01 December 2022: Clinical Research
Clinical Outcomes of Extracranial Carotid Artery-Related Stroke Eligible for Mechanical Reperfusion on Top of Per-Guidelines Thrombolytic Therapy: Analysis from a 6-Month Consecutive Patient Sample in 2 Centers
Karolina Dzierwa12ABDEF*, Magdalena Knapik234ABDEF, Łukasz Tekieli235ABDEF, Adam Mazurek23ABDEF, Małgorzata Urbańczyk-Zawadzka26BCD, Artur Klecha7ABD, Tomasz Kowalczyk7ABD, Teresa Koźmik7ABD, Łukasz WiewiórkaDOI: 10.12659/MSM.938549
Med Sci Monit 2022; 28:e938549
Abstract
BACKGROUND: Systemic intravenous thrombolysis and mechanical thrombectomy (MT) are guideline-recommended reperfusion therapies in large-vessel-occlusion ischemic stroke. However, for acute ischemic stroke of extracranial carotid artery origin (AIS-CA) there have been no specific trials, resulting in a data gap.
MATERIAL AND METHODS: We evaluated referral/treatment pathways, serial imaging, and neurologic 90-day outcomes in consecutive patients, presenting in a real-life series in 2 stroke centers over a period of 6 months, with AIS-CA eligible for emergency mechanical reperfusion (EMR) on top of thrombolysis as per guideline criteria.
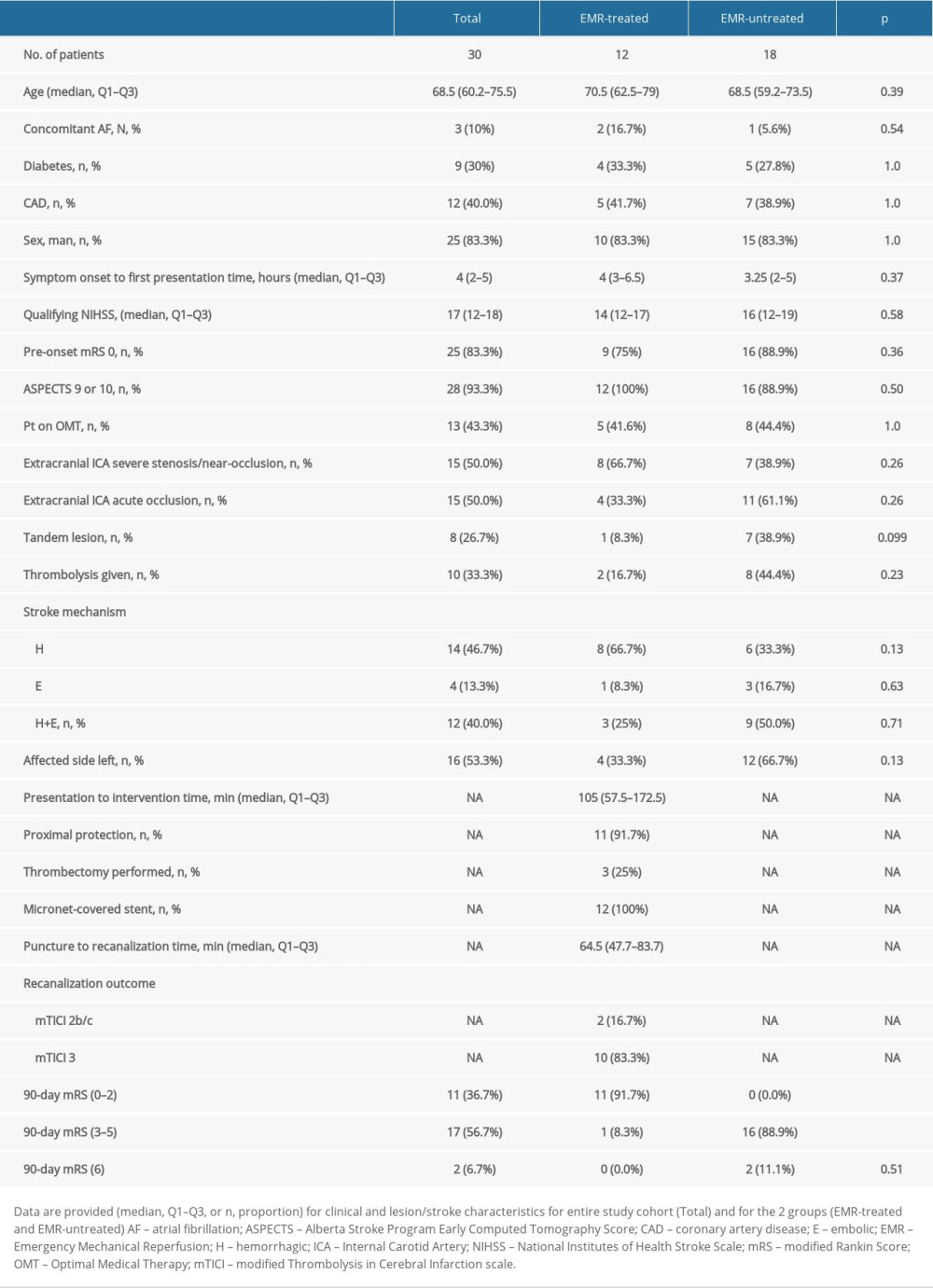
RESULTS: Of 30 EMR-eligible patients (33.3% in-window for thrombolysis and thrombolysed, 73.3% male, age 39-87 years, median Alberta Stroke Program Early Computed Tomography Score (ASPECTS) 10, pre-stroke mRS 0-1 in all, tandem lesions 26.7%), 20 (66.7%) were EMR-referred (60% – endovascular, 6.7% – surgery referrals). Only 40% received EMR, nearly exclusively in stroke centers with carotid artery stenting (CAS) expertise (100% eligible patient acceptance rate, 100% treatment delivery involving CAS±MT with culprit lesion sequestration using micronet-covered stents). The emergency surgery rate was 0%. Baseline clinical and imaging characteristics did not differ between EMR-treated and EMR-untreated patients. Ninety-day neurologic status was profoundly better in EMR-treated patients: mRS 0-2 (91.7% vs 0%; P<0.001); mRS 3-5 (8.3% vs 88.9%; P<0.001), mRS 6 (0% vs 11.1%; P<0.001).
CONCLUSIONS: In a real-life AIS-CA setting, the referral rate of EMR-eligible patients for EMR was low, and the treatment rate was even lower. AIS-CA revascularization was delivered predominantly in stroke thrombectomy-capable cardioangiology centers, resulting in overwhelmingly superior patient outcome. Large vessel occlusion stroke referral and management pathways should involve centers with proximal-protected CAS expertise. AIS-CA, irrespective of any thrombolysis administration, is a hyperacute cerebral emergency and EMR-eligible patients should be immediately referred for mechanical reperfusion.
Keywords: carotid stenosis, endovascular procedures, ischemic stroke, Thrombectomy, Mechanical Thrombolysis, Humans, Male, adult, Middle Aged, Aged, Aged, 80 and over, Female, Stents, Reperfusion, Thrombolytic Therapy, Carotid Artery, Common, Stroke
Background
Carotid atherosclerotic disease is an important [1–4] yet underestimated [5] cause of ischemic stroke. Strokes of carotid bifurcation origin constitute at least 20% of ischemic strokes [1,6]; some of those present with a co-existing occlusion of the intracranial vessel(s) (tandem lesions) [7,8]. Acute ischemic stroke of extracranial carotid artery origin (AIS-CA) has an unfavorable clinical prognosis due to the large volume of affected brain tissue and the typically large thrombus load, with recanalization rates below 10% using systemic intravenous thrombolysis (IVT) [9–11]. IVT is an established part of stroke reperfusion therapy that, in the setting of large vessel occlusions (LVO), should be combined with mechanical reperfusion [10–16]. There is some evidence in intracranial LVO strokes that IVT administration prior to emergency mechanical reperfusion (EMR) may be associated with increased early recanalization rate [14–16]; but this may be less applicable to AIS-CA due to the typically large thrombus burden in AIS-CA [6,9,17]. Some authors suggest that the bleeding risk with mechanical recanalization may be increased if IVT precedes mechanical thrombectomy (MT) [13,18].
