13 June 2022: Database Analysis
Retrospective Study from a Single Center to Identify Risk Factors Associated with Reinfection in Patients Undergoing 2-Stage Revision Surgery for Periprosthetic Joint Infection
Bingshi Zhang1ABCE, Zeming Liu1CE, Sikai Liu1BC, Bo Liu1BC, Mengnan Li1B, Wenao Li1E, Yongtai Han1ABCDE*DOI: 10.12659/MSM.936973
Med Sci Monit 2022; 28:e936973
Abstract
BACKGROUND: This study identified risk factors associated with reinfection and created a scoring system designed for patients with periprosthetic joint infection (PJI) who have undergone prosthetic resection and spacer implantation.
MATERIAL AND METHODS: Patients who underwent 2-stage revision for PJI from January 2010 to December 2017 were retrospectively analyzed in this study. Patients were divided into 2 groups: infection recurrence and infection cure. Demographic and clinical information, laboratory test results, and radiological images that were potentially associated with infection were obtained and analyzed.
RESULTS: Seven independent risk (protective) factors for infection recurrence in patients with PJI who underwent 2-stage hip revision surgery were identified: C-reactive protein level, type of bacterial infection, age, immunosuppression, albumin/globulin ratio, glucose level, and magnetic resonance imaging findings. Finally, a weighted scoring system of 100-mark system was established and the area under the curve was 0.965 (95% confidential interval=0.947-0.983). The predictive scores for low risk (≤30%), moderate risk (31-69%), and high risk (≥70%) of infection recurrence were ≤45, 46-77, and ≥78, respectively.
CONCLUSIONS: For patients with PJI who had already undergone joint resection and spacer implantation, this newly established scoring system might help determine the accurate risk of infection recurrence after a definitive new prosthesis implantation. Patients with scores greater than 78 points would be considered very likely to have an infection recurrence. Therefore, the second-stage revision surgery should be changed to an additional anti-infection treatment or a debridement surgery instead of a definitive prosthesis implantation surgery.
Keywords: Arthroplasty, Replacement, Hip, Reoperation, Recurrence, Research Design, Anti-Bacterial Agents, Arthritis, Infectious, Humans, Prosthesis-Related Infections, reinfection, Risk Factors
Background
Periprosthetic joint infection (PJI) after total hip arthroplasty (THA) is almost the most challenging postoperative complication for hip surgeons [1]. PJI can largely compromise the prognosis of patients, result in additional hospitalization stays, and in some situations be fatal [2–4]. Delanois et al reported that infection was the most common etiology for revision total knee arthroplasty (20.4%) in the United States [5]. According to the PJI treatment guideline from the Infectious Diseases Society of America and Musculoskeletal Infection Society (MSIS), the predominant treatment for acute PJI is debridement, antibiotics, and implant retention. For subacute and chronic PJI, the criterion standard treatment is 2-stage revision surgery [6]. After initial debridement, an antibiotic-containing cemented spacer is implanted to maintain the extremity length and to release the antibiotics into local tissues. If the infection is under control, the spacer is removed, and a new prosthesis is implanted [7].
However, the proper time interval between spacer implantation and final revision surgery, as well as the definitive indication for second-stage prosthesis implantation, is still unclear. At worst, the new prosthesis is implanted with bone grafting or augmentation, which helps fill the bone defect. However, infection recurrence ultimately results in an early loosening of the prothesis. This might bring a great economic burden and serious physical and psychological pain to the patient [4]. For instance, sequentially performed joint resection and implantation surgeries with existing sepsis could cause systematic inflammation response syndrome and multiple organ dysfunction syndrome. Alternatively, at least the second-stage implanted prosthesis must be removed again [8]. Moreover, the patient has to stay in bed for an additional several weeks and undergo extra surgeries. Furthermore, in some circumstances, the infection might be insidious [9,10]. The erythrocyte sedimentation rate and C-reactive protein level of such patients might be normal. For instance, Pérez-Prieto reported that blood inflammatory markers such as the serum erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) level may not be accurate as diagnostic tools in PJI, particularly to identify low-grade and chronic PJI [11]. Gomez et al reported that the recurrence rate of PJI was 18.6% following 2-stage revision surgery [12]. Akgün et al reached a similar conclusion that 17% of patients undergoing 2-stage revision arthroplasty failed at a mean follow-up of 33 months from reimplantation [13].
To prevent the treatment failure of 2-stage revision surgery in patients with PJI, individuals at high risk of infection recurrence must be identified before definitive revision surgery. Consequently, surgeons need to understand the risk factors for infection recurrence prior to the treatment of patients with PJI. George et al reported that ESR and CRP are risk factors for infection recurrence in patients with PJI [14]. Paziuk et al also reported that patients with high platelet counts are at high risk of infection recurrence [15]. However, these previous studies only focused on 1 or 2 infection parameters. The recurrence of infection might be affected by many factors simultaneously. Therefore, the use of only 1 or 2 parameters to predict the final prognosis of patients was inaccurate. Cierny et al built a staging system that comprehensively included several factors to evaluate patients with osteomyelitis [16]. Similarly, in the present study, by analyzing patients with PJI using a Cox proportional hazard model and subsequently weighting the independent risk factors associated with PJI recurrence, a new scoring system was built that systematically identified the risk factors for infection recurrence in patients with PJI who had already undergone debridement, prosthesis removal, and spacer implantation and were scheduled to undergo definitive prosthesis implantation. The final aim of the present study was to create a new scoring system that would help surgeons estimate the infection recurrence risk for each individual with PJI undergoing 2-stage revision surgery.
Material and Methods
STUDY DESIGN:
This study employed a retrospective cohort design. The main research objectives of this study were (1) to estimate the risk of infection recurrence in patients with PJI undergoing 2-stage revision surgery, (2) to identify the risk factors associated with infection recurrence, and (3) to weight these risk factors and build a new clinical protocol that might help determine the individual risk of infection recurrence.
