09 December 2021: Database Analysis
Low Expression of Long Noncoding RNA SLC26A4 Antisense RNA 1 Is an Independent Prognostic Biomarker and Correlate of Immune Infiltrates in Breast Cancer
Weiwei Yi1ABCEF, Haiqing Shen2BC, Dexi Sun3BC, Yongchao Xu4BC, Yujie Feng1BC, Dongbing Li5BC, Caixia Wang1ABCEF*DOI: 10.12659/MSM.934522
Med Sci Monit 2021; 27:e934522
Abstract
BACKGROUND: Aberrant expression of long noncoding RNA (lncRNA) SLC26A4 antisense RNA 1 (SLC26A4-AS1) plays an important role in some cancer types. However, the clinical significance of SLC26A4-AS1 in patients with breast cancer (BC) and the possible regulatory mechanisms of SLC26A4-AS1 are unclear.
MATERIAL AND METHODS: Statistical analysis was used to assess the correlation between SLC26A4-AS1 expression and patients’ clinical characteristics. The Kaplan-Meier method and Cox regression analysis were used to assess the correlation between SLC26A4-AS1 expression and prognosis. Gene set enrichment analysis (GSEA) and immuno-infiltration analysis were used to investigate the possible regulatory mechanisms of SLC26A4-AS1.
RESULTS: Low SLC26A4-AS1 expression in BC was associated with age (P<0.001), estrogen-receptor status (P<0.001), PAM50 (P<0.001), and menopause status (P<0.001). Low SLC26A4-AS1 expression predicted a poorer overall survival (OS) (hazard ratio [HR]: 0.56; 95% confidence interval [CI]: 0.40-0.78; P=0.001) and disease-specific survival (DSS) (HR: 0.57; 95% CI: 0.37-0.88; P=0.011). Also, SLC26A4-AS1 expression (HR: 0.298; 95% CI: 0.154-0.579; P<0.001) was independently correlated with OS in patients with BC. SLC26A4-AS1 was related to CYP2E1 reactions, protein export, mitochondrial_ciii_assembly, formation of adenosine triphosphate by chemiosmotic coupling, budding and maturation of HIV virion, cristae formation, biocarta proteasome pathway, endosomal sorting complex required for transport, and histone modification. SLC26A4-AS1 expression was associated with some types of immune infiltrating cells.
CONCLUSIONS: SLC26A4-AS1 expression was significantly associated with poor survival and immune infiltration in patients with BC. It may be a promising prognostic biomarker for BC.
Keywords: Biomarkers, Tumor, Breast Neoplasms, Female, Humans, RNA, Antisense, Sulfate Transporters
Background
Breast cancer (BC) is the second leading cause of cancer death in women [1] with 2.1 million new cases and 627 000 deaths yearly, making it the most common malignancy and the second leading cause of cancer-related deaths in women [2]. Although significant progress has been made in the personalized treatment of BC, the 5-year overall survival (OS) rate remains relatively low due to the heterogeneity of tumors [3]. Although screening for BC, such as through mammograms and serum tumor markers, is becoming increasingly popular, limited health service infrastructure is still a problem in some areas, hindering early diagnosis [4]. Therefore, new prognostic biomarkers of BC and their molecular mechanisms need to be further explored.
Long noncoding RNAs (lncRNAs), RNA molecules with transcripts longer than 200 nucleotides, have limited coding potential [5]. They are emerging as a new class of important regulators of cell biological behavior and are indispensable players involved in tumorigenesis, progression, and metastasis [6]. For easy access and detection, lncRNA can be encapsulated in membrane vesicles, such as exosomes and apoptotic bodies, or stably bound to specific proteins and released into body fluids, such as serum and urine [7]. The aberrant expression of lncRNAs is associated with the development and progression of BC [8–10]. Therefore, screening for the lncRNAs associated with BC prognosis is important for patients with BC.
The lncRNA SLC26A4-AS1 inhibits the ability of pancreatic cancer to migrate, invade, and metastasize [11]. Overexpression of SLC26A4-AS1 inhibits the aggressiveness of glioma cells and their pro-angiogenic capacity [12]. Downregulated SLC26A4-AS1 is significantly associated with lower OS in patients with BC [13]. The upregulation of SLC26A4-AS1 was associated with its promoter hypermethylation in BC [14]. However, the clinical significance of SLC26A4-AS1 in patients with breast cancer and the possible regulatory mechanisms of SLC26A4-AS1 are unclear.
The tumor microenvironment (TME) includes cellular and non-cellular components [15]. Inflammatory cells, including neutrophils and myeloid-derived suppressor cells, suppress the beneficial immune function of TME, preventing normal immune cells from attacking tumor cells and promoting tumor growth [16]. Infiltration of the TME by immune cells is a strategy used by tumor cells to evade immune-mediated killing [17]. The BC TME is rich in immune infiltrates with different functions [18]. However, the role of SLC26A4-AS1 in BC tumor immunity is unclear.
In this study, the clinical significance of SLC26A4-AS1 was analyzed based on data from The Cancer Genome Atlas (TCGA) database, and the possible regulatory network of SLC26A4-AS1 was investigated based on gene set enrichment analysis (GSEA) and immune infiltration analysis. This study may provide promising prognostic biomarkers for patients with BC.
Material and Methods
DIFFERENCES IN SLC26A4-AS2 EXPRESSION:
For molecule SLC26A4-AS1 [ENSG00000227199], the following analyses were conducted as described in the literature: patient baseline information [19]; unpaired samples [20]; paired samples [20]; and receiver operating characteristics analysis [19].
CORRELATION BETWEEN SLC26A4-AS2 AND CLINICAL CHARACTERISTICS:
Analysis of clinical relevance for molecule SLC26A4-AS1 was performed as described in the literature [20]. The clinical variables included were age, progesterone-receptor (PR) status, estrogen-receptor (ER) status, PAM50, and menopause status. RNAseq data and clinical data in level 3 HTSeq-FPKM format were from The Cancer Genome Atlas Breast Invasive Carcinoma (TCGA-BRCA) project.
With SLC26A4-AS1 as the dependent variable, logistics analysis was performed as described in the literature [20].
CORRELATION BETWEEN SLC26A4-AS2 AND PROGNOSIS:
Kaplan-Meier curve was performed for SLC26A4-AS1 according to data analysis methods described in the literature [19,21]. Prognosis included OS and disease-specific survival (DSS).
Cox regression analysis was performed as described in the literature [20,21] and included the variables tumor (T) stage, lymph node (N) stage, pathologic stage, race, age, histological type, PR status, ER status, human epidermal growth factor receptor 2 (HER2) status, PAM50, menopause status, anatomic neoplasm subdivisions, radiation therapy, and SLC26A4-AS1.
GSEA ANALYSIS:
Single-gene differential analysis of SLC26A4-AS1 was performed as described in the literature [20,22]. GSEA analysis was performed as described in the literature [20,23,24].
IMMUNE INFILTRATION ANALYSIS BY SINGLE-SAMPLE GSEA:
Data analysis by single-sample GSEA of SLC26A4-AS1 was performed by methods described in the literature [20,25,26].
Results
CLINICAL CHARACTERISTICS:
A total of 1083 patients were included in the present study (Table 1). The T stages of the patients were as follows: 277 were T1 (25.6%), 629 were T2 (58.2%), 139 were T3 (12.9%), and 35 were T4 (3.2%). The N stage of the patients were as follows: 514 were N0 (48.3%), 358 were N1 (33.6%), and were 116 N2 (10.9%). There were 902 patients with metastasis (M) stage 0 (97.8%) and 20 with stage M1 (2.2%). The pathologic stages were as follows: 181 in stage I (17.1%), 619 in stage II (58.4%), 242 in stage III (22.8%), and 18 in stage IV (1.7%). Regarding race, 753 patients were White, 60 patients were Asian, and 181 patients were Black or African American. The age range was 48.5 to 67 years, with a median of 58 years; 601 patients were ≤60 years (55.5%) and 482 patients were >60 years (44.5%). Histological types were 772 infiltrating ductal carcinoma (79%) and 205 infiltrating lobular carcinoma (21%). PR status was negative in 342 (33.1%) patients, indeterminate in 4 (0.4%) patients, and positive in 688 (66.5%) patients. ER status was negative in 240 (23.2%) patients, indeterminate in 2 (0.2%) patients, and positive in 793 (76.6%) patients. HER2 status was negative in 558 (76.8%) patients, indeterminate in 12 (1.7%) patients, and positive in 157 (21.6%) patients. The PAM50 was normal in 40 (3.7%) patients, luminal A in 562 (51.9%) patients, luminal B in 204 (18.8%) patients, HER2 in 82 (7.6%) patients, and basal in 195 (18%) patients. A total of 229 (23.6%) patients were in pre-menopause, 40 (4.1%) were in peri-menopause, and 703 (72.3%) were in menopause. The anatomic neoplasm subdivisions were 563 left (52%) and 520 right (48%). A total of 553 (56%) patients underwent radiation therapy and 434 (44%) did not.
