21 June 2021: Clinical Research
Association of Blood Pressure, White Matter Lesions, and Regional Cerebral Blood Flow
Dong Zhang1ABCDEF*, Jianyu Zhang1CEF, Bo Zhang2ABD, Jin Zhang1BDE, Mingli He1ABDEGDOI: 10.12659/MSM.929958
Med Sci Monit 2021; 27:e929958
Abstract
BACKGROUND: At present, the association between blood pressure, regional cerebral blood flow, and white matter lesions is not well understood.
MATERIAL AND METHODS: A total of 147 subjects aged from 40 to 80 years were assessed by the Fazekas score for white matter lesions, CT perfusion imaging for regional cerebral blood flow, and 24-h ambulatory blood pressure monitoring for blood pressure level and rhythm. Logistic regression analysis was used to obtain the odds ratio and 95% confidence interval between Fazekas scores and relevant factors. The relationship between blood pressure index and regional cerebral blood flow was analyzed through cubic curve estimation.
RESULTS: Fazekas score was negatively correlated with regional cerebral blood flow (r=-0.801; r=-0.831, P<0.001). For subcortical lesion, the regional cerebral blood flow of Fazekas grade 0 was 1.976 times that of Fazekas grade 3 (OR=1.976, 95% CI=1.576-2.477), and for periventricular lesion, the regional cerebral blood flow of Fazekas grade 0 was 2.034 times that of Fazekas grade 3 (OR=2.034, 95% CI=1.602-2.583). Increased nighttime systolic blood pressure may be more dangerous (OR=1.112, 95% CI=1.059-1.169). The day-night systolic blood pressure ratio (OR=0.801, 95% CI 0.711-0.902) and the day-night diastolic blood pressure ratio (OR=0.876, 95% CI 0.807-0.950) were significantly correlated with Fazekas score.
CONCLUSIONS: The decrease of white matter regional cerebral blood flow caused by hypertension is probably one of the important causes of white matter lesions. Patients with white matter lesions should also pay attention to the rhythm of blood pressure when controlling hypertension, especially if their blood pressure is too high or too low at night.
Keywords: Blood Pressure Monitors, Hypertension, Leukoencephalopathies, Regional Blood Flow, Aged, 80 and over, Blood Pressure, Blood Pressure Monitoring, Ambulatory, Cerebrovascular Circulation, Tomography, X-Ray Computed, white matter
Background
White matter lesions (WMLs) found by brain MRI are common among elderly people. WMLs is often asymptomatic in its early stages, but as the disease progresses, patients develop cognitive and motor executive dysfunction [1], and even dementia [2]. There are many risk factors for WMLs, including age, hypertension, and diabetes. Age and hypertension have been identified as the most important risk factors for WMLs. Compared with people with normal blood pressure, patients with hypertension have a higher incidence rate of WMLs, as well as a higher rate of relevant disease progression [3].
The pathophysiology of WMLs has not been fully elucidated. It has been reported that WMLs may be the result of mild chronic white matter hypoperfusion [4]. Results of relevant Japanese studies showed that Alzheimer disease patients with WMLs had significantly decreased regional cerebral blood flow (rCBF) in the memory- and learning-related brain areas, and cognitive function decreased rapidly within 2 years compared with those without WMLs [5]. Similar studies reported that cerebral perfusion in the posterior cerebral artery supply area was associated with WMLs severity and cognitive decline [6]. This regional hypoperfusion is probably caused by cerebral small vascular lesions, and risk factors like hypertension are the cause of vascular lesions [7].
The above results show that hypertension is an important cause of WMLs, and leads to cerebral microvascular disease, which in turn causes brain white matter damage [8]. However, most recent studies failed to clarify the interaction between blood pressure level, rCBF, and WMLs severity at the same time, and often used MRI (eg, in artery spin labeling technology) for measuring CBF, but this method was not approved by the FDA for relatively low signal noise [9]. Therefore, this study utilized craniocerebral computed tomography perfusion (CTP) imaging to record the patients’ white matter rCBF more accurately, and assess the patients’ dynamic blood pressure changes, so as to explore the interaction between blood pressure level, WMLs, and rCBF, and discuss the possible mechanism for the occurrence and development of WMLs from the perspective of blood pressure. Therefore, we hypothesized that: (a) BP is associated with decreased cerebral blood flow in WMLs area and (b) Blood pressure rhythm is associated with decreased cerebral blood flow in the WMLs area.
Material and Methods
SUBJECTS:
This study included 154 patients who were found to have WMLs without any history of cerebral infarction in the Department of Neurology, Lianyungang First People’s Hospital from November 2019 to October 2020. According to the inclusion and exclusion criteria, data anomalies were excluded in 5 patients due to extreme values and other reasons, and 2 were excluded due to MRI intolerance. A total of 147 patients were included. Among them, 135 completed 24-h ambulatory blood pressure monitoring (24hABPM), and 137 completed a craniocerebral CTP examination. For all of the subjects, relevant medical history collection and multiple examinations were conducted. The examinations included routine blood biochemistry, echocardiography, carotid ultrasonography, cognitive function assessment, 24hABPM, and routine cranial MR scan. We performed a prospective observational cohort study to explore the relationship between blood pressure level, rCBF, and severity of WMLs. This study was approved by the Ethics Committee of Lianyungang First People’s Hospital, and all subjects or their legal representatives signed the informed consent form.
INCLUSION CRITERIA:
The inclusion criteria were: (1) 40–80 years old; (2) definite WMLs found by cranial MRI (WMLs were defined as areas of brain parenchyma with an increased signal on both T2-weighted and FLAIR images without corresponding hypointense lesions on the T1-weighted scans).
