07 June 2021: Lab/In Vitro Research
Downregulation of Prolactin-Induced Protein Promotes Osteogenic Differentiation of Periodontal Ligament Stem Cells
Xiaomeng Li12BFG, Yunpeng Zhang13DEF, Linglu Jia1BC, Yixiao Xing1BC, Bin Zhao1BC, Lei Sui2CF, Dayong Liu2EF, Xin Xu1AD*DOI: 10.12659/MSM.930610
Med Sci Monit 2021; 27:e930610
Abstract
BACKGROUND: Periodontal ligament stem cells (PDLSCs) are promising seed cells for bone tissue engineering and periodontal regeneration applications. However, the mechanism underlying the osteogenic differentiation process remains largely unknown. Previous reports showed that prolactin-induced protein (PIP) was upregulated after PDLSCs osteogenic induction. However, few studies have reported on the function of PIP in osteogenic differentiation. The purpose of the present study was to investigate the effect of PIP on osteogenic differentiation of PDLSCs.
MATERIAL AND METHODS: The expression pattern of PIP during PDLSCs osteogenic differentiation was detected and the effect of each component in the osteogenic induction medium on PIP was also tested by qRT-PCR. Then, the PIP knockdown cells were established using lentivirus. The knockdown efficiency was measured and the proliferation, apoptosis, and osteogenic differentiation ability were examined to determine the functional role of PIP on PDLSCs.
RESULTS: QRT-PCR showed that PIP was sustainedly upregulated during the osteogenic induction process and the phenomenon was mainly caused by the stimulation of dexamethasone in the induction medium. CCK-8 and flow cytometer showed that knocking down PIP had no influence on proliferation and apoptosis of PDLSCs. ALP staining and activity, Alizarin Red staining, and western blot analysis demonstrated PIP knockdown enhanced the osteogenic differentiation and mineralization of PDLSCs.
CONCLUSIONS: PIP was upregulated after osteogenic induction; however, PIP knockdown promoted PDLSCs osteogenic differentiation. PIP might be a by-product of osteogenic induction, and downregulating of PIP might be a new target in bone tissue engineering applications.
Keywords: Osteogenesis, mesenchymal stromal cells, Down-Regulation, Guided Tissue Regeneration, Periodontal, Membrane Transport Proteins, Periodontal Ligament, Tissue Engineering
Background
Periodontium, including alveolar bone, gingiva, periodontal ligament (PDL), and cementum, is the supporting tissue of the teeth and is essential for the preservation and function of teeth. The PDL tissue, which is located between the alveolar bone and cementum, plays an active role in maintaining periodontal homeostasis [1–3]. Many factors, such as infections, trauma, orthodontic tooth movement, and systemic and genetic diseases, can cause the destruction of periodontal tissues and eventually lead to tooth loss [4–6]. The treatment of periodontal disease, especially the functional regeneration of alveolar bone and PDL tissue, is a major focus of oral regenerative medicine research [7,8].
In tissue engineering regenerative medicine, a recent research focus had been inoculation of cells with scaffold materials [9]. In this area, seed cells and the property of scaffolds are 2 major concerns [10]. Periodontal ligament stem cells (PDLSCs), which are adult stem cells that can be easily obtained from PDL and have great potential to differentiate into multilineage cells, have been extensively studied and are considered to be good seed cells for use in oral tissue engineering [11–14].
However, the specific regulatory mechanism of osteogenic differentiation of periodontal ligament stem cells remains unclear. Over the years, although several studies reported the use of high-throughput methods to detect and analyze the expression changes between osteogenic differentiated and undifferentiated PDLSCs, including mRNAs, microRNAs, long noncoding RNAs, and circular RNAs [2,15–18], few of them did further research to elucidate the function of these differentially expressed molecules.
Prolactin-induced protein (PIP), which is also known as gross cystic disease fluid protein 15 (GCDFP-15), is a 17-kDa glycoprotein that is usually secreted by many kinds of cells [19,20]. PIP expression has been widely reported in breast cancer studies and is one of the biomarkers in diagnosis of breast cancer [21]. Several studies have reported that PIP participates in various cell behaviors, including proliferation, invasion, and apoptosis [22–24]. PIP can be induced by several factors, such as prolactin, androgens, growth hormone, and glucocorticoids [22,25,26]. A gene expression profile study of PDLSCs osteogenic differentiation reported that PIP was upregulated by more than 5-fold, which suggests that PIP has a functional role in this process [2]. It was also reported that PIP is an aspartic-type protease of which the substrate is fibronectin [27]. Fibronectin is a principal component of the extracellular matrix (ECM), and the property of ECM affects the differentiation of stem cells through many mechanisms. For instance, ECM components bind to cell surfaces directly, ECM activates other functional molecules, and the stiffness of ECM also can affect cell fate [28]. Therefore, we speculated that PIP affects the osteogenic differentiation of PDLSCs through the management of ECM.
In the present study, we assessed PIP expression during osteogenic differentiation of PDLSCs, and established PIP knockdown PDLSCs. We further detected the proliferation, apoptosis, and osteogenic differentiation of PDLSCs to investigate the functional role of PIP in PDLSCs.
Material and Methods
PRIMARY PDLSCS CULTURE:
The PDLSCs were isolated as previously described [13,14]. Briefly, premolars were collected from medical wastes of patients 12–25 years old with orthodontic extractions. Teeth with caries and periodontal disease were excluded. The periodontal ligament tissues were scraped off from the middle third of the tooth root and chopped. Collagenase type I (3 mg/mL, Sigma, USA) and dispase (4 mg/ml, Sigma, USA) were used to digest the tissues for 60 min at 37°C. Single-cell suspensions were obtained using a 70-μm cell strainer (BD Falcon). Cell suspensions were seeded into culture dishes and cultured with complete culture medium consisting of α-minimum essential medium (α-MEM) (Corning, USA) supplemented with 10% fetal bovine serum (FBS) (Gibco, USA) at 37°C in 5% CO2. Cells at passages 3–6 were used.
IMMUNOPHENOTYPE ANALYSIS:
As described before [29], we used a BD Stemflow™ hMSC Analysis kit (BD Biosciences, USA). Briefly, after digestion and centrifugation, cells were incubated with CD90-FITC, CD105-PerCP-Cy, CD73-APC, CD44-PE, PE-negative cocktail (CD34-PE, CD11b-PE, CD19-PE, CD45-PE, and HLA-DR-PE) antibodies in the dark at 4°C for 20 min, and the respective isotype control antibodies were used simultaneously as negative controls. The cells were then detected by flow cytometry (BD Biosciences, USA) and the results were analyzed using FlowJo software (version 10.0.6, FlowJo LLC, Ashland, USA).
