02 February 2023: Database Analysis
Study of 52 Pediatric Patients Who Required 78 Hospital Admissions for Cerebral Venous Sinus Thrombosis from 2013 to 2020 Using Data from the Polish National Health Fund Registry
Beata Sarecka-Hujar1ADEF*, Mariusz Gujski2DEF, Michał Maluchnik34BCE, Jarosław Bartosiński5DEF, Agnieszka Chłoń-Domińczak6ADEF, Ilona Kopyta7DEF, Dorota Raczkiewicz8ACDEGDOI: 10.12659/MSM.939176
Med Sci Monit 2023; 29:e939176
Abstract
BACKGROUND: Cerebral venous sinus thrombosis (CVST) in children is a rare disease with a complex, multifactorial etiopathogenesis. The Polish National Health Fund (NHF) Registry [Narodowy Fundusz Zdrowia (NFZ)], contains health insurance data from all 16 national provinces, or voivodeships. This study used data from the Polish NHF Registry to evaluate 52 pediatric patients who required 78 hospital admissions for CVST from 2013 to 2020.
MATERIAL AND METHODS: The data in the Polish NHF Registry were acquired based on the disease code I67.6 from the International Classification of Diseases, Tenth Revision (ICD-10), and the patients’ age (up to 18 years old).
RESULTS: We identified 78 hospitalizations of 52 pediatric patients due to CVST in Poland from 2013 to 2020 (63.5% boys and 36.5% girls, mean age 9.7±5.8 years old). The mean duration of hospitalization was 10.5±11.7 days, the mean cost of hospitalization was 3273±2191 Polish zloty (PLN). The most common age subgroup was adolescents (27%). Ten percent of patients were hospitalized in a region other than their region of residence. The duration and cost of hospitalization were positively correlated with each other (r=0.512, P<0.001). The most common type of admission was an emergency (51%), and the most common discharge was referral for further outpatient treatment (50%).
CONCLUSIONS: Polish registry data showed that from 2013 to 2020, CVST was more commonly diagnosed in male adolescents from 15 to 18 years of age who presented as emergency hospital admissions. There were regional differences in incidence and duration of hospital stay and healthcare costs between patients.
Keywords: Hospitalization, Intracranial Thrombosis, Pediatrics, Registries, Female, Adolescent, Humans, Male, Child, Child, Preschool, Poland, Sinus Thrombosis, Intracranial, Hospitals
Background
Cerebral venous sinus thrombosis (CVST) is a rare disease in the pediatric population and most often has a multifactorial background [1]. The true rate of CVST is underestimated. In the literature data, approximately 1 case of CVST is most commonly diagnosed in 100 000 children every year [1,2]. Most cases of CVST in the pediatric population concern the neonatal period and account for up to 40% of all the diagnosed cases of CVST. The frequency of CVST at the age of less than 28 days is 2.6 per 100 000 children per year [3]. Previously, such a high frequency of neonates with CVST was suggested to result from multiple risk factors in this age group [4].
Risk factors for CVST differ between neonates and older children [1,5–7]. Neonatal thrombosis can be caused by maternal factors (eg, hypertension, diabetes, inflammation of the membranes), congenital infections, birth defects, and perinatal hypoxia [5]. In turn, in infants and older children, the most important risk factors for CVST can be divided into several groups: general causes (dehydration, fever), head/neck infections, other pathologies of head and neck area (eg, injuries, treatment with radiant energy, proliferative processes, neurosurgical procedure), congenital heart defects, disorders of the hematopoietic system, kidney diseases, certain medications (corticosteroids), thrombophilia, and complicated delivery [2,3,6,7].
The diagnosis of CVST is based on the results of imaging tests; currently, the method of choice is venography by computed tomography (CT) or magnetic resonance venography (MRV) [8–10]. The treatment of CVST involves the use of anticoagulants, and recommendations in this regard are taken from the experience with adults. According to the guidelines of the American Society of Hematology, currently, in the case of venous thrombosis in newborns and children, unfractionated heparin, low-molecular-weight heparin, and oral vitamin K antagonists are recommended [11,12]. The most important long-term consequences of CVST in childhood are permanent visual impairment, seizures, and headaches. Mortality in the acute phase of the disease affects up to 10% of patients [1,13].
Therefore, the present study used data from the Polish National Health Fund Registry to evaluate 52 pediatric patients who required 78 hospital admissions for CVST from 2013 to 2020. In Poland, hospitalizations are both financed by public funds and paid directly by patients. The NHF data include only hospitalizations financed by public funds. However, CVST in children is unlikely to be treated privately. The Polish NHF registry is a useful tool in the population analysis of various disease entities, especially the rare ones, as well as in the population distributions of demographic and hospitalization-related characteristics. There is a growing amount of research analyzing various diseases based on NHF data [14–16].
Material and Methods
ETHICS STATEMENT:
Since database analysis is not considered a medical experiment according to Polish law, no formal approval of the local ethics committee was required. During the analysis, we did not have direct contact with the patients, and therefore the patients’ consent was not required. The data used for analysis were maintained by the Polish Ministry of Health and the Polish National Health Fund. The individual data were anonymous and pseudonymized and reviewed only by one of the co-authors of the present study who is an authorized employee from the Ministry of Health.
STUDY DESIGN:
This retrospective study involved individual, anonymous, and pseudonymized data from the Polish NHF concerning the patients’ hospitalizations financed by public funds in the years 2013 to 2020. The data before 2013 are incomparable due to the fact that in Poland at that time there was a different system of recording and financing hospitalizations. The formal approval for using the data was obtained from the NHF as part of the “Maps of Health Needs” project.
