09 April 2022: Review Articles
A Review of the Neurobiological Mechanisms that Distinguish Between Loudness Recruitment and Hyperacusis
Lin Shi1EF, Rui Zhao1C, Xinxin Li1C, Wei Sun2A*, Xiuli Liu1ADDOI: 10.12659/MSM.936373
Med Sci Monit 2022; 28:e936373
Abstract
ABSTRACT: Loudness recruitment is a common symptom of hearing loss induced by cochlear lesions, which is defined as an abnormally fast growth of loudness perception of sound intensity. This is different from hyperacusis, which is defined as “abnormal intolerance to regular noises” or “extreme amplification of sounds that are comfortable to the average individual”. Although both are characterized by abnormally high sound amplification, the mechanisms of occurrence are distinct. Damage to the outer hair cells alters the nonlinear characteristics of the basilar membrane, resulting in aberrant auditory nerve responses that may be connected to loudness recruitment. In contrast, hyperacusis is an aberrant condition characterized by maladaptation of the central auditory system. Peripheral injury can produce fluctuations in loudness recruitment, but this is not always the source of hyperacusis. Hyperacusis can also be accompanied by aversion to sound and fear of sound stimuli, in which the limbic system may play a critical role. This brief review aims to present the current status of the neurobiological mechanisms that distinguish between loudness recruitment and hyperacusis.
Keywords: Auditory Diseases, Central, Hearing Loss, Sensorineural, Hyperacusis, Loudness Perception, Acoustic Stimulation, Cochlear Nerve, Hearing Loss, Humans
Background
Loudness of sound is the subjective perception of the strength and weakness of a sound, and is mainly influenced by the sound intensity or pressure level. Sounds at and just above their threshold are perceived as weak and soft sound. The loudness increases exponentially as the sound intensity increases (loudness=coefficient×sound intensity) [1]. When a patient develops sensorineural hearing loss, there is a rapid growth of the loudness at high-intensity sounds, even though the patient cannot hear low-intensity sounds. This phenomenon is often clinically referred to as loudness recruitment. Patients with recruitment often report a history of noise exposure, developed age-related hearing loss, using of ototoxic drugs, or developed inflammatory diseases that cause hair cell damage [1]. There is an auditory disorder that differs with patients with loudness recruitment, primarily described as reduction in sound tolerance, named hyperacusis.
When we introduce loudness recruitment and hyperacusis, it is important to first discuss the uncomfortable level (UCL), which refers to the lowest sound intensity level that causes discomfort [2]. The UCL for a normal person is ~100 decibels (dB HL), and patients with loudness recruitment often have sensorineural hearing loss, but the UCL remains essentially unchanged. In some patients with hyperacusis, the hearing threshold is maintained at 0 dB HL, but the UCL decreases to 70–80 dB, thus their hearing dynamic range or comfortable hearing range is significantly reduced (Figure 1) [3]. Loudness recruitment has long been considered a specific indicator of a cochlear lesion, showing only that the cochlea perceives abnormal amplification as the sound signal increases [4]. Loudness recruitment is therefore a clinical disorder rather than an independent disease. Hyperacusis, in contrast, is often a diagnosis describing a patient’s intolerance to “loud” sound. Because of the similarities between these 2 symptoms, some clinicians have used “loudness recruitment” and “hyperacusis” interchangeably. This brief review aims to present the current status of the neurobiological mechanisms that can distinguish between loudness recruitment and hyperacusis.
Definition and Pathological Mechanism of Loudness Recruitment
Loudness recruitment means that the affected ear perceives an abnormally rapid increase in loudness as the sound intensity increases [5, 6]. These patients have a higher hearing threshold than normal subjects, yet they perceive a faster increase in loudness than normal subjects when the volume of external sound increases. When the test sound reaches a certain intensity, the perceived loudness of a person with hearing loss is virtually the same that of a person with normal hearing. There are 5 types of loudness growth functions based on intensity-loudness function measured in patients with sensorineural hearing loss [7–9] (Figure 2): (1) No recruitment, in which the affected ear and the normal ear have equal changes in loudness to same sound intensity and the intensity-loudness function of the ear with hearing loss is parallel with the normal ear; (2) Partial recruitment, in which the slope of intensity-loudness function of the ear with hearing loss is higher than normal ear, but the loudness of the ear with hearing loss never reaches to the normal ear; (3) Complete recruitment, in which the loudness reaches to a certain level and the affected ear perceives the same intensity as the normal ear [6], which is the most common type of recruitment; (4) Excessive recruitment, in which the patient perceives an even louder response in the ear with hearing loss, greater than the sound perceived by the normal ear, and this change in loudness profile is the same as the hyperacusis described by Cox et al; (5) Counter-recruitment, in which the loudness increase in the affected ear increased slower than in the normal ear.