STUDY POPULATION:
We conducted a single-center, retrospective cohort study that was approved by the Institutional Review Board of the Third Hospital of Hebei Medical University and was conducted in accordance with the Declaration of Helsinki. As this study used a retrospective design and all patient information was deidentified before analysis, informed consent was not needed. This exemption from requiring informed consent was granted by the Ethics Committee of the Third Hospital of Hebei Medical University. Data from patients who underwent 2-stage revision for PJI from January 2010 to December 2017 were retrospectively analyzed in this study. Inclusion criteria were as follows: (1) subacute or chronic PJI, (2) patients undergoing joint prosthesis resection and spacer implantation, and (3) the time interval between spacer implantation and definitive prosthesis implantation was more than 2 weeks. The exclusion criteria were as follows: (1) malignant diseases, (2) metabolic bone diseases, and (3) incomplete medical records or radiologic images. Patients with infection recurrence after definitive prosthesis implantation were included as patients. Meanwhile, other patients who were cured of infection were considered controls. The definition of PJI is completely consistent with the evidence-based definition for PJI recorded by the MSIS in 2018. Briefly, 2 major and 6 minor criteria were included in the diagnostic criteria for MSIS. Major criteria included (1) 2 positive periprosthetic cultures with phenotypically identical organisms and (2) a sinus tract communicating with the joint. The presence of at least 4 of the following 6 minor criteria was also proposed to suggest PJI: (1) elevated CRP level and ESR, (2) elevated synovial fluid white blood cell (WBC) count or + + change on a leukocyte esterase test strip, (3) elevated synovial fluid polymorphonuclear (PMN), (4) positive histological analysis of periprosthetic tissue, (5) presence of purulence in the affected joint, and (6) a single positive culture [17].
INITIAL SAMPLE SIZE:
We used the sample size estimation method for the survival analysis to estimate the sample size required for this study. In many previous studies, infection occurred in approximately 10% of patients exposed to risk factors and in approximately 5% not exposed [18–21]. Therefore, we set the risk of infection to 0.05 and the test efficiency to 0.2. The required sample size was approximately 37. A total of 355 patients were included in this study, and thus the sample size was very sufficient.
TREATMENT STRATEGY:
All patients underwent 2-stage hip revision surgery for PJI. Debridement was performed on the previous surgical site, and the prosthesis was finally resected. Next, a spacer was planted to maintain hip function. After surgery, all patients received antibiotic therapy and underwent definitive prosthetic implantation when their condition allowed. The surgeries were performed by the same group of surgeons (YH, BL, WM, SL, and HL). All surgical treatments were performed electively when the patient’s condition was stable. No emergency surgical procedures were performed.
DATA COLLECTION:
All demographic and clinical information, laboratory test results and radiological images were obtained and analyzed.
Demographic and clinical information, such as sex, age, body mass index, smoking and alcohol status, steroid use, and other complications, of patients with PJI infection undergoing 2-stage hip revision surgery were retrieved from medical records.
In addition to demographic characteristics, the treatment history, laboratory test results, and MRI findings before definitive prosthesis implantation were investigated. The hematological tests we used to assess the condition of patients before the definitive prosthesis implantation were hemoglobin, ESR, CRP, albumin, globulin, fibrinogen, D-dimer, platelet counts, and glucose levels. These laboratory tests were performed using a hematology analyzer (Sysmex, Kobe, Japan), automatic biochemical analyzer (Beckman Coulter, CA, USA), and automatic blood coagulation analyzer (Sysmex, Kobe, Japan) in our hospital. CRP levels were measured using a turbidimetric inhibition immunoassay (Beckman Coulter). The ESR was measured using the Westergren method. Magnetic resonance imaging (MRI) was performed using one 1.5 T MR scanner (Siemens, Munich, Germany) and recorded in the picture archiving and communication system (H3C, Hangzhou, China). The MRIs before the definitive prosthesis implantation were read by 2 experienced surgeons, and the characteristics were summarized and classified as follows: 1) negative finding, 2) only lamellated hyperintense synovitis, 3) only soft tissue edema, and 4) lamellated hyperintense synovitis and soft tissue edema. The lamellated hyperintense synovitis was characterized by thickened synovial tissue around the joint, and soft tissue edema was characterized by a high signal in the T2WI sequence image. Blood transfusion in the definitive prosthesis implantation and treatment of antibiotics after implantation were analyzed.
We also collected information about debridement with prosthetic resection from the electric medical record system (Mediway, Beijing, China). Before debridement with prosthetic resection, a bacteriological smear was performed to determine the type of infectious bacteria. During resection, the selection of spacers with or without antibiotics was also analyzed. The time interval between debridement with prosthetic resection and definitive prosthesis implantation was recorded.
OUTCOMES OF INTEREST:
The primary outcome of interest was PJI recurrence, which was based on the MSIS criteria [22]. Secondary outcomes included the time period between second-stage surgery and PJI recurrence, the main clinical manifestation of PJI, and whether the prosthesis was loosened. Some infections were potentially latent, so a negative result of a single follow-up evaluation was not reliable. In other words, patients whose results of each follow-up were negative were considered cured of infection. Once a patient was found to have infectious clinical manifestations followed by a positive bacteriological smear, he or she was considered to have infection recurrence.
Censoring was defined if the patient had no evidence to confirm PJI recurrence at the last follow-up (at least 2 years).
STATISTICAL ANALYSIS:
Statistical analyses were performed using SPSS version 19.0 statistical software for Windows (IBM, Armonk, NY, USA). Continuous variables are expressed as the mean±standard deviation, and categorical variables are expressed as frequencies. The Mann-Whitney U test was performed to compare continuous variables between different groups. The chi-square test was performed for categorical variables. A Kaplan-Meier curve was drawn to show the survivorship of the implanted prostheses.
A new scoring system was built using the steps described below to estimate the infection recurrence risk of each individual undergoing 2-stage revision surgery following PJI. (1) A Cox proportional hazards model was built to determine the associations between potential risk factors and the recurrence of infection. All potential risk factors, including demographic characteristics, infection and surgical characteristics, and laboratory examination results (ie, all variables reported in Tables 1–3), were initially included in the regression models. The stepwise regression method was used to determine the final independent variables in the equation. (2) Risk factors that were continuous variables were converted to categorical variables by constructing receiver operating characteristic (ROC) curves. For each variable, 2 cutoff points were established at the estimated risks of 30% and 70% for infection recurrence from the coordinate points of the ROC curve. (3) We rebuilt the Cox regression model to establish the diagnostic model, including the finally converted categorical variables. The hazard ratio, 95% confidence interval (95% CI) and β-coefficient were calculated. (4) According to the β-coefficient, the independent risk factors were weighted. A scoring system for predicting infection recurrence was built to estimate the risk for each individual with PJI undergoing 2-stage revision surgery. The total score of the system was set to 100 points. Notably, the weight of each factor might be adjusted slightly according to the clinical significance and convenience of calculation. (5) The ROC curve for this scoring system was created. Some cutoff points were also identified to estimate the risk of infection recurrence. These cutoff points were selected from the coordinate points of the ROC curve.
