28 November 2022: Clinical Research
Ultrasonographic Carotid Artery Flow Measurements as Predictors of Spinal Anesthesia-Induced Hypotension in Elderly Patients: A Prospective Observational Study
Hyae Jin Kim1ABEF, Ah-Reum Cho12ACDE, Hanbit Lee1ABEF*, Haekyu Kim12DEF, Jae-Young Kwon12DEF, Hyeon-Jeong Lee12DEF, Christine Kang3BE, Soeun Jeon4BDEF, Nara Kim1BEDOI: 10.12659/MSM.938714
Med Sci Monit 2022; 28:e938714
Abstract
BACKGROUND: In elderly patients, spinal anesthesia-induced hypotension (SAH) can be frequently caused by reduced preload and stiff ventricles. The primary purpose of this study was to investigate the ability of ultrasonographic carotid artery flow measurements during the passive leg raise (PLR) test to predict SAH in elderly patients. The correlation between preoperative transthoracic echocardiography (TTE) measurements and SAH was also investigated.
MATERIAL AND METHODS: The patients aged over 65 years scheduled for elective surgery under spinal anesthesia were recruited. Preoperative TTE was performed in all patients. Corrected carotid flow time and carotid blood flow were measured in the supine, semirecumbent, and PLR positions. Ultrasonographic carotid artery flow and preoperative TTE measurements were compared between patients who developed SAH and those who did not. Receiver operating characteristic (ROC) curve analysis and logistic regression analysis were used to test the association with SAH.
RESULTS: SAH occurred in 17 of 50 patients. Carotid blood flow in the semirecumbent position and preoperative mitral inflow E velocity could predict SAH, showing an area under the ROC curve of 0.754 (95% CI, 0.612-0.865) and 0.775 (95% CI, 0.634-0.881), respectively. However, according to the multivariate analysis, the independent risk factor for SAH was mitral inflow E velocity (OR 0.918, 95% CI 0.858-0.982, P=0.013).
CONCLUSIONS: In elderly patients, ultrasonographic carotid artery flow measurements failed to predict the occurrence of SAH. Only preoperative mitral inflow E velocity of TTE was selected as an independent risk factor for SAH.
Keywords: Anesthesia, Spinal, Echocardiography, Geriatrics, Hypotension, Ultrasonography, Aged, Humans, Hypotension, Controlled, Carotid Arteries, Carotid Artery, Common, Prospective Studies
Background
The main cause of spinal anesthesia-induced hypotension (SAH) is sympathetic blockade, which results in vasodilation with relative hypovolemia and reduced cardiac output. Many elderly patients arrive at the operating room in a relatively hypovolemic state due to preoperative fasting, reduced thirst mechanisms, and diminished renal capacity to conserve water and salt [1]. Moreover, venodilation after the intrathecal injection decreases preload through blood redistribution to the splanchnic bed and lower limbs [2]. Considering that the elderly heart is very sensitive to preload to maintain cardiac output because of stiff ventricles and blunted reflex tachycardia [3], it is easy to understand why elderly patients undergoing spinal anesthesia are at increased risk of hypotension, with reported incidences of 25% to 80% [4].
Recently, transthoracic echocardiography (TTE) findings before and after spinal anesthesia were compared between patients older and younger than 70 years of age [5]. The study supports that SAH in elderly patients is mainly caused by reduced preload due to vasodilation with underlying cardiac dysfunction that further reduces cardiac output. Many elderly patients develop SAH without any existing cardiac symptoms. In particular, left ventricular (LV) diastolic dysfunction is asymptomatic at rest, and symptoms appears under stress conditions [6]. Cardiac output would be reduced when LV diastolic function is impaired, as with LV systolic function. Cardiac output decreases early in the diastolic dysfunction before left atrial (LA) pressure is increased by the compensatory mechanism, which represents grade I diastolic dysfunction [6].
Measuring cardiac output with arterial waveform or echocardiography is invasive and requires extensive training. Ultrasonographic carotid artery flow measurements, such as corrected carotid flow time (cCFT) and carotid blood flow (CBF), are noninvasive, objective, and easy to perform at the bedside. Several studies have shown that they are correlated with invasive cardiac output measurements [7,8]. Furthermore, cCFT and CBF have been studied to detect changes in cardiac output during a passive leg raise (PLR) test and were revealed as good parameters in fluid responsiveness during the PLR test [9–11].
The hypothesis of this study was that elderly patients with reduced preload and stiff ventricles are at risk of SAH. The primary purpose of this study was to investigate the ability of ultrasonographic carotid artery flow measurements during the PLR test, which reflect cardiac output, to predict SAH in elderly patients. The secondary purpose was to find the contributors of SAH, including preoperative TTE measurements.
Material and Methods
ETHICS STATEMENT:
This study was approved by the Institutional Review Board of Pusan National University Hospital, Republic of Korea (Busan, Republic of Korea, H-1711-020-061) and registered prior to patient enrollment at the WHO-ICTRP, Clinical Research Information Service (ID: KCT0002676; principal investigator: Ah-Reum Cho; date of registration: February 2, 2018). The prospective observational study was conducted from February 2018 to January 2019. This manuscript adheres to the applicable STROBE guidelines. Patients received oral and written information about the protocol and provided written consent the day before the surgery.
ENROLLMENT:
Patients over 65 years of age with American Society of Anesthesiologists physical status I–III who were scheduled for elective surgery under spinal anesthesia were invited to participate. Exclusion criteria were morbid obesity (body mass index >35 kg/m2); carotid disease; symptomatic cardiac disease (arrhythmia, valvular disease, ischemic heart disease); lack of preoperative TTE; uncontrolled hypertension; contraindications to spinal anesthesia (bleeding tendency, allergy to local anesthetics, sepsis, infection of the needle insertion site); inability to tolerate a PLR (unable to lie supine or had pain with PLR); inability to understand the study information sheet; and refusal. Patients were excluded if spinal anesthesia failed or the angle of insonation was >60° after recruitment.
