15 April 2022: Clinical Research
A Single-Center Retrospective Study of Selected Clinical Parameters and Intraoperative Fluid Management of Patients Undergoing Pancreatoduodenectomy
Patryk Zemła1BCDEF*, Justyna Bajak1BEF, Bartosz MolasyDOI: 10.12659/MSM.936114
Med Sci Monit 2022; 28:e936114
Abstract
BACKGROUND: Pancreatoduodenectomy is an extensive procedure with a very high risk of complications. Appropriate intraoperative fluid therapy is a subject of ongoing debate. The aim of this retrospective study was to analyze the relationship between selected preoperative parameters, intraoperative fluid therapy, and catecholamines administration during pancreatoduodenectomy.
MATERIAL AND METHODS: From 2011 through 2017, among pancreatoduodenectomies performed at a single university hospital, 192 patients met the inclusion criteria of the study: 105 (54.7%) males and 87 (45.3%) females with a mean age of 60.06 (±11.63) years. Correlations were assessed between sex, age, body mass index (BMI), selected comorbidities, surgery duration, American Society of Anesthesiologists (ASA) Physical Status (PS) scale, preoperative endoscopic retrograde cholangiopancreatography (ERCP) and intraoperative catecholamine administration, intraoperative fluid supply, red blood cell (RBC) concentrate and fresh frozen plasma (FFP) supply, blood loss, and diuresis.
RESULTS: A need for catecholamines has been shown to be more frequent in smokers (P=0.01), patients with cardiovascular comorbidities (P=0.037), high ASA PS scores (P=0.003), and preoperative ERCP (P=0.011). The need for intraoperative transfusion of RBC concentrate was more frequent in smokers (P=0.005). Surgical time was significantly longer in males (P=0.014). Among females, liberal intraoperative fluid therapy (>7.9 ml/kg/h) was more frequent in patients with thyroid comorbidities (P=0.003).
CONCLUSIONS: The findings of this retrospective study demonstrate the influence of comorbidities, ASA PS class, and catecholamine use on fluid therapy during pancreatoduodenectomy.
Keywords: Catecholamines, Comorbidity, Fluid Therapy, Pancreatic Cancer, Adult, Pancreaticoduodenectomy, Blood Transfusion, Female, Humans, Male, operative time
Background
Pancreatoduodenectomy is an extensive procedure with a very high risk of complications [1]. It is mainly used to treat tumors in the pancreaticoduodenal region. It has been shown that one of the primary methods to reduce perioperative and postoperative mortality is to have the procedure performed by a team of experienced surgeons in highly specialized surgical centers [2]. Intraoperative fluid supply is among the factors that can result in postoperative complications. Appropriate perioperative fluid management remains a subject of controversy [3–5]. Very low fluid volumes cause dehydration and electrolyte disturbances, while too much fluid reloading can lead to complications from the circulatory, respiratory, and gastrointestinal systems [6]. In the past, the recommended range of fluid administration in patients undergoing intra-abdominal surgery was 10 to 15 mL/kg/h with additional fluid supplementation depending on the volume of blood loss [7]. Many studies were performed comparing restrictive with liberal administration of fluid therapy, usually in favor of the former [8–13]. Few studies have compared the effects of preoperative factors on intraoperative fluid therapy and the need for intraoperative catecholamines usage. The aim of this study was to analyze the relationship between selected preoperative factors, including body mass index (BMI), comorbidities, the ASA PS (American Society of Anesthesiologists Physical Status classification system [14]), and intraoperative fluid therapy (restrictive and liberal), as well as the need for catecholamine administration during pancreatoduodenectomy. Our goal was to analyze preoperative factors that can influence intraoperative fluid therapy and catecholamine administration in order to be able to take them into account when planning and performing a major surgery such as pancreatoduodenectomy.
Material and Methods
PATIENTS’ CHARACTERISTICS:
Patients were treated in accordance with the most recent ESMO guidelines [18]. Multidisciplinary treatment included collaboration of a surgeon with a gastroenterologist, radiologist, and oncologist. Prior to surgical treatment, radiological and endoscopic examinations (endoscopic ultrasound [EUS] and computed tomography) were carried out. Pancreatoduodenectomies were performed by 3 experienced pancreatic surgeons.
ANALYSIS OF INTRAOPERATIVE FACTORS:
The relationships between sex, age, BMI, patients’ comorbidities, ASA PS score, duration of surgery and fluid administration (colloids, crystalloids), blood loss, and diuresis, and the need for red blood cell (RBC) concentrate and catecholamine supply during and after surgery were analyzed. The ASA PS classification was used to assess the clinical status of the patients before surgery [14]. Total fluid supply divided into colloids and crystalloids was determined. Fluid administration was also determined in ml/kg/h of surgery time, which allowed a BMI-independent analysis. Both the total and the ml/kg/h fluid supply were statistically analyzed. Indications for blood transfusion, catecholamine administration, and intraoperative anesthetic care were consistent with the ERAS (Enhanced Recovery After Surgery) protocol and NICE (National Institute for Health and Care Excellence) principles [19,20]. The indication for intraoperative transfusion of RBC concentrate was hemodynamic instability of the patient. Catecholamines (norepinephrine, epinephrine, dopamine, dobutamine) were administered during surgery due to patients’ hemodynamic instability associated with a drop in the mean blood pressure or stroke volume. Plasma (fresh frozen plasma [FFP]) was transfused for blood loss accompanied by coagulation disorders. The use of colloids (hydroxyethyl starch [HES]) was according to the guidelines for fluid therapy [21,22]. Values defining the adequate volume of fluids administered during pancreatoduodenectomy are not clearly defined in literature [8–13,23,24]. The median total fluid supply per kilogram of the patient’s weight per hour of surgery time [ml/kg/h] has been used as a criterion to differentiate between restrictive and liberal fluid therapy [4,25].
COMORBIDITIES:
We collected data on the prevalence of diabetes and cardiovascular diseases, including hypertension, ischemic heart disease, heart failure, arrhythmias, aortic aneurysms, thrombosis, atherosclerosis, severe regurgitation, and valvular stenosis. We also considered the prevalence of respiratory diseases such as asthma, chronic obstructive pulmonary disease (COPD), and emphysema. Considering speculations that active smoking before surgery can cause postoperative complications [26], regular smoking during the year preceding the surgery was included in the analysis as an independent risk factor. Obesity was defined by a BMI value ≥30 [27]. Renal conditions included in analysis were chronic kidney disease, need for dialysis, and post-nephrectomy status. Central nervous system (CNS) comorbidities included post-stroke status and CNS tumors. Pancreatic diseases other than adenocarcinoma were also analyzed, including chronic pancreatitis (CP) and a history of acute pancreatitis (AP). Thyroid diseases taken into account were hypothyroidism, hyperthyroidism, and autoimmune thyroid diseases. Endoscopic retrograde cholangiopancreatography (ERCP) with prosthesis placement (at least 2 weeks before surgery) was also analyzed.
