28 March 2022: Clinical Research
A Retrospective Study to Evaluate the Effect of Dynamic Fracture Mobility on Cement Leakage in Percutaneous Vertebroplasty and Percutaneous Kyphoplasty in 286 Patients with Osteoporotic Vertebral Compression Fractures
Hu Ren1AEF, Tao Feng1AEF*, Jianhui Cao1BC, Yaning Hu1BCD, Dahai Yu1CD, Shuo Pan1BCD, Guangqing Yao1EFDOI: 10.12659/MSM.935080
Med Sci Monit 2022; 28:e935080
Abstract
BACKGROUND: Cement leakage is the most common complication following percutaneous vertebroplasty (PVP) and percutaneous kyphoplasty (PKP) for osteoporotic vertebral compression fractures (OVCFs). Dynamic fracture mobility was determined by comparing preoperative standing lateral radiographs with intraoperative prone lateral radiographs. This retrospective study from a single center aimed to evaluate the effect of dynamic fracture mobility on cement leakage in PVP and PKP in 286 patients with OVCFs.
MATERIAL AND METHODS: Records of patients who underwent PVP or PKP in our department between January 2016 and December 2019 were retrospectively analyzed, showing that 156 patients received PVP and 130 patients received PKP. Variables that were significantly related to presence of cement leakage in the univariate analysis were subsequently included in a multivariate logistic regression analysis for determining the independent risk factors for cement leakage.
RESULTS: The univariate analysis showed that dynamic fracture mobility (P<0.001), operative approach (P=0.026), peripheral vertebrae wall damage (P<0.001), intravertebral cleft (P<0.001), and cement volume injected (P<0.001) were correlated with cement leakage. Factors that showed differences by univariate analysis underwent multivariate logistic regression analysis, showing that peripheral vertebrae wall damage (OR=11.774,95% CI 4.384-31.619, P=0.000), dynamic fracture mobility (OR=5.884, 95% CI 2.295-15.087, P=0.000), operative approach (OR=3.143, 95% CI 1.136-8.698, P=0.027), and cement volume injected (OR=1.486, 95% CI 1.119-1.973, P=0.006) were independent risk factors for postoperative cement leakage.
CONCLUSIONS: This retrospective study showed that dynamic fracture mobility, peripheral vertebrae wall damage, operative approach, and cement volume injected were risk factors for cement leak following PVP and PKP.
Keywords: Bone Cements, Minimally Invasive Surgical Procedures, Osteoporotic Fractures, Risk Factors, Fractures, Compression, Humans, kyphoplasty, Spinal Fractures, vertebroplasty
Background
With the increasing number of osteoporotic vertebral compression fractures (OVCFs), the quality of life of the elderly has been seriously affected [1]. Percutaneous vertebroplasty (PVP) and percutaneous kyphoplasty (PKP) both involve injecting bone cement into the fractured vertebra. This procedure not only stabilizes the fractured vertebra, but also significantly relieves pain [2]. Although both techniques have shown good results for OVCFs, complications associated with surgery are inevitable. Among these complications, cement leakage is the most common [3]. Although it was asymptomatic in most cases, cement leakage can cause severe consequences [4,5]; therefore, it is very important to identify the risk factors of cement leakage before surgery. Many studies have attempted to reveal the risk factors of cement leakage, including cement viscosity, cement volume, intravertebral cleft, the degree of compression of the fractured vertebrae, the type of the fractured vertebrae, and operative approach [6–9].
The previously unrecognized occurrence of “dynamic fracture mobility” in many OVCFs was first demonstrated by McKiernan et al in 2003 [10]. Spontaneous reduction in deformity may occur when the patient is placed in prone position. Dynamic fracture mobility was determined by comparing preoperative standing lateral radiographs with intraoperative prone lateral radiographs [11]. Many authors have reported that the spinal deformity correction achieved with percutaneous vertebral augmentation techniques is greatly influenced by the presence of dynamic fracture mobility, and it was reported to be an important parameter in the evaluation of vertebral fractures [11–14]. Unfortunately, there is no research on the effect of dynamic fracture mobility on cement leakage. Therefore, this retrospective study from a single center aimed to evaluate the effect of dynamic fracture mobility on cement leakage in PVP and PKP in 286 patients with OVCFs. Our results may help to in making detailed plans for a unique OVCF and may reduce surgical complications.
Material and Methods
STUDY DESIGN:
Records of patients who underwent PVP or PKP in our department between January 2016 and December 2019 were retrospectively analyzed. To reduce confounding factors caused by the inclusion of multiple OVCFs patients, only patients with a single-level acute OVCF were selected in this study. This study protocol was approved by the Ethics Committee of Shijiazhuang People’s Hospital (no. 2021-006) before acquiring and analyzing patients’ information.
INCLUSION AND EXCLUSION CRITERIA:
Inclusion criteria were: (1) persistent back pain resulting from a single-level OVCF that was not responsive to nonoperative treatment; (2) fracture duration ≤6 weeks; (3) treated with PVP or PKP; (4) complete imaging and surgical data were available. Exclusion criteria were: (1) fracture duration >6 weeks; (2) fracture not caused by OVCFs; (3) presence of a previously treated OVCF; (4) patients with radicular symptoms.
PATIENTS:
Finally, 286 patients (232 females, 54 males, mean age 72.87 years, range 51–89 years) were included in our study: 156 (54.5%) patients received PVP and 130 (45.5%) patients received PKP. Among the 286 vertebrae studied, 86 (30.1%) involved the thoracic spine, 174 (60.8%) involved the thoracolumbar spine, and 26 (9.1%) involved the lumbar spine.
