12 August 2021: Clinical Research
Renal Cortical Perfusion Estimated in Color Doppler Dynamic Tissue Perfusion Measurement in Patients Treated with Levothyroxine Following Total Thyroidectomy for Resectable Thyroid Cancer Is Independently Associated with Free Thyroxine: A Single-Center Prospective Study
Arkadiusz Lubas1ABCDEF*, Anna Grzywacz1DEF, Stanisław Niemczyk1BEG, Grzegorz Kamiński2ABE, Marek Saracyn2ABDEFDOI: 10.12659/MSM.932096
Med Sci Monit 2021; 27:e932096
Abstract
BACKGROUND: The thyroid state significantly influences renal function. However, a direct link between thyroid and kidney dysfunction has not been identified. Thyroid hormones affect cardiac output and vascular resistance, and thus can modify kidney perfusion. This prospective study aimed to test the association between renal cortical perfusion (RCP) estimated in color Doppler sonographic dynamic tissue perfusion measurement (DTPM) with thyroid hormones in 36 patients treated with levothyroxine following total thyroidectomy for resectable thyroid cancer.
MATERIAL AND METHODS: Blood tests, blood pressure monitoring, and DTPM of the renal cortex were performed. To exclude possible reading errors, the intrarater reliability of the ultrasound perfusion measurement method was estimated.
RESULTS: The absolute difference between the 2 ultrasound RCP measurements was 5.2±4.4%. RCP correlated significantly with free thyroxine (FT₄) (r=0.46; p=0.006) but not with triiodothyronine and thyroid-stimulating hormone. In the adjusted to age backward stepwise multivariable regression analysis model, including estimated glomerular filtration rate, mean arterial pressure, and FT₄, only FT₄ was independently associated with RCP (R²=0.21; p=0.006).
CONCLUSIONS: Renal cortical perfusion is independently associated with free thyroxine, which can contribute to renal function abnormalities in the condition of impaired thyroid function. This small prospective study from a single center showed that the renal cortex’s color Doppler sonographic dynamic tissue perfusion measurement had very good intraobserver reproducibility.
Keywords: perfusion imaging, Ultrasonography, Doppler, thyroxine, Renal Circulation, Adolescent, Feasibility Studies, Glomerular Filtration Rate, Kidney Cortex, Observer Variation, Postoperative Complications, Prospective Studies, renal insufficiency, Reproducibility of Results, Thyroid Function Tests, Thyroidectomy, Ultrasonography, Doppler, Color, young adult
Background
Renal function is significantly affected by the thyroid state [1]. An increase in thyroid stimulation hormone (TSH) concentration, even within its normal limits, is related to a higher incidence of chronic kidney disease (CKD), and thyroid replacement therapy with levothyroxine is related to slowing decline or even an improvement of the kidney function [1,2]. However, the direct association between thyroid abnormalities and renal dysfunction remains widely unknown [3].
Although triiodothyronine is the most active thyroid hormone, thyroid secrets mostly thyroxine and only 20% of triiodothyronine [4]. Peripheral tissue conversion of thyroxine by 5′-deiodinase delivers lacking 80% of triiodothyronine. Thus, monotherapy with synthetic levothyroxine, which is indistinguishable from natural thyroxine, is sufficient in hypothyroidism and covers accessibility of both thyroid hormones. In patients after total thyroidectomy due to differentiated thyroid cancer (DTC), endogenous production of thyroid hormones decreases, and their lowering concentrations trigger TSH secretion. To avoid the stimulation of DTC proliferation through its TSH receptors, patients have to be treated with high, supraphysiologic doses of levothyroxine, which suppress TSH production [5,6].
Hyperthyroidism and an excess of thyroid hormones accelerate cardiac output and renal blood flow, increasing glomerular filtration rate [7,8]. However, long-standing thyrotoxicosis can be associated with reduced kidney function [8].
Hypothyroidism leads to significant functional and structural alterations of the heart (negative chronotropic and inotropic effects, decreased cardiac contractility and output, myocardial fibrosis), as well as the rise in peripheral vascular resistance and blood pressure [9–11]. On the other hand, adequately preserved renal perfusion, which is essential for proper renal function, is closely associated with cardiac sufficiency [12,13]. The influence of thyroid hormones on organ perfusion likely is one of the important mechanisms affecting kidney function. Recently, a significant worsening of renal perfusion and function after the recombinant human thyrotropin administration was shown [14]. However, in hypothyroidism, high TSH concentrations coexist with low concentrations of free thyroid hormones, which can also be associated with renal perfusion and function [8]. Thyroidectomy followed by exogenous thyroid hormones supplementation seems to be an appropriate and unique model for testing thyroid hormones and renal function relations. In the last years, an ultrasound method of color Doppler dynamic tissue perfusion measurement (DTPM) was introduced for quantification of renal cortical perfusion (RCP) [15].
Therefore, this prospective study aimed to test the association between renal cortical perfusion estimated in color Doppler sonographic dynamic tissue perfusion measurement with thyroid hormones in patients treated with levothyroxine following total thyroidectomy for resectable thyroid cancer.