ULTRASONOGRAPHIC CAROTID ARTERY FLOW MEASUREMENTS:
Real-time measurement of ultrasound carotid images was taken using a 12L-RS, 5–13 MHz linear probe on a bedside ultrasound system (GE Vivid I; Trisonics, Harrisburg, PA, USA) by a trained anesthesiologist. In all cases, an ultrasound evaluation of carotid artery flow was performed by assessing the right common carotid artery, which was scanned in the transverse and longitudinal planes. Pulsed-wave Doppler tracings were then obtained by placement of a 0.5-mm sample gate through the center of vessel within 2 to 3 cm proximal to the carotid bulb in the longitudinal plane (Figure 1), in accordance with standard guidelines [12]. The angle correction cursor was placed parallel to the direction of blood flow.
The cCFT was calculated as follows: cCFT=systole time/√cycle time [13]. Systole time was measured from the start of systolic upstroke to the start of the dicrotic notch (Figure 1, a), while cycle time was measured from the beginning of the current beat to the beginning of the adjacent beat (Figure 1, b). CBF was calculated as follows: blood flow=π×(carotid diameter)2/4×velocity time integral (VTI)×heart rate (HR). VTI of the Doppler signal was measured using automatic tracings (Figure 1, c). Intimal-to-intimal carotid diameter was measured at the level of the sample gate (Figure 1, d). All carotid measurements (systole time, cycle time, VTI, diameter) were measured on 3 waveforms by a single investigator to calculate mean cCFT and CBF. To establish interrater reliability of the measurements, all measurements on the obtained images were repeated by a second independent investigator. Results from the second investigator were used to provide interrater reliability estimates.
STUDY PROTOCOL:
All patients received maintenance fluid therapy during the fasting period. Upon operating room arrival, patients were monitored in the supine position with electrocardiography, noninvasive blood pressure (BP), and pulse oximetry (SpO2). After 5 min of stabilization, baseline BP, HR, and SpO2 were recorded. Hypotension was defined as a systolic BP <90 mm Hg and a decrease in baseline systolic BP >20%. Baseline ultrasound carotid images were also obtained. Prior to PLR, patients were placed in the semirecumbent position with the head of bed at 45° for 3 min. A PLR test was then performed with the patient’s leg passively raised at 45° and trunk horizontal for 90 s. BP, HR, SpO2, and ultrasound carotid artery images were obtained in the semirecumbent and PLR positions. Then, patients were returned to the supine position.
Intrathecal injection was administered to the patient in the right lateral decubitus position, lumbar puncture preferably midline at L3–4 with a Quincke 25-gauge needle. A standard dose of hyperbaric bupivacaine 0.5% (12–15 mg depending on surgery and patient condition) was injected into the intrathecal space, with the needle orifice oriented cranially. Immediately after the intrathecal injection, the patients were returned to the supine position. Balanced crystalloid solution (5 mL/kg) was administered during the procedure. After the intrathecal injection, mean BP, HR, and SpO2 were recorded simultaneously at 1-min intervals for the first 15 min. One minute after the intrathecal injection, ultrasound carotid artery images were obtained. The level of sensory block was checked at 5 and 15 min after the intrathecal injection with a cold swab. When hypotension occurred, the patient was initially treated with 200 mL of balanced crystalloid solution. If the hypotension persisted for 3 min, 5 mg of ephedrine was administered intravenously and repeated every 3 min depending on the subsequent improvement. Bradycardia was defined as HR <50 beats/min and treated with 0.5 mg of intravenous atropine.
STATISTICAL ANALYSIS:
The sample size was calculated based on the areas under the receiver operating characteristic (ROC) curves. After reviewing the parameters to predict SAH in previous studies, the expected area under the ROC curve was >0.75. A power calculation determined a sample size of 50 patients with an α risk of 5% and a β risk of 10%. Considering a dropout rate of 10%, 55 patients were required.
All continuous variables were tested for normal distribution using a Q-Q plot and Kolmogorov-Smirnov test. Data are expressed as mean±standard deviation, median (interquartile range), or number (%), as appropriate. The
The accuracy of the ultrasonographic carotid artery flow measurements for predicting SAH was analyzed using ROC curves with a 95% CI. The optimal cut-off was selected to maximize the Youden index. If some preoperative TTE measurements were significantly different between patients who did and did not develop hypotension, their ability to predict SAH was also tested using the ROC curves.
Association of the occurrence of SAH was analyzed using univariate and multivariate logistic regression analyses. We selected those factors significantly associated with hypotension in the univariate analysis (
Results
Of the 76 eligible patients, 55 patients were recruited. Five patients were excluded after recruitment due to atheroma in the carotid artery (n=2), angle of insonation >60°, and failed spinal anesthesia. Finally, 50 patients were included in the analysis (Figure 2). The patients’ baseline characteristics are shown in Table 1. Hypotension occurred in 17 patients (34.0%) and was first detected at 7.2±3.7 min after the intrathecal injection. Preoperative TTE demonstrated significantly lower mitral inflow E velocity and medial E velocity in patients who developed hypotension (Table 2). The ICC and 95% CI of the ultrasound carotid artery flow measurements for single and mean measures are presented in Table 3. All ICC for the measurements were greater than 0.9, indicating good interrater reproducibility.
Ultrasonographic carotid artery flow measurements of patients with and without SAH are shown in Table 4. The cCFT in different positions were similar between patients who did and patients who did not develop hypotension. CBF in the semirecumbent position was significantly lower in patients who developed hypotension compared with those who did not (
For the primary outcome, the area under the ROC curve of the ultrasonographic carotid artery flow measurements for predicting SAH are shown in Table 5. The area under the ROC curve for CBF in the semirecumbent position was 0.754 (
As seen in Table 6, we identified risk factors for SAH using logistic regression analysis. According to the multivariate analysis, the independent risk factors for SAH were mitral inflow E velocity (OR 0.918, 95% CI 0.858–0.982,
Discussion
In the present study of elderly patients, lower CBF values in the semirecumbent position could predict SAH. However, it failed to be selected as an independent risk factor for the development of SAH. Also, neither CBF nor cCFT changes during the PLR test differed between patients who did and patients who did not develop SAH. For the secondary outcome, mitral inflow E velocity showed fair ability to predict SAH and was selected as an independent risk factor.
