28 February 2023: Clinical Research
Proposal for a Simple Equation for Limb Muscle Weight Calculation
Yasutaka Kurokawa12ABCDE*, Takayuki Kurokawa3BCDEF, Misato Fujii4B, Masatoshi Tanifuji5B, Takashi Nakajin6B, Tsukasa Sato7B, Ikuko Machida4BDOI: 10.12659/MSM.938606
Med Sci Monit 2023; 29:e938606
Abstract
BACKGROUND: Although body mass index (BMI) is currently being utilized frequently as an indicator of obesity, it provides little information concerning body composition; key components such as fat and muscle cannot be differentiated. It is especially non-sensitive in identifying muscle mass, which can be challenging to examine without the use of radiologic methods. We sought to identify whether biometric values such as upper arm subcutaneous fat thickness/circumference could provide an adequate indicator of muscle mass.
MATERIAL AND METHODS: Patients admitted to our clinic for various causes were retrospectively studied in 95 consecutive cases. Physical parameters including upper arm subcutaneous fat thickness, upper arm circumference, weight, and height were measured. Then, values such as limb muscle weight (LMWDXA) and total fat weight (FWDXA) were obtained from dual-energy X-ray absorptiometry. Pearson’s correlation coefficients were calculated and linear regression analysis was conducted.
RESULTS: Neither upper arm subcutaneous fat thickness nor upper arm circumference was correlated with LMWDXA. FWDXA also showed a correlation with BMI (r=0.823, P<0.001). LMWDXA also significantly correlated with measured body weight (BWm)-BMI (r=0.719, P<0.001).
CONCLUSIONS: From our analytic data we propose an equation for calculating muscle mass, designated the Simple Muscle Weight (SMW): SMW=289.2×(BWm-BMI)+3631. SMW calculation has potential for use as an easy and simple first-line diagnostic tool to identify diminished muscle mass.
Keywords: Body Mass Index, sarcopenia, Humans, Muscles, Upper Extremity, Body Weight
Background
Overall health in older patients is influenced by the degree of decrease in muscle mass [1]. Despite the fact that methods of diagnosis vary, from measuring walking speeds and grip strength to radiological investigations, the majority of these methods have faced difficulties, due to the myriad of tests or parameters required [2,3].
Theoretically, diminished muscle volume can be diagnosed easily by radiological modalities such as computed tomography, magnetic resonance imaging, and/or dual-energy X-ray absorptiometry (DXA). These methods are practically not available in the majority of smaller clinics, and the demand for a simple method is high.
We aimed to assess the statistical correlations between biometric values such as weight, height, upper arm subcutaneous fat thickness, and upper arm circumference to find a combination of parameters that correlates with the values obtained by DXA scans such as total fat weight (FWDXA) and limb muscle weight (LMWDXA).
Material and Methods
LIMB MUSCLE WEIGHT MEASUREMENT:
FWDXA and LMWDXA of all 4 limbs were measured by DXA [5] using a PRODIGY Fuga Advance system (GE Healthcare, Chicago, Illinois, USA) with enCORE: Ver. 17 SP1 software.
STATISTICAL ANALYSIS:
Statistical analysis was performed using JSTAT: Ver. 22.0E [6]. The Pearson’s correlation coefficient and probability values were determined for each of the physical parameters compared with the values obtained using DXA. Probability less than 0.01 was regarded as significant.
Results
PATIENT BACKGROUND AND MEASURED PARAMETERS:
The gender distribution in the 95 cases was 27: 68 for Men: Women, respectively. BMI, upper arm subcutaneous fat thickness, and upper arm circumference were measured (mean±standard deviation) as seen in Table 2.
Age ranged from 47 to 102 years of age (85±8.3). Measured body weight (BWm) ranged from 26.8 to 64.0 kg (43.1±8.17). Height ranged from 134 to 175 cm (153±8.85). BMI ranged from 11.6 to 28.1 kg/m2 (18.4±3.18). Upper arm subcutaneous fat thickness ranged from 0.6 to 18 mm (7.9±4.0). Upper arm circumference ranged from 12 to 27 cm (20±3.0).
The mean values as measured by DXA were as follows: FWDXA was 3563–25 823 g (11 823±5203.9) and LMWDXA was 6951–17 595 g (10 772±2388.2).
STATISTICAL ANALYSES FOR CORRELATION BETWEEN PHYSICAL PARAMETERS AND DXA VALUES:
Upper arm subcutaneous fat thickness was significantly correlated with BMI (r=0.558, P<0.001) and FWDXA (r=0.666, P<0.001). Upper arm circumference was also significantly correlated with BMI (r=0.629, P<0.001) and FWDXA (r=0.743, P<0.001) (Table 3).