SURGICAL TECHNIQUE:
All operations were performed by experienced spinal surgeons. Local anesthesia was used during all procedures. Patients were monitored and placed in prone position using Cawley’s technique [15]. Supports were positioned under the iliac crests and either side of the upper thorax. The arms were abducted. Two columns of pillows were positioned under the legs so that the hips were maximally extended. The table could be flexed as needed, and this position may help to correct the fracture deformity to some extent. While in this position, a prone position radiograph centered on the fractured vertebra was obtained for each patient.
Then, the standard PVP or PKP surgery was performed by the bilateral transpedicular approach under lateral and anteroposterior fluoroscopic guidance. During PKP, the inflatable bone tamp (IBT, Changzhou Bai-long Medical Utensils Co., Changzhou, China) was inflated until the fracture was reduced, the IBT contacted a vertebral body end-plate, the balloon volume reached maximum, or it was felt unsafe to continue. The IBT was then removed and polymethylmethacrylate bone cement (PMMA, Osteopal V, Heraeus Medical GmbH, Wehrheim, Germany) was injected at the “toothpaste-like” stage after being mixed to reduce cement leakage. When cement was evenly distributed in the fractured vertebra or when leakage was observed, the cement injection was discontinued.
After the surgery, the patient was transferred to the ward for further monitoring. The degree of focal back pain was assessed by visual analog scale (VAS) (where 0 represents no pain and 10 represents the worst pain) before surgery, 3 days after surgery, and at last follow-up.
RADIOGRAPHIC EVALUATION:
All patients underwent preoperative standing anterior-posterior and lateral radiographs centered on the fractured vertebral body. The maximum compression point of the preoperative vertebral body was selected for vertebral height measurement [16,17]. Assessing the preoperative vertebral body compression ratios according to the equation, preoperative vertebral compression ratio=preoperative vertebral body height/estimated original vertebral body height. The mean value of the vertical height of the vertebrae above and below the fractured vertebra was used to estimate original vertebral body height. If the adjacent vertebrae were fractured previously, the nearest intact vertebra was used to make an estimation. Kyphotic angle was measured from the superior and inferior end-plates of the fractured vertebrae [18]. Dynamic fracture mobility was considered to be present when any measurable change in vertebral body kyphotic angle or vertebral body height occurred between preoperative standing lateral radiographs and intraoperative prone position radiographs [11] (Figure 1A, 1B). Fractures with no measurable changes were considered fixed.
Magnetic resonance imaging (MRI) was used to evaluate for persistent edema in the fractured vertebrae and to exclude other causes besides OVCFs. An intravertebral cleft was defined as an intravertebral, abnormal, well-demarcated, linear or cystic hypointensity similar to air on preoperative radiography or T1-weighted MRI sequences, and an abnormal, well-demarcated, linear or cystic hyperintensity was defined as being similar to cerebrospinal fluid on T2-weighted MRI sequences or STIR sequences [19] (Figure 1C, 1D). Peripheral vertebrae wall damage was defined as the presence of a fracture line and destruction of cortical bone at the vertebral wall or end-plate by preoperative computed tomography (CT) and MRI [20] (Figure 1E). Bone mineral density (BMD) of lumbar vertebrae was measured by dual-energy X-ray absorptiometry (DEXA) prior to surgery.
Patients were divided into a leakage group and a no leakage group according to postoperative radiographs and CT examination. Cement leakage was defined as the presence of any extravertebral cement (Figure 1F).
POTENTIAL RISK FACTORS:
To provide a more comprehensive prediction model for cement leakage, all possible correlation factors were collected, including: age, sex, fracture duration, bone mineral density, dynamic fracture mobility, operative approach, spinal segment of fracture, peripheral vertebrae wall damage, intravertebral cleft, cement volume injected, preoperative vertebral height, and preoperative kyphotic angle.
STATISTICAL ANALYSIS:
Statistical software SPSS (version 13.0, SPSS, Inc, Chicago, IL) was used for the statistical analysis. Continuous variables were expressed as mean±standard deviation and categorical variables were expressed as the number of cases. Univariate analysis was carried out with
Results
CHARACTERISTICS OF STUDY PATIENTS:
Almost all of the patients had rapid and significant improvement in back pain following surgery. VAS was from 7.56±1.43 preoperatively to 2.35±1.77 at 3 days after surgery, and 2.58±1.73 at last follow-up (
UNIVARIATE ANALYSIS BETWEEN 2 GROUPS:
The univariate analysis showed that dynamic fracture mobility (P<0.001), operative approach (P=0.026), peripheral vertebrae wall damage (P<0.001), intravertebral cleft (P<0.001), and cement volume injected (P<0.001) were correlated with cement leakage. The presence of peripheral vertebrae wall damage, dynamic fracture mobility, and intravertebral cleft exhibited a 6.6-fold (49.15% vs 7.49%), 4.7-fold (30.17% vs 6.47%), and 3.7-fold (35.14% vs 9.43%) higher cement leakage risk, respectively (Figure 2). The average volumes of injected cement in the leakage group were greater than in the no leakage group (5.17±1.24 ml vs 4.16±1.49 ml, t=4.330, P<0.001). The use of PKP was significantly associated with a lower cement leakage risk than PVP (10.77% vs 20.51%, χ2=4.988, P=0.026) (Figure 2). Additionally, univariate analysis showed there was no statistically significant difference between the 2 groups with regard to age, sex, fracture duration, bone mineral density, spinal segment of fracture, preoperative vertebral height, and preoperative kyphotic angle (P>0.05). The clinical and radiological features of the leakage and no leakage groups are shown in Table 1.
In addition, of the 116 vertebrae with dynamic fracture mobility, 62 vertebrae (53%) had an intravertebral cleft and 45 vertebrae (39%) had peripheral vertebrae wall damage. Of the 170 vertebrae without dynamic fracture mobility, 12 vertebrae (7%) had an intravertebral cleft and 14 vertebrae (8%) had peripheral vertebrae wall damage (Figure 3).