Most patients in this study had impaired ventricular relaxation with normal LA pressure (grade I diastolic dysfunction), which can be asymptomatic at rest. In the early phase of diastolic dysfunction, mitral inflow E velocity decreases because patients with diastolic dysfunction have a steeper and upward end-diastolic pressure – volume relationship, in which small changes in end-diastolic volume would lead to a reduced stroke volume [14]. Therefore, the higher the LV end-diastolic pressure, the lower the mitral inflow E velocity before the LA pressure increases due to a reduced transmitral pressure gradient. Although CBF in the semirecumbent position was not selected as an independent risk factor of SAH, the significant difference between patients who did and those who did not develop SAH may have clinical implications. The head-up position reduces preload and cardiac output, which would be further aggravated in patients with stiff ventricles with a higher LV end-diastolic pressure, which represent a lower inflow mitral E velocity on echocardiography, and these patients are more likely to develop SAH [15,16].
A recent study showed an interesting finding in elderly patients with SAH. Cardiac output decreased from baseline before and during intrathecal injection after the patients changed to the lateral decubitus or seated position and progressively decreased after intrathecal injection [17]. On the other hand, cardiac output increased initially before the intrathecal injection in patients who did not develop SAH. The authors hypothesized that diastolic dysfunction could be the reason for the absence of this biphasic change in cardiac output in patients who develop SAH. Their hypothesis could partially explain the results of the present study. Although the ability of preoperative TTE measurements to predict SAH was a secondary outcome, and while its statistical power was small, mitral inflow E velocity showed good ability to predict SAH. Moreover, mitral inflow E velocity was significantly correlated with ephedrine dose, which would represent the severity of SAH.
Another independent risk factor for SAH was the level of sensory block of ≥T6 at 5 min after intrathecal injection. A previous study showed that ascension range and rate are important predictor for SAH [18]. In our study, the level of sensory block at 15 min after intrathecal injection was not significantly different between patients who did and did not develop hypotension. Although extensive and rapidly raised sensory block is an apparent risk factor, there is no definite strategies to prevent higher and rapid sensory block [19]. Also, when it is noticed, there is not much time to apply strategies to prevent SAH.
The cCFT value failed to predict the occurrence of SAH during the PLR test. One study demonstrated that invasive measurements of cardiac output were significantly correlated with CBF but weakly correlated with cCFT in patients undergoing cardiac catheterization [8]. This is because the CBF calculation includes more clinical parameters, such as carotid diameter, VTI, and HR, which may be less sensitive to the changes of a single parameter. PLR is believed to result in the autotransfusion of approximately 300 mL of blood from the legs to the central blood volume [20]. This reversible and transient autotransfusion increases cardiac output without excess fluid administration. Fluid responsiveness can be predicted by changes in the index of cardiac output during the PLR test. The reason the PLR test was not useful in the present study might have been that patients were not in a hypovolemic state enough to respond to the PLR test. Previous studies that showed positive fluid responsiveness with CBF and cCFT during the PLR test included critically ill patients and patients with dialysis or acute blood loss [11,21,22]. On the other hand, patients in this study were relatively healthy and received maintenance fluid therapy during the fasting period and fluid preloading during intrathecal injection. This is in accordance with the relatively lower incidence of SAH in elderly patients in the present study than in those previously reported [23,24]. Another assumption is that patients with diastolic dysfunction may have a reduced response to the PLR test owing to the steep end-diastolic pressure–volume relationship that leads to reduced stroke volume.
There are several limitations to this study. First, reduced cardiac output and mitral inflow E velocity in patients with LV diastolic dysfunction only occurs in grade I diastolic dysfunction. When LV diastolic dysfunction becomes more severe, mitral inflow E velocity increases as LA pressure rises to compensate. Therefore, our results could apply only to the patients with grade I diastolic dysfunction. In our study, SAH occurred in only 17 patients. Moreover, this study was not designed to investigate the mechanism of the relationship among CBF, LV diastolic dysfunction, and SAH. Therefore, further large studies are needed to confirm the causal relationship between LV diastolic dysfunction and SAH.
Conclusions
According to this study, ultrasonographic carotid artery flow measurements failed to predict the occurrence of SAH. However, only mitral inflow E velocity and the level of sensory block at 5 min after intrathecal injection were selected as independent risk factors for SAH. Our results suggest that preoperative TTE may identify patients with a high risk of SAH. Larger studies are needed to identify the relationship between LV diastolic dysfunction and SAH.
Figures
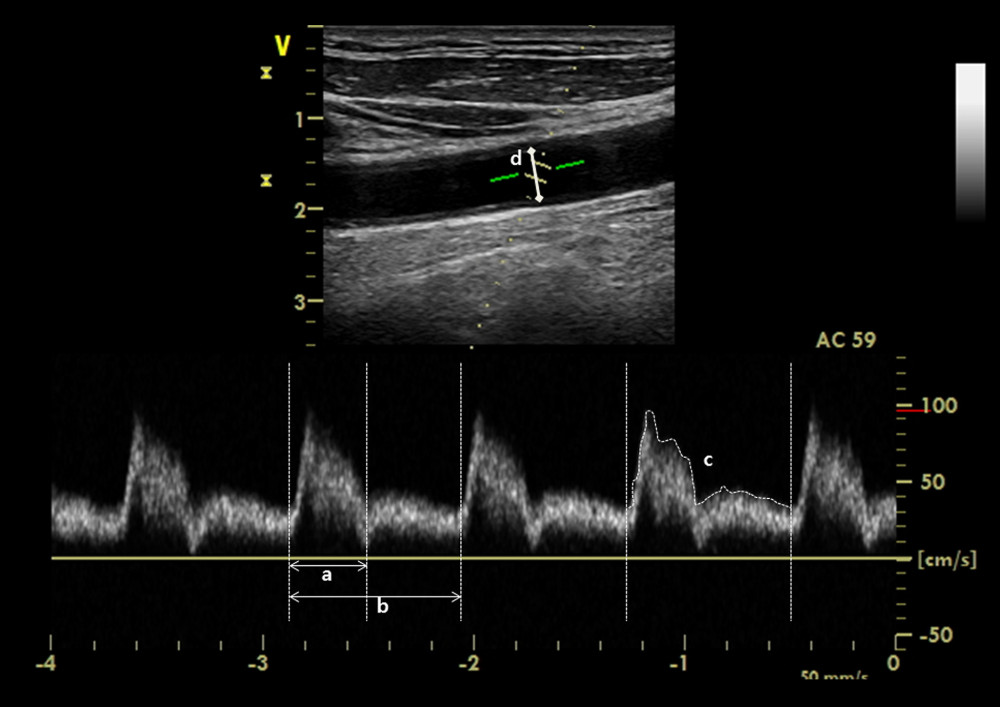 Figure 1. Corrected carotid flow time was calculated by the measurement of interval between the start of systolic upstroke and the dicrotic notch of the spectral Doppler waveform: a) systole time; b) cycle time. Carotid blood flow was calculated by the measurement of velocity time integral tracing of the c) spectral Doppler signal and d) carotid diameter.
