06 September 2022: Clinical Research
Predictive Value of Red Blood Cell Distribution Width and Atrial Diameter in Paroxysmal Atrial Fibrillation: A Cross-Sectional Study
Guoli LinDOI: 10.12659/MSM.937802
Med Sci Monit 2022; 28:e937802
Abstract
BACKGROUND: Atrial fibrillation (AF) is the most common arrhythmia and is associated with deleterious consequences. In addition to worsening a patient’s quality of life, AF is associated with stroke, heart failure, and increased mortality. Red blood cell distribution width (RDW) has been associated with an increased risk of death and adverse cardiovascular outcomes, while left atrial enlargement has been linked to atrial fibrillation (AF). However, the relationship among RDW, atrial diameter (AD), and paroxysmal AF is uncertain. The aim of this study was to investigate the relationship among RDW, atrial diameter, and paroxysmal AF.
MATERIAL AND METHODS: A total of 22 patients with paroxysmal AF and 100 patients with non-AF were included in the study. The demographic variables and baseline clinical characteristics of both groups were analyzed.
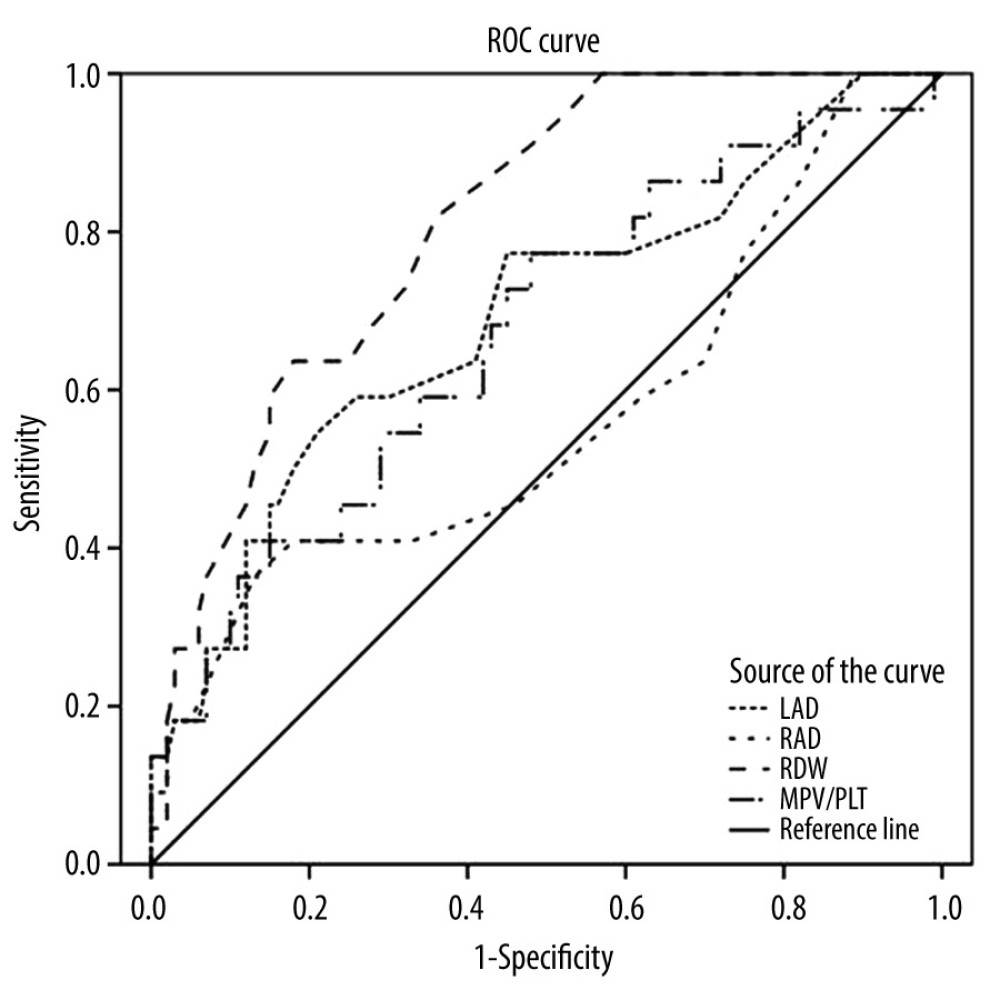
RESULTS: The demographics and comorbidities were comparable between the paroxysmal AF and control groups, except for BMI (body mass index). RDW, high-sensitivity C-reactive protein (hs-CRP) levels, NT-pro-BNP levels, MPV/PLT (mean platelet volume/total platelet count), LAD, RAD, and CHA2DS2-VASc score were higher in the paroxysmal AF group versus the control group (P<0.05). Binary logistic regression analyses demonstrated that RDW (OR: 2.557, 95% CI: 1.481~4.414), Hs-CRP(OR: 1.445, 95% CI: 1.144~1.825), MPV/PLT (OR: 1.342, 95% CI: 1.047~1.720), LAD (OR: 1.068, 95% CI: 1.007~1.132), and CHA₂DS₂-VASc score (OR: 1.645, 95% CI: 1.042~2.597) were independent predictors for paroxysmal AF (P<0.05, respectively). The ROC analysis showed that the area under the curve for LAD was 0.692, the area under the curve for RAD was 0.566, the area under the curve for RDW was 0.811, and the area under the curve for MPV/PLT was 0.671.