A diagnostic test evaluation model was then built to establish the predictive value of the new scoring system. The area under the curve (AUC) was calculated to estimate the predictive accuracy of this scoring system. Sensitivity, specificity, accuracy and Youden’s index were calculated to demonstrate the predictive value of the new scoring system.
A
Results
GENERAL INFORMATION:
A total of 355 patients were ultimately included in this study. Among them, 25 patients with infection recurrence were identified, and the other 330 patients were finally cured. The mean follow-up time was 4.8 years (range from 3 to 7 years). According to their outcomes, these 355 patients were divided into 2 groups: infection recurrence (n=25) and infection cure (n=330). The comparison of general information between the 2 groups is shown in Table 1. The average age and the 3 most common complications (diabetes, immunosuppression, rheumatoid) were different between the 2 groups. Patients who suffered from infection recurrence were older (65.17±19.84 vs 62.43±17.81, P=0.007) and tended to have comorbidities such as diabetes (11/25 vs 70/330, P=0.009), immunosuppression (13/25 vs 97/330, P=0.018), and rheumatism (6/25 vs 33/330, P=0.031). No other differences in other general characteristics, such as sex (11/14 vs 152/178, P=0.842), body mass index (BMI) (24.24±4.84 vs 24.04±3.43, P=0.840), current smoking (22/3 vs 296/34, P=0.789), current alcohol use (21/4 vs 304/26, P=0.159), steroid use (17/8 vs 254/76, P=0.309), or type of implanted endoprosthesis (9/16 vs 79/251, P=0.178), were observed between groups (Table 1).
LABORATORY TESTS AND RADIOLOGICAL EXAMINATIONS:
The laboratory tests and MRIs of these 355 patients before the definitive prosthesis implantation were compared between the 2 groups (Table 2). Patients with abnormal laboratory tests, such as an increased CRP concentration (32.63±14.59 vs 26.70±11.59, P=0.002), decreased albumin level (32.37±8.25 vs 36.01±7.41, P=0.029), increased globulin level (29.83±6.19 vs 24.37±5.87, P=0.004), increased albumin/globulin ratio (1.12±0.29 vs 1.49±0.36, P<0.001) and increased glucose level (normal glucose level/increased glucose level: 7/18 vs 251/79, P<0.001), were likely to have infection recurrence. No additional differences were observed in the results for other laboratory tests, such as the hemoglobin level (119.56±19.31 vs 121.72±17.68, P=0.827), ESR (52.54±11.53 vs 50.71±16.37, P=0.392), fibrinogen level (3.95±0.88 vs 3.89±0.97, P=0.722), D-dimer level (0.91±0.37 vs 0.86±0.32, P=0.109), and platelet count (341.70±99.65 vs 327.23±85.82, P=0.510). Based on the features of the MRI scans, most patients with infection recurrence had positive MRI findings, such as lamellated hyperintense synovitis (8 patients, 32%), soft tissue edema (9 patients, 36%), and lamellated hyperintense synovitis with soft tissue edema (6 patients, 24%). In contrast, most patients who were cured had negative MRI findings (157 patients, 47.6%). Moreover, infections caused by gram-positive bacteria were more likely to recur (P<0.001). However, regardless of whether the bacteria were antibiotic-resistant (P=0.120), the period of antibiotic use (P=0.939) and time of remission (28.04±10.36 vs 26.14±11.95, P=0.602) were not associated with infection recurrence. Similarly, comparisons of whether the spacer was used with antibiotics and the time interval between prosthetic resection and hip revision were also not statistically significant (Table 2).
CHARACTERISTICS OF INFECTION RECURRENCE:
Infection recurrence was identified in 25 patients, and the incidence of infection recurrence was 7.04 per 100 patients (Table 3). Among these 25 patients, 19 patients had deep infection (76.00%), and 6 patients had superficial infection (24.00%). In terms of recurrence time, only 1 case each was found at less than 1 month (4.00%) and within 12 to 24 months (4.00%), 8 cases were found within 1 to 3 months (32.00%), 10 cases were found within 3 to 6 months (40.00%), and 5 cases were found within 6 to 12 months (20.00%). The clinical manifestations appeared to be sinus tract formation (36.00%, n=9), abscess formation (28.00%, n=7), and both (36.00%, n=9). At the last follow-up, 7 of 25 patients (28.00%) were found to have loosened prostheses due to infection recurrence (Table 3).
RISK FACTORS FOR INFECTION RECURRENCE:
A Cox regression model was built to identify the independent risk factors for infection recurrence. Seven independent risk (protective) factors for infection reoccurrence in patients with PJI who underwent 2-stage hip revision surgery were identified (Table 4). The glucose level before the definitive prosthesis implantation was the first risk factor: every 1-parameter increase in the glucose level increased the infection recurrence by 9.5% (HR=1.095, 95% CI=1.008–1.245). Aging was the second risk factor: every 1-year increase in age increased the hazard ratio of infection recurrence by 8.5% (HR=1.085, 95% CI=1.053–1.201). CRP was the third factor: every 1-parameter increase in CRP increased infection recurrence by 5.9% (HR=1.059, 95% CI=1.012–1.185). The type of infected bacteria was tested by bacteriological smear before debridement with prosthetic resection. Compared with patients with negative smear results, patients with a previous gram-negative bacterial infection had a more than 2-fold increased risk for infection recurrence (HR=2.165, 95% CI=1.452–3.022). Compared with patients without, patients with immunosuppression/immunocompromised status had a more than 3-fold increased risk for infection recurrence (HR=3.208, 95% CI=1.121–8.254). Additionally, patients with both lamellated hyperintense synovitis and soft tissue edema on MRI before definitive prosthesis implantation had an approximately 4-fold increased risk for infection recurrence (HR=3.980, 95% CI=2.250–6.233). Apart from these risk factors, a protective factor was also found in this study. Infection reoccurrence was less likely in patients who had a higher albumin and globulin ratio (HR=0.928, 95% CI=0.875–0.945) (Table 4).
SCORING SYSTEM FOR PREDICTING INFECTION RECURRENCE:
A new scoring system was built to help surgeons rapidly screen patients who would have a high risk of infection recurrence among all patients with PJI and 2-stage revision, as shown in Table 5. A total of 7 factors were involved in this scoring system. For each factor, 1 or 2 weighted scores were assigned. The total score of each factor was the final score of the patient, and the maximum score was 100 points. The AUC of this scoring system was 0.965 (95% CI=0.947–0.983). According to previously described standards, the predictive scores for a low (≤30%), moderate (31–69%), and high risk (≥70%) of infection recurrence were ≤45.0 points, 46.0 to 77.0 points, and ≥78.0 points, respectively (Table 5).
