07 January 2023: Clinical Research
Serum Vitamin D Levels, Disease Activity Score-28 for Rheumatoid Arthritis with C-Reactive Protein (DAS28-CRP), and Cardiac Remodeling Determined by Ventricular Dimensions and Left Atrium Diameter in Patients with Rheumatoid Arthritis: A Prospective Observational Study
Andrea A.P. Batista1ABCDEF, Taline Lazzarin1ABCDEF*, Filipe W.L. Pereira1BC, Antonio Baccaro1B, Oswaldo M. Rocha1B, Karina L. Narimatsu2B, Juli T. Souza1B, Pamela N. Modesto2B, Silméia Garcia Zanati Bazan1B, Suzana E. Tanni1CD, Marina P. Okoshi1DEG, Bertha F. Polegato2E, Sergio A.R. PaivaDOI: 10.12659/MSM.938989
Med Sci Monit 2023; 29:e938989
Abstract
BACKGROUND: Rheumatoid arthritis (RA) can cause extra-articular manifestations, and the myocardium can be a target. This study aimed to describe structural and functional cardiac echocardiographic variables in RA patients and to evaluate whether vitamin D (VD) levels and inflammation markers, evaluated by Disease Activity Score-28 for Rheumatoid Arthritis with C-reactive protein (DAS28-CRP), are associated with cardiac remodeling (CR) in this population.
MATERIAL AND METHODS: This prospective observational study evaluated 90 patients with RA in Botucatu University Hospital wards from 2014 to 2017. Clinical data were recorded, including demographic information, comorbidities, length of disease, and treatment type. Serum VD and C-reactive protein levels were measured, and the DAS28-CRP was calculated. A transthoracic echocardiography study was performed. The outcome evaluated was CR. This parameter was assessed by left ventricular geometric patterns and left atrium diameter.
RESULTS: We evaluated 90 RA patients. The mean age was 52.9±10.8 years, and 17.8% were male. The length of the disease was 96 (60-180) months. Serum VD levels were 30.7±10.4 ng/mL and the DAS28 was 2.7±0.9. Regarding the CR parameters, 56.7% had altered left ventricular geometric patterns and 25.8% had enlargement of left atrium diameter. Even in multivariate analysis, the left ventricular geometric patterns were not associated with the VD levels and the inflammation marker used. However, sufficient VD levels protect from left atrium enlargement (OR: 0.905; IC 95%: 0.843-0.973; P=0.007).
CONCLUSIONS: Low serum vitamin D values, but not inflammation, are associated with CR in patients with RA.
Keywords: 25-hydroxyvitamin D, Arthritis, Rheumatoid, Echocardiography, Heart Atria, Humans, Male, Adult, Middle Aged, Female, C-Reactive Protein, Ventricular Remodeling, Inflammation, Vitamin D, Severity of Illness Index
Background
Rheumatoid arthritis (RA) is a chronic systemic inflammatory disease characterized by joint destruction [1]. However, extra-articular manifestations are common and usually responsible for reducing RA patients’ survival. Compared with the general population, RA patients have a 1.5-fold increase in mortality rate due to cardiovascular (CV) events [2]. The primary CV compromise is due to ischemic heart disease [3]. However, RA can also silently affect myocardial tissue independent of vascular disease [4]. The relationship between RA and cardiac structural changes suggests that some RA patients have the cardiac remodeling (CR) process [3–6]. Cardiac remodeling is characterized as molecular, cellular, and interstitial changes that manifest clinically as changes in size, mass, geometry, and function of the heart [5].
Some cardiac alterations that have been described in RA patients are left ventricular (LV) hypertrophy [6–8] and atrial dilatation [9]. Hypertrophy occurs through an increase in the mass of the LV. The evaluation of LV mass combined with the relative wall thickness (RWT) characterizes 4 geometric echocardiographic patterns: standard, concentric remodeling, concentric hypertrophy, and eccentric hypertrophy. Although alterations in ventricular geometry may be clinically asymptomatic, they significantly enhance the risk of cardiac failure and death [10].
