04 May 2022: Clinical Research
Comparison of the Prevalence of Antibodies to SARS-CoV-2 in 9954 Recruits in the Korean Army Training Center with the General Korean Population of Equivalent Age Between September and November, 2020
Kwang Ho Mun1CDEF, Haebong Jang2ABG, Hwanyeong Jeong3BC, Jangkyu Choi4B, June-Woo Lee5A, Ah-Ra KimDOI: 10.12659/MSM.934926
Med Sci Monit 2022; 28:e934926
Abstract
BACKGROUND: Understanding the seroprevalence of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) can be a useful tool when studying spread of the disease. This study aimed to compare the prevalence of antibodies to SARS-CoV-2 in 9954 recruits in the Korean Army Training Center with the general Korean population age <30 years between September and November, 2020.
MATERIAL AND METHODS: At the Korean Army Training Center, samples were taken from 9954 men from September to November, 2020. Participants were randomly enlisted healthy adult men. The data were compared with 4,205,265 samples from the Korean general population. Men age <30 years were used, as this is similar to the age range of the military recruits.
RESULTS: Among military recruits, 31 subjects (0.31%) were positive for the antibody, while the Korean male population had 3757 (0.09%) positive individuals. Among these 31 men, 13 were previously diagnosed by PCR, while 18 (58.06%) had no history related to the disease. Positive military recruits were mostly from 2 regional clusters. The first cluster was Daegu and Gyeongbuk areas (1.97% and 0.80%, respectively), which had an outbreak in March, 2020. The second cluster was Gyeonggi and Seoul, or capital areas (0.23% and 0.20%, respectively), which currently has high PCR positivity. Overall, seroprevalence was 3.49 times higher in study subjects.
CONCLUSIONS: The high seroprevalence of antibodies to SARS-CoV-2 between September and November 2020 in a densely populated military academy in Korea may have been an indicator for the resulting outbreak of COVID-19 in winter 2020-21, which highlights the importance of asymptomatic spread from the young and healthy to the general population.
Keywords: Antibodies, COVID-19, SARS-CoV-2, Public Health, Population, Adult, COVID-19, Female, Humans, Immunoglobulin G, Male, Military Personnel, Prevalence, Seroepidemiologic Studies
Background
Symptomatic and asymptomatic infection with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) both trigger antibody responses in humans [1–4]. Determining the total incidence of SARS-CoV-2 infection, including asymptomatic cases, allows us to understand the exact extent of the pandemic and its associated morbidity. This is important for developing suitable policies and tactics against the disease, such as modifying social restriction plans [5]. Therefore, seroprevalence of SARS-CoV-2 antibodies can be a useful tool when studying spread of the disease in a population.
The first case of COVID-19 diagnosed in Korea was on January 20, 2020 [6]. During the first 2 months, most cases were caused by infection from abroad. After February, however, a large-scale outbreak of more than 8000 cases occurred in the Daegu and Gyeongbuk regions [6]. After the outbreak, domestic infections increased, followed by government lead nation-wide interventions and testing [7]. During the second outbreak, in August and September, social distancing was not strict, with high rate of unknown transmission routes (23.5% of the cases) [8]. Therefore, it is safe to assume local spread of the disease was present in the community, although the scale is unknown.
In Korea, a few SARS-CoV-2 antibody seroprevalence studies were done [9,10]. The Korea National Health and Nutrition Examination Survey (KNHANES) reported the seroprevalence in Korea twice, from April to June and June to August. The first tested 1555 people, with no positive results, while the second tested 1440 people, with 1 positive for the antibody [9]. Another study, limited to Daegu city, found that 15 out of 198 tested individuals were positive [10]. However, these studies were performed in specific regions of Korea, as the KNHANES study was limited to the capital and people might have refused to participate in the study if they belonged to a high-risk group [9]. Another study was limited to Daegu city, limiting the representativeness of the study [10].
Korean army recruits of might be an ideal population in which to determine SARS-CoV-2 seroprevalence, as most healthy young Korean men in their 20s are required to serve in the military, recruits are randomly selected, without social or geographic considerations. Furthermore, blood samples are acquired within 1 week of enlistment, reducing the chance of internal spread after enlistment. Moreover, young, relatively healthy men are recruited, allowing detection of unknown past asymptomatic SARS-CoV-2 infection.
