04 June 2021: Animal Study
Effects of β-Asarone on Ischemic Stroke in Middle Cerebral Artery Occlusion Rats by an Nrf2-Antioxidant Response Elements (ARE) Pathway-Dependent Mechanism
Huiying Pan1BC, Yi Xu2D, Qian Cai1E, Meiling Wu1ABD, Mingxing Ding1AG*DOI: 10.12659/MSM.931884
Med Sci Monit 2021; 27:e931884
Abstract
BACKGROUND: This study assessed the effects and underlying molecular mechanisms of β-asarone on ischemic stroke model rats.
MATERIAL AND METHODS: Ischemic stroke was induced by middle cerebral artery occlusion (MCAO) in rats. Before and after modeling, cognitive function was evaluated via fear conditioning test and neurological deficit was determined via Longa and Bederson scores. Following treatment with β-asarone or nuclear factor erythroid 2-related factor 2 (Nrf2) inhibitor for 20 consecutive days, the cerebral infarction was detected via TTC staining and Cresyl Violet staining in brain tissues. TUNEL staining and western blot analysis for apoptosis-related proteins were performed to assess the apoptosis of neurons. Nrf2-antioxidant response elements (ARE) pathway-related proteins were examined by RT-qPCR or western blot.
RESULTS: The cognitive and neurological function was defective in MCAO rats. The infarction volumes and the apoptosis of cortical neurons were significantly increased in brain tissues of model rats, which were ameliorated after treatment with β-asarone. Meanwhile, the increase in pro-apoptotic proteins and decrease in anti-apoptotic proteins were found in brain tissues of model rats, which were markedly ameliorated by β-asarone treatment. However, Nrf2 inhibitor worsened the cerebral infarction and the apoptosis of neurons. Western blot results showed that β-asarone treatment activated the Nrf2-ARE pathway-related proteins in model rats, which was inhibited by Nrf2 inhibitor.
CONCLUSIONS: Our findings suggest that β-asarone treatment ameliorated the cerebral infarction in MCAO rats, which could be related to activation of the Nrf2-ARE pathway.
Keywords: Antioxidant Response Elements, Infarction, Middle Cerebral Artery, Stroke, Allylbenzene Derivatives, Anisoles, Apoptosis Regulatory Proteins, Asarum, Behavior, Animal, Fibrinolytic Agents, ischemic stroke, NF-E2-Related Factor 2, Neuroprotective Agents
Background
Stroke is a life-threatening cerebrovascular thrombotic disease globally. Ischemic stroke is mainly characterized by insufficient blood flow to the brain; ≤4.5 h for thrombolytic and ≤6 h for thrombectomy is the main therapeutic strategy for ischemic stroke [1]. The endovascular treatment time can vary from 6 to 12 h, depending on the artery and the patient. Due to the narrow therapeutic time window and many complicated factors, most patients cannot get timely treatment, especially in underdeveloped countries. There is still a lack of effective drugs for treating ischemic stroke patients. Recombinant tissue plasminogen activator targeting the occlusion to induce reperfusion has gained FDA approval as the only drug for treating ischemic stroke [2]. Nevertheless, oxidative damage and neuronal apoptosis after ischemia contribute to neurological function deficits [2]. Despite much progress in recent years, effective treatment strategies are still lacking for targeting neurological deficits induced by ischemic stroke. Hence, it is clinically important to discover novel drugs and targets for ischemic stroke.
Oxidative stress plays a critical role in the pathophysiology of ischemic stroke [3]. The Nrf2-ARE pathway may mediate the transcription of many antioxidant genes, thereby preserving cellular homeostasis [4]. Nrf2, as a transcription factor, is made up of 7 functional domains that are named Neh1-7 [5]. It is involved in mediating cellular defense mechanisms against endogenous and exogenous stresses [6]. Increasing evidence suggests that Nrf2 activation and its downstream genes are a promising therapeutic strategy for preventing and treating ischemic stroke [7]. For example, activation of Nrf2 alleviates neuroinflammation [8], cerebral ischemia/reperfusion damage [9], and neurological deficits [10] for ischemic stroke rats. Thus, Nrf2 is an available therapeutic target for ischemic stroke.
β-asarone (1,2,4-trimethoxy-5-[(Z)-prop-1-enyl]benzene) is primarily extracted from
Material and Methods
ANIMALS:
Twenty healthy male Sprague-Dawley rats aged 6–7 weeks and weighing 250–280 g were purchased from Hangzhou Scientific Cloud Biotechnology Co., Ltd. (China). All rats were adaptively fed for 1 week. Then, these animals were randomly separated into 4 groups (n=5 each group): control, ischemic stroke model, model+β-asarone, and model+β-asarone+Nrf2 inhibitor groups. These animals were housed at 21±2°C, 30–70% relative humidity, and 12/12-h light cycle. This study was approved by the Ethics Committee of the Medical College, Jinhua Polytechnic (2019011), in line with the guidance of the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
MCAO MODEL:
The MCAO models were established according to the following steps: rats were intraperitoneally injected with 3% sodium pentobarbital (30 mg/kg). The center of the neck was incised for exposure of the right common, internal, and external carotid arteries. We used 6-0 silk thread to ligate the distal end of the external carotid artery 4 mm from the bifurcation of the common carotid artery. Another 6-0 silk thread was threaded into the external carotid artery. The common carotid artery was clamped by the arterial clamp. A small opening was cut on the external carotid artery 3 mm from the bifurcation of the common carotid artery, and a 0.3-mm-diameter nylon thread through the small opening was inserted into the internal carotid artery and the middle cerebral artery inward. The insertion depth of the nylon thread was about 16±1 mm from the bifurcation of the common carotid artery. After 90 min of ischemia, the thread plug was pulled out. We used a 6-0 silk thread to ligate the proximal end of the external artery, and the neck wound was sutured with 3-0 silk thread. For the control (sham-operation) rats, the common carotid artery and the right external carotid artery were occluded. The wound was by disinfected by iodine, and the rats were put on a heating pad in a constant-temperature nursing box after waking up. After 24 h, the rats were scored for neurological function. The rats in the β-asarone group were administered with β-asarone (10 ml/kg) once a day for 20 consecutive days. The model group and the normal control group were given the same amount of saline intragastrically. After being intraperitoneally injected with 3% sodium pentobarbital, the brain tissues were taken for subsequence experiments.
NEUROLOGICAL DEFICIT ASSESSMENT:
Neurological deficit was assessed in line with Longa score and Bederson score [17]. The rats were scored before the operation and 24 h after the rats awoke from anesthesia. The higher the score, the more serious the animal behavior disorder. The scoring criteria were 0 points: without neurological damage symptom; 1 point: inability to completely extend the contralateral front paw; 2 points: turning to the opposite side in a circle; 3 points: slumping to the opposite side; 4 points: inability to walk spontaneously; 5 points: without consciousness.
FEAR CONDITIONING TEST:
The fear conditioning test was used for evaluation of cognitive function. The rats were placed in a fear box 1 day in advance to adapt autonomously for 3 min. Then, the rats were given 3 consecutive single-tone stimulation (2000 Hz, 90 dB, 30 s)+electrical stimulation (1 mA, 2 s), that is, a single tone of 90 dB lasted for 30 s, and the last 2 s were given 1 mA electrical stimulation at the same time, with a 60-s interval between each stimulation. At 24 h before modeling and after drug treatment 1 day after modeling, the rats were placed in the previous cages for 3 min, during which no stimulation was given. The time that the rat stayed still during this period was recorded as an index to measure the scene fear memory of the rats.