Pivotal trials of mechanical intervention in AIS have been focused on intracranial LVO; patients with AIS due to extracranial carotid artery (CA) occlusion were routinely excluded or severely underrepresented [17,19,20]. As a result of trials, MT of the intracranial CA occlusion causing AIS is today recommended in all eligible patients (class I, level of evidence A recommendation) [12,17]. This contrasts with the data gap regarding EMR of causative occlusions of the extracranial internal CA, including tandem lesions. Today, stroke guidelines from the American Heart Association/American Stroke Association [12] indicate that emergent interventional management of extracranial CA lesions causing stroke “may be considered” (class IIb recommendation), in the absence of clarification regarding which treatment method (ie, endovascular [7,14,21], or carotid surgery±intracranial thrombectomy [21,22]) should be used. Surprisingly, the current European Stroke Organization guideline on carotid stenosis management states that it does “not address carotid revascularization done as part of acute stroke therapy”, leaving European stroke physicians without a guideline on AIS-CA patient management [23]. The relevance of those gaps for everyday clinical practice decision-making remains unknown.
Recent technological advances allow thrombus extraction using aspiration and/or stentrievers, with a high recanalization rate [11,17], from the extracranial internal CA (similarly to the intracranial segments of internal CA [24]), as well as a full reconstruction of the CA lumen with lesion sequestration [25]. However, data on the implementation of these new technologies in acute stroke are scarce [24].
In a sample of consecutive EMR-eligible patients in 2 centers, we performed an assessment of the real-life impact of randomized evidence gaps (resulting in class IIb guideline recommendation level) on contemporary referral and treatment pathways, as well as neurologic outcomes, in AIS-CA.
Material and Methods
ETHICS APPROVAL:
The study was approved by the Regional Ethics Committee (OIL/KBL/75/2021). Individual patient consents were waived due to retrospective analysis of anonymized data.
STUDY CENTERS:
Prospective data collection involved all consecutive patients with AIS receiving cerebral and vascular imaging in 2 collaborating centers with acute stroke neurology. Study Center 1 is a high-volume major tertiary 24/7 cardiovascular center with carotid artery stenting (CAS) expertise with a case load of 350–400 CAS procedures per year [26–28] and MT service under World Federation for Stroke Treatment (WIST) certification and supervision (Level-2 stroke center – Thrombectomy-Capable Stroke Center, TCSC) [24,29]. In addition, vascular surgery treatments (including carotid endarterectomy of ≈200 cases per year) are performed [26–28]. A multispecialty local Stroke Endovascular Mechanical Reperfusion Team with interventional cardiology and angiology, stroke neurology, radiology, and anesthesia is operational within the Level-2 WIST stroke center framework [24,29].
Study Center 2 is a major district hospital (MDH) incorporating regional stroke neurology and on-site 24/7 interventional cardiovascular facilities with neuroradiology procedures performed on an elective basis. Center 2 has developed a CAS program and is presently en route to serve as a stroke MT (Level-2) center for patients in the region. Study Center 2 has a well-established cardiovascular collaboration with Study Center 1. However, its LVO stroke patients are typically referred to a regional Comprehensive Stroke Center (CSC, Level-1 stroke center). Travel time from Study Center 2 to the CSC is ≈90 min, resulting in a minimum transfer time of 120 min.
There are 5 Level-3 stroke centers in the region; these collaborate with both the CSC and TCSC. The CSC is the only Level 1 stroke center in an administrative area of about 3.5 million inhabitants.
PATIENT ELIGIBILITY:
In a prospectively-run stroke imaging database in Center 1 and Center 2, 32 consecutive patients with AIS-CA were identified over a 6-month period. For the 2 patients accepted for EMR by the CSC, no final treatment status (ie, EMR delivered vs not performed) and no clinical outcome data could be obtained, resulting in 30 subjects in the present analysis.
A clinical committee consisting of a neurologist, diagnostic radiologist, and stroke management-certified interventionist reviewed the clinical and imaging data in all consecutive AIS-CA stroke patients. Eligibility for EMR [12] was defined as: (1) NIHSS (National Institutes of Health Stroke Scale) ≥6 or a significant neurologic deficit (eg, aphasia), (2) ASPECTS (Alberta Stroke Program Early Computed Tomography Score) ≥6, (3) pre-stroke modified Rankin scale (mRS) ≤2, and (4) evidence of internal carotid artery occlusion/sub-occlusion on computed tomography angiography (CTA). For each study patient, their EMR eligibility was verified by a senior interventional neuroradiologist with over 15 years of experience in acute stroke interventions [30]. Administration of IV -thrombolysis, referral for EMR (surgical or endovascular), and the treatment received were evaluated.
ENDOVASCULAR TREATMENT STRATEGY:
The endovascular EMR strategy involved internal CA revascularization under routine flow-reversal cerebral protection, which reduces cerebral embolism in elective CAS [31–34] and in stroke intracranial interventions [35,36]. Aspiration or carotid-dedicated large-diameter thrombectomy device [24] was used in case of CA large thrombus. Intracranial LVO MT [19] was performed in tandem lesions. The culprit CA lesion sequestration was performed using micronet-covered stents [4,25,28,37,38]. Functional status was routinely assessed at 90 days.
PERIPROCEDURAL IMAGING AND PHARMACOLOGICAL TREATMENT REGIMEN:
Guideline-recommended [12] study imaging involved: plain cerebral computed tomography (cCT), CTA, cerebral magnetic resonance imaging (MRI) with diffusion-weighted imaging (DWI), apparent diffusion coefficient (ADC); fluid-attenuated inversion recovery (FLAIR) sequences, carotid Doppler ultrasound (if performed), and (in patients with endovascular treatment) catheter angiography (Figures 1, 2). Perfusion imaging was performed as clinically indicated or as required to establish EMR eligibility.
Optimal (as per current criteria) medical therapy (OMT) prior to stroke onset was defined as pharmacological treatment involving at least 1 antiplatelet agent, statin dose titrated to achieve guideline-indicated LDL cholesterol level (or maximally tolerated dose), and angiotensin-converting enzyme inhibitor/receptor blocker [39].
At the index event, IVT was administered in eligible patients (ie, those without contraindications to thrombolysis and presenting within 4.5 h after stroke onset) [12]. In the group receiving endovascular treatment, peri-procedural pharmacotherapy included heparinization at a reduced or full dose (at operator’s discretion) and a single periprocedural antiplatelet agent (IV acetylsalicylic acid), followed by a second antiplatelet agent after the intervention [12].
Care was taken to provide a tight periprocedural blood pressure control with: (1) systolic blood pressure of 160–180 mmHg prior to and during the intervention (to enhance flow through the stroke-related CA and enhance collateral supply), followed by (2) systolic blood pressure reduction to 100–120 mmHg after reperfusion of the intracranial vessels to reduce the risk of cerebral bleed [40].
STATISTICAL ANALYSIS:
Continuous variables were reported as median (Q1–Q3) and categorical data were expressed as numbers and proportions. The Mann-Whitney U test or Wilcoxon matched pairs test was used for between-group and within-group comparisons. Bonferroni correction was applied for multiple comparisons. Statistical significance was defined as
Results
STUDY COHORT:
Table 1 shows clinical data in consecutive AIS-CA patients in the study (age 39–87 years, 26.7% women): 93.3% had ASPECTS ≥9; NIHSS was 12–22 (median 17); 83.3% had pre-stroke mRS-0, 16.7% mRS-1; all eligible patients received thrombolysis (33.3%); and 20 (66.7%) were referred for EMR (18 for emergency endovascular management, 2 for emergency carotid endarterectomy). Figures 1 and 2 present typical examples of the disease course in EMR-untreated and EMR-treated patients on top of IVT as per guidelines.