MULTILINEAGE DIFFERENTIATION:
The multilineage differentiation capacities of cells were detected as previously described [30,31], and cells were directed to osteogenic, adipogenic, and chondrocyte differentiation. Osteogenic induction medium contained α-MEM (Corning, USA) supplemented with 10% FBS (Gibco, USA), 10 nM dexamethasone (Dex) (Solarbio, China), 10 mM β-glycerophosphate (Solarbio, China), and 50 mg/L-ascorbic acid (Sigma, USA). Adipogenic medium was α-MEM (Corning, USA) containing 10% FBS (Gibco, USA), 2 μM Dex (Solarbio, China), 0.2 mM indomethacin (Sigma, USA), 0.01g/L insulin (Sigma, USA), and 0.5 mM isobutyl-methylxanthine (Sigma, USA). The medium was refreshed every 3 days. Alizarin Red staining was used to detect the mineralized nodules after 3 weeks of osteogenic induction and oil red O staining was utilized to show the lipid droplets after 2 weeks of adipogenic induction. A particle culture method was applied for chondrocyte differentiation. The medium was α-MEM (Corning, USA) supplemented with 10% FBS (Gibco, USA), 2 ng/mL transforming growth factor-β1 (TGF-β1; R & D Systems, Minneapolis, MN, USA), 1×ITS+Premix (BD Biosciences, San Jose, CA, USA), 50 mg/mL L-ascorbic acid (Sigma Aldrich, St Louis, MO, USA), 100 mg/mL sodium pyruvate (Wako, Japan), 100 nM Dex (Solarbio, China), and 100 U/mL penicillin/streptomycin. The medium was refreshed every 3 days. After 4 weeks, Alcian Blue staining was used to detect the sulfated proteoglycans by paraffin sectioning.
LENTIVIRUS TRANSFECTION:
The recombinant lentivirus was constructed and produced by Shanghai GenePharma Company (Shanghai, China). The vector used for lentivirus transfection was LV3(H1/GFP&Puro). Short hairpin RNA (shRNA) targeting PIP mRNA was named shPIP and nontargeting negative control was named shNC. The PIP target sequence was: 5′-GCAAATTGCAGCCGTCGTT-3′ and the target sequence of negative control was: 5′-TTCTCCGAACGTGTCACGT-3′. PDLSCs were infected with recombinant lentivirus according to the manufacturer’s instructions. After 6 h of infection, the medium containing lentivirus (MOI=10) was refreshed to complete culture medium. Knockdown efficiency of PIP was measured on gene transcription after 2 days and protein expression levels after 3 days.
CELL PROLIFERATION ASSAY:
The proliferation of cells was detected by Cell Counting Kit-8 (CCK-8) (Dojindo Laboratories, Japan). Cells were plated in a 96-well plate at a density of 4×103 cells/well and incubated for 1, 2, 3, 4, 5, 6, and 7 days, respectively. At each time point, the medium was replaced with fresh medium supplemented with 10% CCK-8 solution and then incubated for an additional 3 h. The absorbance of the medium was measured using a microplate reader (SPECTROstar Nano; BMG Labtech, Germany) at 450 nm wavelength.
CELL CYCLE ANALYSIS:
We plated 5×104 cells into 6-well plates. After 48 h of culture, cell cycle was detected using PI/RNase cell cycle analysis kit (Sungene Biotech, Tianjin, China) by flow cytometer (BD Accuri TM, USA) and analyzed by BD Accuri TM C6 Plus software.
CELL APOPTOSIS ASSAY:
Cell apoptosis assay was performed as previously described [14]. We seeded 1×105 cells into 6-well plates. After reached 80–90% confluency, the cells were treated with 250 μM H2O2 for 4 h. Then, an Annexin V-APC/7-AAD staining kit (Sungene Biotech, China) was used to label cells. Cells were then detected by flow cytometer (BD Accuri TM, USA) and analyzed by BD Accuri TM C6 Plus software.
ALP STAINING AND ALP ACTIVITY ASSAY:
Alkaline phosphatase (ALP) activity assay kit (Nanjing Jiancheng Bioengineering Institute, China) was used to measure ALP activity according to manufacturer’s instructions. The protein of each group was extracted and incubated with detection reagent for 15 min at 37°C. The absorbance at 520 nm wavelength was measured with a microplate reader (SPECTROstar Nano; BMG Labtech). ALP staining was achieved using an NBT/BCIP staining kit (Beyotime, China). Briefly, the cells after 14 days induction were stained with BCIP/NBT solution evade light.
RNA ISOLATION AND QUANTITATIVE REAL-TIME PCR (QRT-PCR) ANALYSIS:
Total RNA of cells was extracted using Trizol (Takara, Tokyo, Japan) method, and PrimeScript™ RT reagent kit with gDNA Eraser (Takara) was used to reverse-transcribe mRNA to cDNA. qRT-PCR assay was performed using the SYBR Green PCR Core kit (Takara). Primers for PIP were (forward: 5′-TAC TGC CTG CCT ATG TGA CG-3′, reverse: 5′-TCA GGG CAG ATG CCT AAT TC-3′), and GAPDH (forward: 5′-GCA CCG TCA AGG CTG AGA AC-3′, reverse: 5′-TGG TGA AGA CGC CAG TGG A-3′). The expression levels of mRNAs were calculated by the 2−ΔΔCt method using GAPDH as an internal reference.
PROTEIN EXTRACTION AND WESTERN BLOT ANALYSIS:
Cells were lysed using RIPA lysis buffer (Solarbio, China) containing 1% PMSF (Solarbio, China). The BCA Protein Assay kit (Solarbio, China) was used to measure the protein concentration. Proteins were separated by SDS-PAGE and then transferred to polyvinylidene difluoride membranes. After blocking with non-fat dry milk for 1 h, the membranes were incubated with primary antibodies at 4°C overnight and incubated with respective secondary antibodies. The following primary antibodies were used: PIP (1: 200, ab218480, abcam), CDK2 (1: 1000, #2546, Cell Signaling Technology), CDK4 (1: 1000, #12790, Cell Signaling Technology), CDK6 (1: 1000, #3136, Cell Signaling Technology), Cyclin B1 (1: 1000, #12231, Cell Signaling Technology), ALP (1: 20000, ab108337, abcam), COL1A1 (1: 1000, #84336, Cell Signaling Technology), OCN (1: 500, ab93876, abcam), GAPDH (1: 20000, HRP-60004, Proteintech). The protein bands were detected by enhanced chemiluminescence (Millipore, USA) under Amersham Imager 600, and the intensities of the bands were analyzed by ImageJ software.
STATISTICAL ANALYSIS:
All experiments were performed 3 times individually from 3 samples and all data are presented as mean±standard deviations. The normal distribution was confirmed using SPSS Statistics 20 (IBM, Armonk, NY, USA). We used the
Results
IDENTIFICATION OF PDLSCS:
Immunophenotype analysis showed that PDLSCs were positive for CD90, CD105, CD73, and CD44, and negative for CD34, CD11b, CD19, CD45, and HLA-DR (Figure 1A). Multilineage differentiation assay showed that PDLSCs could differentiate into osteoblasts, adipocytes, and chondrocytes (Figure 1B–1D). Therefore, PDLSCs were identified as multi-potent mesenchymal stem cells (MSCs) and were used in the following experiments.