Material and Methods
COMPLIANCE WITH ETHICS STANDARDS:
All procedures performed in studies involving human participants were in accordance with the ethics standards of the local institutional research committee (reference number 9/WIM/2019, approval date 20/02/2019) and with the 1964 Helsinki Declaration and its later amendments or comparable ethics standards.
Informed consent was obtained from all individual participants included in the study.
STUDY POPULATION:
For 13 months of study duration, 36 consecutive patients (9M, 27F; age 38.2±11.4) 3–6 months after total thyroidectomy due to the differentiated thyroid cancer (34 papillary, 2 follicular) admitted to the Endocrinology and Isotope Therapy Department were included in this cohort prospective observational study. All investigated patients were diagnosed with DTC, underwent thyroidectomy, and were qualified for isotope ablation therapy in other centers. Exclusion criteria were age over 55 years, pregnancy, acute or chronic disease other than thyroid cancer qualified for the treatment, abnormalities of kidneys in a prior ultrasound or computed tomography examination, and the use of any drug other than levothyroxine. Investigated patients were hospitalized to undergo ablation with radioiodine of potentially remnant thyroid tissue, and the study was performed before isotope treatment.
LABORATORY TESTS:
Blood tests were performed in the early morning after fasting. They included TSH, free thyroxine (FT4), free triiodothyronine (FT3), and serum creatinine with an estimation of glomerular filtration rate (eGFR) based on the CKD-EPI (chronic kidney disease-epidemiology collaboration) equation [16]. The concentration of TSH was measured with the use of the electrochemiluminescence immunoassay (ECLIA) method (Cobas e601 analyzer, Roche), with a reference range of 0.4–4.2 μIU/mL. Concentrations of FT4 and FT3 were measured using the same ECLIA method, with a reference range of 12.0–22.0 pmol/L and 3.2–6.9 pmol/L, respectively. Serum creatinine was estimated using the enzymatic method (Cobas c 601 analyzer, Roche) with a reference range of 61.9–106.1 μmol/L.
ULTRASOUND EXAMINATION:
Kidney 2D ultrasound examination (Logiq P6 with a curved array probe of 2–5 MHz (GE Healthcare, Seoul, Korea)) included measurement of the right kidney length and cortical thickness with the careful assessment of parenchymal echoic structure and exclusion of pathologic changes in the renal sinus and calyces. A Dynamic Tissue Perfusion Measurement (DTPM) of the renal cortex was performed with the use of stable gain of color Doppler (constant frequency of 3.3 MHz), which was described before [13,15]. Briefly, the renal perfusion had been estimated in the middle segment of the longitudinal projection of the right kidney with blood pressure measurement (Omron 705IT, Omron Corporation, Kyoto, Japan) on the left arm, just before the ultrasound examination. A cortical segment between 2 medullar pyramids was visualized, and then a color Doppler frame was inserted, encompassing the area between the pyramids and renal capsule. Color Doppler flow was recorded for about 3 s. The mean value of arterial and venous flow intensity in the entire cortex in the examined segment was calculated in an external medical device (PixelFlux, Chameleon Software, Leipzig, Germany) as the Renal Cortical Perfusion (RCP) and then considered for statistical analysis. For the assessment of the repeatability of the DTPM method, in 20 participants (5M, 15F) with well-defined color Doppler flow in the inner and outer renal cortex, a second measurement was performed. After the first measurement, the transducer was moved away from the skin. Then the second attempt, with a reproduced transducer position, was accomplished in the same region of interest and ultrasound presets. An analysis of the repeated RCP measurements showed very good repeatability of the method. The intraclass correlation coefficient for single measures was 0.980 (95% CI: 0.950–0.992) and the absolute difference between measurements was only 5.2±4.4%. A Bland-Altman plot indicated that 95% of repeated measurements were within ±1.96SD=95% limits of agreement (Figure 1).
STATISTICAL ANALYSIS:
The results of the performed tests were presented as a mean with a standard deviation (SD) and a median with an interquartile range (IQR). The quantitative variables distribution was checked with the Shapiro-Wilk test. Depending on the type of distribution, correlations between variables were analyzed with Pearson’s or Spearman’s test. The backward stepwise multivariable linear regression analysis was used to identify factors independently associated with RCP. Missing data were not replaced by the mean but were omitted. In all presented analyses, p-value <0.05 was considered as significant.
To present intraobserver variability, a Bland-Altman plot was calculated. Statistical tests were performed with the use of Statistica 12 software (StatSoft Inc., Tulsa, OK, USA). The intraclass correlation coefficient (ICC) (model 3, form 1, absolute agreement) and the absolute value of the difference between the 2 measurements were calculated using an Excel spreadsheet [17,18].
For sample size calculation, data from 14 patients investigated in the first 2 months of the study were analyzed. In this small group, RCP correlated significantly with FT4 (r=0.516), and for p<0.05, the power of test 0.9, the estimated sample size was 35.