One of the potential mechanisms for cardiac structural changes in RA patients is the pro-inflammatory status [11]. Cytokines such as IL-6 and tumor necrosis factor (TNF)-α, known to exert crucial roles in RA, are associated with cardiac hypertrophy. IL-6 participates in the Janus kinases (JAK)/signal transducer and activator of transcription proteins (STAT) pathways of inflammation and cardiac hypertrophy signaling. In experimental studies, TNF-α induced cardiac hypertrophy and fibrosis, dilated cardiomyopathy, and cardiac dysfunction [3,6,11,12]. Importantly, inflammation is associated with RA activity. This activity can be measured by different parameters, including Disease Activity Score-28 for Rheumatoid Arthritis with C-reactive protein (DAS28-CRP) [13,14].
Another potential mechanism of cardiac alterations in RA is vitamin D (VD) deficiency. The role of vitamin D in cardiovascular disease (CVD) is an emerging topic. This nutrient has a well-described relationship with cardiac structure and function, but it still requires further clinical investigation [15,16].
Although the relationship between VD deficiency and inflammation with cardiac changes is already known, no study has yet evaluated the association between CR, VD status, and inflammation in RA patients. Therefore, this study aimed to describe structural and functional cardiac echocardiographic variables in rheumatoid arthritis patients and to evaluate whether VD levels and inflammation (evaluated by DAS28-CRP score) are associated with CR in RA patients.
Material and Methods
ETHICS STATEMENT:
The study was approved by the Ethics Committee of Botucatu Medical School, São Paulo State University, UNESP (protocol: 4135-2012). Written informed consent was obtained from all patients before their inclusion.
STUDY DESIGN:
This prospective observational study was conducted with patients diagnosed with RA and followed by the Rheumatology Service from Botucatu Medical School from 2014 until 2017. The sample size was calculated using the expected difference in the means of the LV mass index (LVMI) from RA and control patients, which was 8.6, and the standard deviation was 15.5 [8]. Considering α=0.05 and the desired power of 80%, the estimated sample size was 53 patients. Considering the multiple factors that can influence CR patterns, the sample size was raised to 90 patients. All patients met the 2010 American College of Rheumatology/European League Against Rheumatism criteria for RA to be included [17]. The exclusion criteria were previous coronary artery disease, heart failure symptoms or signs, chest pain, non-sinus cardiac rhythm, and renal and liver failure history.
VITAMIN D AND C-REACTIVE PROTEIN EVALUATION:
A baseline medical history was recorded, and a blood sample was collected. The following clinical variables were analyzed: age, gender, previous diseases, smoking status, RA activity, length of disease, and treatment. Blood samples were taken to determine the 25-hydroxyvitamin D levels by chemiluminescence method [18]; in our laboratory, the normal reference range is 9.5 to 55.5 ng/dL. C-reactive protein (CRP) was assessed using the dry chemistry method.
RA ACTIVITY EVALUATION:
Inflammation was evaluated by grade disease activity calculated by DAS28-CRP score [19]. DAS28-CRP was calculated using tender joint count, swollen joint count, the visual analog scale of the patient’s global health, and CRP measurement. The DAS28 cut-off points were considered remission lower than 2.6, low disease activity between 2.6 and 3.2, moderate disease activity between 3.2 and 5.1, and high disease activity if higher than 5 [13,14].
ECHOCARDIOGRAPHIC EVALUATION:
Cardiac structural and functional evaluation was performed by Doppler transthoracic echocardiogram. Exams were performed by the same researcher using General Electric (GE) Vivid S6 equipment, with a 2.0–3.5 MHz multifrequency ultrasonic transducer and image recording system [20].
Echocardiographic alterations describe various CR findings as previously described changes in size, mass, geometry, and function of the heart [5]. As dysfunction may not be present at early stages, we evaluated cardiac remodeling through morphological parameters, ventricular geometry, and LA dimension [5,21]. Regarding the geometry pattern, 4 types were defined based on LVMI and RWT. The normal value of LVMI was less or equal to 95 g/m2 for women and 115 g/m2 for men, and normal RWT was less or equal to 0.42. Thus, the 4 types are: normal, concentric remodeling if normal LVMI and high RWT, concentric hypertrophy if high LVMI and high RWT, and eccentric hypertrophy if high LVMI and normal RWT [20]. We used the LA diameter adjusted by body surface area, which was considered increased when its diameter was more than 2.3 cm/m2 [22].