PIP EXPRESSION DURING PDLSCS OSTEOGENIC DIFFERENTIATION:
To understand the role of PIP in osteogenesis, qRT-PCR was used to detect the mRNA expression of PIP in osteo-differentiated and undifferentiated PDLSCs on days 3, 7, 14, and 21 during PDLSCs osteogenic differentiation. As shown in Figure 2A, at all time points, the PIP expression level of the osteogenic induction group was higher than that of the non-induced group.
Since there are Dex, β-glycerophosphate, and ascorbic acid in the osteogenic induction system, we further explored which one mainly contributed to the expression change of PIP. After incubating cells with Dex, β-glycerophosphate, or ascorbic acid, respectively, for 7 days, we detected the expression of PIP and confirmed that the Dex group had the highest PIP level (Figure 2B).
KNOCKDOWN EFFICIENCY OF PIP:
The GFP expression under the microscope showed that more than 90% of the PDLSCs were successfully transfected with lentivirus in the shNC and shPIP groups (Figure 3A). The results of qRT-PCR and western blot analysis confirmed that the expression level of PIP was successfully knocked down (Figure 3B–3D).
KNOCKING DOWN PIP HAD NO INFLUENCE ON PDLSCS PROLIFERATION:
To explore the effects of PIP knockdown on cell growth, we used CCK-8 to compare the cell proliferation abilities between the shNC and shPIP groups. As shown in Figure 4A, there was no significant difference in cell numbers between the 2 groups at each time point up to 7 days. Simultaneously, the results of cell cycle and relative proteins showed that PIP knockdown had no influence on PDLSCs cell cycle (Figure 4B, 4C).
PIP EXPRESSION LEVEL DID NOT AFFECT PDLSCS APOPTOSIS:
In shNC PDLSCs, the early and late apoptotic rate was 4.2% and 3.2%, respectively, while in the shPIP group the rate was 4.2% and 3.9%, respectively (Figure 5A). After statistical analysis, no significant difference was shown in apoptotic rate between the 2 groups. We further detected the expression of several cell apoptosis related genes in cells after H2O2 treatment, and found that the protein levels of BAX, Bcl-2, c-IAP1, and c-IAP2 were unchanged in the PIP knockdown group compared with the control group (Figure 5B).
DOWNREGULATION OF PIP ENHANCED PDLSCS OSTEOGENIC DIFFERENTIATION:
After knocking down PIP, the ALP staining was enhanced (Figure 6A), and the ALP activity increased (Figure 6B) compared to the control group on 14 days. The Alizarin Red staining of shPIP PDLSCs was also stronger than in the shNC group (Figure 6C) after osteogenic induction for 21 days. In addition, the expression of osteogenesis-related genes, COL1 and ALP, was improved when PIP was knocked down (Figure 6D, 6E).
Discussion
PIP is a small secreted glycoprotein which was first reported as GCDFP-15 detected in the cystic fluid from mastopathy and now has been verified to exist in many physiological secretions, including saliva, seminal plasma, amniotic fluid, lacrimal fluid, and sweat gland secretions [19,20,32,33]. The currently accepted name, PIP, came from research demonstrating the expression of this protein could be significantly induced by human prolactin (hPRL) or human growth hormone, a hormone structurally and functionally similar to hPRL, in breast cancer T-47D cells [26].
Studies have shown that the abnormal expression of PIP is related to some pathological conditions. For instance, PIP is overexpressed in luminal A and molecular apocrine subtypes of breast cancer [34]. In keratoconus (KC) disease, tear samples from healthy subjects had significantly higher intensity of PIP than those from KC subjects [35]. Another study reported that the amount of PIP was lower in tape-stripped stratum corneum extracts from atopic dermatitis patients compared to health controls and the same trend was observed in sweat samples [36]. The existence of PIP in physiological and pathological conditions suggests that it might be involved in multiple functional activities of cells and organisms. However, its exact functions have not been fully elucidated either in cancerous or normal cells [37] and its role in MSCs differentiation has never been reported.
In the present study, we demonstrated that PIP was significantly upregulated in PDLSCs after osteogenic differentiation and this phenomenon might mainly due to the stimulation of Dex. Dex is a clinically used synthetic glucocorticoid for anti-inflammation and immunosuppression, and is a glucocorticoid receptor (GR)-specific ligand. GR belongs to the nuclear receptors superfamily, which is a group of ligand-regulated transcriptional regulatory factors (TRF), it regulates transcriptional activity of target genes through interacting with specific DNA sequences in their promoter regions [38,39]. There are several patterns that GR directly or indirectly binding DNA sequences in vitro [40]. A half-site DNA sequence of canonical GR-binding sequence, 5′-AGAACA-3′, is found at the glucocorticoid response element in the PIP promotor region (data not shown), which suggests that Dex promotes the expression of PIP through a GR-dependent pathway.
Dex can also stimulate the expression and activation of Runx2, an important osteogenic differentiation-related TRF (reviewed in [41]). Runx2 can participate in the upregulation of PIP expression together with androgen receptor (AR), another nuclear receptor subfamily member, in T-47D breast cancer cells [42]. Considering the conservatism between GR and AR, it could be conjectured that Runx2 promotes PIP expression under osteogenic induction and may work synergistically with GR. However, further experimental research is needed to determine the exact mechanism controlling the expression of PIP.
Exogenous PIP was reported to possess a mitogenic effect on both breast cancer cell lines and immortal breast-cell lines; however, this effect was not observed on colon-cancer cell lines, neuroblastoma cell lines, and small-cell lung carcinoma cells [43]. These results indicated that the function of PIP might be cell type-specific. Another study demonstrated that PIP is required for cell cycle progression, including the progression through G1 phase, mitosis, and cytokinesis, through regulating genes controlling these processes, such as cyclin D1, cyclin B1, BUB1, and FOXM1 [44]. Sugiura et al found that PIP could induce the proliferation of epidermal keratinocytes in a human 3-D skin model and speculated that this phenomenon might be caused by an indirect effect of PIP through stratum corneum damage [45]. Other researchers reported the opposite finding that androgens could increase the expression of PIP and strongly inhibits proliferation of breast cancer cells in an estrogen-sensitive ZR-75-1 cell line [46]. The paradoxical results show the multiple functions of PIP in regulating cell proliferation. However, the results of the present study showed that knockdown of PIP did not influence the number of PDLSCs cells during a 7-day continuous culture, and PIP downregulation had no effect on cell cycle and relative protein levels. These results suggested that PIP might not directly regulate the proliferation of mesenchymal stem cells, at least PDLSCs.