EXCLUSION CRITERIA:
The exclusion criteria were: (1) Mental disorder or disturbance of consciousness; (2) Secondary hypertension (including increased blood pressure caused by various clear causes, such as renal hypertension, etc); (3) Newly discovered acute stroke (MRI showed high signal on DWI, low signal on T1, and high signal on T2) or past history of severe stroke (at admission, NIH Stroke Scale >5 or infarction that may affect cognitive function and MRI showed low signal on T1, high signal on T2, and low signal on T2 FLAIR); (4) Congenital vascular disease or cerebral artery occlusion; (5) WMLs for other reasons, including multiple sclerosis, hypoxia, and intoxication; (6) Past history of contrast agent allergy or at least 2 time experiences of allergy to other drugs; (7) Heart, liver, or kidney dysfunction; and (8) History of craniocerebral injury or surgery.
CTP SCANNING:
The dual-source Siemens CT imaging system of (Somatzom Definition Flash) was used; its plain scan can cover the whole brain. For the cranial CT volume perfusion imaging (VPCT), a VPCT mode was used for imaging. The z axis whole-brain CTP images were obtained with 0.6 mm of collimation and 100 mm of scan coverage, 100 kV tube voltage, and 80 mAs tube current. A high-pressure syringe was used to inject 50 ml of iodixanol as contrast agent, flow rate: 5~6 ml/s. The scanning was started after a delay of 4 s, and then 40 ml of normal saline was injected at the same flow rate for flushing, recombination thickness: 1 mm. The volume data of 24 phases were obtained, tube voltage: 100 kV, and tube current: 140 mA. After image processing with the Syngo software by the imaging professional operator, according to the MRI images, we selected 2–3 lesion areas from the most obvious levels of WMLS in both left and right subcortical and paraventricular areas to measure CBF. We calculated the average value of all selected areas on one side as the CBF of that side.
MRI SCANNING:
The superconducting nuclear magnetic resonance (GE Signa HDxt 3.0T, the U.S.A) and standard orthonormal coil were used for MRI. During the examination, the patients were on their back, with heads on the headrest. Then, a plain scan was first performed with the following parameters: T1-weighted scan: TR 1900 s, TE 20 ms, slice thickness 5 mm, with 1 mm for the interior, and FOV 230×185 mm; T2-weighted scanning: TR 4000 ms, TE 104 ms, slice thickness 5 mm, with 1 mm of interval, and FOV 230×209 mm; FLAIR scan: TR 9000 ms, TE 120 ms, TI 2500 ms, slice thickness 5 mm, with 1 mm of interval. The report was given by a doctor from the imaging department.
WMLS ANALYSIS:
The scale for WMLs severity rating-Fazekas scale (range: 0–6) was used to score paraventricular and deep white matter lesions separately, and then the total score of the 2 parts was calculated as the final score. Paraventricular or deep white matter high signal score: 0: no lesion, 1: cap-like or pencil-like thin layer lesion, 2: lesion of smooth halo appearance, 3: irregular paraventricular high signal extending to deep white matter. Fazekas grade (0–3): Grade 0: no lesion, grade 1 (mild lesion, 0–1), grade 2 (moderate lesion, 2–3), and grade 3 (severe lesion, 4–6).
BP RECORD:
After bed rest for at least 5 min, a validated electronic sphygmomanometer was used to record the brachial BP of the patient’s left upper limb every 3 min and measured blood pressure 3 times in total. The mean of the 3 measurements was taken as the BP at admission.
After other tests were completed, the patients performed 24hABPM (Spacelabs Healthcare 90217A, US), and the mean SBP and mean DBP were recorded during the day and night. Blood pressure should be measured every 20 min between 6: 00 and 22: 00 (considered as daytime), and every 30 min between 22: 00 and 6: 00 the next day (considered as nighttime). We recorded the mean systolic blood pressure (SBP), mean diastolic blood pressure (DBP), and the standard deviation of blood pressure over the corresponding period. According to the 24-h mean SBP quartile, they were divided into a higher systolic blood pressure group (SBP >133 mmHg) and a lower systolic blood pressure group (SBP ≤133 mmHg). The blood pressure ratio=(daytime blood pressure value–nighttime blood pressure value)/daytime blood pressure value. The blood pressure variation factor (BPV) is defined as the ratio of SD to mean SBP or DBP over the same period and calculated by the following formula: CV=SD/mean BP [10]. During the examination, patients were only allowed to carry out normal activities necessary for life, vigorous exercises or other activities that may cause mental stimulation were prohibited, and other relevant examinations were not carried out temporarily, so as to prevent patients from having blood pressure fluctuations due to stimulation. If a patient was taking antihypertensive drugs, in order to continue to maintain the patient’s BP status at home and to avoid BPV caused by sudden withdrawal or change of medication, we decided not to stop and did not change the patient’s current antihypertensive plan.
COGNITIVE FUNCTION ASSESSMENT:
The cognitive function of all subjects was evaluated using Montreal Cognitive Assessment Scale (MoCA Scale) [11]. During the test, in a quiet and private room, the patient was examined by a professional psychosomatic doctor or neurologist. The tests were conducted strictly following the standard protocols. Additionally, when participants had less than 12 years of education, the original score was increased by 1 point [12].
