20 January 2023: Clinical Research
Monitoring of Autoantibodies Following Autologous Hematopoietic Stem Cell Transplantation in 6 Children with Recently Diagnosed Type 1 Diabetes Mellitus
Alejandro D. Mesples1ABCDEFG*, Desiree C.T. Cox23ACDEF, Harriet D. Lundy1ADE, Sheena Antonio-Collie1CDG, Charles W. Diggiss1A, Jonathan R.T. Lakey45ACDEFDOI: 10.12659/MSM.938979
Med Sci Monit 2023; 29:e938979
Abstract
BACKGROUND: Bone marrow stem cells have been shown to be a promising therapeutic strategy for autoimmune diseases. This study aimed to assess the safety and efficacy of autologous hematopoietic stem cell (ABMSC) transplantation without immunoablation used to suppress the autoimmune reaction in 6 children with newly diagnosed autoimmune diabetes mellitus. We monitored the levels of islet cell antibodies (ICA), antibodies against islet antigen-related tyrosine phosphatase 2 (IA2), glutamic acid-decarboxylase (GAD) antibodies, and anti-insulin antibodies (AIA).
MATERIAL AND METHODS: Between 2018 and 2022, 6 children (age 6-10 years, average 8 years) recently diagnosed with type 1 diabetes mellitus with the presence of ICA, IA2, GAD, AIA and ketoacidosis, were treated with an ABMSC stimulated with Filgrastim, granulocyte colony-stimulating factor (G-CSF), 10 ug/kg/day for 4 days. Bone marrow was harvested on day 5, collected by puncture and identified as mononuclear cells >180×10⁶/kg, CD34+ >0.22%, and transplanted by intravenous (i.v.) infusion. Patients were monitored with ICA, IA2, GAD, AIA, C-peptide, blood glucose, and glycosylated hemoglobin A1c (HbA1C) 6 months after the procedure.
RESULTS: At 6-month follow-up, we observed a negative value of the ICA, which was previously positive (P<0.001). The IA2 (p=0.037) and GAD (P=0.377) antibodies decreased slowly but were significantly lower. AIA remained high. A decrease in blood glucose and HbA1C levels was observed (P<0.001). No complications occurred during follow-up.
CONCLUSIONS: Autologous hematopoietic stem cell transplantation without immunoablation was safe and effective in significantly decreasing the production and effect of autoantibodies against ICA, GAD, and IA2, as well as decreasing blood sugar levels and HbA1c.
Keywords: Humans, Child, Diabetes Mellitus, Type 1, Autoantibodies, glycated hemoglobin, Blood Glucose, Hematopoietic Stem Cell Transplantation, Glutamate Decarboxylase, Islets of Langerhans
Background
Diabetes is a chronic and degenerative disease with short- and long-term multi-functional complications and significant associated mortality and morbidity [1,2]. Autoimmune T1D is a metabolic disorder characterized by elevated blood glucose levels as a result of decreased insulin secretion. This decrease in insulin secretion is secondary to the destruction of the β cells of the pancreatic islets. The World Health Organization recently declared diabetes to be a pandemic. It is a significant global health problem with millions of patients worldwide. Individuals with diabetes have a 2–4-fold higher chance of having a stroke, myocardial infarction, or peripheral artery disease in comparison to individuals without diabetes. Currently, diabetes lacks effective treatments. America, and in particularly the Caribbean and Central America regions, are seriously affected by the disease. The Caribbean region has almost twice the incidence (12%) of type 1 and type 2 diabetes compared to other regions in America [3]. In the Bahamas, 50–60% of all citizens will develop diabetes at some point in their lives and 50% of all deaths in the Bahamas are due to a condition related to diabetes [3]. Diabetes is the leading cause of blindness in individuals aged 20–74 years in the Bahamas. Moreover, diabetes creates a serious financial burden, with 80% of the total drug costs for the National Prescription Drug Plan currently spent on treatment for diabetes and hypertension [3].