Figure 1. Corrected carotid flow time was calculated by the measurement of interval between the start of systolic upstroke and the dicrotic notch of the spectral Doppler waveform: a) systole time; b) cycle time. Carotid blood flow was calculated by the measurement of velocity time integral tracing of the c) spectral Doppler signal and d) carotid diameter. 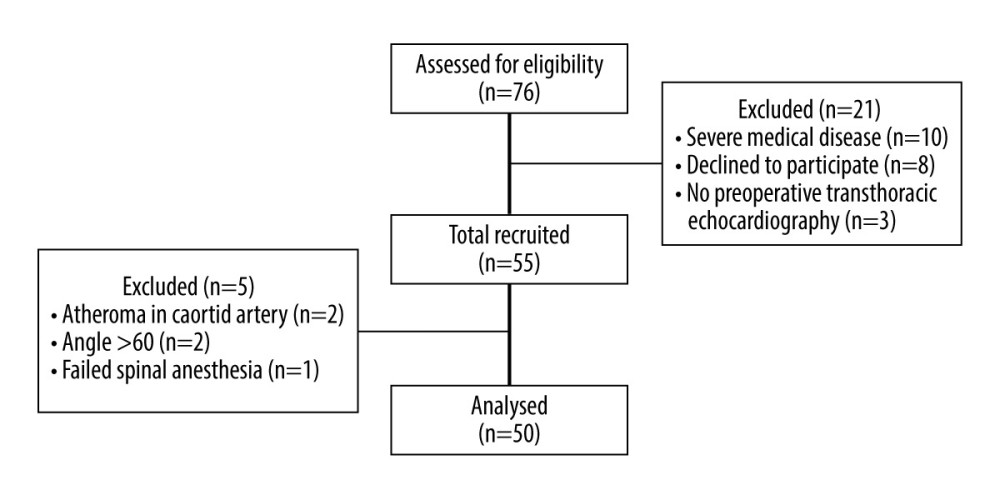 Figure 2. Consort flow diagram.
Figure 2. Consort flow diagram. 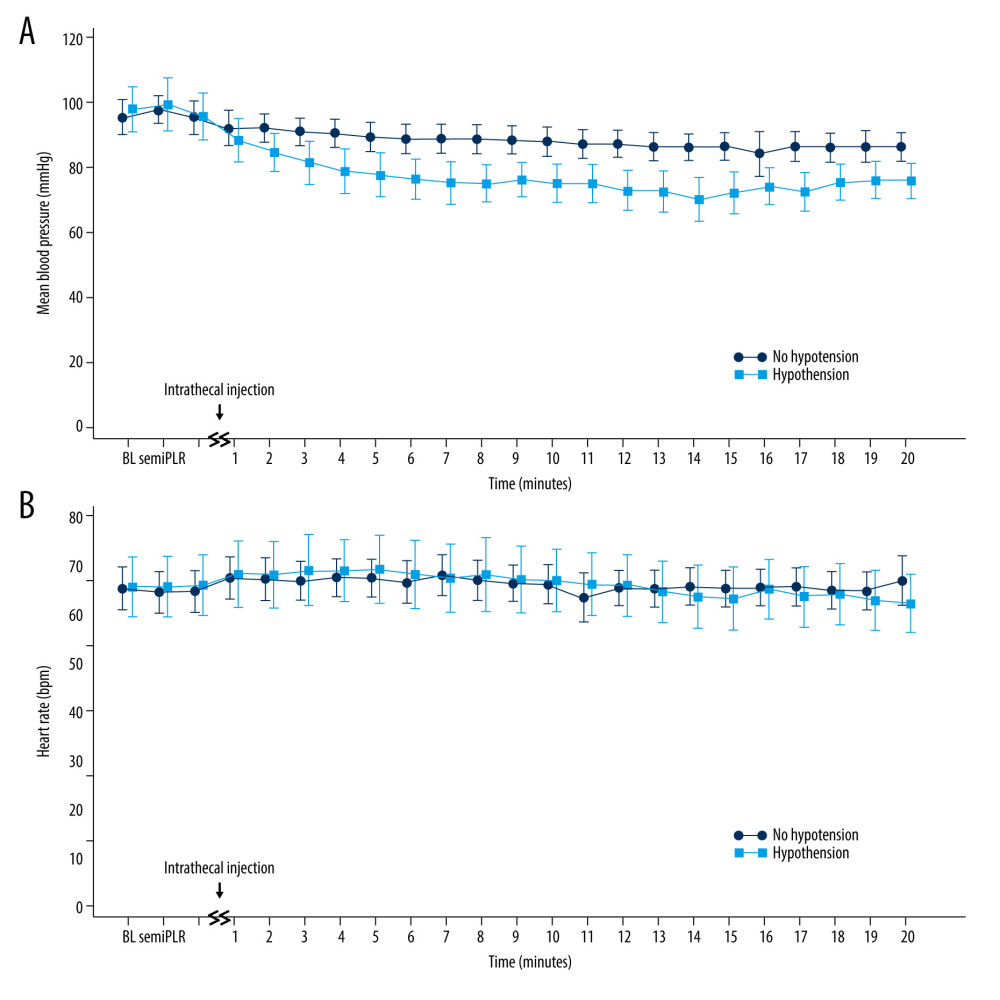 Figure 3. Hemodynamic parameters. (A) Mean blood pressure in the patients who developed hypotension decreased significantly compared with the baseline value and remained lower until the end of the study compared with patients who did not (P=0.006). (B) However, HR was not significantly changed, remaining close to baseline values in all patients (P=0.972). BL – baseline; semi, semirecumbent position; PLR – passive leg raise position. This figure was created with MedCalc® Statistical Software (version 13.2, MedCalc Software Ltd.).