DATA ANALYSIS:
All data are expressed as mean±standard deviation, percentage, or inter-quartile range depending on corresponding types. Kolmogorov-Smirnov test was used for normality test of data, the Mann-Whitney
Results
GENERAL INFORMATION:
There were 147 cases, including 72 males and 75 females, average age: 67.4 years. There were 59 cases with mild cognitive impairment and 74 cases with significant cognitive impairment (Table 1). Among the 147 patients, 71 (48.3%) had relatively serious periventricular or deep WMLs (Fazekas grade 2 or greater). For age, blood lipid, sex, blood glucose, coronary heart disease, level of education, smoking, and drinking, there was no statistically significant difference between the quartile groups, while for history of high blood pressure (P=0.002), Fazekas score (P<0.001) and MOCA score (P<0.001), the differences were significant. For SBP, DBP, and 24-h average DBP, the differences between groups were significant (P<0.001). For daytime and nighttime SBP and DBP, there were significant differences between groups (for all, P<0.05).
ANALYSIS OF THE RELATIONSHIP BETWEEN THE SEVERITY OF WMLS AND VARIOUS FACTORS:
Single-factor binary logistic regression analysis on the relationship between Fazekas score and various factors showed that age, body mass index, 24-h mean SBP and DBP, daytime mean SBP, and nighttime mean SBP and DBP were all associated with WMLs, but daytime mean DBP was not. Binary multivariate logistic regression analysis showed that the previously significant association remained after accounting for potential confounding factors (eg, age, fasting blood glucose, and blood pressure on admission). The single-factor multivariate logic analysis of the relationship between Fazekas score and rCBF showed that with the reduction of the Fazekas score, the rCBF at the lesion site gradually increased. The multivariate logistic regression analysis found that the OR value was increased after adjustment, and for the subcortical lesions, Fazekas grade 0 rCBF was 1.976 times higher than Fazekas grade 3 rCBF (OR=1.976, 95% CI=1.576 2.477, P<0.001); for the periventricular lesions, Fazekas grade 0 rCBF was 2.034 times higher than Fazekas grade 3 BF (OR=2.034, 95% CI=1.602–2.583, P<0.001) (Table 2).
ANALYSIS OF THE RELATIONSHIP BETWEEN BLOOD PRESSURE AND RCBF:
As shown in Figures 1 and 2, the cubic simulation curves of blood pressure and cerebral blood flow were roughly in a “S” shape. Each curve had 2 inflection points, and there was a rCBF value interval of relatively stable curve between the 2 inflection points. We roughly estimated the possible range for this value: when the subcortical rCBF was 35–55 ml/100 g/min, the corresponding daytime mean SBP/DBP was 130–135/78–83 mmHg, and nighttime mean SBP/DBP was 120–130/70–75 mmHg. When the periventricular CBF was 50–65 ml/100 g/min, the daytime mean SBP/DBP was 130–135/78–81 mmHg, and the nighttime mean SBP/DBP was 120–130/70–75 mmHg.
RELATIONSHIP BETWEEN THE SEVERITY OF WMLS AND CBF:
Spearman correlation analysis results are shown in Figure 3 The subcortical and paraventricular Fazekas scores were negatively correlated with rCBF (r=−0.801; r=−0.831, P<0.001).
Discussion
The present results show that among patients with high blood pressure, the severity of WMLs was significantly correlated with rCBF, and with increase of Fazekas score, rCBF dropped significantly in the subcortical and periventricular lesion areas. The day-night SBP ratio and the day-night DBP ratio were significantly correlated with Fazekas score, which indicated that a low blood pressure drop rate from daytime to nighttime had a protective effect on WMLs.
The participants in this study were screened out from inpatients complicated with various underlying diseases. However, the results still showed that the high blood pressure group was more likely to develop WMLs and had more severe cognitive impairment. Logistic regression analysis showed that both high 24-h mean SBP and high 24-h DBP were independent risk factors for WMLs, where high SBP at nighttime was more dangerous than high SBP at daytime, and high DBP at nighttime was more dangerous than high DBP at daytime. However, the results failed to show that DBP was more dangerous than SBP [13]. In terms of blood pressure circadian rhythm, systolic blood pressure ratio (SBPR) and diastolic blood pressure (DBPR) were protective factors in WMLs. The higher the proportion of normal circadian rhythm, the more likely the patients had fewer WMLs. A study in China showed that WMLs volume increased significantly in patients with long-term deep dipper blood pressure, non-dipper blood pressure, or reversed dipper blood pressure as compared to those with dipper blood pressure [14]. Therefore, we assumed that a good blood pressure rhythm (normal dipper blood pressure) can delay WMLs, while the disturbance of biological rhythm often leads to increased blood pressure at nighttime, which was probably more dangerous than increased blood pressure at daytime. Many previous cross-sectional and longitudinal cohort studies have shown that the strongest risk factor for increase of WMLs with age is hypertension, and the alleviation of hypertension can delay the progression of WMLs [15]. However, there are few published studies indicating how to properly control blood pressure to prevent progression of WMLs, and there is a particular scarcity of large randomized controlled clinical trials. Moreover, when patients stay at home, they often take antihypertensive drugs only once, in the morning. Although this can maintain a stable blood pressure during the day, it is difficult to take effect at night. Therefore, patients tend to pay more attention to the daytime blood pressure value when they are awake and neglect the control of nighttime blood pressure, which may quietly promote the occurrence or progress of WMLs.