The inclusion criteria for the analysis and simultaneous queries to search for data in the database were as follows: I67.6 as ICD-10 code for nonpyogenic thrombosis of intracranial venous system; age up to 18 years old at admission to hospital; and hospitalization at pediatric neurology or neurosurgery wards. We used SQL language for queries and R language for grouping.
In total, in the years 2013 to 2020, 105 hospital records met the inclusion criteria (78 hospitalizations for 52 patients). Figure 1 presents the flow chart of the included 105 hospital records, which concerned 78 hospitalizations due to CVST in 52 patients.
The following data were available for each pediatric hospitalization: the regional branch of the NHF in which the patient was hospitalized, the regional branch of the NHF in which the patient was registered, age, sex, the ICD-10 classification code, date and type of admission, date and type of discharge, procedure provided, and cost of the procedure provided. Additionally, using the data from the registry, we calculated the duration of hospitalization, month and season of the year at the time of admission, and total cost of hospitalization (in Polish zloty, PLN). We also determined how many hospitalizations were done in the region other than the patient’s region of residence.
DIVISION OF THE PATIENTS ACCORDING TO AGE:
The pediatric patients with CVST hospitalized in Poland during 2013–2020 were divided into 5 subgroups according to age: (1) neonates or infants (0–12 months), (2) toddlers and preschoolers (1–6 years), (3) pre-pubertal children (7–10 years), (4) early pubertal children (11–14 years), and (5) adolescents (15–18 years).
STATISTICAL ANALYSIS:
The data were analyzed using STATISTICA 13 software (StatSoft Tulsa, OK, USA). The mean and standard deviation were estimated for numerical variables, as well as absolute numbers (n) and percentages (%) of the occurrence of items for categorical variables.
The following statistical tests were used:
A regression analysis of hospitalization cost against hospitalization duration was used.
The significance level was assumed as 0.05 in all the statistical tests.
Results
DEMOGRAPHIC CHARACTERISTICS OF PEDIATRIC PATIENTS HOSPITALIZED DUE TO CVST IN POLAND IN THE YEARS 2013 TO 2020:
Among the 52 pediatric patients hospitalized for CVST in Poland in the years 2013 to 2020, 33 were boys (63.46%) and 19 were girls (36.54%).
The mean age of children hospitalized for the first time owing to CVST during the 2013–2020 period in Poland, was 9.7±5.8 years. The distribution of the patients’ age is shown in Figure 2. The most frequent age group was adolescents (26.92%) followed by toddlers or preschoolers (21.15%), pre-pubertal children (21.15%), and early pubertal children (19.23%), while the least frequent was the group of neonates and infants (11.54%).
Table 1 presents the regions of residence where the CVST pediatric patients lived. The highest percentage lived in the Śląskie region (14 patients, 26.92%), followed by the Dolnośląskie region (9 patients, 17.31%), and the Mazowieckie region (7 patients, 13.42%). These 3 regions are the most highly populated in Poland. The region of residence of the patients hospitalized for pediatric CVST did not significantly differ between the years analyzed (P=0.920).
REGION OF HOSPITALIZATION OF PATIENTS FOR PEDIATRIC CVST IN POLAND FROM 2013 TO 2020:
Among the 78 hospitalizations for pediatric CVST registered in Poland in years 2013 to 2020, the number of annual hospitalizations differed between the analyzed years. The highest numbers of hospitalizations were registered in 2013 and 2019 (n=15 each), followed by 2015 (n=13), and 2018 (n=13), and then 2014 and 2017 (n=8 each); and the lowest numbers were registered in 2016 (n=3) and 2020 (n=4).
Table 1 also presents the regions where the pediatric patients were hospitalized for CVST. The highest percentage of CVST pediatric patients in total from 2013 to 2020 lived in and were hospitalized in 3 regions: Dolnośląskie (17% of the patients lived here and 26% of them were hospitalized here), Mazowieckie (13% and 15%, respectively), and Śląskie (27% and 32%, respectively).
The region of hospitalization of pediatric patients for CVST did not significantly differ between the years analyzed (
A total of 8 cases out of 78 hospitalizations (10%) were in a region other than the patient’s region of residence. The percentage of CVST pediatric hospitalizations in the region other than the patient’s region of residence did not differ significantly between the years analyzed (
SEASONS OF YEAR AND MONTHS AT ADMISSION TO HOSPITALS FOR PEDIATRIC CVST IN POLAND FROM 2013 TO 2020:
The same number of pediatric patients were admitted to hospitals for CVST in every season of the year (Table 2). January, July, and November were dominate months of pediatric CVST hospitalizations (9 hospitalizations each), followed by February, May, and June (8 hospitalizations each). The lowest number of hospitalizations were registered in August (2), and March and October (3 hospitalizations each). Season of the year and month at admission did not significantly differ between the years analyzed (P=0.868 and P=0.460, respectively).
TYPES OF ADMISSION AND DISCHARGE FROM HOSPITALIZATION FOR PEDIATRIC CVST IN POLAND FROM 2013 TO 2020:
The type of admission did not significantly differ between the years of hospitalization for pediatric CVST (P=0.113; Table 3). The most common admission to hospital for CVST was emergency (51%), including emergency admission as a result of a transfer by the emergency medical team (4%) and other emergency admissions (47%). Scheduled admissions on the basis of a referral accounted for 46% of all admissions, while the less common type of admission was transfer from another hospital (3%).
The type of discharge did not significantly differ between the years of hospitalization for pediatric CVST (P=0.166; Table 4). The most common type of discharge was referral for further outpatient treatment (50%), followed by completion of the therapeutic or diagnostic process (31%), while referral for further treatment in another hospital and discharge at own request were the least common types of discharges (8% and 10%, respectively). One patient died in a hospital.