CONCLUSIONS: LAD, RDW, and MPV/PLT were associated with paroxysmal AF.
Keywords: Atrial Fibrillation, Erythrocyte Indices, C-Reactive Protein, Cross-Sectional Studies, Erythrocytes, Humans, Quality of Life
Background
Atrial fibrillation (AF) is a common clinical arrhythmia, and its prevalence increases with age, leading to cardiac dysfunction and stroke. It has been estimated that 6–12 million people worldwide will have AF in the US by 2050 and 17.9 million people in Europe by 2060 [1]. Current epidemiological studies indicate that AF is more prevalent in White patients than in other racial and ethnic groups [2]. In the Multi-Ethnic Study of Atherosclerosis (MESA), after adjusting for age and sex, non-Hispanic blacks, Hispanics, and Chinese Americans had 49%, 46%, and 65% lower rates of AF, respectively, compared with non-Hispanic White patients. In China the age-adjusted prevalence rate of AF was 0.75% [3]. The prevalence of AF in males and females aged <60 years was 0.43% and 0.44%, respectively, while the prevalence of AF in those aged >60 years was 1.83% and 1.92%, respectively [4]. The main causes of death in patients are heart failure, cardiac arrest, and stroke [5].
In patients with diabetes, their risk of developing AF was increased by 40%, probably due to autonomic nervous system abnormalities as an important mechanism for the development and maintenance of AF. Diabetic autonomic neuropathy (DAN) is a common complication of diabetes, with a prevalence of 46% in patients who have had diabetes for less than 5 years and 70% in patients who have had diabetes for more than 20 years. DAN had a significant negative impact on the cardiovascular system, and a study by Rizzo et al found a significant association between autonomic dysfunction and asymptomatic AF episodes in patients with type 2 diabetes [6]. Studies had shown that the occurrence and development of AF were closely related to the size of the atrium [7]. Red blood cell distribution width (RDW), a parameter that reflects the heterogeneity of red blood cell volume and which is part of the standard complete blood count [8], was mainly used in the differential diagnosis of anemia [9]. However, previous studies had shown that RDW was closely related to the clinical outcomes of cardiovascular events, such as heart failure [10], acute myocardial infarction [11], and stable angina. Some studies demonstrated that increasing levels of RDW were associated with chronic inflammation [12]. Inflammation was considered as a potential factor for AF recurrence after catheter ablation (CA), and was associated with post-CA fibrosis and remodeling processes that alter atrial shape and function. This electroanatomical remodeling affects the generation, duration, and propagation of action potentials in the atria, making this tissue more susceptible to factors that trigger arrhythmias [13]. Studies found that patients without AF recurrence after epicardial ablation had increased sarcoplasmic endoplasmic reticulum calcium ATPase (SERCA) levels, which is a protein involved in the handling of intracellular Ca2+. Reducing Ca2+ overload in atrial myocytes could reduce AF duration and atrial remodeling [14]. Mean platelet volume/platelet count ratio (MPV/PLT) is an indicator of platelet activation and aggregation, as well as inflammation [15]. Metabolic syndrome (MS) was a clinical condition that is associated with a pro-inflammatory and pro-thrombotic state. It was associated with a higher rate of recurrence of arrhythmias, higher rates of hospitalization for worsening heart failure, and a worse clinical prognosis [16–18]. Few studies have focused on predicting the risk of paroxysmal AF. However, the diagnosis of paroxysmal AF relies on 24-h Holter monitoring, so a simple and easy index is needed to predict it. The aim of this study was to investigate the relationship among atrial diameter, RDW, MPV/PLT, and paroxysmal atrial fibrillation.