2,3,5-TRIPHENYLTETRAZOLIUM CHLORIDE (TTC) STAINING:
We prepared 1% TTC (T8877-25G; Sigma, USA; W/V) with PBS, and dissolved the mixture at 37°C. The brain tissue section was incubated with 10 ml TTC solution at 37°C for 10 min to stain the tissue evenly. The normal brain tissue was stained bright red while the infarct area was stained pale white. The whole-infarct volumes and whole-brain volumes were determined via Image-Pro Plus 6.0 analysis software. The infarct volumes were calculated according to the ratio of the whole-infarct volumes to the whole-brain volumes.
CRESYL VIOLET STAINING:
Paraffin sections were treated by a series of xylene (10023418; Macklin, Wuhan, China). Afterwards, the sections were stained by Toluidine Blue Dye solution (G1032; Servicebio, Wuhan) lasting 5 min. After washing, the sections were differentiated by 1% glacial acetic acid (G10000218; Servicebio), and the reaction was terminated by washing with tap water. The degree of differentiation was controlled under a microscope (BX53; Olympus, Japan). After washing with tap water, the slices were put into the oven to dry. The slices were transparentized with clean xylene for 5 min, and covered with neutral gum, followed by microscope inspection, image acquisition, and analysis. The Nissl body of the brain tissue was dark blue around a light blue background.
TERMINAL-DEOXYNUCLEOTIDYL TRANSFERASE MEDIATED NICK-END LABELING (TUNEL) STAINING:
The paraffin tissue sections were deparaffinized in xylene for 10–20 min at room temperature. Then, the sections were soaked in gradient ethanol (100%, 100%, 95%, 80%, 70%) for 5 min at room temperature and were permeated by the Proteinase K solution with a final concentration of 0.02 μg/μl for 15–20 min. TUNEL test solution (ATK00001; Atagenix; Wuhan, China) was used for incubation of the sections for 60 min in the dark. We added 0.05 μg/μl 4′, 6-diamidino-2-phenylindole (DAPI) solution to the sections, followed by incubation for 10 min in a humid chamber at room temperature in the dark. The slices were immersed in PBS solution at room temperature 3 times. The slides were mounted with anti-fluorescence quenching mounting tablet. The results were observed and pictures were taken under a fluorescence microscope.
REAL-TIME QUANTITATIVE POLYMERASE CHAIN REACTION (RT-QPCR):
Total RNA was extracted from brain tissues via TRIzol reagent (Takara, Japan), which was then reverse-transcribed into cDNA according to the procedures as follows: 25°C for 5 min, 42°C for 30 min, and 85°C for 5 min. The reverse transcription reaction system included 10 μL RNase free ddH2O, 2 μL 5×gDNA digester buffer, 1 μL gDNA digester, 1 μg total RNA, and 10 μL 2×Hifair® II SuperMix plus. In the reaction system of 1 μL primer (forward), 1 μL primer (reverse), 10 μL 2×mix, 7 μL H2O, and 1 μL cDNA, RT-qPCR was presented to detect the mRNA expression levels of target genes on the 12K fluorescenceRT-qPCR instrument (ABI, USA). The primer sequences are listed in Table 1. GAPDH served as a reference control. Data were calculated with the 2−ΔΔCt method.
WESTERN BLOT:
The tissue samples were lysed on ice for 30 min with RIPA lysis solution (P0013B; Beyotime, Shanghai, China), ultrasonicated in the ice bath for 3 min, and centrifuged at 4°C at 12 000 rpm for 10 min. Then, the supernatant was transferred to a new EP tube. The protein concentration was assessed via BCA method (P0009; Beyotime). We added 5×SDS loading buffer according to the volume of the lysate, and boiled it at 100°C for 5 min. The glass plate (1.0 mm or 1.5 mm) filled with polyacrylamide gel was clamped, and the currently configured SDS polyacrylamide gel (8%, 10% or 12%) was quickly added into the gap of the glass plate, and isopropanol was used to seal the glue. After the separation gel was solidified, the isopropanol was poured out and the remaining liquid was removed with filter paper. We added 5% of the laminated glue to the separating glue and the comb was inserted immediately. After the glue was completely solidified, the comb was pulled out and the sample hole was rinsed with water. After fixing the electrophoresis device, Tris-glycine electrophoresis buffer and pre-stained markers (#26616; Thermo scientific) were added as required. The electrophoresis device was connected to the power supply, and the voltage was adjusted to 80 V. When the bromophenol blue reached the separation gel, the voltage was adjusted to 120 V until the bromophenol blue reached the bottom of the separation gel. After removing the gel, samples were moved to PVDF membranes and were sealed with a 5% milk/TBST at room temperature for 1 h. After rinsing with TBST for 2 min, the membranes were incubated with primary antibodies against NQO1 (11451-1-AP; Proteintech, Wuhan; 1: 1000), Nrf2 (16396-1-AP; Proteintech; 1: 1000), BAX (60267-1-Ig; Proteintech; 1: 3000), BCL2 (12789-1-AP; Proteintech; 1: 1000), CASP3 (19677-1-AP; Proteintech; 1: 1000), CASP9 (66169-1-Ig; Proteintech; 1: 1000), HO-1 (10701-1-AP; Proteintech; 1: 1000), GCLM (14241-1-AP; Proteintech; 1: 1000), GCLC (12601-1-AP; Proteintech; 1: 2000), and GAPDH (ATA00013Rb; Atagenix; Wuhan; 1: 5000). The primary antibody was diluted with 1% BSA/PBST according to the recommended dilution ratio. The membrane was sealed with a hybridization bag, and refrigerated overnight at 4°C. Following washing with PBST 3 times, PVDF membrane was put into the horseradish peroxidase-labeled secondary antibody (SA00001-2; Proteintech; 1: 5000) diluted with 5% milk/PBST, and incubated at room temperature for 1 h. The same amounts of Enhanced Luminol Reagent and Oxidizing Reagent were mixed and diluted with an appropriate amount of ddH2O, which was then added dropwise to the sealing film. The front side of the PVDF membrane was exposed to the luminescent reagent, and the color was developed for 2 min. The images were observed with a gel imaging system.
STATISTICAL ANALYSIS:
Statistical analysis was performed via SPSS 21.0 software (SPSS, Inc., Chicago, IL, USA). Data are presented as the mean ± standard deviation. One-way ANOVA followed by Tukey’s post hoc test was performed for comparisons between groups. Contextual fear conditioning test scores were compared before and after the intervention in each group by two-way ANOVA followed by Sidak’s multiple comparisons test. Differences with P<0.05 were significant.
