15 December 2021: Clinical Research
Risk Factors for Cryptococcal Meningitis Recurrence in Human Immunodeficiency Virus (HIV)-Infected Patients in a Large Chinese Acquired Immune Deficiency Syndrome (AIDS) Treatment Center
Yue Zhou1AEF, Feng Li1BCD, Rui Li1BCD, Yuan Peng1CD, Mei He1CD, Fengjun Sun2ADEF*, Ming Yang12AEFGDOI: 10.12659/MSM.933688
Med Sci Monit 2021; 27:e933688
Abstract
BACKGROUND: Cryptococcal meningitis (CM) is one of the most common opportunistic neuroinfections in patients with HIV. Most studies have focused on non-HIV CM and there are only a few studies on HIV CM in China. The purpose of the present study was to evaluate the characteristics and risk factors for CM recurrence in patients infected with HIV in the Chongqing Public Health Treatment Center in China.
MATERIAL AND METHODS: From January 2014 to December 2017, all patients with CM aged 18 years or older were enrolled and a case-control study was performed to determine the risk factors associated with recurrence of CM. Antimicrobial susceptibility was determined with a fungal drug sensitivity kit and the sequence types (STs) were analyzed with multilocus sequence typing.
RESULTS: The incidence of CM in the 5185 HIV-infected patients was 3.5% (179). Follow-up data were available for 82 of the patients for whom complete medical records were available and they were included in the present study. There were 7 STs among 82 Cryptococcus neoformans isolates; ST5 and ST31 were the most prevalent genotypes. Testing showed that C. neoformans had high sensitivity to 5 antifungal drugs and no differences in resistance were observed, even when different STs were tested. Risk factors for recurrence were analyzed in 69 patients, excluding those who died. The results of multivariate analysis showed that only hospital stay was associated with recurrence of CM.
CONCLUSIONS: Our results indicated that combining education about medication with clinical treatment could help prevent recurrence of CM.
Keywords: Cryptococcus neoformans, Drug Resistance, Fungal, Meningitis, Cryptococcal, Multilocus Sequence Typing, Population Characteristics, AIDS-Related Opportunistic Infections, Adult, Antifungal Agents, Case-Control Studies, Female, Humans, Male, Microbial Sensitivity Tests, Recurrence, Risk Factors
Background
Cryptococcal meningitis (CM) is one of the most common opportunistic infections in patients with immunodeficiency, especially those who are infected with HIV. CM is the second most common opportunistic neuroinfection in patients with HIV [1]. A study estimated that in low-income countries, 1-year mortality after CM was 70% (uncertainty interval 59–81) in those who received care and 100% in those who did not receive care [2–4]. The lethality of cryptococcosis is high, even with routine use of antifungal drugs. The 1-year mortality rate is 20% in those who receive care and 30% in those who do not receive care in the United States. Annual deaths from CM are estimated at 181 100 (95% confidence interval [CI] 119 400 to 234 300) and CM accounts for 10% to 15% of AIDS-related mortality [2,5]. Although global improvements have been made in access to antiretroviral therapy (ART) and most patients consent to medical treatment, the number of HIV-infected people with a CD4 count <100 cells/μL is still substantial: 20% to 25% among those presenting for care [6]. The global prevalence of cryptococcal antigenemia in HIV-seropositive individuals with CD4 cell counts <100 cells/μL is estimated at 6%, with 223 100 (95% CI, 150 600 to 282 400) incident cases of CM occurring annually [2].
Material and Methods
PATIENTS AND DATA COLLECTION:
All patients with CM aged 18 years or older admitted to the Chongqing Public Health Treatment Center from January 1, 2014 to December 31, 2017 were enrolled. Located in Chongqing, it is the largest AIDS treatment center in China, with 160 beds. Patients were included in the study if they: were HIV-positive; had culture-proven
The present study was approved by the Ethics Committee of Chongqing Public Health Center and was registered with the hospital registration system for clinical trials (2018-006KY).
DIAGNOSTICS AND TREATMENT REGIMENS:
HIV screening and confirmation were performed using an enzyme-linked immunosorbent assay (ELISA) and western blot. Patients were diagnosed as HIV-positive if their blood samples were positive on both tests. The tests were performed following standard laboratory procedures from the Chinese Center for Disease Control and Prevention. Clinical staging of HIV infection was defined based on the diagnosis and treatment of AIDS in China (2018). In accordance with Chinese policy, all HIV-positive patients who met national treatment guidelines of a CD4 cell count <350 cells/mm3 or had World Health Organization stage III or IV disease were eligible to receive ART [11,12].
Current non-culture methods of diagnosing CM can produce false-positive and false-negative results. To ensure the accuracy of our study, we chose the culture method as the criterion standard. HIV-infected patients were diagnosed with CM disease if their CSF culture was positive for Cryptococcus. Recurrence was defined as a positive CSF culture after at least 2 negative cultures, with a 30-day asymptomatic period [13,14]. Patients with CM were screened for cryptococcal antigen, and to prevent development of cryptococcal disease, those who were positive for cryptococcal antigen were preemptively treated with antifungal therapy. According to the Chinese expert consensus on CM, treatment with amphotericin B (AMB) combined with fluorocytosine (5-FC) is recommended during the induction period and fluconazole (FLU) during the consolidation period.