Therefore, this study aimed to compare the prevalence of antibodies to SARS-CoV-2 in 9954 recruits in the Korean Army Training Center with the general Korean population, who had a mean age <30 years, between September and November 2020.
Material and Methods
STUDY DESIGN AND PARTICIPANTS:
Study subjects were recruited biweekly, from September to November 2020. All of the participants were newly enlisted personnel at the Korean Army Training Center, Nonsan, Chungnam, Korea. The study population was all adult men, who were enlisted due to mandatory draft, aged 18 or older. As of November 2020, 9954 subjects were recruited. Data on region and age of each encrypted identification number were obtained from Republic of Korean Army Headquarters, concealing subjects’ personal information. The region of each subject was classified by provinces and metropolitan cities, resulting in 17 groups.
The data on the Korean general population was used as a comparison. Only adult males age <30 years were used, as military recruits are all men, with similar age range. Data on 4,205,265 men were obtained from Statistics Korea as of year 2020. Among these, 3757 tested positive for SARS-CoV-2 by polymerase chain reaction (PCR) test as of midnight November 24, 2020, according to the Korea Disease Control and Prevention Agency. This time was selected as final study sample was obtained then. The study was conducted with the approval of the Ethics Committee of the Armed Force Medical Command (IRB No. AFMC-20069-IRB-20-069).
MEASUREMENT:
After enlistment, a blood sample was taken from each subject within 1 week as part of the routine physical examination. After routine examinations, leftover blood samples were used to test for SARS-CoV-2 antibodies.
Antibodies were first studied using the Elecsys anti-SARS-CoV-2 IgG ASSAY on a Cobas e801 analyzer (Roche Diagnostics, Vilvoorde, Belgium). The kit was selected as it showed the highest sensitivity of 99.5% (after 14 days or more of PCR confirmation) and specificity of 99.8% [11]. Positive cases were analyzed again by 3 of the following tests; Abbott SARS-CoV-2 IgG assay (Abbott Park, IL, USA), SARS-CoV-2 IgG assay (Beckman Coulter, USA), R-FIND COVID-19 ELISA (SG medical, Seoul, Korea), and SGTi-flex COVID-19 IgM/IgG (Sugentech, Daejeon, Korea). This method was chosen through an expert advisory meeting of the Korean Society for Laboratory Medicine board, as no standard measure has been established and to compare the results between current study and other Korean studies ongoing at the time. Also, the serum was sent to the Korea Center for Disease Control and Prevention, where plaque reduction neutralization antibody test (PRNT), and the subject’s history of SARS-CoV-2 infection was studied. When at least 1 of the other 5 tests was positive, the subject was considered to be positive for SARS-CoV-2 antibodies.
PLAQUE REDUCTION NEUTRALIZATION TEST (PRNT):
All test sera were heat inactivated at 56°C for 30 min, and underwent serial 2-fold dilution starting from 1/4. Diluted serum was incubated with the 50 pfu of SARS-CoV-2 virus (BetaCoV/Korea/KCDC03/2020) for 1 h at 37°C and 5% CO2. The virus-serum mixtures were added to Vero cell confluent in a 12-well plate, and incubated at 37°C and 5% CO2 for 1 h, with shaking every 10 min, after which virus-serum mixtures were removed and 1 ml of overlay medium (0.75% agarose, 2% 2×MEM) was added. The plate was incubated at 37°C and 5% CO2 for 3 days. At the end of the incubation period, the cells were fixed and stained with crystal violet mixture (0.07% crystal violet, 10% formaldehyde solution, 5% ethanol) for 5–6 h at room temperature, and then overlaid agar was decanted and plaques were visualized and counted. The 50% neutralizing dose (ND50) were calculated using the Kärber formula:
where m is log10 of the highest dilution, Δ is log10 of the dilution coefficient, and Σp is the sum of the number of plaques produced by the virus-serum inoculum/the average number of plaques produced by the virus controls.
STATISTICAL ANALYSIS:
Seroprevalence, both unweighted and weighted by region, was determined among all subjects with 95% confidence interval (CI), using z-statistics. The bootstrap procedure was used for both prevalences, considering statistical error, sensitivity, and specificity of the tests. All analyses were performed by R version 3.5.1 (
Results
As of November 23, 2020, 9954 subjects were tested. A comparison of the subjects and general Korean male population (age 30 or younger) are shown in Table 1. Compared to the general population, our subjects had a higher percentage of population from Daegu and Gyeongbuk, which were the areas where the first pandemic occurred during March 2020.