Results
β-ASARONE AMELIORATES ISCHEMIC BRAIN INJURY IN MCAO RAT MODELS:
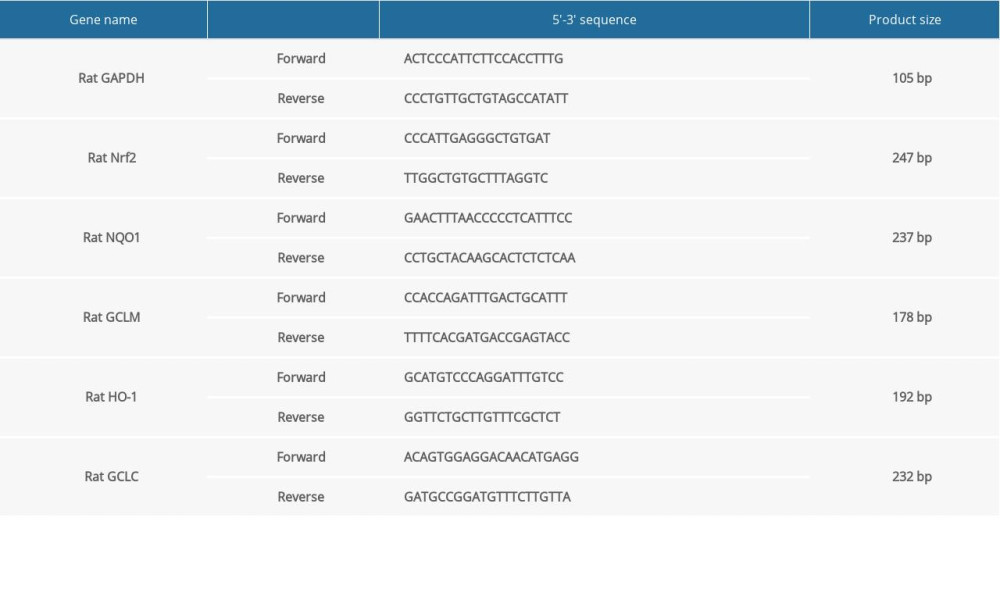
To observe whether β-asarone could improve the ischemic brain injury, we first established the MCAO rat models. We compared the cognitive function of rats 24 h before modeling and 24 h after modeling via contextual fear conditioning test. Our data showed that in the control group, the rigid time was distinctly prolonged after modeling compared to before modeling (p<0.05; Figure 1A). For the model rats, the rigid time was significantly shorter 24 h after modeling than before modeling (p<0.01), suggesting the defective cognitive function of the rat models. No significant differences were found between 24 h after modeling and before modeling for model rats treated with β-asarone and/or Nrf2 inhibitor, indicating that β-asarone could protect model rats from the cognitive function defect. Furthermore, we assessed the neurological function deficits of rats for each group. Before modeling, the neurological function scores were all zero for each group, suggesting good neurological function. MCAO rat models were then constructed with neurological function scores of 4, which were randomly divided into a model group, a model+β-asarone group, and a model+β-asarone+Nrf2 inhibitor group. Following 24 h of modeling, the neurological function scores were still zero for control rats. However, the neurological function scores of MCAO rats were 4, suggesting that the model rats had severe neurological function deficits. However, for the model rats treated with β-asarone and/or Nrf2 inhibitor, the neurological function scores were 1 and 2, respectively. After treatment with β-asarone and/or Nrf2 inhibitor for 20 days, the neurological function scores were 0 for MCAO rats, but the neurological function scores were 2 in the model group. These data suggested that β-asarone treatment could improve the neurological function of the model rats. Following treatment with β-asarone for consecutive 20 days, the infarct volumes were determined in brain tissues via TTC staining. The data showed that the infarct volumes were distinctly higher in the model rats than in controls (p<0.0001; Figure 1B, 1C). β-asarone treatment significantly lowered the infarct volumes of MCAO rats (p<0.0001). However, co-treatment with Nrf2 inhibitor significantly deteriorated the infarct volumes for the models compared to the model+β-asarone group (p<0.0001). Thus, the ischemic brain injury was ameliorated by β-asarone treatment, which was deteriorated following treatment with Nrf2 inhibitor.
β-ASARONE IMPROVES CEREBRAL INFARCT IN MCAO RAT MODELS:
To investigate the effects of β-asarone on the cerebral infarct for in MCAO rat models in detail, we performed Cresyl Violet staining in brain tissue sections. Compared to the control group, we found that cytoplasmic Nissl body disappeared and nucleus was pyknotic in brain tissues of MCAO rats. After treatment with β-asarone, there were Nissl body in brain tissues. Our data showed that β-asarone distinctly ameliorated the cerebral infarct in MCAO rats (Figure 2). Nevertheless, Nrf2 inhibitor weakened the therapeutic effects of β-asarone on cerebral infarct.
β-ASARONE SUPPRESSES NEURONAL APOPTOSIS IN MCAO RAT MODELS:
The neuronal apoptosis in rats was detected via TUNEL staining. Compared to the control group, apoptosis of cortical neurons was enhanced for the model group (Figure 3). Following treatment with β-asarone for 20 days, the apoptosis of cortical neurons was significantly ameliorated in brain tissues of the model rats. However, Nrf2 inhibitor weakened the therapeutic effects of β-asarone the protection of cortical neurons. Hence, our data suggest that β-asarone suppressed apoptosis of cortical neurons in MCAO model rats.
β-ASARONE TREATMENT ACTIVATES THE NRF2-ARE PATHWAY IN BRAIN TISSUES OF MCAO MODEL RATS:
During ischemic stroke, oxidative stress produces and aggravates brain damage [18]. Targeting Nrf2-ARE pathway activation could alleviate oxidative stress damage in ischemic stroke [19–21]. As a neuroprotective pathway, the Nrf2-ARE pathway regulates complex neuroprotection against stroke, which has become a promising therapeutic strategy in ischemic stroke [22]. Therefore, we further detected whether β-asarone treatment could affect the activation of the Nrf2-ARE pathway in brain tissues of MCAO model rats. Our RT-qPCR results showed Nrf2 (p<0.001; Figure 4A), NQO1 (p<0.01; Figure 4B), GCLM (p<0.01; Figure 4C), HO-1 (p<0.05; Figure 4D), and GCLC (p<0.001; Figure 4E) expression were distinctly higher in the model group than in controls. After treatment with β-asarone for 20 days, the expression of Nrf2 (p<0.001), NQO1 (p<0.01), GCLM (p<0.05), HO-1 (p<0.05), and GCLC (p<0.001) was significantly higher than in the model group. There was no significant difference in Nrf2, NQO1, GCLM, and HO-1 in the model rats treated with β-asarone and/or Nrf2 inhibitor. However, compared to the model rats treated with β-asarone, GCLC expression was markedly lowered after co-treatment with β-asarone and Nrf2 inhibitor (p<0.001). Furthermore, the expression of these proteins was examined by western blot (Figure 5A). Consistent with the RT-qPCR results, Nrf2 (p<0.001; Figure 5B), NQO1 (p<0.001; Figure 5C), GCLM (p<0.0001; Figure 5D), HO-1 (p<0.0001; Figure 5E), and GCLC (p<0.0001; Figure 5F) expression was distinctly elevated in brain tissues from the model group than in the control group. The treatment with β-asarone for 20 days significantly increased the expression of Nrf2 (p<0.001), NQO1 (p<0.001), GCLM (p<0.0001), HO-1 (p<0.0001), and GCLC (p<0.0001) in brain tissues of the model rats. Collectively, β-asarone treatment could enhance the Nrf2-ARE pathway activation in brain tissues of MCAO model rats. However, we found that Nrf2 inhibitor significantly weakened the effects of β-asarone treatment on Nrf2 (p<0.001), NQO1 (p<0.001), GCLM (p<0.0001), HO-1 (p<0.0001), and GCLC (p<0.0001) in brain tissues of the model rats. Hence, β-asarone treatment may elevate the Nrf2-ARE pathway activation in brain specimens from MCAO model rats.