STRAINS AND DRUG RESISTANCE ASSAYS:
All clinical isolates were obtained from patients suspected of having CM. Identification of Cryptococcus was carried out with a bacterial detection system. Then, the isolates were stored as glycerol stocks and cultured on yeast peptone dextrose medium before they were used [15]. In strict accordance with the manufacturer’s instructions, a Sensititre Yeastone Colorimetric Antifungal Susceptibility Panel (Trek Diagnostic Systems, Thermo Fisher Scientific, West Sussex, UK) was used to test for AMB, 5-FC, FLU, voriconazole (VOR), and itraconazole (ITR) susceptibility. Candida parapsilosis ATCC 22019 was used as the control strain for the susceptibility assays. Based on domestic and foreign research reports, we set the epidemiological cut-off values (ECVs) at 8 mg/L for 5-FC and FLU, 0.25 mg/L for ITR and VOR, and 1 mg/L for AMB for Cryptococcus neoformans [16–18]. In the present study, isolates with minimum inhibitory concentration values above the ECVs were considered resistant.
MULTILOCUS SEQUENCE TYPING:
Genomic DNA was extracted following the manufacturer’s instructions for the Fungal Genomic DNA Extraction kit (Solarbio, Beijing, China). Multilocus sequence typing (MLST) analysis consists of 7 unlinked loci, including 6 housekeeping genes (CAP59, SOD1, GPD1, LAC1, PLB1 and URA5) and the non-coding region IGS1. Both forward and reverse amplicons of each locus were amplified according to the International Society for Human and Animal Mycology consensus MLST scheme for Cryptococcus and then subjected to bidirectional sequencing for all isolates [19]. All sequences were submitted to the C. neoformans/C. gattii species complex database (http://mlst.mycologylab.org) and an allele number was assigned to each of them. Seven allele type numbers and a sequence type (ST) number were given to each specimen after being compared to the MLST database website. The MLST data were analyzed with the hierarchical clustering algorithm in PHYLOViZ 2.0 software (http://www.phyloviz.net/) to construct a rooted tree (dendrogram) that reflected the structure present in a pairwise dissimilarity matrix. At each step, the nearest 2 clusters were combined into a higher-level cluster. The Hamming distance was used as a measure of genetic distance.
STATISTICAL ANALYSIS:
All analyses were performed using the Statistical Package for the Social Sciences software, version 17.0 (SPSS Inc., Chicago, Illinois, United States). Data were expressed as numbers and percentages for categorical variables and continuous variables with normal distribution were presented as mean±standard deviation; non-normal variables were reported as medians with interquartile ranges. In a first analysis, an independent-sample
Results
During the present study, records from 5185 HIV-infected patients at the center were examined. In total, 179 of them (3.5%) who had a CSF culture positive for
Demographic and clinical characteristics of patients with CM who had first onset or recurrence are shown in Table 1. Regardless of recurrence or non-recurrence, the majority were men. At first admission, the mean age of patients with recurrence was greater than that of patients without recurrence. Except for cough, hospital stay, and length of medication continuation after discharge, there were no significant differences in other characteristics between the first-onset and recurrence groups. Univariate analysis was performed to determine whether demographic and abnormal clinical characteristics were risk factors for recurrence of CM (Table 2). Except for cough and liver disease, there was no correlation between clinical symptoms and recurrence. The statistical analysis showed that fungal resistance was not associated with recurrence. In the present study, time-related indicators (hospital stay, duration of medication use after discharge) were associated with recurrence of CM. A multivariate analysis of risk factors was undertaken after adjustment of the logistic regression model for liver disease, cough, hospital stay, and duration of medication use. The results showed that only hospital stay was an independent risk factor for recurrence of CM (Table 3).
The results of the univariate and multivariate analyses indicated that time of administration was an important risk factor for recurrence of CM. Therefore, we divided the total medication time for patients with CM recurrence into 2 groups: >75 days and <75 days. Then, time to recurrence after treatment termination was analyzed, as shown in Figure 2. There were 21 patients with recurrence whose total medication time was <75 days. Of them, 4 (19.05%), 7 (33.33%), 2 (9.52%), and 2 (9.52%) patients presented with recurrence in the 1st, 2nd, 3rd, and 4th months after discharge, respectively. There were 11 patients with recurrence whose total medication time was >75 days. Of them, 2 (18.18%), 2 (18.18%), and 3 (27.27%) patients experience recurrence in the 7th, 8th, and 9th months after discharge, respectively.