Among the study subjects, 31 (0.31%) were positive for the antibody, while Korean the general male population showed 3757 (0.09%) positivity (Table 2). Apart from regions without any or with just 1 positive individual, positives were limited to 2 regions. First were the Daegu and Gyeongbuk areas, which had a SARS-CoV-2 outbreak in March and showed the highest positive seroprevalence in study subjects (1.97% vs 0.80%, respectively). Gyeongbuk especially was noted to have 7.23 times higher levels of positives compared to the general Korean male population. Other areas with high positivity were Gyeonggi and Seoul, with 6 (0.23%) and 4 (0.20%) positives, respectively. Overall, seroprevalence was 3.49 times higher in study subjects compared to the general Korean male population. Therefore, the estimated SARS-CoV-2 seroprevalence was 13.096, or 9.340 not confirmed by PCR, based on the current study.
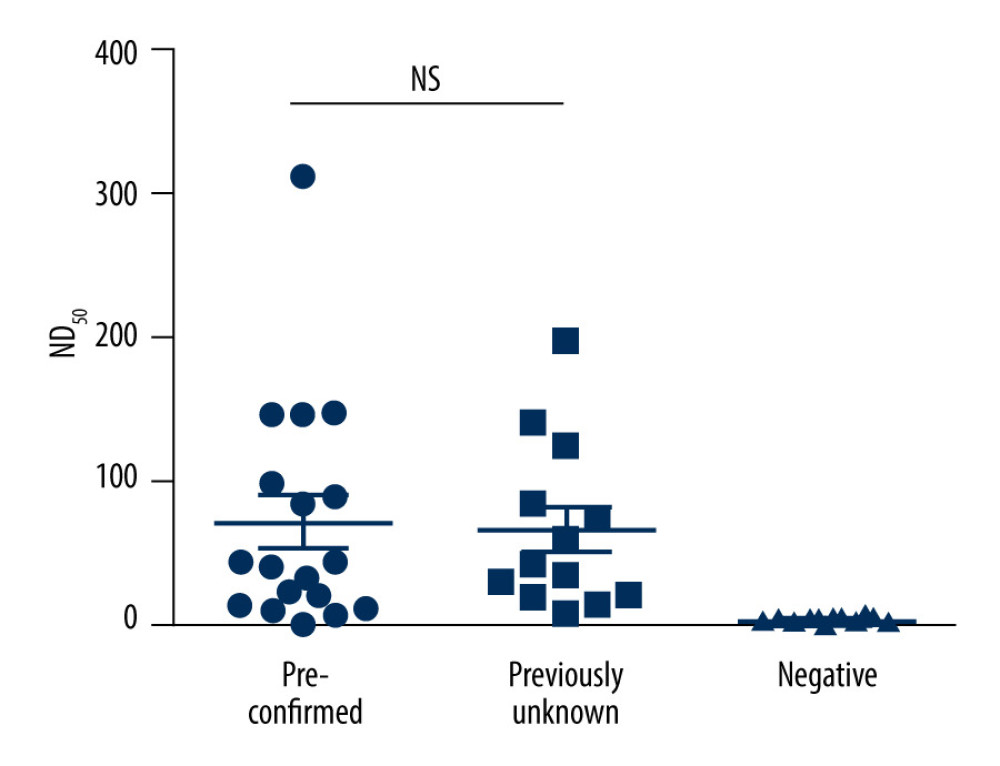
Thirteen out of 31 subjects were reported to have been confirmed by PCR previously (pre-confirmed group), with 18 (58.06%) never known to be infected and had no close contact with people known to have SARS-CoV-2 infection (the previously unknown group) as shown in Table 3. The average time between the PCR-positive date and antibody-positive date was 207.4±51.7 days, with a maximum of 267 days. Neutralizing titers (ND50) of the pre-confirmed group and the unknown seropositive group were the same (both 41), as seen in Figure 1.
Discussion
Our study compared the seroprevalence of SARS-CoV-2 antibodies from young, male military recruits with PCR-positive people in the general Korean male population. The results suggest that there are many undetected infections in communities, despite high numbers of PCR tests. Although most Koreans are highly active in society, most do not have antibodies or any signs of exposure. However, compared to the general population, military recruits showed 5 times higher seroprevalence, suggesting asymptomatic infection in the past, or denial of diagnosis during symptoms.
