ranked first in Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment terms.
Figure 10. The estrogen signaling pathway ranked first in Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment terms. Figure 11. The osteoclast differentiation signaling pathway ranked twenty-first in Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment terms.
Figure 11. The osteoclast differentiation signaling pathway ranked twenty-first in Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment terms. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387