In contrast, upper arm subcutaneous fat thickness had no correlation with LMWDXA (r=0.163,
:
Since upper arm subcutaneous fat thickness and circumference did not significantly correlate with LMWDXA but did correlate with FWDXA, we tested the correlation between BMI and FWDXA. As a result, we found that BMI does correlate significantly with FWDXA (r=0.823, P<0.001) (Figure 1). The linear correlation suggests that BMI can mathematically substitute for FWDXA in the studied population. Therefore, we additionally hypothesized that there may be a correlation between LMWDXA and the difference between BWm and BMI (BWm-BMI) (Appendix 1).
Since LMWDXA correlated significantly with (BWm-BMI) (r=0.719, P<0.001), in our subjects, we can regard (BWm-BMI) as muscle mass, which might help to predict sarcopenia (Figure 2).
In conclusion, we propose the Simple Muscle Weight (SMW) equation [SMW=289.2×(BWm-BMI)+3631] as a possible first-step indicator of muscle mass.
Discussion
CURRENT STRATEGIES FOR MEASURING MUSCLE MASS: RADIOLOGICAL METHODS:
While DXA remains unchallenged as the criterion standard for measuring muscle mass, emerging modalities have been identified to be useful in the identification of sarcopenia, but with certain difficulties [12]. Although ethnic and disease-specific modifications apply, the accuracy of DXA has been backed by numerous research studies in the literature [13].
The use of radiological measurement to obtain accurate values requires special equipment and qualifications, compared with the cheaper and lighter devices used for impedance adipometry [14] and ultrasonic measurement of muscle thickness [15].
CURRENT STRATEGIES FOR MEASURING MUSCLE MASS: FUNCTIONAL TESTING:
A common way to start the initial investigation for sarcopenia is measurement of the patient’s calf circumference, grip strength, and ambulatory abilities, which can be inaccurate and inconsistent in patients with physical disabilities. Functional tests such as the Sit to Stand test, 30-second Chair Stand Test, muscle strength, physical performance, and physical functioning have also been examined to determine the loss of muscle mass in the elderly [16]. These functional tests give quantitative and descriptive analyses of the patient population, with numerous modifications requiring adjustment for different patient populations [17].
BIOCHEMICAL MARKERS:
The lack of availability of a way to easily measure muscle mass has resulted in increased use of non-radiological methods. Fayh et al [18] and Evans et al [19] have proposed methods using biochemical markers. Other markers, such as blood albumin levels and molecular markers, have been speculated to reflect the existence of diminished muscle mass. Although these biomarkers have the potential to aid in the diagnosis of decreased muscle mass, difficulties remain, including coverage of the costs, difficulty/lack of accessibility, and patient cooperation.
UNDERSTANDING THE CORRELATION BETWEEN BMI AND FAT WEIGHT:
While there is a substantial body of literature examining the relationship between BMI and fat weight, the estimation of body composition without the use of radiological or biochemical methods seems to be challenging.
Our study showed significant correlations between FWDXA and upper arm subcutaneous fat thickness and/or circumference. More importantly, BMI was significantly correlated with FWDXA. These findings are in accord with the international literature, especially when measured by impedance adipometry or DXA [20].
To better understand the dynamic relationship between FWDXA and BMI, we re-examined the correlations between these parameters, and found that LMWDXA is also significantly correlated with (BWm-BMI). From this correlation and the linear regression, the equation SMW=289.2×(BWm-BMI)+3631 was devised. This equation serves as a way, by applying it as a relative index, to identify underlying muscle mass. In Figure 3 we see a large portion of patients’ LMWDXA indicating low muscle mass. Even when BMI offered no information on sarcopenia in these patients, the results of the SMW calculation showed that most patients were sarcopenic. Indeed, the majority of the patients in our study were admitted with limb fractures caused by falls, and pneumonia – conditions which are highly associated with sarcopenia [21,22].
On the other hand, patients with low BMI did not necessarily show low LMWDXA, as can be seen in Figure 4. High BMI did not necessarily mean that the patients did not have diminished muscle mass; some patients with relatively high BMI even presented with low LMWDXA. This is important, since obesity coexisting with sarcopenia, known as sarcopenic obesity, can lead to especially high morbidity in the elderly [23].
LIMITATIONS AND FURTHER PROSPECTS:
Although our study suggested that muscle weight could be roughly calculated without radiographic, biochemical, or functional data, there were a few factors that it did not take into consideration. While SMW appears to be able to be calculated on the basis of body weight and BMI for a homogenous population, we have yet to understand modifying factors, as well as the accuracy of SMW in different ethnicities. Our subject population was mostly elderly patients, but it would be interesting to observe whether the same principles apply in younger, healthier patients as well. Male and female differences should also be considered, and modifying factors must be addressed and identified.
Clinical pictures based on multiple parameters are of the utmost importance, and assessment for risk of falls, debilitation, and ambulatory function should also be considered if SMW is to be applied in a realistic clinical situation, where indication of therapy would be determined on its basis.