STATISTICAL ANALYSIS:
All statistical analyses were performed with SigmaPlot software for Windows v12.0 (Systat Software, Inc., San Jose, CA, USA). Data are expressed as the percentage, mean values, standard deviation, or medians, with 25% and 75% percentiles, where appropriate. Comparisons between 2 groups for continuous variables were performed using the
Results
GEOMETRIC PATTERN:
Table 1 shows the association of demographic and echocardiographic variables with left ventricular geometric patterns (LVGP). The factors related to this outcome were age, arterial hypertension, and diastolic dysfunction. Echocardiographic parameters such as LVMI, LV diastolic diameter (LVDD), and RWT were significantly associated with geometric patterns. However, because these parameters are used to classify the type of geometric alteration, they were not used in the multiple logistic regression described in Table 2. As shown in Table 2, VD and DAS28-CRP had no association with the alteration of LVGP.
LEFT ATRIUM ENLARGEMENT:
The association between demographic and echocardiographic variables with LA enlargement is shown in Table 3. The factors associated were VD level and LVMI. VD serum levels in patients with LA enlargement were 6 ng/mL smaller than in patients with normal atrial diameter. In multivariate analysis (Table 4), adjusted by all factors with P value <0.1 (age, smoking, and LVMI), the association between VD and LA diameter was maintained (OR: 0.905; IC 95%: 0.843–0.973; P=0.007).
Discussion
This study’s initial objective was to describe the echocardiographic parameters in RA patients. Summarizing our finding, high-frequency alterations compatible with CR were observed. In our study, more than 50% of patients had altered ventricular geometry. Of those, more than 95% had concentric remodeling and concentric hypertrophy patterns. Ventricular geometry alteration, even asymptomatic, is a marker of progression to heart failure and death [5,23], reinforcing the importance of identifying preclinical echocardiographic alterations.
LA enlargement was found in 27% of our patients and can be considered an early sign of CR and diastolic dysfunction. A study of a cohort of more than 340 hypertensive patients showed that LA enlargement occurred before left ventricular enlargement and that LA measurement can be an early marker for heart failure [24].
Another study objective was to assess the possible correlation of CR findings with the inflammatory status of the patients (assessed by the DAS28-CRP score) and with serum concentrations of VD. Inflammatory status is important because inflammation is an essential mechanistic pathway in CR pathophysiology [25], and VD is an easily measured marker that is increasingly documented to be associated with cardiovascular diseases.
In patients with ventricular geometry alterations, age and arterial hypertension were the only positive associations observed. Evidence indicates a close relationship between senescence and CR, but age is a non-modifiable intrinsic factor [26]. On the other hand, hypertension is known to influence the CR process, which has been demonstrated to be prevalent in patients with RA. In our study, 45% were hypertensive. Hypertension in RA patients may be increased due to the inflammatory process that can increase blood pressure [27], and the therapy itself when using corticosteroids.
We found no association between ventricular geometry alteration with inflammatory activity or VD levels. Unlike our findings, Midtbo et al [11] found that patients with active RA had a higher RWT than patients with RA remission, assessed in the present study using the Simple Disease Activity Index (SDAI) score. One explanation of these results is that the mean DAS28-CRP score of our patients was lower than in the study discussed above, reflecting lower disease activity in our sample. Another explanation for this discrepancy may be the different scores used to assess the inflammatory state (SDAI versus DAS28-CRP). Despite having a good correlation at initial assessment, recent studies have shown that this correlation decreases over time [28]. In addition, a large observational cohort study in Japan showed that DAS28-CRP underestimates disease activity and overestimates response criteria compared to DAS28 erythrocyte sedimentation rate [19].