DURATION AND COST OF HOSPITALIZATION FOR PEDIATRIC CVST IN POLAND FROM 2013 TO 2020:
The duration of CVST pediatric hospitalization did not differ significantly between the analyzed years (P=0.335) and lasted 10.5±11.7 days, on average (Table 5).
The cost of CVST pediatric hospitalization did not differ significantly between the analyzed years (
Among all hospitalizations, the duration and cost of pediatric CVST hospitalizations were positively correlated (r=0.512, P<0.001; Figure 3). The longer the hospitalization lasted, the higher the average costs of hospitalization. Each extra day of hospitalization increased the cost of hospitalization by 101 PLN, on average ceteris paribus (black fitted line).
The patient’s age did not correlate significantly with the duration of CVST pediatric hospitalizations (r=−0.116,
Discussion
Based on data from the Polish NHF registry, we demonstrated that in the years 2013 to 2020 CVST was more common in boys than in girls, and in adolescents (15–18 years old) than in younger children. The region of residence and the region of hospitalization due to pediatric CVST did not differ between the years analyzed. Hospitalizations outside the region of residence also did not differ significantly between the years analyzed. A total of 8 out of 78 hospitalizations (10%) were in a region other than the patient’s region of residence. The duration of CVST pediatric hospitalization lasted 11 days, on average, and the mean cost of hospitalization was about 3273 PLN. The cost of CVST pediatric hospitalization did not differ significantly between the analyzed years. As expected, we observed a positive correlation between the duration and the cost of CVST pediatric hospitalizations: the longer the hospitalization, the higher the average costs of hospitalization. In the present registry-based analysis, the most common type of admission was an emergency admission, and the most common discharge was a referral for further outpatient treatment. The number of annual hospitalizations in our study differed between the analyzed years, the highest numbers of hospitalizations were registered in 2013 and in 2019 and the lowest numbers were in 2016 and in 2020 (n=15, each vs n=3, and n=4, respectively). The small number of registered hospitalizations in 2020 may have been caused by restrictions on hospital admissions in Poland due to the COVID-19 pandemic. However, in other years there is no possible reason to explain the different numbers of admissions of pediatric patients for CVST. The season or month of the year at admission did not differ between the years.
The size of the cohort analyzed in the present study (52 pediatric patients within 8 years) is comparable to the several previously analyzed groups of CVST patients [17,18]. Vieira et al [17] studied 53 Portuguese children and adolescents with CVST within 7 years, from 2001 to 2007. Moreover, 60 CVST pediatric patients with a median age of 4.79 years old were identified within 13 years in the United States [18]. In turn, Wang et al [19] analyzed a smaller number of Chinese children with CVST (n=30), recruited within the period of 2008 to 2018. Kar et al [20] retrospectively analyzed the medical records of 29 pediatric CVST patients treated between 2011 and 2018 in Turkey. Anderst et al [21] in the International Pediatric Stroke Study enrolled 216 children with CVST (160 childhood and 56 perinatal).
In our total group, 63.5% of the pediatric patients with CVST were boys. A recent study by Rodrigues et al [22] demonstrated that 29 of 53 CVST pediatric patients (54.7%) were boys. A frequency of 53% of male patients with CVST was reported by Wang et al [19] and by Kuhle et al [23] and 56% by Heller et al [3]. Sébire et al, in a study based on 5 European registries [24], reported a similar percentage of boys with CVST (64%) to ours, which was than the Chinese and Canadian studies. On the other hand, the study from Turkey had a lower number of boys with CVST 43.5% [25]. However, the study was based on a significantly lower overall number of patients than was our study (23 vs 52, respectively). The Polish CVST patients included in the present analysis were 10 years old, on average. The most frequent age subgroup was adolescents (27%). The least frequent was the group of neonates or infants (11.5%), which is contrary to the study by deVeber et al [2], which showed 43% of patients with CVST were neonates. In the study by Kuhle et al [23], 23% of the CVST children were newborns. Contrary to our study, Wasay et al [4] analyzed children with CVST below 12 years old, out of whom 90% were younger than 12 months old (35%). On the other hand, the percentage of neonates was significantly lower in the study based on pediatric patients with CVST from Portugal (11%) [17]. The Chinese children with CVST were slightly younger than our patients (median age 8.33 years old) [13]. The study by Kalita et al [26] was performed in a group of CVST patients with ages ranging from 3 to 76 years old. Children (<18 years) in this group accounted for 17.7%, whereas 9% were elderly patients (>65 years). In this sample, almost 52% of the CVST patients were female [26].
The patients analyzed in the study were hospitalized during every season of the year with the same frequency. This may be a bit surprising since some infections, such as otitis media, suggested as a risk factor for CVST, were found to occur more often between November and April each year [27]. A study performed in young adults with CVST (mean age 36.71±12.44 years) demonstrated the highest frequency of CVST within 3 months from July to September, while the lowest frequency of CVST was from December to April [28]. In addition, the authors found a significant correlation between the average temperature in each month and the frequency of CVST occurrence. What is interesting, an extremely high percentage of women (78.31%) were observed in the study group [28] and most of them developed CVST in the winter months. In the study from Pakistan [29], infections were the common cause of CVST in children, followed by anemia and dehydration. The authors also found that fever was the most common presentation [29].
In our study, the number of pediatric CVST hospitalizations was different in each region of Poland. This could be connected with the different size of each region’s population as well as the access to referral hospitals.