β-ASARONE TREATMENT SUPPRESSED PRO-APOPTOTIC PROTEIN EXPRESSION AND INCREASES ANTI-APOPTOTIC PROTEIN EXPRESSION IN BRAIN TISSUES OF MCAO RAT MODELS:
Our further study assessed whether β-asarone could attenuate neuronal apoptosis in brain tissues from MCAO rats. Western blot was utilized for assessment of the expression of pro- (Bax, CASP3 and CASP9) and anti-apoptotic (Bcl2) markers in brain tissues of MCAO model rats. Our results showed that compared to the control group, the expression of pro-apoptotic proteins including BAX (p<0.001; Figure 5G), CASP3 (p<0.001; Figure 5H), and CASP9 (p<0.001; Figure 5I) was significantly elevated in brain tissues of the model group. On the contrary, β-asarone treatment markedly lowered the expression of BAX (p<0.001), CASP3 (p<0.001), and CASP9 (p<0.001) in brain tissues of the model group. The inhibitory functions of β-asarone on BAX (p<0.001), CASP3 (p<0.001) and CASP9 (p<0.001) expression were weakened by Nrf2 inhibitor in brain tissues of MCAO rats. We also found that anti-apoptotic protein Bcl2 had a significantly lower expression in brain specimens from the model rats than controls (p<0.001; Figure 5J). Its high expression in the model rats was markedly elevated after treatment with β-asarone (p<0.001). However, Nrf2 inhibitor weakened the effects of β-asarone treatment on Bcl2 expression in brain tissues from MCAO rats.
Discussion
In this study, our results demonstrated that β-asarone improved the cognitive functions and cerebral infarct and suppressed apoptosis of neurons in MCAO model rats, which was closely linked with activation of the Nrf2-ARE pathway. β-asarone exerts an excellent therapeutic effect on ischemic stroke.
β-asarone has been found to readily pass through the blood-brain barrier [23]. Herein, this study suggested that β-asarone treatment significantly improved the cognitive functions for MCAO rats. Consistently, a previous study has reported that β-asarone could significantly improve the neurological outcome after cerebral ischemia and reperfusion in rats; however, the mechanism of β-asarone in protection of cerebral ischemia was not further studied [24]. It has been reported that β-asarone can improve social interaction and cognitive function deficits for MK-801-induced mice [16]. Moreover, β-asarone ameliorates cognitive functions via suppression of neuronal apoptosis for Alzheimer disease rats [25]. A previous study has found that β-asarone can prevent autophagy as well as synaptic loss via decreasing ROCK expression for Alzheimer disease mice [16]. Additionally, β-asarone improves spatial working memory in rats with long-term exposure to corticosterone [26]. Combining these findings, β-asarone could be used for improvement of the cognitive effects of ischemic stroke. Following administration of β-asarone for 20 consecutive days, TTC staining demonstrated that β-asarone distinctly decreased the infarct volume in MCAO model rats. Furthermore, our Cresyl Violet staining demonstrated that β-asarone improved cerebral infarct in brain tissues of MCAO rats. A previous study found that β-asarone ameliorated neuronal damage through weakening Beclin-1-dependent autophagy in an in vitro ischemic stroke model [27]. Hence, β-asarone could be a promising drug for treatment of cerebral infarct. However, further studies on this are needed.
It has been confirmed that β-asarone exerts distinct pharmacological effects on central system diseases [28]. Herein, we found that β-asarone attenuated neuronal apoptosis in brain tissues from MCAO rats. It significantly lowered the expression of pro-apoptotic proteins including Bax, CASP3, and CASP9, and elevated the expression of anti-apoptotic protein Bcl2 in brain tissues of MCAO rats. Consistently, β-asarone treatment suppresses neuronal apoptosis through CaMKII/CREB/Bcl-2 pathway in Alzheimer disease models [29]. Moreover, it can induce apoptosis of glioma cells [30]. Thus, β-asarone could attenuate neuronal apoptosis in ischemic stroke.
Our findings suggest that β-asarone can activate the expression of Nrf2 and its downstream genes in brain tissues of MCAO rats. Following co-treatment with Nrf2 inhibitor, β-asarone treatment had therapeutic effects on cognitive function, infarct volume, and neuronal apoptosis in MCAO rats. The activation of Nrf2 can reduce brain inflammatory responses following ischemic stroke via enhancement of microglia M2 polarization [31]. Furthermore, the Nrf2/ARE pathway alleviated nerve cell apoptosis and improved cognition function in ischemic stroke rat models [32]. These findings indicate that β-asarone exerts a therapeutic effect on ischemic stroke via activation of the Nrf2-ARE pathway. The present study confirmed the therapeutic efficacy of β-asarone on ischemic brain injury via an Nrf2-dependent mechanism. Our findings propose an innovative potential drug for treating ischemic stroke.
Conclusions
This study demonstrated that β-asarone could alleviate cognitive functions, infarct volume, and neuronal apoptosis in MCAO rats, which was closely related to the Nrf2-ARE signaling pathway. Taken together, these results show that β-asarone could be a novel drug for treating ischemic stroke via an Nrf2-dependent mechanism.