MLST has been established as the criterion standard for identifying the major molecular types or populations of
Discussion
Previous studies from other regions have shown that most patients with CM are HIV-positive [2,5]. In China, most study results and viewpoints suggest that immunocompetent individuals are the most likely to develop CM, and researchers believe that the Chinese population may be more susceptible to cryptococcal infections than other ethnic populations [20–22]. The null hypothesis was that HIV patients have a low incidence of CM, but related studies are limited. During the present study, 3.5% of HIV-positive inpatients with a CSF culture positive for
AMB in combination with FC during the induction period followed by FLU during the consolidation period is the recommended antifungal regimen for management of acute CM [28]. In our center, only 39.1% of patients adhered to the recommended therapeutic regimen. Although the recurrence rate in patients after receiving the recommended treatment regimen was lower than that for those who did not follow the recommended treatment regimen, there was no significant difference in the recurrence rate between the patients who followed the recommended treatment regimen and those who did not. The reason may be an insufficient number of samples; additional samples will help us obtain more accurate results. Moreover, cryptococcal resistance was found to be associated with recurrence in other reports [29,30], in contrast with the results of our study. This may be because there were few resistant cryptococci. In the present study, the rate of
Our results showed that only hospital stay was an independent risk factor for recurrence of CM. Considering the follow-up data, we believe that a short hospitalization time and limited medication use outside the hospital resulted in the low compliance rates in patients. The reasons for poor medication compliance were as follows: patients mistakenly believed that they had fully recovered because their symptoms had improved and they were concerned about the cost of the treatment. There may be additional reasons for noncompliance; however, poor adherence to antifungal drug prophylaxis leading to recurrence has been reported in previous research [34]. The costs of antifungal drug treatment result in a heavy economic burden for patients, regardless of hospitalization or discharge. In contrast, patients generally have good compliance with free ART drugs. These results suggest that economic support was an important factor in treatment.
In our study, ST5 and ST31 were the most common epidemic genotypes, similar to results from domestic research reports [16,35]. Notably, in the adjacent province of Sichuan, the ST5 prevalence is 82.9% (34/41) [36]. In contrast to international reports [37,38], ourresults suggest that there were regional differences in genotypes. MLST genotypes are helpful in identifying the source of
Our study had the following limitations. First, the clinical information and isolates were collected from a single center and the sample size was small. Second, only a 4-year period was considered, which was short.
Conclusions
In summary, the present study showed that
Figures
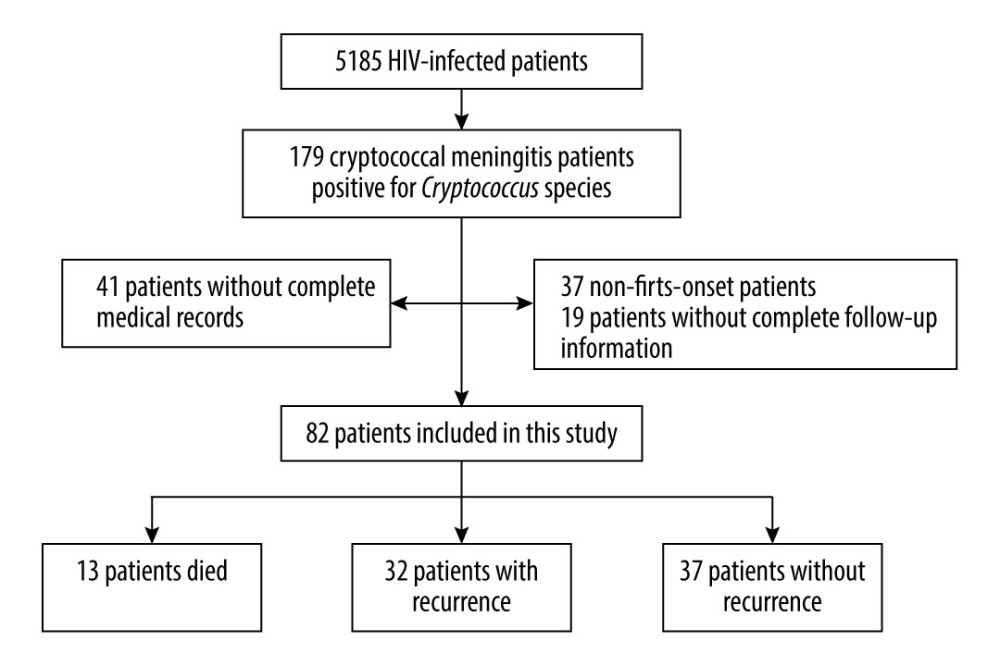 Figure 1. Flowchart showing the flow of patients through the study.