Results
Results of blood tests, blood pressure monitoring, and Doppler examinations are shown in Table 1.
Median TSH concentration was 0.24 μIU/mL (range: 0.01–32.39). Six patients had TSH above the upper limit of the reference range (0.27–4.20 μIU/mL), and one of them had a low concentration of FT3 (3.0 pmol/L), whereas FT4 was in the reference range. Twenty patients had lowered TSH <0.27 μIU/mL, and 10 of them had high concentrations of FT4, but FT3 was not elevated.
To test an association of thyroid hormones and the renal perfusion, an analysis of correlation was performed and showed a significant association between RCP and levothyroxine dose (r=0.340; p=0.042) and FT4 (r=0.457; p=0.006, power of the test 82,6%), but not with FT3 (r=0.012; p=0.945), and TSH (r=−0.108, p=0.531) (Figure 2).
In the model of stepwise multivariable regression analysis adjusted to age and considering estimated glomerular filtration rate, mean arterial pressure, and FT4 (Table 2), only FT4 concentration was independently associated with RCP (r=0.46; R2=0.21, p=0.006). Differences in perfusion intensity between patients with high or low concentrations of FT4 are presented in Figure 3A, 3B.
Discussion
In the presented study, we show for the first time the significant and independent relation between free thyroxine and renal cortical perfusion measured by the dynamic ultrasound method. Moreover, to the best of our knowledge, this is the first report assessing the repeatability of the DTPM method used for renal cortex perfusion assessment. In our study, the DTPM had very good intraobserver reproducibility, and the absolute difference between the 2 ultrasound measurements was only 5.2±4.4%. This finding is comparable to the reported variability of ultrasound renal resistive index (RRI), in which differences between measurements taken in renal segmental arteries by well-trained staff ranged from 2.1 to 5.1% [19]. However, recent findings suggest that RRI does not sufficiently represent renal parenchymal perfusion as opposed to the dynamic cortical indexes [20].
For many years, a simple assessment of renal perfusion has been challenging [21]. Although the use of different kinds of contrast media (iodinated, paramagnetic, isotope, air bubble) is very helpful in perfusion quantification, these radiologic methods require specific types of equipment and staff. Widely accessible ultrasound examination using the color Doppler option can only assess renal cortical perfusion qualitatively. However, in the DTPM method using dedicated software, careful scanning of renal parenchyma with color Doppler option enables simple quantitative perfusion assessment without use of contrast media [15].
As has been shown earlier, the thyroid state has a specific influence on the blood vessel wall. Thyroid hormones, by genomic and underlying genomic mechanisms, increase local nitric oxide production, transmembrane transport of sodium, potassium, and calcium, and induce relaxation in vascular smooth muscles, thereby improving vascular reactivity [9–11]. Studies concerning the influence of thyroid hormones on renal perfusion are sparse, while most of the investigations report the higher incidence of chronic kidney disease (CKD) in thyroid abnormalities [3]. In an experimental study, Sabio et al showed an increased reactivity on vasoconstrictors in hyperthyroid rat kidneys, but in hypothyroid animals, renal vasculature showed a significantly reduced sensitivity [22]. This work proved the influence of the thyroid state on renal vessels, which could be explained by the increased density of β-adrenergic receptors due to elevated thyroid function [23]. Inrecent work, Stock et al examined 42 hyperthyroid cats before and after radioiodine (131I) therapy using contrast-enhanced ultrasound. The authors showed a significant decrease in renal cortical perfusion after treatment, which corresponded with the resolution of the hyperthyroid state and diminished renal function [24]. In an experimental human study, Tsuda et al investigated 26 patients and showed a significant proportional relation of TSH (r=0.609, p=0.001) with the renal afferent arteriole resistance, but a negative association with renal blood flow (RBF) and inulin-based GFR [25]. However, neither FT3 nor FT4 was related to renal arterial resistance, RBF, and GFR. Although Tsuda et al’s findings are essential, the lack of the impact of free thyroid hormones on RBF and GFR could be dissembled by glomerular hyperfiltration corresponding to diabetes mellitus in 24 participants. Moreover, recently, we showed a significant worsening of renal perfusion followed by a reduction in eGFR after the recombinant human thyrotropin administration in a selected population of patients after thyroidectomy [14]. Thus, the concentration of TSH could have an exclusive influence on renal perfusion and function, regardless of free thyroid hormones. Additionally, in the presented work, we show that in normotensive, mostly euthyroid patients without renal function abnormalities, the concentration of FT4 is independently correlated with RCP and explains about 21% of renal cortical perfusion changeability. An exclusive contribution of FT4 in this relation can be supported by the fact that FT4 is the main thyroid hormone circulating in the blood, whereas FT3 is chiefly generating intracellularly in the deiodination process [4]. Total daily production, fraction releasing from thyroid, and concentration of both total and free form of FT4 is much higher than the FT3 ones. Thereby, the systemic FT4 changes could have a more substantial relation with renal perfusion than the variability of FT3 concentration. The next factor that could influence the presented result is the total thyroidectomy that patients underwent earlier. In this situation, supplementation of thyroid hormones is obligatory [5]. Oral treatment with levothyroxine alone is sufficient for maintaining serum concentrations of both thyroid hormones in a close to physiologic pattern [4]. Moreover, to suppress TSH production and diminish the risk of DTC remnants and potential metastases proliferation, even higher than optimal doses of levothyroxine alone are recommended [5,6]. Oral supplementation of levothyroxine in one prescribed dose probably provides a more stable serum concentration of free thyroxine compared to TSH or triiodothyronine.