Figure 3. Hemodynamic parameters. (A) Mean blood pressure in the patients who developed hypotension decreased significantly compared with the baseline value and remained lower until the end of the study compared with patients who did not (P=0.006). (B) However, HR was not significantly changed, remaining close to baseline values in all patients (P=0.972). BL – baseline; semi, semirecumbent position; PLR – passive leg raise position. This figure was created with MedCalc® Statistical Software (version 13.2, MedCalc Software Ltd.). Tables
Table 1. Patient characteristics.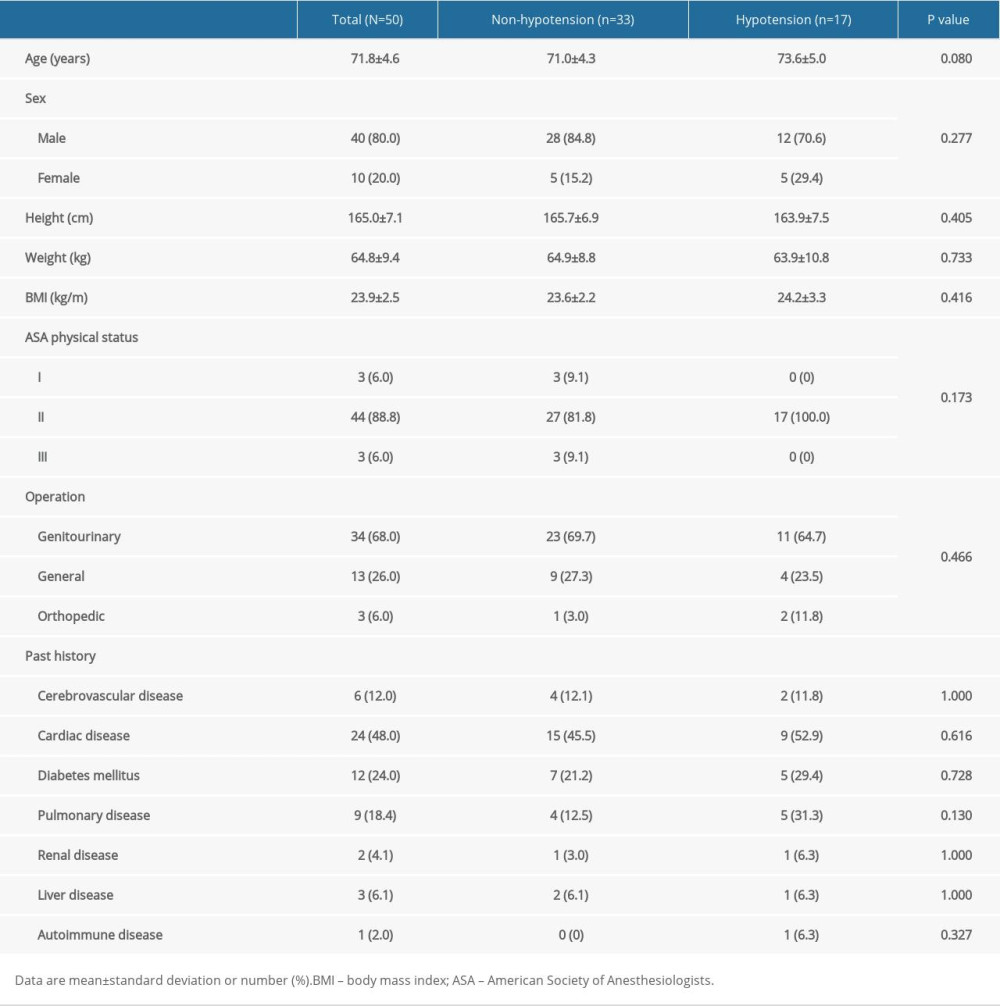 Table 2. Preoperative transthoracic echocardiography.
Table 2. Preoperative transthoracic echocardiography.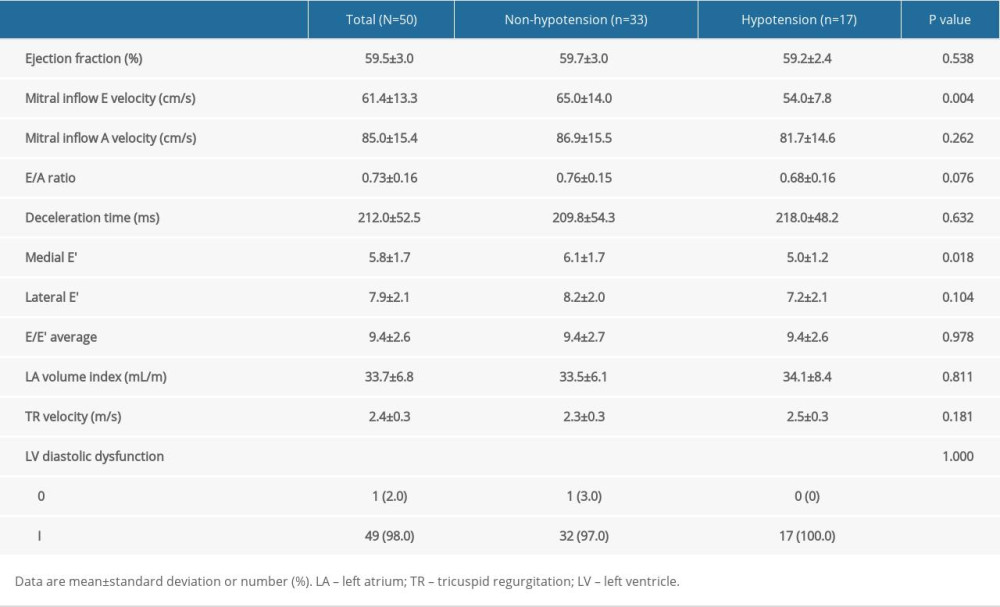 Table 3. Intraclass correlation coefficients.