As our study subjects were randomly recruited due to conscription, selection bias due to social status, religion, or other factors was avoided. Furthermore, subjects were young healthy men and therefore were more active in society, increasing risk of transmission. Thus, we could assume there are many undetected infections present in communities.
Sampling young healthy people might result in overestimating community seroprevalence. However, in Korea most cases were not related to age or social active group, but rather were part of group outbreaks (38.4%) [12]. Other studies have also shown that compared to diagnosed cases of COVID-19, there are many more undiagnosed cases. Even early in the pandemic, in Santa Clara County, USA, estimated cases were more than 50 times higher than the number of diagnosed cases [13]. In a study of dialysis patients in the United States, 8~9% of the population was estimated to be infected, but less than 2% were confirmed cases [14]. In a cross-sectional study of outpatients in Kobe, Japan, seroprevalence was 396 to 858 times higher than the number of confirmed cases [15].
The study of cruise ship outbreaks suggests 20–40% of SARS-CoV-2 infection are asymptomatic [16]. However, as shown above, the estimated number of infections is 5–100 times higher than the number of actual cases. The present study found there were 5 times more people testing positive than the number of confirmed cases. This suggests that the Korean quarantine system detected most of the symptomatic cases.
Moreover, despite high urbanization and population density, there were relatively few missed cases. Kobe, Japan had an estimated seroprevalence of 2.7%, while Santa Clara County had 1.2% [13,15]. However, highly urbanized New York state showed an average of 12.5% positives, and numbers increased in densely populated areas such as New York City, with 20.2% [17].
A few studies have shown antibodies persisting for long periods. A study in Iran showed Ig G was still present after 150 days in 84% of the patients [18]. Another study, by Isho et al, showed Ig G lasting in serum for at least 90 days [19]. A study in hospitalized patients and volunteers found antibodies lasting for 40–200 days after infection, especially among those with more severe disease [20]. In the current study, we found subjects to be seropositive after a mean of 207.4 days. Although further studies should be performed to assess how many have seroreverted, results show that antibodies could last for more than 6 months. Moreover, all of the 13 subjects were PRNT-positive, thus providing a foundation of future herd immunity strategies.
Our study has several strengths and limitations. This is the first large study in Korea with randomized study subjects. Due to the military mandatory draft system, samples could be acquired randomly, across all of the regions. This could have lowered risk of selection bias, compared to the Korean National Health and Nutrition Examination’s result of 0.09%, which requires active participation [10]. However, as study subjects are military recruits, all of the subjects are healthy young men. As SARS-CoV-2 more easily infects unhealthy subjects, the results might be too low, or as SARS-CoV-2 is more easily spread in socially active populations, the results might be too high. Further studies should be performed to further explore these topics.
Also, we tested for SARS-CoV-2 antibodies using several kits selected by Korean Society for Laboratory Medicine and Korean Disease Control and Prevention Agency during spring of 2020. Several diagnostic measures have been developed since, with similar or better results [21–23]. Some include cost-efficient, faster detection methods, with some giving results within 45 min [23]. However, the current study was designed as a national project, with some studies started in spring of 2020. To compare and obtain consistent results between studies, diagnostic measures were not changed. This has been reinforced in ongoing 2021 studies.
Conclusions
This study highlights the importance of continuous regional serological infection monitoring during the COVID-19 pandemic. The high seroprevalence of antibodies to SARS-CoV-2 between September and November 2020 in a densely populated military academy in Korea may have been a predictor of the subsequent outbreak of COVID-19 in winter 2020–21. Also, the findings from this study may help design and reinforce future quarantine strategies and reduce the public health burden of SARS-CoV-2 in the general population.