Based on the approximate interval of the 2 inflection points, we estimated the corresponding blood pressure interval with an ideal CBF value. In the subcortical and periventricular regions, when the CBF value was roughly within 40–60 ml/100 g/min, the mean daytime systolic/diastolic blood pressure was 130–135/75–85 mmHg, and the mean nighttime systolic/diastolic blood pressure was 120–130/70–75 mmHg. As our blood pressure is measured at the time of hospitalization, the patient’s blood pressure will inevitably have some environmental impact, leading higher values than the recorded value of home blood pressure patients. Therefore, the data indicate that patients can have a good cerebral perfusion level when their daytime/nighttime blood pressure values are slightly lower than the above values. For patients with high blood pressure accompanied by WMLs, it is critical to reduce the blood pressure to a reasonable range. A study published in JAMA indicated that for adult patients with hypertension, after intensive antihypertensive treatment, the WMLs volume was lower and the total cerebral blood volume was higher in the group of target SBP lower than 120 mmHg than in the group of target SBP lower than 140 mmHg, which was close to our predicted blood pressure interval. However, they also suggested that the strategies to reduce blood pressure in other hypertensive populations should be used cautiously because the study did not include patients with baseline diabetes, stroke, serious heart failure, dementia, advanced or severe proteinuria, or those living in nursing homes [16]. WMLs have been proved to be a risk factor for ischemic stroke, and also influence relevant outcomes [17]. Park et al reported that intracranial atherosclerotic stenosis was more correlated with WMLs than extracranial atherosclerotic stenosis [18]. Therefore, many patients have both vascular stenosis and a past history of IS, and our estimated blood pressure range is within the normal blood pressure range, and may even need to maintain a lower blood pressure to effectively delay the progression of WMLs, so this goal may not be applicable for all patients with WMLs. To avoid excessive blood pressure reduction causing transient ischemic attacks and other more serious consequences, we need to weigh the pros and cons, and individually recommend reasonable blood pressure reduction programs to achieve “early detection, early diagnosis, and early treatment”.
Our analysis verified the hypothesis that chronic hypoperfusion with reduced local rCBF is caused by hypertension. According to the results of logistic regression analysis, the rCBF of Fazekas0 patients is significantly higher than that of Fazekas3 patients in both the paraventricular and subcortical regions (Table 2), which indicated that CBF at white matter lesions was significantly lower than in healthy people. This is consistent with the results of van Dalen et al, who concluded that WMLs in elderly hypertensive patients was associated with reduced CBF at the lesion sites, but not associated with peripheral normal white matter or whole-brain low flow perfusion [19]. This also verified the hypothesis that a pathologic mechanism of vascular damage at white matter lesion sites leads to local hypoperfusion. When this reduction in focal cerebral perfusion occurs in the frontal lobe, parietal lobe, middle temporal lobe, and putamen, WMLs gradually develop and damage cognitive functioning [5]. However, some studies have also shown that the causes of WMLs in patients with Alzheimer disease cannot be explained only by the hypoperfusion resulting from small vessel disease because they may also be related to Wallerian degeneration triggered by pathological lesions in cortical AD, especially for WMLs in the parietal lobe [20].
There are many theories concerning the pathogenesis of WMLs, including the theories of chronic cerebral hypoperfusion [21], cerebral blood barrier disorder [22], inflammatory damage [23], and age-related white matter lesions [24], and there is no consensus. Some studies have found that decreased cerebral perfusion can cause bilateral cortical ischemia and hypoxia, thus resulting in microcirculation disorder and worsened neurodegenerative changes [25], and damage to the connections between white matter fibers can lead to secondary cortical damage [26]. Chronic hypertension can thicken the intima of cerebral arterioles, thus reducing cerebral perfusion and leading to demyelination and degeneration of nerve fibers [6]. Anatomically, the blood in the white matter is supplied by the long perforating arteries located in the subarachnoid space without branching. This relatively unique blood supply mode is considered as the anatomical basis for the increased vulnerability of blood vessels in the watershed area to the damage from risk factors such as hypertension, which can result in decreased cerebral perfusion [27]. Studies on large vessels have shown that high blood pressure is one of the important risk factors for carotid atherosclerosis [28]. Therefore, the production of WMLs is likely to be the result of the long-term synergy of the above pathophysiological mechanisms, which are likely to cascade based on hypertension, the recognized risk factor. A study at the University of California showed that even if the duration of hypertension is short or the blood pressure is well controlled, the adverse reactions caused by hypertension cannot be alleviated; furthermore, the microstructural changes of white matter appear in the early stage of hypertension, for which the harm may persist even if the treatment is adequate. Therefore, they believed that for hypertension, prevention was the most important [29].
This study has some limitations. First, only a small number of patients were included, and the patients were all from the wards, which may have biased the blood pressure data. Second, no follow-up was conducted. A reasonable antihypertensive program should be provided for follow-up observation. Third, it is still necessary to verify the range of blood pressure values that can maintain good perfusion.
Conclusions
In conclusion, this study found supporting evidence that the decrease in rCBF caused by increased blood pressure is likely to be one of the causes of WMLs. Patients with WMLs should also pay attention to the rhythm of blood pressure in controlling hypertension, especially when hyperpiesia or hypopiesia occurs at night.