ETHICS STATEMENT:
This was a retrospective study of medical records and all data had been fully anonymized before we accessed them. Written informed consent was obtained from all participants. All procedures were in accordance with the 1964 Helsinki Declaration regarding medical research involving human subjects and its later amendments or comparable ethical standards. Our retrospective analysis of patients’ medical records fell into a category exempt from Institutional Review Board (IRB) approval.
STATISTICAL ANALYSIS:
A descriptive analysis was performed. A confidence interval of 95% was used. The distribution of quantitative variables was analyzed. Variables closely following a normal distribution are expressed as means and standard deviation; non-normally distributed variables are given as medians with interquartile range. Correlation analysis was performed between sex, age, BMI, surgery time, comorbidities, ASA PS score and fluid supply (colloids, crystalloids), blood loss, diuresis, need for RBC transfusion during and after surgery, and need for catecholamine administration. The level of statistical significance was set at
Results
PATIENTS’ CHARACTERISTICS AND COMORBIDITIES:
The study group consisted of 105 males and 87 females with a mean age of 60.06 (±11.63) years; 159 (82.8%) patients were over 50 years of age and 101 (52.6%) were over 60 years of age. The characteristics of the patients and the prevalence of comorbidities are presented in Tables 1 and 2. The most common comorbidities were cardiovascular diseases, occurring in 107 (55.7%) patients.
PERIOPERATIVE PARAMETERS:
Perioperative parameters are shown in Tables 1 and 2. Most patients were in ASA PS class II (110 patients – 57.3%). The median fluid supply was 7.9 ml/kg/h and this value was considered the cut-off point between restrictive (≤7.9 ml/kg/h) and liberal (>7.9 ml/kg/h) fluid therapy [4].
CORRELATIONS BETWEEN PREOPERATIVE AND INTRAOPERATIVE PARAMETERS:
The analyzed correlations are presented in Tables 3–6. Only statistically significant results are discussed below.
A statistically significant difference in surgery duration was found between the males and females (
Catecholamine administration was more frequent in patients with preoperative cardiovascular disease (P=0.037), those after preoperative ERCP (P=0.011) and in smokers (P=0.01) (Table 4). The frequency of catecholamine administration increased along with the ASA PS class (P=0.003).
In the female group, a positive correlation was revealed between smoking and the supply of RBC concentrate after surgery (P=0.017). Thyroid comorbidities significantly increased the intraoperative fluid supply (P=0.001). In females, liberal fluid therapy (>7.9 ml/kg/h) was significantly more common compared to the male group (P=0.001). In contrast, males exhibited a correlation between obesity and the use of restrictive (≤7.9 ml/kg/h) fluid therapy (P=0.041). All qualitative correlations, including sex-dependent ones, are shown in Table 4.
CNS and respiratory diseases as well as smoking were associated with higher colloid supply in males (P=0.022, P=0.018, and P=0.028, respectively) (Table 5). CNS and pancreatic diseases other than adenocarcinoma were associated with higher crystalloid supply in females (P=0.026, P<0.001, respectively).
An analysis of parameters correlated with fluid supply revealed that higher supply (ml/kg/h) was related to female sex (P<0.001), obesity (P=0.03), thyroid disease (P=0.001), concomitant pancreatic disease (P<0.001), and ASA PS class (P<0.05) (fluid supply increased along with the ASA PS score). Additionally, in females, higher fluid reloading (ml/kg/h) was associated with smoking (P=0.044), CNS disease (P =0.059), ASA PS class (P<0.05) (fluid supply increased along with the ASA PS score), and thyroid disease (P=0.003). Intraoperative blood loss was higher in females with a history of smoking (P=0.051) and renal comorbidities (P=0.037). All quantitative-qualitative correlations are presented in Tables 5 and 6.
There was no correlation between FFP intraoperative supply and preoperative parameters. Age, diabetes, CNS diseases, pancreatic diseases did not correlate with intraoperative administration of catecholamines and perioperative RBC concentrate supply. Crystalloids supply (ml) was not correlated with preoperative variables. Diabetes, cardiovascular diseases, and preoperative ERCP did not correlate with surgery duration, fluid therapy, intraoperative blood loss, and diuresis.
Discussion
There are numerous reports on the relationships between perioperative parameters and postoperative complications as well as survival of post-pancreatoduodenectomy patients [1,9,11,25,28–30]. However, few of these considered preoperative parameters and their correlations with intraoperative factors that can affect postoperative complications. Our analysis also includes sex-dependent relationships, and this has rarely been done before.
The association between intra-pancreatoduodenectomy catecholamines and preoperative parameters was rarely investigated. In our study, we noted several correlations between comorbidities and the need for catecholamine supply. Table 4 shows the association of chronic diseases with intraoperative catecholamine administration.
In extensive procedures like pancreatoduodenectomy, intraoperative blood supply has a significant impact on the prevalence of postoperative complications such as delayed gastric emptying, pancreatic fistulas, or the length of postoperative hospital stay and overall survival [31–40]. Although Barreto et al [41] concluded that intraoperative blood transfusion was not associated with postoperative morbidity, they did find that patients with renal comorbidities required intraoperative transfusions more frequently, which is consistent with our observations. We also confirmed, consistent with the results obtained by Barreto et al, that higher ASA PS scores were associated with more frequent need for intraoperative blood supply. In males, higher BMIs were correlated with greater intraoperative blood loss (
The duration of surgery is a very important factor that can affect postoperative prognosis [34]. According to the literature, longer surgery is associated with increases in blood loss and fluid supply; these, in turn, might result in higher postoperative mortality [34,42,43]. It is therefore crucial to identify preoperative clinical factors that influence surgery duration (Tables 3 and 5). We found a statistically significant difference (
The determination of appropriate fluid therapy for extensive abdominal procedures, including pancreatoduodenectomy, remains controversial [4]. According to Myles et al [11], restrictive (≤3 liters of total fluid volume) and liberal fluid therapy (≥5 liters of total fluid volume) have both advantages and drawbacks. According to Gottin et al [45], significantly fewer postoperative complications (eg, postoperative fistula, abdominal collection, and hemorrhage) were found in the restrictive fluid therapy group (≤4 ml/kg/h) than in patients who had received liberal fluid therapy (≥12 ml/kg/h). Gilgien et al [46] also concluded that patients who received ≥4400 ml intravenous fluids during the first 24 h after surgery had more complications, especially pulmonary complications. Liberal fluid therapy may also lead to an increase in postoperative pancreatic fistula (POFF) formation [47]. Some authors argue that both ultra-restrictive and ultra-liberal fluid therapy are not optimal [48]. Patients managed with ultra-restrictive fluid regimens are prone to hypotension, acidosis, and reduced interstitial volume. In contrast, patients receiving high volumes of fluids can develop interstitial tissue edema and hemoglobin dilution. It is worth pointing out that a more restrictive fluid regimen during pancreatoduodenectomy does not lead to an increase in postoperative acute kidney injury [49]. However, it has also been suggested that there is no association between the amount of perioperative intravenous fluid administered and postoperative complications (including the length of hospital stay, estimated blood loss, pancreas specific complications or 30-day mortality) in patients undergoing pancreatoduodenectomy [50,51].