Discussion
INCIDENCE AND CHARACTERISTICS OF INFECTION RECURRENCE:
Infection recurrence was identified in 25 patients, namely, 7.04% of all patients who underwent 2-stage revision surgery. This is at a moderate level compared to that reported by other studies. Sanchez-Sotelo et al found that 7.1% of 169 patients experienced infection recurrence [28. Petis demonstrated that the incidence of recurrence of infection was 10% at 1 year [29]. Furthermore, the results suggested that most infection recurrences were subacute or chronic (>1 month), which means that once the infection recurrence occurred, debridement with implant retention surgery would not be the proper treatment. In such situations, the reimplanted prosthesis must be removed again. However, the results showed that loosening of the newly implanted prosthesis was identified in only 7 of 28 patients (28%). Consequently, it must be emphasized again that patients who are at high risk of infection recurrence should be better identified before a planned second-revision surgery. The other characteristics of infection recurrence were not remarkable.
RISK FACTORS FOR INFECTION RECURRENCE:
Seven independent risk factors were identified in this study. The first one was aging. Advanced age was commonly associated with decreased immunity and poor nutritional status. Meanwhile, comorbidities such as diabetes or rheumatism were more commonly seen in elderly patients. These would all increase the risk of infection. In particular, in addition to aging, blood glucose and plasma albumin/globulin levels were also independent risk factors for infection recurrence. Shohat et al carried out a study that included 2360 patients who had undergone THA and found that higher glycemic variability was associated with an increased length of stay, 90-day mortality, and especially PJI and surgical site infection [30]. Another meta-analysis demonstrated that an albumin level <3.5 g/dL had an almost 2.5-fold increased risk of surgical site infection. These previous conclusions were similar to the findings in this study [31]. The serum CRP concentration, which is a well-established parameter for infection and inflammation prediction, was also associated with infection recurrence in this study. Alijanipour et al [21] reported that the optimal threshold for CRP was 13.5 mg/L, and the median values for CRP in early-postoperative and late-chronic hip infections were 143 mg/L and 56 mg/L, respectively. Additionally, positive Gram staining, as reported by Ghanem et al, was also a risk factor for infection recurrence [32]. No differences in the success rate of infection revision caused by organisms that showed resistance to antibiotics and those that were sensitive to antibiotics were reported in the study by Volin et al [24]. Similar results were also obtained in the present study. Moreover, the immunosuppression status of the patient was also correlated with infection recurrence. The last risk factor was the MRI findings. This was considered to be the most important predictive factor for infection. However, in this study, the sole predictive value of MRI findings was not investigated. Gao et al found that the sensitivity of lamellated hyperintense synovitis for infection was 0.80 to 0.88 and the specificity was 0.84 to 0.92 [33]. Another study showed that the sensitivity of lamellated hyperintense synovitis for infection was 0.86 to 0.92 and the specificity was 0.85 to 0.87, with almost perfect interobserver agreement and intraobserver agreement [34].
SCORING SYSTEM FOR PREDICTING INFECTION RECURRENCE:
According to the method of transparent reporting of a multivariable prediction model for individual prognosis or diagnosis (TRIPOD statement), a new prediction system was established in this study. All the independent risk factors were included in this scoring system and weighted. Consequently, the accuracy of this comprehensive predictive system was higher than that in some previous predictive methods, which were characterized as single-parameter prediction. For instance, Wang et al, Alijanipour et al, and Cichos et al used the solo ESR to predict infection and concluded that the AUC was limited to 0.877 [21,35,36]. In another study, the AUC of CRP levels was 0.9 [37]. Gao found that the sensitivity and specificity of MRI in the prediction of infection were only 0.80 and 0.84, respectively [33]. In the present study, the AUC of this new scoring system was 0.965, which was the highest among the scoring systems and demonstrated the strength of this comprehensive predictive system. Additionally, 3 intervals of estimated risk were given, which could help determine the risk of infection recurrence in this special group of patients in detail. This consideration is crucial for making clinical decisions.
Although this investigation attempted to identify the appropriate time for 2-stage revision surgery by establishing a scoring system, some limitations still exist. The average follow-up time of all patients was 4.8 years. Garvin et al [38] followed 97 patients with 2-stage revision for an average of 11 years. Twelve patients had recurrence of infection, and cases were identified in 5 of these patients after more than 3.5 years. Therefore, the long-term clinical outcomes of our patients are uncertain. The second limitation is that retrospective studies often have selection bias; however, all patients in the present study had consistent indications, and thus we do not believe that selection bias existed. The third limitation is that this retrospective study was conducted at a single center, and the assessment method has not been verified in clinical practice. In our subsequent studies, relevant clinical trials will be conducted to verify the feasibility of this scoring scale.
Conclusions
For each patient with PJI who had already undergone joint resection and spacer implantation, this newly established scoring system might help determine the accurate risk of infection recurrence after a definitive new prosthesis implantation. If a patient has a score greater than 78 points, the patient would be considered highly likely to have a recurrence of infection. Therefore, the second-stage revision surgery should be changed to an additional anti-infection treatment or a debridement surgery instead of a definitive prosthesis implantation surgery.