Another reason for this particular relation of renal cortical perfusion with FT4 may be that FT4 has its own exclusive cellular membrane transporter, OATP1C1 (organic anion transporting peptide 1C1), which is mainly located in capillary vessels and also in the glomerular capillary wall [26]. Hence, systemic changes in FT4 could transfer to renal perfusion.
On the other hand, previously performed studies showed a significant association of RCP with renal and cardiac systolic function in hypertensive patients with CKD [12,27]. It is well known that thyroid hormones increase cardiac output and reduce systemic vascular resistance, which indirectly contributes to an increase in renal blood flow [8]. Thus, it is clear that the influence of the thyroid state on RCP is simultaneously direct and indirect. Moreover, we can hypothesize that the significant and independent association between FT4 and RCP shown in our study could be responsible for the increased prevalence of CKD in a hypothyroid state.
Despite promising results, our work has several limitations. Due to the very strict qualification criteria, only 36 patients were recruited, which is a relatively small sample size investigated in a single center. Although the repeatability of the DTPM method was assigned as very good/excellent, we have not assessed inter-observer variability due to only one rater. Operator and equipment dependency could be the main reason for DTPM result discrepancies and inconsistency in multicenter studies [28]. Moreover, accessibility of this method is limited by the specific software, which unfortunately is still not implemented in the ultrasound systems. On the other hand, as each ultrasound method requires experience in its use and interpretation, we have successfully performed ultrasound DTPM in the renal cortex for many years. Thus, we believe that the results presented in this article are as reliable as they can be. Finally, this study shows only the association between thyroxine concentrations and renal cortical perfusion at the moment of investigation, without time-dependent observation and evidence of kidney function change. Due to the rather small sample size, further longitudinal studies with a larger group of patients are needed to confirm our findings.
Conclusions
Renal cortical perfusion is independently associated with free thyroxine, which can contribute to renal function abnormalities in the condition of impaired thyroid function. Our small prospective study from a single center showed that color Doppler sonographic dynamic tissue perfusion measurement of the renal cortex had very good intraobserver reproducibility. The integration of the dynamic tissue perfusion measurement with ultrasound software could exclude equipment-dependent variability and extend the availability of the method.
Figures
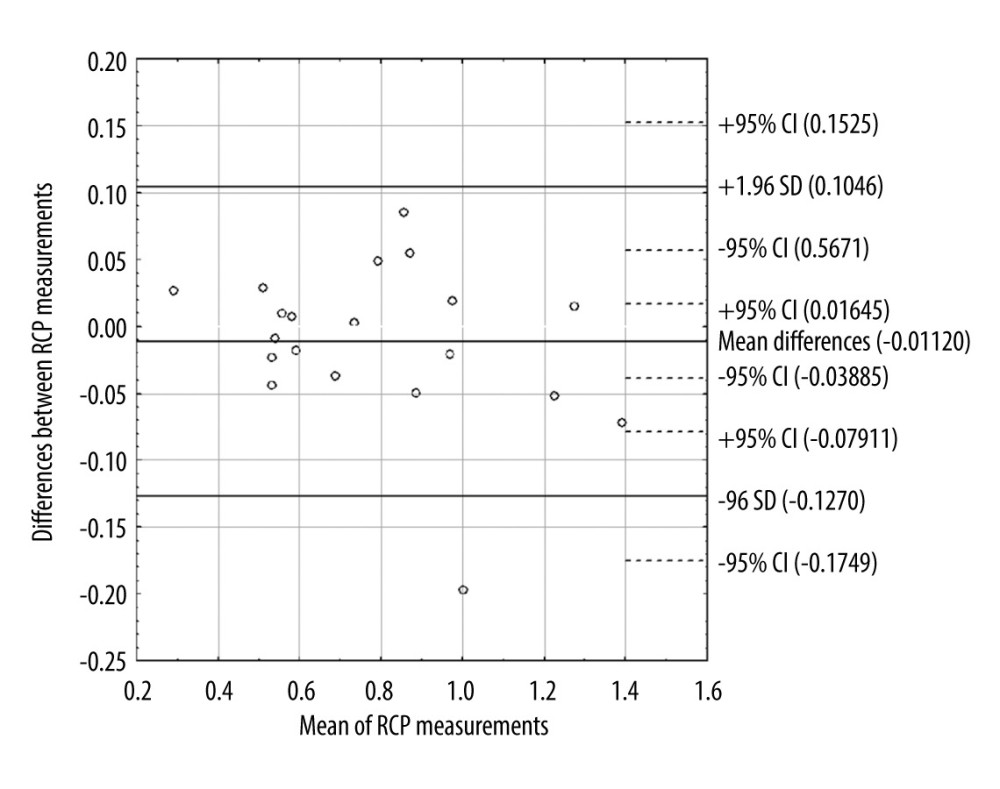 Figure 1. Bland-Altman plot of differences between 2 renal cortical perfusion measurements. CI – confidence interval; SD – standard deviation; RCP – renal cortical perfusion.