Figures
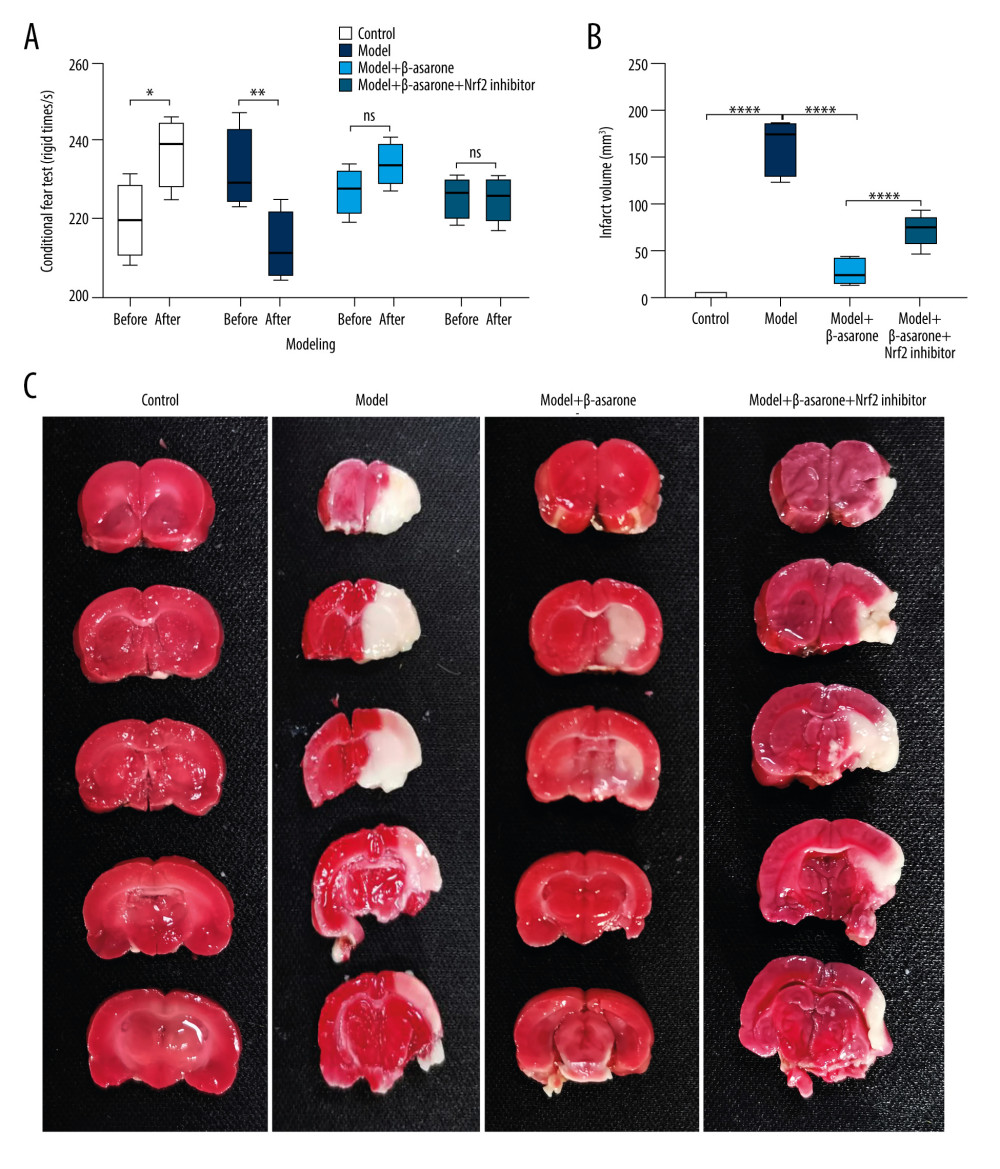 Figure 1. β-asarone ameliorates ischemic brain injury in MCAO rat models. (A) Contextual fear conditioning test for rats 24 h before modeling and 24 h after modeling. (B, C) TTC staining for assessment of the infarct volumes in brain tissues for control, model, and model rats treated with β-asarone and/or Nrf2 inhibitor for consecutive 20 days. Ns – not significant; * p<0.05, ** p<0.01, **** p<0.0001.
Figure 1. β-asarone ameliorates ischemic brain injury in MCAO rat models. (A) Contextual fear conditioning test for rats 24 h before modeling and 24 h after modeling. (B, C) TTC staining for assessment of the infarct volumes in brain tissues for control, model, and model rats treated with β-asarone and/or Nrf2 inhibitor for consecutive 20 days. Ns – not significant; * p<0.05, ** p<0.01, **** p<0.0001. 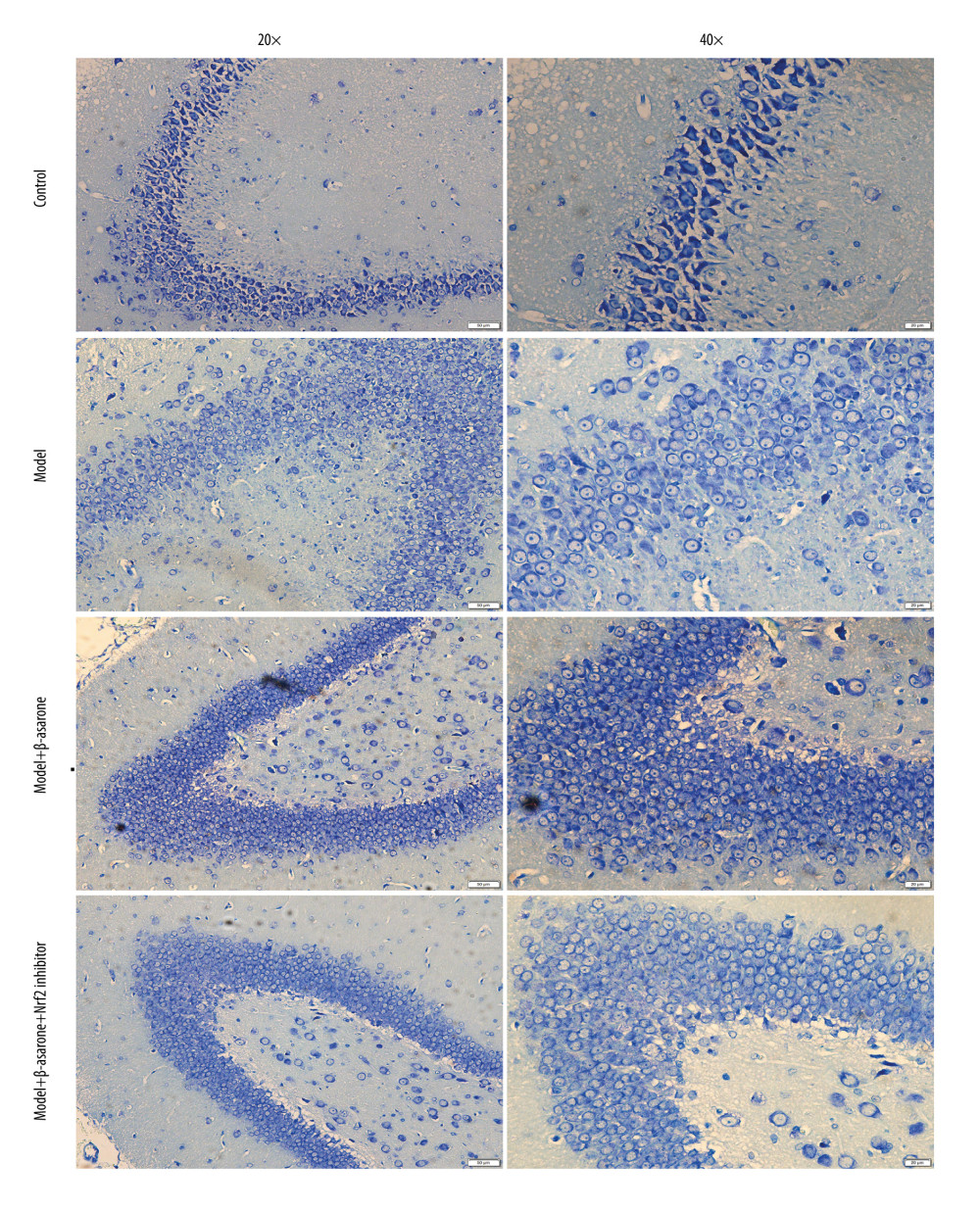 Figure 2. Cresyl Violet staining for the pathologic changes in Nissl bodies brain tissues of control, model, model treated with β-asarone and/or Nrf2 inhibitor groups. Bar=50 or 20 μm. Magnification: 20×; 40×.