We next assessed whether changes in PIP expression levels will affect PDLSCs apoptosis. The results showed that downregulation of PIP had no influence on the early and late apoptosis rate of PDLSCs under H2O2 stimulation. The expression level of pro-apoptotic protein BAX and anti-apoptotic protein Bcl-2, c-IAP1, and c-IAP2 showed no difference from that of the control group. These data suggested that PIP might not participate in the regulation of mesenchymal stem cells apoptosis. PIP can strongly inhibit T lymphocyte programmed cell death and upregulate the expression of Bcl-2 in these cells through binding to CD4 [24].
The performance of osteogenic differentiation is one of the most important abilities of PDLSCs in tissue engineering and periodontal regeneration therapy. Our results indicated that PIP was significantly and persistently upregulated during osteogenic induction, and knocking down its expression enhanced osteogenic differentiation of PDLSCs. However, the underlying mechanism is still unknown. As PIP is a secreted protein which possesses an aspartyl protease activity and contains fibronectin binding domain, it is proved that PIP could degrade fibronectin to peptides [27,47]. Fibronectin is a glycoprotein widely distributed in most ECMs, especially in active morphogenesis regions, and played a prominent role in matrix assembly through binding cell surface receptors to extracellular compounds [48]. Thus, PIP is believed to participate in ECM degradation. Nevertheless, the progressive development of the osteogenic differentiation and osteogenesis involves the production, maturation, and mineralization of ECM [49]. Meanwhile, a recent study also reported that a fibronectin-treated plate can enhance osteoblast differentiation of human bone marrow-derived mesenchymal stem cells [10]. Therefore, we could speculate that the ECM degradation effect caused by PIP-generated fibronectin cleavage might potentially impact the differentiation and mineralization during PDLSCs osteogenic induction and this process might explain why knocking down PIP promoted PDLSCs osteogenic differentiation. However, this mechanism needs further research to confirm.
We generally believe that when we use some drugs to induce cell differentiation, the elevated genes will play a role in promotion. In the present study, we initially thought that PIP would promote osteogenic differentiation, but we found the opposite. In view of the fact that this study used Dex as an osteoinductive system to conduct experiments and the cells responded to the stimuli received, we have reason to believe that not all of these responses are positive, just like the side effects of drugs. The increased expression of PIP may be a side effect of Dex and the function of PIP happens to inhibit osteogenic differentiation. This phenomenon provides us with new strategies for regulating cell differentiation.
Conclusions
The present study demonstrated that the expression of PIP was obviously increased under the osteogenic induction in PDLSCs and this phenomenon was mainly caused by Dex in the osteogenic induction medium. Although downregulation of PIP had no influence on PDLSCs proliferation and apoptosis rate, the osteogenic differentiation and mineralization of PDLSCs was enhanced. We hypothesized that Dex stimulates the expression of PIP through a GR-dependent transcriptional activation and that increased PIP functions as a fibronectin-degrading protein mediating the ECM degradation, which affects osteogenic differentiation and mineralization. The present findings suggest that some genetic changes are by-products of induction, which will have side effects on the induction process. These results also provide a new strategy in the application of PDLSCs.
Figures
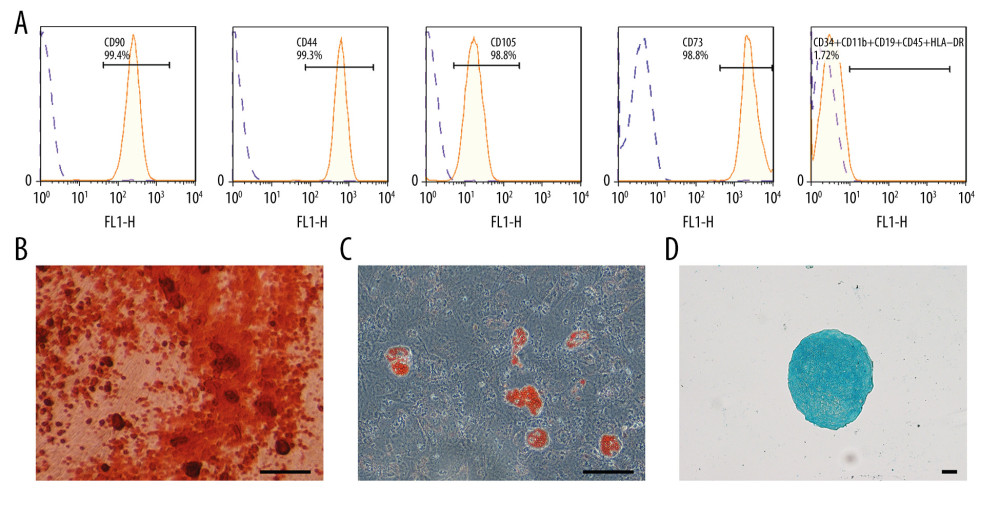 Figure 1. Cultivation and identification of PDLSCs. (A) The immunophenotype analysis of PDLSCs. The detected markers included CD90-FITC, CD105-PerCP-Cy, CD73-APC, CD44-PE, and PE-negative cocktail (CD34-PE, CD11b-PE, CD19-PE, CD45-PE, and HLA-DR-PE). (B) Alizarin Red staining of PDLSCs after 3 weeks of osteogenic induction. (C) Oil red O staining of PDLSCs after 2 weeks of adipogenic induction. (D) Alcian Blue staining of PDLSCs after 4 weeks of chondrogenic induction. (scale bar: 100 μm).
Figure 1. Cultivation and identification of PDLSCs. (A) The immunophenotype analysis of PDLSCs. The detected markers included CD90-FITC, CD105-PerCP-Cy, CD73-APC, CD44-PE, and PE-negative cocktail (CD34-PE, CD11b-PE, CD19-PE, CD45-PE, and HLA-DR-PE). (B) Alizarin Red staining of PDLSCs after 3 weeks of osteogenic induction. (C) Oil red O staining of PDLSCs after 2 weeks of adipogenic induction. (D) Alcian Blue staining of PDLSCs after 4 weeks of chondrogenic induction. (scale bar: 100 μm). 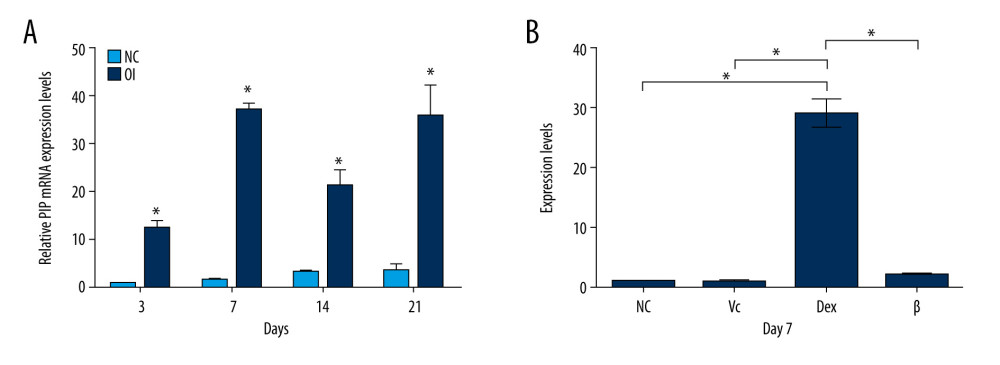 Figure 2. Verification of the PIP expression during PDLSCs osteogenic differentiation. (A) The mRNA expression of PIP in osteo-differentiated (OI) and undifferentiated (NC) PDLSCs on day 3, 7, 14, and 21. (B) The mRNA expression of PIP in PDLSCs incubated in dexamethasone (Dex), β-glycerophosphate (β), ascorbic acid (Vc) and negative control (NC) respectively for 7 days. * P<0.05.