Figures
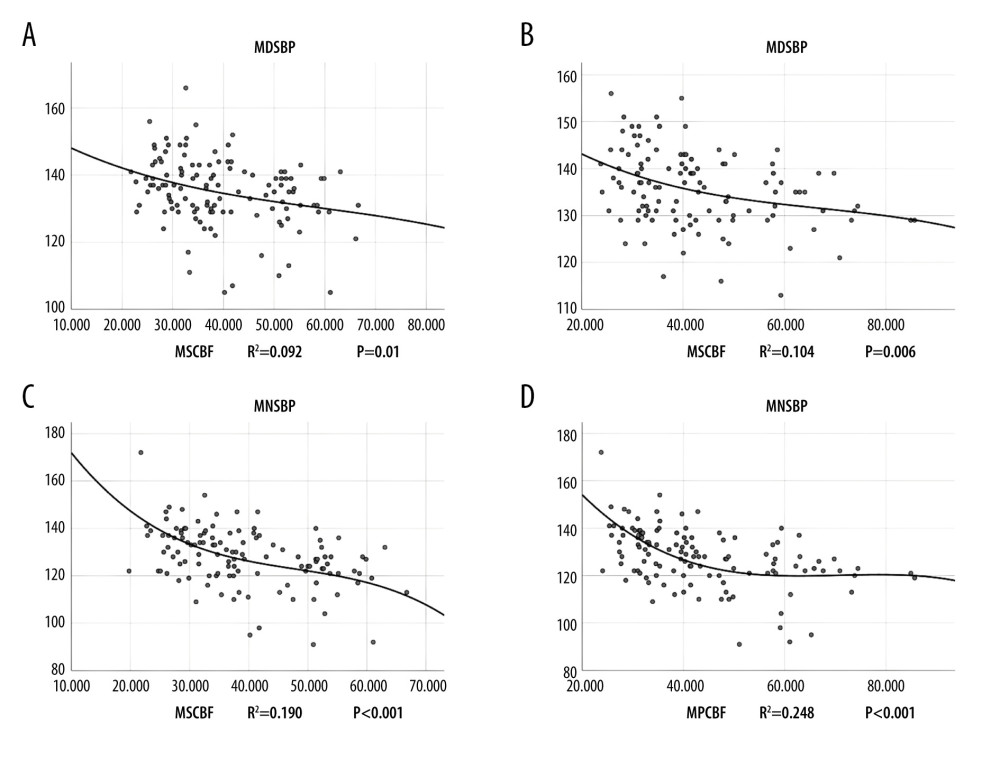 Figure 1. (A–D) The cubic simulation curves of circadian systolic blood pressure. MDSBP – daytime mean systolic blood pressure; MNSBP – mean systolic blood pressure at night; MSCBF – bilateral mean subcortical local cerebral blood flow; MPCBF – bilateral mean paraventricular cerebral blood flow.
Figure 1. (A–D) The cubic simulation curves of circadian systolic blood pressure. MDSBP – daytime mean systolic blood pressure; MNSBP – mean systolic blood pressure at night; MSCBF – bilateral mean subcortical local cerebral blood flow; MPCBF – bilateral mean paraventricular cerebral blood flow. 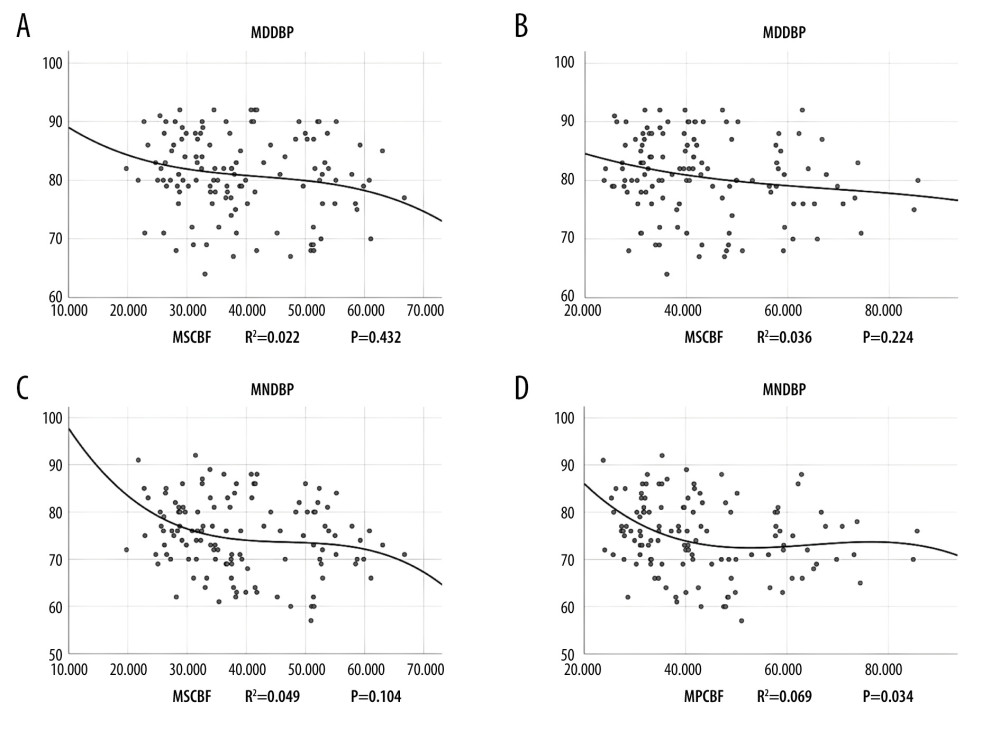 Figure 2. (A–D) The cubic simulation curves of circadian diastolic blood pressure. MDDBP – daytime mean diastolic blood pressure; MNDBP – mean diastolic pressure at night; MSCBF – bilateral mean subcortical local cerebral blood flow; MPCBF – bilateral mean paraventricular cerebral blood flow
Figure 2. (A–D) The cubic simulation curves of circadian diastolic blood pressure. MDDBP – daytime mean diastolic blood pressure; MNDBP – mean diastolic pressure at night; MSCBF – bilateral mean subcortical local cerebral blood flow; MPCBF – bilateral mean paraventricular cerebral blood flow 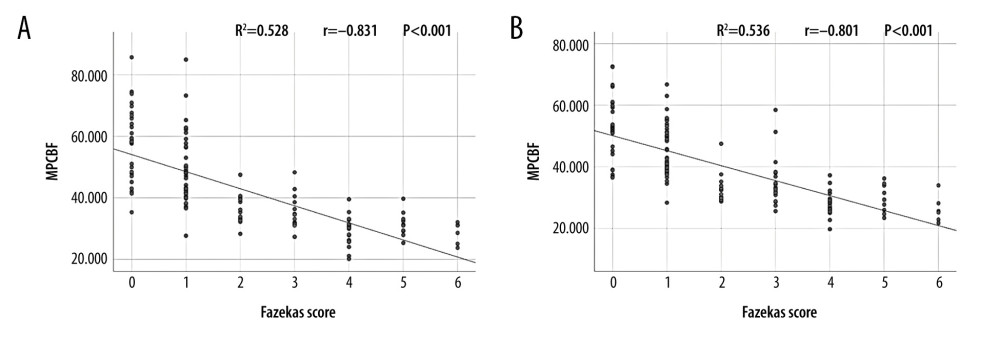 Figure 3. (A, B) The linear relationship between rCBF and Fazekas score. MSCBF – bilateral mean subcortical local cerebral blood flow; MPCBF – bilateral mean paraventricular cerebral blood flow.