LOW EXPRESSION OF SLC26A4-AS1 PREDICTED POOR CLINICAL CHARACTERISTICS IN PATIENTS WITH BC:
SLC26A4-AS1 was found to be significantly less expressed in BC tissues than in normal breast tissues (0.267±0.015 vs 0.808±0.062, P<0.001), based on the TCGA database of 1109 BC tissues and 113 normal breast tissues (Figure 1A). The expression of SLC26A4-AS1 in 113 BC tissues and their matched normal breast tissues showed that BC tissues had a low expression of SLC26A4-AS1 (0.231±0.036 vs 0.808±0.062, P<0.001, Figure 1B). The area under curve (AUC) of SLC26A4-AS1 was 0.805 (Figure 1C). SLC26A4-AS1 could be used to differentiate between BC and normal breast tissue. SLC26A4-AS1 expression was associated with age (P<0.001), PR status (P<0.001), ER status (P<0.001), PAM50 (P<0.001), and menopause status (P=0.002) (Table 2). SLC26A4-AS1 was significantly related to age (P<0.001), ER status (P<0.001), PAM50 (P<0.001), and menopause status (P<0.001) (Figure 2, Table 3).
CORRELATION BETWEEN SLC26A4-AS1 EXPRESSION AND PATIENT PROGNOSIS:
SLC26A4-AS1 expression was significantly associated with poor OS (hazard ratio [HR]: 0.56; 95% confidence interval [CI]: 0.40–0.78; P=0.001) and DSS (HR: 0.57; 95% CI: 0.37–0.88; P=0.011) of patients with BC (Figure 3). Low SLC26A4-AS1 expression (HR: 0.560; 95% CI: 0.403–0.777; P<0.001) was associated with T stage (HR: 1.482; 95% CI: 1.007–2.182; P=0.046), N stage (HR: 2.239; 95% CI: 1.567–3.199; P<0.001), pathologic stage (HR: 2.391; 95% CI: 1.703–3.355; P<0.001), age (HR: 2.020; 95% CI: 1.465–2.784; P<0.001), menopause status (HR: 0.426; 95% CI: 0.259–0.700; P<0.001), and radiation therapy (HR: 0.576; 95% CI: 0.394–0.841; P=0.004) (Table 4). SLC26A4-AS1 expression (HR: 0.298; 95% CI: 0.154–0.579; P<0.001), pathologic stage (HR: 5.876; 95% CI: 2.271–15.205, P<0.001), and radiation therapy (HR: 0.448; 95% CI: 0.243–0.823, P=0.004) were independent prognostic factors for patients with BC (Table 4).
SLC26A4-AS1-ASSOCIATED PATHWAY:
A total of 124 datasets showed significant differential enrichment in SLC26A4-AS1 high-expression phenotypes, while 50 datasets showed significant differential enrichment in SLC26A4-AS1 low-expression phenotypes. The top 9 data sets with a low P value included CYP2E1 reactions, protein export, mitochondrial_ciii_assembly, formation of adenosine triphosphate (ATP) by chemiosmotic coupling, histone modification, budding and maturation of HIV virion, cristae formation, biocarta proteasome pathway, and endosomal sorting complex required for transport (Table 5, Figure 4).
CORRELATION OF SLC26A4-AS1 EXPRESSION WITH IMMUNE INFILTRATION:
For aDC, the mean level of the SLC26A4-AS1 low-expression group (0.284±0.109) was significantly lower than that of the high-expression group (0.314±0.126) (P<0.001) (Figure 5A). The results (r=0.150, P<0.001) indicated a positive correlation between SLC26A4-AS1 and aDC (Figures 6A and 7). For B cells, the mean level of the SLC26A4-AS1 low-expression group (0.167±0.084) was significantly lower than that of the high-expression group (0.199±0.091, P<0.001) (Figure 5B). The results (r=0.220, P<0.001) indicated a positive correlation between SLC26A4-AS1 and B cells (Figures 6B and 7). For CD8 T cells, the mean level of the SLC26A4-AS1 low-expression group (0.522±0.018) was significantly lower than that of the high-expression group (0.523±0.023) (P=0.003) (Figure 5C). The results (r=0.067, P=0.027) indicated a positive correlation between SLC26A4-AS1 and CD8 T cells (Figures 6C and 7). For cytotoxic cells, the mean level of the SLC26A4-AS1 low-expression group (0.319±0.083) was significantly lower than that of the high-expression group (0.337±0.09) (P<0.001) (Figure 5D). The results (r=0.130, P<0.001) indicated a positive correlation between SLC26A4-AS1 and CD8 T cells (Figures 6D and 7). For DC, the mean level of the SLC26A4-AS1 low-expression group (0.281±0.089) was significantly lower than that of the high-expression group (0.303±0.098) (P<0.001) (Figure 5E). The results (r=0.160, P<0.001) indicated a positive correlation between SLC26A4-AS1 and CD8 T cells (Figures 6E and 7). For macrophages, the mean level of the SLC26A4-AS1 low-expression group (0.445±0.054) was significantly lower than that of the high-expression group (0.456±0.052) (P<0.001) (Figure 5F). The results (r=0.140, P<0.001) indicated a positive correlation between SLC26A4-AS1 and macrophages (Figures 6F and 7). For neutrophils, the mean level of the SLC26A4-AS1 low-expression group (0.231±0.045) was significantly lower than that of the high-expression group (0.239±0.053) (P=0.005) (Figure 5G). The results (r=0.130, P<0.001) indicated a positive correlation between SLC26A4-AS1 and neutrophils (Figures 6G and 7). For NK CD56bright cells, the mean level of the SLC26A4-AS1 low-expression group (0.348±0.050) was significantly higher than that of the high-expression group (0.331±0.056) (P=0.005) (Figure 5H). The results (r=−0.170, P<0.001) indicated a negative correlation between SLC26A4-AS1 and NK CD56bright cells (Figures 6H and 7). For NK CD56dim cells, the mean level of the SLC26A4-AS1 low-expression group (0.180±0.061) was significantly lower than that of the high-expression group (0.193±0.072) (P<0.001) (Figure 5I). The results (r=0.130, P<0.001) indicated a positive correlation between SLC26A4-AS1 and NK CD56dim cells (Figures 6I and 7). For T cells, the mean level of the SLC26A4-AS1 low-expression group (0.312±0.110) was significantly lower than that of the high-expression group (0.339±0.116) (P<0.001) (Figure 5J). The results (r=0.160, P<0.001) indicated a positive correlation between SLC26A4-AS1 and T cells (Figures 6J and 7). For T helper cells, the mean level of the SLC26A4-AS1 low-expression group (0.554±0.023) was significantly lower than that of the high-expression group (0.559±0.023) (P<0.001) (Figure 5K). The results (r=0.120, P<0.001) indicated a positive correlation between SLC26A4-AS1 and T helper cells (Figures 6K and 7). For Tcm, the mean level of the SLC26A4-AS1 low-expression group (0.356±0.028) was significantly lower than that of the high-expression group (0.364±0.031) (P<0.001) (Figure 5L). The results (r=0.150, P<0.001) indicated a positive correlation between SLC26A4-AS1 and Tcm (Figures 6L and 7). For Tem, the mean level of the SLC26A4-AS1 low-expression group (0.362±0.026, P=0.008) was significantly lower than the mean level of the high-expression group (0.366±0.029, P=0.008) (P=0.008) (Figure 5M). The results (r=0.100, P=0.001) indicated a positive correlation between SLC26A4-AS1 and Tem (Figures 6M and 7). For TFH, the mean level of the SLC26A4-AS1 low-expression group (0.345±0.029) was significantly lower than that of the high-expression group (0.350±0.033) (P<0.001) (Figure 5N). The results (r=0.120, P<0.001) indicated a positive correlation between SLC26A4-AS1 and TFH (Figures 6N and 7). For Th1 cells, the mean level of the SLC26A4-AS1 low-expression group (0.304±0.048) was significantly lower than that of the high-expression group (0.317±0.05) (P<0.001) (Figure 5O). The results (r=0.180, P<0.001) indicated a positive correlation between SLC26A4-AS1 and Th1 cells (Figures 6O and 7).
Discussion
A growing number of studies have shown that lncRNAs play an important role in tumorigenesis and development [4]. The lncRNA MALAT1 is used as a potential indicator for early diagnosis and prognosis prediction in patients with BC [8]. The serum lncRNA ATB is a noninvasive diagnostic marker in the early stages of BC [9]. INC00978 is an oncogene in BC and can be used as a potential prognosis biomarker for patients with BC [10]. Low expression of LINC00944 is associated with poor prognosis in patients with BC [27]. Therefore, it is crucial to explore promising novel lncRNAs as prognostic biomarkers for BC.