Conclusions
From our analytic data we propose the possibility of calculating muscle mass as a variable called the Simple Muscle Weight. The equation is: SMW=289.2×(BWm-BMI)+3631. SMW calculation has the potential to be used as an easy and simple first-line diagnostic tool to identify diminished muscle mass.
Figures
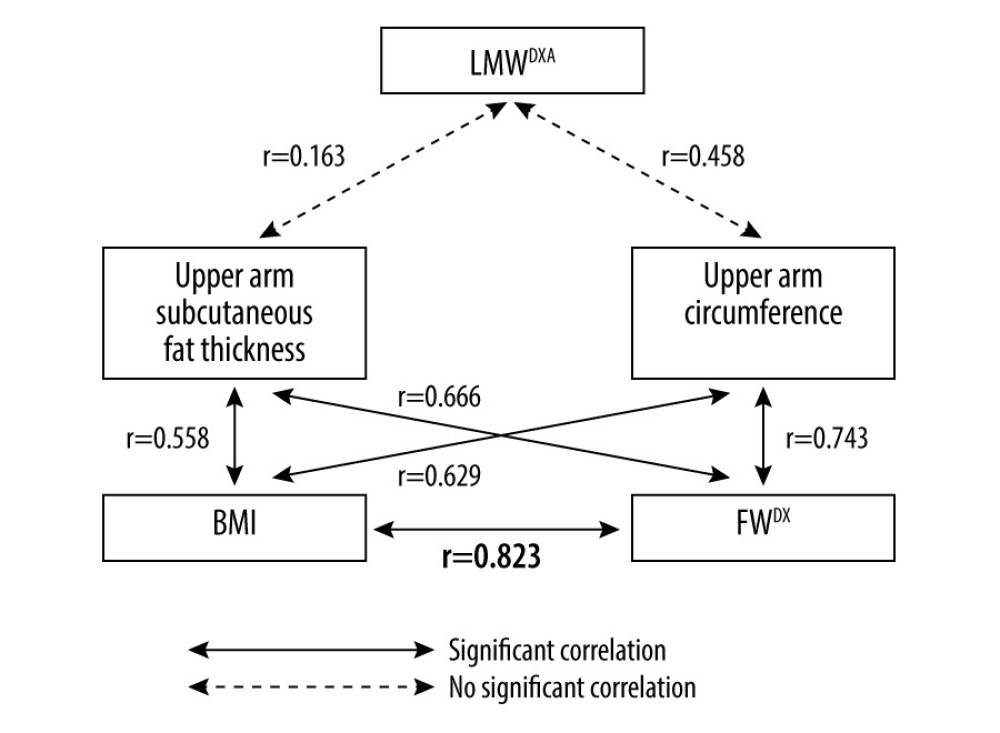 Figure 1. Statistical analysis for each correlation. Total fat weight (FWDXA) obtained by DXA strongly correlates with BMI. BMI – body mass index; DXA – dual-energy X-ray absorptiometry; FWDXA – total fat weight obtained by DXA; LMWDXA – limb muscle weight obtained by DXA; r – correlation coefficient.
Figure 1. Statistical analysis for each correlation. Total fat weight (FWDXA) obtained by DXA strongly correlates with BMI. BMI – body mass index; DXA – dual-energy X-ray absorptiometry; FWDXA – total fat weight obtained by DXA; LMWDXA – limb muscle weight obtained by DXA; r – correlation coefficient. 