IMAGING AND CLINICAL PATHWAYS:
Seventy five percent EMR-treatment-eligible referrals were rejected by the CSC. Out of the 2 patients referred for emergency surgery (carotid endarterectomy, CEA), none were treated (1 rejected on referral, 1 initially accepted for CEA but surgery was not performed). Once rejected, an alternative center was not contacted or alternative reperfusion method referral was not attempted in any of the EMR-eligible patients rejected by CSC or vascular surgery.
Only 40% of all study participants (60% of all EMR referrals, n=12 patients) received EMR. The EMR-untreated group included EMR-eligible patients who were either not referred or were referred but rejected (n=18; 60% of the study cohort). There were no significant clinical, demographic, or lesion-related differences between the EMR-untreated group and EMR-treated group, including NIHSS and functional status prior to stroke onset (Table 1). Prior to stroke onset, 13 patients (43.3%) were on documented OMT.
Eighty percent of patients who (subsequently) did not receive EMR presented within 6 h of symptom onset and had a favorable ASPECTS ≥6. A further 20% of the EMR-untreated group were wake-up strokes with favorable DWI/FLAIR mismatch and an ASPECTS ≥6, consistent with EMR eligibility. Similarly, 75% of EMR-treated patients presented within the 6-h window. Cerebral perfusion imaging confirmed EMR eligibility in a further 12.5% who presented with stuttering symptoms. Another 12.5% of EMR-treated patients initially presented with a “low” NIHSS (NIHSS of 3 and 4) in peripheral hospitals, without an initial referral. These patients were referred for EMR when their NIHSS exceeded 6; imaging repeated within 1 h of EMR still showed ASPECTS ≥6.
In the patients who did not receive EMR despite eligibility, the reasons were (1) lack of referral (56%), (2) being referred but not accepted for EMR treatment by CSC (40.7%), or carotid endarterectomy treatment not being performed (3.3%, no patient referred for CEA or referred and accepted for CEA received CEA).
REPERFUSION STRATEGY:
IVT was administered in all eligible patients (10, 33.3%), of which only 2 were immediately transferred for EMR (Table 1).
Proximal cerebral protection with transient flow reversal was applied if feasible (91.7%; distal filter use in 8.3% of interventions). In patients with particularly large thrombus burden at the carotid bifurcation, thrombectomy was performed using a dedicated large-diameter (9-mm) stentriever on top of aspiration (Figure 2B) [24]. Tandem lesion presence mandated intracranial thrombectomy performed with aspiration as the technique of 1st choice (followed, if needed, by stentriever). Micronet-covered stents were routinely used for culprit lesion sequestration and lumen reconstruction (length 30 or 40 mm, diameters 9.0 or 10.0 mm). Stent post-dilatation optimization was routinely performed to ensure device embedding. Modified Thrombolysis in Cerebral Infarction (TICI) reperfusion grade 2b–c/3 was achieved in all EMR-treated cases.
NEUROLOGIC OUTCOMES:
Good neurologic outcomes (mRS 0–2, functional independence) at 90 days occurred exclusively in the EMR-treated group (91.7% vs 0%, P<0.001, Figure 3B). Poor functional outcome (mRS 3–5) prevalence was 8.3% vs 88.9% (P<0.001, EMR-treated vs untreated, Figure 3B). Intracerebral bleeding occurred in 1 patient (EMR-untreated group, thrombolysis given). There were 2 deaths by 90 days after the intervention (11.1%, mRS 6), and both occurred in the EMR-untreated group. The neurologic outcome difference between the study groups (Figure 3) was driven by patients who reached mRS 0–2 (P<0.001).
Discussion
LIMITATIONS:
Limitations include our retrospective analysis of prospectively collected data and the sample size. In the era of MT, obtaining further randomized evidence with regard to AIS-CA management in patients who qualify for EMR on top of thrombolysis is unlikely, as this would be unethical today. While the sample size may be considered moderate, it is similar to other AIS-CA cohort studies published [7,41,46,47]. An important element of our present work is the comparison of contemporary outcomes in the treated and untreated AIS-CA patients (Figure 3). With the moderate study size, however, the present comparison needs to be regarded as a pilot study with respect to the size of the treatment effect. As it would be unethical today to randomize AIS-CA patients to emergency intervention vs no intervention (or delayed intervention) [12], it is crucial to expand the body of knowledge by analysis of real-life datasets such as ours. Next, larger datasets, such as data from multiple centers and/or regional or national databases, are needed. However, it must be noted that detailed patient-level verification of criteria for emergency mechanical recanalization (which we achieved through detailed analysis using access to full imaging and clinical data) would be difficult for large patient cohorts with limited access to source data.
Regrettably, we could not obtain any specific information regarding reasons for non-referral/non-acceptance for EMR. Likely contributing factors include: (1) “symptomatic” carotid disease being historically considered a primarily surgical disease, to be addressed within 14 days from symptom onset [23]; (2) CSCs primary focus on intracranial LVO management [8,19,20]; (3) unfamiliarity with the treatment of carotid origin disease under flow reversal [7,21,45]; (4) shortage of stroke MT centers[50,51]; and (5) lack of established pathways for emergency AIS-CA stroke referrals that would involve centers with expertise in endovascular carotid revascularization [49]. Furthermore, clinical practice suggests that rejection of a particular patient type will impact further referrals of similar patients. Thus, “symptomatic” (elective) CA stenosis guidelines should not be applied blindly to AIS-CA, but should rather be used together with the acute stroke guidelines that demand emergency reperfusion not only in intracranial LVO but also in carotid LVO [12].
Finally, it is important to note that in many patients with large carotid-related strokes, once they reach the post-acute stroke stage, any potential intervention “within 14 days” [39] (whether surgical or endovascular) becomes futile due to the large irreversible loss of brain tissue and an unacceptable risk of intracranial hemorrhage. Such patients are not included in most statistics on symptomatic carotid stenosis and its revascularization.
Conclusions
An acute stroke due to an underlying carotid lesion represents a cerebral emergency, irrespective of the presence/absence of co-existing intracranial LVO and irrespective of any thrombolysis administration. Our analysis suggests that patients receiving emergency mechanical reperfusion have overwhelmingly better clinical outcomes. To increase the proportion of AIS-CA patients who receive mechanical reperfusion in acute stroke, cardioangiology centers with expertise in endovascular carotid revascularization and with knowledge of proximal cerebral protection and stenting should actively participate in delivering mechanical reperfusion for patients with acute stroke [55].