References
1. Pollán M, Pérez-Gómez B, Pastor-Barriuso R, Prevalence of SARS-CoV-2 in Spain (ENE-COVID): A nationwide, population-based seroepidemiological study: Lancet, 2020; 396; 535-44
2. Sakurai A, Sasaki T, Kato S, Natural history of asymptomatic SARS-CoV-2 infection: N Engl J Med, 2020; 383; 885-86
3. Payne DC, Smith-Jeffcoat SE, Nowak G, SARS-CoV-2 infections and serologic responses from a sample of U.S. navy service members-USS Theodore Roosevelt, April 2020: MMWR Morb Mortal Wkly Rep, 2020; 69; 714-21
4. Hung IF, Cheng VC, Li X, SARS-CoV-2 shedding and seroconversion among passengers quarantined after disembarking a cruise ship: A case series: Lancet Infect Dis, 2020; 20; 1051-60
5. https://www.governor.ny.gov/news/amid-ongoing-covid-19-pandemic-governor-cuomo-outlines-blueprint-un-pause-new-york
6. Korean Society of Infectious Diseases, Korean Society of Pediatric Infectious Diseases, Korean Society of Epidemiology, Korean Society for Antimicrobial Therapy, Korean Society for Healthcare-associated Infection Control and Prevention, Korea Centers for Disease Control and Prevention, Report on the epidemiological features of coronavirus disease 2019 (COVID-19) outbreak in the Republic of Korea from January 19 to March 2, 2020: J Korean Med Sci, 2020; 35; e112
7. Choi S, Ki M, Analyzing the effect of social distancing for novel coronavirus disease 2019 (COVID-19) in South Korea: Epidemiol Health, 2020; 42; e2020064
8. Seong H, Hyun HJ, Yun JG, Comparison of the second and third waves of the COVID-19 pandemic in South Korea: Importance of early public health intervention: Int J Infect Dis, 2021; 104; 742-45
9. https://kosis.kr/statHtml/statHtml.do?orgId=101&tblId=DT_1B040M1