Figure 3. (A, B) The linear relationship between rCBF and Fazekas score. MSCBF – bilateral mean subcortical local cerebral blood flow; MPCBF – bilateral mean paraventricular cerebral blood flow. References
1. Jokinen H, Goncalves N, Vigario R, Early-stage white matter lesions detected by multispectral MRI segmentation predict progressive cognitive decline: Front Neurosci, 2015; 9; 455
2. Abraham HM, Wolfson L, Moscufo N, Cardiovascular risk factors and small vessel disease of the brain: Blood pressure, white matter lesions, and functional decline in older persons: J Cereb Blood Flow Metab, 2016; 36; 132-42
3. Hannawi Y, Yanek LR, Kral BG, Hypertension is associated with white matter disruption in apparently healthy middle-aged individuals: Am J Neuroradiol, 2018; 39; 2243-48
4. van Dalen JW, Mutsaerts H, Nederveen AJ, White matter hyperintensity volume and cerebral perfusion in older individuals with hypertension using arterial spin-labeling: Am J Neuroradiol, 2016; 37; 1824-30
5. Ishibashi M, Kimura N, Aso Y, Matsubara E, Effects of white matter lesions on brain perfusion in patients with mild cognitive impairment: Clin Neurol Neurosurg, 2018; 168; 7-11
6. Liu M, Nie ZY, Li RR, Correlation of brain perfusion with white matter hyperintensity, brain atrophy, and cognition in patients with posterior cerebral artery stenosis and subjective cognitive decline: Med Sci Monit, 2018; 24; 5729-38
7. Nilsson PM, Early vascular aging in hypertension: Front Cardiovasc Med, 2020; 7; 6
8. Chen X, Zhu Y, Geng S, Li Q, Jiang H, Association of blood pressure variability and intima-media thickness with white matter hyperintensities in hypertensive patients: Front Aging Neurosci, 2019; 11; 192
9. Jahng GH, Song E, Zhu XP, Human brain: Reliability and reproducibility of pulsed arterial spin-labeling perfusion MR imaging: Radiology, 2005; 234; 909-16
10. Filomena J, Riba-Llena I, Vinyoles E, Short-term blood pressure variability relates to the presence of subclinical brain small vessel disease in primary hypertension: Hypertension, 2015; 66; 634-640 discussion 645
11. Crum RM, Anthony JC, Bassett SS, Folstein MF, Population-based norms for the Mini-Mental State Examination by age and educational level: JAMA, 1993; 269; 2386-91
12. Nasreddine ZS, Phillips NA, Bedirian V, The Montreal Cognitive Assessment, MoCA: A brief screening tool for mild cognitive impairment: J Am Geriatr Soc, 2005; 53; 695-99
13. Zhang D, Tang Y, Ge J, Age and diastolic blood pressure play an important role in the progression of white matter lesions: A meta-analysis: Eur Neurol, 2020; 83; 351-59