Figures
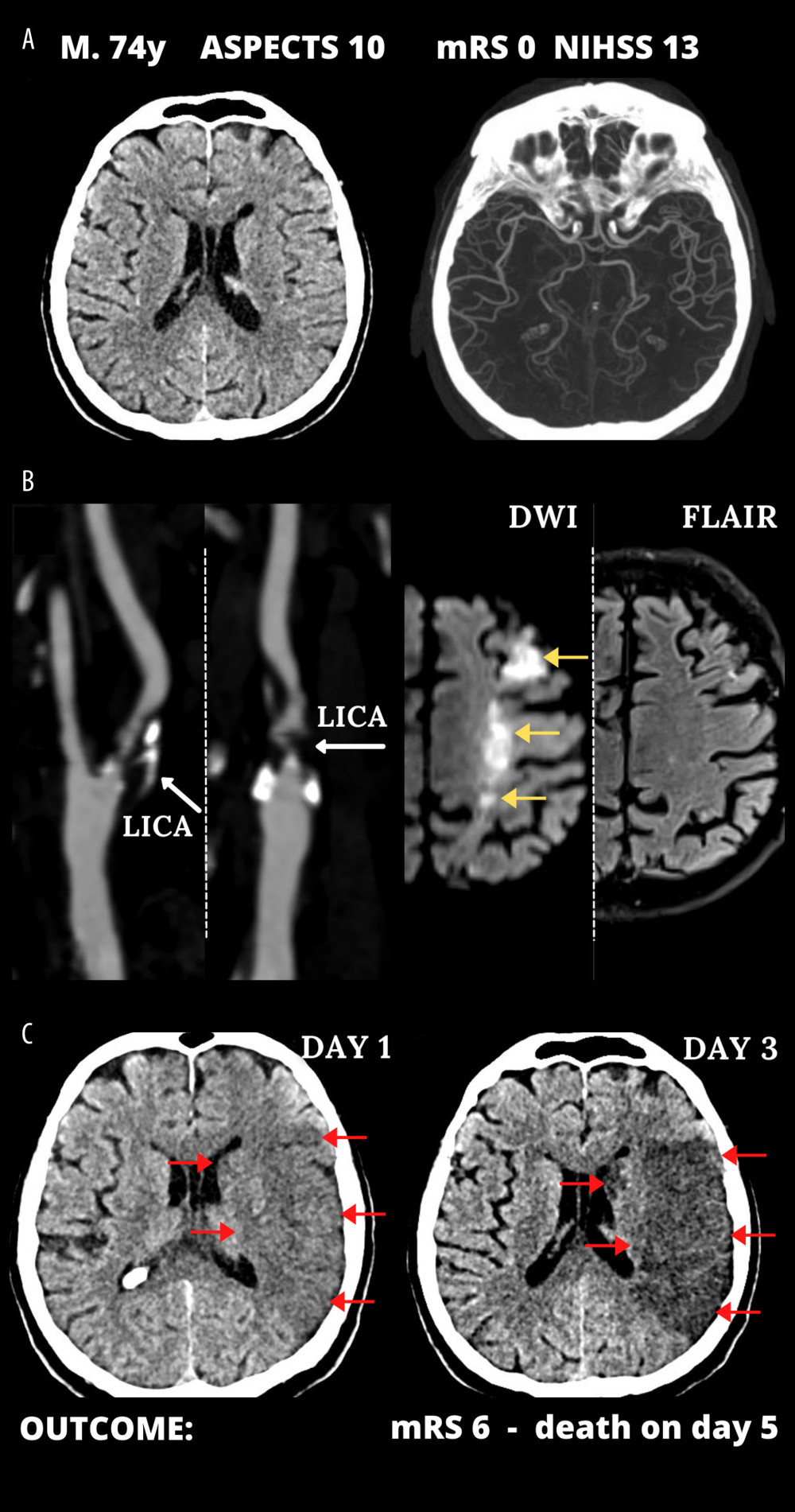 Figure 1. Typical cerebral infarct evolution in an acute ischemic stroke of the carotid artery origin in emergency mechanical reperfusion (EMR) eligible patient that did not receive EMR (EMR-untreated). (A) Admission cerebral computed tomography (day 0) was normal; Alberta Stroke Program Early Computed Tomography Score (ASPECTS) 10 in a man presenting with left hemispheric stroke symptoms of increasing severity National Institutes of Health Stroke Scale (NIHSS) – 13 (left) and computed tomography angiography showed no intracranial artery occlusion and good collaterals (Tan 3) (right). (B) Sub-occlusive left internal carotid artery (LICA) stenosis (left); magnetic resonance imaging demonstrated potentially reversible hyperacute left-sided diffusion restriction on diffusion-weighted imaging (DWI, yellow arrows) which are absent on the fluid-attenuated inversion recovery (FLAIR) sequence (right). IV thrombolysis was started, and the patient was observed for thrombolysis effect; there was no referral for EMR. Neurologic status gradually deteriorated. (C) Large cerebral tissue loss (red arrows) seen on control computed tomography 12 hours after 1st scan (left, thrombolysis ineffective, collateral supply exhaustion) and on day 3 (right). Figure was created with the use of Canva (Perth, Australia).
Figure 1. Typical cerebral infarct evolution in an acute ischemic stroke of the carotid artery origin in emergency mechanical reperfusion (EMR) eligible patient that did not receive EMR (EMR-untreated). (A) Admission cerebral computed tomography (day 0) was normal; Alberta Stroke Program Early Computed Tomography Score (ASPECTS) 10 in a man presenting with left hemispheric stroke symptoms of increasing severity National Institutes of Health Stroke Scale (NIHSS) – 13 (left) and computed tomography angiography showed no intracranial artery occlusion and good collaterals (Tan 3) (right). (B) Sub-occlusive left internal carotid artery (LICA) stenosis (left); magnetic resonance imaging demonstrated potentially reversible hyperacute left-sided diffusion restriction on diffusion-weighted imaging (DWI, yellow arrows) which are absent on the fluid-attenuated inversion recovery (FLAIR) sequence (right). IV thrombolysis was started, and the patient was observed for thrombolysis effect; there was no referral for EMR. Neurologic status gradually deteriorated. (C) Large cerebral tissue loss (red arrows) seen on control computed tomography 12 hours after 1st scan (left, thrombolysis ineffective, collateral supply exhaustion) and on day 3 (right). Figure was created with the use of Canva (Perth, Australia). 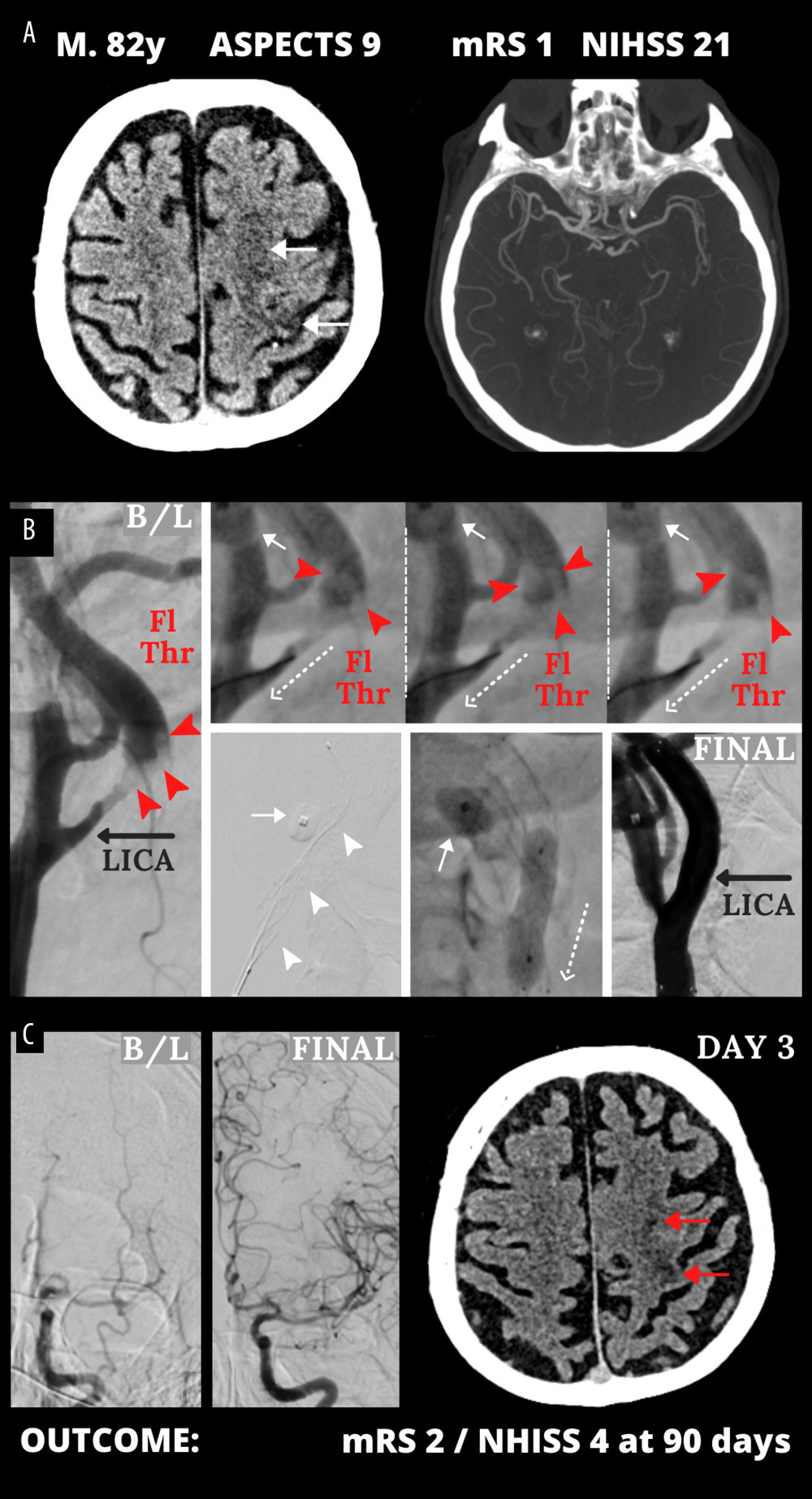 Figure 2. Emergency mechanical reperfusion in acute carotid artery origin ischemic stroke. (A), (left panel) – mild vascular changes (white arrows) seen on admission cerebral computed tomography Alberta Stroke Program Early Computed Tomography Score (ASPECTS) 9 in a patient who was emergency-transferred to a cardioangiology-based Thrombectomy-Capable Stroke Center from external Neurology; (right panel) – diminished flow to the left hemisphere on computed tomography angiography (compare left vs right). (B) Catheter angiography – left internal carotid artery (LICA) near-occlusion with a floating thrombus (Fl Thr, red arrowheads) and next stages of emergency mechanical reperfusion (from left to right, upper and lower panels): transient flow reversal (dotted arrows; enhanced by active aspirations at the procedure