The low expression of SLC26A4-AS1 in thyroid cancer was significantly associated with poor patient prognosis [11]. SLC26A4-AS1 is downregulated in human glioma tissues and cells [12]. In the present study, low expression of SLC26A4-AS1 in patients with BC was correlated with age (
SLC26A4-AS1 interacts with DDX5 and the E3 ligase TRIM25 to promote DDX5 degradation via the ubiquitin-proteasome pathway [11]. SLC26A4-AS1 is downregulated in human glioma tissues and cells [12]. SLC26A4-AS1 promotes the transcriptional activity of NPTX1 through the recruitment of NFKB1, thereby exerting an anti-angiogenic effect on glioma cells [12]. Overexpression of lncRNA SLC26A4-AS1 exerts anticancer effects on papillary thyroid carcinoma through inactivation of the MAPK pathway [28]. In the present study, SLC26A4-AS1 was related to the following pathways: CYP2E1 reactions, protein export, mitochondrial_ciii_assembly, formation of ATP by chemiosmotic coupling, histone modification, budding and maturation of HIV virion, cristae formation, biocarta proteasome pathway, and endosomal sorting complex required for transport.
The TME, consisting of tumor-infiltrating lymphocytes and other immune cells, is an important part of tumor development [29,30]. The number and proportion of mesenchymal and immune cells in tumor tissue are associated with clinical features and prognosis [31]. Screening valuable genes from immune and stromal cells in tumor tissues may be a target for cancer therapy [32]. Therefore, there is a need to gain insight into the significance of genes associated with the TME for accurate assessment and treatment. The present study investigated the relationship between SLC26A4-AS1 expression and the level of multiple immune infiltrations in BC. The results showed modest correlations between SLC26A4-AS1 expression and infiltration levels of aDC, B cells, CD8 T cells, cytotoxic cells, DC, eosinophils, macrophages, neutrophils, NK CD56bright cells, NK CD56dim cells, T cells, T helper cells, Tcm, Tem, TFH, Tgd, Th1 cells, and TReg in BC. The present findings suggest that SLC26A4-AS1 may mediate the development and progression of BC by negatively regulating eosinophils and NK CD56bright cells and positively regulating aDC, B cells, CD8 T cells, cytotoxic cells, DCs, macrophages, neutrophils, NK CD56dim cells, T cells, T helper cells, Tcm, Tem, TFH, and Th1 cells.
This study is the first to examine the relationship between SLC26A4-AS1 and BC. However, there are some limitations. This study was conducted based on the TCGA database and bioinformatics analysis, but the specific mechanism by which SLC26A4-AS1 mediates the development and progression of BC could not be clarified. In the real world setting, the clinical significance and specific mechanisms of SLC26A4-AS1 in patients with BC need to be further investigated.
Conclusions
SLC26A4-AS1 expression was significantly lower in BC tissue than in normal breast tissue. Low expression of SLC26A4-AS1 was associated with poor OS and DSS in patients with BC. SLC26A4-AS1 may be related to the following pathways: CYP2E1 reactions, protein export, mitochondrial_ciii_assembly, formation of ATP by chemiosmotic coupling, histone modification, budding and maturation of HIV virion, cristae formation, biocarta proteasome pathway, and endosomal sorting complex required for transport. SLC26A4-AS1 expression was associated with several immune infiltrating cells. This study provides a promising prognostic biomarker for patients with BC.
Figures
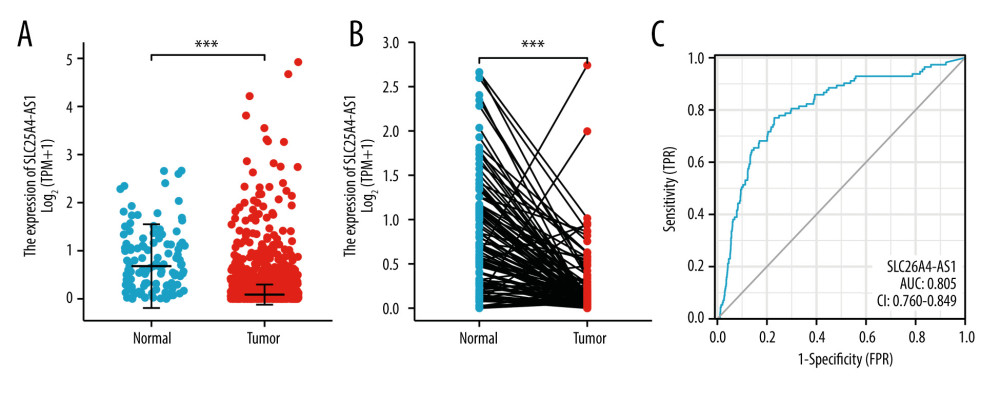 Figure 1. Expression of SLC26A4-AS1 in breast cancer (BC) tissues compared to normal tissues. (A) Differential expression of SLC26A4-AS1 in BC tissues and normal breast tissues. (B) Differential expression of SLC26A4-AS1 in BC tissues and matched normal breast tissues. (C) receiver operating characteristic curves showed the efficiency of SLC26A4-AS1 expression levels in distinguishing BC tissues from non-tumor tissues. Significance markers: *** P<0.001.
Figure 1. Expression of SLC26A4-AS1 in breast cancer (BC) tissues compared to normal tissues. (A) Differential expression of SLC26A4-AS1 in BC tissues and normal breast tissues. (B) Differential expression of SLC26A4-AS1 in BC tissues and matched normal breast tissues. (C) receiver operating characteristic curves showed the efficiency of SLC26A4-AS1 expression levels in distinguishing BC tissues from non-tumor tissues. Significance markers: *** P<0.001. 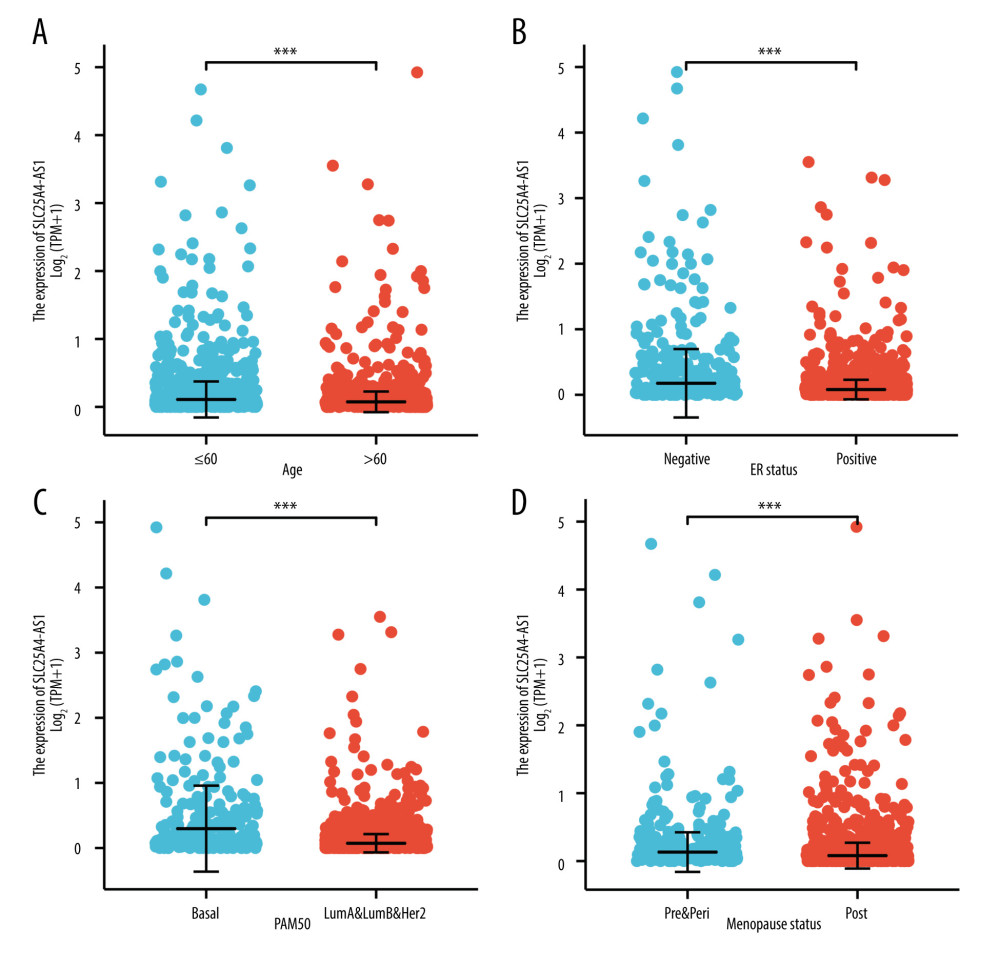 Figure 2. SLC26A4-AS1 expression is associated with the clinical characteristics of patients with breast cancer. (A) Age, (B) estrogen-receptor status, (C) PAM50, (D) menopause. Significance markers: *** P<0.001.