Figure 1. Flowchart showing the flow of patients through the study. 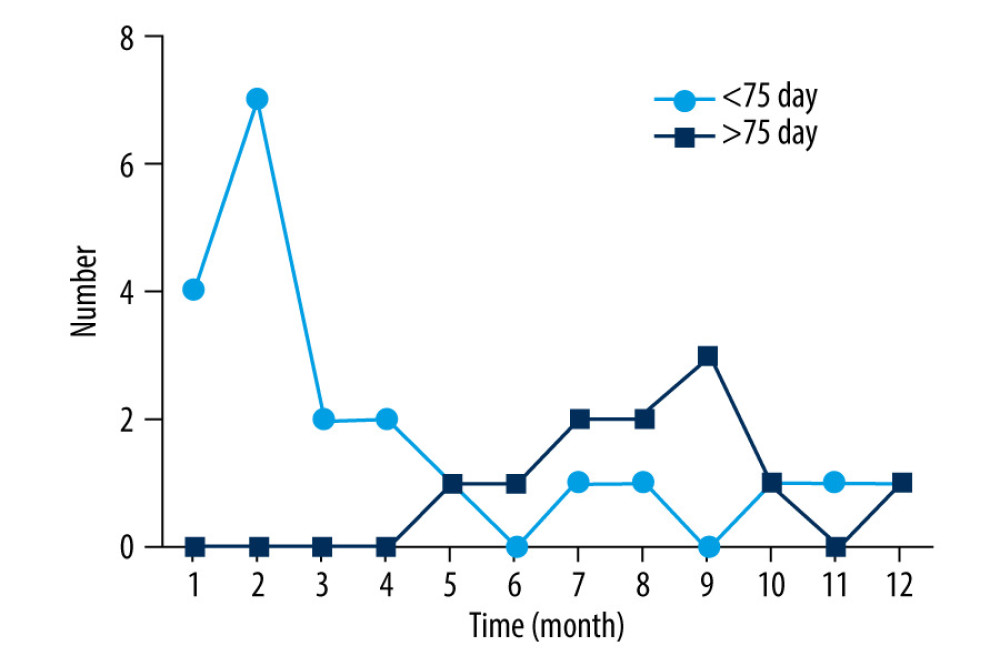 Figure 2. Distribution of patients with recurrence by month after discharge. The patients with recurrence were divided into 2 groups according to total medication time. Of the patients, 21 had total medication times >75 d and 11 had total medication times <75 d.
Figure 2. Distribution of patients with recurrence by month after discharge. The patients with recurrence were divided into 2 groups according to total medication time. Of the patients, 21 had total medication times >75 d and 11 had total medication times <75 d. 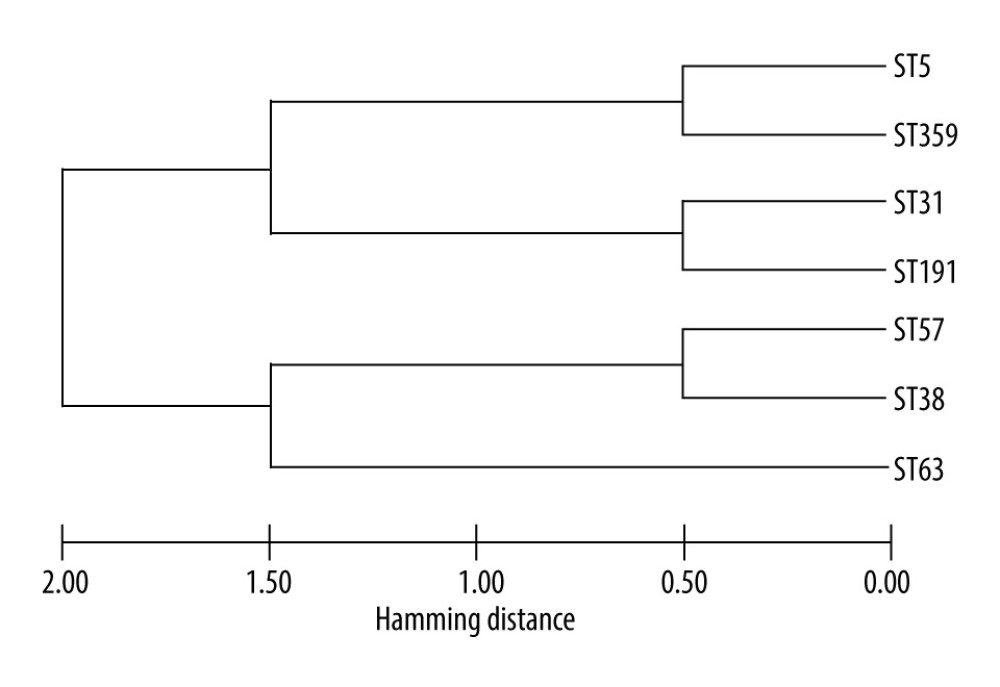 Figure 3. Maximum likelihood phylogenetic analysis of Cryptococcus neoformans.