Since CVST is uncommon in children, its diagnosis can be delayed, which results in an increased risk of mortality. Sebire et al [24] reported 5 deaths in CVST patients (11.9%), 3 of whom died at once and 2 died later. In turn, Javed et al [29] reported the deaths of 6.25% of children with CVST. An especially high frequency of deaths was found by Ozcan et al [25]. In our database, 1 death was noted. Previously, the data suggested that a long follow-up should be done for children with CVST since 10% to 20% of them will experience recurrent events [30].
To the best of our knowledge, the present study is the first to analyze hospital admissions for pediatric CVST based on the data from the Polish NHF, which registers all hospitalizations in Poland. Using this registry, we were able to analyze a sizeable group of pediatric patients who were hospitalized for CVST in every region/voivodship of Poland within an 8-year period. Because our analysis covers the entire population of patients with CVST, it provides valuable knowledge for clinicians about the distribution of patients by sex, age, and other factors. This offers an opportunity to expand the knowledge of CVST resulting from this single-center analysis. The data on CVST obtained from the NHF Registry in Poland are comparable with the data obtained in Europe.
The present study had some limitations. The retrospective and registry-based design of the study allowed us to analyze only the variables entered into the registry. Thus, many important clinical data, such as risk factors and treatment were not analyzed. Another limitation was the small number of patients with CVST; however, due to the rarity of the disease, we found the sample size sufficient, especially in reference to the other available data. In Poland, there is no unique registry for pediatric CVST; therefore, in the present study, the NHF database was screened using the ICD-10 I67.6 (nonpyogenic thrombosis of an intracranial venous system) code. This may have resulted in some coverage errors, such as misclassification or omission of some patients. NHF data are collected for accounting purposes and could be subjected to bias resulting from limited reporting of the patient’s situation in the facility, which concentrates mainly on financial settlements, but lacks clinical and laboratory data on the disease for which the patient is currently hospitalized and comorbidities.
Conclusions
Polish registry data showed that from 2013 to 2020, CVST was more commonly diagnosed in male adolescents between 15 and 18 years of age who presented as emergency hospital admissions. There were regional differences in incidence and duration of hospital stay and healthcare costs between patients.
Figures
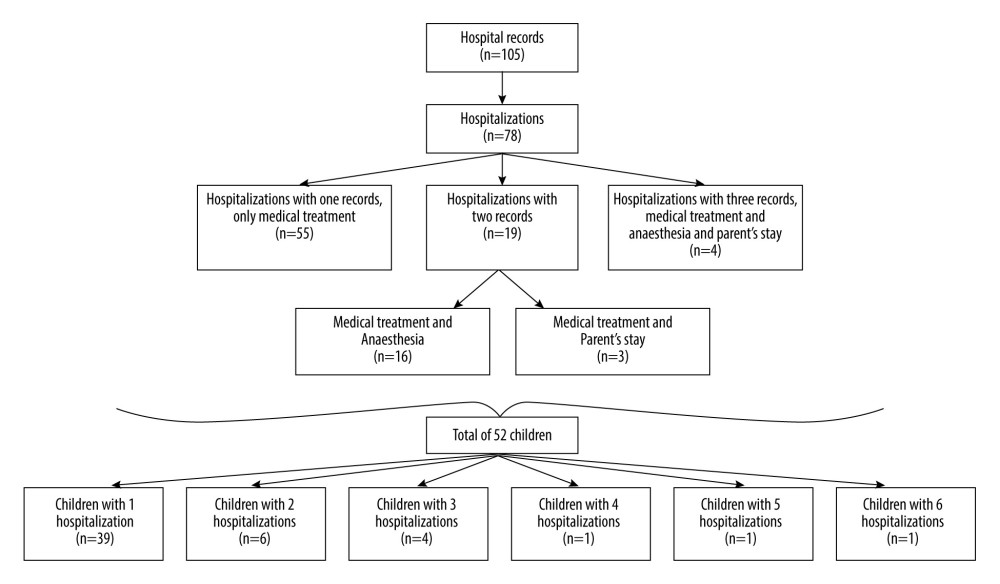 Figure 1. Flow chart presenting hospital records, hospitalizations, and patients hospitalized for pediatric cerebral venous sinus thrombosis in Poland from 2013 to 2020. Figure was made in Word, Office 365, Microsoft Corporation, USA.
Figure 1. Flow chart presenting hospital records, hospitalizations, and patients hospitalized for pediatric cerebral venous sinus thrombosis in Poland from 2013 to 2020. Figure was made in Word, Office 365, Microsoft Corporation, USA. 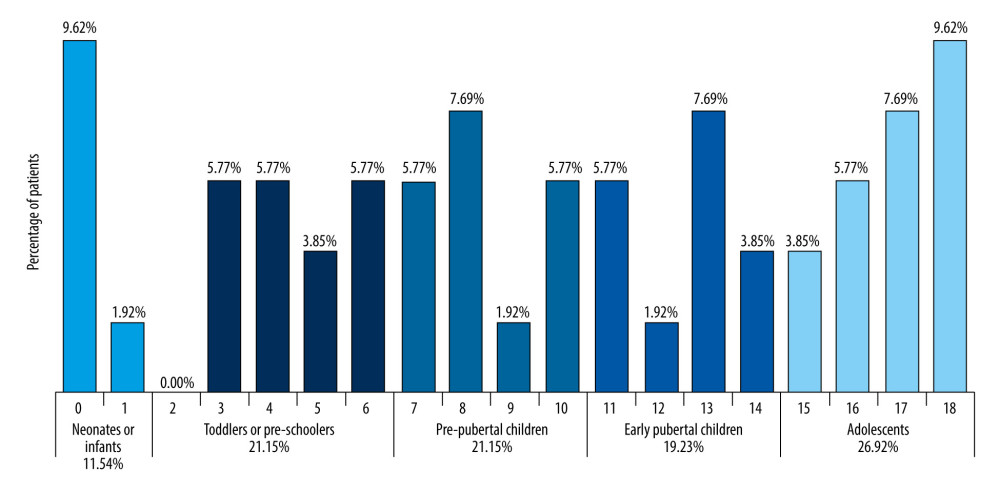 Figure 2. Age of pediatric patients hospitalized due to cerebral venous sinus thrombosis in Poland from 2013 to 2020 (n=52). Figure was made in Excel, Office 365, Microsoft Corporation, USA.