The mechanism of loudness recruitment is still controversial, and most scholars believe that it may be related to a lesion of the cochlea. As outer hair cells act as an active amplifier on the basilar membrane movement, damage to the outer hair cells will lead to reduction of amplification of the input-output function of the cochlea with low-intensity sounds. As the outer hair cell motility contributes less effect to high-intensity sounds, this will result in a steep input-output function of the cochlea (Figure 3).Therefore, some scholars have suggested that impaired cochlear amplification due to outer hair cell damage is the main mechanism of loudness recruitment [5]. This speculation mainly focuses on assessing the effect of cochlear lesions on loudness, considering there is less influence of the auditory central system on auditory loudness perception.
As cochlear signals are transmitted from the spiral ganglion neurons to the brain, it has been hypothesized that loudness recruitment is the result of an increase in overall auditory nerve firing if “loudness perception” is represented by cochlear response.
Heinz et al investigated the firing rate of the auditory nerve after noise exposure in cats and found that the firing rate of the peripheral nerve did not increase with noise frequencies and the input-output curve did not become steeper [10,11]. Their results suggested that the equal-loudness phenomena found in “complete or excessive recruitment” cannot be explained by cochlear output measured by auditory nerve firing rates after noise exposure [11].
Furthermore, the study by Young et al refuted the assumption that loudness recruitment is induced by an increase in auditory nerve firing because no steepening of the auditory nerve firing rate function was observed, despite the increase of the basilar membrane function. The theory that loudness recruitment is caused by injury to the auditory nerves, resulting in a drop in the auditory nerve response threshold, has also been rule out as there was no change in the auditory nerve response threshold [11].
To study the central auditory system changes after cochlear damage, Cai et al [12] studied the effects of 112 dB SPL noise on cochlear nucleus activity in cats for 4 h. They found that the input-output function of firing rate of non-primary-like neurons in the ventral cochlear nucleus, especially chopper, were consistent with the changes in loudness recruitment. The chopper cells were responsible for identifying brief pure tones, and there is an abnormally rapid increase in firing rates of the choppers to stimulation of tone-burst tones during cochlear injury, supporting the argument that there is a central involvement in loudness recruitment. However, there are still questions about when and where the precise site of increased sound perception occurs after a peripheral lesion forms. Although most previous studies suggested that recruitment is caused by reduction of cochlear amplification due to outer hair cell loss, a recent study by Lefler et al demonstrated that endolymphatic hydrops can induce loudness recruitment without causing outer hair cell damage. This study provides a new mechanism of recruitment that may present in patients with Meniere’s disease [13].
Definition and Mechanism of Hyperacusis
Hyperacusis is referred to as “abnormal intolerance to ordinary sounds” or “excessive amplification of sounds that are comfortable to normal people” (Figure 2). It is suggested that hyperacusis is evoked by low-intensity or common sounds, rather than specific sounds, which distinguishes it from “acoustophobia” and “anosmia” [14].
Many studies have suggested that hyperacusis has a centrally-driven source, as opposed to the “passive central involvement” theory of loudness recruitment. For example, when normal people wear earplugs for a few weeks, they will perceive the surrounding sound as noisy when they remove the earplugs, whereas when these subjects wear hearing aids, the UCL increases when they remove them [15]. Studies have suggested that the abnormal response of the central auditory system plays a critical role in sound perception changes. This hypothesis is referred to as central plasticity or central gain change. Central gain suggests that a rapid compensatory increase in firing rate will occur in the central auditory system as peripheral input decreases. It may be caused by the regulation of the balance of central inhibition and excitatory neurotransmission. When the output of the auditory afferents is reduced, it reduces the input of the inhibitory circuits in the central system and increases central output. This can compensate for the rapid reduction of afferent impulses from the peripheral system [16].