critical steps) – proximal cerebral protection device – Mo.Ma (Medtronic, Tolochenaz, Switzerland), external carotid artery balloon (white arrow). Following thrombectomy (carotid-dedicated adjustable-diameter stentriever – TigerTrieverXL (Rapid Medical, Yokneam, Israel), the culprit lesion was sequestrated (white arrowheads), using a micronet-covered stent – C-Guard (InspireMD, Tel Aviv, Israel) with post-dilatation embedding. (C), (left panel) – effective lumen reconstruction resulted in normalized left hemispheric cerebral blood supply symptoms regressed, (right panel) – discharge cerebral computed tomography showed a minor cerebral infarct (red arrows). B/L indicates baseline, (mag) = magnified image. Figure was created with the use of Canva (Perth, Australia).
Figure 2. Emergency mechanical reperfusion in acute carotid artery origin ischemic stroke. (A), (left panel) – mild vascular changes (white arrows) seen on admission cerebral computed tomography Alberta Stroke Program Early Computed Tomography Score (ASPECTS) 9 in a patient who was emergency-transferred to a cardioangiology-based Thrombectomy-Capable Stroke Center from external Neurology; (right panel) – diminished flow to the left hemisphere on computed tomography angiography (compare left vs right). (B) Catheter angiography – left internal carotid artery (LICA) near-occlusion with a floating thrombus (Fl Thr, red arrowheads) and next stages of emergency mechanical reperfusion (from left to right, upper and lower panels): transient flow reversal (dotted arrows; enhanced by active aspirations at the procedure critical steps) – proximal cerebral protection device – Mo.Ma (Medtronic, Tolochenaz, Switzerland), external carotid artery balloon (white arrow). Following thrombectomy (carotid-dedicated adjustable-diameter stentriever – TigerTrieverXL (Rapid Medical, Yokneam, Israel), the culprit lesion was sequestrated (white arrowheads), using a micronet-covered stent – C-Guard (InspireMD, Tel Aviv, Israel) with post-dilatation embedding. (C), (left panel) – effective lumen reconstruction resulted in normalized left hemispheric cerebral blood supply symptoms regressed, (right panel) – discharge cerebral computed tomography showed a minor cerebral infarct (red arrows). B/L indicates baseline, (mag) = magnified image. Figure was created with the use of Canva (Perth, Australia). 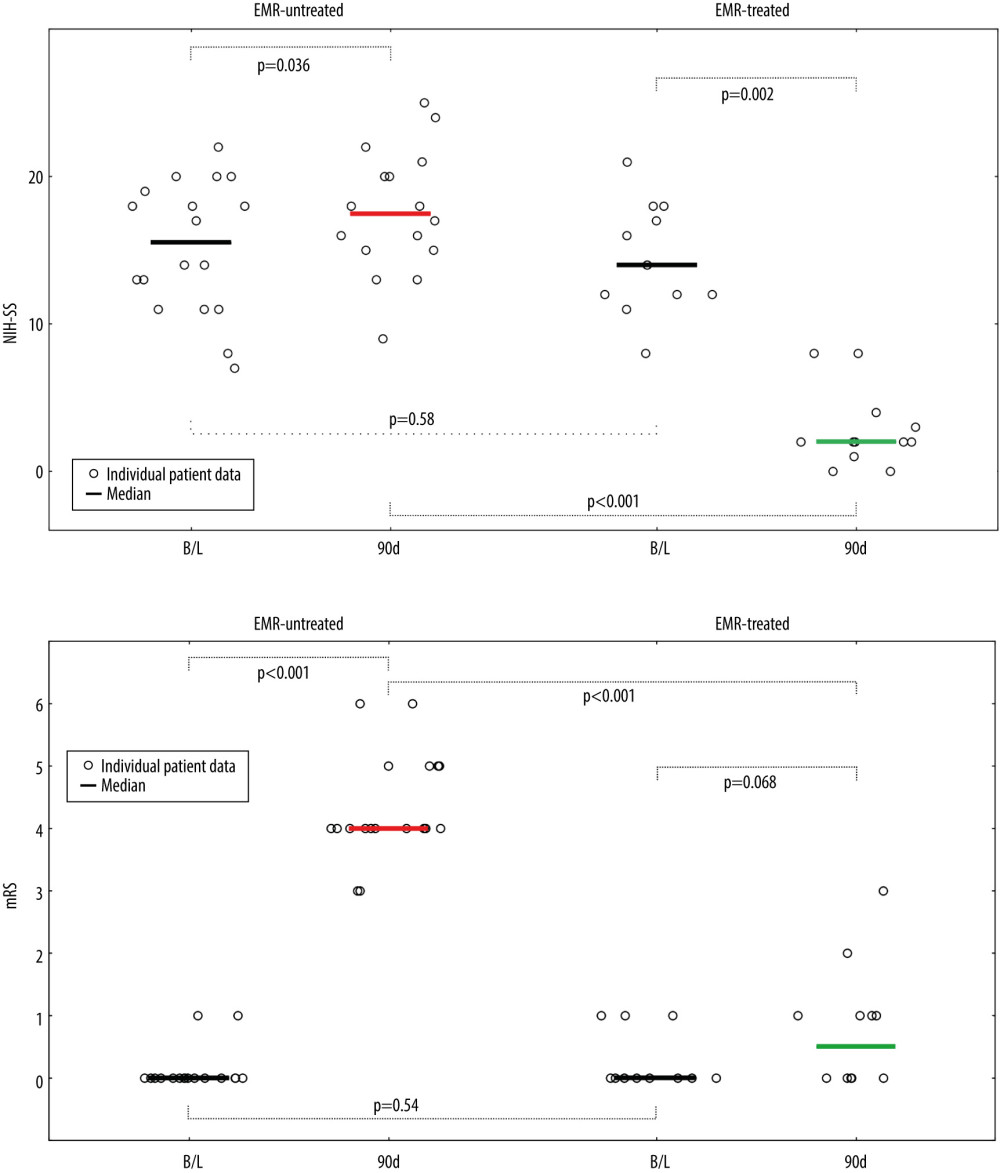 Figure 3. Evolution of National Institutes of Health Stroke Scale (NIHSS), (A) and functional status (modified Rankin Score – mRS), (B) in patients not treated and treated with EMR (Emergency Mechanical Reperfusion). Individual patient and group data on acute ischemic stroke of carotid artery origin (AIS-CA) clinical severity (NIHSS, A) and patient functional status (mRS, B) are provided at baseline at 90 days. Emergency mechanical reperfusion (EMR) treatment effect is demonstrated by comparison of EMR-untreated (natural history, left) and EMR-treated patients (right). With the particularly large volume of cerebral tissue-at-risk in AIS-CA, EMR profoundly impacts clinical outcomes. Note the striking difference in NIHSS and mRS at 90 days in EMR-treated patients versus those who did not receive mechanical reperfusion. The EMR-untreated patients would have been accepted for treatment in the cardioangiology cathlab-based Thrombectomy-Capable Stroke Center (operator team with experience in proximal-protected carotid artery stenting). The figure was created with the use of Statistica 10 (StatSoft GmBH, Hamburg, Germany).