10. Song SK, Lee DH, Nam JH, IgG seroprevalence of COVID-19 among individuals without a history of the coronavirus disease infection in Daegu, Korea: J Korean Med Sci, 2020; 35; e269
11. https://diagnostics.roche.com/global/en/products/params/elecsys-anti-sars-cov-2.html
12. http://ncov.mohw.go.kr/bdBoardList_Real.do?brdId=1&brdGubun=11&ncvContSeq=&contSeq=&board_id=&gubun=
13. Bendavid E, Mulaney B, Sood N, COVID-19 antibody seroprevalence in Santa Clara County, California: Int J Epidemiol, 2021; 50; 410-19
14. Anand S, Montez-Rath M, Han J, Prevalence of SARS-CoV-2 antibodies in a large nationwide sample of patients on dialysis in the USA: A cross-sectional study: Lancet, 2020; 396; 1335-44
15. Doi A, Iwata K, Kuroda H, Estimation of seroprevalence of novel coronavirus disease (COVID-19) using preserved serum at an outpatient setting in Kobe, Japan: A cross-sectional study: Clin Epidemiol Glob Health, 2021; 11; 100747
16. Mizumoto K, Kagaya K, Zarebski A, Chowell G, Estimating the asymptomatic proportion of coronavirus disease 2019 (COVID-19) cases on board the Diamond Princess cruise ship, Yokohama, Japan, 2020: Euro Surveill, 2020; 25; 2000180
17. Rosenberg ES, Tesoriero JM, Rosenthal EM, Cumulative incidence and diagnosis of SARS-CoV-2 infection in New York: Ann Epidemiol, 2020; 48; 23-29
18. Fotouhi F, Salehi-Vaziri M, Farahmand B, Prolonged viral shedding and antibody persistence in patients with COVID-19: Microbes Infect, 2021; 23; 104810
19. Isho B, Abe KT, Zuo M, Persistence of serum and saliva antibody responses to SARS-CoV-2 spike antigens in COVID-19 patients: Sci Immunol, 2020; 5; eabe5511
20. Figueiredo-Campos P, Blankenhaus B, Mota C, Seroprevalence of anti-SARS-CoV-2 antibodies in COVID-19 patients and healthy volunteers up to 6 months post disease onset: Eur J Immunol, 2020; 50; 2025-40