Figure 2. Age of pediatric patients hospitalized due to cerebral venous sinus thrombosis in Poland from 2013 to 2020 (n=52). Figure was made in Excel, Office 365, Microsoft Corporation, USA. 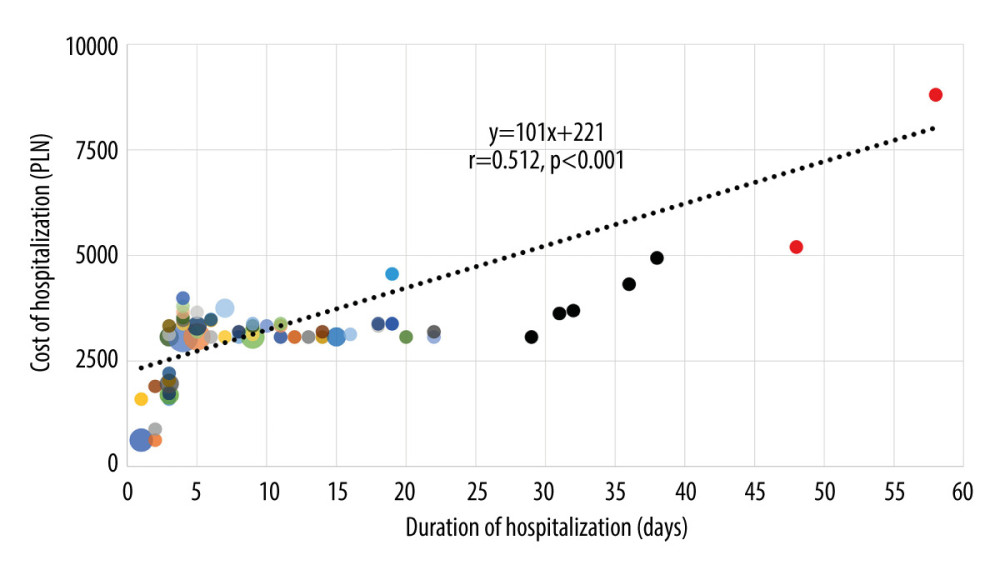 Figure 3. Scatter diagram to illustrate the cost of hospitalization against the duration of hospitalization for cerebral venous sinus thrombosis in Poland from 2013 to 2020 (n=77 hospitalizations). PLN – Polish zloty. One hospitalization is not included in the figure due to the outlier cost of 19759 PLN and the duration of hospitalization of 29 days. The size of the bubbles corresponds to the number of patients. Figure was made in Excel, Office 365, Microsoft Corporation, USA.
Figure 3. Scatter diagram to illustrate the cost of hospitalization against the duration of hospitalization for cerebral venous sinus thrombosis in Poland from 2013 to 2020 (n=77 hospitalizations). PLN – Polish zloty. One hospitalization is not included in the figure due to the outlier cost of 19759 PLN and the duration of hospitalization of 29 days. The size of the bubbles corresponds to the number of patients. Figure was made in Excel, Office 365, Microsoft Corporation, USA. Tables
Table 1. Region of residence and hospitalization of pediatric patients hospitalized for cerebral venous sinus thrombosis in Poland from 2013 to 2020.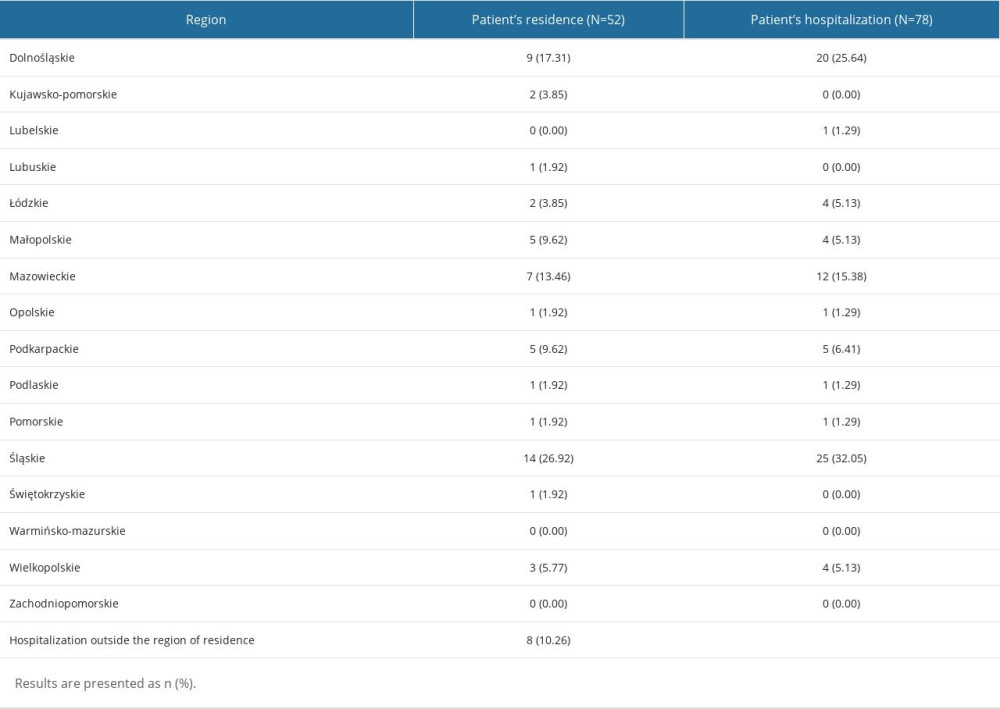 Table 2. Season of a year and month at the admission of pediatric patients hospitalized due to cerebral venous sinus thrombosis in Poland from 2013 to 2020 (n=78 hospitalizations).