Figure 2. Verification of the PIP expression during PDLSCs osteogenic differentiation. (A) The mRNA expression of PIP in osteo-differentiated (OI) and undifferentiated (NC) PDLSCs on day 3, 7, 14, and 21. (B) The mRNA expression of PIP in PDLSCs incubated in dexamethasone (Dex), β-glycerophosphate (β), ascorbic acid (Vc) and negative control (NC) respectively for 7 days. * P<0.05. 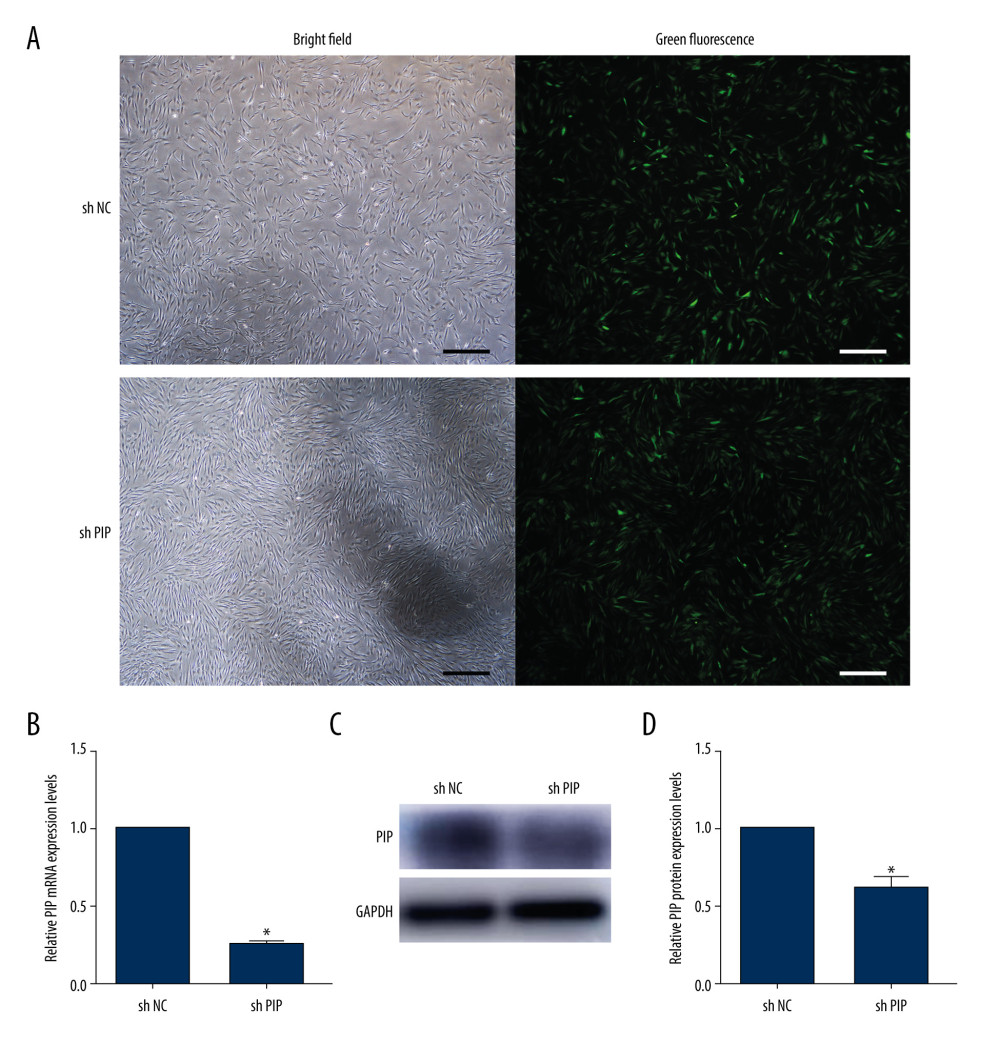 Figure 3. Detection of knockdown efficiency. (A) PDLSCs were transfected with lentivirus and observed under a microscope (scale bar: 200 μm). (B) The expression of PIP mRNA in shNC and shPIP group. (C) The expression of PIP protein in shNC and shPIP group. (D) The quantitative analysis of western blot. * P<0.05.
Figure 3. Detection of knockdown efficiency. (A) PDLSCs were transfected with lentivirus and observed under a microscope (scale bar: 200 μm). (B) The expression of PIP mRNA in shNC and shPIP group. (C) The expression of PIP protein in shNC and shPIP group. (D) The quantitative analysis of western blot. * P<0.05. 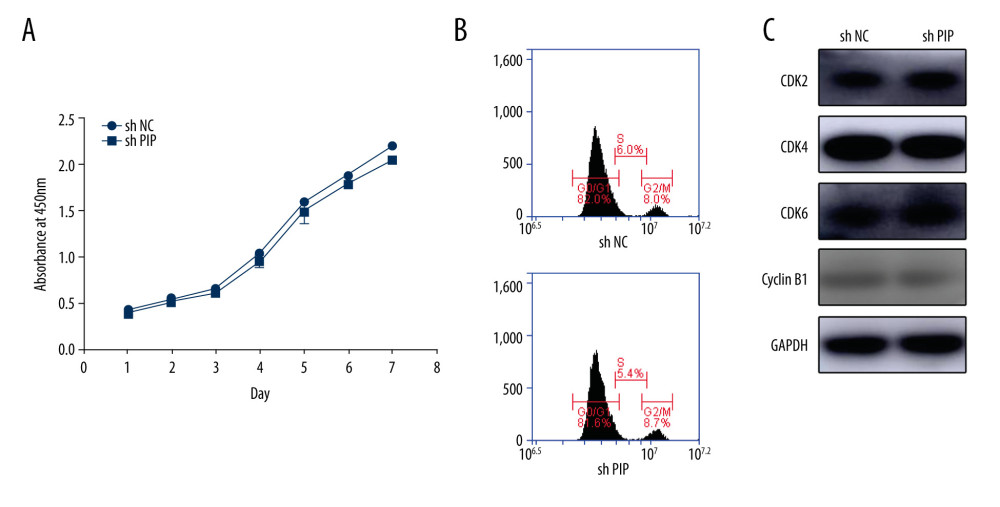 Figure 4. Knocking down PIP had no influence on PDLSCs proliferation. (A) The CCK-8 analysis of shNC and shPIP PDLSCs during the 7 days of cell proliferation. (B) The cell cycle analysis of shNC and shPIP PDLSCs. (C) The expression level of cell cycle relative proteins of shNC and shPIP PDLSCs.