Figure 2. SLC26A4-AS1 expression is associated with the clinical characteristics of patients with breast cancer. (A) Age, (B) estrogen-receptor status, (C) PAM50, (D) menopause. Significance markers: *** P<0.001. 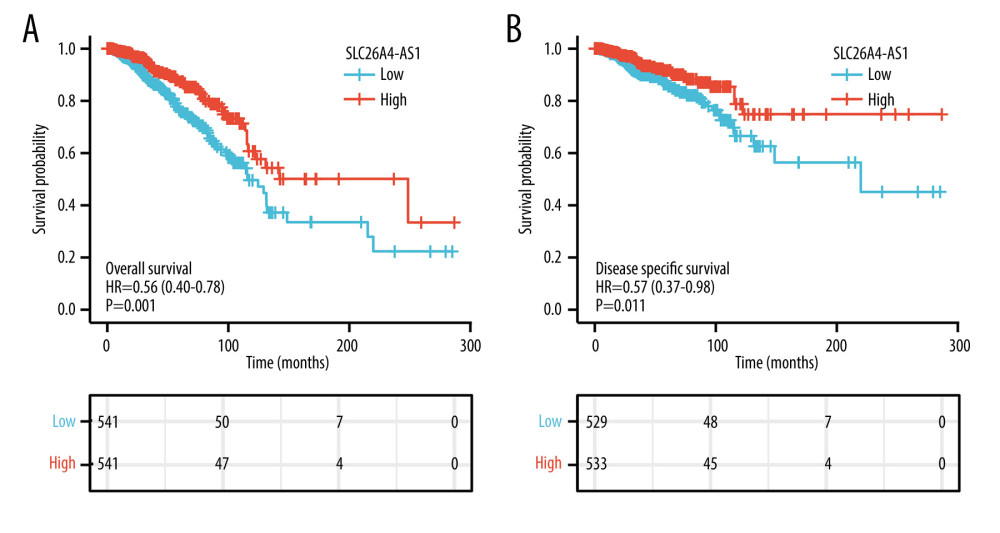 Figure 3. Low expression of SLC26A4-AS1 is associated with poor overall survival and disease-specific survival in patients with breast cancer. (A) Overall survival. (B) Disease-specific survival.
Figure 3. Low expression of SLC26A4-AS1 is associated with poor overall survival and disease-specific survival in patients with breast cancer. (A) Overall survival. (B) Disease-specific survival. 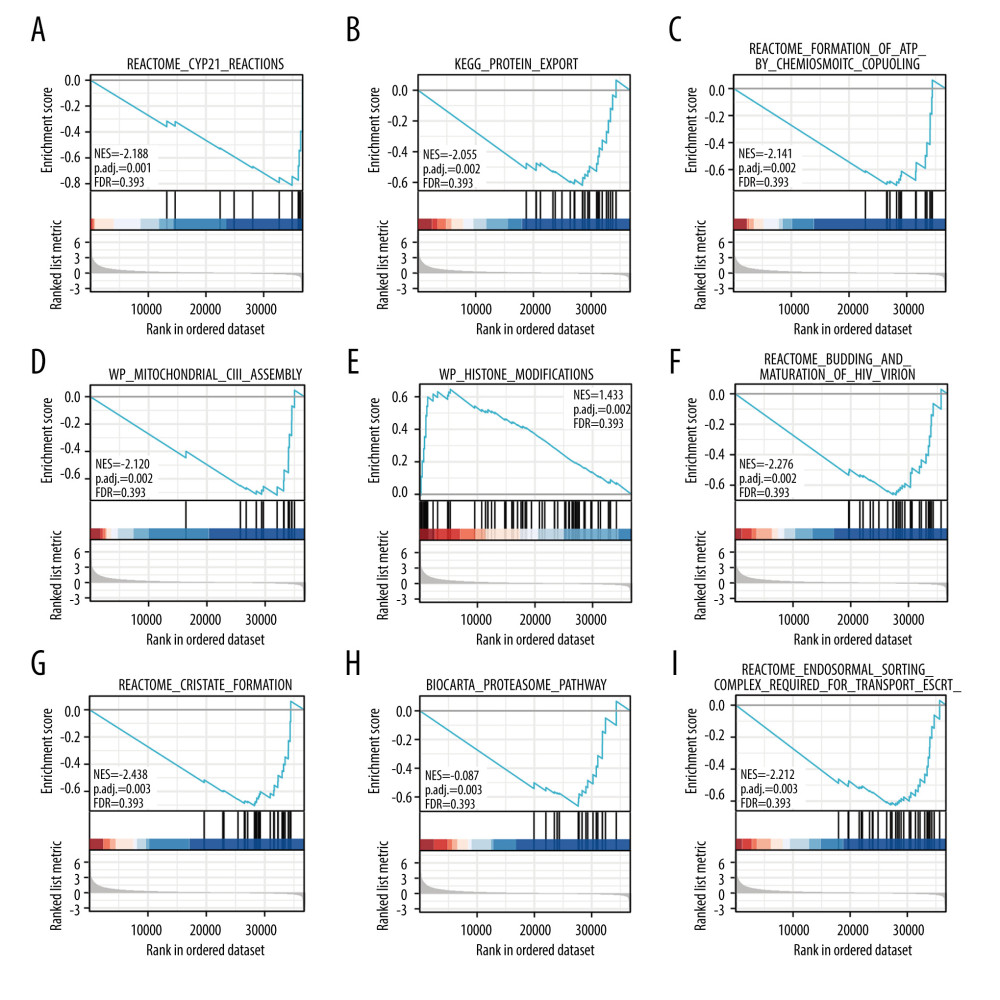 Figure 4. Enrichment plots for gene set enrichment analysis. (A) CYP2E1 reactions, (B) protein export, (C) mitochondrial_ciii_assembly, (D) formation of ATP by chemiosmotic coupling, (E) histone modification, (F) budding and maturation of HIV virion, (G) cristae formation, (H) biocarta proteasome pathway, and (I) endosomal sorting complex required for transport. NES – normalized enrichment score; FDR – false discovery rate
Figure 4. Enrichment plots for gene set enrichment analysis. (A) CYP2E1 reactions, (B) protein export, (C) mitochondrial_ciii_assembly, (D) formation of ATP by chemiosmotic coupling, (E) histone modification, (F) budding and maturation of HIV virion, (G) cristae formation, (H) biocarta proteasome pathway, and (I) endosomal sorting complex required for transport. NES – normalized enrichment score; FDR – false discovery rate 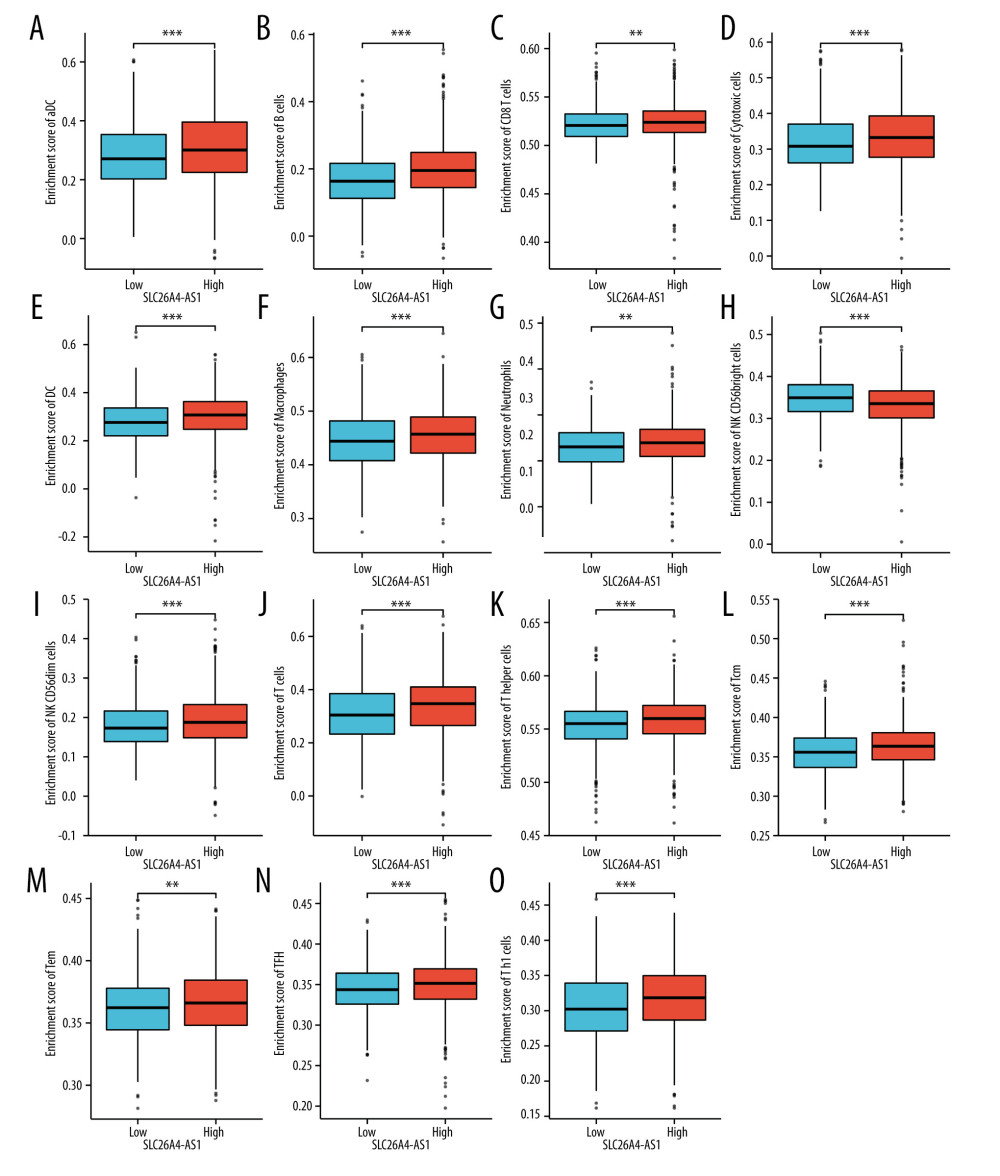 Figure 5. Expression of SLC26A4-AS1 correlated with immune cells in patients with breast cancer (grouped comparison chart). (A) aDC, (B) B cells, (C) CD8 T cells, (D) cytotoxic cells, (E) DC, (F) macrophages, (G) neutrophils, (H) NK CD56bright cells, (I) NK CD56dim cells, (J) T cells, (K) T helper cells, (L) Tcm, (M) Tem, (N) TFH, and (O) Th1 cells.