Figure 2. Cresyl Violet staining for the pathologic changes in Nissl bodies brain tissues of control, model, model treated with β-asarone and/or Nrf2 inhibitor groups. Bar=50 or 20 μm. Magnification: 20×; 40×. 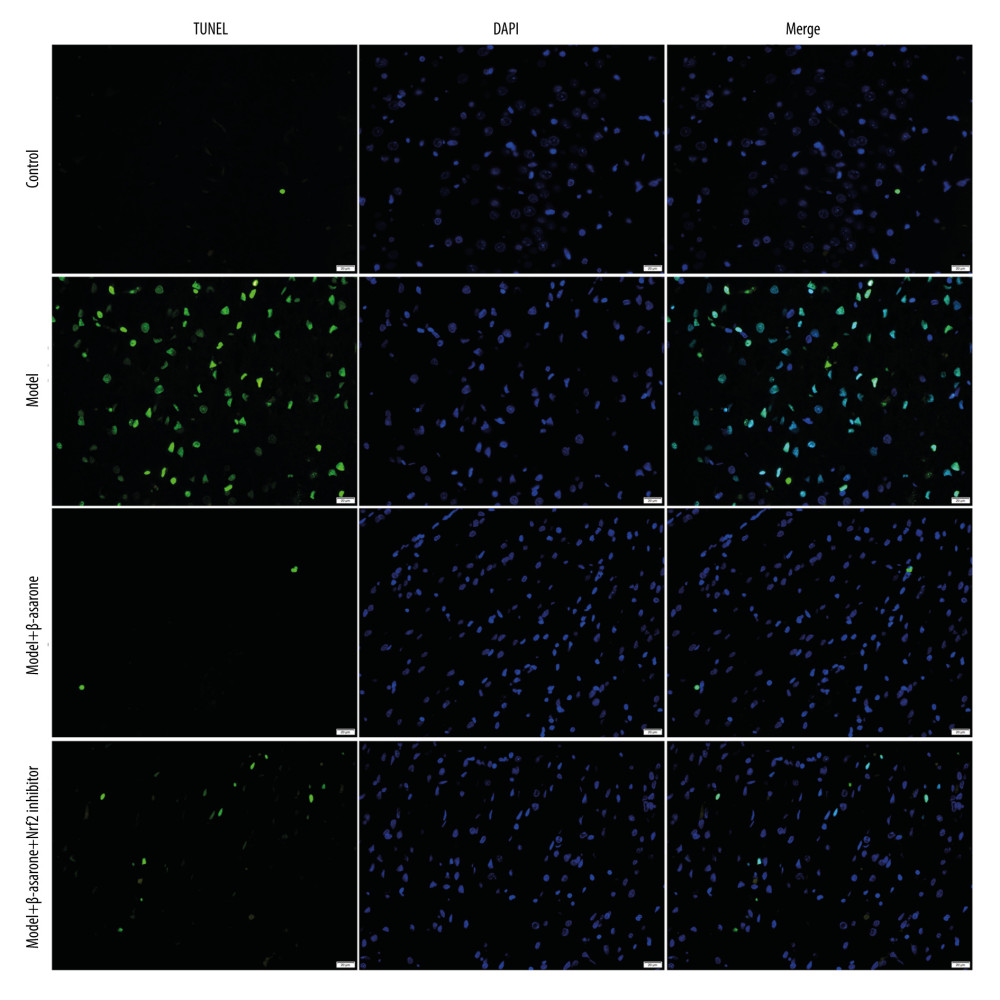 Figure 3. TUNEL staining for the apoptosis of neurons in brain tissues from control, model, model treated with β-asarone, and/or Nrf2 inhibitor. Bar=20 μm.
Figure 3. TUNEL staining for the apoptosis of neurons in brain tissues from control, model, model treated with β-asarone, and/or Nrf2 inhibitor. Bar=20 μm. 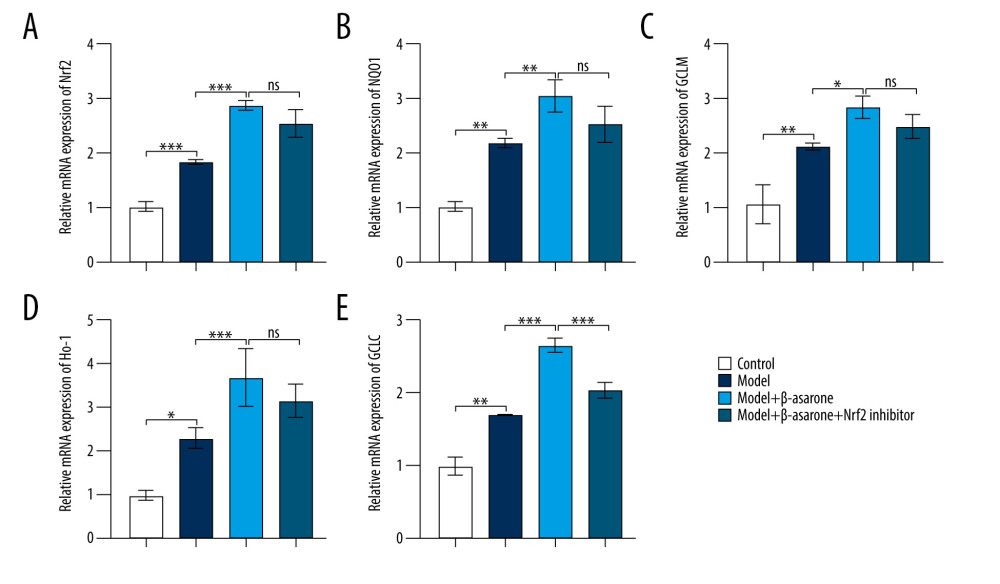 Figure 4. RT-qPCR assay for detection of the mRNA expression of Nrf2-ARE pathway-related proteins in brain tissues of MCAO rat models. (A) Nrf2; (B) NQO1; (C) GCLM; (D) HO-1 and (E) GCLC. Ns – not significant; * p<0.05, ** p<0.01, *** p<0.001.