Tables
Table 1. Demographic information of patients with periprosthetic joint infection undergoing 2-stage hip revision surgery.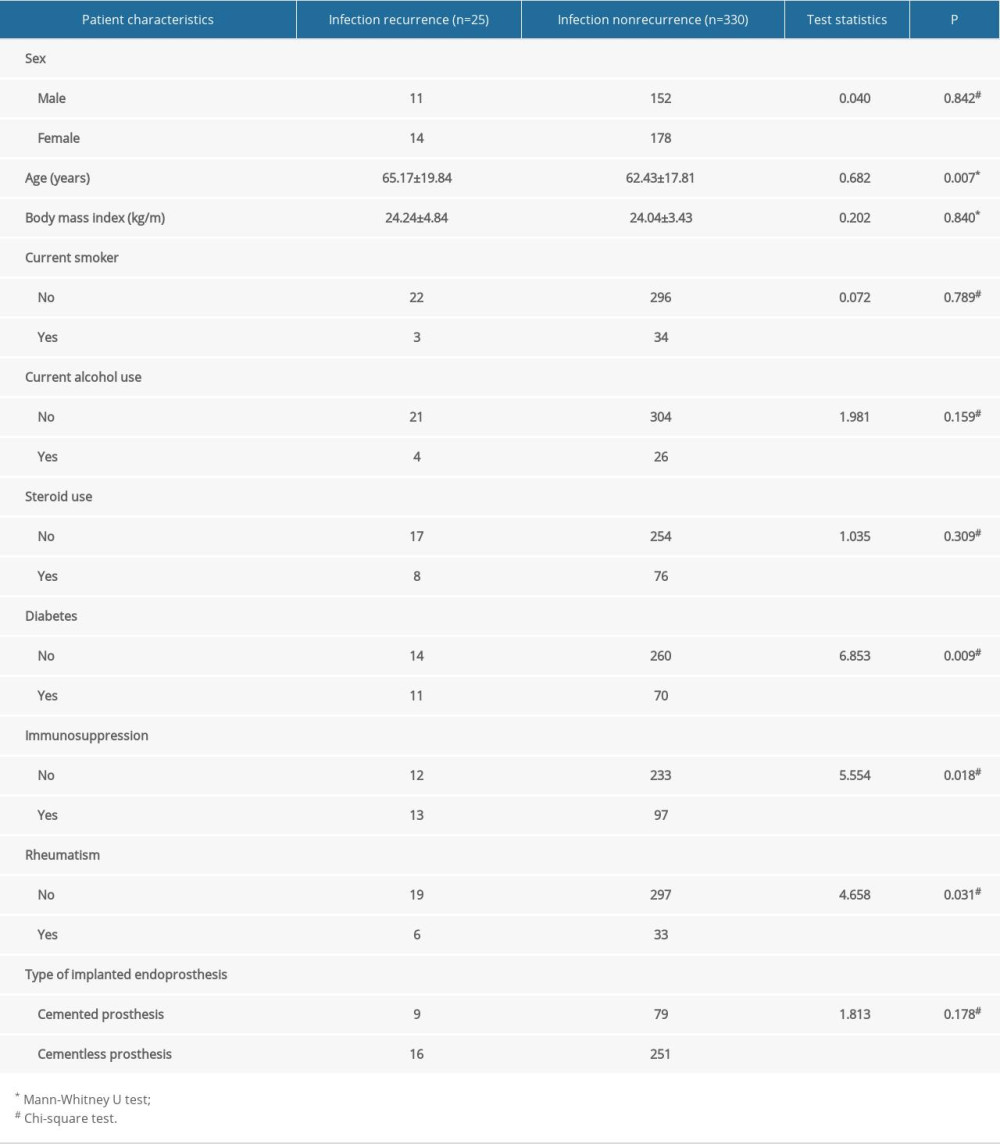 Table 2. Results of hematological, bacteriological, and laboratory examinations of patients with periprosthetic joint infection undergoing 2-stage hip revision surgery.
Table 2. Results of hematological, bacteriological, and laboratory examinations of patients with periprosthetic joint infection undergoing 2-stage hip revision surgery.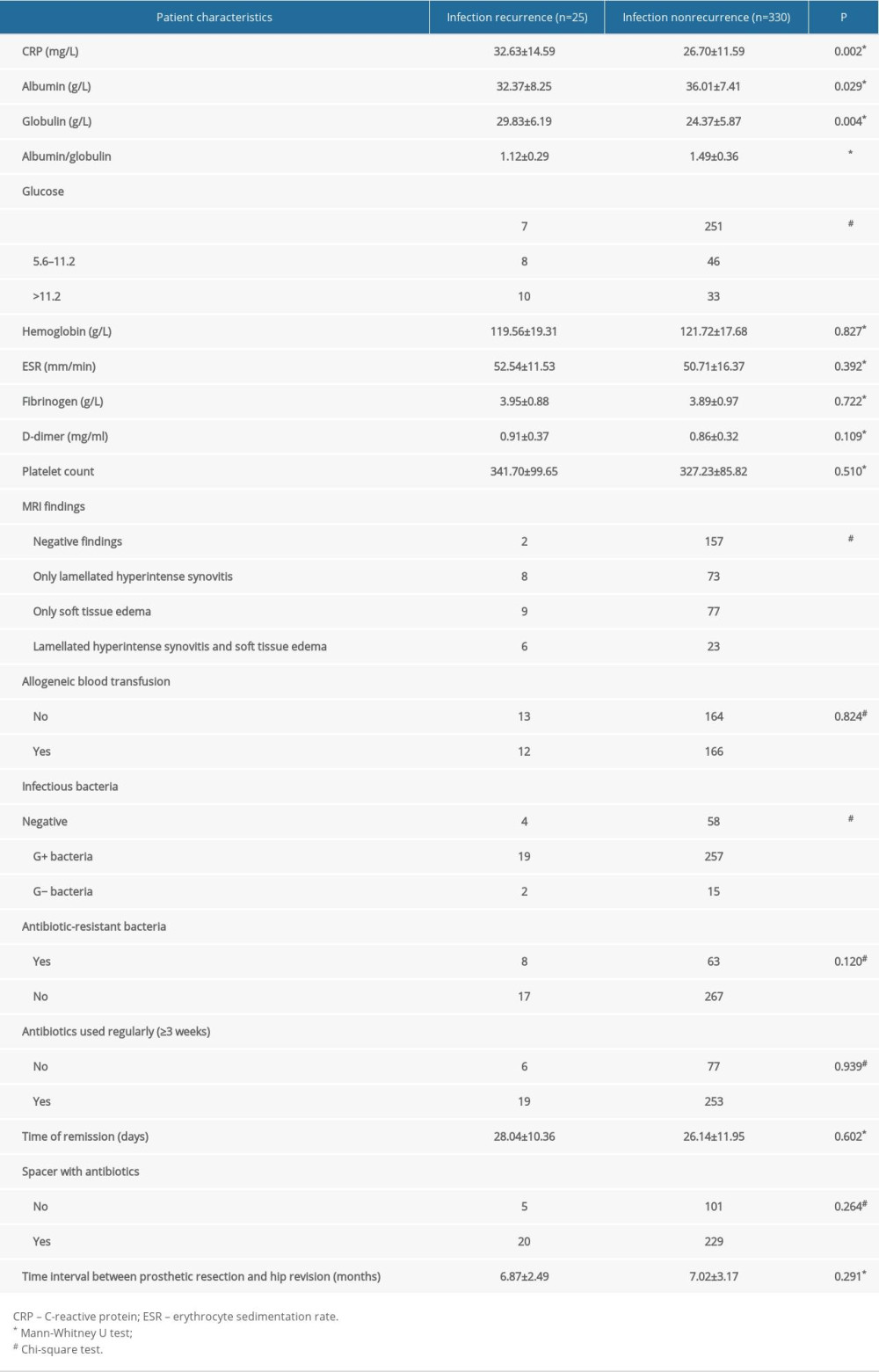 Table 3. Incidence, recurrence time, and characteristics of infection recurrence in patients with periprosthetic joint infection undergoing 2-stage hip revision surgery.