Table 2. Season of a year and month at the admission of pediatric patients hospitalized due to cerebral venous sinus thrombosis in Poland from 2013 to 2020 (n=78 hospitalizations).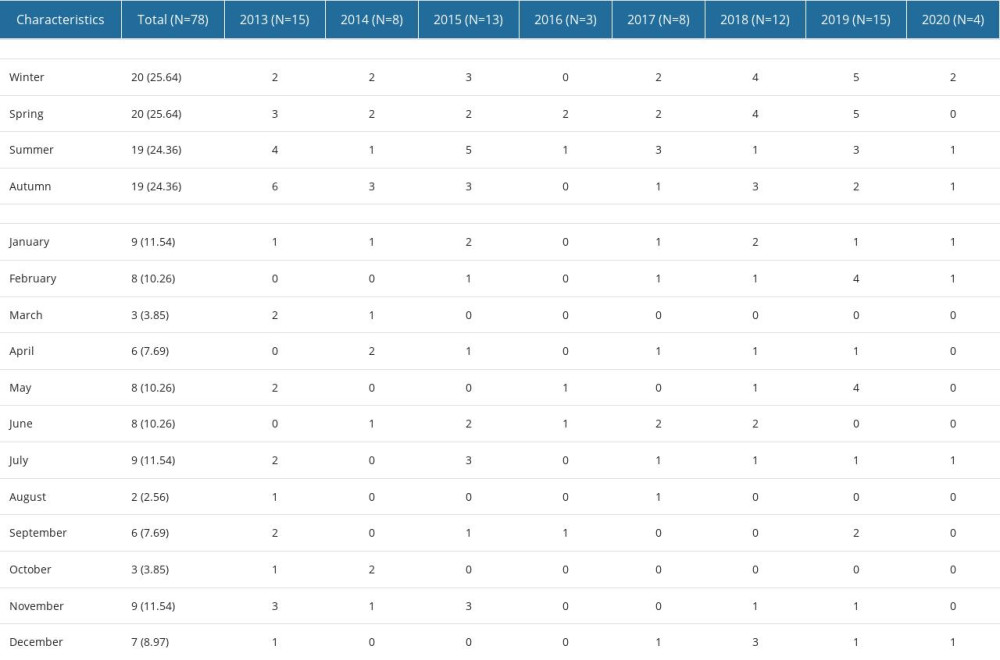 Table 3. Admission procedures of pediatric patients hospitalized due to cerebral venous sinus thrombosis in Poland from 2013 to 2020 (n=78 hospitalizations).
Table 3. Admission procedures of pediatric patients hospitalized due to cerebral venous sinus thrombosis in Poland from 2013 to 2020 (n=78 hospitalizations).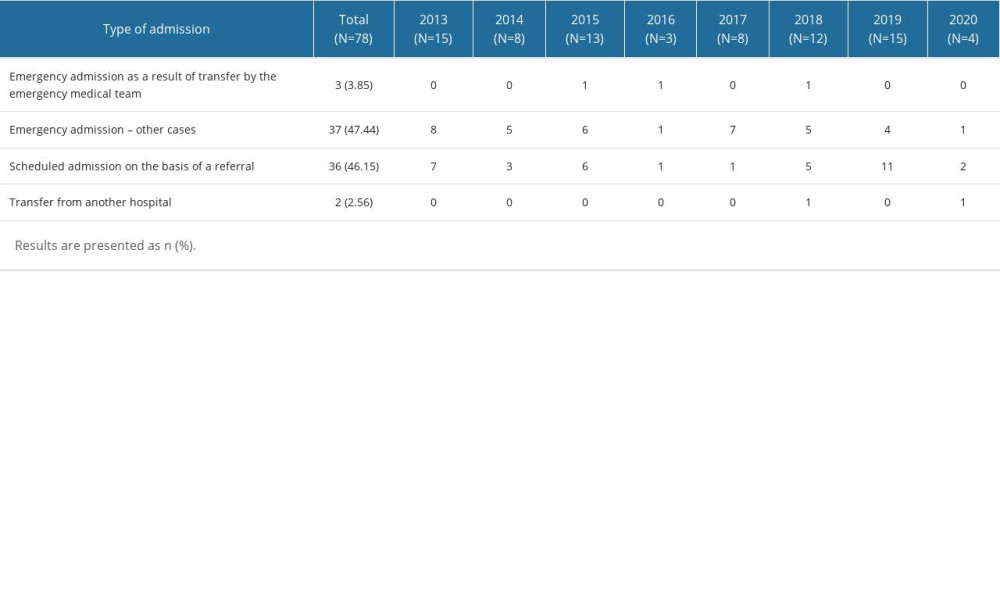 Table 4. Discharge procedures of pediatric patients hospitalized due to cerebral venous sinus thrombosis in Poland from 2013 to 2020 (n=78 hospitalizations).