Figure 5. Expression of SLC26A4-AS1 correlated with immune cells in patients with breast cancer (grouped comparison chart). (A) aDC, (B) B cells, (C) CD8 T cells, (D) cytotoxic cells, (E) DC, (F) macrophages, (G) neutrophils, (H) NK CD56bright cells, (I) NK CD56dim cells, (J) T cells, (K) T helper cells, (L) Tcm, (M) Tem, (N) TFH, and (O) Th1 cells. 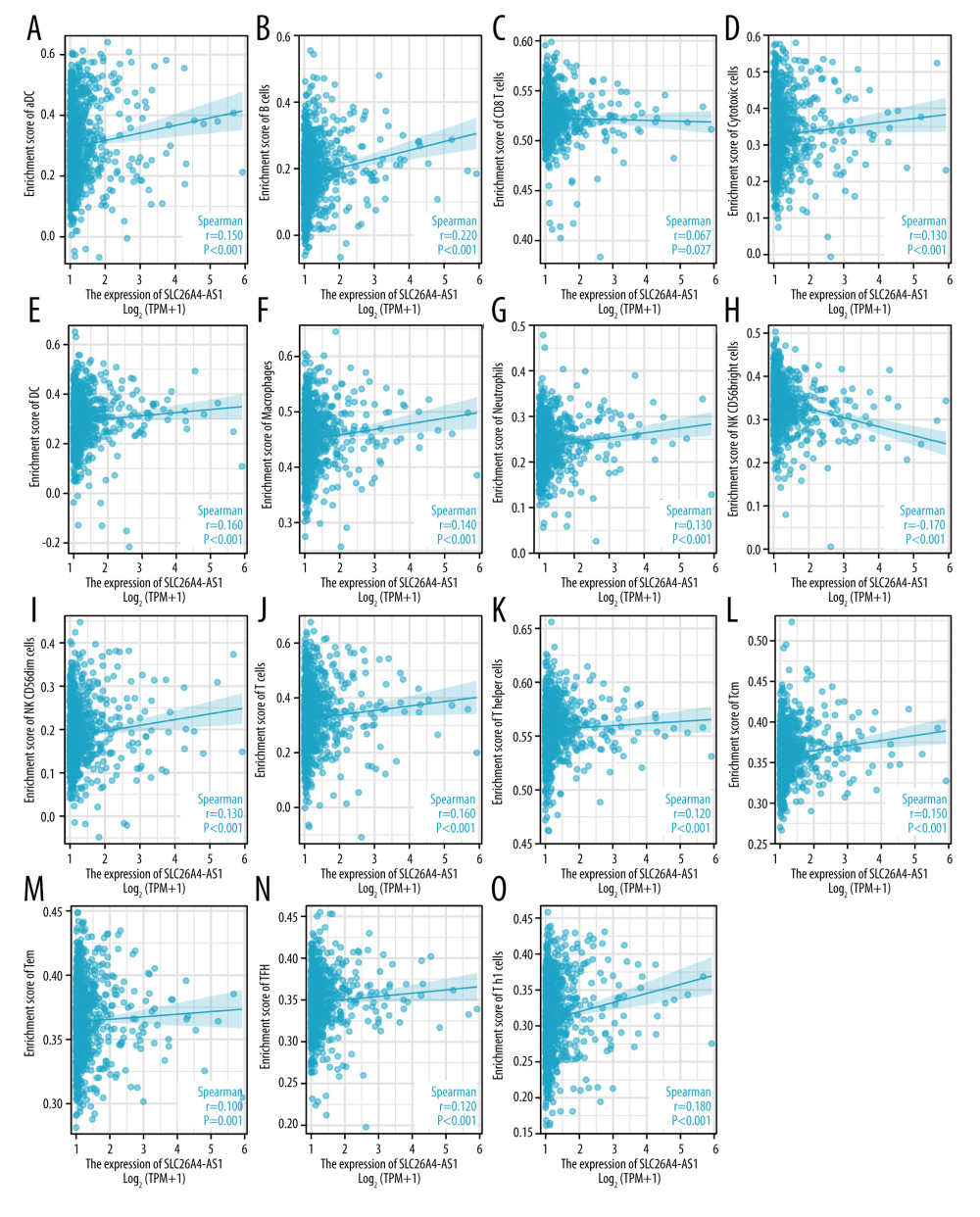 Figure 6. Correlation of SLC26A4-AS1 expression with 24 immune cells in patients with breast cancer (scatter plot). (A) aDC, (B) B cells, (C) CD8 T cells, (D) cytotoxic cells, (E) DC, (F) macrophages, (G) neutrophils, (H) NK CD56bright cells, (I) NK CD56dim cells, (J) T cells, (K) T helper cells, (L) Tcm, (M) Tem, (N) TFH, and (O) Th1 cells.
Figure 6. Correlation of SLC26A4-AS1 expression with 24 immune cells in patients with breast cancer (scatter plot). (A) aDC, (B) B cells, (C) CD8 T cells, (D) cytotoxic cells, (E) DC, (F) macrophages, (G) neutrophils, (H) NK CD56bright cells, (I) NK CD56dim cells, (J) T cells, (K) T helper cells, (L) Tcm, (M) Tem, (N) TFH, and (O) Th1 cells. 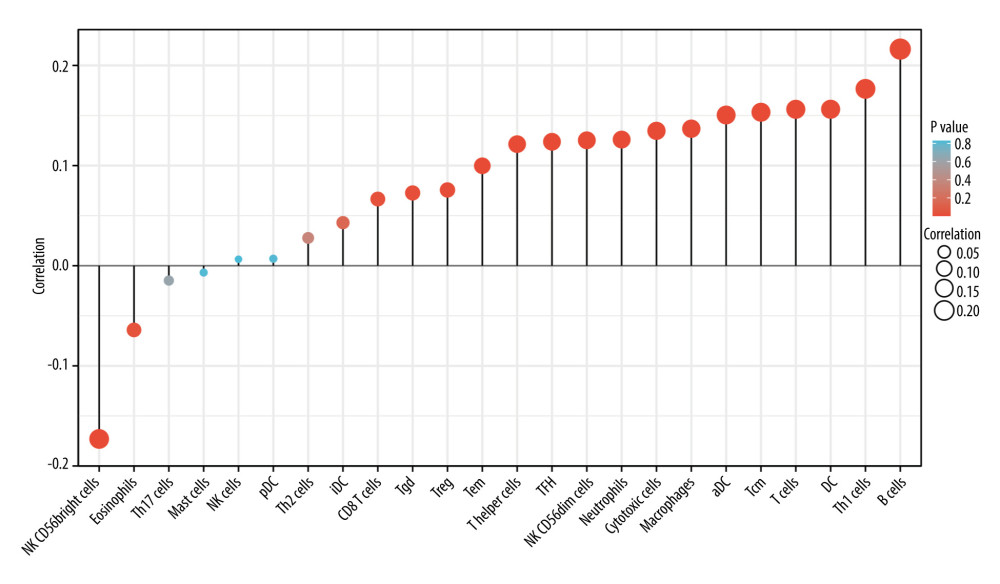 Figure 7. Correlation of SLC26A4-AS1 expression with 24 immune cells in patients with breast cancer (lollipop chart).