Table 3. Incidence, recurrence time, and characteristics of infection recurrence in patients with periprosthetic joint infection undergoing 2-stage hip revision surgery.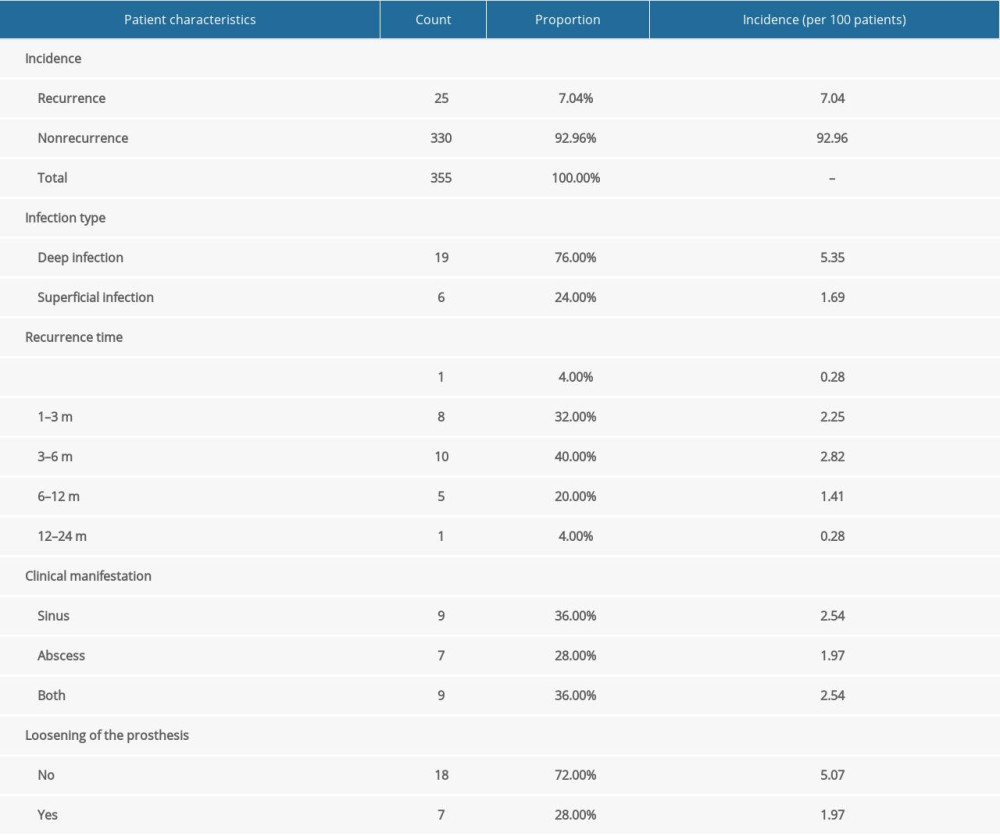 Table 4. Independent risk factors for infection recurrence in patients with periprosthetic joint infection undergoing 2-stage hip revision surgery.
Table 4. Independent risk factors for infection recurrence in patients with periprosthetic joint infection undergoing 2-stage hip revision surgery.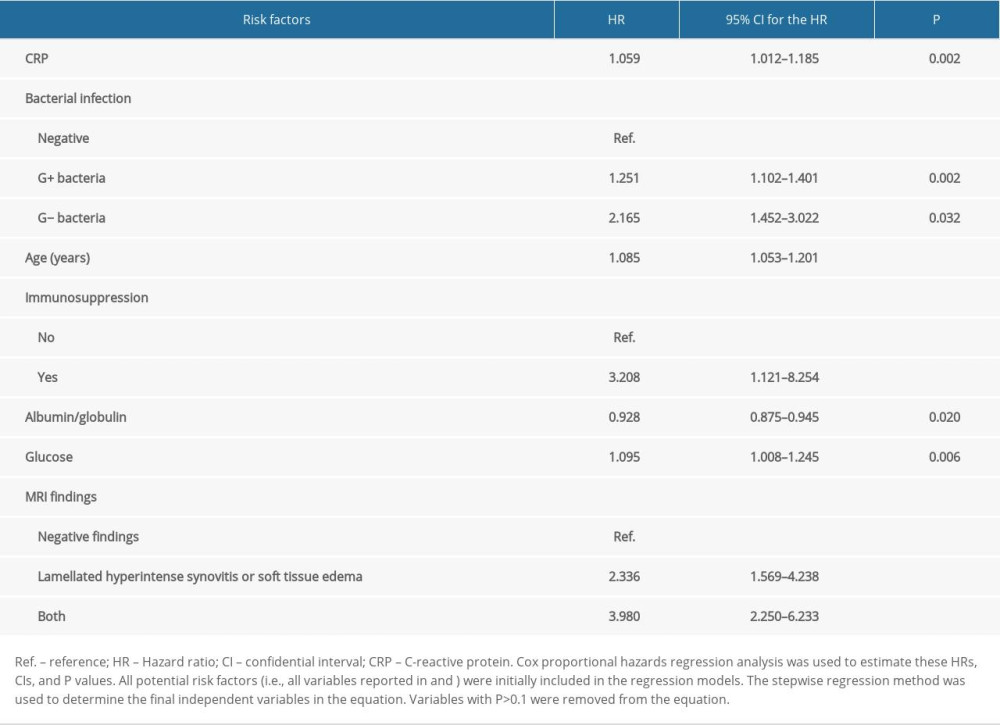 Table 5. Scoring system for predicting infection recurrence in patients with PJI undergoing 2-stage hip revision surgery.