Material and Methods
STUDY DESIGN AND POPULATIONS:
The study was approved by the Ethics Committee of the participating institution (the Affiliated Hospital of Putian University, Putian University, Fujian, China).
This study was a retrospective study, and patient data were collected through the hospital digital information system. The data did not affect the patient’s personal privacy. Therefore, informed consent was not required for the study.
We retrospectively analyzed medical records of 22 consecutive non-valvular paroxysmal AF patients who were admitted to our inpatient clinic in the Department of Cardiovascular Medicine, The Affiliated Hospital of Putian University between January 2020 and December 2020. The control group was recruited from hospitalized patients without atrial fibrillation. The exclusion criteria were as follows: recent acute infection or chronic inflammatory disease; malignant tumor; valvular heart disease; thyroid dysfunction; anemia; and acute myocardial infarction. These patients underwent a detailed history and physical examination, a 12-lead electrocardiogram (ECG), 24-h Holter electrocardiogram, laboratory analysis, and transthoracic echocardiography. Electrocardiography was performed immediately when patients had palpitations. Patients with transient ischemic attack (TIA) or ischemic stroke underwent 72-h continuous Holter monitoring. Paroxysmal AF was defined as AF that terminated spontaneously or by cardioversion within 7 days of onset. The ECG of atrial fibrillation showed the disappearance of the P wave, replaced by the f wave, with a frequency of 350–600 beats/min. The QRS wave rhythm was absolutely irregular, showing uneven RR intervals and a normal QRS wave pattern. The diagnosis of paroxysmal atrial fibrillation was in accordance with the diagnostic criteria developed by the 2018 Chinese Medical Association Electrophysiology and Pacing Section, “Atrial fibrillation: current understanding and treatment recommendations – 2018” [2].
DATA COLLECTION:
All data were retrieved from the digital information system of the hospital. Baseline demographic and clinical characteristics, including age, sex, hypertension, diabetes mellitus, and heart failure were recorded for all patients.
CLINICAL MEASUREMENTS:
Blood samples were drawn by an expert nurse in vacuum blood tubes containing ethylene diamine tetra-acetic acid-treated. Complete blood count, parameters such as RDW, platelet count, and MPV, were measured by electrical impedance method using an automated hematology analyzer (CAL8000; Mindray Corporation, Shenzhen, China). The MPV/PLT was calculated by dividing the MPV by the PLT. Determination of N-terminal B-type natriuretic peptide precursor (NT-pro-BNP) levels was by dry immunofluorescence assay using an automated fluorescence immunoassay analyzer (Getein1600; Geteinbiotech, Nanjing, China).
ECHOCARDIOGRAPHY:
All patients were examined using a Vivid 7 echocardiography device (General Electric, Boston, Massachusetts, USA). Two-dimensional, M-mode, and subsequent transthoracic Doppler echocardiographic examinations were performed on each patient. The left atrial diameter (LAD), right atrial diameter (RAD), and left ventricular ejection fraction (LVEF) were measured.
STATISTICAL ANALYSIS:
All statistical analyses were conducted using SPSS 19.0 for Windows (IBM, USA). Descriptive statistics are calculated as the mean±standard deviation of continuous variables, and the number and percentage of categorical variables. Continuous variables were compared using the independent samples
Results
PATIENT CHARACTERISTICS:
A total of 22 patients with paroxysmal AF (54.5% males; mean age: 73.59±12.82 years) and 100 controls (57% males; mean age: 71.62±7.79 years) were included in the study. The demographic and clinical characteristics of the patients are summarized in Table 1. There were no significant differences between patients with paroxysmal AF and controls in terms of demographic characteristics, except BMI. Although there was no statistical difference between the diabetic patients in the paroxysmal AF group and the control group, the P value was 0.636, which may be related to the fact that diabetic patients are more prone to AF. BMI was higher in the paroxysmal atrial fibrillation group than in the control group, which may be related to the increased risk of metabolic syndrome (Table 1).