Table 4. Discharge procedures of pediatric patients hospitalized due to cerebral venous sinus thrombosis in Poland from 2013 to 2020 (n=78 hospitalizations).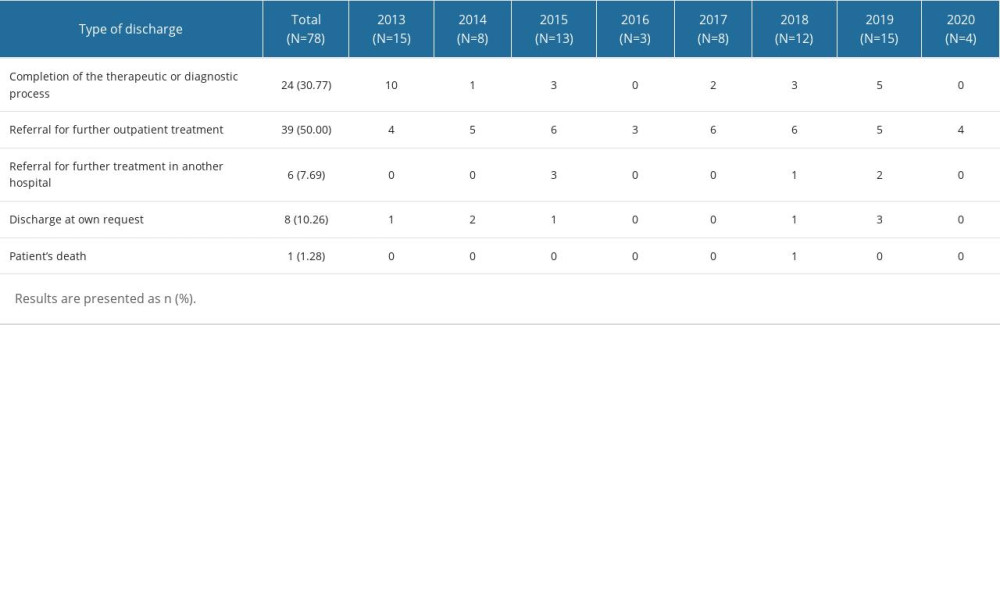 Table 5. Duration and cost of hospitalization due to cerebral venous sinus thrombosis in Poland from 2013 to 2020 (n=78 hospitalizations).
Table 5. Duration and cost of hospitalization due to cerebral venous sinus thrombosis in Poland from 2013 to 2020 (n=78 hospitalizations).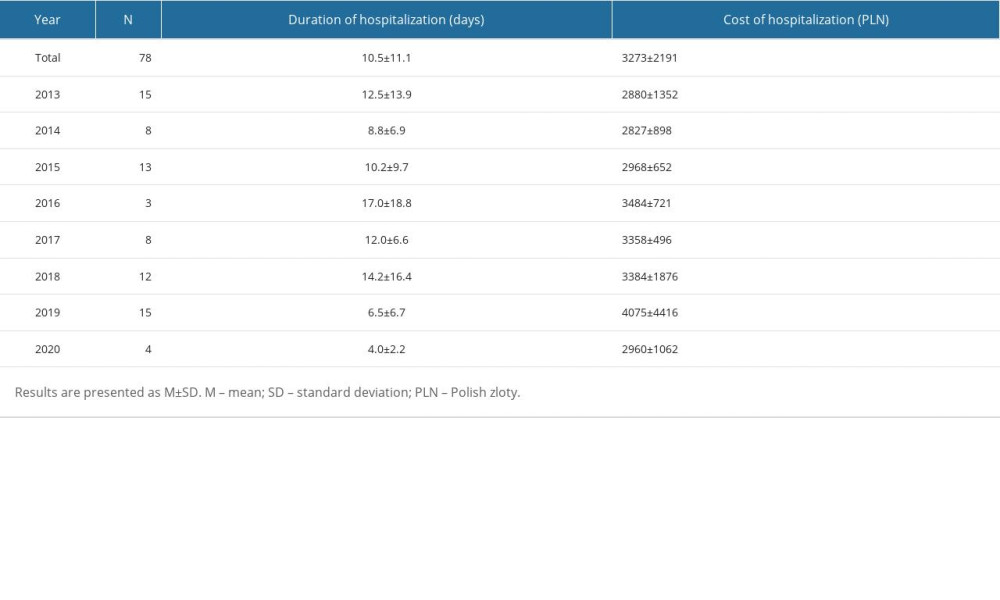
References
1. Dlamini N, Billinghurst L, Kirkham FJ, Cerebral venous sinus (sinovenous) thrombosis children: Neurosurg Clin N Am, 2010; 21(3); 511-27
2. deVeber G, Andrew M, Adams C, Canadian Pediatric Ischemic Stroke Study Group. Cerebral sinovenous thrombosis in children: N Engl J Med, 2001; 345(6); 417-23
3. Heller C, Heinecke A, Junker R, Childhood Stroke Study Group. Cerebral venous thrombosis in children: A multifactorial origin: Circulation, 2003; 108(11); 1362-67
4. Wasay M, Dai AI, Ansari M, Cerebral venous sinus thrombosis in children: A multicenter cohort from the United States: J Child Neurol, 2008; 23(1); 26-31
5. Filip C, Zonda GI, Vasilache IA, Neonatal Cerebral Sinovenous Thrombosis and the main perinatal risk factors – a retrospective unicentric study: Children (Basel), 2022; 9(8); 1182
6. Narcy L, Durand S, Grimaud M, Cerebral sinovenous thrombosis associated with head/neck infection in children: Clues for improved management: Dev Med Child Neurol, 2022; 65(2); 215-22
7. Sellers A, Meoded A, Quintana J, Risk factors for pediatric cerebral sinovenous thrombosis: A case-control study with case validation: Thromb Res, 2020; 194; 8-15
8. Zubkov AY, McBane RD, Brown RD, Rabinstein AA, Brain lesions in cerebral venous sinus thrombosis: Stroke, 2009; 40(4); 1509-11
9. Kenet G, Waldman D, Lubetsky A, Paediatric cerebral sinus vein thrombosis. A multi-center, case-controlled study: Thromb Haemost, 2004; 92(4); 713-18
10. Barnes C, Newall F, Furmedge J, Cerebral sinus venous thrombosis in children: J Paediatr Child Health, 2004; 40(1–2); 53-55
11. Monagle P, Cuello CA, Augustine C, American Society of Hematology 2018 Guidelines for management of venous thromboembolism: treatment of pediatric venous thromboembolism: Blood Adv, 2018; 2(22); 3292-316
12. Monagle P, Newall F, Management of thrombosis in children and neonates: Practical use of anticoagulants in children: Hematology Am Soc Hematol Educ Program, 2018; 2018(1); 399-404
13. deVeber GA, MacGregor D, Curtis R, Mayank S, Neurologic outcome in survivors of childhood arterial ischemic stroke and sinovenous thrombosis: J Child Neurol, 2000; 15(5); 316-24
14. Jahnz-Rozyk K, Raciborski F, Śliwczyński AM, Kłak A, Pinkas J, Anaphylaxis in Poland: The epidemiology and direct costs: Postepy Dermatol Alergol, 2017; 34(6); 573-79
15. Sarecka-Hujar B, Maluchnik M, Bartosiński J, Analysis of 622 pediatric hospitalizations due to arterial ischemic stroke in Poland – National Health Fund registry-based study from 2011 to 2020: Arch Med Sci, 2022 [Epub ahead of print]
16. Wierzba W, Wierzba A, Śliwczyński A, Analysis of National Health and Insurance Registers for all-cause mortality in patients with heart failure with and without diabetes mellitus in Poland in 2012: Med Sci Monit, 2019; 26; e921138