Figure 3. Evolution of National Institutes of Health Stroke Scale (NIHSS), (A) and functional status (modified Rankin Score – mRS), (B) in patients not treated and treated with EMR (Emergency Mechanical Reperfusion). Individual patient and group data on acute ischemic stroke of carotid artery origin (AIS-CA) clinical severity (NIHSS, A) and patient functional status (mRS, B) are provided at baseline at 90 days. Emergency mechanical reperfusion (EMR) treatment effect is demonstrated by comparison of EMR-untreated (natural history, left) and EMR-treated patients (right). With the particularly large volume of cerebral tissue-at-risk in AIS-CA, EMR profoundly impacts clinical outcomes. Note the striking difference in NIHSS and mRS at 90 days in EMR-treated patients versus those who did not receive mechanical reperfusion. The EMR-untreated patients would have been accepted for treatment in the cardioangiology cathlab-based Thrombectomy-Capable Stroke Center (operator team with experience in proximal-protected carotid artery stenting). The figure was created with the use of Statistica 10 (StatSoft GmBH, Hamburg, Germany). References
1. Marsh JD, Keyrouz SG, Stroke prevention and treatment: J Am Coll Cardiol, 2010; 56; 683-91
2. Spagnoli LG, Mauriello A, Sangiorgi G, Extracranial thrombotically active carotid plaque as a risk factor for ischemic stroke: JAMA, 2004; 292; 1845-52
3. Alagoz AN, Acar BA, Acar T, Relationship between carotid stenosis and infarct volume in ischemic stroke patients: Med Sci Monit, 2016; 22; 4954-59
4. Musialek P, Rosenfield K, Siddiqui AH, Grunwald IQ, Carotid stenosis and stroke: Medicines, stents, surgery – “wait-and-see” or protect?: Thromb Haemost, 2022 [Online ahead of print]
5. Abbott AL, Nederkoorn PJ, Outcomes are improving for patients with carotid stenosis: Neurology, 2015; 85; 302-3
6. Rubiera M, Ribo M, Delgado-Mederos R, Tandem internal carotid artery/middle cerebral artery occlusion: An independent predictor of poor outcome after systemic thrombolysis: Stroke, 2006; 37; 2301-5
7. Mizowaki T, Fujita A, Inoue S, Outcome and effect of endovascular treatment in stroke associated with acute extracranial internal carotid artery occlusion: Single-center experience in Japan: J Stroke Cerebrovasc Dis, 2020; 29; 104824
8. Cirillo L, Romano DG, Vornetti G, Acute ischemic stroke with cervical internal carotid artery steno-occlusive lesion: Multicenter analysis of endovascular approaches: BMC Neurol, 2021; 21; 362
9. Bhatia R, Hill MD, Shobha N, Low rates of acute recanalization with intravenous recombinant tissue plasminogen activator in ischemic stroke: Real-world experience and a call for action: Stroke, 2010; 41; 2254-58
10. Saver JL, Adeoye O, Intravenous thrombolysis before endovascular thrombectomy for acute ischemic stroke: JAMA, 2021; 325; 229
11. Imran R, Mohamed GA, Nahab F, Acute reperfusion therapies for acute ischemic stroke: J Clin Med, 2021; 10; 3677
12. Powers WJ, Rabinstein AA, Ackerson T, 2019 update to the 2018 guidelines for the early management of acute ischemic stroke: Stroke, 2019; 50; e344-e418
13. Suzuki K, Matsumaru Y, Takeuchi M, Effect of mechanical thrombectomy without vs with intravenous thrombolysis on functional outcome among patients with acute ischemic stroke: The SKIP randomized clinical trial: JAMA, 2021; 325; 244
14. Romoli M, Mosconi MG, Pierini P, Reperfusion strategies in stroke due to isolated cervical internal carotid artery occlusion: Systematic review and treatment comparison: Neurol Sci, 2021; 42; 2301-8
15. Podlasek A, Dhillon PS, Butt W, To bridge or not to bridge: Summary of the new evidence in endovascular stroke treatment: Stroke Vasc Neurol, 2022; 7; 179-81
16. Procházka V, Jonszta T, Czerny D, Comparison of mechanical thrombectomy with contact aspiration, stent retriever, and combined procedures in patients with large-vessel occlusion in acute ischemic stroke: Med Sci Monit, 2018; 24; 9342-53
17. Jadhav AP, Desai SM, Jovin TG, Indications for mechanical thrombectomy for acute ischemic stroke: Current guidelines and beyond: Neurology, 2021; 97(20 Suppl 2); S126-36
18. Furlan NE, Luvizutto GJ, Hamamoto Filho PT, Clinical and functional outcomes of patients receiving cerebral reperfusion therapy: A Stroke Databank Study in Brazil: Front Surg, 2022; 9; 799485
19. Rajkumar CA, Ganesananthan S, Ahmad Y, Mechanical thrombectomy with retrievable stents and aspiration catheters for acute ischaemic stroke: A meta-analysis of randomised controlled trials: Eurointervention, 2022; 17; e1425-e34
20. Sheth KN, Saver JL, Starkman S, Enrollment bias: Frequency and impact on patient selection in endovascular stroke trials: J Neurointerv Surg, 2016; 8; 353-59
21. Knappich C, Kuehnl A, Tsantilas P, Patient characteristics and in-hospital outcomes of emergency carotid endarterectomy and carotid stenting after stroke in evolution: J Vasc Surg, 2018; 68; 436-44
22. Gajin P, Radak DJ, Tanaskovic S, Urgent carotid endarterectomy in patients with acute neurological ischemic events within six hours after symptoms onset: Vascular, 2014; 22; 167-73
23. Bonati LH, Kakkos S, Berkefeld J, European Stroke Organisation guideline on endarterectomy and stenting for carotid artery stenosis: Eur Stroke J, 2021; 62; I-XLVII
24. Tekieli L, Banaszkiewicz K, Moczulski Z, Novel large-diameter controlled-expansion stentriever, embolic-prevention stent and flow reversal in large-thrombus-burden ICA proximal occlusion stroke: JACC Cardiovasc Interv, 2021; 14; e287-e91
25. Karpenko A, Bugurov S, Ignatenko P, Randomized controlled trial of conventional versus MicroNet-Covered stent in carotid artery revascularization: JACC Cardiovasc Interv, 2021; 14; 2377-87
26. Tekieli L, Musialek P, Kablak-Ziembicka A, Severe, recurrent in-stent carotid restenosis: endovascular approach, risk factors. Results from a prospective academic registry of 2637 consecutive carotid artery stenting procedures (TARGET-CAS): Adv Interv Cardiol, 2019; 15; 465-71
27. Dzierwa K, Piatek J, Paluszek P, One-day, sequential carotid artery stenting followed by cardiac surgery in patients with severe carotid and cardiac disease: Vasc Med, 2019; 24; 431-38
28. Musialek P, Mazurek A, Trystula M, Novel PARADIGM in carotid revascularisation: Prospective evaluation of All-comer peRcutaneous cArotiD revascularisation in symptomatic and Increased-risk asymptomatic carotid artery stenosis using CGuard MicroNet-covered embolic prevention stent system: EuroIntervention, 2016; 12; 658-70
29. Tekieli L, Tomaszewski T, Musial R, Large-vessel occlusion, large thrombus burden acute stroke in acute pulmonary embolism: A single multi-specialty multi-skill team treatment optimization: Pol Heart J, 2021; 79; 1382-84
30. Grunwald IQ, Walter S, Papanagiotou P, Revascularization in acute ischaemic stroke using the Penumbra system: The first single center experience: Eur J Neurol, 2009; 16(11); 1210-16