21. Yan S, Ahmad KZ, Warden AR, One-pot pre-coated interface proximity extension assay for ultrasensitive co-detection of anti-SARS-CoV-2 antibodies and viral RNA: Biosens Bioelectron, 2021; 193; 113535
22. Yan S, Ahmad KZ, Li S, Pre-coated interface proximity extension reaction assay enables trace protein detection with single-digit accuracy: Biosens Bioelectron, 2021; 183; 113211
23. Li S, Huang S, Ke Y, A HiPAD integrated with rGO/MWCNTs nano-circuit heater for visual point-of-care testing of SARS-CoV-2: Adv Funct Mater, 2021; 31; 2100801
Tables
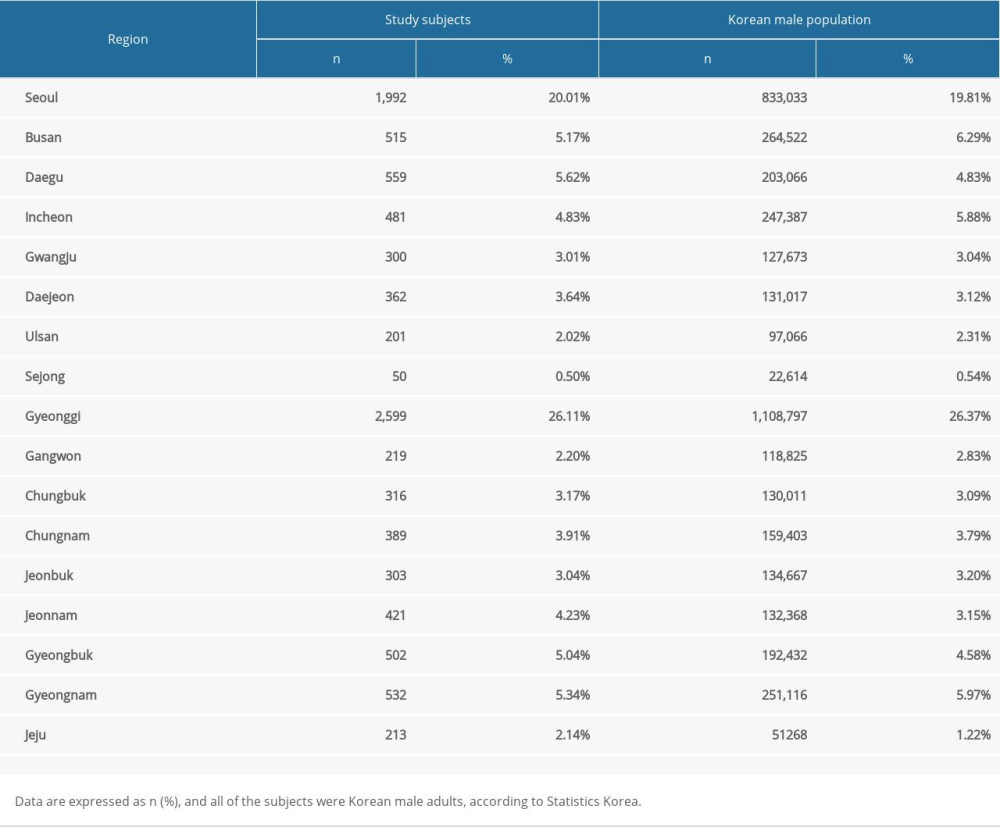 Table 1. Comparison of Study Subjects and Korean Male Population (age <30), by region.
Table 1. Comparison of Study Subjects and Korean Male Population (age <30), by region.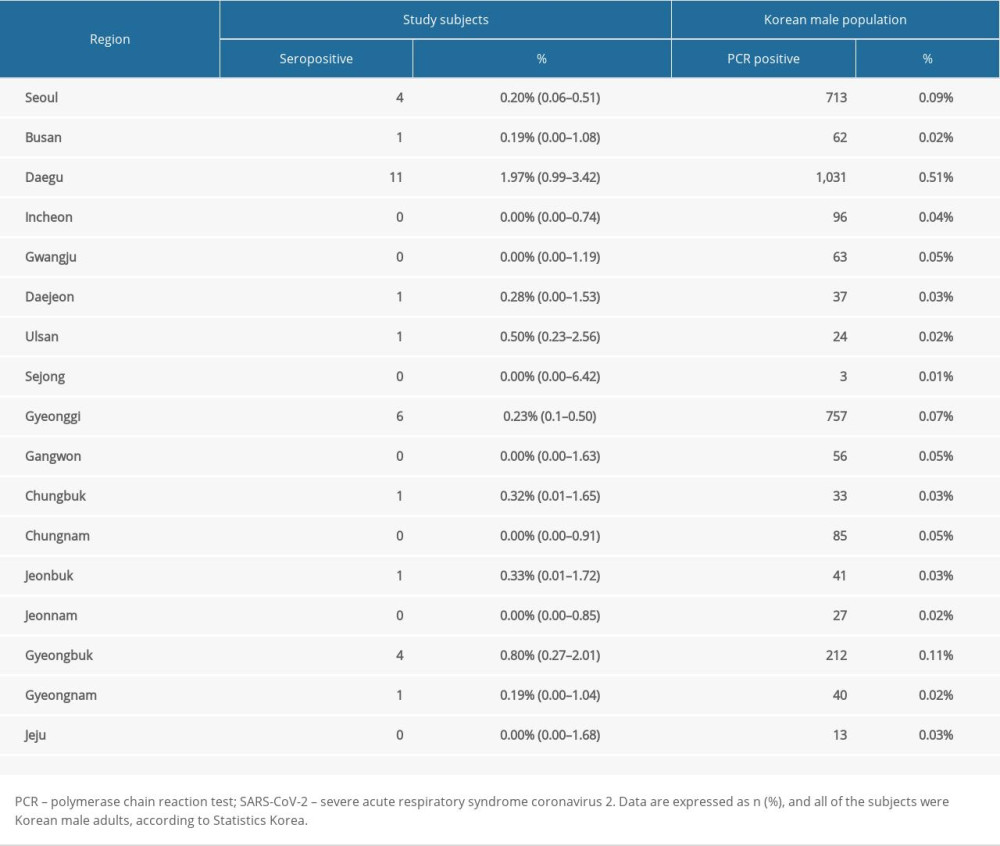 Table 2. Seroprevalence of SARS-CoV-2 antibodies in study subjects and Korean male population.
Table 2. Seroprevalence of SARS-CoV-2 antibodies in study subjects and Korean male population.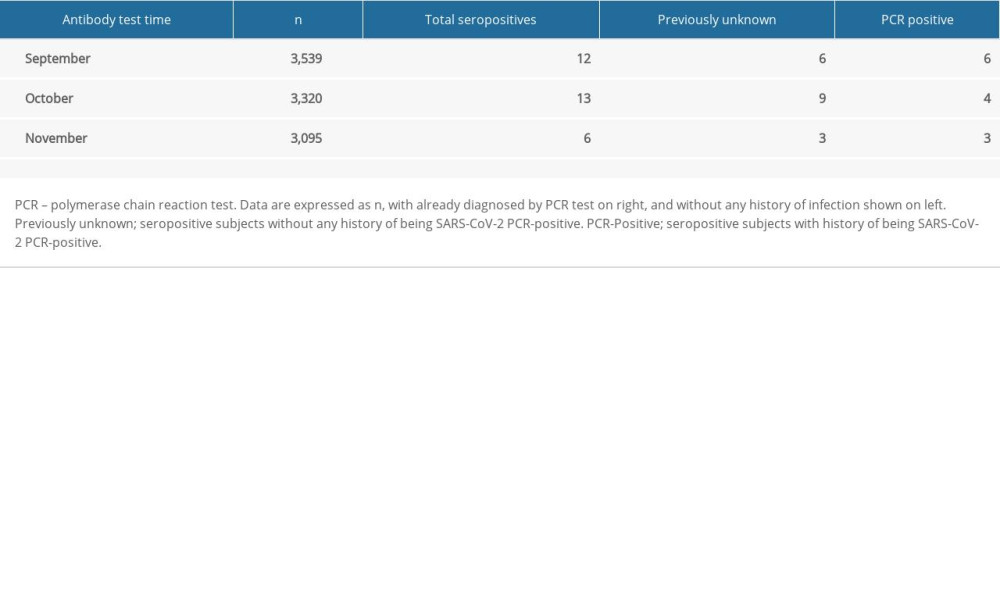 Table 3. Seropositive subjects with previous positive PCR results.
Table 3. Seropositive subjects with previous positive PCR results. Table 1. Comparison of Study Subjects and Korean Male Population (age <30), by region.
Table 1. Comparison of Study Subjects and Korean Male Population (age <30), by region. Table 2. Seroprevalence of SARS-CoV-2 antibodies in study subjects and Korean male population.
Table 2. Seroprevalence of SARS-CoV-2 antibodies in study subjects and Korean male population. Table 3. Seropositive subjects with previous positive PCR results.
Table 3. Seropositive subjects with previous positive PCR results. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387