Autoimmune diabetes is characterized by the presence of anti-islet antibodies (ICA), anti-CMV IE2 antibodies, anti-glutamic acid decarboxylase antibodies (anti-GAD antibodies), and anti-insulin antibodies [4]. Type 1 diabetes mellitus (T1D) affects children and young adults. Specific autoantibodies and killer T cells attack insulin-secreting pancreatic islet β cells and cause their destruction. This causes a decrease in insulin secretion and leads to the need for exogenous insulin to regulate glucose metabolism. Islet cell autoantibodies are strongly associated with the development of T1D. The specificity of autoantibodies against pancreatic islet cells (ICA), islet cell antigen 2 (IA2), and antibodies against insulin is very high in autoimmune diabetes [5,6]. The attack of these antibodies is the characteristic feature of the first stage of T1D and the sum of the antibodies involved has important prognostic implications, since it is directly related to the severity of the disease [5,6] therefore, the antibody level is reported as an independent risk variable [6]. Antibodies destroy insulin-producing pancreatic cells, T cells, and regulatory T cells (Tregs). We know that effector T cells are activated against and attack pancreatic cells, while Treg cells, which normally maintain the balance of cellular reaction, homeostasis, and self-tolerance through their inhibitory impact on autoreactive effector T cells [7], are decreased. There is evidence that Treg cell abnormalities, either in number or function, are associated with the onset and progression of T1D [8].
Autoimmune diabetes confronts us with a challenge because children not only require the supply of insulin and glycemic control, but also need correct nutritional intake, physical activity, and a social environment that allows them to grow properly and develop fully [9]. Children have different needs compared to those of adults with diabetes. For this reason, the different therapeutic alternatives with the different types of insulin and control devices must be analyzed individually to be effective in a broad sense. Currently, the main goals of treating autoimmune diabetes in children include controlling and maintaining glucose levels as close to normal as possible, avoiding acute complications, and preventing long-term complications [9]. Recent reports describe the results of these strategies and warn of an increase in complications and the cost-benefit ratio [10]. Undoubtedly, the treatment of autoimmune diabetes presents a complex problem and requires innovative alternatives for its management.
Different authors have shown that hematopoietic stem cells and mesenchymal cells have properties that allow them to regulate the action of the immune system [11,12]. Several studies have shown that mesenchymal cells (MSCs) can secrete peptides, such as hepatocyte growth factor, with local immunosuppressive capacity [12]. Transforming growth factor-1 has also been shown to interact with hepatocyte growth factor and cause T cell suppression [13]. Interleukin-10 (IL-10) also appears to be expressed by MSCs and has a proven role in regulating T cells and promoting the suppressive phenotype by antagonizing the action of interleukin-12 (IL-12) during induction of the inflammatory immune response [14–16].
Bone marrow stem cells (BMSCs) also display immunomodulatory properties via CD4+ and CD8+ T cells via surface molecular program death ligand 1 (PD-L1), secrete nitric oxide (NO), and control lymphocyte proliferation by mitogen suppression [17]. Another group of researchers has shown a significant decrease in the proliferation of T cell clones after stimulation by antigen-presenting cells (APCs) and glutamic acid peptide decarboxylase (GAD), suggesting that BMSCs can suppress the proliferation of GAD-specific T cells [18].
Many studies have shown that control of diabetes is correlated with systemic immune disturbances, such as restoration of T-lymphocyte helper cells (Th 1 and 2), balance of cytokines in the blood, and local immune regulation in pancreatic islets through transforming growth factor −b1, which can protect pancreatic islet β cells against these lymphocytes [19–21].
Based on these research findings, adult stem cells are now emerging as a valid alternative treatment for autoimmune diabetes [22]. Hussein et al have shown that adult stem cells can migrate and differentiate into cells of different lineages. Using human pancreatic islets as an in vitro model of peripheral tissues, it has been observed that these islets release factors that attract stem cells and this attraction is mainly mediated by the chemokines CX3CL1 and CXCL12 [23,24].
Many in vitro studies have demonstrated the immunomodulatory and anti-inflammatory properties of stem cells and their mechanisms of action [25–28]. Recently, Lakey et al demonstrated that mesenchymal cells, acting through their exosomes, decreased demyelination, decreased neuroinflammation, and increased the number of CD4+CD25+FOXP3+ regulatory T cells (Tregs) in the spinal cords of mice with autoimmune encephalomyelitis [29].