Studies in animal models of noise and ototoxic drugs have found plastic changes in multiple levels of the central auditory system, including enhanced neural synchronization, increased spontaneous excitability in the cochlear nucleus and the inferior colliculus, and expansion and reorganization of cortical response areas [17,18]. Because of the multiple transit points in sound transmission, the onset of central gain has been investigated using noise exposure models. An increase of the amplitude of acoustic startle reflex (ASR) was found in noise-treated animals compared to the controls, even those with hearing loss [19–21]. One study discovered that after 105-dB noise exposure, guinea pigs’ hearing threshold increased, but the electrophysiological responses in the inferior colliculus (IC) was significantly higher than before the exposure [17]. In another study, a decrease in the compound action potential (CAP) of the cochlea, no change in the cochlear nucleus, and a significant increase in the IC and the AC discharges were found after 120-dB noise exposure in guinea pigs [20]. This study suggested that the onset of loss of central gain may lie between the cochlear nucleus and the inferior colliculus.
Studies have shown that the site of central gain sometimes changes dynamically with the degree and duration of hearing loss. A study of animals exposed to noise at 120 dB for 2 weeks found that the CAP, IC, and AC potentials also tended to change dynamically with increasing noise exposure time [21–23]. These nuclei do not simply transmit from bottom to top; instead, the central gain in the AC occurs first, followed by compensations in the inferior colliculus. Therefore, it has been suggested that the central gain is due to a weakened inhibitory projection from the auditory cortex to the inferior auditory nuclei, which leads to an increase in the firing rate [24] and even hyperacusis.
The Difference and Connection Between Loudness Recruitment and Hyperacusis
Loudness recruitment and hyperacusis are both changes in the perception of sound loudness. However, there is a difference between them. Loudness recruitment is generally manifested after the onset of sensorineural deafness, which is characterized by an abnormal amplification of the perception of medium- to high-intensity sounds, whereas hyperacusis does not necessarily present with hearing loss, but there is a decrease in tolerance to everyday sounds, either medium-to-high-intensity sounds or sounds that others do not find loud. In addition, loudness recruitment does not account for the patient’s subjective emotions regarding sound, and the degree of hyperacusis often varies with the patient’s emotions. For example, exertion and anxiety can exacerbate hyperacusis, while hyperacusis can be triggered by seasonal or other factors.
In recent years, variation in the limbic system has also been studied [25]. The amygdala, striatum, and hippocampus are also involved in the projections from the medial geniculate body (MGB) to the auditory cortex (AC) in sound signaling. Loudness recruitment occurs mainly with peripheral hearing damage, while hyperacusis may result from lesions in the auditory or limbic centers, such as Williams’ syndrome, a genetic defective disorder occurring in the vasculature, connective tissue, and central nervous system, but 60% of patients present with hyperacusis, in which more than half of patients have normal hearing [26,27].
Since noise exposure causes damage to outer hair cells first, the animal models of noise-induced hyperacusis explained the mechanism by which hyperacusis occurs after outer hair cell damage. In contrast, a study of ototoxic hearing loss caused by exposure to carboplatin found that the specific destruction of inner hair cells resulted in normal DPOAE and a significant decrease in CAP in mice, with a small increase in the IC discharge and a more pronounced increase in the AC. This suggests that peripheral hearing loss, regardless of lesions in the outer or inner hair cells, will cause cortical compensatory changes [28].
In a recent study, rats were exposed to noise of 16–20 kHz 104 dB SPL for 12 weeks. Behavioral sound responses measured by reaction time were assessed before, during, and 2 months after the noise exposure. Loudness recruitment was observed in the area of hearing loss during exposure, but clear signs of hyperacusis began to appear at the edges of the hearing loss frequencies. The above experiments suggest that loudness recruitment and hyperacusis can occur simultaneously [29].
Conclusions
This review has shown that although cochlear injury results in abnormal conduction in the auditory nerve, the mechanisms leading to loudness recruitment require further study. It has been speculated that the nonlinear properties of the cochlear response are altered by lesions of outer hair cells, leading to loudness recruitment.
Hyperacusis, on the other hand, is an abnormal sound perception change dominated by central active compensation, which differs from loudness recruitment. Hyperacusis may also involve the limbic system, accompanied by fear and aversion to sound.