Table 5. Scoring system for predicting infection recurrence in patients with PJI undergoing 2-stage hip revision surgery.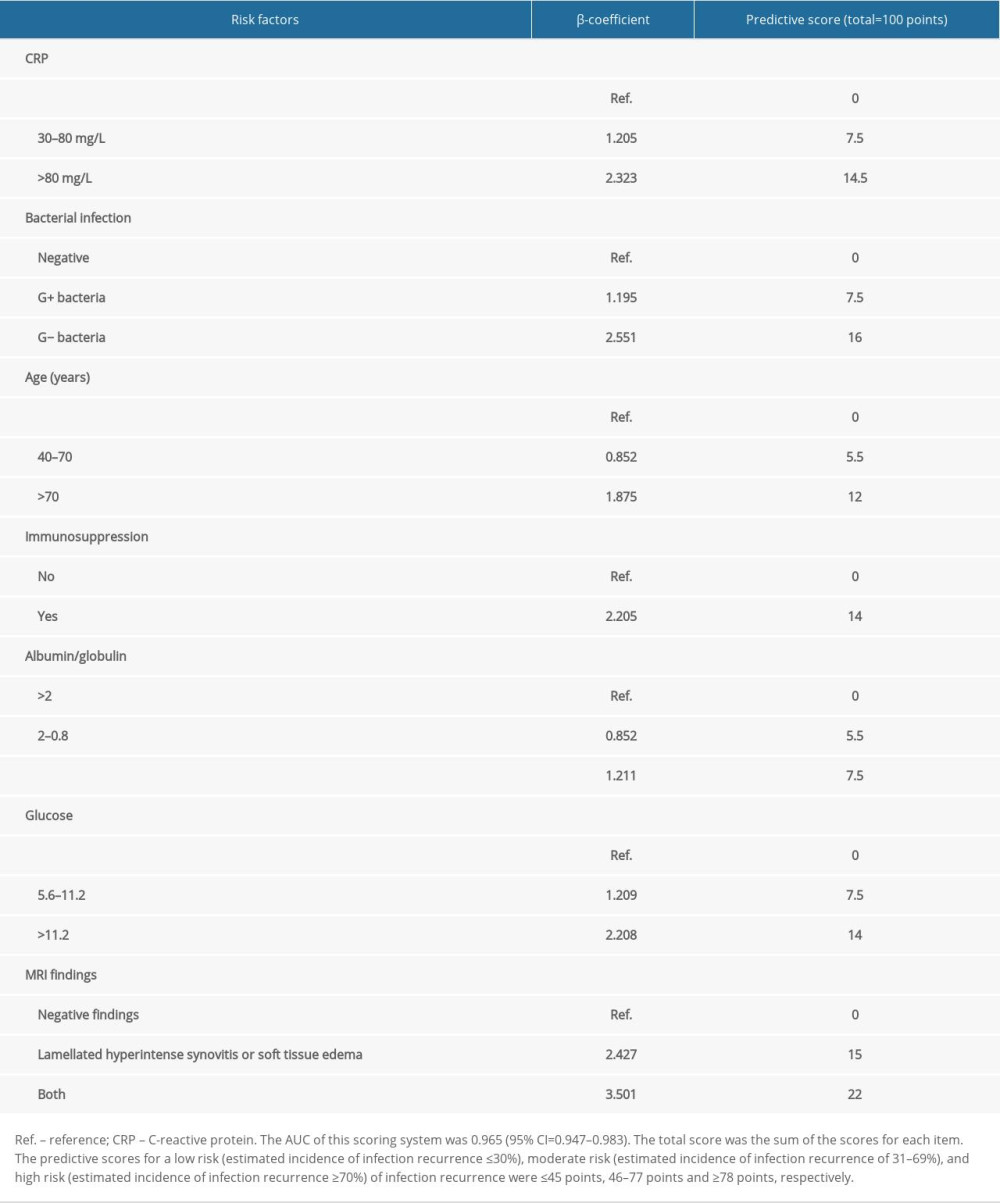
References
1. Bori G, Soriano A, Garcia S, Mallofre C, Usefulness of histological analysis for predicting the presence of microorganisms at the time of reimplantation after hip resection arthroplasty for the treatment of infection: J Bone Joint Surg Am, 2007; 89(6); 1232-37
2. Eka A, Chen AF, Patient-related medical risk factors for periprosthetic joint infection of the hip and knee: Ann Transl Med, 2015; 3(16); 233
3. Shohat N, Fleischman A, Tarabichi M, Weighing in on body mass index and infection after total joint arthroplasty: Is there evidence for a body mass index threshold?: Clin Orthop Relat Res, 2018; 476(10); 1964-69
4. Yao JJ, Hevesi M, Visscher SL, Direct inpatient medical costs of operative treatment of periprosthetic hip and knee infections are twofold higher than those of aseptic revisions: J Bone Joint Surg Am, 2021; 103(4); 312-18
5. Delanois RE, Mistry JB, Gwam CU, Current epidemiology of revision total knee arthroplasty in the United States: J Arthroplasty, 2017; 32(9); 2663-68
6. Lazic I, Scheele C, Pohlig F, von Eisenhart-Rothe R, Suren C, Treatment options in PJI – is two-stage still gold standard?: J Orthop, 2021; 23; 180-84
7. Cai YQ, Fang XY, Huang CY, Destination joint spacers: A similar infection-relief rate but higher complication rate compared with two-stage revision: Orthop Surg, 2021; 13(3); 884-91
8. Renz N, Trampuz A, Perka C, Rakow A, Outcome and failure analysis of 132 episodes of hematogenous periprosthetic joint infections-a cohort study: Open Forum Infect Dis, 2022; 9(4); ofac094
9. Portillo ME, Salvado M, Trampuz A, Improved diagnosis of orthopedic implant-associated infection by inoculation of sonication fluid into blood culture bottles: J Clin Microbiol, 2015; 53(5); 1622-27
10. Trampuz A, Hanssen AD, Osmon DR, Synovial fluid leukocyte count and differential for the diagnosis of prosthetic knee infection: Am J Med, 2004; 117(8); 556-62
11. Perez-Prieto D, Portillo ME, Puig-Verdie L, C-reactive protein may misdiagnose prosthetic joint infections, particularly chronic and low-grade infections: Int Orthop, 2017; 41(7); 1315-19
12. Gomez MM, Tan TL, Manrique J, The fate of spacers in the treatment of periprosthetic joint infection: J Bone Joint Surg Am, 2015; 97(18); 1495-502
13. Akgün D, Müller M, Perka C, A positive bacterial culture during re-implantation is associated with a poor outcome in two-stage exchange arthroplasty for deep infection: Bone Joint J, 2017; 99-B(11); 1490-95
14. George J, Jawad M, Curtis GL, Utility of serological markers for detecting persistent infection in two-stage revision arthroplasty in patients with inflammatory arthritis: J Arthroplasty, 2018; 33(7S); S205-8
15. Paziuk T, Rondon AJ, Goswami K, A novel adjunct indicator of periprosthetic joint infection: Platelet count and mean platelet volume: J Arthroplasty, 2020; 35(3); 836-39
16. Cierny G, Mader JT, Penninck JJ, A clinical staging system for adult osteomyelitis: Clin Orthop Relat Res, 2003(414); 7-24
17. Parvizi J, Tan TL, Goswami K, The 2018 definition of periprosthetic hip and knee infection: An evidence-based and validated criteria: J Arthroplasty, 2018; 33(5); 1309-14e2
18. Windisch C, Brodt S, Roehner E, Matziolis G, C-reactive protein course during the first 5 days after total knee arthroplasty cannot predict early prosthetic joint infection: Arch Orthop Trauma Surg, 2017; 137(8); 1115-19
19. Shi W, Wang Y, Zhao X, CRP/albumin has a promising prospect as a new biomarker for the diagnosis of periprosthetic joint infection: Infect Drug Resist, 2021; 14; 5145-51
20. Zajonz D, Brand A, Lycke C, Risk factors for early infection following hemiarthroplasty in elderly patients with a femoral neck fracture: Eur J Trauma Emerg Surg, 2019; 45(2); 207-12
21. Alijanipour P, Bakhshi H, Parvizi J, Diagnosis of periprosthetic joint infection: The threshold for serological markers: Clin Orthop Relat Res, 2013; 471(10); 3186-95
22. Workgroup Convened by the Musculoskeletal Infection Society, New definition for periprosthetic joint infection: J Arthroplasty, 2011; 26(8); 1136-38