Regarding atrial assessment, age and smoking participated in multivariate analysis because the
Despite finding no association with the DAS28-CRP score during the atrial evaluation, an association with VD levels was found. The role of vitamin D role in calcium homeostasis is well established, but its deficiency is emerging as a new risk factor for CVD. Vitamin D deficiency is prevalent in 5.9–13% of the population [32]. In our study, the mean of VD was 30.73 ng/dL (±10.42), and when evaluated separately, approximately 36% had values compatible with insufficiency and 11% with deficiency of this nutrient.
Previous studies have established VD as a critical cardiac structure and function regulator. Detecting a nuclear VD receptor in endothelial cells and cardiomyocytes suggests direct involvement of VD in the development and progression of cardiovascular disease [33]. At the practical level, multiple pathways through which VD can act have been described, including inflammatory regulation and CR attenuation [34]. In clinical scenarios, several studies have reported a close association between low levels of VD and CR-associated diseases [33].
However, despite growing evidence, VD supplementation in CVD is still controversial. Some studies suggest that VD supplementation attenuates human ventricular remodeling [35]. However, the toxicity risk is a problem. Despite playing an essential role in the CV system, VD appears to exert a biphasic “dose-response” curve in cardiovascular pathophysiology and can cause deleterious effects, even at non-toxic doses, as recently demonstrated in experimental models [36].
We should consider some limitations of this study. We only included patients from a single medical center, with a small number of patients. In addition, RA patients can have a range of associated cardiac diseases, including pericardial and myocardial disease, which could have affected the results [37]. However, we emphasize the importance of VD in CR in patients with rheumatoid arthritis and encourage the development of trials with VD supplementation in patients with RA.
Conclusions
In conclusion, low serum vitamin D values, but not inflammation, are associated with cardiac remodeling in patients with RA.
Tables
Table 1. Demographic and echocardiographic data of 90 patients with rheumatoid arthritis according to altered geometry patterns.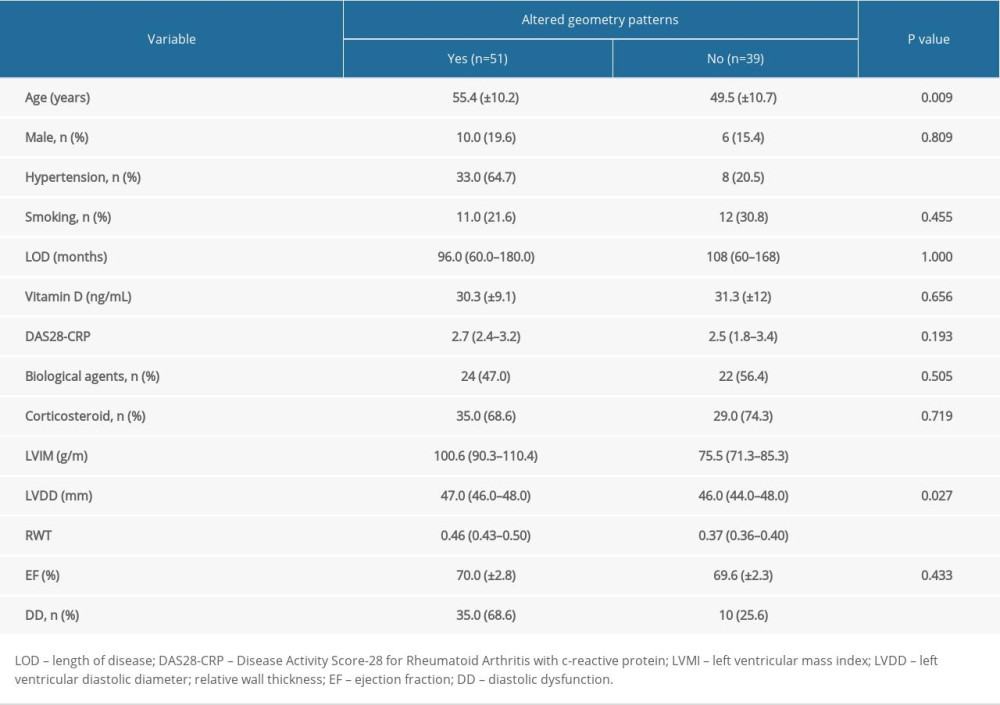 Table 2. Logistic regression model for the prediction of association between altered geometry patterns, vitamin D levels, DAS28-CRP, and biological agents.