Autologous hematopoietic stem cell transplantation has been tested in several clinical trials to treat autoimmune diseases, such as multiple sclerosis (MS), systemic sclerosis (SSc), systemic lupus erythematosus (SLE), Crohn’s disease (CD), type 1 diabetes (T1D), and a variety of other immune-mediated disorders [30–32].
In autoimmune diabetes there are clinical experiences using hematopoietic stem cells (HSC) with results that demonstrate their effectiveness, even recent studies report that immunomodulatory and trafficking abilities can be improved in genetically modulated HSC and genetically modified HSC can be seen as a new therapy in type 1 diabetes. However, there are still doubts regarding the durability of its effects [33]. There is evidence showing significant independence from insulin with metabolic control and increased C-peptide in short- and medium-term follow-up. Even a cost-effectiveness analysis conducted in type 1 diabetic patients who underwent autologous hematopoietic cell transplantation, compared to type 1 diabetic patients who remained on insulin therapy, showed cost-effectiveness benefits [33].
Recently, Madani et al [34] published a meta-analysis showing that 26% of treated patients were insulin-free for a period of 1 to 80 months and 68% of patients showed a decrease in the daily dose of insulin after autologous hematopoietic cell transplantation. The combination of hematopoietic stem cells (HSC) with mesenchymal stem cell (MSC) transplantation was significantly associated with improvement in daily insulin dose, with a decrease in HbA1C and an increase in C-peptide. However, they found many serious adverse events secondary to immune ablation before transplant, which improves the safety of the treatment [34].
In the present study, we chose to measure antibodies because other authors have already verified the influence of hematopoietic stem cells in inactivating T cells, or the cellular pathway for the destruction of pancreatic B cells, but not the antibody-mediated humoral pathway. Also, the levels of ICA, GADA, and IA2 are high at the onset of diabetes and their determination is simple, routine, and effective in the diagnosis of autoimmune diabetes and checking the humoral immune reaction. In addition, follow-up can quantify the effectiveness of the proposed treatment. We have previously published our experience in the measurement of antibodies in patients with autoimmune diabetes and treatment with hematopoietic stem cells but implanted via liver puncture [35].
Our present study describes the changes observed in the measurement of ICA, anti-GAD, anti-CMV IA2, and anti-insulin antibodies during the 6-month follow-up in 6 children with recently diagnosed autoimmune diabetes after administration of Filgrastim-stimulated autologous hematopoietic stem cells from bone marrow transplantation and intravenous infusion, without immune ablation, with ex vivo or cell cultures. To the best of our knowledge, this is the first report on this topic.
Material and Methods
PATIENT CHARACTERISTICS:
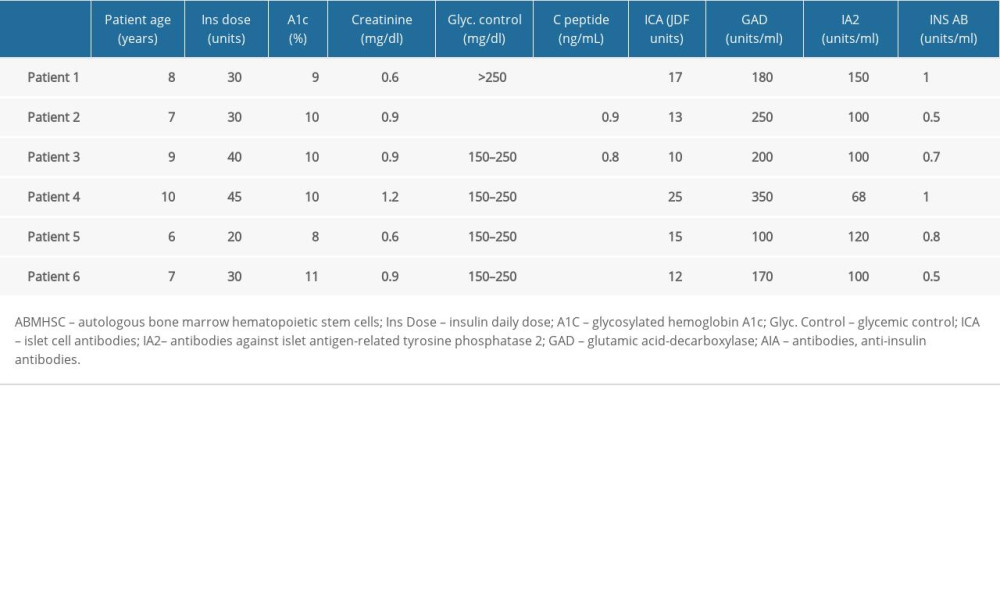