Figures
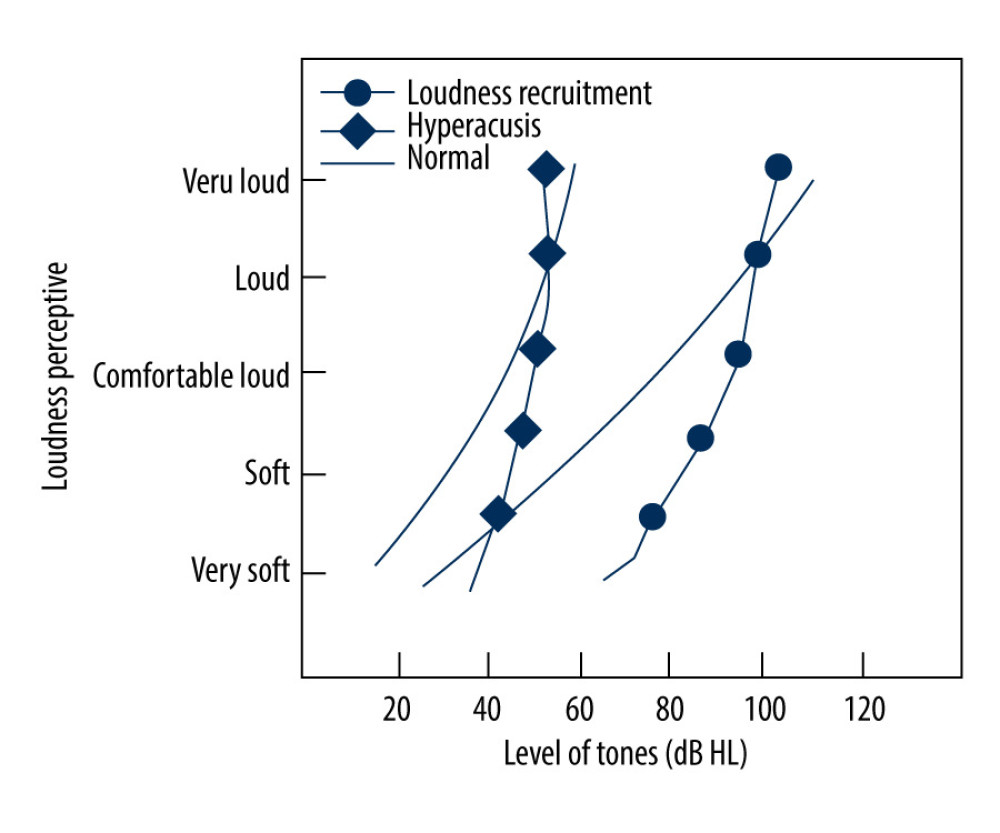 Figure 1. Loudness models for normal subjects, patients with loudness recruitment, and patients with hyperacusis (Cox, 1997). The maximum discomfort threshold for a normal person is 100 dB HL. Patients with loudness recruitment have a largely unchanged maximum discomfort threshold, although they often have sensorineural deafness, while some patients with hyperacusis have an increased hearing threshold, the maximum discomfort threshold falls to 70–80 dB instead, thus the range of hearing tolerance is significantly reduced.
Figure 1. Loudness models for normal subjects, patients with loudness recruitment, and patients with hyperacusis (Cox, 1997). The maximum discomfort threshold for a normal person is 100 dB HL. Patients with loudness recruitment have a largely unchanged maximum discomfort threshold, although they often have sensorineural deafness, while some patients with hyperacusis have an increased hearing threshold, the maximum discomfort threshold falls to 70–80 dB instead, thus the range of hearing tolerance is significantly reduced. 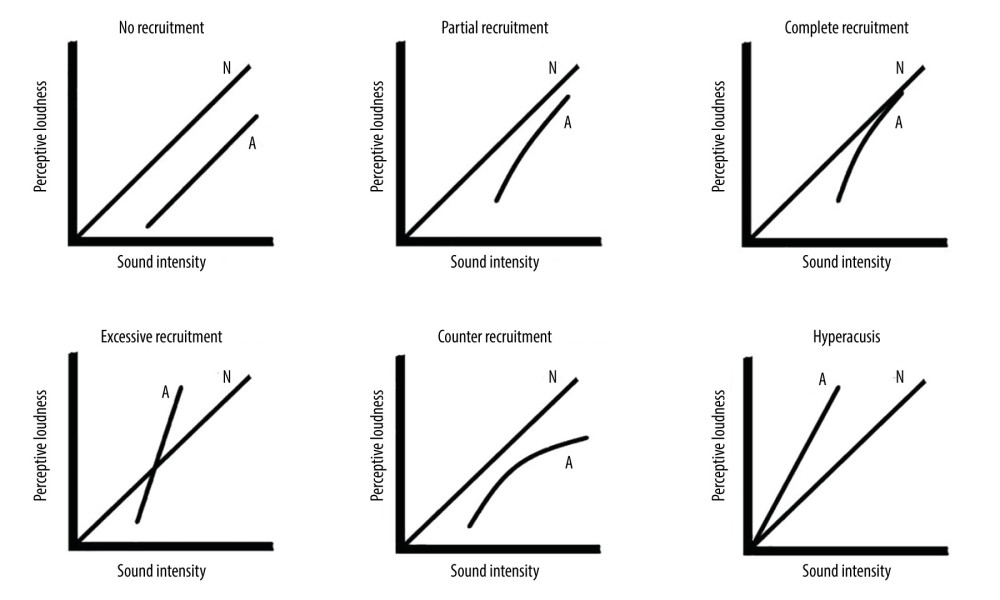 Figure 2. Diagram of Loudness recruitment classification and hyperacusis. N – normal individuals, A – abnormal patients.