31. Grunwald IQ, Reith W, Kühn AL, Proximal protection with the Gore PAES can reduce DWI lesion size in high-grade stenosis during carotid stenting: Eurointervention, 2014; 10; 271-76
32. Bijuklic K, Wandler A, Hazizi F, Schofer J, The PROFI study (Prevention of Cerebral Embolization by Proximal Balloon Occlusion Compared to Filter Protection During Carotid Artery Stenting): A prospective randomized trial: J Am Coll Cardiol, 2012; 59; 1383-89
33. Montorsi P, Caputi L, Galli S, Microembolization during carotid artery stenting in patients with high-risk, lipid-rich plaque. A randomized trial of proximal versus distal cerebral protection: J Am Coll Cardiol, 2011; 58; 1656-63
34. Stabile E, Sannino A, Schiattarella GC, Cerebral embolic lesions detected with diffusion-weighted magnetic resonance imaging following carotid artery stenting: JACC Cardiovasc Interv, 2014; 7; 1177-83
35. Pederson JM, Reierson NL, Hardy N, Comparison of balloon guide catheters and standard guide catheters for acute ischemic stroke: A systematic review and meta-analysis: World Neurosurg, 2021; 154; 144-53
36. Brinjikji W, Starke RM, Murad MH, Impact of balloon guide catheter on technical and clinical outcomes: A systematic review and metaanalysis: J Neurointervent Surg, 2018; 10; 335-39
37. Schofer J, Musialek P, Bijuklic K, A prospective, multicenter study of a novel mesh-covered carotid stent: The CGuard CARENET Trial (Carotid Embolic Protection Using MicroNet): JACC Cardiovasc Interv, 2015; 8; 1229-34
38. Musialek P, Mazurek A, Kolvenbach R, 5-year clinical and ultrasound outcomes in CARENET Prospective Multicenter Trial of CGuard MicroNET-Covered Carotid Stent (CARotid Embolic Prevention using MicroNET-Covered Stent System in Patients with Symptomatic and Asymptomatic Carotid Artery Stenosis): JACC Cardiovasc Interv, 2022; 15; 1889-91
39. Aboyans V, Ricco JB, Bartelink MEL, 2017 ESC guidelines on the diagnosis and treatment of peripheral arterial diseases, in collaboration with the European Society for Vascular Surgery (ESVS): Document covering atherosclerotic disease of extracranial carotid and vertebral, mesenteric, renal, upper and lower extremity arteries. Endorsed by: The European Stroke Organization (ESO)The Task Force for the Diagnosis and Treatment of Peripheral Arterial Diseases of the European Society of Cardiology (ESC) and of the European Society for Vascular Surgery (ESVS): Eur Heart J, 2018; 39; 763-816
40. Han B, Sun X, Tong X, Early blood pressure management for endovascular therapy in acute ischemic stroke: A review of the literature: Interv Neuroradiol, 2020; 26; 785-92
41. Kappelhof M, Marquering HA, Berkhemer OA, Majoie CB, Intra-arterial treatment of patients with acute ischemic stroke and internal carotid artery occlusion: A literature review: J Neurointerv Surg, 2015; 7; 8-15
42. Jonsson M, Gillgren P, Wanhainen A, Peri-procedural risk with urgent carotid artery stenting: A population based SWEDVASC study: Eur J Vasc Endovasc Surg, 2015; 49; 506-12
43. Natera-Villalbaa E, Cruz-Culebrasa A, García-Madronaa S, Mechanical thrombectomy beyond 6 hours in acute ischaemic stroke with large vessel occlusion in the carotid artery territory: Experience at a tertiary hospital: Neurología, 2021 [Online ahead of print]
44. Jankowitz BT, Tonetti DA, Kenmuir C, Urgent treatment for symptomatic carotid stenosis: The Pittsburgh Revascularization and Treatment Emergently After Stroke (PIRATES) Protocol: Neurosurgery, 2020; 87; 811-15
45. Quispe-Orozco D, Limaye K, Zevallos CB, Safety and efficacy of symptomatic carotid artery stenting performed in an emergency setting: Interv Neuroradiol, 2021; 27; 411-18
46. Papanagiotou P, Roth C, Walter S, Carotid artery stenting in acute stroke: J Am Coll Cardiol, 2011; 58; 2363-69
47. Nikas D, Reimers B, Elisabetta M, Percutaneous interventions in patients with acute ischemic stroke related to obstructive atherosclerotic disease or dissection of the extracranial carotid artery: J Endovasc Ther, 2007; 14; 279-88
48. Mazurek A, Malinowski K, Rosenfield K, Clinical outcomes of second-versus first-generation carotid stents: A systematic review and meta-analysis: J Clin Med, 2022; 11; 4819
49. Lanzer P, Cremonesi A, Widimsky P, Perspectives on training requirements for interventional cardiologists to perform endovascular interventions for acute ischaemic stroke: Eurointervention, 2019; 14; 1357-60
50. Holmes DR, Hopkins N, Interventional cardiology and acute stroke care going forward: J Am Coll Cardiol, 2019; 73; 1483-90
51. Musialek P, Nizankowski R, Hopkins LN, Interdisciplinary management of acute ischaemic stroke – current evidence on training requirements for endovascular stroke treatment. Position Paper from the ESC Council on Stroke and the European Association for Percutaneous Cardiovascular Interventions with the support of the European Board of Neurointervention: A step forward: Adv Interv Cardiol, 2021; 17; 245-50
52. Musialek P, Kowalczyk ST, Klecha A, Where and how to treat a man presenting up to 4 hours after cerebral large-vessel occlusion to a thrombectomy-capable major regional hospital: Pol Heart J, 2020; 78; 354-56
53. Bob-Manuel T, Hornung M, Guidera S, Outcomes following endovascular therapy for acute stroke by interventional cardiologists: Catheter Cardiovasc Interv, 2020; 96; 1296-303
54. Hornung M, Bertog SC, Grunwald I, Acute stroke interventions performed by cardiologists: Initial experience in a single center: JACC Cardiovasc Interv, 2019; 17; 1703-10
55. Alvarez CA, Mechanical thrombectomy for ischemic stroke: Interventional cardiology fills the fundamental gap in the system: Pol Heart J, 2020; 78; 804-6
Figures
 Figure 1. Typical cerebral infarct evolution in an acute ischemic stroke of the carotid artery origin in emergency mechanical reperfusion (EMR) eligible patient that did not receive EMR (EMR-untreated). (A) Admission cerebral computed tomography (day 0) was normal; Alberta Stroke Program Early Computed Tomography Score (ASPECTS) 10 in a man presenting with left hemispheric stroke symptoms of increasing severity National Institutes of Health Stroke Scale (NIHSS) – 13 (left) and computed tomography angiography showed no intracranial artery occlusion and good collaterals (Tan 3) (right). (B) Sub-occlusive left internal carotid artery (LICA) stenosis (left); magnetic resonance imaging demonstrated potentially reversible hyperacute left-sided diffusion restriction on diffusion-weighted imaging (DWI, yellow arrows) which are absent on the fluid-attenuated inversion recovery (FLAIR) sequence (right). IV thrombolysis was started, and the patient was observed for thrombolysis effect; there was no referral for EMR. Neurologic status gradually deteriorated. (C) Large cerebral tissue loss (red arrows) seen on control computed tomography 12 hours after 1st scan (left, thrombolysis ineffective, collateral supply exhaustion) and on day 3 (right). Figure was created with the use of Canva (Perth, Australia).