MULTIVARIATE LOGISTIC REGRESSION ANALYSIS OF RISK FACTORS:
Factors that showed differences by univariate analysis underwent multivariate logistic regression analysis, showing that peripheral vertebrae wall damage (OR=11.774, 95% CI 4.384–31.619, P=0.000), dynamic fracture mobility (OR=5.884, 95% CI 2.295–15.087, P=0.000), operative approach (OR=3.143, 95% CI 1.136–8.698, P=0.027) and cement volume injected (OR=1.486, 95% CI 1.119–1.973, P=0.006) were independent risk factors for postoperative cement leakage (Table 2, Figure 4).
Discussion
This retrospective study confirmed that dynamic fracture mobility was one of the most influential factors for cement leakage after PVP and PKP for OVCFs. In addition, peripheral vertebrae wall damage, operative approach, and cement volume injected were also be identified as risk factors associated with cement leakage.
Most of the intraoperative and postoperative adverse effects in PVP and PKP were associated with cement leakage [21]. To reduce the occurrence of complications, there is an urgent need to identify the risk factors for cement leakage. A variety of clinical information and procedural characteristics have been collected and analyzed [6–9]. However, there is no consensus on the risk factors for bone cement leakage.
Since dynamic fracture mobility was first demonstrated by McKiernan et al in 35% of treated vertebrae [10], many studies have explored its effect on deformity correction in PVP and PKP for OVCFs. In addition, many scholars consider dynamic fracture mobility as an important index for characterizing individual fractured vertebrae and engaging in PVP and PKP [11–14]. In the present study, the final results confirmed it was also one of the most influential factors affecting cement leakage.
Dynamic fracture mobility, peripheral vertebrae wall damage, and intravertebral cleft were 3 important indicators for evaluating fracture characteristics. It has been widely accepted that the presence of peripheral vertebrae wall damage is a strong risk predictor for cement leakage [4,6–8,17], which was also confirmed by the present study. However, the effect of an intravertebral cleft remains controversial to date. Some scholars reported that because of the frequently present connection with the peri-vertebral tissue, intravertebral cleft was an independent risk factor for cement leakage [22]. This view was also confirmed by Gao et al [6]. They found that the presence of an intravertebral cleft was a risk factor for intradiscal and paravertebral subtype of cortical leakages, but it was not an independent risk factor for overall level cement leakage. On the contrary, Krauss et al found that patients with intravertebral clefts showed a significant lower risk of cement leakage compared to non-cleft patients during vertebroplasty (18.2% vs 46%) [23]. They speculated the reason might be that an intravertebral cleft is an avascular process surrounded by a fibrocartilaginous membrane; cement leakage is thus only possible when vital bone tissue is filled in addition to the intravertebral cleft. Tomé-Bermejo et al also expressed a similar opinion [24]. In our study, the presence of peripheral vertebrae wall damage, dynamic fracture mobility, and intravertebral cleft exhibited a 6.6-fold, 4.7-fold, and 3.7-fold higher cement leakage risk, respectively. After further analysis, we discovered that of the 116 vertebrae that showed dynamic fracture mobility, 53% of the vertebrae had an intravertebral cleft and 39% of the vertebrae had peripheral vertebrae wall damage, and this proportion was only 7% and 8% in the vertebrae that lack dynamic fracture mobility, respectively. These 3 factors may intersect and coexist in a particular fractured vertebral body. After controlling for confounding factors, subsequent more precise multivariate logistic regression analysis excluded intravertebral cleft from the independent risk factors due to its relatively weak influence, and we found that dynamic fracture mobility could be used as an independent risk index to predict cement leakage.
Dynamic fracture mobility initially attracted attention because it implied complete corticocancellous disruption and allowed height recovery of the vertebra after postural reduction [25,26]. Liu et al reported that placement of the patient in prone hyperextended position produced a significant reduction of anterior vertebral height by 5.51±2.64 mm, middle vertebral height by 4.35±2.73 mm, and posterior vertebral height by 3.79±3.22 mm [27]. In turn, these vertebral height changes also aggravate damage to the peri-vertebral wall, thereby increasing the risk of cement leakage.
Percutaneous injection of bone cement into the fractured vertebrae is the most important step in PVP and PKP. Therefore, the characteristics of bone cement itself would play an important role in the effect of the operation. Both cement viscosity and cement volume have been widely demonstrated to be the major risk factors associated with cement leakage [28,29]. With the increase of injection volume of bone cement, the risk of cement leakage would also increase. The volume of bone cement injected was relatively small (mean volume was 4.32 ml) in our study, so the leakage incidence was relatively lower compared with some other reports. This result was consistent with Sun’s research [20]. They suggested that the optimal amount of cement injection in a single thoracolumbar fractured vertebral body was 4–6 ml. The optimal intravertebral cement volume/fractured vertebral body volume was 19.78%, which could achieve satisfactory cement distribution, rapid relief of pain, and reduce occurrence of cement leakage.
From the literature, we know cement viscosity is another crucial parameter associated with cement leakage risk. Because there was no objective standard to evaluate the cement viscosity, it was not analyzed as an impact indicator in this study. Optimal cement viscosity is subjective and multiple factors could influence it, such as storage conditions, mixed methods, operating room temperature, and surgeon experience [24,30,31]. To reduce the occurrence of bone cement leakage, the cement was injected during the phase of “toothpaste-like” consistency after mixing, according to most previous reports [32].