COMPARISON OF CLINICAL AND ECHOCARDIOGRAPHIC PARAMETERS BETWEEN THE 2 GROUPS:
There were significant differences in terms of RDW, MPV, PLT, high-sensitivity C-reactive protein (hs-CRP) levels, NT-pro-BNP levels, MPV/PLT, LAD, RAD, and CHA2DS2-VASc score. There was no significant differences in terms of LVEF (Table 2).
PREDICTORS OF PAROXYSMAL AF:
Binary logistic regression analyses demonstrated that RDW (OR: 2.557, 95% CI: 1.481~4.414), Hs-CRP (OR: 1.445, 95% CI: 1.144~1.825), MPV/PLT (OR: 1.342, 95% CI: 1.047~1.720), LAD (OR: 1.068, 95% CI: 1.007~1.132), and CHA2DS2-VASc score (OR: 1.645, 95% CI: 1.042~2.597) were independent predictors for paroxysmal AF (P<0.05, all) (Table 3).
In correlation analysis, the LAD level was correlated with RDW (r=0.196, P<0.05) and CHA2DS2-VASc (r=0.180, P<0.05). The RDW level was correlated with MPV/PLT (r=0.637, P<0.05). The Hs-CRP level was correlated with CHA2DS2-VASc (r=0.305, P<0.05). The CHA2DS2-VASc was correlated with LAD (r=0.180, P<0.05), Hs-CRP (r=0.305, P<0.05). The MPV/PLT was correlated with RDW (r=0.637, P<0.05) (Table 4).
ROC CURVE:
For the prediction of paroxysmal AF, ROC analysis showed that the area under the curve for LAD was 0.692 (95% CI: 0.563–0.821, P<0.01). The best cut-off value of LAD was 38.5 mm (sensitivity: 54.6%; specificity: 79%). The area under the curve for RAD was 0.566 (95% CI: 0.420–0.713, P>0.05). The best cut-off value of RAD was 36.5 mm (sensitivity: 36.4%; specificity: 87%). The area under the curve for RDW was 0.811(95% CI: 0.725–0.898, P<0.01). The best cut-off value of RDW was 12.55% (sensitivity: 81.8%; specificity: 64%). The area under the curve for MPV/PLT was 0.671 (95% CI: 0.543–0.800, P<0.01). The best cut-off value of MPV/PLT was 0.408 (sensitivity: 77.3%; specificity: 52%) (Figure 1). The best cut-off value was usually determined by the “Youden index”, which was the sum of sensitivity and specificity minus 1, and the value of the index with the highest value was the best cut-off value.
Discussion
Our study evaluated the association between RDW, MPV/PLT, atrial diameter, and paroxysmal AF. The study showed higher RDW, MPV/PLT, and LAD in patients with paroxysmal AF than in non-AF patients. RDW, MPV/PLT, and LAD were independently associated with paroxysmal AF.
Atrial fibrillation, whether paroxysmal or sustained, can cause hemodynamic instability, thromboembolism, stroke, and even death. Various mechanisms, such as inflammation, oxidative stress, and hypercoagulable states, are involved in the development of AF [19]. Compared with patients in sinus rhythm, atrial pathology in patients with AF showed infiltration of lymphomonocytes and necrosis of adjacent myocytes, higher levels of inflammatory markers (such as CRP) in patients with AF, and higher levels of CRP were associated with the presence of AF. In addition, baseline CRP could be used to predict the risk of developing atrial fibrillation. Thus, a persistent inflammatory state could promote atrial fibrillation [20]. Atrial pressure overload and atrial muscle dysfunction during AF lead to an increase in the levels of BNP [21]. In this study, the levels of hs-CRP and NT-pro-BNP in patients with combined paroxysmal AF were significantly higher than those measured in the control group, suggesting that hs-CRP and NT-pro-BNP are associated with paroxysmal AF.
Complete blood count reflected the hemodynamic status and was a predictor of the patient’s clinical prognosis [22]. Numerous studies had shown that elevated RDW levels were associated with adverse cardiovascular events, but not with hemoglobin and hematocrit levels [23]. Our study found that RDW had some predictive value for the occurrence of paroxysmal AF (OR: 2.557, 95% CI: 1.481~4.414). A possible mechanism for RDW-induced paroxysmal atrial fibrillation was that red blood cells play an important role in cardiovascular regulation and inflammation increases RDW levels by altering iron metabolism, inhibiting erythropoietin, and shortening the lifespan of red blood cells. Increased RDW may reflect underlying chronic inflammation [24].