Table 3. Intraclass correlation coefficients.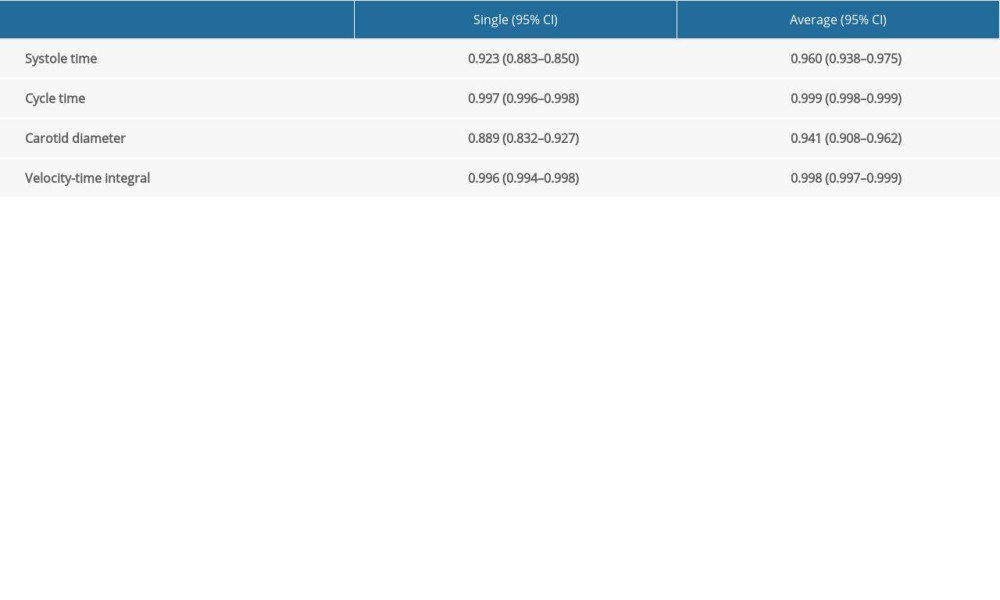 Table 4. Ultrasonographic carotid artery measurements and intraoperative data in patients with and without spinal anesthesia-induced hypotension.
Table 4. Ultrasonographic carotid artery measurements and intraoperative data in patients with and without spinal anesthesia-induced hypotension.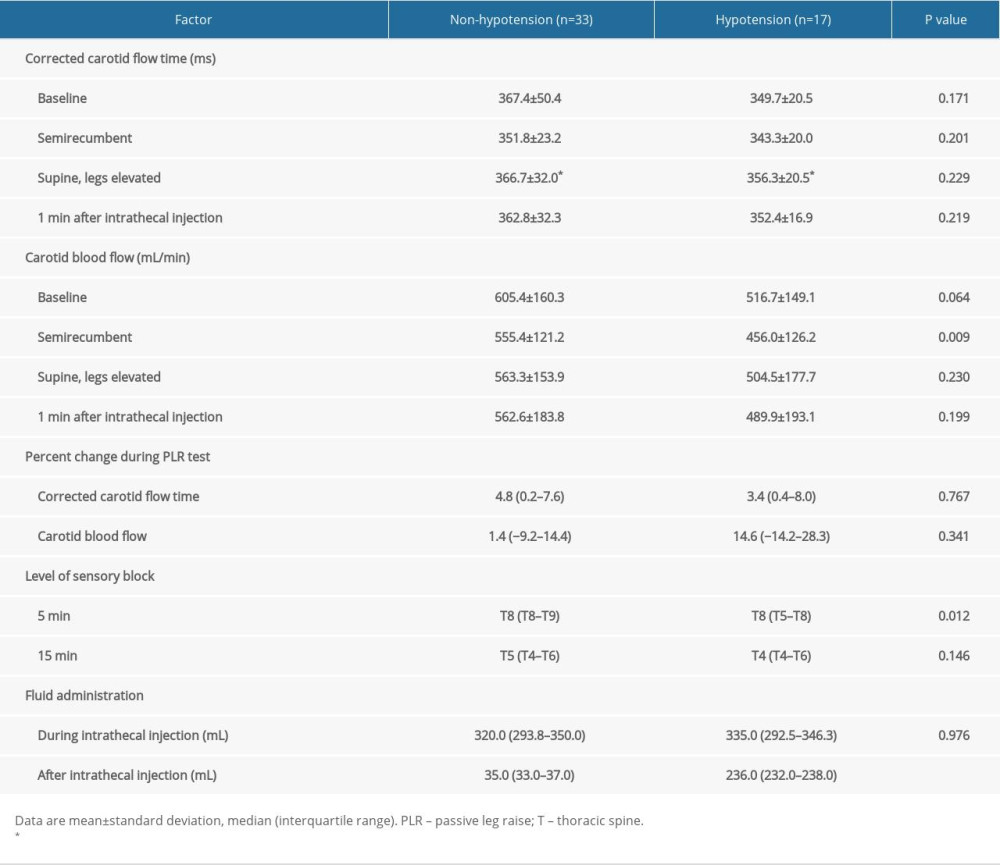 Table 5. Receiver operating characteristic (ROC) analysis to predict spinal anesthesia-induced hypotension.