Many studies have confirmed that PKP has significant advantages over PVP in terms of vertebral height restoration, kyphosis correction, and prevention of bone cement leakage [33]. The use of an expandable balloon during PKP provided a low-pressure cavity for the cement injection, which could reduce the risk of cement leakage. However, PKP has not completely replaced PVP due to its higher cost, greater complexity, and longer operation time [34,35]. Even so, when risk factors, such as peripheral vertebrae wall damage and dynamic fracture mobility, for cement leakage are present, PKP is recommend because of its ability to reduce the incidence of complications associated with cement leakage. In addition, the importance of C-arm fluoroscopy for monitoring possible leakage and keeping the needle tip away from the cortical disruption should also be emphasized [6].
The present study has several limitations. First, we did not distinguish the type of cement leakage and did not further explore the unique risk factors for each type of leakage because of concerns that a relatively small sample size could lead to inaccurate results, but we believe that any pattern of leakage should be given sufficient attention. Second, this was a retrospective study with a small number of patients from a single center. Nevertheless, we believe that this study provides some meaningful information for practical work and future research directions. Further large-sample multicenter studies are needed to confirm this result.
Conclusions
This retrospective study showed that dynamic fracture mobility, peripheral vertebrae wall damage, operative approach, and cement volume injected were risk factors for cement leak following PVP and PKP. Surgeons should pay more attention to these factors and make a detailed plan before the operation to reduce the complications associated with cement leakage.
Figures
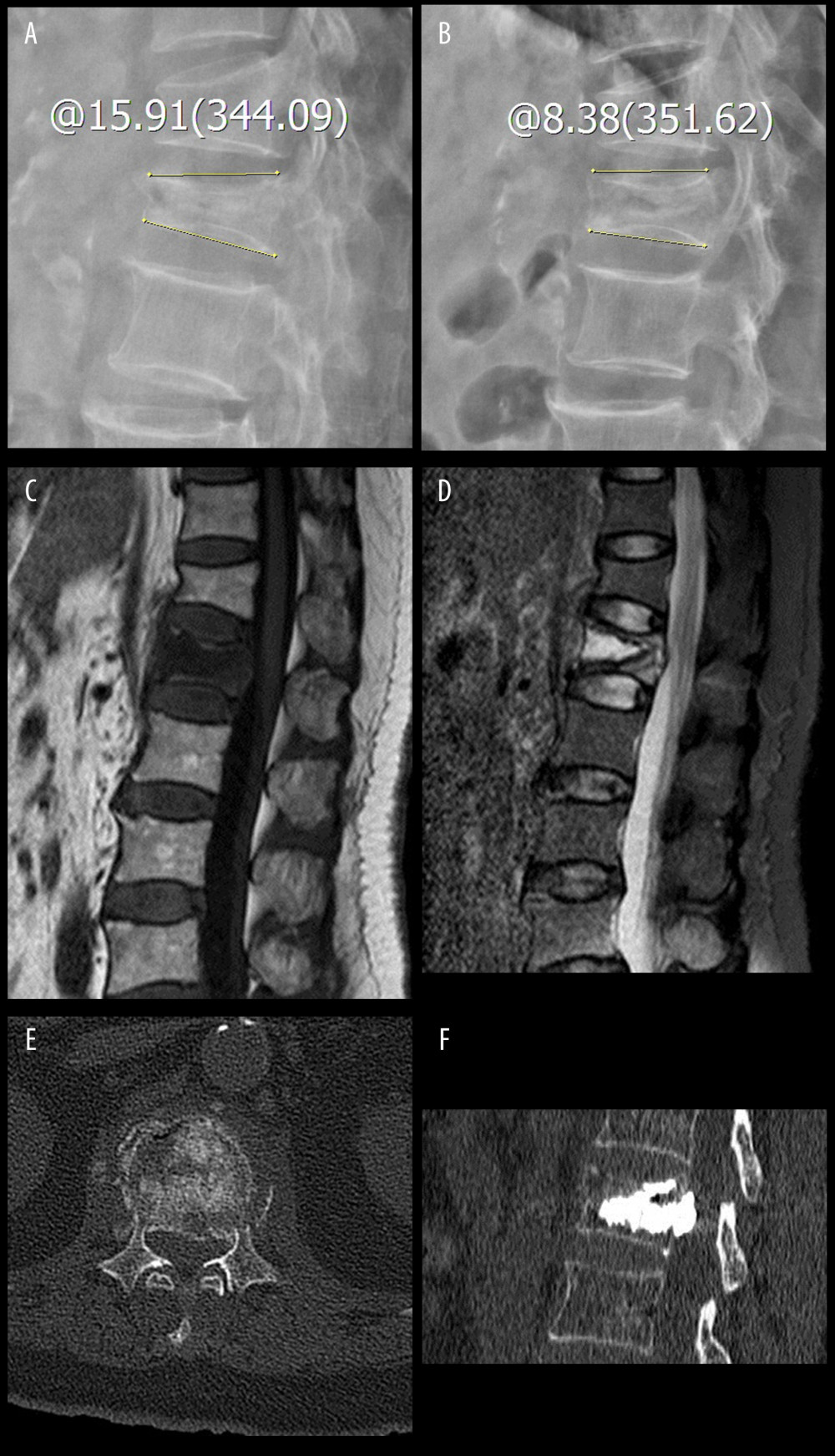 Figure 1. The patient was a 74-year-old woman with an L1 osteoporotic vertebral compression fracture and percutaneous vertebroplasty was performed. The yellow line through A and B indicates the kyphotic angle. (A, B) The kyphotic angle changed from 15.91° in preoperative standing lateral radiographs to 8.38° in intraoperative prone lateral radiographs. Thus, the dynamic fracture mobility was present. (C, D) Preoperative MRI showing the L1 osteoporotic vertebral compression fracture existed intravertebral cleft. (E) Preoperative CT showing the peripheral vertebrae wall damage. (F) Postoperative CT showing the occurrence of cement leakage.