Figure 4. Knocking down PIP had no influence on PDLSCs proliferation. (A) The CCK-8 analysis of shNC and shPIP PDLSCs during the 7 days of cell proliferation. (B) The cell cycle analysis of shNC and shPIP PDLSCs. (C) The expression level of cell cycle relative proteins of shNC and shPIP PDLSCs. 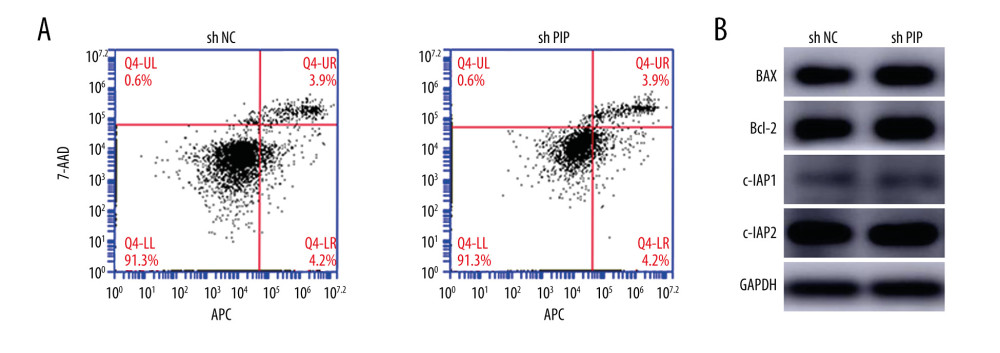 Figure 5. PIP expression level did not affect PDLSCs apoptosis. (A) Cell apoptosis assay of shNC and shPIP PDLSCs after 250 μM H2O2 incubation for 4 h. (B) The protein level of BAX, Bcl-2, c-IAP1, and c-IAP2 in shNC and shPIP PDLSCs.
Figure 5. PIP expression level did not affect PDLSCs apoptosis. (A) Cell apoptosis assay of shNC and shPIP PDLSCs after 250 μM H2O2 incubation for 4 h. (B) The protein level of BAX, Bcl-2, c-IAP1, and c-IAP2 in shNC and shPIP PDLSCs. 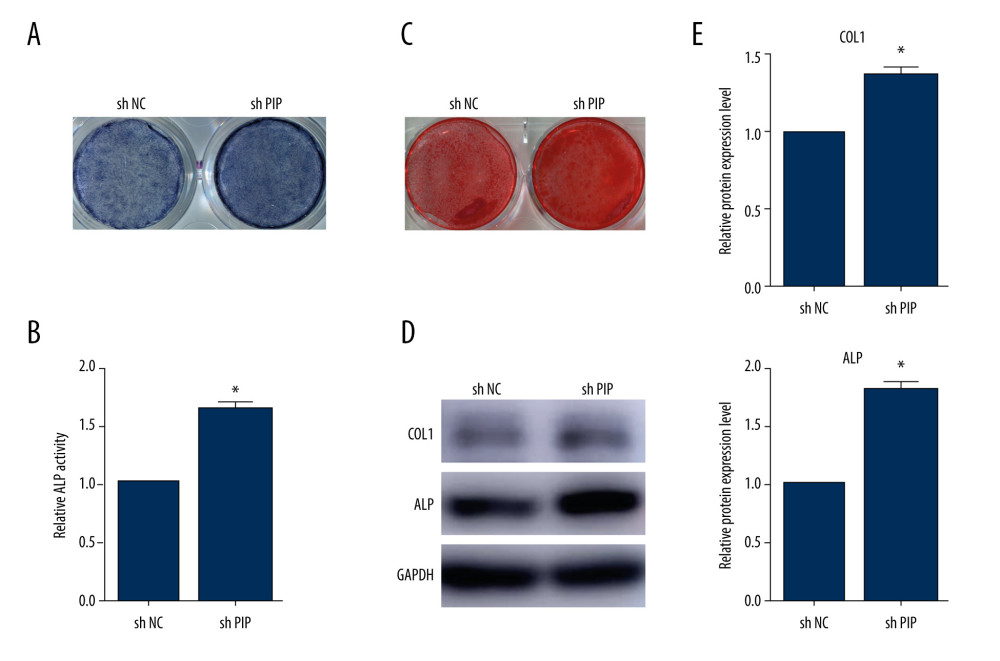 Figure 6. Downregulation of PIP enhanced PDLSCs osteogenic differentiation. (A) ALP staining of PDLSCs after osteogenic induction for 14 days. (B) ALP activity assay of PDLSCs after osteogenic induction for 14 days. (C) Alizarin Red staining of PDLSCs after osteogenic induction for 21 days. (D) The protein level of ALP and COL1 in shNC and shPIP PDLSCs after 14 days of induction. (E) The quantitative analysis of western blot. * P<0.05.