Diabetes was diagnosed by an independent endocrinologist with a non-fasting C-peptide below 1.0 mg/dL and hyperglycemia above 300 mg/dL, along with glycated hemoglobin A1C (HbA1C) and a physical exam. The patients had a diagnosis of type 1 diabetes for <120 days (60–120 days) and were on average aged 8 years [6–10]. Of the subjects, there were 5 female patients and 1 male patient. For all 6 patients, body mass index was normal at <22 kg/m2, with a normal complete blood count and coagulation, normal renal function, no lesions in target organs, HbA1c level < 12% (6–12), C-peptide level <0.9 ng/ml, and positive results for antibodies to islets cells (ICAs), tyrosine phosphatase-related islet antigen 2 (IA2), glutamic acid decarboxylase (GAD), and insulin antibody (Table 1). The families of the patients provided informed consent.
The subjects were treated with autologous bone marrow stem cells stimulated with Filgrastim for 4 days (10 ug/kg/day, subcutaneous) and then delivered through i.v. infusion as immune modulators on Day 5. Subjects were then monitored and any adverse effects were noted and managed by the physician/nursing team. All patients continued to receive the standard treatment with blood glucose monitoring multiple times per day concurrently with a continuous glucose monitoring device and subcutaneous insulin delivery to maintain non-fasting blood glucose <20 mg/dL.
BONE MARROW PREPARATION AND TRANSPLANTATION:
Bone marrow stimulation was performed using Filgrastim (10 ug/kg/day) subcutaneously, and granulocyte colony-stimulating factor (G-CSF) daily from Day 1 through Day 4. Bone marrow was aspirated on Day 5 under general anesthesia and stem cells were extracted by a hip puncture from the anterior superior crest. The BMSCs were transplanted by i.v. infusion in a volume of 300 ml. The qualified autologous bone marrow stem cells were collected and identified as mononuclear cells >180×106/kg, CD34+ cells >0.22%.
STATISTICAL EVALUATION:
Testing was standardized for each peripheral blood sample (baseline, when Filgrastim stimulation and 6 months follow-up). Data were analyzed using ANOVA, with
FOLLOW-UP:
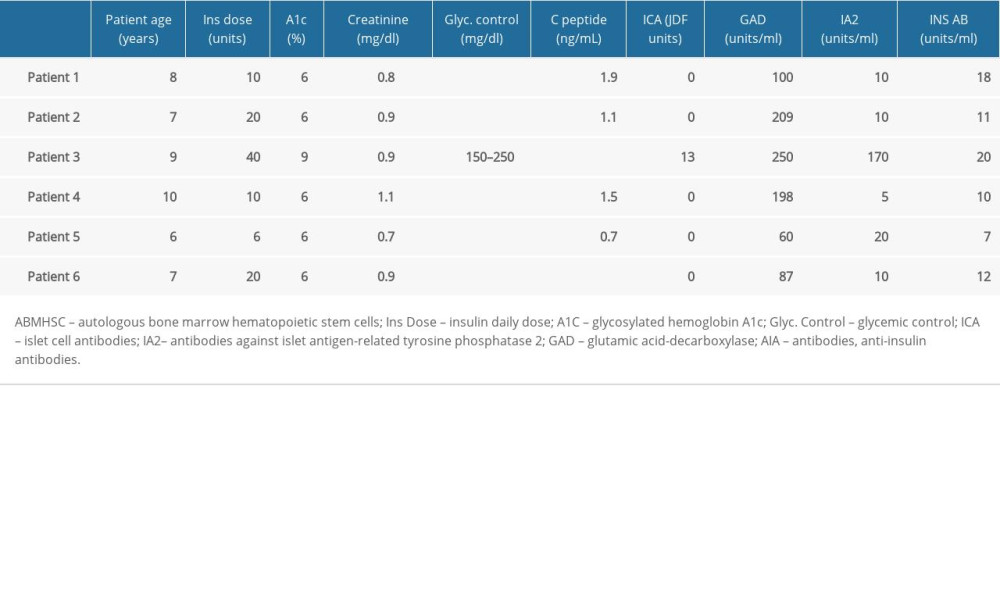
The subjects were contacted by phone every 48 h in the first week after the stem cell infusion. Clinical evaluations were performed at baseline (pre-treatment Table 1) and at 6 months (post-treatment Table 2). These included measurements of daily insulin dose, complete blood counts, creatinine clearance test, C-peptide by chemiluminescence (normal value 0.90–7.10 ng/ml), glycosylated hemoglobin (HbA1C) by liquid chromatography (normal value 4.20–6.00%), ICA anti-islet antibody by immunofluorescence: normal value negative, Juvenile Diabetes Foundation Units (JDF), GAD antibody by radio immune analysis (normal value: equal to or less than 1.0 U/ml), and anti-insulin antibody by radio immune analysis (normal value: equal to or less than 0.4 U/ml).
Results
At 6-month follow-up after treatment, 5 of the 6 treated subjects presented negative values of anti-pancreatic islet antibodies (ICA), having previously been positive before treatment (