Figure 3. Maximum likelihood phylogenetic analysis of Cryptococcus neoformans. Tables
Table 1. Demographic and clinical characteristics of 69 cryptococcal meningitis patients with first onset or recurrence.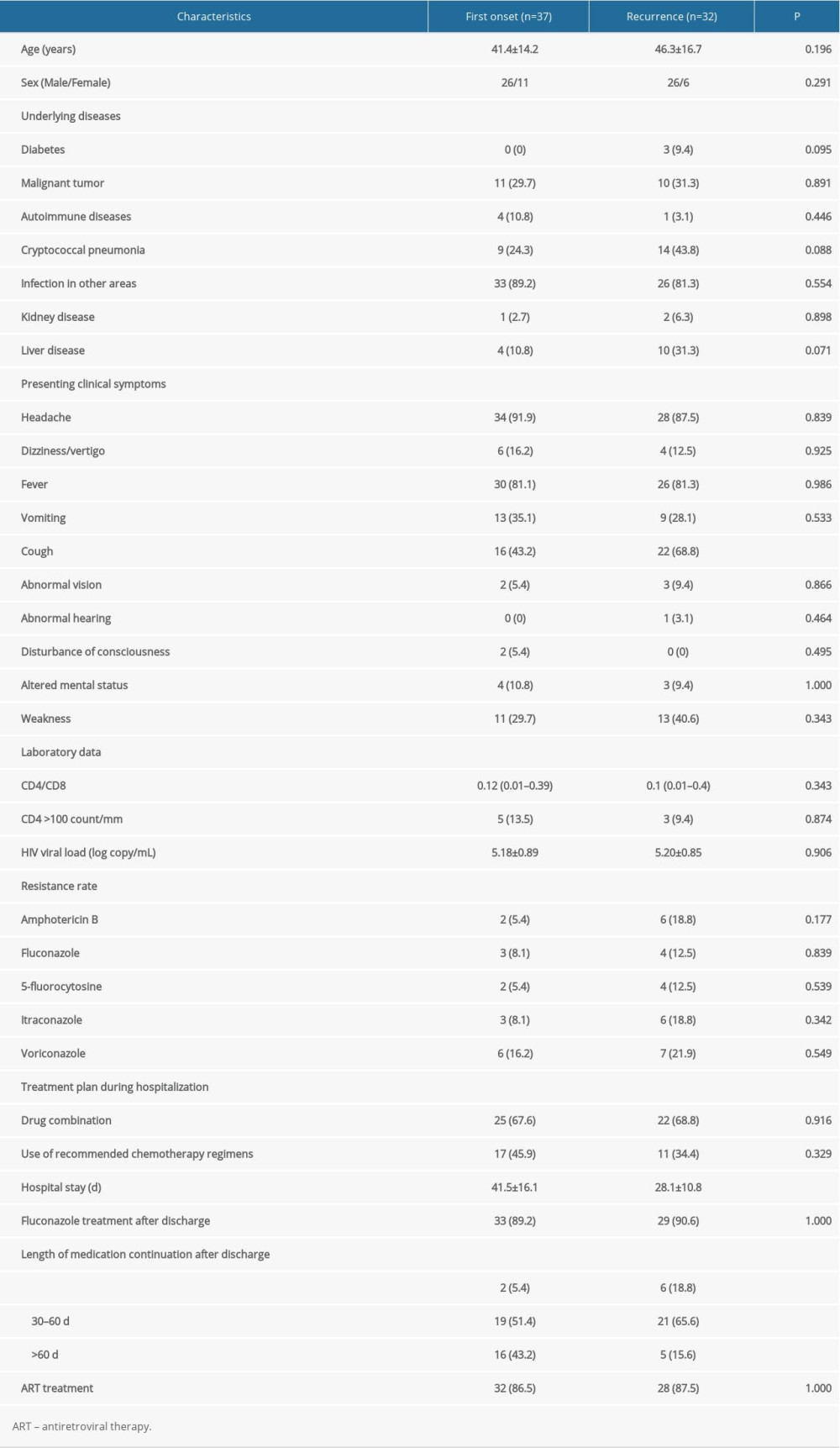 Table 2. Univariate analysis of risk factors for recurrence in cryptococcal meningitis patients.