The factors determining the type of fluid therapy during the procedure are still a matter of debate [52]. In our study, we analyzed the relationship between preoperative parameters and the fluid therapy administered, with an additional division into colloids and crystalloids. Similar to Wang et al [25], we separated restrictive and liberal fluid therapy, taking as a cut-off point the median volume of fluids administered per kilogram of patient weight per hour of surgery (ml/kg/h), with a result of 7.9 ml/kg/h [25].
Iwasaki et al designed an interesting pilot study that compared the intraoperative fluid balance and perioperative complications in patients undergoing hepato-biliary-pancreatic surgery with or without stroke volume variation (SVV)-guided fluid management [53]. They concluded that the use of the SVV-guided fluid management protocol did not reduce intraoperative fluid balance, but increased the intraoperative fluid administration and might worsen postoperative oxygenation (
As seen in recently published work of Lapisatepun et al, postoperative fluid balance was a modifiable risk factor reducing the incidence of clinically relevant postoperative pancreatic fistula (CR-POFF) [54]. Similar findings were reported by Winer et al [55], in which CR-POFF was correlated with high 72-h net fluid balance. Higher positive postoperative fluid balance at postoperative day 3 should be avoided, especially in higher CR-POPF risk patients. However, a prospective randomized controlled trial shows that in high-volume centers there is not any difference in major postoperative complications such as 60-day mortality or clinically relevant complications (≥3) in Clavien-Dindo scale between restrictive (≤6 ml/kg/h) and liberal (≥12 ml/kg/h) fluid regimens [56]. Sandini et al evaluated 9 different fluid regimens [57], concluding that total volume administration of >5000 ml and >6000 ml of fluids was associated with increased complications and >6000 ml was associated with increased risk of sepsis. Additionally, a rate of <5 ml/kg/h was associated with increased risk of postoperative pancreatic fistula and sepsis, <6.8 ml/kg/h fluid regimen was associated with increased major morbidity and sepsis, and <8.2 ml/kg/h was associated with increased POFF. There were no effects observed on pulmonary complications, surgical site infections, length of stay, or mortality. Differing fluid therapy regimens do not have any significant effect on occurrence of delayed gastric emptying and its emptying time [58].
Patients’ BMI is another parameter considered in studies on intraoperative fluid therapy. Obesity can lead to a number of post-pancreatoduodenectomy complications, including wound infection, septic shock, and death [12,43,59,60], but other authors found no such relationship [4,61]. However, the relationship between BMI and fluid administration has only rarely been considered. Our study revealed that obesity in males was associated with restrictive (≤7.9 ml/kg/h) fluid therapy (
The relationship between sex and intraoperative fluid supply has not been clarified so far. According to some authors, females require higher fluid volumes more often than do male patients [9,62]. There are also reports that males, require higher fluid supply up to twice as often [63]. Our study demonstrated that intra-pancreatoduodenectomy liberal fluid therapy (>7.9 ml/kg/h) was significantly more frequent in females (
The risk for intraoperative and postoperative complications increases with higher ASA PS scores [14]. If surgery becomes complicated due to increased blood loss, patients are more likely to require increased fluid supply [11]. According to the literature, patients whose ASA PS score is ≥3 are about 1.5 times more likely to require increased fluid therapy [62,63]. In our study, this relationship was demonstrated with respect to colloids (Eta=0.28,
In our analysis, higher ASA PS scores were associated with greater intraoperative blood loss; in females, urine excretion increased as well (Eta=0.295,
Patients undergoing pancreatoduodenectomy are frequently older individuals (the mean age of our patients was 60.06±11.63 years) with concomitant diseases. Consequently, the rates of postoperative complications and mortality tend to be higher [66–68]. We determined the relationships between comorbidities and intraoperative fluid supply. Co-occurrence of thyroid disease was significantly correlated with higher fluid reloading during surgery (
To sum up:
A need for catecholamine use was more frequent in smokers and in patients with cardiovascular diseases, higher ASA PS scores, and pre-surgery ERCP. Intraoperative RBC transfusions were more frequently used in patients with renal diseases and higher ASA PS scores. Post-surgery RBC administration was more frequent in smokers and patients with respiratory diseases. In males, higher BMIs were associated with greater intraoperative blood loss, whereas in females, blood loss was greater in patients with renal comorbidities and smoking. The duration of surgery was significantly longer in males. Liberal intraoperative fluid therapy (>7.9 ml/kg/h) was significantly more frequent in women with higher ASA PS and those with thyroid disease.
There are some limitations to our research. It was a single-center retrospective study and it was restricted to 192 patients. There may be some bias that could have been introduced by the long duration of the study. Our results might have been influenced by comorbidities other than those included in the analysis. Despite the defined standards of intraoperative anesthesia, patient management could have included administration of some other medications that could also affect the patient’s condition. Two types of anastomoses were used – duct-to-mucosa and end-to-end invagination. However, as surgery duration and fluid therapy were comparable, we believe anastomotic techniques did not have any significant effect on the patients’ outcomes.
Conclusions
There are some preoperative factors that can influence intraoperative fluid therapy and catecholamine administration. Those that might have a significant impact on the intraoperative care are the ASA PS score, BMI, smoking, and certain comorbidities. These should therefore be taken into account when planning and performing a major surgery such as pancreatoduodenectomy.