There were no significant complications observed in any subjects during the bone marrow puncture from hip, aspiration, or i.v. infusion immediately following the procedure or in the 6 months after the procedure.
Results show significant changes in patients’ ICA antibody and IA2 antibody levels and insulin daily dose, HbA1C level, and glycemic control, and no significant changes in creatinine, C-peptide, GAD antibody, or insulin antibody levels comparing the change in these variables at 6 months after treatment with the autologous hematopoietic bone marrow stem cell transplant, as shown in Table 2.
Discussion
In type 1 diabetes mellitus, activated T cells, anti-islet antibodies (ICA), anti-IA2 antibodies, glutamic acid decarboxylase antibodies (anti-GAD antibodies), and anti-insulin antibodies can be found in 90% of cases, and about 10% of cases are idiopathic. These antibodies rise during the first years of the course of the disease and spontaneously follow a downward curve when the number of pancreatic islets, which are antigenic for T cells and autoantibodies, decreases [36–38].
The prevalence of diabetes mellitus worldwide was estimated at 2.8% in 2000 and will increase to 4.4% by 2030 [38]. These statistics are convincing and show an increase in the number of patients suffering from the disease and also an increase in serious complications without curative treatment. Currently, no medical treatment is available to change the course of autoimmune diabetes. This justifies the research and development of alternative therapeutic strategies.
Our procedure consisted of stimulating bone marrow stem cells with colony-stimulating factors (Filgrastim), with the aim of increasing the number of BMSCs, and their subsequent i.v. infusion into the systemic circulation. This protocol does not require immune ablation with cyclophosphamide or cell culture procedure, and, advantageously, can be immediately implanted into the subject.
Our work succeeded in altering the natural history curve of autoimmune diabetes-specific antibodies. We were able to verify that ICA was negative at 6-month follow-up, while GAD and IA2 decreased significantly more slowly. In addition, patients who responded to treatment with negative AHF achieved metabolic stability measured with normal HbA1c at 6-month follow-up. The anti-insulin antibodies remained high, probably secondary to the exogenous insulin administration.
We know that the stimulation of the bone marrow with Filgrastim does not increase the production of mesenchymal cells, but our data suggests that it stimulates hematopoietic stem cells in their entire series of differentiation and that the transplantation of these stem cells acts on the immune system to modulate the immune system, decrease inflammation, and increase T cell regulation. It has been hypothesized that implanted bone marrow cells can stimulate the secretion of chemokines, which can modulate the action of T cells, antigen-presenting cells, and lymphocytes that could reverse antibody production, as described in clinical experiences [30–32].