Figure 1. Typical cerebral infarct evolution in an acute ischemic stroke of the carotid artery origin in emergency mechanical reperfusion (EMR) eligible patient that did not receive EMR (EMR-untreated). (A) Admission cerebral computed tomography (day 0) was normal; Alberta Stroke Program Early Computed Tomography Score (ASPECTS) 10 in a man presenting with left hemispheric stroke symptoms of increasing severity National Institutes of Health Stroke Scale (NIHSS) – 13 (left) and computed tomography angiography showed no intracranial artery occlusion and good collaterals (Tan 3) (right). (B) Sub-occlusive left internal carotid artery (LICA) stenosis (left); magnetic resonance imaging demonstrated potentially reversible hyperacute left-sided diffusion restriction on diffusion-weighted imaging (DWI, yellow arrows) which are absent on the fluid-attenuated inversion recovery (FLAIR) sequence (right). IV thrombolysis was started, and the patient was observed for thrombolysis effect; there was no referral for EMR. Neurologic status gradually deteriorated. (C) Large cerebral tissue loss (red arrows) seen on control computed tomography 12 hours after 1st scan (left, thrombolysis ineffective, collateral supply exhaustion) and on day 3 (right). Figure was created with the use of Canva (Perth, Australia). Figure 2. Emergency mechanical reperfusion in acute carotid artery origin ischemic stroke. (A), (left panel) – mild vascular changes (white arrows) seen on admission cerebral computed tomography Alberta Stroke Program Early Computed Tomography Score (ASPECTS) 9 in a patient who was emergency-transferred to a cardioangiology-based Thrombectomy-Capable Stroke Center from external Neurology; (right panel) – diminished flow to the left hemisphere on computed tomography angiography (compare left vs right). (B) Catheter angiography – left internal carotid artery (LICA) near-occlusion with a floating thrombus (Fl Thr, red arrowheads) and next stages of emergency mechanical reperfusion (from left to right, upper and lower panels): transient flow reversal (dotted arrows; enhanced by active aspirations at the procedure critical steps) – proximal cerebral protection device – Mo.Ma (Medtronic, Tolochenaz, Switzerland), external carotid artery balloon (white arrow). Following thrombectomy (carotid-dedicated adjustable-diameter stentriever – TigerTrieverXL (Rapid Medical, Yokneam, Israel), the culprit lesion was sequestrated (white arrowheads), using a micronet-covered stent – C-Guard (InspireMD, Tel Aviv, Israel) with post-dilatation embedding. (C), (left panel) – effective lumen reconstruction resulted in normalized left hemispheric cerebral blood supply symptoms regressed, (right panel) – discharge cerebral computed tomography showed a minor cerebral infarct (red arrows). B/L indicates baseline, (mag) = magnified image. Figure was created with the use of Canva (Perth, Australia).
Figure 2. Emergency mechanical reperfusion in acute carotid artery origin ischemic stroke. (A), (left panel) – mild vascular changes (white arrows) seen on admission cerebral computed tomography Alberta Stroke Program Early Computed Tomography Score (ASPECTS) 9 in a patient who was emergency-transferred to a cardioangiology-based Thrombectomy-Capable Stroke Center from external Neurology; (right panel) – diminished flow to the left hemisphere on computed tomography angiography (compare left vs right). (B) Catheter angiography – left internal carotid artery (LICA) near-occlusion with a floating thrombus (Fl Thr, red arrowheads) and next stages of emergency mechanical reperfusion (from left to right, upper and lower panels): transient flow reversal (dotted arrows; enhanced by active aspirations at the procedure critical steps) – proximal cerebral protection device – Mo.Ma (Medtronic, Tolochenaz, Switzerland), external carotid artery balloon (white arrow). Following thrombectomy (carotid-dedicated adjustable-diameter stentriever – TigerTrieverXL (Rapid Medical, Yokneam, Israel), the culprit lesion was sequestrated (white arrowheads), using a micronet-covered stent – C-Guard (InspireMD, Tel Aviv, Israel) with post-dilatation embedding. (C), (left panel) – effective lumen reconstruction resulted in normalized left hemispheric cerebral blood supply symptoms regressed, (right panel) – discharge cerebral computed tomography showed a minor cerebral infarct (red arrows). B/L indicates baseline, (mag) = magnified image. Figure was created with the use of Canva (Perth, Australia). Figure 3. Evolution of National Institutes of Health Stroke Scale (NIHSS), (A) and functional status (modified Rankin Score – mRS), (B) in patients not treated and treated with EMR (Emergency Mechanical Reperfusion). Individual patient and group data on acute ischemic stroke of carotid artery origin (AIS-CA) clinical severity (NIHSS, A) and patient functional status (mRS, B) are provided at baseline at 90 days. Emergency mechanical reperfusion (EMR) treatment effect is demonstrated by comparison of EMR-untreated (natural history, left) and EMR-treated patients (right). With the particularly large volume of cerebral tissue-at-risk in AIS-CA, EMR profoundly impacts clinical outcomes. Note the striking difference in NIHSS and mRS at 90 days in EMR-treated patients versus those who did not receive mechanical reperfusion. The EMR-untreated patients would have been accepted for treatment in the cardioangiology cathlab-based Thrombectomy-Capable Stroke Center (operator team with experience in proximal-protected carotid artery stenting). The figure was created with the use of Statistica 10 (StatSoft GmBH, Hamburg, Germany).
Figure 3. Evolution of National Institutes of Health Stroke Scale (NIHSS), (A) and functional status (modified Rankin Score – mRS), (B) in patients not treated and treated with EMR (Emergency Mechanical Reperfusion). Individual patient and group data on acute ischemic stroke of carotid artery origin (AIS-CA) clinical severity (NIHSS, A) and patient functional status (mRS, B) are provided at baseline at 90 days. Emergency mechanical reperfusion (EMR) treatment effect is demonstrated by comparison of EMR-untreated (natural history, left) and EMR-treated patients (right). With the particularly large volume of cerebral tissue-at-risk in AIS-CA, EMR profoundly impacts clinical outcomes. Note the striking difference in NIHSS and mRS at 90 days in EMR-treated patients versus those who did not receive mechanical reperfusion. The EMR-untreated patients would have been accepted for treatment in the cardioangiology cathlab-based Thrombectomy-Capable Stroke Center (operator team with experience in proximal-protected carotid artery stenting). The figure was created with the use of Statistica 10 (StatSoft GmBH, Hamburg, Germany). In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387