Table 5. Receiver operating characteristic (ROC) analysis to predict spinal anesthesia-induced hypotension.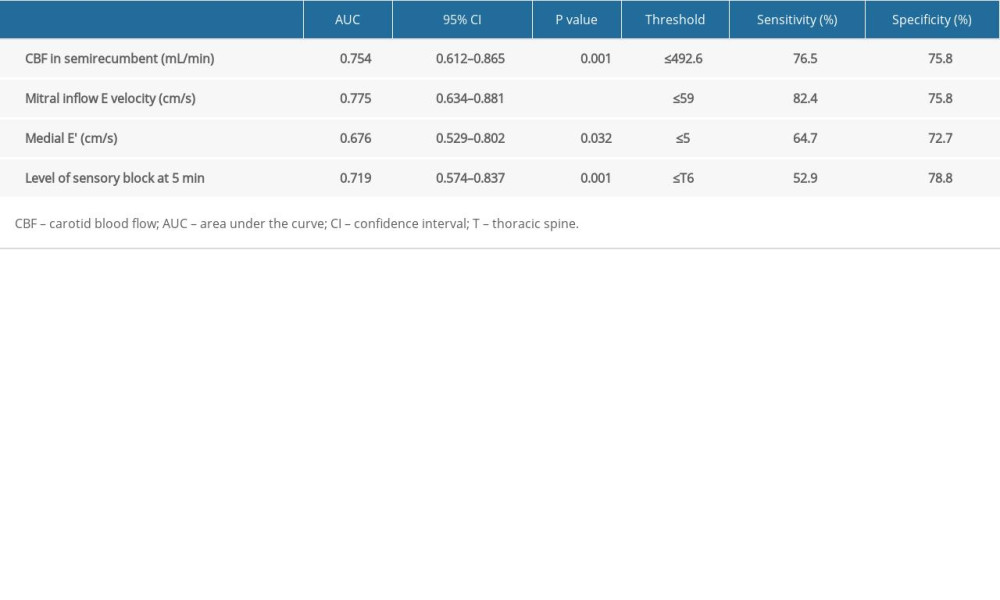 Table 6. Univariate and multivariate logistic regression of variables associated with spinal anesthesia-induced hypotension.
Table 6. Univariate and multivariate logistic regression of variables associated with spinal anesthesia-induced hypotension.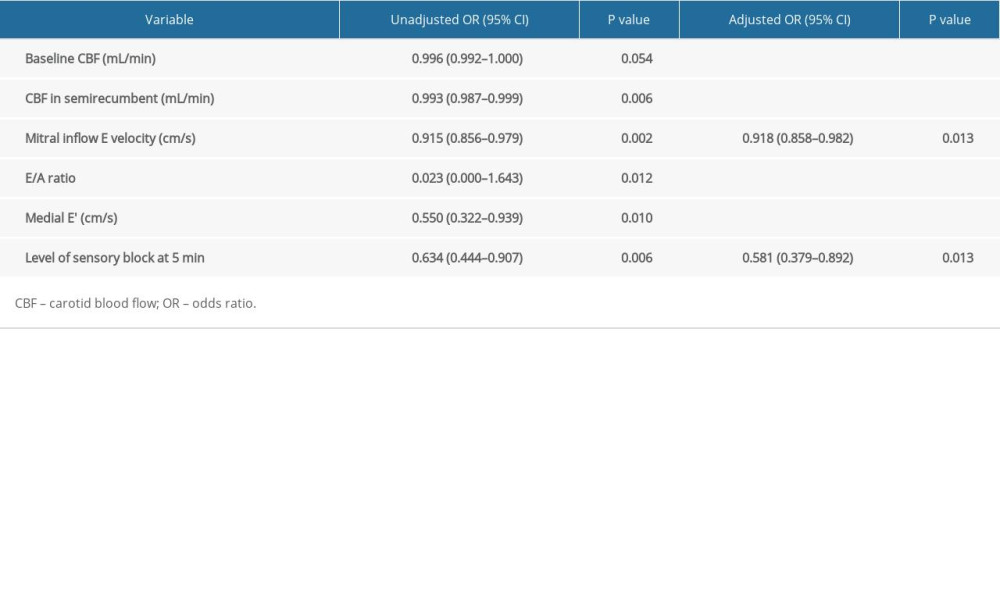
References
1. Cowen LE, Hodak SP, Verbalis JG, Age-associated abnormalities of water homeostasis: Endocrinol Metab Clin North Am, 2013; 42(2); 349-70
2. Rooke GA, Freund PR, Jacobson AF, Hemodynamic response and change in organ blood volume during spinal anesthesia in elderly men with cardiac disease: Anesth Analg, 1997; 85(1); 99-105
3. Dai X, Hummel SL, Salazar JB, Cardiovascular physiology in the older adults: J Geriatr Cardiol, 2015; 12(3); 196-201
4. Yap JC, Critchley LA, Yu SC, A comparison of three fluid-vasopressor regimens used to prevent hypotension during subarachnoid anaesthesia in the elderly: Anaesth Intensive Care, 1998; 26(5); 497-502
5. Lairez O, Ferre F, Portet N, Cardiovascular effects of low-dose spinal anaesthesia as a function of age: An observational study using echocardiography: Anaesth Crit Care Pain Med, 2015; 34(5); 271-76
6. Mottram PM, Marwick TH, Assessment of diastolic function: What the general cardiologist needs to know: Heart, 2005; 91(5); 681-95
7. Jalil B, Thompson P, Cavallazzi R, Comparing changes in carotid flow time and stroke volume induced by passive leg raising: Am J Med Sci, 2018; 355(2); 168-73
8. Ma IWY, Caplin JD, Azad A, Correlation of carotid blood flow and corrected carotid flow time with invasive cardiac output measurements: Crit Ultrasound J, 2017; 9(1) 10-017-0065-0
9. Marik PE, Levitov A, Young A, Andrews L, The use of bioreactance and carotid doppler to determine volume responsiveness and blood flow redistribution following passive leg raising in hemodynamically unstable patients: Chest, 2013; 143(2); 364-70
10. Chebl RB, Wuhantu J, Kiblawi S, Corrected carotid flow time and passive leg raise as a measure of volume status: Am J Emerg Med, 2018; 37(8); 1460-65
11. Antiperovitch P, Iliescu E, Chan B, Carotid systolic flow time with passive leg raise correlates with fluid status changes in patients undergoing dialysis: J Crit Care, 2017; 39; 83-86
12. American College of Radiology: ACR-AIUM-SPR-SRU practice parameter for the performance of an ultrasound examination of the extracranial cerebrovascular system, 2016 Available from: https://onlinelibrary.wiley.com//10.1002/jum.15877
13. Blehar DJ, Glazier S, Gaspari RJ, Correlation of corrected flow time in the carotid artery with changes in intravascular volume status: J Crit Care, 2014; 29(4); 486-88
14. Smiseth OA, Evaluation of left ventricular diastolic function: State of the art after 35 years with doppler assessment: J Echocardiogr, 2018; 16(2); 55-64
15. Marshall WK, Bedford RF, Miller ED, Cardiovascular responses in the seated position – impact of four anesthetic techniques: Anesth Analg, 1983; 62(7); 648-53
16. Rowland T, Unnithan V, Barker P, Orthostatic effects on echocardiographic measures of ventricular function: Echocardiography, 2012; 29(5); 523-27
17. Jakobsson J, Kalman SH, Lindeberg-Lindvet M, Bartha E, Is postspinal hypotension a sign of impaired cardiac performance in the elderly? An observational mechanistic study: Br J Anaesth, 2017; 119(6); 1178-85
18. Zhang N, He L, Ni JX, Level of sensory block after spinal anesthesia as a predictor of hypotension in parturient: Medicine (Baltimore), 2017; 96(25); e7184
19. Carpenter RL, Caplan RA, Brown DL, Incidence and risk factors for side effects of spinal anesthesia: Anesthesiology, 1992; 76(6); 906-16
20. Jabot J, Teboul JL, Richard C, Monnet X, Passive leg raising for predicting fluid responsiveness: Importance of the postural change: Intensive Care Med, 2009; 35(1); 85-90
21. Mackenzie DC, Khan NA, Blehar D, Carotid flow time changes with volume status in acute blood loss: Ann Emerg Med, 2015; 66(3); 277-282e1
22. Barjaktarevic I, Toppen WE, Hu S, Ultrasound assessment of the change in carotid corrected flow time in fluid responsiveness in undifferentiated shock: Crit Care Med, 2018; 46(11); e1040-e46