Table 2. Univariate analysis of risk factors for recurrence in cryptococcal meningitis patients.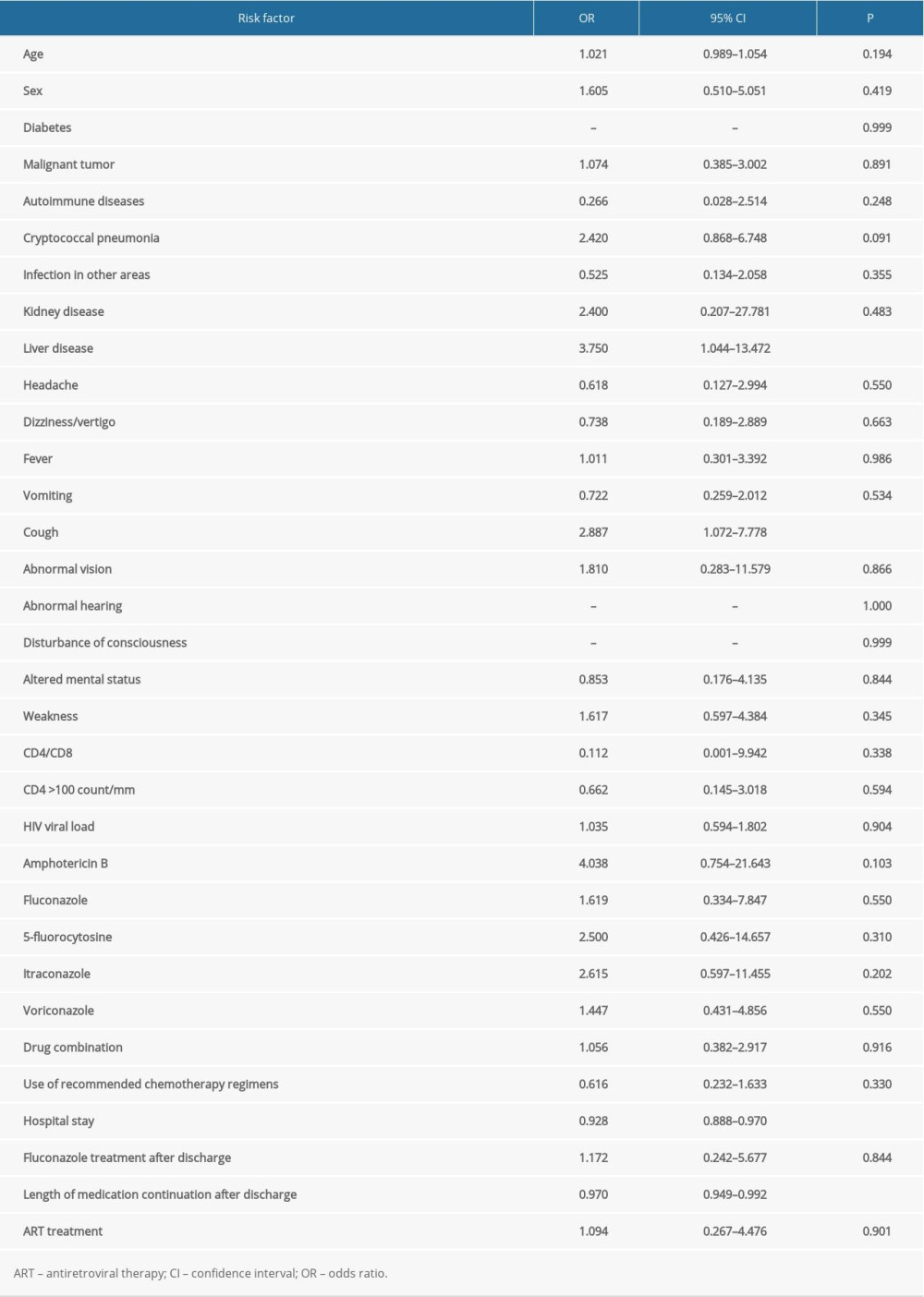 Table 3. Multivariate analysis of risk factors for recurrence in patients with cryptococcal meningitis.
Table 3. Multivariate analysis of risk factors for recurrence in patients with cryptococcal meningitis.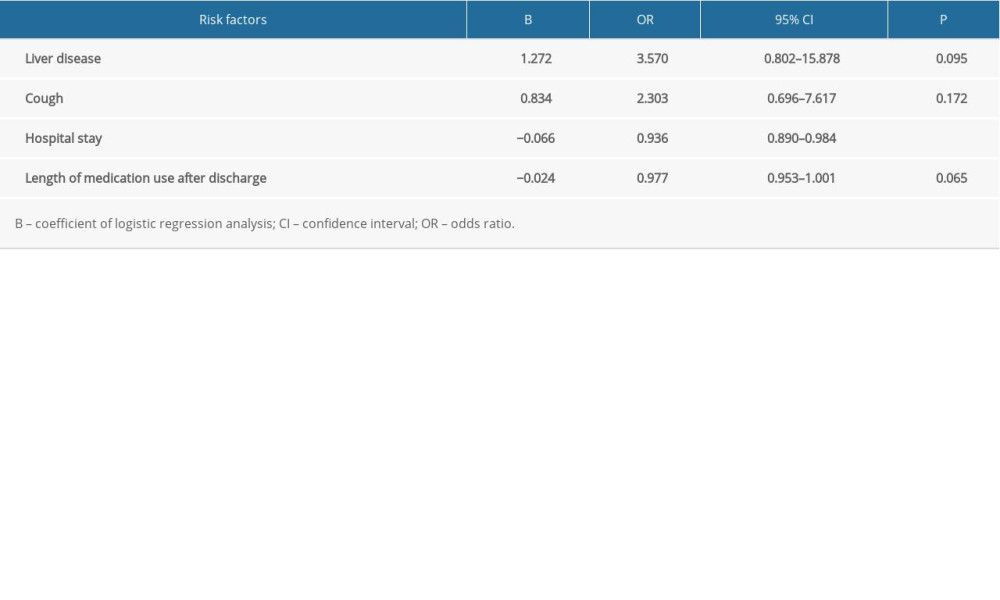 Table 4. Antifungal drug susceptibility results (mg/L).