Figure 2. Diagram of Loudness recruitment classification and hyperacusis. N – normal individuals, A – abnormal patients.  Figure 3. Input-output curve for normal ear and loudness recruitment ear at different frequencies.
Figure 3. Input-output curve for normal ear and loudness recruitment ear at different frequencies. References
1. May BJ, Little N, Saylor S, Loudness perception in the domestic cat: Reaction time estimates of equal loudness contours and recruitment effects: J Assoc Res Otolaryngol, 2009; 10(2); 295-308
2. Mariam M, Delb W, Harris AR, Detection of the uncomfortable loudness level by electroencephalographic data: A novelty detection approach using habituation correlates: Annu Int Conf IEEE Eng Med Biol Soc, 2008; 2008; 4976-79
3. Cox RM, Alexander GC, Taylor IM, Gray GA, The contour test of loudness perception: Ear Hear, 1997; 18(5); 388-400
4. Yankaskas K, Prelude: Noise-induced tinnitus and hearing loss in the military: Hear Res, 2013; 295; 3-8
5. Joris PX, Recruitment of neurons and loudness. Commentary on “Encoding intensity in ventral cochlear nucleus following acoustic trauma: Implications for loudness recruitment” by Cai et al. J Assoc Res Otolaryngol. 2009;10(1):5–22: J Assoc Res Otolaryngol, 2009; 10(1); 1-4
6. Rance G, Starr A, Pathophysiological mechanisms and functional hearing consequences of auditory neuropathy: Brain, 2015; 138(Pt 11); 3141-58
7. Moore BC, Glasberg BR, A revised model of loudness perception applied to cochlear hearing loss: Hear Res, 2004; 188(1–2); 70-88
8. Salmivalli A, The clinical and audiological pattern due to acoustic trauma: Acta Otolaryngol, 1966(Suppl 224); 39
9. Anari M, Axelsson A, Eliasson A, Magnusson L, Hypersensitivity to sound – questionnaire data, audiometry and classification: Scand Audiol, 1999; 28(4); 219-30
10. Shi L, Liu K, Wang H, Noise induced reversible changes of cochlear ribbon synapses contribute to temporary hearing loss in mice: Acta Otolaryngol, 2015; 135(11); 1093-102
11. Heinz MG, Issa JB, Young ED, Auditory-nerve rate responses are inconsistent with common hypotheses for the neural correlates of loudness recruitment: J Assoc Res Otolaryngol, 2005; 6(2); 91-105
12. Cai S, Ma WL, Young ED, Encoding intensity in ventral cochlear nucleus following acoustic trauma: implications for loudness recruitment: J Assoc Res Otolaryngol, 2009; 10(1); 5-22
13. Lefler SM, Duncan RK, Goodman SS, Measurements from ears with endolymphatic hydrops and 2-hydroxypropyl-beta-cyclodextrin provide evidence that loudness recruitment can have a cochlear origin: Front Surg, 2021; 8; 687490
14. Aazh H, McFerran D, Salvi R, Insights from the First International Conference on Hyperacusis: Causes, evaluation, diagnosis and treatment: Noise Health, 2014; 16(69); 123-26
15. Formby C, Sherlock LP, Gold SL, Adaptive plasticity of loudness induced by chronic attenuation and enhancement of the acoustic background: J Acoust Soc Am, 2003; 114(1); 55-58
16. Schaette R, Kempter R, Development of tinnitus-related neuronal hyperactivity through homeostatic plasticity after hearing loss: A computational model: Eur J Neurosci, 2006; 23(11); 3124-38
17. Mulders WH, Robertson D, Hyperactivity in the auditory midbrain after acoustic trauma: Dependence on cochlear activity: Neuroscience, 2009; 164(2); 733-46
18. Weisz N, Muller S, Schlee W, The neural code of auditory phantom perception: J Neurosci, 2007; 27(6); 1479-84
19. Chen GD, Stolzberg D, Lobarinas E, Salicylate-induced cochlear impairments, cortical hyperactivity and re-tuning, and tinnitus: Hear Res, 2013; 295; 100-13
20. Popelar J, Syka J, Berndt H, Effect of noise on auditory evoked responses in awake guinea pigs: Hear Res, 1987; 26(3); 239-47