Figure 7. Correlation of SLC26A4-AS1 expression with 24 immune cells in patients with breast cancer (lollipop chart). Tables
Table 1. The Cancer Genome Atlas-based characterization of patients with breast cancer.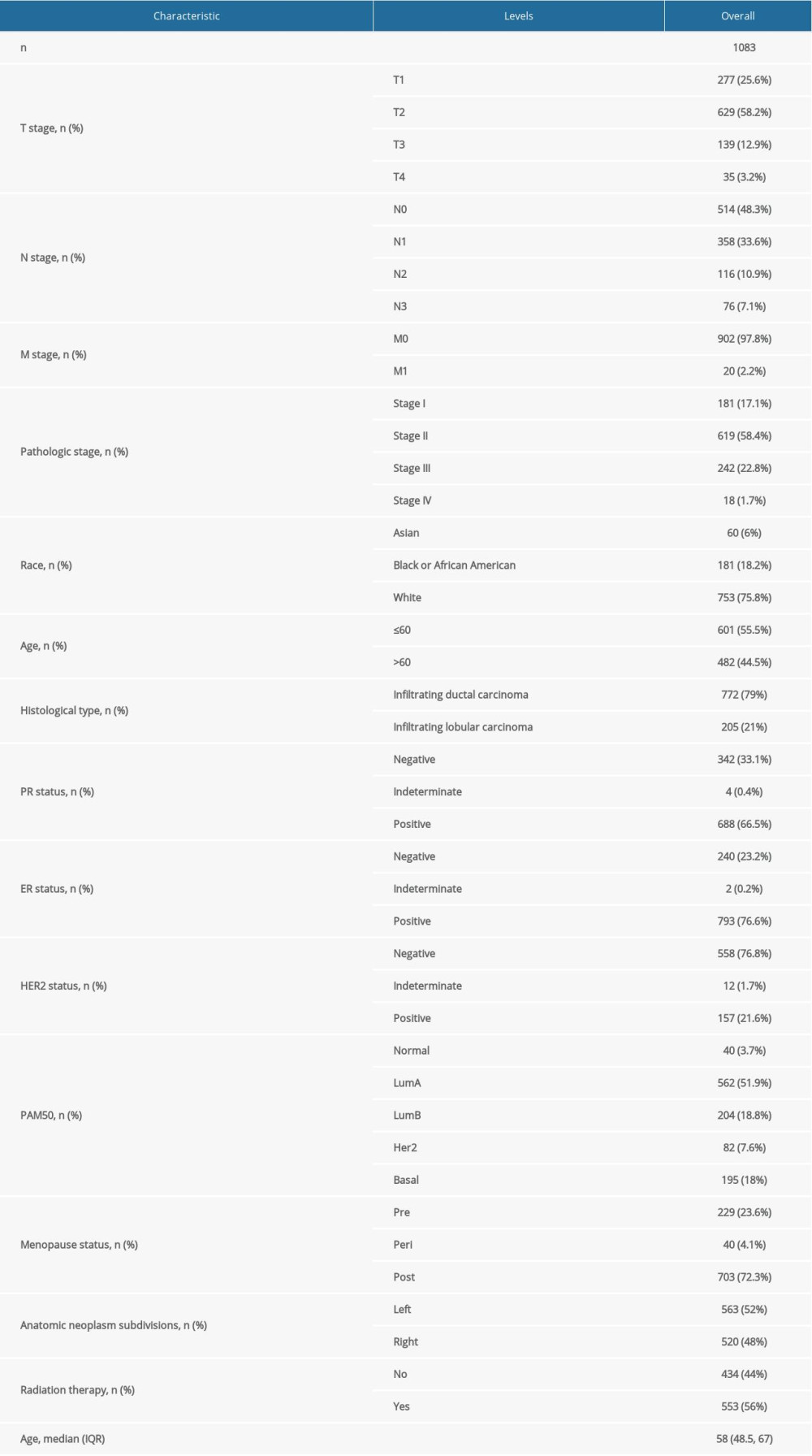 Table 2. Expression of SLC26A4-AS1 in breast cancer is associated with clinical characteristics.
Table 2. Expression of SLC26A4-AS1 in breast cancer is associated with clinical characteristics.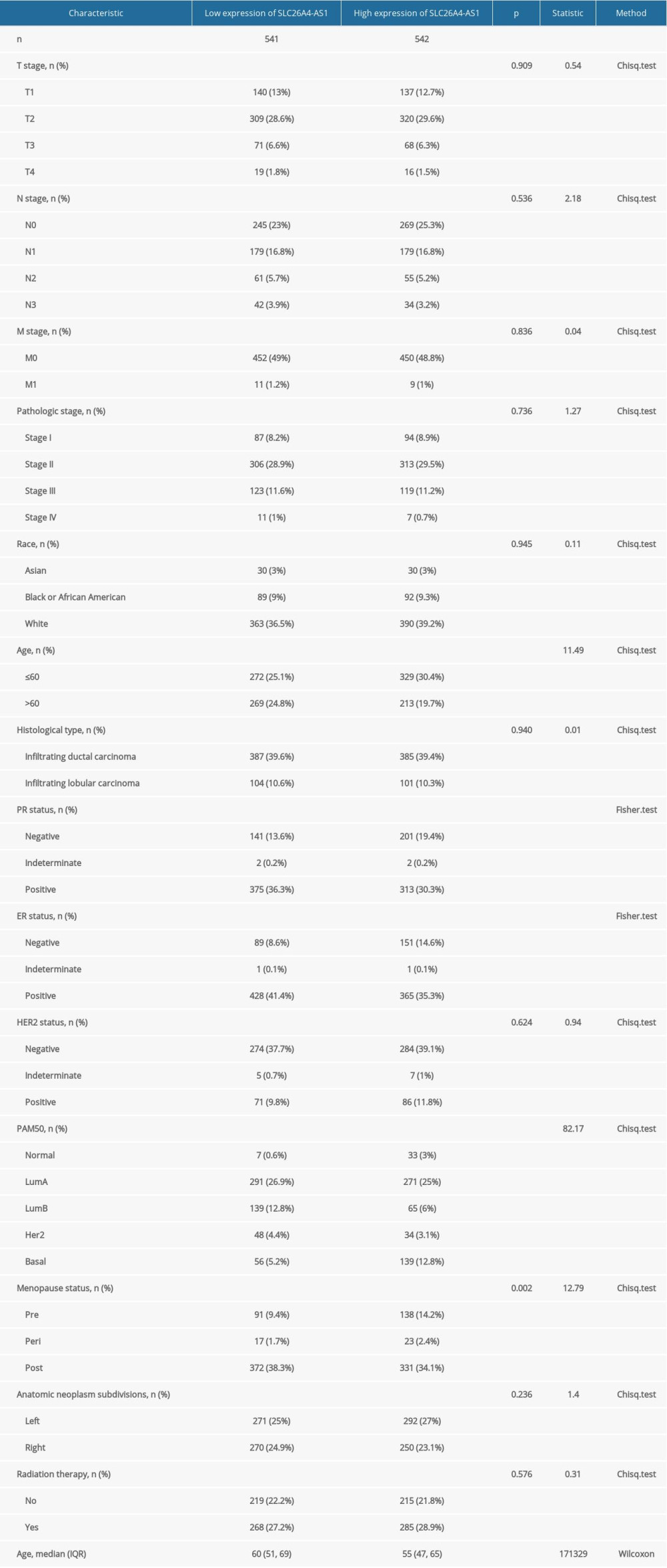 Table 3. Correlation between SLC26A4-AS1 expression and clinical characteristics in patients with breast cancer (logistic regression).
Table 3. Correlation between SLC26A4-AS1 expression and clinical characteristics in patients with breast cancer (logistic regression).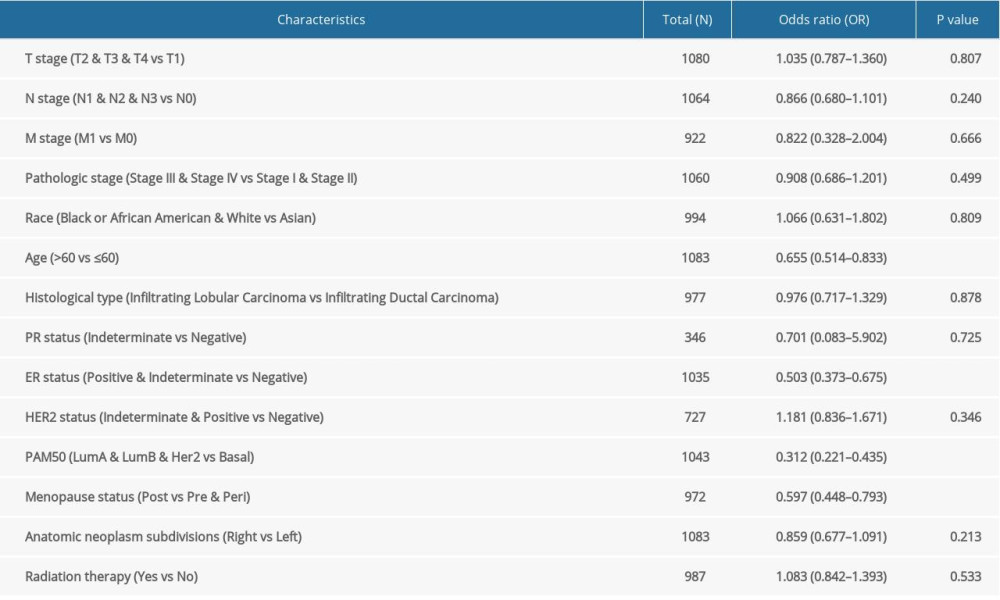 Table 4. Correlation between overall survival and clinical characteristics in patients with breast cancer (Cox regression).