Table 4. Antifungal drug susceptibility results (mg/L).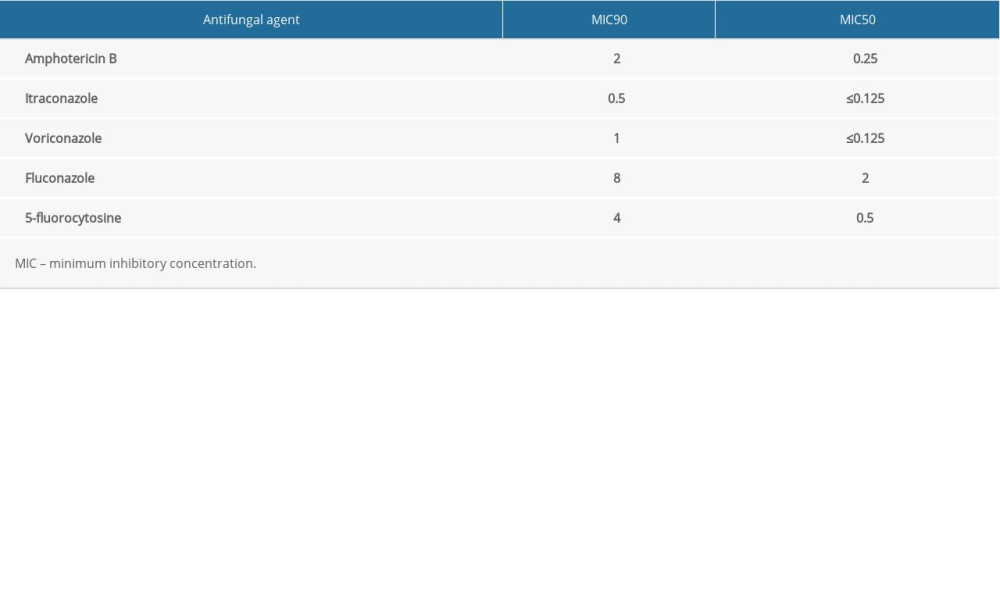
References
1. Currie BP, Casadevall A, Estimation of the prevalence of cryptococcal infection among patients infected with the human immunodeficiency virus in New York City: Clin Infect Dis, 1994; 19(6); 1029-33
2. Rajasingham R, Smith RM, Park BJ, Global burden of disease of HIV-associated cryptococcal meningitis: An updated analysis: Lancet Infect Dis, 2017; 17(8); 873-81
3. Mfinanga S, Chanda D, Kivuyo SL, Cryptococcal meningitis screening and community-based early adherence support in people with advanced HIV infection starting antiretroviral therapy in Tanzania and Zambia: An open-label, randomised controlled trial: Lancet, 2015; 385(9983); 2173-82
4. Govender NP, Roy M, Mendes JF, Evaluation of screening and treatment of cryptococcal antigenaemia among HIV-infected persons in Soweto, South Africa: HIV Med, 2015; 16(8); 468-76
5. Pyrgos V, Seitz AE, Steiner CA, Epidemiology of cryptococcal meningitis in the US: 1997–2009: PLoS One, 2013; 8(2); e56269
6. Lahuerta M, Wu Y, Hoffman S, Advanced HIV disease at entry into HIV care and initiation of antiretroviral therapy during 2006-2011: Findings from four sub-saharan African countries: Clin Infect Dis, 2014; 58(3); 432-41
7. Shen L, Zheng J, Wang Y, Increased activity of the complement system in cerebrospinal fluid of the patients with Non-HIV Cryptococcal meningitis: BMC Infect Dis, 2017; 17(1); 7
8. Qu J, Zhou T, Zhong C, Comparison of clinical features and prognostic factors in HIV-negative adults with cryptococcal meningitis and tuberculous meningitis: A retrospective study: BMC Infect Dis, 2017; 17(1); 51
9. Liao CH, Chi CY, Wang YJ, Different presentations and outcomes between HIV-infected and HIV-uninfected patients with Cryptococcal meningitis: J Microbiol Immunol Infect, 2012; 45(4); 296-304
10. Yuchong C, Fubin C, Jianghan C, Cryptococcosis in China (1985–2010): Review of cases from Chinese database: Mycopathologia, 2012; 173(5–6); 329-35
11. Zhao Y, Sun X, He Y, Progress of the National Pediatric Free Antiretroviral Therapy program in China: AIDS Care, 2010; 22(10); 1182-88
12. Gilks C, Crowley S, Ekpini R, The WHO public-health approach to antiretroviral treatment against HIV in resource-limited settings: Lancet, 2006; 368(9534); 505-10
13. Powderly WG, Saag MS, Cloud GA, A controlled trial of fluconazole or amphotericin B to prevent relapse of cryptococcal meningitis in patients with the acquired immunodeficiency syndrome. The NIAID AIDS Clinical Trials Group and Mycoses Study Group: N Engl J Med, 1992; 326(12); 793-98
14. Bicanic T, Harrison T, Niepieklo A, Symptomatic relapse of HIV-associated cryptococcal meningitis after initial fluconazole monotherapy: The role of fluconazole resistance and immune reconstitution: Clin Infect Dis, 2006; 43(8); 1069-73
15. Wiesner DL, Moskalenko O, Corcoran JM, Cryptococcal genotype influences immunologic response and human clinical outcome after meningitis: mBio, 2012; 3(5); e00196-12
16. Hong N, Chen M, Xu N: Mycoses, 2019; 62(2); 171-80
17. Espinel-Ingroff A, Chowdhary A, Cuenca-Estrella M: Antimicrob Agents Chemother, 2012; 56(6); 3107-13
18. Espinel-Ingroff A, Chowdhary A, Gonzalez GM: Antimicrob Agents Chemother, 2015; 59(1); 666-68
19. Prakash A, Sundar G, Sharma B: Mycoses, 2020; 63(3); 284-93
20. Liu Y, Kang M, Wu SY, Different characteristics of cryptococcal meningitis between HIV-infected and HIV-uninfected patients in the Southwest of China: Med Mycol, 2017; 55(3); 255-61
21. Chen J, Varma A, Diaz MR: Emerg Infect Dis, 2008; 14(5); 755-62
22. Sloan DJ, Parris V, Cryptococcal meningitis: Epidemiology and therapeutic options: Clin Epidemiol, 2014; 6; 169-82
23. Fang W, Fa Z, Liao W: Fungal Genet Biol, 2015; 78; 7-15