Figure 4. RT-qPCR assay for detection of the mRNA expression of Nrf2-ARE pathway-related proteins in brain tissues of MCAO rat models. (A) Nrf2; (B) NQO1; (C) GCLM; (D) HO-1 and (E) GCLC. Ns – not significant; * p<0.05, ** p<0.01, *** p<0.001. 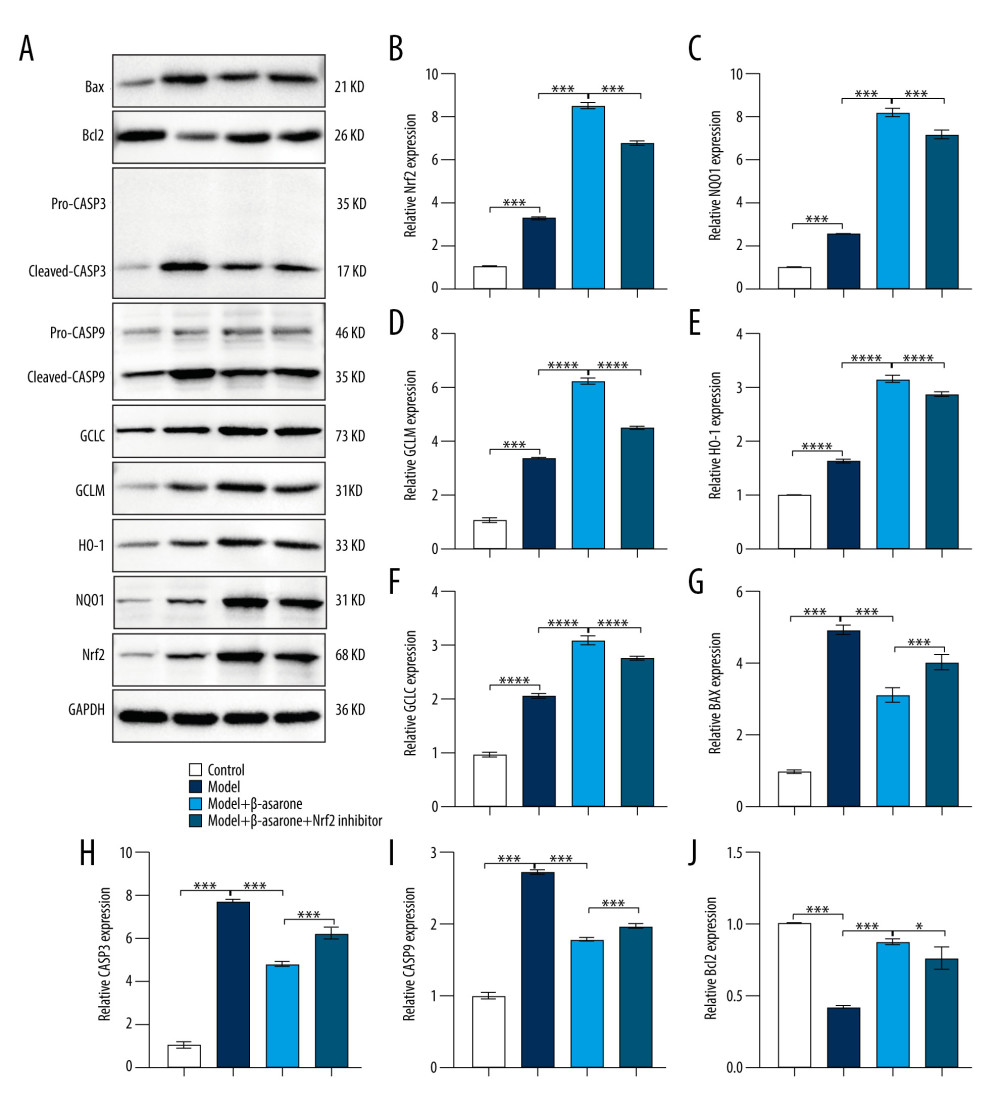 Figure 5. Western blot for detecting Nrf2-ARE pathway- and apoptosis-related protein expression in brain tissues of MCAO rat models. (A) Representative images for western blot results. The expression of Nrf2-ARE pathway-related proteins including (B) Nrf2 (C) NQO1, (D) GCLM, (E) HO-1, (F) GCLC and apoptosis-related proteins including (G) BAX, (H) CASP3, (I) CASP9, and (J) Bcl-2 was quantified according to the western blots. * p<0.01, *** p<0.001, ****p<0.0001.
Figure 5. Western blot for detecting Nrf2-ARE pathway- and apoptosis-related protein expression in brain tissues of MCAO rat models. (A) Representative images for western blot results. The expression of Nrf2-ARE pathway-related proteins including (B) Nrf2 (C) NQO1, (D) GCLM, (E) HO-1, (F) GCLC and apoptosis-related proteins including (G) BAX, (H) CASP3, (I) CASP9, and (J) Bcl-2 was quantified according to the western blots. * p<0.01, *** p<0.001, ****p<0.0001. References
1. Kang R, Gamdzyk M, Tang H, Delayed recanalization-how late is not too late?: Transl Stroke Res, 2021; 12(3); 382-93
2. Cui HY, Zhang XJ, Yang Y, Rosmarinic acid elicits neuroprotection in ischemic stroke via Nrf2 and heme oxygenase 1 signaling: Neural Regen Res, 2018; 13; 2119-28
3. Tian Y, Su Y, Ye Q, Silencing of TXNIP alleviated oxidative stress injury by regulating MAPK-Nrf2 axis in ischemic stroke: Neurochem Res, 2020; 45; 428-36
4. Tu W, Wang H, Li S, The anti-inflammatory and anti-oxidant mechanisms of the Keap1/Nrf2/ARE signaling pathway in chronic diseases: Aging Dis, 2019; 10; 637-51
5. Liu L, Vollmer MK, Ahmad AS, Pretreatment with Korean red ginseng or dimethyl fumarate attenuates reactive gliosis and confers sustained neuroprotection against cerebral hypoxic-ischemic damage by an Nrf2-dependent mechanism: Free Radic Biol Med, 2019; 131; 98-114
6. Liu L, Locascio LM, Doré S, Critical role of Nrf2 in experimental ischemic stroke: Front Pharmacol, 2019; 10; 153
7. Yang T, Sun Y, Li Q, Ischemic preconditioning provides long-lasting neuroprotection against ischemic stroke: The role of Nrf2: Exp Neurol, 2020; 325; 113142
8. Liao S, Wu J, Liu R, A novel compound DBZ ameliorates neuroinflammation in LPS-stimulated microglia and ischemic stroke rats: Role of Akt(Ser473)/GSK3β(Ser9)-mediated Nrf2 activation: Redox Biol, 2020; 36; 101644
9. Guo H, Adah D, James PB, Xueshuantong injection (lyophilized) attenuates cerebral ischemia/reperfusion injury by the activation of Nrf2-VEGF pathway: Neurochem Res, 2018; 43; 1096-103
10. Shi YS, Zhang Y, Liu B, Nomilin protects against cerebral ischemia-reperfusion induced neurological deficits and blood-brain barrier disruption via the Nrf2 pathway: Food Funct, 2019; 10; 5323-32
11. Chellian R, Pandy V, Mohamed Z, Pharmacology and toxicology of α- and β-asarone: A review of preclinical evidence: Phytomedicine, 2017; 32; 41-58
12. Li L, Wu M, Wang C, β-Asarone inhibits invasion and EMT in human glioma U251 cells by suppressing splicing factor HnRNP A2/B1: Molecules, 2018; 23; 671
13. Xiao B, Huang X, Wang Q, Wu Y, beta-Asarone alleviates myocardial ischemia-reperfusion injury by inhibiting inflammatory response and NLRP3 inflammasome mediated pyroptosis: Biol Pharm Bull, 2020; 43; 1046-51
14. Liu SJ, Yang C, Zhang Y, Neuroprotective effect of β-asarone against Alzheimer’s disease: Regulation of synaptic plasticity by increased expression of SYP and GluR1: Drug Des Devel Ther, 2016; 10; 1461-69
15. Xue Z, Guo Y, Zhang S, Beta-asarone attenuates amyloid beta-induced autophagy via Akt/mTOR pathway in PC12 cells: Eur J Pharmacol, 2014; 741; 195-204
16. Xiao X, Xu X, Li F, Xie G, Zhang T, Anti-inflammatory treatment with β-asarone improves impairments in social interaction and cognition in MK-801 treated mice: Brain Res Bull, 2019; 150; 150-59
17. Longa EZ, Weinstein PR, Carlson S, Cummins R, Reversible middle cerebral artery occlusion without craniectomy in rats: Stroke, 1989; 20; 84-91
18. Kuo PC, Yu IC, Scofield BA, 3H-1,2-Dithiole-3-thione as a novel therapeutic agent for the treatment of ischemic stroke through Nrf2 defense pathway: Brain Behav Immun, 2017; 62; 180-92
19. Liu D, Wang H, Zhang Y, Zhang Z, Protective Effects of Chlorogenic Acid on Cerebral Ischemia/Reperfusion Injury Rats by Regulating Oxidative Stress-Related Nrf2 pathway: Drug Des Devel Ther, 2020; 14; 51-60
20. Liu W, Guo Q, Zhao H, Oxidative stress-elicited YY1 potentiates antioxidative response via enhancement of NRF2-driven transcriptional activity: A potential neuronal defensive mechanism against ischemia/reperfusion cerebral injury: Biomed Pharmacother, 2018; 108; 698-706
21. Zhang LL, Zhang ZJ, Sestrin2 aggravates oxidative stress of neurons by decreasing the expression of Nrf2: Eur Rev Med Pharmacol Sci, 2018; 22; 3493-501