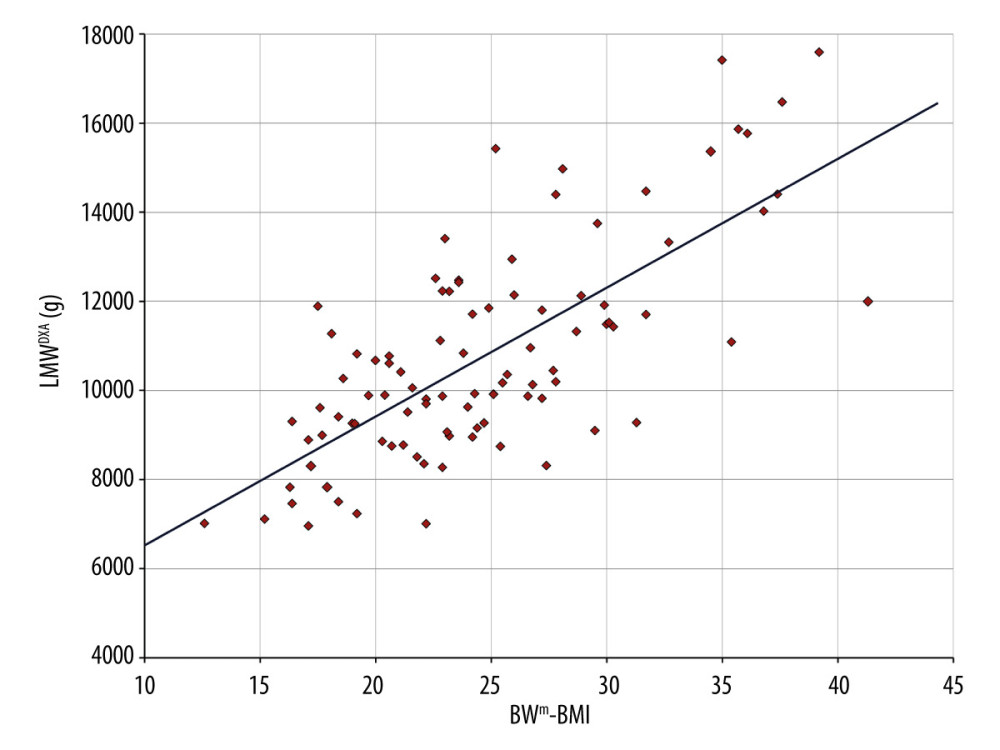 Figure 2. Scatter diagram and regression line of limb muscle weight (LMWDXA) and the difference between BWm and BMI. A strong linear correlation between LMWDXA and (BWm-BMI) can be seen (r=0.719, P<0.001, y=289.2×(BWm-BMI) +3631). This enables us to approximate muscle mass simply by using BWm and height. BMI – body mass index; BWm – measured body weight; DXA – dual-energy X-ray absorptiometry; LMWDXA – limb muscle weight obtained by DXA.
Figure 2. Scatter diagram and regression line of limb muscle weight (LMWDXA) and the difference between BWm and BMI. A strong linear correlation between LMWDXA and (BWm-BMI) can be seen (r=0.719, P<0.001, y=289.2×(BWm-BMI) +3631). This enables us to approximate muscle mass simply by using BWm and height. BMI – body mass index; BWm – measured body weight; DXA – dual-energy X-ray absorptiometry; LMWDXA – limb muscle weight obtained by DXA. 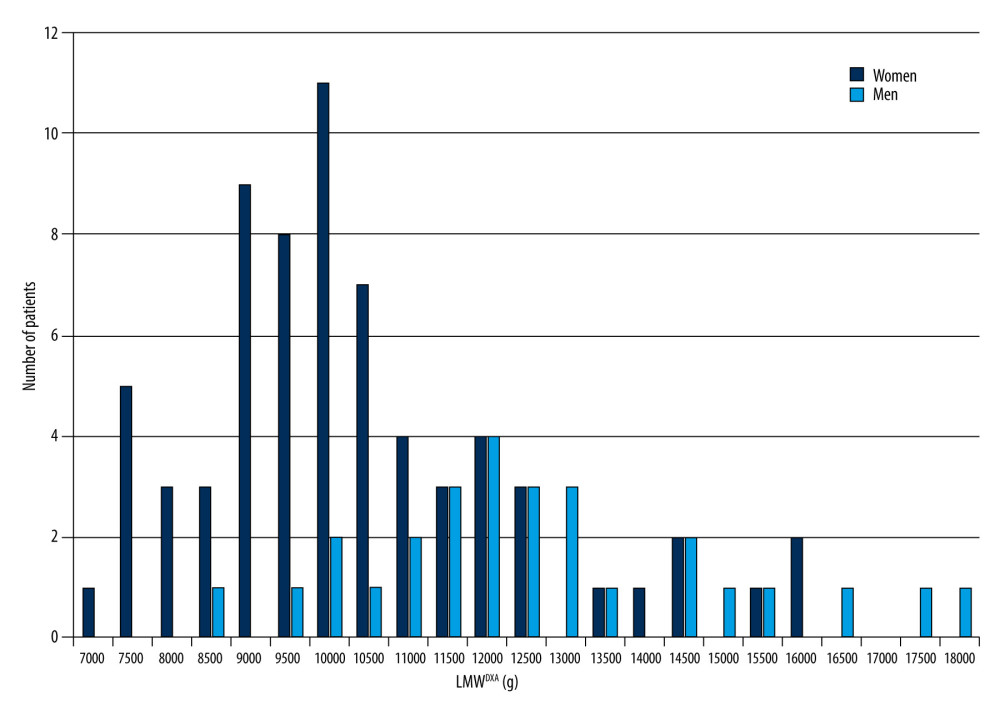 Figure 3. Distribution of limb muscle weight (LMW) in 95 cases. DXA – dual-energy X-ray absorptiometry; LMWDXA – limb muscle weight obtained by DXA.