Table 4. Correlation between overall survival and clinical characteristics in patients with breast cancer (Cox regression).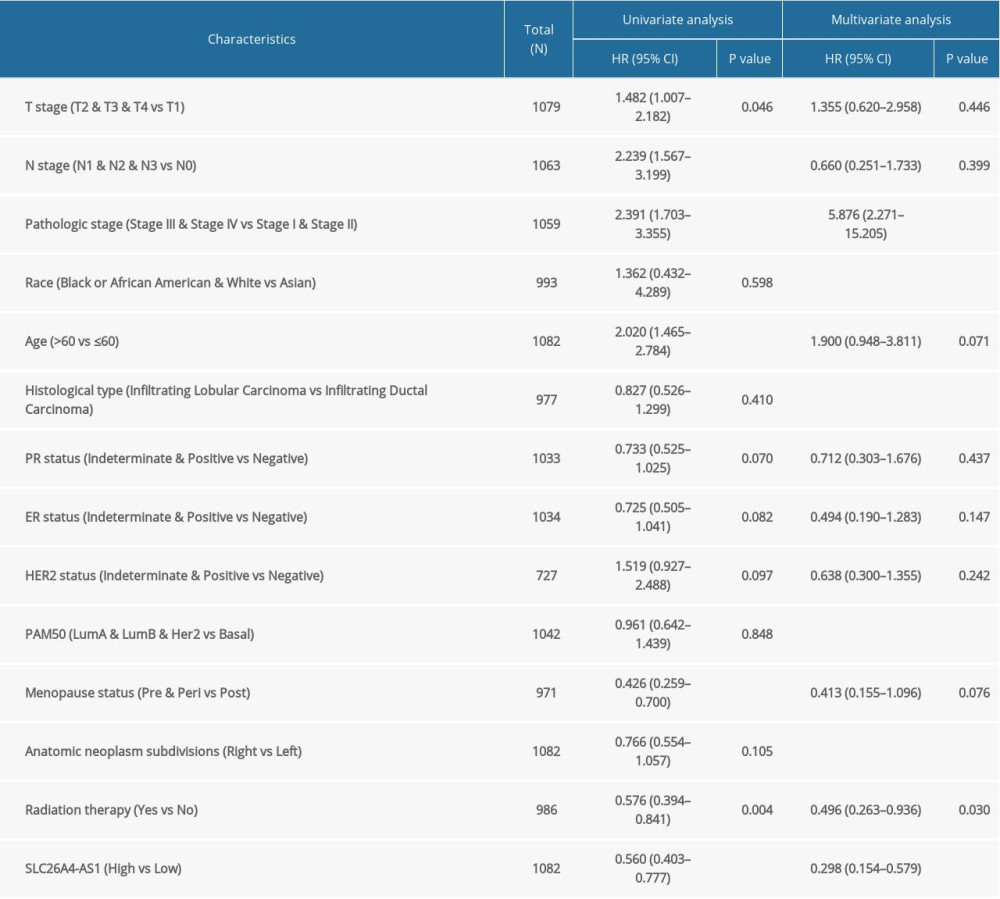 Table 5. Enrichment of SLC26A4-AS1-related pathways (gene set enrichment analysis).
Table 5. Enrichment of SLC26A4-AS1-related pathways (gene set enrichment analysis).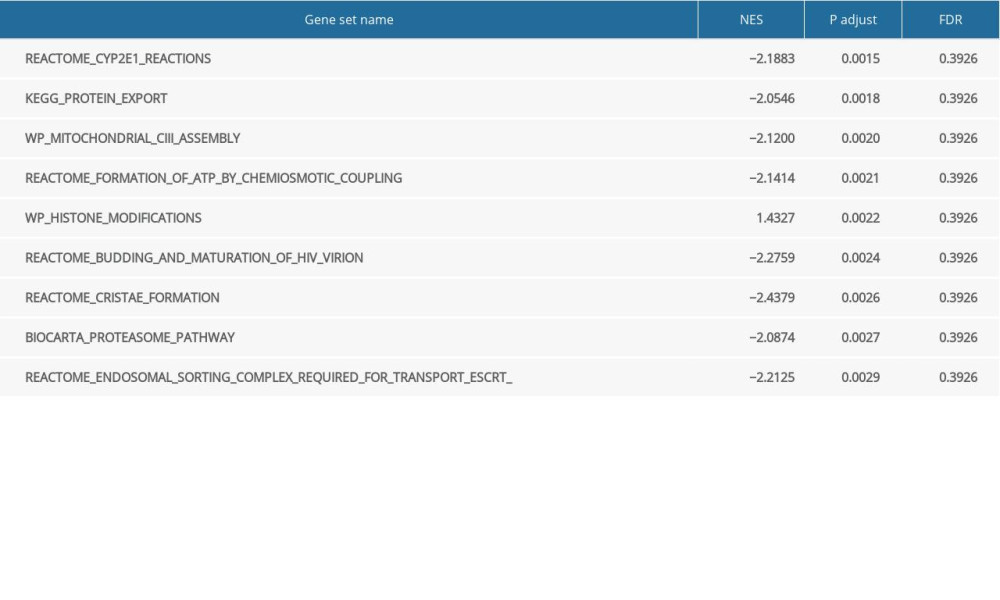
References
1. Zhao E, Lan Y, Quan F, Identification of a six-lncRNA signature with prognostic value for breast cancer patients: Front Genet, 2020; 11; 673
2. Bray F, Ferlay J, Soerjomataram I, Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries: Cancer J Clin, 2018; 68(6); 394-424 [Erratum in: Cancer J Clin. 2020;70(4):313]
3. Li Y, Liang Y, Ma T, Yang Q, Identification of DGUOK-AS1 as a prognostic factor in breast cancer by bioinformatics analysis: Front Oncol, 2020; 10; 1092
4. Zhou T, Lin K, Nie J, LncRNA SPINT1-AS1 promotes breast cancer proliferation and metastasis by sponging let-7 a/b/i-5p: Pathol Res Pract, 2021; 217; 153268
5. Hu RH, Zhang ZT, Wang HX, LncRNA ST7-AS1, by regulating miR-181b-5p/KPNA4 axis, promotes the malignancy of lung adenocarcinoma: Cancer Cell Int, 2020; 20; 568-68
6. Xu M, Chen X, Lin K, The long noncoding RNA SNHG1 regulates colorectal cancer cell growth through interactions with EZH2 and miR-154-5p: Mol Cancer, 2018; 17; 141
7. Shi T, Gao G, Cao Y, Long Noncoding RNAs as novel biomarkers have a promising future in cancer diagnostics: Dis Markers, 2016; 2016; 9085195
8. Sun Z, Liu J, Liu J, The expression of lncRNA-MALAT1 in breast cancer patients and its influences on prognosis: Cell Mol Biol (Noisy-Le-Grand), 2020; 66; 72-78
9. El-Ashmawy NE, Hussien FZ, El-Feky OZ, Serum LncRNA-ATB and FAM83H-AS1 as diagnostic/prognostic non-invasive biomarkers for breast cancer: Life Sci, 2020; 259; 118193
10. Deng LL, Chi YY, Liu L, LINC00978 predicts poor prognosis in breast cancer patients: Sci Rep, 2016; 6; 37936
11. Yuan J, Song Y, Pan W, LncRNA SLC26A4-AS1 suppresses the MRN complex-mediated DNA repair signaling and thyroid cancer metastasis by destabilizing DDX5: Oncogene, 2020; 39; 6664-76
12. Li H, Yan R, Chen W, Long non coding RNA SLC26A4-AS1 exerts antiangiogenic effects in human glioma by upregulating NPTX1 via NFKB1 transcriptional factor: FEBS J, 2021; 288; 212-28
13. Gao S, Lu X, Ma J, Comprehensive analysis of lncRNA and miRNA regulatory network reveals potential prognostic non-coding RNA involved in breast cancer progression: Front Genetic, 2021; 12; 621809
14. Zhao H, Liu X, Yu L, Comprehensive landscape of epigenetic-dysregulated lncRNAs reveals a profound role of enhancers in carcinogenesis in BC subtypes: Mol Ther Nucleic Acids, 2021; 23; 667-81
15. Dysthe M, Parihar R, Myeloid-derived suppressor cells in the tumor microenvironment: Adv Exp Med Biol, 2020; 1224; 117-40
16. Topalian SL, Drake CG, Pardoll DM, Immune checkpoint blockade: A common denominator approach to cancer therapy: Cancer Cell, 2015; 27; 450-61
17. De Palma M, Biziato D, Petrova TV, Microenvironmental regulation of tumour angiogenesis: Nat Rev Cancer, 2017; 17; 457-74
18. Burugu S, Asleh-Aburaya K, Nielsen TO, Immune infiltrates in the breast cancer microenvironment: Detection, characterization and clinical implication: Breast Cancer, 2017; 24; 3-15
19. Lu X, Li G, Liu S, Bioinformatics analysis of KIF1A expression and gene regulation network in ovarian carcinoma: Int J Gen Med, 2021; 14; 3707-17
20. Chen T, Zhu C, Wang X, Pan Y, LncRNA ELF3-AS1 is a prognostic biomarker and correlated with immune infiltrates in hepatocellular carcinoma: Can J Gastroenterol Hepatol, 2021; 2021; 8323487
21. Liu J, Lichtenberg T, Hoadley KA, An integrated TCGA pan-cancer clinical data resource to drive high-quality survival outcome analytics: Cell, 2018; 173; 400-416e411
22. Love MI, Huber W, Anders S, Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2: Genome Biol, 2014; 15; 550
23. Yu G, Wang LG, Han Y, He QY, clusterProfiler: An R package for comparing biological themes among gene clusters: OMICS, 2012; 16; 284-87
24. Subramanian A, Tamayo P, Mootha VK, Mesirov, gene set enrichment analysis: A knowledge-based approach for interpreting genome-wide expression profiles: Proc Natl Acad Sci USA, 2005; 102; 15545-50
25. Hänzelmann S, Castelo R, Guinney J, GSVA: Gene set variation analysis for microarray and RNA-seq data: BMC Bioinformatics, 2013; 14; 7
26. Bindea G, Mlecnik B, Tosolini M, Spatiotemporal dynamics of intratumoral immune cells reveal the immune landscape in human cancer: Immunity, 2013; 39; 782-95
27. de Santiago PR, Blanco A, Morales F, Immune-related IncRNA LINC00944 responds to variations in ADAR1 levels and it is associated with breast cancer prognosis: Life Sci, 2021; 268; 118956
28. Wang DP, Tang XZ, Liang QK, Overexpression of long noncoding RNA SLC26A4-AS1 inhibits the epithelial-mesenchymal transition via the MAPK pathway in papillary thyroid carcinoma: J Cell Physiol, 2020; 235; 2403-13
29. Junttila MR, de Sauvage FJ, Influence of tumour micro-environment heterogeneity on therapeutic response: Nature, 2013; 501; 346-54
30. Wang Z, Song K, Zhao W, Zhao Z, Dendritic cells in tumor microenvironment promoted the neuropathic pain via paracrine inflammatory and growth factors: Bioengineered, 2020; 11; 661-78
31. Zhou ZJ, Xin HY, Li J, Intratumoral plasmacytoid dendritic cells as a poor prognostic factor for hepatocellular carcinoma following curative resection: Cancer Immunol Immunother, 2019; 68; 1223-33
32. Ren H, Hu D, Mao Y, Su X, Identification of genes with prognostic value in the breast cancer microenvironment using bioinformatics analysis: Med Sci Monit, 2020; 26; e920212
Figures
 Figure 1. Expression of SLC26A4-AS1 in breast cancer (BC) tissues compared to normal tissues. (A) Differential expression of SLC26A4-AS1 in BC tissues and normal breast tissues. (B) Differential expression of SLC26A4-AS1 in BC tissues and matched normal breast tissues. (C) receiver operating characteristic curves showed the efficiency of SLC26A4-AS1 expression levels in distinguishing BC tissues from non-tumor tissues. Significance markers: *** P<0.001.