Figure 1. The patient was a 74-year-old woman with an L1 osteoporotic vertebral compression fracture and percutaneous vertebroplasty was performed. The yellow line through A and B indicates the kyphotic angle. (A, B) The kyphotic angle changed from 15.91° in preoperative standing lateral radiographs to 8.38° in intraoperative prone lateral radiographs. Thus, the dynamic fracture mobility was present. (C, D) Preoperative MRI showing the L1 osteoporotic vertebral compression fracture existed intravertebral cleft. (E) Preoperative CT showing the peripheral vertebrae wall damage. (F) Postoperative CT showing the occurrence of cement leakage. 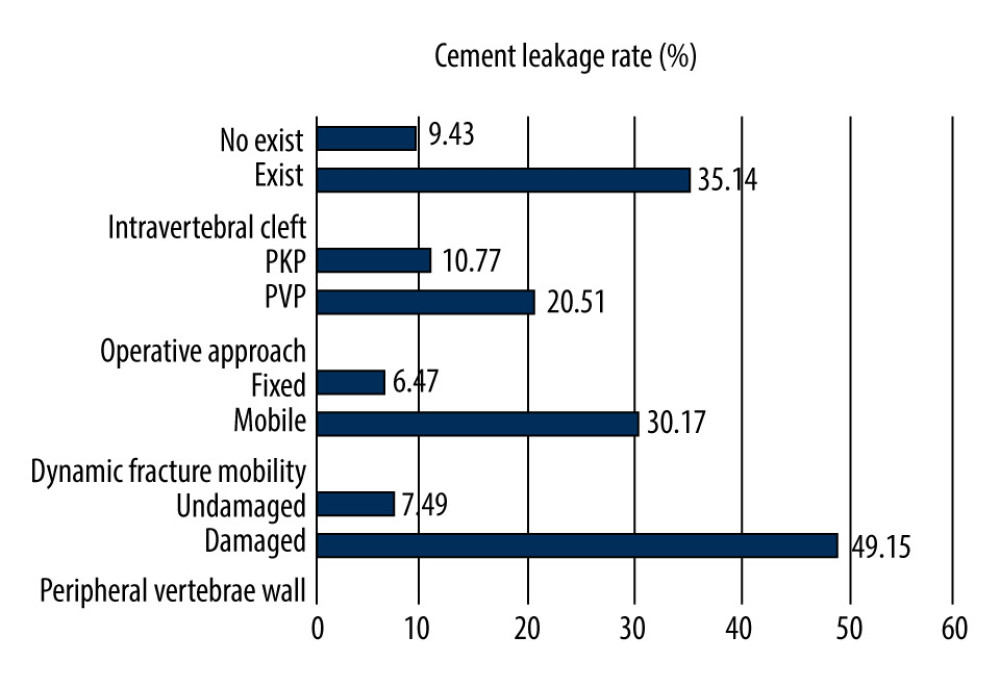 Figure 2. Cement leakage rates compared between different types. PVP – percutaneous vertebroplasty; PKP – percutaneous kyphoplasty.
Figure 2. Cement leakage rates compared between different types. PVP – percutaneous vertebroplasty; PKP – percutaneous kyphoplasty. 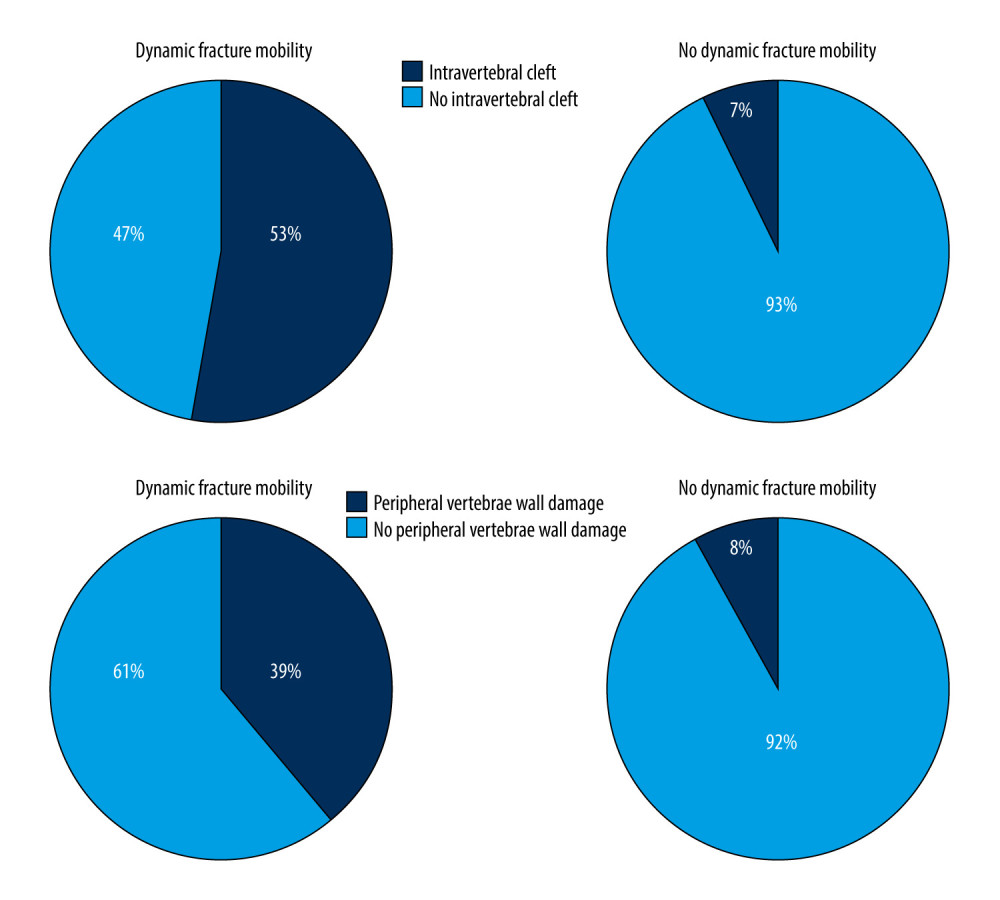 Figure 3. Composition of fractured vertebrae with dynamic fracture mobility.