Figure 6. Downregulation of PIP enhanced PDLSCs osteogenic differentiation. (A) ALP staining of PDLSCs after osteogenic induction for 14 days. (B) ALP activity assay of PDLSCs after osteogenic induction for 14 days. (C) Alizarin Red staining of PDLSCs after osteogenic induction for 21 days. (D) The protein level of ALP and COL1 in shNC and shPIP PDLSCs after 14 days of induction. (E) The quantitative analysis of western blot. * P<0.05. References
1. Koori K, Maeda H, Fujii S, The roles of calcium-sensing receptor and calcium channel in osteogenic differentiation of undifferentiated periodontal ligament cells: Cell Tissue Res, 2014; 357(3); 707-18
2. Zheng Y, Li X, Huang Y, Time series clustering of mRNA and lncRNA expression during osteogenic differentiation of periodontal ligament stem cells: Peer J, 2018; 6; e5214
3. Memmert S, Damanaki A, Nogueira AVB, Regulation of tyrosine hydroxylase in periodontal fibroblasts and tissues by obesity-associated stimuli: Cell Tissue Res, 2019; 375(3); 619-28
4. Nesse W, Abbas F, van der Ploeg I, Periodontal inflamed surface area: Quantifying inflammatory burden: J Clin Periodontol, 2008; 35(8); 668-73
5. Arzate H, Zeichner-David M, Mercado-Celis G, Cementum proteins: Role in cementogenesis, biomineralization, periodontium formation and regeneration: Periodontol, 2000; 67(1); 211-33 2015
6. Goncalves-Anjo N, Leite-Pinheiro F, Ribeiro R, Toll-like receptor 9 gene in periodontal disease – a promising biomarker: Gene, 2019; 687; 207-11
7. Orciani M, Trubiani O, Vignini A, Nitric oxide production during the osteogenic differentiation of human periodontal ligament mesenchymal stem cells: Acta Histochem, 2009; 111(1); 15-24
8. Xiang L, Zhang X, Yu H, Overexpression of alphaCGRP promotes osteogenesis of periodontal ligament cells by regulation of YAP signaling: J Cell Physiol, 2019; 234(4); 5077-85
9. Chen M, Xu Y, Zhang T, Mesenchymal stem cell sheets: A new cell-based strategy for bone repair and regeneration: Biotechnol Lett, 2019; 41(3); 305-18
10. Mathews S, Bhonde R, Gupta PK, Extracellular matrix protein mediated regulation of the osteoblast differentiation of bone marrow derived human mesenchymal stem cells: Differentiation, 2012; 84(2); 185-92
11. Seo BM, Miura M, Gronthos S, Investigation of multipotent postnatal stem cells from human periodontal ligament: Lancet, 2004; 364(9429); 149-55
12. Liu N, Zhou M, Zhang Q, Effect of substrate stiffness on proliferation and differentiation of periodontal ligament stem cells: Cell Prolif, 2018; 51(5); e12478
13. Xing Y, Zhang Y, Wu X, A comprehensive study on donor-matched comparisons of three types of mesenchymal stem cells-containing cells from human dental tissue: J Periodontal Res, 2018; 54(3); 286-99
14. Zhang Y, Xing Y, Jia L, An in vitro comparative study of multisource derived human mesenchymal stem cells for bone tissue engineering: Stem Cells Dev, 2018; 27(23); 1634-45
15. Choi HD, Noh WC, Park JW, Analysis of gene expression during mineralization of cultured human periodontal ligament cells: J Periodontal Implant Sci, 2011; 41(1); 30-43
16. Qu Q, Fang F, Wu B, Potential role of long non-coding RNA in osteogenic differentiation of human periodontal ligament stem cells: J Periodontol, 2016; 87(7); e127-37
17. Gu X, Li M, Jin Y, Identification and integrated analysis of differentially expressed lncRNAs and circRNAs reveal the potential ceRNA networks during PDLSC osteogenic differentiation: BMC Genet, 2017; 18(1); 100
18. Hao Y, Ge Y, Li J, Identification of microRNAs by microarray analysis and prediction of target genes involved in osteogenic differentiation of human periodontal ligament stem cells: J Periodontol, 2017; 88(10); 1105-13
19. Haagensen DE, Mazoujian G, Dilley WG, Breast gross cystic disease fluid analysis. I. Isolation and radioimmunoassay for a major component protein: J Natl Cancer Inst, 1979; 62(2); 239-47
20. Haagensen DE, Gall SA, Brazy JE, Analysis of amniotic fluid, maternal plasma, and cord blood for a human breast gross cystic disease fluid protein: Am J Obstet Gynecol, 1980; 138(1); 25-32
21. Clark JW, Snell L, Shiu RP, The potential role for prolactin-inducible protein (PIP) as a marker of human breast cancer micrometastasis: Br J Cancer, 1999; 81(6); 1002-8
22. Baniwal SK, Chimge NO, Jordan VC, Prolactin-induced protein (PIP) regulates proliferation of luminal A type breast cancer cells in an estrogen-independent manner: PLoS One, 2014; 8(6); e62361
23. Naderi A, Meyer M, Prolactin-induced protein mediates cell invasion and regulates integrin signaling in estrogen receptor-negative breast cancer: Breast Cancer Res, 2012; 14(4); R111
24. Gaubin M, Autiero M, Basmaciogullari S, Potent inhibition of CD4/TCR-mediated T cell apoptosis by a CD4-binding glycoprotein secreted from breast tumor and seminal vesicle cells: J Immunol, 1999; 162(5); 2631-38
25. Murphy LC, Tsuyuki D, Myal Y, Isolation and sequencing of a cDNA clone for a prolactin-inducible protein (PIP). Regulation of PIP gene expression in the human breast cancer cell line, T-47D: J Biol Chem, 1987; 262(31); 15236-41
26. Shiu RP, Iwasiow BM, Prolactin-inducible proteins in human breast cancer cells: J Biol Chem, 1985; 260(20); 11307-13
27. Caputo E, Manco G, Mandrich L, A novel aspartyl proteinase from apocrine epithelia and breast tumors: J Biol Chem, 2000; 275(11); 7935-41
28. Muncie JM, Weaver VM, The physical and biochemical properties of the extracellular matrix regulate cell fate: Curr Top Dev Biol, 2018; 130; 1-37
29. Jia L, Xiong Y, Zhang W, Ma X, Metformin promotes osteogenic differentiation and protects against oxidative stress-induced damage in periodontal ligament stem cells via activation of the Akt/Nrf2 signaling pathway: Exp Cell Res, 2020; 386(2); 111717
30. Xing Y, Zhang Y, Jia L: Mol Oral Microbiol, 2019; 34(1); 12249
31. Jia L, Zhang Y, Ji Y, YAP balances the osteogenic and adipogenic differentiation of hPDLSCs in vitro partly through the Wnt/beta-catenin signaling pathway: Biochem Biophys Res Commun, 2019; 518(1); 154-60
32. Schaller J, Akiyama K, Kimura H, Primary structure of a new actin-binding protein from human seminal plasma: Eur J Biochem, 1991; 196(3); 743-50
33. Schenkels LC, Rathman WM, Veerman EC, Detection of proteins related to a salivary glycoprotein (EP-GP). Concentrations in human secretions (saliva, sweat, tears, nasal mucus, cerumen, seminal plasma): Biol Chem Hoppe Seyler, 1991; 372(5); 325-29
34. Naderi A, Prolactin-induced protein in breast cancer: Adv Exp Med Biol, 2015; 846; 189-200
35. Priyadarsini S, Hjortdal J, Sarker-Nag A, Gross cystic disease fluid protein-15/prolactin-inducible protein as a biomarker for keratoconus disease: PLoS One, 2014; 9(11); e113310
36. Kamiya K, Sakabe J, Yamaguchi H, Gross cystic disease fluid protein 15 in stratum corneum is a potential marker of decreased eccrine sweating for atopic dermatitis: PLoS One, 2015; 10(4); e0125082