Evidence had shown that MPV is closely related to cardiovascular risk factors, such as diabetes, hypertension, and hypercholesterolemia [25]. The studies had shown an inflammatory and thrombotic predictive role of MPV/PLT in many other diseases, such as obesity, type 2 diabetes, and hepatosteatosis [26]. Our results also showed that MPV/PLT in the paroxysmal AF group was significantly higher than that in the control group, suggesting that this indicator can be used to predict the occurrence of paroxysmal AF.
The currently accepted primary mechanism of atrial fibrillation is abnormal electrical activity in the pulmonary veins. Atrial dilatation can promote further development of atrial fibrillation [27]. Some retrospective analyses have shown that the left atrium gradually increases after AF; as the size of the left atrium increases, patients are more likely to transition from paroxysmal AF to permanent AF [28]. This study showed that the LAD was significantly larger in patients with AF versus the control group. The area under the ROC curve was 0.692, suggesting that LAD has a certain diagnostic value for AF.
Some limitations of this study should be acknowledged. Firstly, the study population was small. Secondly, the duration of AF was not analyzed; currently, it is unclear whether the duration of AF affects the levels of RDW. Third, autonomic function was not evaluated. Finally, although it has been confirmed that RDW levels can predict paroxysmal AF, it is unclear whether RDW is a true marker for paroxysmal AF or merely a disease-related marker in the general population. Therefore, further large-scale prospective studies are warranted to clarify the role of RDW and the right atrium in predicting AF.
Conclusions
In summary, LAD, RDW, and MPV/PLT were associated with paroxysmal AF. MPV/PLT is a newly recognized predictor of AF and can help physicians identify patients at high risk for AF and develop strategies to prevent adverse cardiovascular events in this context.
Tables
Table 1. Clinical and demographic properties of 2 groups.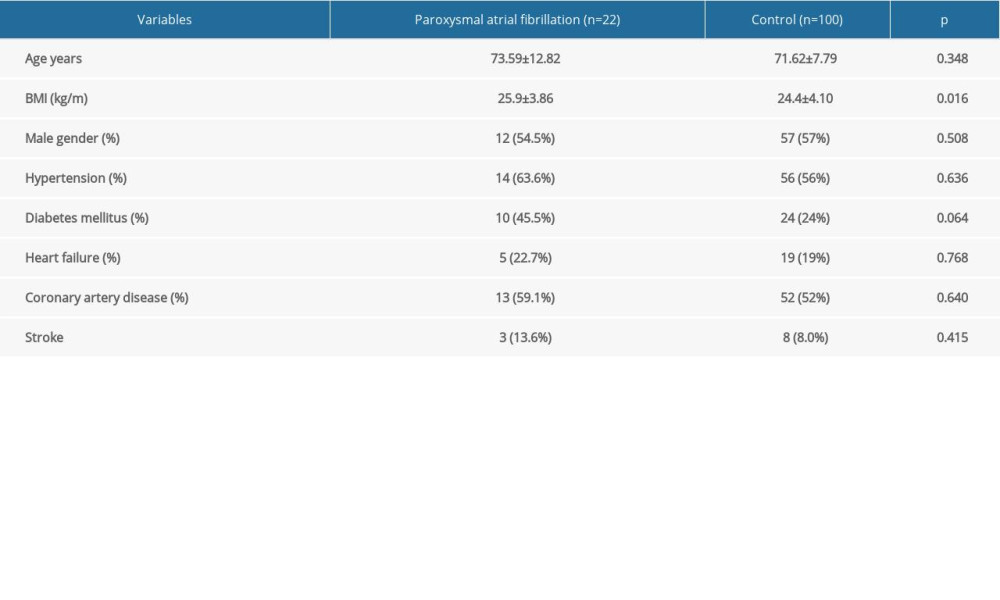 Table 2. Comparison of laboratory parameters and echocardiography parameters in paroxysmal AF and control groups.