Tables
Table 1. Patients’ characteristics and intraoperative parameters by sex.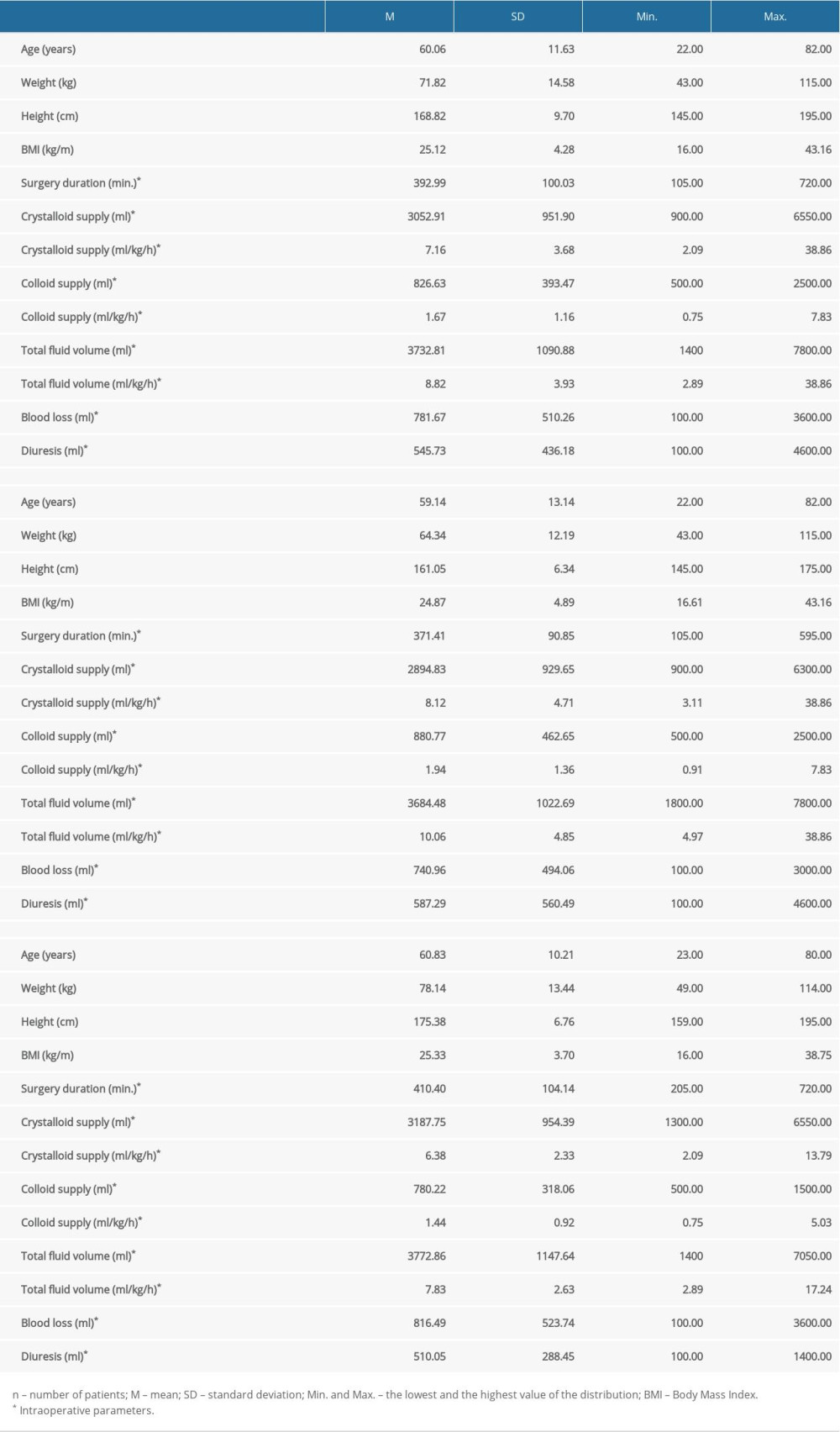 Table 2. Patients’ characteristics, intraoperative parameters, comorbidities, and pre-surgery ERCP by sex.
Table 2. Patients’ characteristics, intraoperative parameters, comorbidities, and pre-surgery ERCP by sex.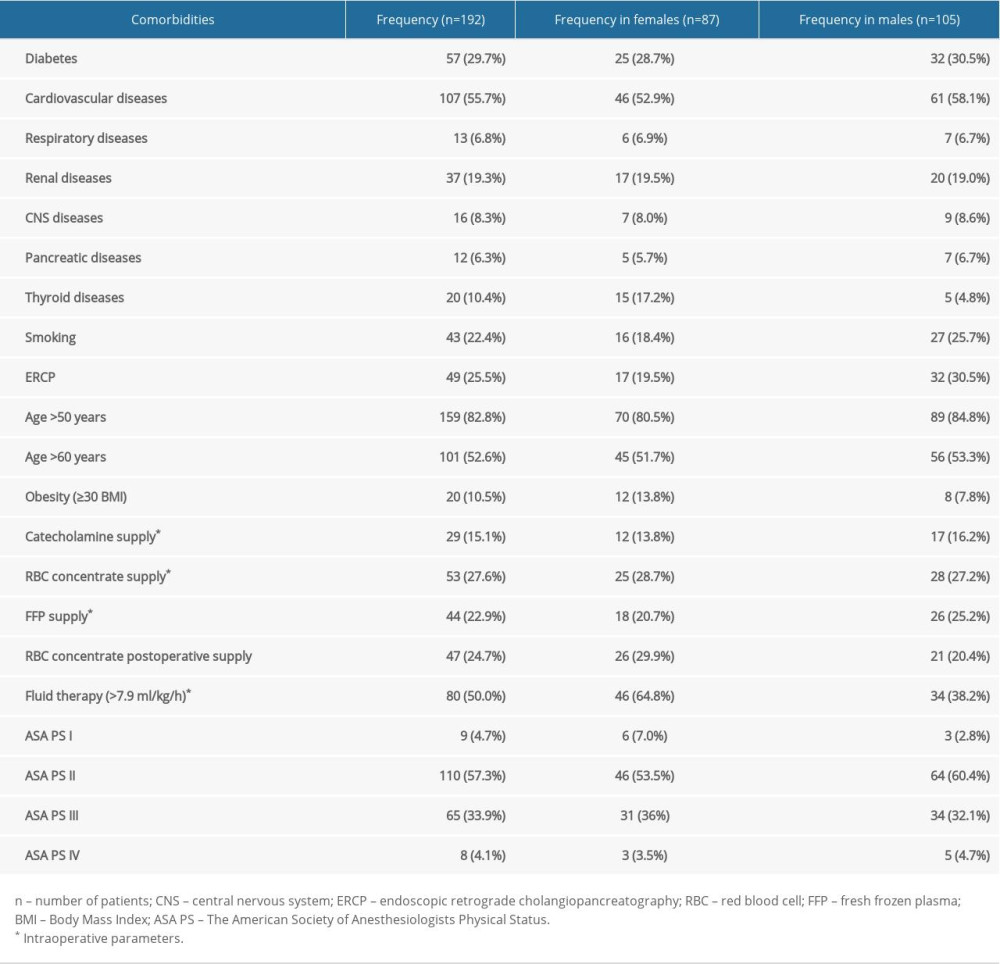 Table 3. Correlation analysis of preoperative versus perioperative variables by sex (numerical data in the table represent P value, quantitative analysis).