Gu et al [39] treated 14 children with newly diagnosed type 1 diabetes with autologous hematopoietic cell transplantation with immunoablation and separation of cell fractions prior to transplantation. This group found no statistically significant differences between the treated group and the placebo group in daily insulin dose, HbA1C, and C-peptide levels at long-term follow-up. ICA, GAD, IA2 antibodies, and T cell activity were not measured in this study due to the prior use of cyclophosphamide in marrow preparation for transplantation [39]. This group observed that 90% of the patients presented serious adverse events after transplantation, secondary to treatment with cyclophosphamide. Although, we consider that the group of treated patients included was very small, and did not allow any definitive conclusion. The main difference was found in the immunoablation with cyclophosphamide, which was not performed in our study.
Izadi et al [40] recently published an interesting clinical study in which 21 patients with recently diagnosed T1D were enrolled and randomly assigned to receive either MSCs twice by i.v. infusion and followed for at least 1 year after transplantation [34]. The results showed that MSC transplantation was safe and significantly reduced the number of hypoglycemic episodes. Further, results showed that MSC transplantation improved glycated hemoglobin (HbA1c), shifted serum cytokine patterns from pro-inflammatory to anti-inflammatory, increased the number of regulatory T cells in the peripheral blood, significantly improved HbA1c and C-peptide levels and, importantly, improved patient quality of life [40]. Although our study did not control the evolution of ICA antibodies, it was able to demonstrate the immunological control of T cells and pro-inflammatory cytokines.
In our work, patients who received treatment showed a significant reduction in anti-islet (ICA) and IA2 antibodies from positive to negative levels at 6-month follow-up. Interestingly, negative results for antibodies were associated with increased C-peptide, although this was not statistically significant; a decreased significant requirement for daily insulin dose; and decreased concentrations of glycosylated hemoglobin (HbA1c).
We believe that the increase in C-peptide, which did not show significance because of the short evolution time, and the decrease in HbA1c is secondary to a decrease in inflammation of pancreatic tissue. We believe that there are inactive pancreatic cells in a niche, acting as “hibernating” cells, that can function again when the inflammation subsides, such as occurs in acute myocardial infarction or chronic cardiac ischemia [41]. Our results showed that hematopoietic bone marrow stem cells transplantation had an effect on the production of pancreatic antibodies and improved pancreatic function, decreased pancreatic tissue inflammation, and improved metabolic control at 6-month follow-up. This treatment protocol is an easy to perform, low-risk procedure, with no ethical problems. No complications were observed during the transplant procedure or during 6-month follow-up, probably because we did not use cyclophosphamide.
We believe that the cell transplantation influenced the suppression of anti-islet antibodies and reversed their effects. Our previous experience in T1D patients who received an autologous bone marrow cell implant through a liver puncture [35] similarly demonstrated ICA antibodies were negative at 6 months of post-procedure evolution. The results were similar to those obtained in the present study, although the elimination of the liver puncture and its replacement with an i.v. delivery of stimulated bone marrow reduces the complexity of the procedure and the risks of an invasive technique such as liver puncture. This therapeutic method appears to be an effective alternative for the treatment of recently diagnosed T1D.
This suggests that using this procedure may be used following screening for islet cell antibodies in patients with newly diagnosed T1D. Early referral of positive patients for autologous bone marrow transplantation could potentially reduce the time of exposure to antibodies, thereby decreasing pancreatic tissue damage and improving patient outcomes.
Despite the small number of subjects treated and short follow-up, our results suggest that we can stop the continued progression and extension of immune attack in a patient’s pancreas. We must continue making strides in clinical research with larger patient groups and long-term monitoring. We suggest that these results should be assessed in multicenter RCTs in different patient groups.
Conclusions
Autologous bone marrow stem cell transplantation in newly diagnosed T1D patients has been shown to decrease levels of ICA and IA2 antibodies and suppress autoimmune attack and pancreatic damage. We believe that these effects, shown 6 months after bone marrow transplantation, imply a change in the natural history of the disease and could imply a delay in the progression of pancreatic damage. Future studies will help to understand the mechanism of this treatment and the duration of expected benefits.