22. Jiang S, Deng C, Lv J, Nrf2 weaves an elaborate network of neuroprotection against stroke: Mol Neurobiol, 2017; 54; 1440-55
23. Ning B, Zhang Q, Wang N, β-Asarone regulates ER stress and autophagy via inhibition of the PERK/CHOP/Bcl-2/Beclin-1 pathway in 6-OHDA-induced parkinsonian rats: Neurochem Res, 2019; 44; 1159-66
24. Yang YX, Chen YT, Zhou XJ: BMC Complement Altern Med, 2013; 13; 236
25. Geng Y, Li C, Liu J, Beta-asarone improves cognitive function by suppressing neuronal apoptosis in the beta-amyloid hippocampus injection rats: Biol Pharm Bull, 2010; 33; 836-43
26. Lee B, Sur B, Cho SG, Effect of beta-asarone on impairment of spatial working memory and apoptosis in the hippocampus of rats exposed to chronic corticosterone administration: Biomol Ther (Seoul), 2015; 23; 571-81
27. Mo ZT, Fang YQ, He YP, Zhang S, β-Asarone protects PC12 cells against OGD/R-induced injury via attenuating Beclin-1-dependent autophagy: Acta Pharmacol Sin, 2012; 33; 737-42
28. Liu L, Fang YQ, Xue ZF, Beta-asarone attenuates ischemia-reperfusion-induced autophagy in rat brains via modulating JNK, p-JNK, Bcl-2 and Beclin 1: Eur J Pharmacol, 2012; 680; 34-40
29. Wei G, Chen YB, Chen DF, β-Asarone inhibits neuronal apoptosis via the CaMKII/CREB/Bcl-2 signaling pathway in an in vitro model and AβPP/PS1 mice: J Alzheimers Dis, 2013; 33; 863-80
30. Wang N, Han Y, Luo L, β-asarone induces cell apoptosis, inhibits cell proliferation and decreases migration and invasion of glioma cells: Biomed Pharmacother, 2018; 106; 655-64
31. Wang Y, Huang Y, Xu Y, A dual AMPK/Nrf2 activator reduces brain inflammation after stroke by enhancing microglia M2 polarization: Antioxid Redox Signal, 2018; 28; 141-63
32. Jin XF, Wang S, Shen M, Effects of rehabilitation training on apoptosis of nerve cells and the recovery of neural and motor functions in rats with ischemic stroke through the PI3K/Akt and Nrf2/ARE signaling pathways: Brain Res Bull, 2017; 134; 236-45
Figures
 Figure 1. β-asarone ameliorates ischemic brain injury in MCAO rat models. (A) Contextual fear conditioning test for rats 24 h before modeling and 24 h after modeling. (B, C) TTC staining for assessment of the infarct volumes in brain tissues for control, model, and model rats treated with β-asarone and/or Nrf2 inhibitor for consecutive 20 days. Ns – not significant; * p<0.05, ** p<0.01, **** p<0.0001.
Figure 1. β-asarone ameliorates ischemic brain injury in MCAO rat models. (A) Contextual fear conditioning test for rats 24 h before modeling and 24 h after modeling. (B, C) TTC staining for assessment of the infarct volumes in brain tissues for control, model, and model rats treated with β-asarone and/or Nrf2 inhibitor for consecutive 20 days. Ns – not significant; * p<0.05, ** p<0.01, **** p<0.0001. Figure 2. Cresyl Violet staining for the pathologic changes in Nissl bodies brain tissues of control, model, model treated with β-asarone and/or Nrf2 inhibitor groups. Bar=50 or 20 μm. Magnification: 20×; 40×.
Figure 2. Cresyl Violet staining for the pathologic changes in Nissl bodies brain tissues of control, model, model treated with β-asarone and/or Nrf2 inhibitor groups. Bar=50 or 20 μm. Magnification: 20×; 40×. Figure 3. TUNEL staining for the apoptosis of neurons in brain tissues from control, model, model treated with β-asarone, and/or Nrf2 inhibitor. Bar=20 μm.
Figure 3. TUNEL staining for the apoptosis of neurons in brain tissues from control, model, model treated with β-asarone, and/or Nrf2 inhibitor. Bar=20 μm. Figure 4. RT-qPCR assay for detection of the mRNA expression of Nrf2-ARE pathway-related proteins in brain tissues of MCAO rat models. (A) Nrf2; (B) NQO1; (C) GCLM; (D) HO-1 and (E) GCLC. Ns – not significant; * p<0.05, ** p<0.01, *** p<0.001.
Figure 4. RT-qPCR assay for detection of the mRNA expression of Nrf2-ARE pathway-related proteins in brain tissues of MCAO rat models. (A) Nrf2; (B) NQO1; (C) GCLM; (D) HO-1 and (E) GCLC. Ns – not significant; * p<0.05, ** p<0.01, *** p<0.001. Figure 5. Western blot for detecting Nrf2-ARE pathway- and apoptosis-related protein expression in brain tissues of MCAO rat models. (A) Representative images for western blot results. The expression of Nrf2-ARE pathway-related proteins including (B) Nrf2 (C) NQO1, (D) GCLM, (E) HO-1, (F) GCLC and apoptosis-related proteins including (G) BAX, (H) CASP3, (I) CASP9, and (J) Bcl-2 was quantified according to the western blots. * p<0.01, *** p<0.001, ****p<0.0001.
Figure 5. Western blot for detecting Nrf2-ARE pathway- and apoptosis-related protein expression in brain tissues of MCAO rat models. (A) Representative images for western blot results. The expression of Nrf2-ARE pathway-related proteins including (B) Nrf2 (C) NQO1, (D) GCLM, (E) HO-1, (F) GCLC and apoptosis-related proteins including (G) BAX, (H) CASP3, (I) CASP9, and (J) Bcl-2 was quantified according to the western blots. * p<0.01, *** p<0.001, ****p<0.0001. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387