23. Muller M, Morawietz L, Hasart O, Strube P, Diagnosis of periprosthetic infection following total hip arthroplasty – evaluation of the diagnostic values of pre- and intraoperative parameters and the associated strategy to preoperatively select patients with a high probability of joint infection: J Orthop Surg Res, 2008; 3; 31
24. Volin SJ, Hinrichs SH, Garvin KL, Two-stage reimplantation of total joint infections: A comparison of resistant and non-resistant organisms: Clin Orthop Relat Res, 2004(427); 94-100
25. Perez-Prieto D, Hinarejos P, Alier A, Adherence to a reliable PJI diagnostic protocol minimizes unsuspected positive cultures rate: BMC Musculoskelet Disord, 2021; 22(1); 653
26. Hemmann P, Schmidutz F, Ahrend MD, Single-stage total hip arthroplasty after failed fixation of proximal femoral fractures: An increased risk for periprosthetic joint infections?: Arch Orthop Trauma Surg, 2021 [Online ahead of print]
27. Chisari E, Parvizi J, Accuracy of blood-tests and synovial fluid-tests in the diagnosis of periprosthetic joint infections: Expert Rev Anti Infect Ther, 2020; 18(11); 1135-42
28. Sanchez-Sotelo J, Berry DJ, Hanssen AD, Cabanela ME, Midterm to long-term followup of staged reimplantation for infected hip arthroplasty: Clin Orthop Relat Res, 2009; 467(1); 219-24
29. Petis SM, Abdel MP, Perry KI, Long-term results of a 2-stage exchange protocol for periprosthetic joint infection following total hip arthroplasty in 164 hips: J Bone Joint Surg Am, 2019; 101(1); 74-84
30. Shohat N, Restrepo C, Allierezaie A, Increased postoperative glucose variability is associated with adverse outcomes following total joint arthroplasty: J Bone Joint Surg Am, 2018; 100(13); 1110-17
31. Yuwen P, Chen W, Lv H, Albumin and surgical site infection risk in orthopaedics: A meta-analysis: BMC Surg, 2017; 17(1); 7
32. Ghanem E, Ketonis C, Restrepo C, Periprosthetic infection: Where do we stand with regard to Gram stain?: Acta Orthop, 2009; 80(1); 37-40
33. Gao Z, Jin Y, Chen X, Diagnostic value of MRI lamellated hyperintense synovitis in periprosthetic infection of hip: Orthop Surg, 2020; 12(6); 1941-46
34. Plodkowski AJ, Hayter CL, Miller TT, Lamellated hyperintense synovitis: Potential MR imaging sign of an infected knee arthroplasty: Radiology, 2013; 266(1); 256-60
35. Wang H, Zhou H, Jiang R, Globulin, the albumin-to-globulin ratio, and fibrinogen perform well in the diagnosis of Periprosthetic joint infection: BMC Musculoskelet Disord, 2021; 22(1); 583
36. Cichos KH, Christie MC, Heatherly AR, The value of serological screening prior to conversion total hip arthroplasty: J Arthroplasty, 2020; 35(6S); S319-24
37. Tetreault MW, Wetters NG, Moric M, Is synovial C-reactive protein a useful marker for periprosthetic joint infection?: Clin Orthop Relat Res, 2014; 472(12); 3997-4003
38. Garvin KL, Miller RE, Gilbert TM, Late reinfection may recur more than 5 years after reimplantation of THA and TKA: Analysis of pathogen factors: Clin Orthop Relat Res, 2018; 476(2); 345-52
Tables
 Table 1. Demographic information of patients with periprosthetic joint infection undergoing 2-stage hip revision surgery.
Table 1. Demographic information of patients with periprosthetic joint infection undergoing 2-stage hip revision surgery. Table 2. Results of hematological, bacteriological, and laboratory examinations of patients with periprosthetic joint infection undergoing 2-stage hip revision surgery.
Table 2. Results of hematological, bacteriological, and laboratory examinations of patients with periprosthetic joint infection undergoing 2-stage hip revision surgery. Table 3. Incidence, recurrence time, and characteristics of infection recurrence in patients with periprosthetic joint infection undergoing 2-stage hip revision surgery.
Table 3. Incidence, recurrence time, and characteristics of infection recurrence in patients with periprosthetic joint infection undergoing 2-stage hip revision surgery. Table 4. Independent risk factors for infection recurrence in patients with periprosthetic joint infection undergoing 2-stage hip revision surgery.
Table 4. Independent risk factors for infection recurrence in patients with periprosthetic joint infection undergoing 2-stage hip revision surgery. Table 5. Scoring system for predicting infection recurrence in patients with PJI undergoing 2-stage hip revision surgery.
Table 5. Scoring system for predicting infection recurrence in patients with PJI undergoing 2-stage hip revision surgery. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