Figure 1. Bland-Altman plot of differences between 2 renal cortical perfusion measurements. CI – confidence interval; SD – standard deviation; RCP – renal cortical perfusion. 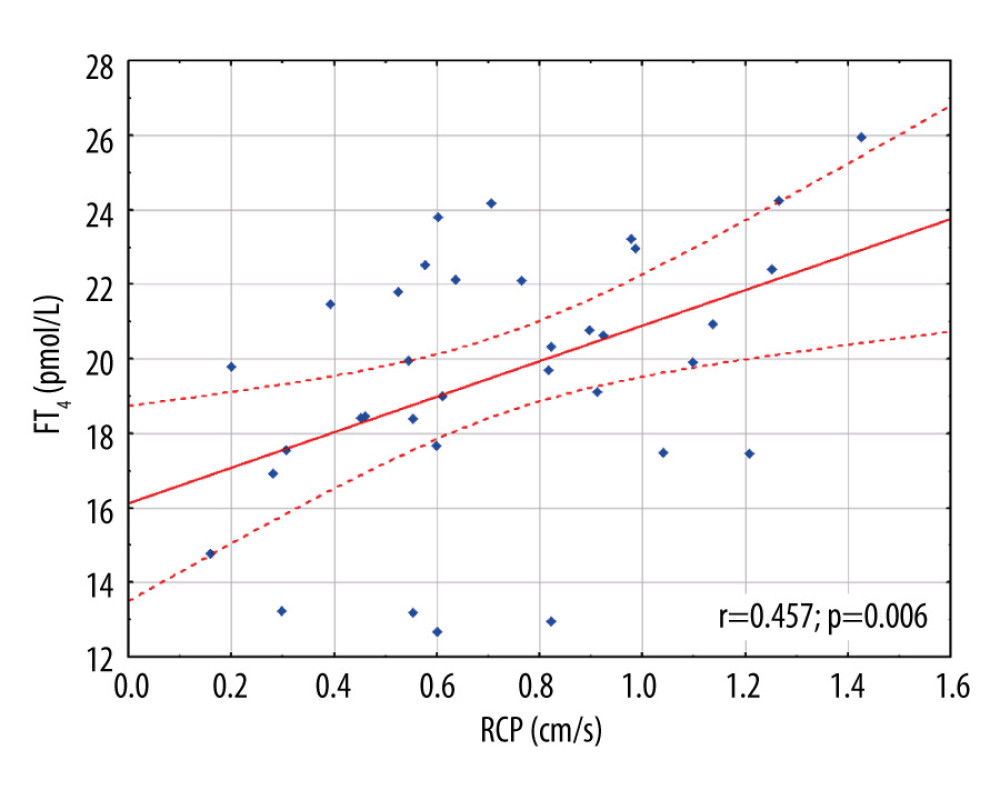 Figure 2. Scatter plot with the linear regression showing association between renal cortical perfusion and free thyroxine. FT4 – free thyroxine; RCP – renal cortical perfusion.
Figure 2. Scatter plot with the linear regression showing association between renal cortical perfusion and free thyroxine. FT4 – free thyroxine; RCP – renal cortical perfusion. 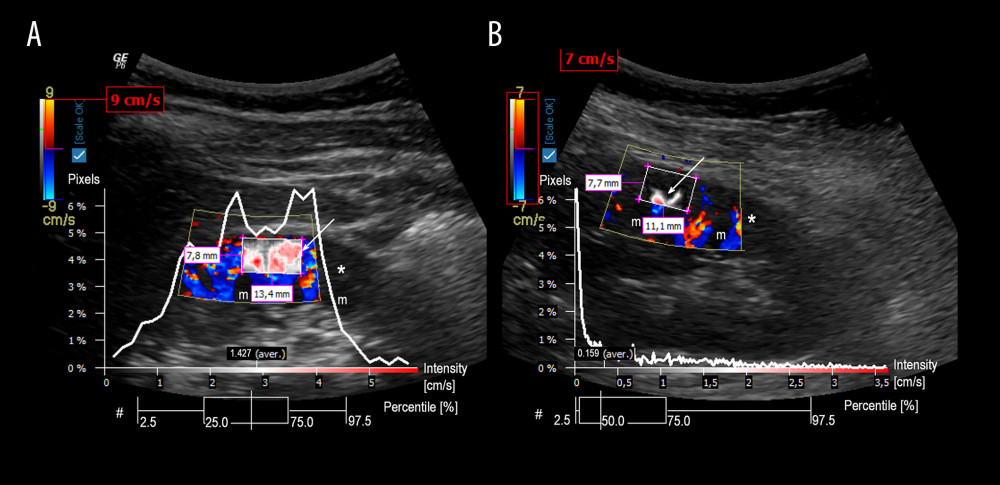 Figure 3. Graphical presentation of the ultrasound examinations and analyses of renal cortical perfusion in the 2 patients with different free thyroxine concentrations. Graphics present the perfusion relief (white arrow) and the perfusion intensity (white curve) assessment in the renal cortex. A diagram Pixels/Intensity is a quantitative evaluation of RCP showing how many pixels within the investigated region of interest are related to the intensity of the respective value. A box-plot diagram (#) expresses the distribution of pixel perfusion intensity, in percentiles (whisker: 2.5% and 97.5%; box 25% and 75%; vertical line in the box 50% – median). A – examination of a patient with FT4 25.93 pmol/L and RCP 1.427 cm/s; B – examination of a patient with FT4 14.77 pmol/L and RCP 0.159 cm/s; * – renal cortex; m – renal medulla.