Table 3. Correlation analysis of preoperative versus perioperative variables by sex (numerical data in the table represent P value, quantitative analysis).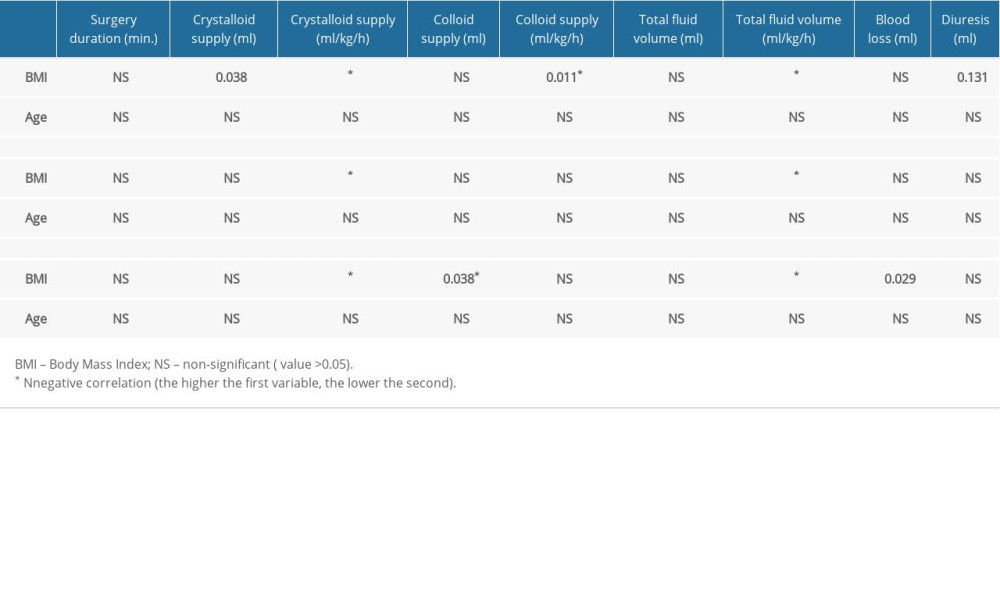 Table 4. Correlation analysis of preoperative versus perioperative variables by sex (numerical data in the table represent P value, qualitative analysis).
Table 4. Correlation analysis of preoperative versus perioperative variables by sex (numerical data in the table represent P value, qualitative analysis).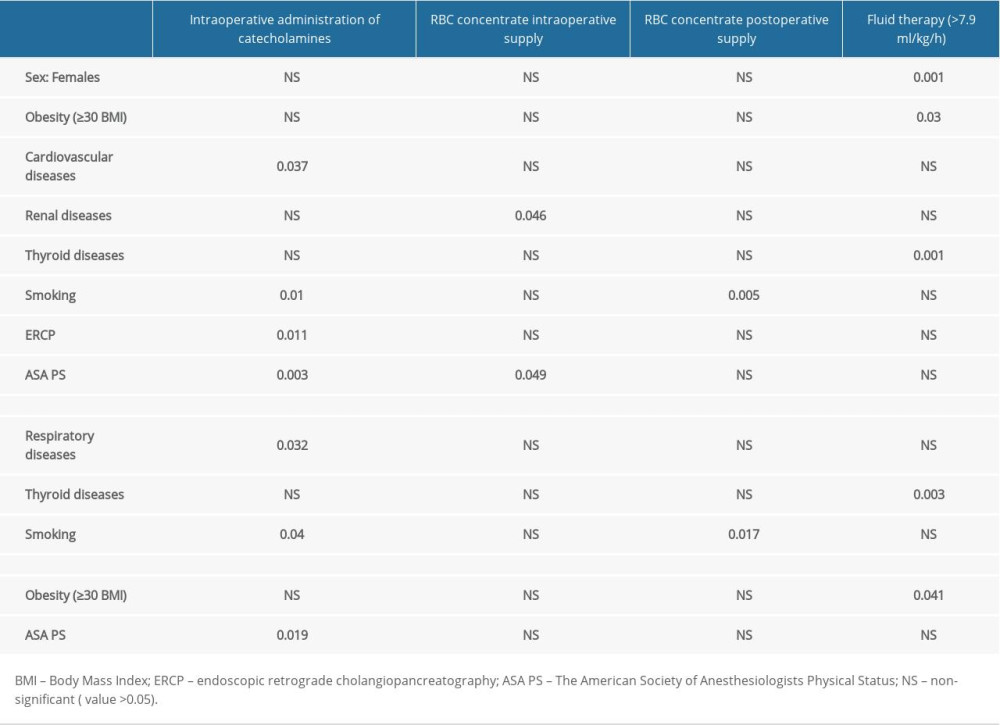 Table 5. Correlation analysis of preoperative versus perioperative variables by sex (numerical data in the table represent P value, qualitative-quantitative analysis).