References
1. Wild S, Roglic G, Green A, Global Prevalence of Diabetes. Estimates for the year 2000 and projections for 2030: Diabetes Care, 2004; 27; 1047-53
2. Onkamo P, Väänänen S, Karvonen M, Tuomilehto J, Worldwide increase in incidence of type I diabetes – the analysis of the data on published incidence trends: Diabetologia, 1999; 42; 1395-403
3. Barcelo A, Aedo C, Rajpathak S, Robles S, The cost of diabetes in Latin America and the Caribbean: Bull World Health Organ, 2003; 81; 1
4. Expert Committee on the Diagnosis and Classification of Diabetes Mellitus, Report of the Expert Committee on the Diagnosis and Classification of Diabetes Mellitus: Diabetes Care, 1997; 20; 1183-97
5. Gianani R, Campbell-Thompson M, Sarkar SA, Dimorphic histopathology of long-standing child-hood-onset diabetes: Diabetologia, 2010; 53; 690-98
6. Sue M, Yoshihara A, Otani T, Characteristics of fulminant type 1 diabetes mellitus: Med Sci Monit, 2008; 14(10); CS97-101
7. Bluestone JA, Tang Q, Sedwick CE, T regulatory cells in autoimmune diabetes: Past challenges, future prospects: J Clin Immunol, 2008; 28; 677-84
8. Alonso N, Soldevila B, Sanmarti A, Regulatory T cells in diabetes and gastritis: Autoimmun Rev, 2009; 8; 659-62
9. Beck JK, Cogen Fran R, Outpatient management of pediatric type 1 diabetes: J Pediatr Pharmacol Ther, 2015; 20(5); 344-57
10. Herman WH, Braffett BH, Kuo Sh, The 30-year cost-effectiveness of alternative strategies to achieve excellent glycemic control in type 1 diabetes: An economic simulation informed by the results of the diabetes control and complications trial/epidemiology of diabetes interventions and complications (DCCT/EDIC): J Diabetes Complications, 2018; 32(10); 934-39
11. Ali T, Behrouz N, Aziz G, Immunopathology of type 1 diabetes and immunomodulatory effects of stem cells: A narrative review of the literature: Endocr Metab Immune Disord Drug Targets, 2022; 22; 169-97
12. Elliott JF, Qin Hui-Yu, Bhatti S, Immunization with the larger isoform of mouse glutamic acid decarboxylase (GAD67) prevents autoimmune diabetes in NOD mice: Diabetes, 1994; 43(12); 1494-99
13. Zhao Y, Huang Z, Qi M, Immune regulation of T lymphocyte by a newly characterized human umbilical cord blood stem cell: Immunol Lett, 2007; 108; 78-87
14. Liu L, He H, Liu A, Therapeutic effects of bone marrow-derived mesenchymal stem cells in models of pulmonary and extrapulmonary acute lung injury: Cell Transplant, 2015; 24; 2629-42
15. Leblanc K, Ringden O, Immunomodulation by mesenchymal stem cells and clinical experience: J Intern Med, 2007; 262; 509-25
16. Tse WT, Pendleton JD, Beyer WM, Suppression of allogeneic T-cell proliferation by human marrow stromal cells: Implications in transplantation: Transplantation, 2003; 75; 389-97
17. LeBlanc K, Tammik L, Sundberg B, Mesenchymal stem cells inhibit and stimulate mixed lymphocyte cultures and mitogenic responses independently of the major histocompatibility complex: Scand J Immunol, 2003; 57; 11-20
18. Becker JC, Andersen MH, Schrama D, Immune-suppressive properties of the tumor microenvironment: Cancer Immunol Immunother, 2013; 62; 1137-48
19. Bluestone JA, Tang Q, Sedwick CE, T regulatory cells in autoimmune diabetes: Past challenges, future prospects: J Clin Immunol, 2008; 28; 677-84
20. Zhao Y, Jiang Z, Zhao T, Reversal of type 1 diabetes via islet beta cell regeneration following immune modulation by cord blood-derived multipotent stem cells: BMC Med, 2012; 10; 3
21. Abdi R, Fiorina P, Adra CN, Immunomodulation by mesenchymal stem cells: A potential therapeutic strategy for type 1 diabetes: Diabetes, 2008; 57; 1759-67