Table 2. Comparison of laboratory parameters and echocardiography parameters in paroxysmal AF and control groups.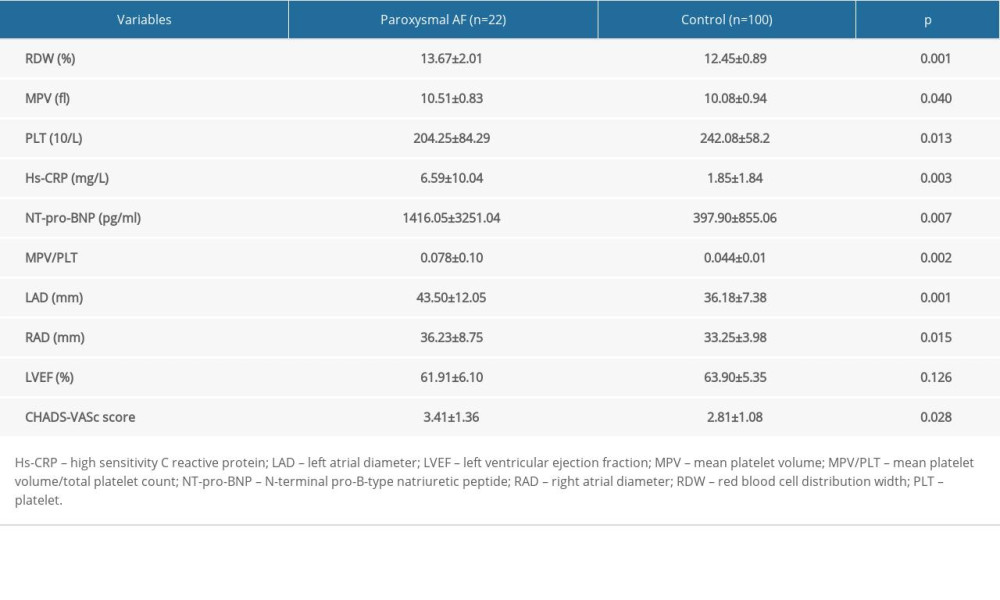 Table 3. Binary logistic regression analysis to detect the independent predictors of paroxysmal atrial fibrillation.
Table 3. Binary logistic regression analysis to detect the independent predictors of paroxysmal atrial fibrillation.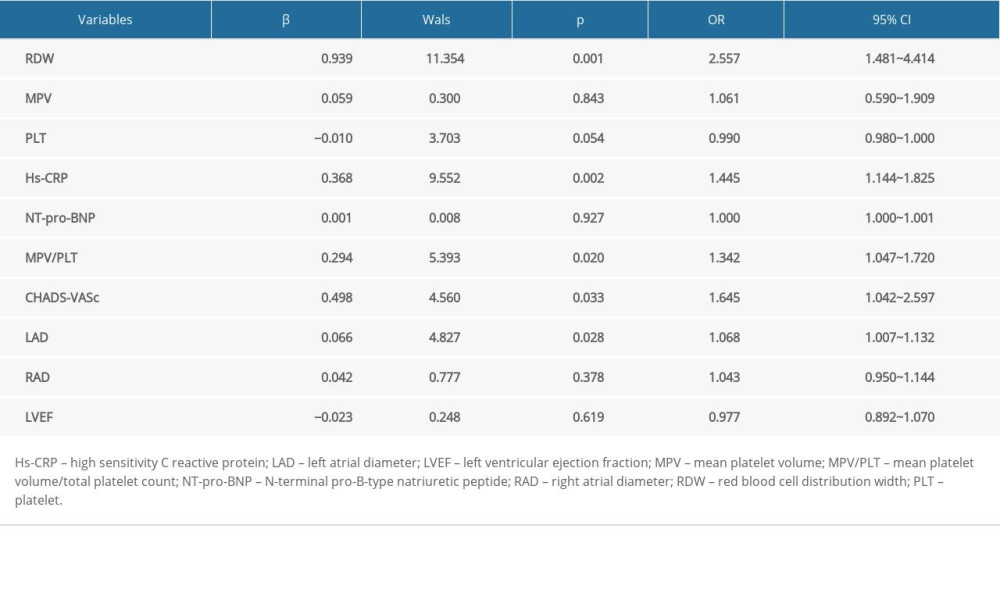 Table 4. Correlation of indicators.