Figure 1. Expression of SLC26A4-AS1 in breast cancer (BC) tissues compared to normal tissues. (A) Differential expression of SLC26A4-AS1 in BC tissues and normal breast tissues. (B) Differential expression of SLC26A4-AS1 in BC tissues and matched normal breast tissues. (C) receiver operating characteristic curves showed the efficiency of SLC26A4-AS1 expression levels in distinguishing BC tissues from non-tumor tissues. Significance markers: *** P<0.001. Figure 2. SLC26A4-AS1 expression is associated with the clinical characteristics of patients with breast cancer. (A) Age, (B) estrogen-receptor status, (C) PAM50, (D) menopause. Significance markers: *** P<0.001.
Figure 2. SLC26A4-AS1 expression is associated with the clinical characteristics of patients with breast cancer. (A) Age, (B) estrogen-receptor status, (C) PAM50, (D) menopause. Significance markers: *** P<0.001. Figure 3. Low expression of SLC26A4-AS1 is associated with poor overall survival and disease-specific survival in patients with breast cancer. (A) Overall survival. (B) Disease-specific survival.
Figure 3. Low expression of SLC26A4-AS1 is associated with poor overall survival and disease-specific survival in patients with breast cancer. (A) Overall survival. (B) Disease-specific survival. Figure 4. Enrichment plots for gene set enrichment analysis. (A) CYP2E1 reactions, (B) protein export, (C) mitochondrial_ciii_assembly, (D) formation of ATP by chemiosmotic coupling, (E) histone modification, (F) budding and maturation of HIV virion, (G) cristae formation, (H) biocarta proteasome pathway, and (I) endosomal sorting complex required for transport. NES – normalized enrichment score; FDR – false discovery rate
Figure 4. Enrichment plots for gene set enrichment analysis. (A) CYP2E1 reactions, (B) protein export, (C) mitochondrial_ciii_assembly, (D) formation of ATP by chemiosmotic coupling, (E) histone modification, (F) budding and maturation of HIV virion, (G) cristae formation, (H) biocarta proteasome pathway, and (I) endosomal sorting complex required for transport. NES – normalized enrichment score; FDR – false discovery rate Figure 5. Expression of SLC26A4-AS1 correlated with immune cells in patients with breast cancer (grouped comparison chart). (A) aDC, (B) B cells, (C) CD8 T cells, (D) cytotoxic cells, (E) DC, (F) macrophages, (G) neutrophils, (H) NK CD56bright cells, (I) NK CD56dim cells, (J) T cells, (K) T helper cells, (L) Tcm, (M) Tem, (N) TFH, and (O) Th1 cells.
Figure 5. Expression of SLC26A4-AS1 correlated with immune cells in patients with breast cancer (grouped comparison chart). (A) aDC, (B) B cells, (C) CD8 T cells, (D) cytotoxic cells, (E) DC, (F) macrophages, (G) neutrophils, (H) NK CD56bright cells, (I) NK CD56dim cells, (J) T cells, (K) T helper cells, (L) Tcm, (M) Tem, (N) TFH, and (O) Th1 cells. Figure 6. Correlation of SLC26A4-AS1 expression with 24 immune cells in patients with breast cancer (scatter plot). (A) aDC, (B) B cells, (C) CD8 T cells, (D) cytotoxic cells, (E) DC, (F) macrophages, (G) neutrophils, (H) NK CD56bright cells, (I) NK CD56dim cells, (J) T cells, (K) T helper cells, (L) Tcm, (M) Tem, (N) TFH, and (O) Th1 cells.
Figure 6. Correlation of SLC26A4-AS1 expression with 24 immune cells in patients with breast cancer (scatter plot). (A) aDC, (B) B cells, (C) CD8 T cells, (D) cytotoxic cells, (E) DC, (F) macrophages, (G) neutrophils, (H) NK CD56bright cells, (I) NK CD56dim cells, (J) T cells, (K) T helper cells, (L) Tcm, (M) Tem, (N) TFH, and (O) Th1 cells. Figure 7. Correlation of SLC26A4-AS1 expression with 24 immune cells in patients with breast cancer (lollipop chart).
Figure 7. Correlation of SLC26A4-AS1 expression with 24 immune cells in patients with breast cancer (lollipop chart). Tables
 Table 1. The Cancer Genome Atlas-based characterization of patients with breast cancer.
Table 1. The Cancer Genome Atlas-based characterization of patients with breast cancer. Table 2. Expression of SLC26A4-AS1 in breast cancer is associated with clinical characteristics.
Table 2. Expression of SLC26A4-AS1 in breast cancer is associated with clinical characteristics. Table 3. Correlation between SLC26A4-AS1 expression and clinical characteristics in patients with breast cancer (logistic regression).
Table 3. Correlation between SLC26A4-AS1 expression and clinical characteristics in patients with breast cancer (logistic regression). Table 4. Correlation between overall survival and clinical characteristics in patients with breast cancer (Cox regression).
Table 4. Correlation between overall survival and clinical characteristics in patients with breast cancer (Cox regression). Table 5. Enrichment of SLC26A4-AS1-related pathways (gene set enrichment analysis).
Table 5. Enrichment of SLC26A4-AS1-related pathways (gene set enrichment analysis). Table 1. The Cancer Genome Atlas-based characterization of patients with breast cancer.
Table 1. The Cancer Genome Atlas-based characterization of patients with breast cancer. Table 2. Expression of SLC26A4-AS1 in breast cancer is associated with clinical characteristics.
Table 2. Expression of SLC26A4-AS1 in breast cancer is associated with clinical characteristics. Table 3. Correlation between SLC26A4-AS1 expression and clinical characteristics in patients with breast cancer (logistic regression).
Table 3. Correlation between SLC26A4-AS1 expression and clinical characteristics in patients with breast cancer (logistic regression). Table 4. Correlation between overall survival and clinical characteristics in patients with breast cancer (Cox regression).
Table 4. Correlation between overall survival and clinical characteristics in patients with breast cancer (Cox regression). Table 5. Enrichment of SLC26A4-AS1-related pathways (gene set enrichment analysis).
Table 5. Enrichment of SLC26A4-AS1-related pathways (gene set enrichment analysis). In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