21. Norena AJ, Moffat G, Blanc JL, Neural changes in the auditory cortex of awake guinea pigs after two tinnitus inducers: salicylate and acoustic trauma: Neuroscience, 2010; 166(4); 1194-209
22. Sun W, Deng A, Jayaram A, Gibson B, Noise exposure enhances auditory cortex responses related to hyperacusis behavior: Brain Res, 2012; 1485; 108-16
23. Sun W, Zhang L, Lu J, Noise exposure-induced enhancement of auditory cortex response and changes in gene expression: Neuroscience, 2008; 156(2); 374-80
24. Auerbach BD, Rodrigues PV, Salvi RJ, Central gain control in tinnitus and hyperacusis: Front Neurol, 2014; 5; 206
25. Gunbey HP, Gunbey E, Aslan K, Limbic-auditory interactions of tinnitus: An evaluation using diffusion tensor imaging: Clin Neuroradiol, 2017; 27(2); 221-30
26. Johnson LB, Comeau M, Clarke KD, Hyperacusis in Williams syndrome: J Otolaryngol, 2001; 30(2); 90-92
27. Miani C, Passon P, Bracale AM, Treatment of hyperacusis in Williams syndrome with bilateral conductive hearing loss: Eur Arch Otorhinolaryngol, 2001; 258(7); 341-44
28. Hofstetter P, Ding D, Salvi R, Induction of spontaneous otoacoustic emissions in chinchillas from carboplatin-induced inner hair cell loss: Hear Res, 2000; 150(1–2); 132-36
29. Radziwon K, Auerbach BD, Ding D, Noise-Induced loudness recruitment and hyperacusis: Insufficient central gain in auditory cortex and amygdala: Neuroscience, 2019; 422; 212-27
Figures
 Figure 1. Loudness models for normal subjects, patients with loudness recruitment, and patients with hyperacusis (Cox, 1997). The maximum discomfort threshold for a normal person is 100 dB HL. Patients with loudness recruitment have a largely unchanged maximum discomfort threshold, although they often have sensorineural deafness, while some patients with hyperacusis have an increased hearing threshold, the maximum discomfort threshold falls to 70–80 dB instead, thus the range of hearing tolerance is significantly reduced.
Figure 1. Loudness models for normal subjects, patients with loudness recruitment, and patients with hyperacusis (Cox, 1997). The maximum discomfort threshold for a normal person is 100 dB HL. Patients with loudness recruitment have a largely unchanged maximum discomfort threshold, although they often have sensorineural deafness, while some patients with hyperacusis have an increased hearing threshold, the maximum discomfort threshold falls to 70–80 dB instead, thus the range of hearing tolerance is significantly reduced. Figure 2. Diagram of Loudness recruitment classification and hyperacusis. N – normal individuals, A – abnormal patients.
Figure 2. Diagram of Loudness recruitment classification and hyperacusis. N – normal individuals, A – abnormal patients. Figure 3. Input-output curve for normal ear and loudness recruitment ear at different frequencies.
Figure 3. Input-output curve for normal ear and loudness recruitment ear at different frequencies. In Press
06 Mar 2024 : Clinical Research
Comparison of Outcomes between Single-Level and Double-Level Corpectomy in Thoracolumbar Reconstruction: A ...Med Sci Monit In Press; DOI: 10.12659/MSM.943797
21 Mar 2024 : Meta-Analysis
Economic Evaluation of COVID-19 Screening Tests and Surveillance Strategies in Low-Income, Middle-Income, a...Med Sci Monit In Press; DOI: 10.12659/MSM.943863
10 Apr 2024 : Clinical Research
Predicting Acute Cardiovascular Complications in COVID-19: Insights from a Specialized Cardiac Referral Dep...Med Sci Monit In Press; DOI: 10.12659/MSM.942612
06 Mar 2024 : Clinical Research
Enhanced Surgical Outcomes of Popliteal Cyst Excision: A Retrospective Study Comparing Arthroscopic Debride...Med Sci Monit In Press; DOI: 10.12659/MSM.941102
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952