Figure 3. Graphical presentation of the ultrasound examinations and analyses of renal cortical perfusion in the 2 patients with different free thyroxine concentrations. Graphics present the perfusion relief (white arrow) and the perfusion intensity (white curve) assessment in the renal cortex. A diagram Pixels/Intensity is a quantitative evaluation of RCP showing how many pixels within the investigated region of interest are related to the intensity of the respective value. A box-plot diagram (#) expresses the distribution of pixel perfusion intensity, in percentiles (whisker: 2.5% and 97.5%; box 25% and 75%; vertical line in the box 50% – median). A – examination of a patient with FT4 25.93 pmol/L and RCP 1.427 cm/s; B – examination of a patient with FT4 14.77 pmol/L and RCP 0.159 cm/s; * – renal cortex; m – renal medulla. References
1. Åsvold BO, Bjøro T, Vatten LJ, Association of thyroid function with estimated glomerular filtration rate in a population-based study: The HUNT study: Eur J Endocrinol, 2011; 164; 101-5
2. Shin DH, Lee MJ, Lee HS, Thyroid hormone replacement therapy attenuates the decline of renal function in chronic kidney disease patients with subclinical hypothyroidism: Thyroid, 2013; 23; 654-61
3. Rhee CM, Brent GA, Kovesdy CP, Thyroid functional disease: An under-recognized cardiovascular risk factor in kidney disease patients: Nephrol Dial Transplant, 2015; 30; 724-37
4. Hennessey JV, The emergence of levothyroxine as a treatment for hypothyroidism: Endocrine, 2017; 55; 6-18
5. Patel KN, Yip L, Lubitz CC, The American Association of Endocrine Surgeons guidelines for the definitive surgical management of thyroid disease in adults: Ann Surg, 2020; 271; e21-93
6. Haugen BR, Alexander EK, Bible KC, 2015 American Thyroid Association management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer The American Thyroid Association Guidelines Task Force on thyroid nodules and differentiated thyroid cancer: Thyroid, 2016; 26; 1-133
7. Kim SH, Min HK, Lee SW, Relationship between thyroid and kidney function: Analysis from the Korea national health and nutrition examination survey between 2013 and 2015. Kidney Blood Press: Res, 2020; 45; 442-54
8. Basu G, Mohapatra A, Interactions between thyroid disorders and kidney disease: Indian J Endocrinol Metab, 2012; 16; 204
9. Napoli R, Biondi B, Guardasole V, Impact of hyperthyroidism and its correction on vascular reactivity in humans: Circulation, 2001; 104; 3076-80
10. Klein I, Ojamaa K, Thyroid hormone and the cardiovascular system: N Engl J Med, 2001; 344; 501-9
11. Kahaly GJ, Dillmann WH, Thyroid hormone action in the heart: Endocr Rev, 2005; 26; 704-28
12. Lubas A, Ryczek R, Kade G, Impact of cardiovascular organ damage on cortical renal perfusion in patients with chronic renal failure: Biomed Res Int, 2013; 2013; 137868
13. Lubas A, Ryczek R, Kade G, Niemczyk S, Renal perfusion index reflects cardiac systolic function in chronic cardio-renal syndrome: Med Sci Monit, 2015; 21; 1089-96
14. Saracyn M, Lubas A, Bober B, Recombinant human thyrotropin worsens renal cortical perfusion and renal function in patients after total thyroidectomy due to differentiated thyroid cancer: Thyroid, 2020; 30(5); 653-60
15. Scholbach T, Dimos I, Scholbach J, A new method of color Doppler perfusion measurement via dynamic sonographic signal quantification in renal parenchyma: Nephron Physiol, 2004; 96; 99-104
16. Levey AS, Stevens LA, Schmid CH, A new equation to estimate glomerular filtration rate: Ann Intern Med, 2009; 150; 604-12