Table 2. Logistic regression model for the prediction of association between altered geometry patterns, vitamin D levels, DAS28-CRP, and biological agents.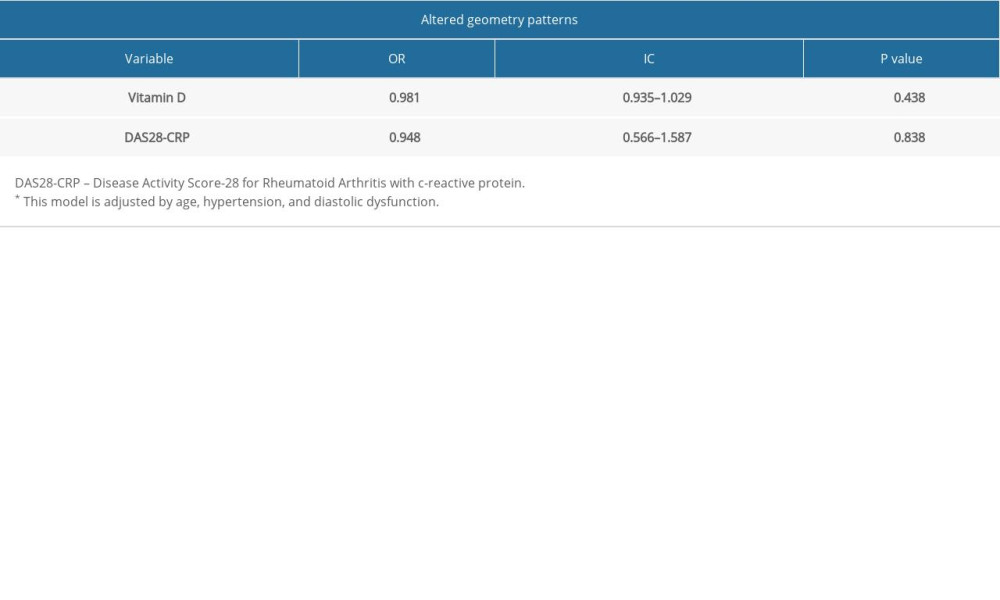 Table 3. Demographic and echocardiographic data of 90 patients with rheumatoid arthritis according to left atrium enlargement.
Table 3. Demographic and echocardiographic data of 90 patients with rheumatoid arthritis according to left atrium enlargement.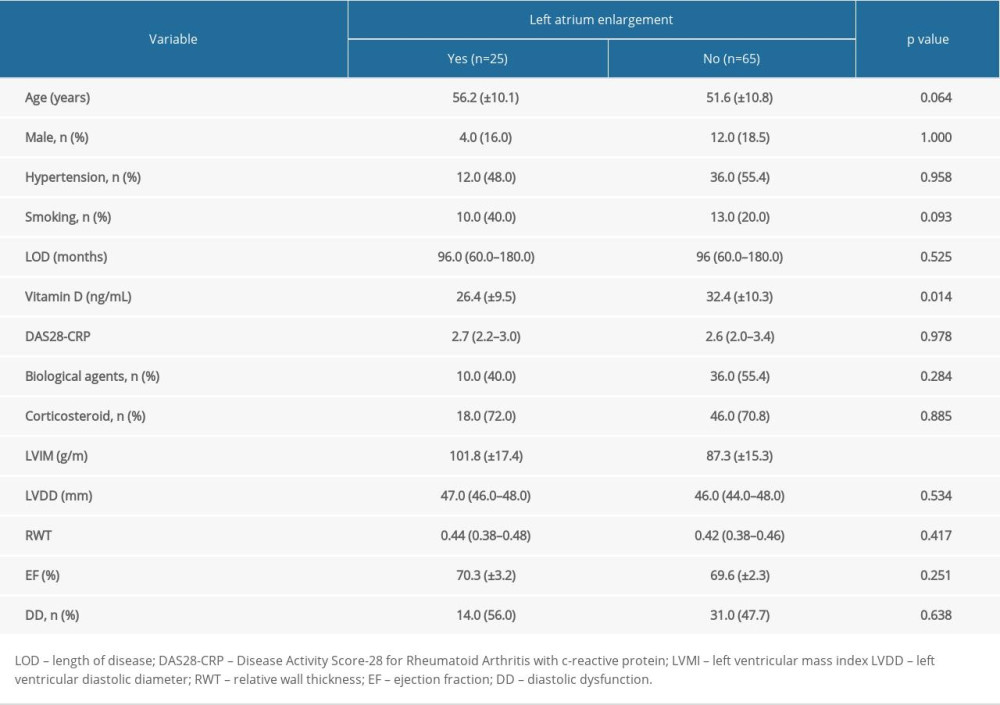 Table 4. Logistic regression model for the prediction of association between Left atrium enlargement, vitamin D levels, DAS28-CRP, and biological agents.