14. Zhang H, Cui Y, Zhao Y, Association of circadian rhythm of blood pressure and cerebral small vessel disease in community-based elderly Population: J Gerontol A Biol Sci Med Sci, 2019; 74; 1322-30
15. Wang T, Li Y, Guo X, Reduced perfusion in normal-appearing white matter in mild to moderate hypertension as revealed by 3D pseudocontinuous arterial spin labeling: J Magn Reson Imaging, 2016; 43; 635-43
16. Nasrallah IM, Pajewski NM, Auchus AP, Association of intensive vs standard blood pressure control with cerebral white matter lesions: JAMA, 2019; 322; 524-34
17. Andersen SD, Larsen TB, Gorst-Rasmussen A, White matter hyperintensities improve ischemic stroke recurrence prediction: Cerebrovasc Dis, 2017; 43; 17-24
18. Park JH, Kwon HM, Lee J, Kim DS, Ovbiagele B, Association of intracranial atherosclerotic stenosis with severity of white matter hyperintensities: Eur J Neurol, 2015; 22; 44-52
19. van Dalen JW, Mutsaerts H, Nederveen AJ, White matter hyperintensity volume and cerebral perfusion in older individuals with hypertension using arterial spin-labeling: Am J Neuroradiol, 2016; 37; 1824-30
20. McAleese KE, Walker L, Graham S, Parietal white matter lesions in Alzheimer’s disease are associated with cortical neurodegenerative pathology, but not with small vessel disease: Acta Neuropathol, 2017; 134; 459-73
21. Liu M, Nie ZY, Li RR, Correlation of brain perfusion with white matter hyperintensity, brain atrophy, and cognition in patients with posterior cerebral artery stenosis and subjective cognitive decline: Med Sci Monit, 2018; 24; 5729-38
22. Kimura M, Tanaka A, Yoshinaga S, Significance of periventricular hemodynamics in normal pressure hydrocephalus: Neurosurgery, 1992; 30; 701-4 , discussion 704–5
23. Wang M, Norman JE, Srinivasan VJ, Rutledge JC, Metabolic, inflammatory, and microvascular determinants of white matter disease and cognitive decline: Am J Neurodegener Dis, 2016; 5; 171-77
24. Dupont PS, Bocti C, Joannette M, Amyloid burden and white matter hyperintensities mediate age-related cognitive differences: Neurobiol Aging, 2020; 86; 16-26
25. Vernieri F, Pasqualetti P, Matteis M, Effect of collateral blood flow and cerebral vasomotor reactivity on the outcome of carotid artery occlusion: Stroke, 2001; 32; 1552-58
26. Schmidt R, Ropele S, Enzinger C, White matter lesion progression, brain atrophy, and cognitive decline: The Austrian stroke prevention study: Ann Neurol, 2005; 58; 610-16
27. Kochunov P, Glahn DC, Hong LE, P-selectin expression tracks cerebral atrophy in Mexican-Americans: Front Genet, 2012; 3; 65
28. Cheng Y, Wang YJ, Yan JC, Effects of carotid artery stenting on cognitive function in patients with mild cognitive impairment and carotid stenosis: Exp Ther Med, 2013; 5; 1019-24