17. Popović ZB, Thomas JD, Assessing observer variability: A user’s guide: Cardiovasc Diagn Ther, 2017; 7; 317-24
18. Trevethan R, Intraclass correlation coefficients: Clearing the air, extending some cautions, and making some requests: Health Services and Outcomes Research Methodology, 2017; 17; 127-43
19. Lubas A, Kade G, Niemczyk S, Renal resistive index as a marker of vascular damage in cardiovascular diseases: Int Urol Nephrol, 2014; 46(2); 395-402
20. Lubas A, Niemczyk S, Doppler indexes of the renal cortex are independently related to renal perfusion in hypertensive CKD patients: J Am Soc Nephrol, 2018; 29; 460-61
21. Lubas A, Kade GImaging methods in quantitative assessment of renal perfusion: Lekarz Wojskowy, 2010; 88; 403-7 [in Polish]
22. Sabio JM, Rodriguez-Maresca M, Luna JD, Vascular reactivity to vasoconstrictors in aorta and renal vasculature of hyperthyroid and hypothyroid rats: Pharmacology, 1994; 49(4); 257-64
23. Vargas F, Moreno JM, Rodríguez-Gómez I, Vascular and renal function in experimental thyroid disorders: Eur J Endocrinol, 2006; 154(2); 197-212
24. Stock E, Daminet S, Paepe D, Evaluation of renal perfusion in hyperthyroid cats before and after radioiodine treatment: J Vet Intern Med, 2017; 31(6); 1658-63
25. Tsuda A, Inaba M, Ichii M, Relationship between serum TSH levels and intrarenal hemodynamic parameters in euthyroid subjects: Eur J Endocrinol, 2013; 169(1); 45-50
26. Strømme P, Groeneweg S, Lima de Souza EC, Mutated thyroid hormone transporter OATP1C1 associates with severe brain hypometabolism and juvenile neurodegeneration: Thyroid, 2018; 28(11); 1406-15
27. Lubas A, Kade G, Ryczek R, Ultrasonic evaluation of renal cortex arterial area enables differentiation between hypertensive and glomerulonephritis-related chronic kidney disease: Int Urol Nephrol, 2017; 49(9); 1627-35
28. Stoperka F, Karger C, Beige J, Limited accuracy of colour Doppler ultrasound dynamic tissue perfusion measurement in diabetic adults: PLoS One, 2016; 11(12); e0168905
Figures
 Figure 1. Bland-Altman plot of differences between 2 renal cortical perfusion measurements. CI – confidence interval; SD – standard deviation; RCP – renal cortical perfusion.
Figure 1. Bland-Altman plot of differences between 2 renal cortical perfusion measurements. CI – confidence interval; SD – standard deviation; RCP – renal cortical perfusion. Figure 2. Scatter plot with the linear regression showing association between renal cortical perfusion and free thyroxine. FT4 – free thyroxine; RCP – renal cortical perfusion.
Figure 2. Scatter plot with the linear regression showing association between renal cortical perfusion and free thyroxine. FT4 – free thyroxine; RCP – renal cortical perfusion. Figure 3. Graphical presentation of the ultrasound examinations and analyses of renal cortical perfusion in the 2 patients with different free thyroxine concentrations. Graphics present the perfusion relief (white arrow) and the perfusion intensity (white curve) assessment in the renal cortex. A diagram Pixels/Intensity is a quantitative evaluation of RCP showing how many pixels within the investigated region of interest are related to the intensity of the respective value. A box-plot diagram (#) expresses the distribution of pixel perfusion intensity, in percentiles (whisker: 2.5% and 97.5%; box 25% and 75%; vertical line in the box 50% – median). A – examination of a patient with FT4 25.93 pmol/L and RCP 1.427 cm/s; B – examination of a patient with FT4 14.77 pmol/L and RCP 0.159 cm/s; * – renal cortex; m – renal medulla.