24. Lawrence DS, Boyer-Chammard T, Jarvis JN, Emerging concepts in HIV-associated cryptococcal meningitis: Curr Opin Infect Dis, 2019; 32(1); 16-23
25. Chen SC, Meyer W, Sorrell TC: Clin Microbiol Rev, 2014; 27(4); 980-1024
26. Lizarazo J, Escandon P, Agudelo CI: PLoS Negl Trop Dis, 2014; 8(11); e3272
27. Krajden S, Summerbell RC, Kane J: J Clin Microbiol, 1991; 29(9); 1883-87
28. Stone NR, Rhodes J, Fisher MC, Dynamic ploidy changes drive fluconazole resistance in human cryptococcal meningitis: J Clin Invest, 2019; 129(3); 999-1014
29. Musubire AK, Boulware DR, Meya DB, Diagnosis and management of cryptococcal relapse: J AIDS Clin Res, 2013; Suppl 3(3); 003
30. Jhamb R, Kashyap B, Das S: Int J STD AIDS, 2014; 25(5); 369-72
31. Bongomin F, Oladele RO, Gago S: Mycoses, 2018; 61(5); 290-97
32. Worasilchai N, Tangwattanachuleeporn M, Meesilpavikkai K: Med Mycol, 2017; 55(6); 680-85
33. Chen YH, Yu F, Bian ZY: Sci Rep, 2018; 8(1); 1495
34. Jarvis JN, Meintjes G, Williams Z, Symptomatic relapse of HIV-associated cryptococcal meningitis in South Africa: The role of inadequate secondary prophylaxis: S Afr Med J, 2010; 100(6); 378-82
35. Dou H, Wang H, Xie S: Med Mycol, 2017; 55(7); 737-47
36. Wu SY, Lei Y, Kang M: Mycoses, 2015; 58(5); 280-87
37. Rocha DFS, Cruz KS, Santos C: PLoS One, 2018; 13(6); e0197841
38. Cogliati M, Zani A, Rickerts V: Fungal Genet Biol, 2016; 87; 22-29
Figures
 Figure 1. Flowchart showing the flow of patients through the study.
Figure 1. Flowchart showing the flow of patients through the study. Figure 2. Distribution of patients with recurrence by month after discharge. The patients with recurrence were divided into 2 groups according to total medication time. Of the patients, 21 had total medication times >75 d and 11 had total medication times <75 d.
Figure 2. Distribution of patients with recurrence by month after discharge. The patients with recurrence were divided into 2 groups according to total medication time. Of the patients, 21 had total medication times >75 d and 11 had total medication times <75 d. Figure 3. Maximum likelihood phylogenetic analysis of Cryptococcus neoformans.
Figure 3. Maximum likelihood phylogenetic analysis of Cryptococcus neoformans. Tables
 Table 1. Demographic and clinical characteristics of 69 cryptococcal meningitis patients with first onset or recurrence.
Table 1. Demographic and clinical characteristics of 69 cryptococcal meningitis patients with first onset or recurrence. Table 2. Univariate analysis of risk factors for recurrence in cryptococcal meningitis patients.
Table 2. Univariate analysis of risk factors for recurrence in cryptococcal meningitis patients. Table 3. Multivariate analysis of risk factors for recurrence in patients with cryptococcal meningitis.
Table 3. Multivariate analysis of risk factors for recurrence in patients with cryptococcal meningitis. Table 4. Antifungal drug susceptibility results (mg/L).
Table 4. Antifungal drug susceptibility results (mg/L). Table 1. Demographic and clinical characteristics of 69 cryptococcal meningitis patients with first onset or recurrence.
Table 1. Demographic and clinical characteristics of 69 cryptococcal meningitis patients with first onset or recurrence. Table 2. Univariate analysis of risk factors for recurrence in cryptococcal meningitis patients.
Table 2. Univariate analysis of risk factors for recurrence in cryptococcal meningitis patients. Table 3. Multivariate analysis of risk factors for recurrence in patients with cryptococcal meningitis.
Table 3. Multivariate analysis of risk factors for recurrence in patients with cryptococcal meningitis. Table 4. Antifungal drug susceptibility results (mg/L).
Table 4. Antifungal drug susceptibility results (mg/L). In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