Table 4. Logistic regression model for the prediction of association between Left atrium enlargement, vitamin D levels, DAS28-CRP, and biological agents.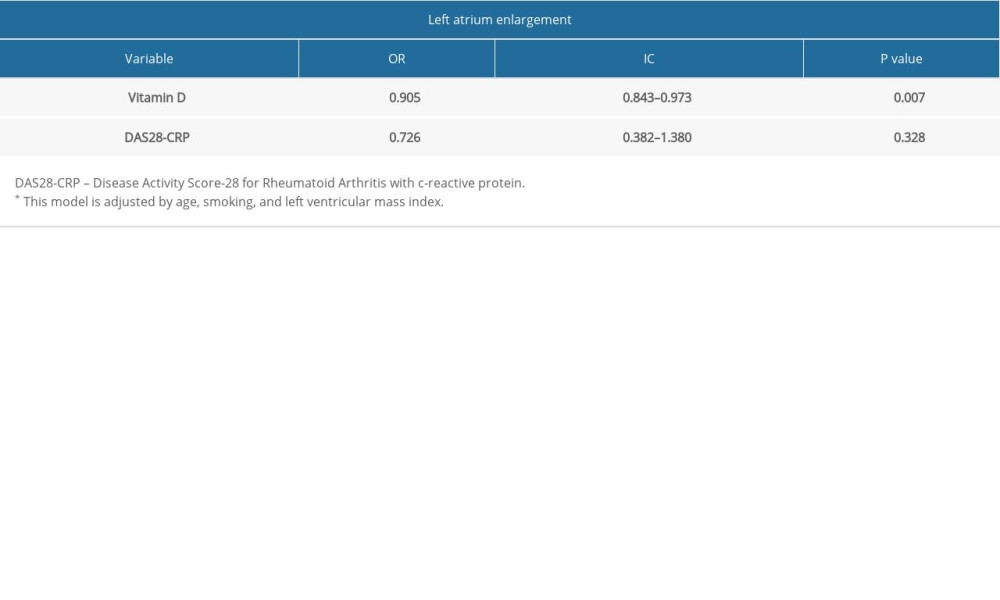
References
1. Schau T, Gottwald M, Arbach O, Increased prevalence of diastolic heart failure in patients with rheumatoid arthritis correlates with active disease, but not with treatment type: J Rheumatol, 2015; 42(11); 2029-37
2. Aviña-Zubieta JA, Choi HK, Sadatsafavi M, Risk of cardiovascular mortality in patients with rheumatoid arthritis: A meta-analysis of observational studies: Arthritis Rheum, 2008; 59(12); 1690-97
3. Kozera L, Andrews J, Morgan AW, Cardiovascular risk and rheumatoid arthritis – the next step: Differentiating true soluble biomarkers of cardiovascular risk from surrogate measures of inflammation: Rheumatology (Oxford), 2011; 50; 1944-54
4. Prasad M, Hermann J, Gabriel SE, Cardiorheumatology: Cardiac involvement in systemic rheumatic disease: Nat Rev Cardiol, 2015; 12; 168-76
5. Azevedo PS, Polegato BF, Minicucci MF, Cardiac remodeling: Concepts, clinical impact, pathophysiological mechanisms and pharmacologic treatment: Arq Bras Cardiol, 2016; 106(1); 62-69
6. Daien CI, Fesler P, du Cailar G, Etanercept normalises left ventricular mass in patients with rheumatoid arthritis: Ann Rheum Dis, 2013; 72; 881-87
7. Schau T, Gottwald M, Arbach O, Increased prevalence of diastolic heart failure in patients with rheumatoid arthritis correlates with active disease but not with treatment type: J Rheumatol, 2015; 42; 2029-37
8. Rudominer RL, Roman MJ, Devereux RB, Independent association of rheumatoid arthritis with increased left ventricular mass but not with reduced ejection fraction: Arthritis Rheum, 2009; 60; 22-29
9. Dawson JK, Goodson NG, Graham DR, Lynch MP, Raised pulmonary artery pressures measured with Doppler echocardiography in rheumatoid arthritis patients: Rheumatology (Oxford), 2000; 39(12); 1320-25
10. Sehgal S, Drazner MH, Left ventricular geometry: Does shape matter?: Am Heart J, 2007; 153; 153-55
11. Midtbo H, Gerdts E, Kvien TK, Disease activity and left ventricular structure in patients with rheumatoid arthritis: Rheumatology (Oxford), 2015; 54; 511-19
12. Lazúrová I, Tomáš L’, Cardiac impairment in rheumatoid arthritis and influence of anti-TNFα treatment: Clin Rev Allergy Immunol, 2017; 52(3); 323-32
13. Wells G, Becker JC, Teng J, Validation of the 28-joint Disease Activity Score (DAS28) and European League Against Rheumatism response criteria based on C-reactive protein against disease progression in patients with rheumatoid arthritis, and comparison with the DAS28 based on erythrocyte sedimentation rate: Ann Rheum Dis, 2009; 68(6); 954-60