Figure 3. Composition of fractured vertebrae with dynamic fracture mobility.  Figure 4. Analysis of risk factors for cement leakage.
Figure 4. Analysis of risk factors for cement leakage. References
1. Ma YH, Tian ZS, Liu HC, Predictive risk factors for recollapse of cemented vertebrae after percutaneous vertebroplasty: A meta-analysis: World J Clin Cases, 2021; 9(12); 2778-90
2. Parreira PCS, Maher CG, Megale RZ, An overview of clinical guidelines for the management of vertebral compression fracture: A systematic review: Spine J, 2017; 17(12); 1932-38
3. Semaan H, Obri T, Bazerbashi M, Clinical outcome and subsequent sequelae of cement extravasation after percutaneous kyphoplasty and vertebroplasty: A comparative review: Acta Radiol, 2018; 59(7); 861-68
4. Fadili Hassani S, Cormier E, Shotar E, Intracardiac cement embolism during percutaneous vertebroplasty: Incidence, risk factors and clinical management: Eur Radiol, 2019; 29(2); 663-73
5. Shridhar P, Chen Y, Khalil R, A review of PMMA bone cement and intra-cardiac embolism: Materials (Basel), 2016; 9(10); 821
6. Gao C, Zong M, Wang WT, Analysis of risk factors causing short-term cement leakages and long-term complications after percutaneous kyphoplasty for osteoporotic vertebral compression fractures: Acta Radiol, 2018; 59(5); 577-85
7. Huang S, Zhu X, Xiao D, Therapeutic effect of percutaneous kyphoplasty combined with anti-osteoporosis drug on postmenopausal women with osteoporotic vertebral compression fracture and analysis of postoperative bone cement leakage risk factors: A retrospective cohort study: J Orthop Surg Res, 2019; 14(1); 452
8. Zhan Y, Jiang J, Liao H, Risk factors for cement leakage after vertebroplasty or kyphoplasty: A meta-Analysis of published evidence: World Neurosurg, 2017; 101; 633-42
9. Tang B, Xu S, Chen X, The impact of intravertebral cleft on cement leakage in percutaneous vertebroplasty for osteoporotic vertebral compression fractures: A case-control study: BMC Musculoskelet Disord, 2021; 22(1); 805
10. McKiernan F, Faciszewski T, Jensen R, Reporting height restoration in vertebral compression fractures: Spine (Phila Pa 1976), 2003; 28(22); 2517-21
11. Voggenreiter G, Balloon kyphoplasty is effective in deformity correction of osteoporotic vertebral compression fractures: Spine (Phila Pa 1976), 2005; 30(24); 2806-12
12. Chiu CK, Lisitha KA, Elias DM, Do the dynamic stress mobility radiographs predict the postoperative vertebral height restoration, kyphosis correction, and cement volume injected after vertebroplasty for osteoporotic thoracolumbar vertebral fractures with intravertebral cleft?: J Orthop Surg, 2018; 26(3); 1-9
13. Yokoyama K, Kawanishi M, Yamada M, In not only vertebroplasty but also kyphoplasty, the resolution of vertebral deformities depends on vertebral mobility: Am J Neuroradiol, 2013; 34(7); 1474-78
14. Chen YJ, Chen HY, Chen HT, Diagnosis of painful cemented vertebrae from failed vertebroplasty: Modified dynamic radiographs play an important role: Eur Spine J, 2017; 26(7); 1953-60
15. Cawley DT, Sexton P, Murphy T, Optimal patient positioning for ligamentotaxis during balloon kyphoplasty of the thoracolumbar and lumbar spine: J Clin Neurosci, 2011; 18(6); 834-36
16. Yu WB, Jiang XB, Liang D, Risk factors and score for recollapse of the augmented vertebrae after percutaneous vertebroplasty in osteoporotic vertebral compression fractures: Osteoporos Int, 2019; 30(2); 423-30
17. Zhang TY, Zhang PX, Xue F, Risk factors for cement leakage and nomogram for predicting the intradiscal cement leakage after the vertebra augmented surgery: BMC Musculoskelet Disord, 2020; 21(1); 792
18. Hsu WE, Su KC, Chen KH, The evaluation of different radiological measurement parameters of the degree of collapse of the vertebral body in vertebral compression fractures: Appl Bionics Biomech, 2019; 2019; 4021640
19. Nieuwenhuijse MJ, van Rijswijk CS, van Erkel AR, The intravertebral cleft in painful long-standing osteoporotic vertebral compression fractures treated with percutaneous vertebroplasty: Diagnostic assessment and clinical significance: Spine (Phila Pa 1976) 15, 2012; 37(11); 974-81
20. Sun HB, Jing XS, Liu YZ, The optimal volume fraction in percutaneous vertebroplasty evaluated by pain relief, cement dispersion, and cement leakage: A prospective cohort study of 130 patients with painful osteoporotic vertebral compression fracture in the thoracolumbar vertebra: World Neurosurg, 2018; 114; e677-88
21. Semaan H, Obri T, Bazerbashi M, Clinical outcome and subsequent sequelae of cement extravasation after percutaneous kyphoplasty and vertebroplasty: A comparative review: Acta Radiol, 2018; 59(7); 861-68