Figure 3. Distribution of limb muscle weight (LMW) in 95 cases. DXA – dual-energy X-ray absorptiometry; LMWDXA – limb muscle weight obtained by DXA. 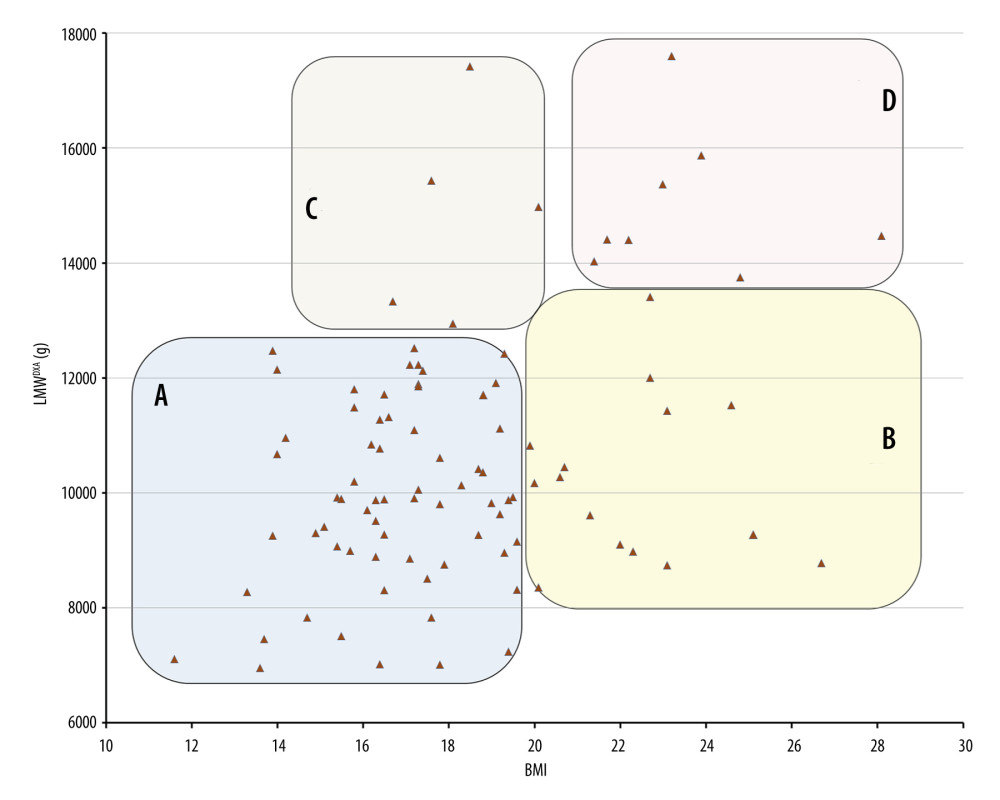 Figure 4. Distribution and relationship of limb muscle weight (LMWDXA) versus BMI. A: Low BMI and low LMWDXA, representing possible sarcopenia. B: High BMI and low LMWDXA, representing possible sarcopenic obesity. C: Low BMI and high LMWDXA, representing muscular body type seen in athletes. D: High BMI and high LMWDXA, representing a large body type. BMI – body mass index; DXA – dual-energy X-ray absorptiometry; LMWDXA – limb muscle weight obtained by DXA.
Figure 4. Distribution and relationship of limb muscle weight (LMWDXA) versus BMI. A: Low BMI and low LMWDXA, representing possible sarcopenia. B: High BMI and low LMWDXA, representing possible sarcopenic obesity. C: Low BMI and high LMWDXA, representing muscular body type seen in athletes. D: High BMI and high LMWDXA, representing a large body type. BMI – body mass index; DXA – dual-energy X-ray absorptiometry; LMWDXA – limb muscle weight obtained by DXA. References
1. Rosenberg IH, Summary comments: Am J Clin Nutr, 1989; 50; 1231-33
2. Chen LK, Woo J, Assantachai P, Asian Working Group for Sarcopenia: 2019 Consensus update on sarcopenia diagnosis and treatment: J Am Med Dir Assoc, 2020; 21; 300-7e2
3. Cruz-Jentoft AJ, Bahat G, Bauer J, Sarcopenia: Revised European consensus on definition and diagnosis: Age Ageing, 2019; 48; 16-31
4. National Institute of Health Research: Procedure for Measuring Adult Circumferences, 2015 Available from https://www.uhs.nhs.uk/Media/Southampton-Clinical-Research/Procedures/BRCProcedures/Procedure-for-adult-circumference-measurements.pdf
5. General Electric Company: Lunar Technology Advantages, 2017 Available fromhttps://www.gehealthcare.com/-/jssmedia/e602102206184a3bac8fc0198f980c73.pdf
6. Sato M: Statistical software development case studies and programming language considerations, 2018 Available from: . Toukeijstat.web.fc2.com
7. Quételet A: Sur l’homme et le développement de ses facultés, ou Essai de physique sociale, 1835(55), Paris, Bachelier, Imprimeur-Libraire, quai de augustins [in French]
8. Keys A, Fidanza F, Karvonen MJ, Indices of relative weight and obesity: Int J Epidemiol, 2014; 43; 655-65
9. Nuttall FQ, Body mass index. Obesity, BMI, and health: A critical review: Nutr Today, 2015; 50; 117-28
10. Caleyachetty R, Barber TM, Mohammed NI, Ethnicity-specific BMI cutoffs for obesity based on type 2 diabetes risk in England: A population-based cohort study: Lancet Diabetes Endocrinol, 2021; 97; 419-26
11. Karasu SR, Adolphe Quetelet and the Evolution of Body Mass Index (BMI): Psychology Today, 2016 Available from:https://www.psychologytoday.com/intl/blog/the-gravity-weight/201603/adolphe-quetelet-and-the-evolution-body-mass-index-bmi
12. Albano D, Messina C, Vitale J, Imaging of sarcopenia: Old evidence and new insights: Eur Radiol, 2020; 30; 2199-208