Table 4. Correlation of indicators.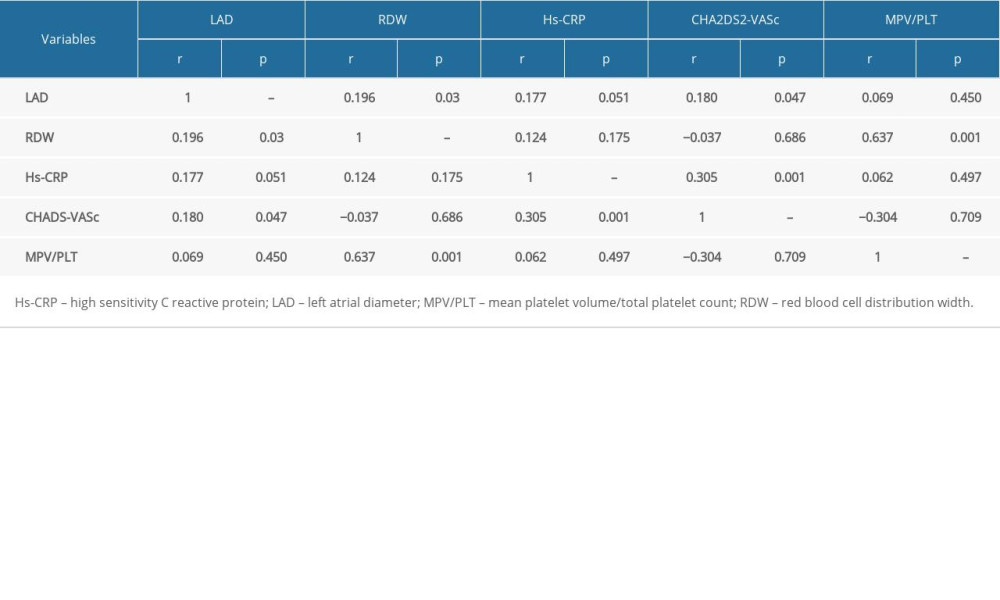
References
1. Lippi G, Sanchis-Gomar F, Cervellin G, Global epidemiology of atrial fibrillation: an increasing epidemic and public health challenge: Int J Stroke Soc, 2021; 16; 217-21
2. Tamirisa KP, Al-Khatib SM, Mohanty S, Racial and ethnic differences in the management of atrial fibrillation: CJC Open, 2021; 3(12 Suppl); S137-48
3. Rodriguez CJ, Soliman EZ, Alonso A, Atrial fibrillation incidence and risk factors in relation to race-ethnicity and the population-attributable fraction of atrial fibrillation risk factors: The Multi-EthnicStudy of Atherosclerosis: Ann Epidemiol, 2015; 25; 71-76
4. Huang CX, Zhang S, Huang DJ, Hua W, AF: Current understanding and treatment recommendations – 2018: Chinese Journal of Cardiac Pacing and Electrophysiology, 2018; 32(4); 315-68
5. Kirchhof P, Benussi S, Kotecha D, 2016 ESC guidelines for the management of atrial fibrillation developed in collaboration with EACTS: Rev Esp Cardiol (Engl Ed), 2017; 70(1); 50
6. Rizzo MR, Sasso FC, Marfella R, Autonomic dysfunction is associated with brief episodes of atrial fibrillation in type 2 diabetes: J Diabetes Complications, 2015; 29(1); 88-92
7. Sun YH, Hu DY, Li YT, Clinical observation on the progression of paroxysmal AF to permanent AF: Chinese Journal of Cardiac Pacing and Electrophysiology, 2004(18); 24-26
8. Sun G, Ma M, Ye N, Diabetes mellitus is an independent risk factor for AF in a general Chinese population: J Diabetes Investig, 2016; 7(5); 791-96
9. Perlstein TS, Weuve J, Pfeffer MA, Beckman JA, Red blood cell distribution width and mortality risk in a community-based prospective cohort: Arch Intern Med, 2009; 169(6); 588-94
10. Kimc J, Kim YD, Song TJ, Red blood cell distribution width is associated with poor clinical outcome in acute cerebral infarction: Thromb Haemost, 2012; 108(2); 349-56
11. Dabbah S, Hammerman H, Markiewicz W, Aronson D, Relation between red cell distribution width and clinical outcomes after acute myocardial infarction: Am J Cardiol, 2010; 105(3); 312-17
12. Emans ME, Gaillard CA, Pfister R, Red cell distribution width is associated with physical inactivity and heart failure, independent of established risk factors, inflammation or iron metabolism; The EPIC-Norfolk study: Int J Cardiol, 2013; 168(4); 3550-55
13. Sardu C, Santulli G, Santamaria M, Effects of alpha lipoic acid on multiple cytokines and biomarkers and recurrence of atrial fibrillation within 1 year of catheter ablation: Am J Cardiol, 2017; 119(9); 1382-86
14. Sardu C, Santulli G, Guerra G, Modulation of SERCA in patients with persistent atrial fibrillation treated by epicardial thoracoscopic ablation: The CAMAF Study: J Clin Med, 2020; 9(2); 544
15. Azab B, Torbey E, Sıngh J, Mean platelet volume/platelet count ratio as a predictor of long-term mortality after non-ST-elevation myocardial infarction: Platelets, 2011; 22; 557-66
16. Sardu C, Carreras G, Katsanos S, Metabolic syndrome is associated with a poor outcome in patients affected by outflow tract premature ventricular contractions treated by catheter ablation: BMC Cardiovasc Disord, 2014; 14; 176
17. Sardu C, Santamaria M, Funaro S, Cardiac electrophysiological alterations and clinical response in cardiac resynchronization therapy with a defibrillator treated patients affected by metabolic syndrome: Medicine (Baltimore), 2017; 96(14); e6558