Table 5. Correlation analysis of preoperative versus perioperative variables by sex (numerical data in the table represent P value, qualitative-quantitative analysis).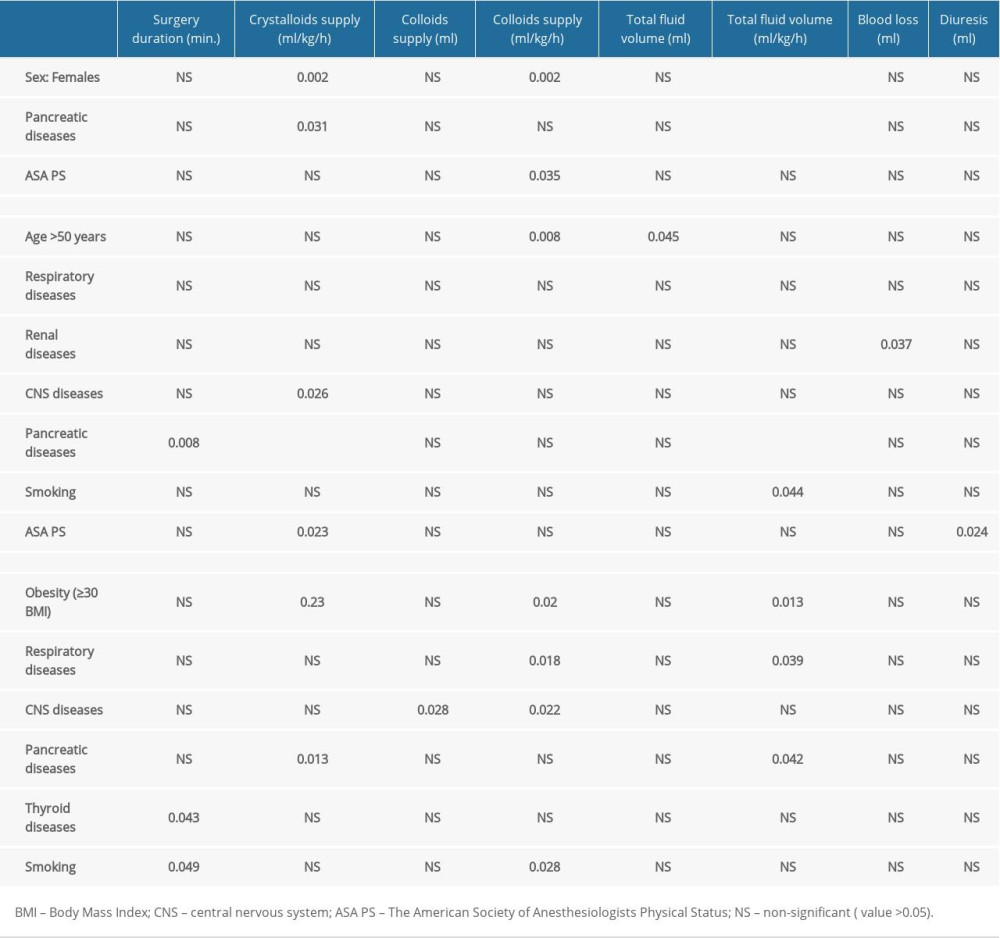 Table 6. Correlation analysis of preoperative versus perioperative variabless by sex (numerical data in the table represent P value, quantitative-qualitative analysis).
Table 6. Correlation analysis of preoperative versus perioperative variabless by sex (numerical data in the table represent P value, quantitative-qualitative analysis).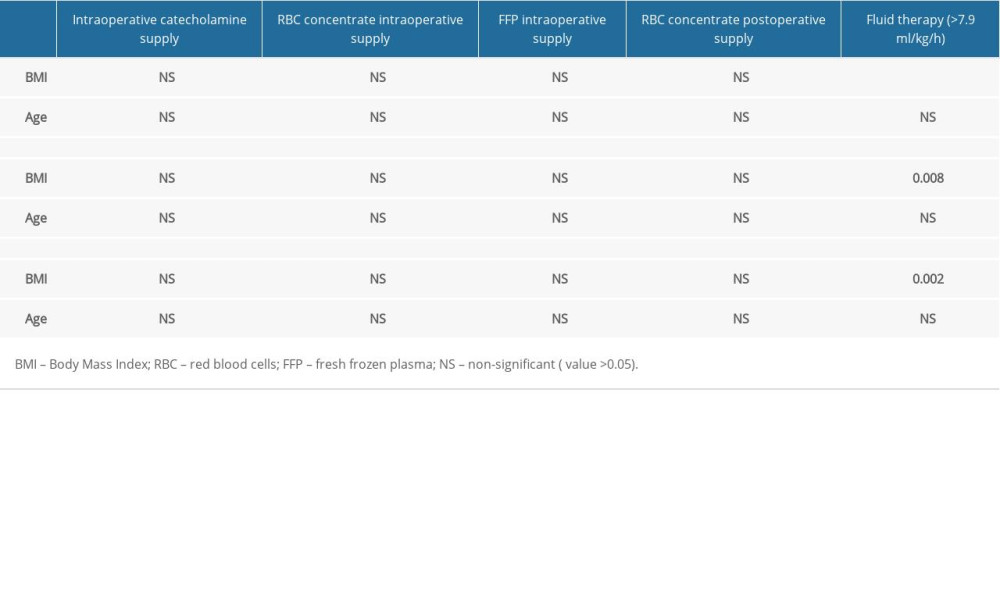
References
1. Karim SAM, Abdulla KS, Abdulkarim QH, Rahim FH, The outcomes and complications of pancreaticoduodenectomy (Whipple procedure): Cross sectional study: Int J Surg, 2018; 52; 383-87
2. Sosa JA, Bowman HM, Gordon TA, Importance of hospital volume in the overall management of pancreatic cancer: Ann Surg, 1998; 228(3); 429-38
3. Joshi G, Intraoperative fluid restriction improves outcome after elective gastrointestinal surgery: Anesth Analg, 2005(101); 601-5
4. Melis M, Marcon F, Masi A, Effect of intra-operative fluid volume on peri-operative outcomes after pancreaticoduodenectomy for pancreatic adenocarcinoma: J Surg Oncol, 2012; 105(1); 81-84
5. Rahbari NN, Zimmermann JB, Schmidt T, Meta-analysis of standard, restrictive and supplemental fluid administration in colorectal surgery: Br J Surg, 2009; 96(4); 331-41
6. Bundgaard-Nielsen M, Secher NH, Kehlet H, ‘Liberal’ vs. ‘restrictive’ perioperative fluid therapy-a critical assessment of the evidence: Acta Anaesthesiol Scand, 2009(53); 843-51
7. Eng OS, Melstrom LG, Carpizo DR, The relationship of perioperative fluid administration to outcomes in colorectal and pancreatic surgery: A review of the literature: J Surg Oncol, 2015; 111(4); 472-77
8. Lavu H, Sell NM, Carter TI, The HYSLAR trial: a prospective randomized controlled trial of the use of a restrictive fluid regimen with 3% hypertonic saline versus lactated Ringers in patients undergoing pancreaticoduodenectomy: Ann Surg, 2014; 260(3); 445-53 discussion 453–55
9. Eng OS, Goswami J, Moore D, Intraoperative fluid administration is associated with perioperative outcomes in pancreaticoduodenectomy: A single center retrospective analysis: J Surg Oncol, 2013; 108(4); 242-47
10. Bruns H, Kortendieck V, Raab HR, Antolovic D, Intraoperative fluid excess is a risk factor for pancreatic fistula after partial pancreaticoduodenectomy: HPB Surg, 2016; 2016; 1601340
11. Myles P, Bellomo R, Corcoran T, Restrictive versus liberal fluid therapy in major abdominal surgery (RELIEF): Rationale and design for a multicentre randomised trial: BMJ Open, 2017; 7(3); e015358
12. Wright GP, Koehler TJ, Davis AT, Chung MH, The drowning whipple: Perioperative fluid balance and outcomes following pancreaticoduodenectomy: J Surg Oncol, 2014; 110(4); 407-11
13. Weinberg L, Wong D, Karalapillai D, The impact of fluid intervention on complications and length of hospital stay after pancreaticoduodenectomy (Whipple’s procedure): BMC Anesthesiology, 2014(14); 35
14. Mayhew D, Mendonca V, Murthy BVS, A review of ASA physical status-historical perspectives and modern developments: Anaesthesia, 2019(74); 373-79
15. NCCN: Clinical Practice Guidelines in Oncology Pancreatic Adenocarcinoma [Internet], 2019 Available from: ttps://www.nccn.org/professionals/physician_gls/pdf/pancreatic.pdf
16. Kennedy GT, McMillan MT, Maggino L, Surgical experience and the practice of pancreatoduodenectomy: Surgery, 2017; 4(162); 812-22
17. Ecker BL, McMillan MT, Allegrini V, Risk factors and mitigation strategies for pancreatic fistula after distal pancreatectomy: Analysis of 2026 resections from the International, Multi-institutional Distal Pancreatectomy Study Group: Ann Surg, 2019; 269(1); 143-49
18. Ducreux M, Cuhna AS, Caramella C, Cancer of the pancreas: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up: Ann Oncol, 2015; 26(Suppl 5); v56-68 [Erratum in: Ann Oncol. 2017;28(Suppl. 4): iv167–68]
19. Melloul E, Lassen K, Roulin D, Guidelines for perioperative care for pancreatoduodenectomy: Enhanced recovery after surgery (ERAS) recommendations 2019: World J Surg, 2020; 44(7); 2056-84
20. National Institute for Health and Care Excellence: Intravenous fluid therapy in adults in hospital [Internet], 2017 Available from: https://www.nice.org.uk/guidance/cg174
21. Joshi Girish P, Intraoperative fluid management: UpToDate [Internet], 2020 Available from: https://www.uptodate.com/contents/intraoperative-fluid-management
22. He H, Liu D, Ince C, Colloids and the microcirculation: Anesth Analg, 2018; 126(5); 1747-54
23. Hwang G: Anesthesia for abdominal surgery, 1997, Philadelphia, Lippincott
24. Sendak M: Monitoring and managment of perioperative fluid and electrolyte therapy, 1993, Mosby, New York