13. Sergi G, Trevisan C, Veronese N, Imaging of sarcopenia: Eur J Radiol, 2016; 85; 1519-24
14. Shafer KJ, Siders WA, Johnson LK, Validity of segmental multiple-frequency bioelectrical impedance analysis to estimate body composition of adults across a range of body mass indexes: Nutrition, 2009; 251; 25-32
15. Madden KM, Feldman B, Arishenkoff S, A rapid point-of-care ultrasound marker for muscle mass and muscle strength in older adults: Age Ageing, 2021; 50; 505-10
16. Björkman MP, Pitkala KH, Jyväkorpi S, Bioimpedance analysis and physical functioning as mortality indicators among older sarcopenic people: Exp Gerontol, 2019; 122; 42-46
17. Kara M, Kaymak B, Frontera W, Diagnosing sarcopenia: Functional perspectives and a new algorithm from the ISarcoPRM: J Rehabil Med, 2021; 53; jrm00209
18. Fayh APT, de Sousa IM, Gonzalez MC, New insights on how and where to measure muscle mass: Curr Opin Support Palliat Care, 2020; 14; 316-23
19. Evans WJ, Hellerstein M, Orwoll E, D3-Creatine dilution and the importance of accuracy in the assessment of skeletal muscle mass: J Cachexia Sarcopenia Muscle, 2019; 10; 14-21
20. Achamrah N, Colange G, Delay J, Comparison of body composition assessment by DXA and BIA according to the body mass index: A retrospective study on 3655 measures: PLoS One, 2018; 13; e0200465
21. Pacifico J, Geerlings MAJ, Reijnierse EM, Prevalence of sarcopenia as a comorbid disease: A systematic review and meta-analysis: Exp Gerontol, 2020; 131; 110801
22. Yeung SSY, Reijnierse EM, Vivien K, Sarcopenia and its association with falls and fractures in older adults: A systematic review and meta-analysis: J Cachexia Sarcopenia Muscle, 2019; 10; 485-500
23. Öztürk ZA, Türkbeyler İH, Abiyev A, Health-related quality of life and fall risk associated with age-related body composition changes; Sarcopenia, obesity and sarcopenic obesity: Intern Med J, 2018; 48; 973-81
Figures
 Figure 1. Statistical analysis for each correlation. Total fat weight (FWDXA) obtained by DXA strongly correlates with BMI. BMI – body mass index; DXA – dual-energy X-ray absorptiometry; FWDXA – total fat weight obtained by DXA; LMWDXA – limb muscle weight obtained by DXA; r – correlation coefficient.
Figure 1. Statistical analysis for each correlation. Total fat weight (FWDXA) obtained by DXA strongly correlates with BMI. BMI – body mass index; DXA – dual-energy X-ray absorptiometry; FWDXA – total fat weight obtained by DXA; LMWDXA – limb muscle weight obtained by DXA; r – correlation coefficient. Figure 2. Scatter diagram and regression line of limb muscle weight (LMWDXA) and the difference between BWm and BMI. A strong linear correlation between LMWDXA and (BWm-BMI) can be seen (r=0.719, P<0.001, y=289.2×(BWm-BMI) +3631). This enables us to approximate muscle mass simply by using BWm and height. BMI – body mass index; BWm – measured body weight; DXA – dual-energy X-ray absorptiometry; LMWDXA – limb muscle weight obtained by DXA.