23. Meyhoff CS, Haarmark C, Kanters JK, Rasmussen LS, Is it possible to predict hypotension during onset of spinal anesthesia in elderly patients?: J Clin Anesth, 2009; 21(1); 23-29
24. Critchley LA, Stuart JC, Short TG, Gin T, Haemodynamic effects of subarachnoid block in elderly patients: Br J Anaesth, 1994; 73(4); 464-70
Figures
 Figure 1. Corrected carotid flow time was calculated by the measurement of interval between the start of systolic upstroke and the dicrotic notch of the spectral Doppler waveform: a) systole time; b) cycle time. Carotid blood flow was calculated by the measurement of velocity time integral tracing of the c) spectral Doppler signal and d) carotid diameter.
Figure 1. Corrected carotid flow time was calculated by the measurement of interval between the start of systolic upstroke and the dicrotic notch of the spectral Doppler waveform: a) systole time; b) cycle time. Carotid blood flow was calculated by the measurement of velocity time integral tracing of the c) spectral Doppler signal and d) carotid diameter. Figure 2. Consort flow diagram.
Figure 2. Consort flow diagram. Figure 3. Hemodynamic parameters. (A) Mean blood pressure in the patients who developed hypotension decreased significantly compared with the baseline value and remained lower until the end of the study compared with patients who did not (P=0.006). (B) However, HR was not significantly changed, remaining close to baseline values in all patients (P=0.972). BL – baseline; semi, semirecumbent position; PLR – passive leg raise position. This figure was created with MedCalc® Statistical Software (version 13.2, MedCalc Software Ltd.).
Figure 3. Hemodynamic parameters. (A) Mean blood pressure in the patients who developed hypotension decreased significantly compared with the baseline value and remained lower until the end of the study compared with patients who did not (P=0.006). (B) However, HR was not significantly changed, remaining close to baseline values in all patients (P=0.972). BL – baseline; semi, semirecumbent position; PLR – passive leg raise position. This figure was created with MedCalc® Statistical Software (version 13.2, MedCalc Software Ltd.). Tables
 Table 1. Patient characteristics.
Table 1. Patient characteristics. Table 2. Preoperative transthoracic echocardiography.
Table 2. Preoperative transthoracic echocardiography. Table 3. Intraclass correlation coefficients.
Table 3. Intraclass correlation coefficients. Table 4. Ultrasonographic carotid artery measurements and intraoperative data in patients with and without spinal anesthesia-induced hypotension.
Table 4. Ultrasonographic carotid artery measurements and intraoperative data in patients with and without spinal anesthesia-induced hypotension. Table 5. Receiver operating characteristic (ROC) analysis to predict spinal anesthesia-induced hypotension.
Table 5. Receiver operating characteristic (ROC) analysis to predict spinal anesthesia-induced hypotension. Table 6. Univariate and multivariate logistic regression of variables associated with spinal anesthesia-induced hypotension.
Table 6. Univariate and multivariate logistic regression of variables associated with spinal anesthesia-induced hypotension. Table 1. Patient characteristics.
Table 1. Patient characteristics. Table 2. Preoperative transthoracic echocardiography.
Table 2. Preoperative transthoracic echocardiography. Table 3. Intraclass correlation coefficients.
Table 3. Intraclass correlation coefficients. Table 4. Ultrasonographic carotid artery measurements and intraoperative data in patients with and without spinal anesthesia-induced hypotension.
Table 4. Ultrasonographic carotid artery measurements and intraoperative data in patients with and without spinal anesthesia-induced hypotension. Table 5. Receiver operating characteristic (ROC) analysis to predict spinal anesthesia-induced hypotension.
Table 5. Receiver operating characteristic (ROC) analysis to predict spinal anesthesia-induced hypotension. Table 6. Univariate and multivariate logistic regression of variables associated with spinal anesthesia-induced hypotension.
Table 6. Univariate and multivariate logistic regression of variables associated with spinal anesthesia-induced hypotension. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