25. Wang S, Wang X, Dai H, The effect of intraoperative fluid volume administration on pancreatic fistulas after pancreaticoduodenectomy: J Invest Surg, 2014; 27(2); 88-94
26. Thomsen T, Villebro N, M⊘ller AM, Interventions for preoperative smoking cessation: Cochrane Database Syst Rev, 2014; 2014(3); CD0002294
27. Apovian CM, Obesity: Definition, comorbidities, causes, and burden: Am J Manag Care, 2016; 22(7); 176-85
28. Kulemann B, Fritz M, Glatz T, Complications after pancreaticoduodenectomy are associated with higher amounts of intra- and postoperative fluid therapy: A single center retrospective cohort study: Ann Med Surg (Lond), 2017; 16; 23-29
29. Han IW, Kim H, Heo J, Excess intraoperative fluid volume administration is associated with pancreatic fistula after pancreaticoduodenectomy: A retrospective multicenter study: Medicine (Baltimore), 2017; 96(22); e6893
30. Wrzosek A, Jakowicka-Wordliczek J, Zajaczkowska R, Perioperative restrictive versus goal-directed fluid therapy for adults undergoing major non-cardiac surgery: Cochrane Database Syst Rev, 2019; 12(12); CD012767
31. Park HM, Park SJ, Shim JR, Perioperative transfusion in pancreatoduodenectomy: The double-edged sword of pancreatic surgeons: Medicine (Baltimore), 2017; 96(49); e9019
32. Seykora TF, Ecker BL, McMillan MT, The beneficial effects of minimizing blood loss in pancreatoduodenectomy: Ann Surg, 2019; 270(1); 147-57
33. Ross A, Mohammed S, Vanburen G, An assessment of the necessity of transfusion during pancreatoduodenectomy: Surgery, 2013; 154(3); 504-11
34. Ball CG, Pitt HA, Kilbane ME, Peri-operative blood transfusion and operative time are quality indicators for pancreatoduodenectomy: HPB (Oxford), 2010; 12(7); 465-71
35. Dosch AR, Grigorian A, Delaplain PT, Perioperative blood transfusion is associated with an increased risk for post-surgical infection following pancreaticoduodenectomy: HPB (Oxford), 2019; 21(11); 1577-84
36. Kneuertz PJ, Patel SH, Chu CK, Effects of perioperative red blood cell transfusion on disease recurrence and survival after pancreaticoduodenectomy for ductal adenocarcinoma: Ann Surg Oncol, 2011; 18(5); 1327-34
37. Zhang L, Liao Q, Zhang T, Blood transfusion is an independent risk factor for postoperative serious infectious complications after pancreaticoduodenectomy: World J Surg, 2016; 40(10); 2507-12
38. Yeh JJ, Gonen M, Tomlinson JS, Effect of blood transfusion on outcome after pancreaticoduodenectomy for exocrine tumour of the pancreas: Br J Surg, 2007; 94(4); 466-72
39. Sutton JM, Kooby DA, Wilson GC, Perioperative blood transfusion is associated with decreased survival in patients undergoing pancreaticoduodenectomy for pancreatic adenocarcinoma: A multi-institutional study: J Gastrointest Surg, 2014; 18(9); 1575-87
40. Zarzavadjian Le Bian A, Fuks D, Montali F, Predicting the severity of pancreatic fistula after pancreaticoduodenectomy: Overweight and blood loss as independent risk factors: Retrospective analysis of 277 patients: Surg Infect (Larchmt), 2019; 20(6); 486-91
41. Barreto SG, Singh A, Perwaiz A, Maximum surgical blood order schedule for pancreatoduodenectomy: A long way from uniform applicability!: Future Oncol, 2017; 13(9); 799-807
42. Chipaila J, Kato H, Iizawa Y, Prolonged operating time is a significant perioperative risk factor for arterial pseudoaneurysm formation and patient death following hemorrhage after pancreaticoduodenectomy: Pancreatology, 2020; 20(7); 1540-49
43. Chang EH, Sugiyama G, Smith MC, Obesity and surgical complications of pancreaticoduodenectomy: An observation study utilizing ACS NSQIP: Am J Surg, 2020; 220(1); 135-39
44. Tang T, Tan Y, Xiao B, Influence of body mass index on perioperative outcomes following pancreaticoduodenectomy: J Laparoendosc Adv Surg Tech A, 2021; 31(9); 999-1005
45. Gottin L, Martini A, Menestrina N, Perioperative fluid administration in pancreatic surgery: A comparison of three regimens: J Gastrointest Surg, 2020; 24(3); 569-77
46. Gilgien J, Hübner M, Halkic N, Perioperative fluids and complications after pancreatoduodenectomy within an enhanced recovery pathway: Sci Rep, 2020; 10(1); 17898
47. Cao X, Wang X, Zhao B, Correlation between intraoperative fluid administration and outcomes of pancreatoduodenectomy: Gastroenterol Res Pract, 2020; 2020; 8914367
48. Lavu H, Pitt HA, Optimal fluid management for patients undergoing pancreatoduodenectomy: Am J Surg, 2020; 220(2); 262-63
49. Mahmooth Z, Jajja MR, Maxwell D, Ultrarestrictive intraoperative intravenous fluids during pancreatoduodenectomy is not associated with an increase in post-operative acute kidney injury: Am J Surg, 2020; 220(2); 264-69
50. Gill P, Chua TC, Huang Y, Pancreatoduodenectomy and the risk of complications from perioperative fluid administration: ANZ J Surg, 2018; 88(4); E318-23
51. Huang Y, Chua TC, Gill AJ, Samra JS, Impact of perioperative fluid administration on early outcomes after pancreatoduodenectomy: A meta-analysis: Pancreatology, 2017; 17(3); 334-41