14. Inoue E, Yamanaka H, Hara M, Comparison of Disease Activity Score (DAS)28-erythrocyte sedimentation rate and DAS28-C-reactive protein threshold values: Ann Rheum Dis, 2007; 66(3); 407-9
15. Wen H, Baker JF, Vitamin D, immunoregulation, and rheumatoid arthritis: J Clin Rheumatol, 2011; 17(2); 102-7
16. Lavie CJ, Lee JH, Milani RV, Vitamin D and cardiovascular disease: Will it live up to its hype?: J Am Coll Cardiol, 2011; 58(15); 1547-56
17. Van der Linden MP, Knevel R, Huizinga TW, van der Helm-van Mil AH, Classification of rheumatoid arthritis: Comparison of the 1987 American College of Rheumatology criteria and the 2010 American College of Rheumatology/European League Against Rheumatism criteria: Arthritis Rheum, 2011; 63; 37-42
18. Hyppönen E, Turner S, Cumberland P, Serum 25-hydroxyvitamin D measurement in a large population survey with statistical harmonization of assay variation to an international standard: J Clin Endocrinol Metab, 2007; 92(12); 4615-22
19. Matsui T, Kuga Y, Kaneko A, Disease Activity Score 28 (DAS28) using C-reactive protein underestimates disease activity and overestimates EULAR response criteria compared with DAS28 using erythrocyte sedimentation rate in a large observational cohort of rheumatoid arthritis patients in Japan: Ann Rheum Dis, 2007; 66; 1221-26
20. Lang RM, Badano LP, Mor-Avi V, Recommendations for cardiac chamber quantification by echocardiography in adults: An update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging: J Am Soc Echocardiogr, 2015; 28(1); 1-39
21. Polovina MM, Coats A, Seferovic P, Is left atrium the best kept secret of the heart? Left atrial dilatation and cardiovascular outcomes: Heart, 2019; 105(24); 1848-49
22. Nagueh SF, Smiseth OA, Appleton CP, Recommendations for the evaluation of left ventricular diastolic function by echocardiography: An update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging: Eur Heart J Cardiovasc Imaging, 2016; 17; 1321-60
23. Milani RV, Drazner MH, Lavie CJ, Progression from concentric left ventricular hypertrophy and normal ejection fraction to left ventricular dysfunction: Am J Cardiol, 2011; 108; 992-96
24. Su G, Cao H, Xu S, Left atrial enlargement in the early stage of hypertensive heart disease: A common but ignored condition: J Clin Hypertens (Greenwich), 2014; 16(3); 192-97
25. Wu QQ, Xiao Y, Yuan Y, Mechanisms contributing to cardiac remodeling: Clin Sci (Lond), 2017; 131(18); 2319-45
26. Shimizu I, Minamino T, Cellular senescence in cardiac diseases: J Cardiol, 2019; 74(4); 313-19
27. Jagpal A, Navarro-Millán I, Cardiovascular co-morbidity in patients with rheumatoid arthritis: A narrative review of risk factors, cardiovascular risk assessment and treatment: BMC Rheumatol, 2018; 11(2); 10