37. Urbaniak A, Jablonska K, Podhorska-Okolow M, Prolactin-induced protein (PIP)-characterization and role in breast cancer progression: Am J Cancer Res, 2018; 8(11); 2150-64
38. Mangelsdorf DJ, Thummel C, Beato M, The nuclear receptor superfamily: The second decade: Cell, 1995; 83(6); 835-39
39. Grad I, Picard D, The glucocorticoid responses are shaped by molecular chaperones: Mol Cell Endocrinol, 2007; 275(1–2); 2-12
40. Weikum ER, Knuesel MT, Ortlund EA, Glucocorticoid receptor control of transcription: Precision and plasticity via allostery: Nat Rev Mol Cell Biol, 2017; 18(3); 159-74
41. Langenbach F, Handschel J, Effects of dexamethasone, ascorbic acid and beta-glycerophosphate on the osteogenic differentiation of stem cells in vitro: Stem Cell Res Ther, 2013; 4(5); 117
42. Baniwal SK, Little GH, Chimge NO, Runx2 controls a feed-forward loop between androgen and prolactin-induced protein (PIP) in stimulating T47D cell proliferation: J Cell Physiol 20; 227(5); 2276-82
43. Cassoni P, Sapino A, Haagensen DE, Mitogenic effect of the 15-kDa gross cystic disease fluid protein (GCDFP-15) on breast-cancer cell lines and on immortal mammary cells: Int J Cancer, 1995; 60(2); 216-20
44. Naderi A, Vanneste M, Prolactin-induced protein is required for cell cycle progression in breast cancer: Neoplasia, 2014; 16(4); 329-42.e1–4
45. Sugiura S, Tazuke M, Ueno S, Effect of prolactin-induced protein on human skin: New insight into the digestive action of this aspartic peptidase on the stratum corneum and its induction of keratinocyte proliferation: J Invest Dermatol, 2015; 135(3); 776-85
46. Labrie F, Simard J, Poulin R, Potent antagonism between estrogens and androgens on GCDFP-15 expression and cell growth in the ZR-75-1 human breast cancer cells: Ann NY Acad Sci, 1990; 586; 174-87
47. Caputo E, Camarca A, Moharram R, Structural study of GCDFP-15/gp17 in disease versus physiological conditions using a proteomic approach: Biochemistry, 2003; 42(20); 6169-78
48. Halper J, Kjaer M, Basic components of connective tissues and extracellular matrix: Elastin, fibrillin, fibulins, fibrinogen, fibronectin, laminin, tenascins and thrombospondins: Adv Exp Med Biol, 2014; 802; 31-47
49. Stein GS, Lian JB, Molecular mechanisms mediating proliferation/differentiation interrelationships during progressive development of the osteoblast phenotype: Endocr Rev, 1993; 14(4); 424-42
Figures
 Figure 1. Cultivation and identification of PDLSCs. (A) The immunophenotype analysis of PDLSCs. The detected markers included CD90-FITC, CD105-PerCP-Cy, CD73-APC, CD44-PE, and PE-negative cocktail (CD34-PE, CD11b-PE, CD19-PE, CD45-PE, and HLA-DR-PE). (B) Alizarin Red staining of PDLSCs after 3 weeks of osteogenic induction. (C) Oil red O staining of PDLSCs after 2 weeks of adipogenic induction. (D) Alcian Blue staining of PDLSCs after 4 weeks of chondrogenic induction. (scale bar: 100 μm).
Figure 1. Cultivation and identification of PDLSCs. (A) The immunophenotype analysis of PDLSCs. The detected markers included CD90-FITC, CD105-PerCP-Cy, CD73-APC, CD44-PE, and PE-negative cocktail (CD34-PE, CD11b-PE, CD19-PE, CD45-PE, and HLA-DR-PE). (B) Alizarin Red staining of PDLSCs after 3 weeks of osteogenic induction. (C) Oil red O staining of PDLSCs after 2 weeks of adipogenic induction. (D) Alcian Blue staining of PDLSCs after 4 weeks of chondrogenic induction. (scale bar: 100 μm). Figure 2. Verification of the PIP expression during PDLSCs osteogenic differentiation. (A) The mRNA expression of PIP in osteo-differentiated (OI) and undifferentiated (NC) PDLSCs on day 3, 7, 14, and 21. (B) The mRNA expression of PIP in PDLSCs incubated in dexamethasone (Dex), β-glycerophosphate (β), ascorbic acid (Vc) and negative control (NC) respectively for 7 days. * P<0.05.
Figure 2. Verification of the PIP expression during PDLSCs osteogenic differentiation. (A) The mRNA expression of PIP in osteo-differentiated (OI) and undifferentiated (NC) PDLSCs on day 3, 7, 14, and 21. (B) The mRNA expression of PIP in PDLSCs incubated in dexamethasone (Dex), β-glycerophosphate (β), ascorbic acid (Vc) and negative control (NC) respectively for 7 days. * P<0.05. Figure 3. Detection of knockdown efficiency. (A) PDLSCs were transfected with lentivirus and observed under a microscope (scale bar: 200 μm). (B) The expression of PIP mRNA in shNC and shPIP group. (C) The expression of PIP protein in shNC and shPIP group. (D) The quantitative analysis of western blot. * P<0.05.
Figure 3. Detection of knockdown efficiency. (A) PDLSCs were transfected with lentivirus and observed under a microscope (scale bar: 200 μm). (B) The expression of PIP mRNA in shNC and shPIP group. (C) The expression of PIP protein in shNC and shPIP group. (D) The quantitative analysis of western blot. * P<0.05. Figure 4. Knocking down PIP had no influence on PDLSCs proliferation. (A) The CCK-8 analysis of shNC and shPIP PDLSCs during the 7 days of cell proliferation. (B) The cell cycle analysis of shNC and shPIP PDLSCs. (C) The expression level of cell cycle relative proteins of shNC and shPIP PDLSCs.
Figure 4. Knocking down PIP had no influence on PDLSCs proliferation. (A) The CCK-8 analysis of shNC and shPIP PDLSCs during the 7 days of cell proliferation. (B) The cell cycle analysis of shNC and shPIP PDLSCs. (C) The expression level of cell cycle relative proteins of shNC and shPIP PDLSCs. Figure 5. PIP expression level did not affect PDLSCs apoptosis. (A) Cell apoptosis assay of shNC and shPIP PDLSCs after 250 μM H2O2 incubation for 4 h. (B) The protein level of BAX, Bcl-2, c-IAP1, and c-IAP2 in shNC and shPIP PDLSCs.
Figure 5. PIP expression level did not affect PDLSCs apoptosis. (A) Cell apoptosis assay of shNC and shPIP PDLSCs after 250 μM H2O2 incubation for 4 h. (B) The protein level of BAX, Bcl-2, c-IAP1, and c-IAP2 in shNC and shPIP PDLSCs. Figure 6. Downregulation of PIP enhanced PDLSCs osteogenic differentiation. (A) ALP staining of PDLSCs after osteogenic induction for 14 days. (B) ALP activity assay of PDLSCs after osteogenic induction for 14 days. (C) Alizarin Red staining of PDLSCs after osteogenic induction for 21 days. (D) The protein level of ALP and COL1 in shNC and shPIP PDLSCs after 14 days of induction. (E) The quantitative analysis of western blot. * P<0.05.
Figure 6. Downregulation of PIP enhanced PDLSCs osteogenic differentiation. (A) ALP staining of PDLSCs after osteogenic induction for 14 days. (B) ALP activity assay of PDLSCs after osteogenic induction for 14 days. (C) Alizarin Red staining of PDLSCs after osteogenic induction for 21 days. (D) The protein level of ALP and COL1 in shNC and shPIP PDLSCs after 14 days of induction. (E) The quantitative analysis of western blot. * P<0.05. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