Figure 2. Scatter diagram and regression line of limb muscle weight (LMWDXA) and the difference between BWm and BMI. A strong linear correlation between LMWDXA and (BWm-BMI) can be seen (r=0.719, P<0.001, y=289.2×(BWm-BMI) +3631). This enables us to approximate muscle mass simply by using BWm and height. BMI – body mass index; BWm – measured body weight; DXA – dual-energy X-ray absorptiometry; LMWDXA – limb muscle weight obtained by DXA. Figure 3. Distribution of limb muscle weight (LMW) in 95 cases. DXA – dual-energy X-ray absorptiometry; LMWDXA – limb muscle weight obtained by DXA.
Figure 3. Distribution of limb muscle weight (LMW) in 95 cases. DXA – dual-energy X-ray absorptiometry; LMWDXA – limb muscle weight obtained by DXA. Figure 4. Distribution and relationship of limb muscle weight (LMWDXA) versus BMI. A: Low BMI and low LMWDXA, representing possible sarcopenia. B: High BMI and low LMWDXA, representing possible sarcopenic obesity. C: Low BMI and high LMWDXA, representing muscular body type seen in athletes. D: High BMI and high LMWDXA, representing a large body type. BMI – body mass index; DXA – dual-energy X-ray absorptiometry; LMWDXA – limb muscle weight obtained by DXA.
Figure 4. Distribution and relationship of limb muscle weight (LMWDXA) versus BMI. A: Low BMI and low LMWDXA, representing possible sarcopenia. B: High BMI and low LMWDXA, representing possible sarcopenic obesity. C: Low BMI and high LMWDXA, representing muscular body type seen in athletes. D: High BMI and high LMWDXA, representing a large body type. BMI – body mass index; DXA – dual-energy X-ray absorptiometry; LMWDXA – limb muscle weight obtained by DXA. Tables
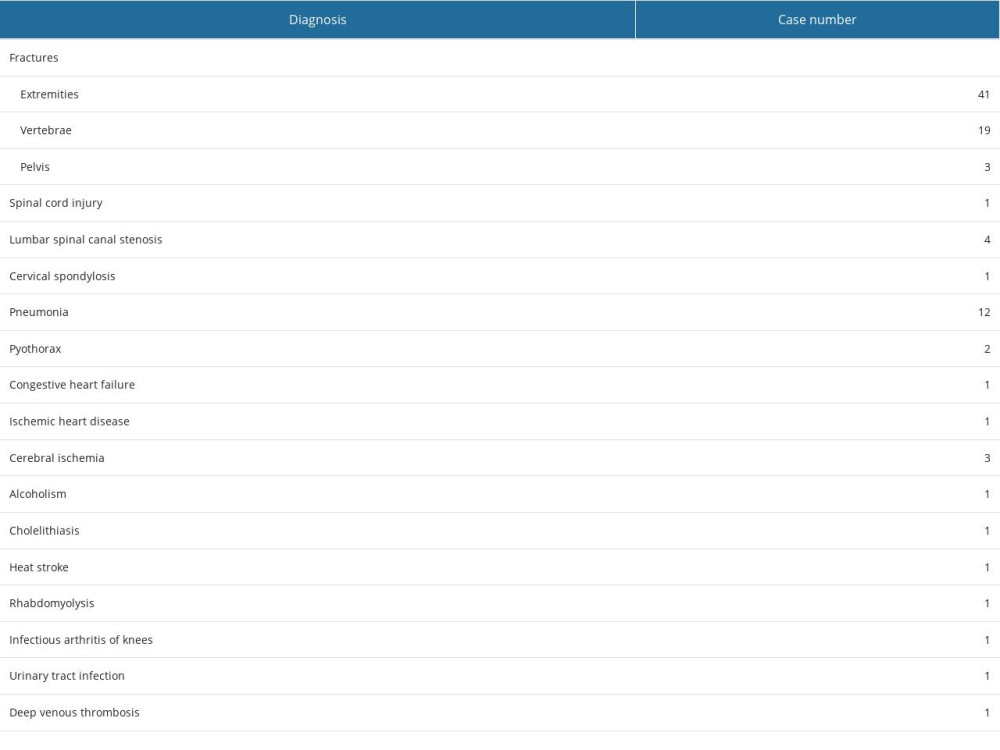 Table 1. Primary reasons for hospitalization.