28. Dhaon P, Das SK, Srivastava R, Dhakad U, Performances of Clinical Disease Activity Index (CDAI) and Simplified Disease Activity Index (SDAI) appear to be better than the gold standard Disease Assessment Score (DAS-28-CRP) to assess rheumatoid arthritis patients: Int J Rheum Dis, 2018; 21(11); 1933-39
29. Minicucci MF, Azevedo PS, Polegato BF, Cardiac remodeling induced by smoking: Concepts, relevance, and potential mechanisms: Inflamm Allergy Drug Targets, 2012; 11(6); 442-47
30. Seko Y, Kato T, Haruna T, Association between atrial fibrillation, atrial enlargement, and left ventricular geometric remodeling: Sci Rep, 2018; 8(1); 6366
31. Lavie CJ, Lee JH, Milani RV, Vitamin D and cardiovascular disease will it live up to its hype?: J Am Coll Cardiol, 2011; 58; 1547-56
32. Cashman KD, Vitamin D deficiency: Defining, prevalence, causes, and strategies of addressing: Calcif Tissue Int, 2020; 106(1); 14-29
33. Cosentino N, Campodonico J, Milazzo V, Vitamin D and cardiovascular disease: Current evidence and future perspectives: Nutrients, 2021; 13(10); 3603
34. Kim IM, Norris KC, Artaza JN, Vitamin D and cardiac differentiation: Vitam Horm, 2016; 100; 299-320
35. Zhao JD, Jia JJ, Dong PS, Effect of vitamin D on ventricular remodelling in heart failure: A meta-analysis of randomised controlled trials: BMJ Open, 2018; 8(8); e020545
36. Santos PPD, Rafacho BPM, Gonçalves AF, Vitamin D Supplementation induces cardiac remodeling in rats: Association with thioredoxin-interacting protein and thioredoxin: Arq Bras Cardiol, 2021; 116(5); 970-78
37. Buleu F, Sirbu E, Caraba A, Dragan S, Heart involvement in inflammatory rheumatic diseases: A systematic literature review: Medicina (Kaunas), 2019; 55(6); 249
Tables
 Table 1. Demographic and echocardiographic data of 90 patients with rheumatoid arthritis according to altered geometry patterns.
Table 1. Demographic and echocardiographic data of 90 patients with rheumatoid arthritis according to altered geometry patterns. Table 2. Logistic regression model for the prediction of association between altered geometry patterns, vitamin D levels, DAS28-CRP, and biological agents.
Table 2. Logistic regression model for the prediction of association between altered geometry patterns, vitamin D levels, DAS28-CRP, and biological agents. Table 3. Demographic and echocardiographic data of 90 patients with rheumatoid arthritis according to left atrium enlargement.
Table 3. Demographic and echocardiographic data of 90 patients with rheumatoid arthritis according to left atrium enlargement. Table 4. Logistic regression model for the prediction of association between Left atrium enlargement, vitamin D levels, DAS28-CRP, and biological agents.
Table 4. Logistic regression model for the prediction of association between Left atrium enlargement, vitamin D levels, DAS28-CRP, and biological agents. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