52. Voldby AWBB, Fluid therapy in the perioperative setting – a clinical review: J Intensive Care, 2016; 4; 27
53. Iwasaki Y, Ono Y, Inokuchi R, Intraoperative fluid management in hepato-biliary-pancreatic operation using stroke volume variation monitoring: A single-center, open-label, randomized pilot study: Medicine, 2020; 99(50); e23617
54. Lapisatepun W, Wongsa W, Chanthima P, Higher cumulative fluid following a pancreaticoduodenectomy as a single modifiable factor for post-operative pancreatic fistula: An analysis of risk factor: Asian J Surg, 2022; 45(1); 401-6
55. Winer LK, Dhar VK, Wima K, Perioperative net fluid balance predicts pancreatic fistula after pancreaticoduodenectomy: J Gastrointest Surg, 2018; 22(10); 1743-51
56. Grant F, Brennan MF, Allen PJ, Prospective randomized controlled trial of liberal vs restricted perioperative fluid management in patients undergoing pancreatectomy: Ann Surg, 2016; 264(4); 591-98
57. Sandini M, Fernández-Del Castillo C, Ferrone CR, Intraoperative fluid administration and surgical outcomes following pancreaticoduodenectomy: External validation at a tertiary referral center: World J Surg, 2019; 43(3); 929-36
58. Van Samkar G, Eshuis WJ, Bennink RJ, Intraoperative fluid restriction in pancreatic surgery: A double blinded randomised controlled trial: PLoS One, 2015; 10(10); e0140294
59. Lattimore CM, Kane WJ, Turrentine FE, Zaydfudim VM, The impact of obesity and severe obesity on postoperative outcomes after pancreatoduodenectomy: Surgery, 2021; 170(5); 1538-45
60. Zorbas K, Wu J, Reddy S, Obesity affects outcomes of pancreatoduodenectomy: Pancreatology, 2021; 21(4); 824-32
61. Ito Y, Kenmochi T, Irino T, The impact of obesity on perioperative outcomes of pancreaticoduodenectomy: Hepatogastroenterology, 2012; 59(120); 2618-22
62. Gill P, Chua TC, Huang Y, Pancreatoduodenectomy and the risk of complications from perioperative fluid administration: ANZ J Surg, 2018; 88(4); E318-23
63. Andrianello S, Marchegiani G, Bannone E, Clinical implications of intraoperative fluid therapy in pancreatic surgery: J Gastrointest Surg, 2018; 22(12); 2072-79
64. Ishihara S, Yokoyama T, Katayama K, Goal-directed therapy reduces fluid balance while maintaining hemodynamic stability in intraoperative management of pancreaticoduodenectomy: A retrospective comparative study: JA Clin Rep, 2018; 4(1); 7
65. Czajka S, Marczenko K, Włodarczyk M, Fluid therapy in patients undergoing abdominal surgery: A bumpy road towards individualized management: Adv Exp Med Biol, 2021; 1324; 63-72
66. Shia BC, Qin L, Lin KC, Age comorbidity scores as risk factors for 90-day mortality in patients with a pancreatic head adenocarcinoma receiving a pancreaticoduodenectomy: A National Population-Based Study: Cancer Med, 2020; 9(2); 562-74
67. Wiltberger G, Muhl B, Benzing C, Pancreaticoduodenectomy in the elderly patient: age-adapted risk assessment: Dig Surg, 2017; 34(1); 43-51
68. Uggeri F, Nespoli L, Sandini M, Analysis of risk factors for hemorrhage and related outcome after pancreatoduodenectomy in an intermediate-volume center: Updates Surg, 2019; 71(4); 659-67
Tables
 Table 1. Patients’ characteristics and intraoperative parameters by sex.
Table 1. Patients’ characteristics and intraoperative parameters by sex. Table 2. Patients’ characteristics, intraoperative parameters, comorbidities, and pre-surgery ERCP by sex.
Table 2. Patients’ characteristics, intraoperative parameters, comorbidities, and pre-surgery ERCP by sex. Table 3. Correlation analysis of preoperative versus perioperative variables by sex (numerical data in the table represent P value, quantitative analysis).
Table 3. Correlation analysis of preoperative versus perioperative variables by sex (numerical data in the table represent P value, quantitative analysis). Table 4. Correlation analysis of preoperative versus perioperative variables by sex (numerical data in the table represent P value, qualitative analysis).
Table 4. Correlation analysis of preoperative versus perioperative variables by sex (numerical data in the table represent P value, qualitative analysis). Table 5. Correlation analysis of preoperative versus perioperative variables by sex (numerical data in the table represent P value, qualitative-quantitative analysis).
Table 5. Correlation analysis of preoperative versus perioperative variables by sex (numerical data in the table represent P value, qualitative-quantitative analysis). Table 6. Correlation analysis of preoperative versus perioperative variabless by sex (numerical data in the table represent P value, quantitative-qualitative analysis).
Table 6. Correlation analysis of preoperative versus perioperative variabless by sex (numerical data in the table represent P value, quantitative-qualitative analysis). In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