18. Sardu C, Marfella R, Santamaria M, Stretch, injury and inflammation markers evaluation to predict clinical outcomes after implantable cardioverter defibrillator therapy in heart failure patients with metabolic syndrome: Front Physiol, 2018; 9; 758
19. Tan C, Ou Yang M, Kong D, Zhou X, Association between the left atrial and left atrial appendages systole strain rate in patients with AF: Med Sci Monit, 2016; 22; 4974-77
20. Hu YF, Chen YJ, Lin YJ, Chen SA, Inflammation and the pathogenesis of atrial fibrillation: Nat Rev Cardiol, 2015; 12(4); 230-43
21. Sahin T, Acar E, Celikyurt U, Relation of hs-CRP and BNP levels with the atrial spontaneous echo contrast and thrombi in permanent AF patients with different etiologies: Med Sci Monit, 2012; 18(2); CR78-87
22. Mirhosseini SJ, Ali-Hassan-Sayegh S, Forouzannia SK, What is the exact predictive role of preoperative white blood cell count for new-onset AF following open heart surgery?: Saudi J Anaesth, 2013; 7; 40-42
23. Perlstein TS, Weuve J, Pfeffer MA, Beckman JA, Red blood cell distribution width and mortality risk in a community based prospective cohort: Arch Intern Med, 2009; 169; 588-94
24. Li H, Gu Y, Liu M, The relationship between red blood cell distribution width and AF in Asian population: A cross-sectional study: Pacing Clin Electrophysiol, 2019; 42(9); 1197-203
25. Ünübol M, Ayhan M, Güney E, The relationship between mean platelet volume with microalbuminuria and glycemic control in patients with type II diabetes mellitus: Platelets, 2012; 23; 475-80
26. Vukicevic P, Klisic A, Neskovic V, New markers of platelet activation and reactivity and oxidative stress parameters in patients undergoing coronary artery bypass grafting: Oxid Med Cell Longev, 2021; 2021 8915253
27. Mah DY, Shakti D, Gauvreau K, Relation of left atrial size to AF in patients aged ≤22 years: Am J Cardiol, 2017; 119(1); 52-56
28. Jurin I, Hadžibegović I, Durlen I, Atrium size and red cell distribution width predict AF progression from paroxysmal or persistent to permanent: Acta Clin Belg, 2019; 1-7
Tables
 Table 1. Clinical and demographic properties of 2 groups.
Table 1. Clinical and demographic properties of 2 groups. Table 2. Comparison of laboratory parameters and echocardiography parameters in paroxysmal AF and control groups.
Table 2. Comparison of laboratory parameters and echocardiography parameters in paroxysmal AF and control groups. Table 3. Binary logistic regression analysis to detect the independent predictors of paroxysmal atrial fibrillation.
Table 3. Binary logistic regression analysis to detect the independent predictors of paroxysmal atrial fibrillation. Table 4. Correlation of indicators.
Table 4. Correlation of indicators. Table 1. Clinical and demographic properties of 2 groups.
Table 1. Clinical and demographic properties of 2 groups. Table 2. Comparison of laboratory parameters and echocardiography parameters in paroxysmal AF and control groups.
Table 2. Comparison of laboratory parameters and echocardiography parameters in paroxysmal AF and control groups. Table 3. Binary logistic regression analysis to detect the independent predictors of paroxysmal atrial fibrillation.
Table 3. Binary logistic regression analysis to detect the independent predictors of paroxysmal atrial fibrillation. Table 4. Correlation of indicators.
Table 4. Correlation of indicators. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387