22. Nieuwenhuijse MJ, Van Erkel AR, Dijkstra PD, Cement leakage in percutaneous vertebroplasty for osteoporotic vertebral compression fractures: Identification of risk factors: Spine J, 2011; 11(9); 839-48
23. Krauss M, Hirschfelder H, Tomandl B, Kyphosis reduction and the rate of cement leaks after vertebroplasty of intravertebral clefts: Eur Radiol, 2006; 16(5); 1015-21
24. Tomé-Bermejo F, Piñera AR, Duran-Álvarez C, Identification of risk factors for the occurrence of cement leakage during percutaneous vertebroplasty for painful osteoporotic or malignant vertebral fracture: Spine (Phila Pa 1976), 2014; 39(11); E693-700
25. Chen YJ, Lo DF, Chang CH, The value of dynamic radiographs in diagnosing painful vertebrae in osteoporotic compression fractures: Am J Neuroradiol, 2011; 32(1); 121-24
26. McKiernan F, Jensen R, Faciszewski T, The dynamic mobility of vertebral compression fractures: J Bone Miner Res, 2003; 18(1); 24-29
27. Liu HX, Xu C, Shang P, The effect of postural correction and subsequent balloon inflation in deformity correction of acute osteoporotic vertebral fractures: J Korean Neurosurg Soc, 2014; 55(6); 337-42
28. Chen C, Fan P, Xie X, Risk factors for cement leakage and adjacent vertebral fractures in kyphoplasty for osteoporotic vertebral fractures: Clin Spine Surg, 2020; 33(6); E251-55
29. Alhashash M, Shousha M, Barakat AS, Effects of polymethylmethacrylate cement viscosity and bone porosity on cement leakage and new vertebral fractures after percutaneous vertebroplasty: A prospective study: Global Spine J, 2019; 9(7); 754-60
30. Zhu SY, Zhong ZM, Wu Q, Risk factors for bone cement leakage in percutaneous vertebroplasty: A retrospective study of four hundred and eighty-five patients: Int Orthop, 2016; 40(6); 1205-10
31. Loeffel M, Ferguson SJ, Nolte LP, Vertebroplasty: Experimental characterization of polymethylmethacrylate bone cement spreading as a function of viscosity, bone porosity, and flow rate: Spine (Phila Pa 1976), 2008; 33(12); 1352-59
32. Martikos K, Greggi T, Vommaro F, Vertebroplasty in the treatment of osteoporotic vertebral compression fractures: Patient selection and perspectives: Open Access Rheumatol, 2019; 11; 157-61
33. Kim KH, Kuh SU, Chin DK, Kyphoplasty versus vertebroplasty: Restoration of vertebral body height and correction of kyphotic deformity with special attention to the shape of the fractured vertebrae: J Spinal Disord Tech, 2012; 25(6); 338-44
34. Yang D, Zhang Y, Ma X, Resources utilisation and economic burden of percutaneous vertebroplasty or percutaneous kyphoplasty for treatment of osteoporotic vertebral compression fractures in China: A retrospective claim database study: BMC Musculoskelet Disord, 2020; 21(1); 255
35. Rabei R, Patel K, Ginsburg M, Percutaneous vertebral augmentation for vertebral compression fractures: National trends in the medicare population (2005–2015): Spine (Phila Pa 1976), 2019; 44(2); 123-33
Figures
 Figure 1. The patient was a 74-year-old woman with an L1 osteoporotic vertebral compression fracture and percutaneous vertebroplasty was performed. The yellow line through A and B indicates the kyphotic angle. (A, B) The kyphotic angle changed from 15.91° in preoperative standing lateral radiographs to 8.38° in intraoperative prone lateral radiographs. Thus, the dynamic fracture mobility was present. (C, D) Preoperative MRI showing the L1 osteoporotic vertebral compression fracture existed intravertebral cleft. (E) Preoperative CT showing the peripheral vertebrae wall damage. (F) Postoperative CT showing the occurrence of cement leakage.
Figure 1. The patient was a 74-year-old woman with an L1 osteoporotic vertebral compression fracture and percutaneous vertebroplasty was performed. The yellow line through A and B indicates the kyphotic angle. (A, B) The kyphotic angle changed from 15.91° in preoperative standing lateral radiographs to 8.38° in intraoperative prone lateral radiographs. Thus, the dynamic fracture mobility was present. (C, D) Preoperative MRI showing the L1 osteoporotic vertebral compression fracture existed intravertebral cleft. (E) Preoperative CT showing the peripheral vertebrae wall damage. (F) Postoperative CT showing the occurrence of cement leakage. Figure 2. Cement leakage rates compared between different types. PVP – percutaneous vertebroplasty; PKP – percutaneous kyphoplasty.
Figure 2. Cement leakage rates compared between different types. PVP – percutaneous vertebroplasty; PKP – percutaneous kyphoplasty. Figure 3. Composition of fractured vertebrae with dynamic fracture mobility.
Figure 3. Composition of fractured vertebrae with dynamic fracture mobility. Figure 4. Analysis of risk factors for cement leakage.