29. McEvoy LK, Fennema-Notestine C, Eyler LT, Hypertension-related alterations in white matter microstructure detectable in middle age: Hypertension, 2015; 66; 317-23
Figures
 Figure 1. (A–D) The cubic simulation curves of circadian systolic blood pressure. MDSBP – daytime mean systolic blood pressure; MNSBP – mean systolic blood pressure at night; MSCBF – bilateral mean subcortical local cerebral blood flow; MPCBF – bilateral mean paraventricular cerebral blood flow.
Figure 1. (A–D) The cubic simulation curves of circadian systolic blood pressure. MDSBP – daytime mean systolic blood pressure; MNSBP – mean systolic blood pressure at night; MSCBF – bilateral mean subcortical local cerebral blood flow; MPCBF – bilateral mean paraventricular cerebral blood flow. Figure 2. (A–D) The cubic simulation curves of circadian diastolic blood pressure. MDDBP – daytime mean diastolic blood pressure; MNDBP – mean diastolic pressure at night; MSCBF – bilateral mean subcortical local cerebral blood flow; MPCBF – bilateral mean paraventricular cerebral blood flow
Figure 2. (A–D) The cubic simulation curves of circadian diastolic blood pressure. MDDBP – daytime mean diastolic blood pressure; MNDBP – mean diastolic pressure at night; MSCBF – bilateral mean subcortical local cerebral blood flow; MPCBF – bilateral mean paraventricular cerebral blood flow Figure 3. (A, B) The linear relationship between rCBF and Fazekas score. MSCBF – bilateral mean subcortical local cerebral blood flow; MPCBF – bilateral mean paraventricular cerebral blood flow.
Figure 3. (A, B) The linear relationship between rCBF and Fazekas score. MSCBF – bilateral mean subcortical local cerebral blood flow; MPCBF – bilateral mean paraventricular cerebral blood flow. Tables
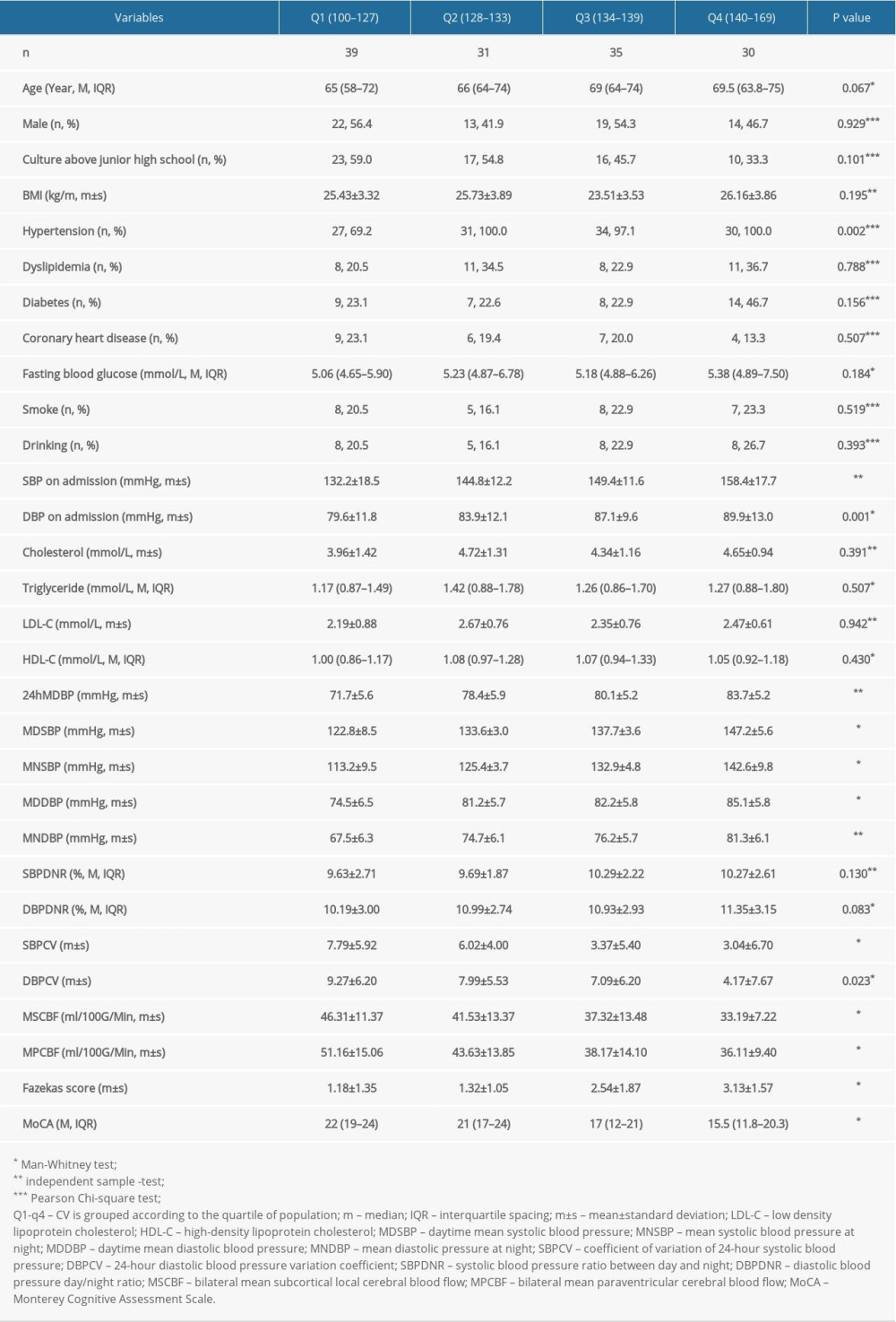 Table 1. Comparison of background data of patients based on 24h mean SBP quartile grouping.
Table 1. Comparison of background data of patients based on 24h mean SBP quartile grouping.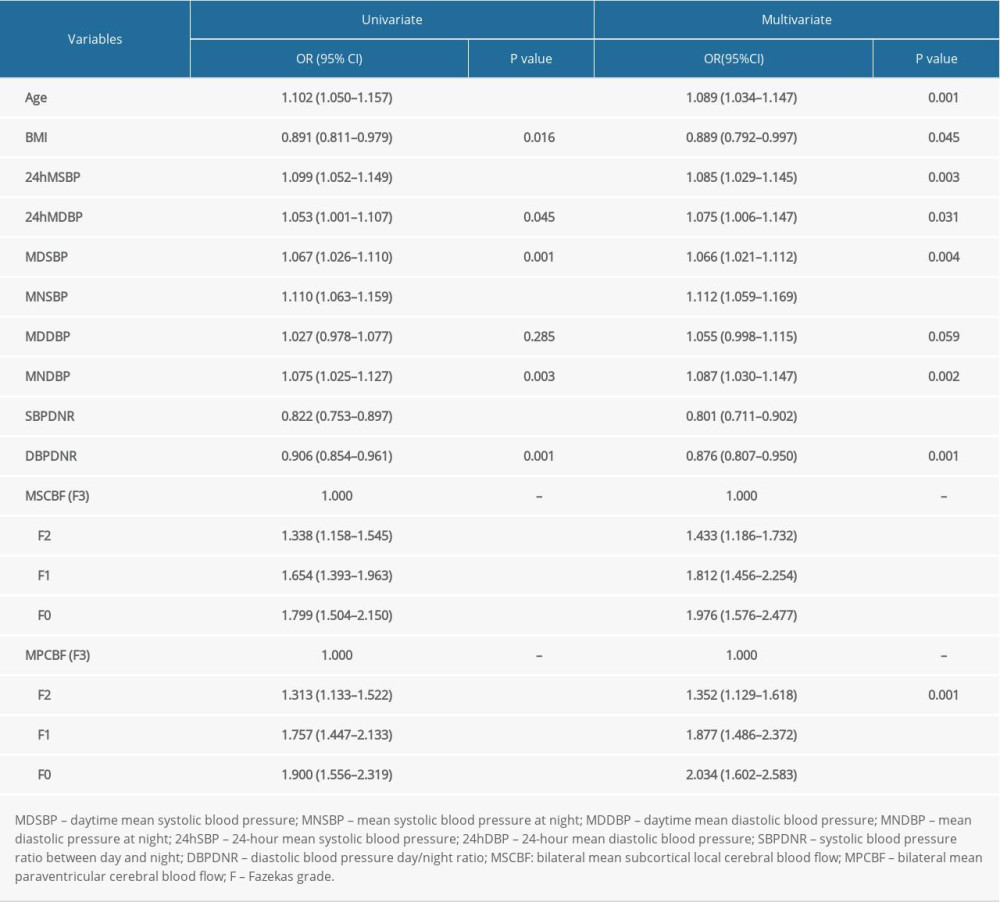 Table 2. Logistics regression analysis of Fazekas grades and various factors.
Table 2. Logistics regression analysis of Fazekas grades and various factors. Table 1. Comparison of background data of patients based on 24h mean SBP quartile grouping.
Table 1. Comparison of background data of patients based on 24h mean SBP quartile grouping. Table 2. Logistics regression analysis of Fazekas grades and various factors.
Table 2. Logistics regression analysis of Fazekas grades and various factors. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