22. Aguayo-Mazzucato C, Bonner-Weir S, Stem cell therapy for type 1 diabetes mellitus: Nat Rev Endocrinol, 2010; 6; 139-48
23. de la Tour D, Halvorsen T, Demeterco C, Beta-cell differentiation from a human pancreatic cell line in vitro and in vivo: Mol Endocrinol, 2001; 15; 476-83
24. Ianus A, Holz GG, Theise ND, Hussain MA, In vivo derivation of glucose-competent pancreatic endocrine cells from bone marrow without evidence of cell fusion: J Clin Invest, 2003; 111(6); 843-50
25. Moodley Y, Vaghjiani V, Chan J, Anti-inflammatory effects of adult stem cells in sustained lung injury: A comparative study: PLoS One, 2013; 8(8); e0069299
26. Sánchez L, Gutierrez-Aranda I, Ligero G, Enrichment of human ESC-derived multipotent mesenchymal stem cells with immunosuppressive and anti-inflammatory properties capable to protect against experimental inflammatory bowel disease: Stem Cells, 2011; 29; 251-62
27. Salari V, Mengoni F, Del Gallo F, The anti-inflammatory properties of mesenchymal stem cells in epilepsy: Possible treatments and future perspectives: Int J Mol Sci, 2020; 21(24); 9683
28. Wang S, Hu S, Zhang Q, Mesenchymal stem cells stabilize atherosclerotic vulnerable plaque by anti-inflammatory properties: PloS One, 2015; 10(8); e0136026
29. Riazifar M, Mohammadi MR, Pone EJ, Stem cell-derived exosomes as nanotherapeutics for autoimmune and neurodegenerative disorders: ACS Nano, 2019; 13(6); 6670-88
30. Burt R, Fassas A, Snowden J, Collection of hematopoietic stem cells from patients with autoimmune diseases: Bone Marrow Transplant, 2001; 28; 1-12
31. Tyndall A, Gratwohl A, Haemopoietic stem and progenitor cells in the treatment of severe autoimmune diseases: Ann Rheum Dis, 1996; 55(3); 149-51
32. Tsukamoto H, Nagafuji K, Horiuchi T, A phase I–II trial of autologous peripheral blood stem cell transplantation in the treatment of refractory autoimmune disease: Ann Rheum Dis, 2006; 65; 508-14
33. Pastore I, Assi E, Ben Nasr M, Hematopoietic stem cells in type 1 diabetes: Front Immunol, 2021; 12; 694118
34. Madani S, Amanzadi M, Aghayan HR, Investigating the safety and efficacy of hematopoietic and mesenchymal stem cell transplantation for treatment of T1DM: A systematic review and meta-analysis: Syst Rev, 2022; 11; 82
35. Mesples A, Majed N, Zhang Y, Hu X, Early immunotherapy using autologous adult stem cells reversed the effect of anti-pancreatic islets in recently diagnosed type 1 diabetes mellitus: Preliminary results: Med Sci Monit, 2013; 19; 852-57
36. Achenbach P, Bonifacio E, Koczwara K, Ziegler A-G, Natural history of type 1 diabetes: Diabetes, 2005; 54(Suppl 2); S25-31
37. Palmer JP, C-peptide in the natural history of type 1 diabetes: Diabetes Metab Res Rev, 2009; 25(4); 325-28
38. Wild S, Roglic G, Green A, Global Prevalence of Diabetes. Estimates for the year 2000 and projections for 2030: Diabetes Care, 2004; 27; 1047-53
39. Gu Y, Gong C, Peng X, Autologous hematopoietic stem cell transplantation and conventional insulin therapy in the treatment of children with newly diagnosed type 1 diabetes: Long term follow-up: Chin Med J (Engl), 2014; 127(14); 2618-22
40. Izadi M, Sadr Hashemi Nejad A, Moazenchi M, Mesenchymal stem cell transplantation in newly diagnosed type-1 diabetes patients: A phase I/II randomized placebo-controlled clinical trial: Stem Cell Res Ther, 2022; 13; 264
41. Heusch G, Schulz R, Hibernating myocardium: A review: J Mol Cell Cardiol, 1996; 28(12); 2359-72
In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387