Figure 4. Analysis of risk factors for cement leakage. Tables
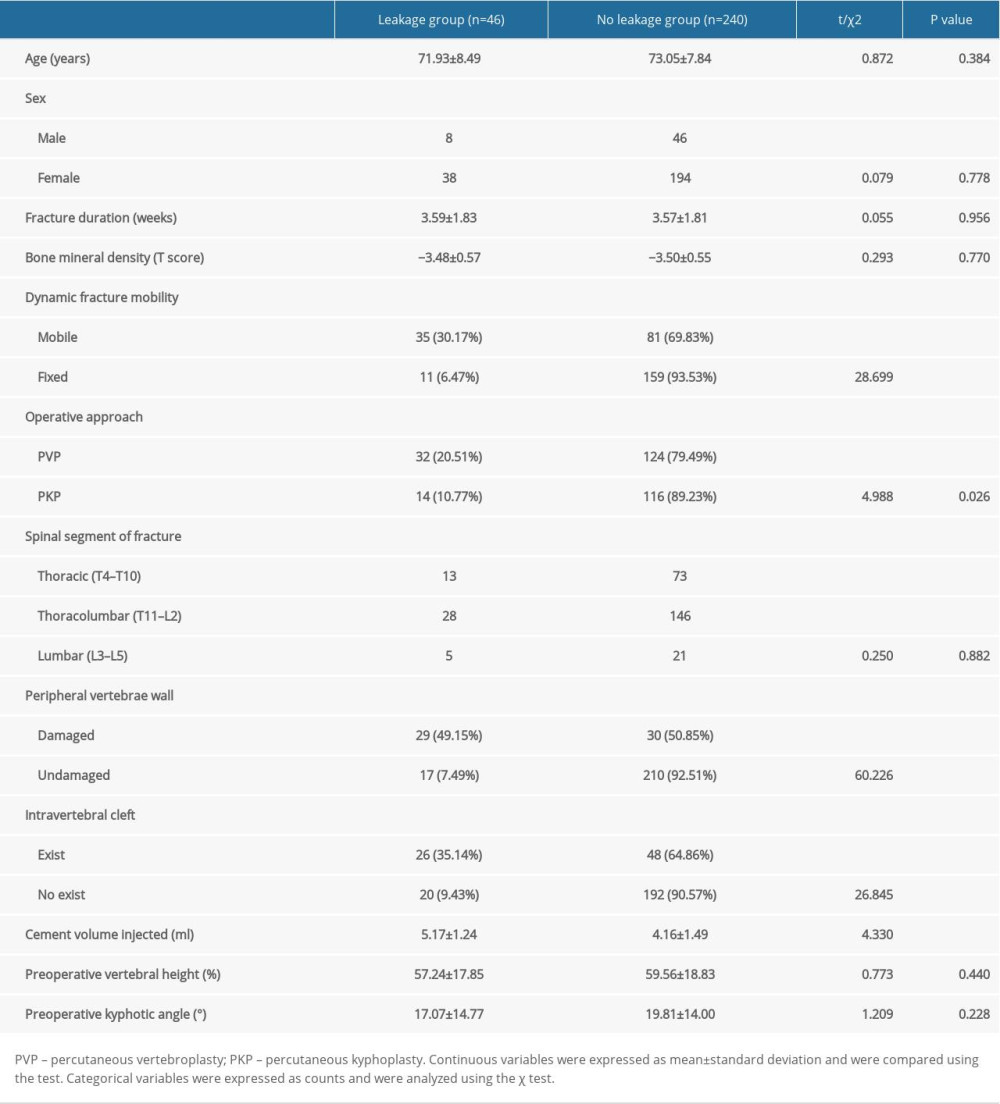 Table 1. Univariate analysis of risk factors for cement leakage.
Table 1. Univariate analysis of risk factors for cement leakage.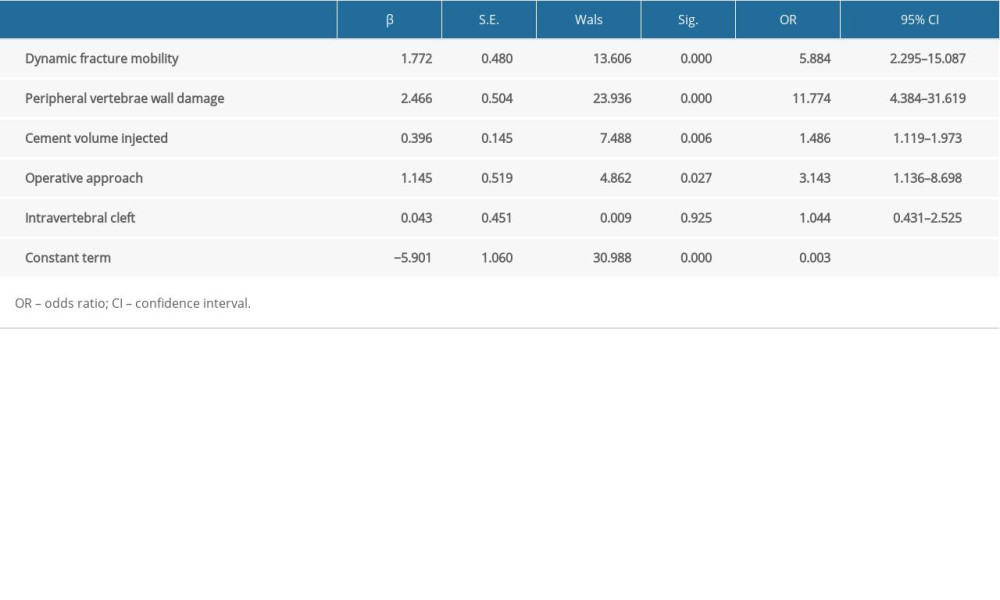 Table 2. Multivariate logistic regression analysis of risk factors for cement leakage.
Table 2. Multivariate logistic regression analysis of risk factors for cement leakage. Table 1. Univariate analysis of risk factors for cement leakage.
Table 1. Univariate analysis of risk factors for cement leakage. Table 2. Multivariate logistic regression analysis of risk factors for cement leakage.
Table 2. Multivariate logistic regression analysis of risk factors for cement leakage. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