Figure 3. Graphical presentation of the ultrasound examinations and analyses of renal cortical perfusion in the 2 patients with different free thyroxine concentrations. Graphics present the perfusion relief (white arrow) and the perfusion intensity (white curve) assessment in the renal cortex. A diagram Pixels/Intensity is a quantitative evaluation of RCP showing how many pixels within the investigated region of interest are related to the intensity of the respective value. A box-plot diagram (#) expresses the distribution of pixel perfusion intensity, in percentiles (whisker: 2.5% and 97.5%; box 25% and 75%; vertical line in the box 50% – median). A – examination of a patient with FT4 25.93 pmol/L and RCP 1.427 cm/s; B – examination of a patient with FT4 14.77 pmol/L and RCP 0.159 cm/s; * – renal cortex; m – renal medulla. Tables
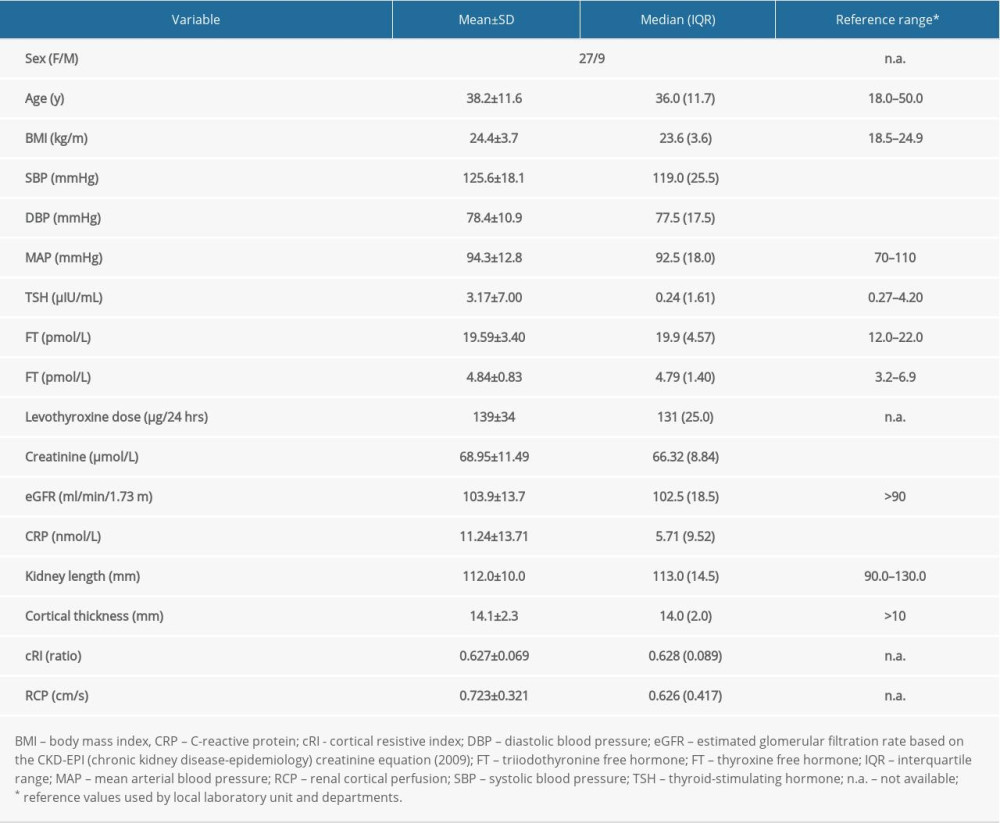 Table 1. Demographic data and results of performed tests in all investigated patients.
Table 1. Demographic data and results of performed tests in all investigated patients.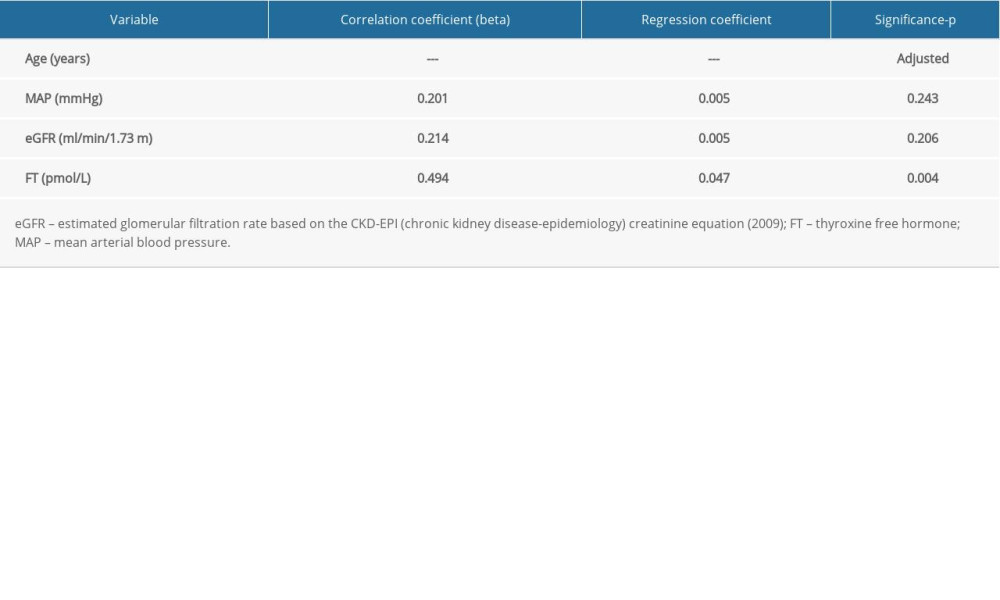 Table 2. Results of multivariable regression analysis for renal cortical perfusion prediction before backward elimination of insignificant variables.
Table 2. Results of multivariable regression analysis for renal cortical perfusion prediction before backward elimination of insignificant variables. Table 1. Demographic data and results of performed tests in all investigated patients.
Table 1. Demographic data and results of performed tests in all investigated patients. Table 2. Results of multivariable regression analysis for renal cortical perfusion prediction before backward elimination of insignificant variables.
Table 2. Results of multivariable regression analysis for renal cortical perfusion prediction before backward elimination of insignificant variables. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