Table 1. Primary reasons for hospitalization.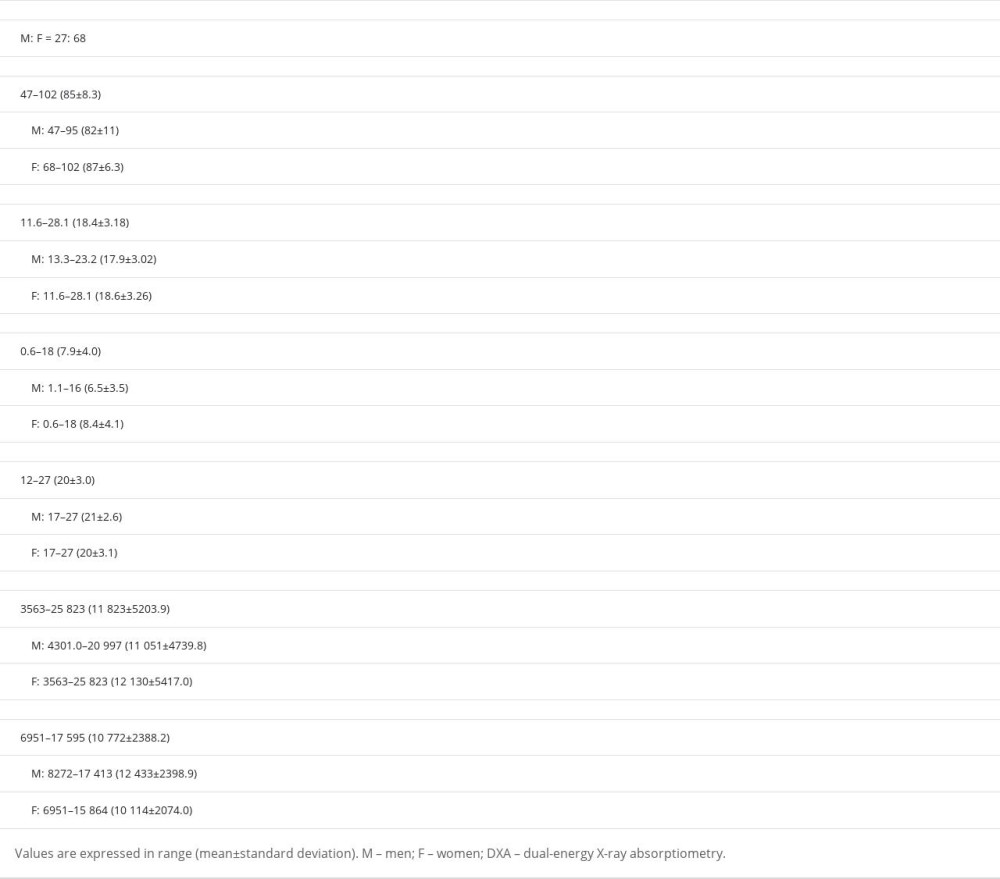 Table 2. Measured physical parameters and radiological values for the patients.
Table 2. Measured physical parameters and radiological values for the patients.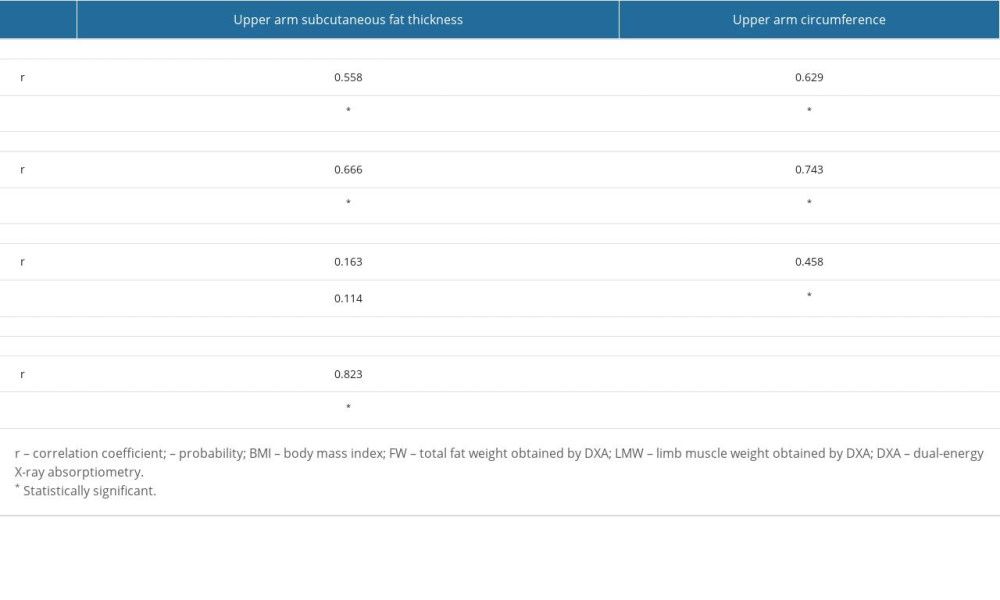 Table 3. Statistical analysis for correlation of measured physical and radiological factors.
Table 3. Statistical analysis for correlation of measured physical and radiological factors. Table 1. Primary reasons for hospitalization.
Table 1. Primary reasons for hospitalization. Table 2. Measured physical parameters and radiological values for the patients.
Table 2. Measured physical parameters and radiological values for the patients. Table 3. Statistical analysis for correlation of measured physical and radiological factors.
Table 3. Statistical analysis for correlation of measured physical and radiological factors. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