17. Vieira JP, Luis C, Monteiro JP, Cerebral sinovenous thrombosis in children: Clinical presentation and extension, localization and recanalization of thrombosis: Eur J Paediatr Neurol, 2010; 14(1); 80-85
18. Sellers AR, Nguyen AT, Betensky M, Risk factors for pediatric cerebral sinovenous thrombosis: A case-control study with pediatric neuroradiologist case validation: Blood, 2019; 134(Suppl 1); 2427
19. Wang XH, Zhang LM, Chai YM, Clinical characteristics and outcomes of pediatric cerebral venous sinus thrombosis: An analysis of 30 cases in China: Front Pediatr, 2019; 7; 364
20. Kar YD, Özdemir ZC, Çarman KB, Cerebral sinovenous thrombosis in children: Clinical presentation, locations, and acquired and inherited prothrombotic risk factors: Turk J Pediatr, 2021; 63(6); 1028-37
21. Anderst J, Carpenter S, Frazier T, International Pediatric Stroke Study Investigators. Subdural hemorrhage in a cohort with cerebral sinovenous thrombosis: Application to abusive head trauma: Child Abuse Negl, 2021; 117; 105119
22. Rodrigues SG, Vieira D, Bernardo F, Pediatric cerebral sinus venous thrombosis: Clinical characterization of a Portuguese cohort: Acta Neurol Belg, 2022; 122(5); 1211-18
23. Kuhle S, Mitchell L, Andrew M, Urgent clinical challenges in children with ischemic stroke: Analysis of 1065 patients from the 1-800-NOCLOTS pediatric stroke telephone consultation service: Stroke, 2006; 37(1); 116-22
24. Sébire G, Tabarki B, Saunders DE, Cerebral venous sinus thrombosis in children: Risk factors, presentation, diagnosis and outcome: Brain, 2005; 128(Pt 3); 477-89
25. Ozcan A, Canpolat M, Doganay S, Cerebral sinus venous thrombosis and prothrombotic risk factors in children: A single-center experience from Turkey: J Pediatr Hematol Oncol, 2018; 40(6); e369-e72
26. Kalita J, Misra UK, Singh RK, Do the risk factors determine the severity and outcome of cerebral venous sinus thrombosis?: Transl Stroke Res, 2018; 9(6); 575-81
27. Stockmann C, Ampofo K, Hersh AL, Seasonality of acute otitis media and the role of respiratory viral activity in children: Pediatr Infect Dis J, 2013; 32(4); 314-19
28. Salehi G, Sarraf P, Fatehi F, Cerebral venous sinus thrombosis may follow a seasonal pattern: J Stroke Cerebrovasc Dis, 2016; 25(12); 2838-43
29. Javed I, Sultan T, Rehman ZU, Yaseen MR, Clinical spectrum and outcome of cerebral venous sinus thrombosis in children: J Coll Physicians Surg Pak, 2018; 28(5); 390-93
30. Ashrafzadeh F, Akhondian J, Beiraghi Toosi M, Hashemi N, Extensive cerebral sinovenous thrombosis in a 5 year old girl, following mild dehydration. (Case report and review of literature): Iran Red Crescent Med J, 2013; 15(12); e6418
Figures
 Figure 1. Flow chart presenting hospital records, hospitalizations, and patients hospitalized for pediatric cerebral venous sinus thrombosis in Poland from 2013 to 2020. Figure was made in Word, Office 365, Microsoft Corporation, USA.
Figure 1. Flow chart presenting hospital records, hospitalizations, and patients hospitalized for pediatric cerebral venous sinus thrombosis in Poland from 2013 to 2020. Figure was made in Word, Office 365, Microsoft Corporation, USA. Figure 2. Age of pediatric patients hospitalized due to cerebral venous sinus thrombosis in Poland from 2013 to 2020 (n=52). Figure was made in Excel, Office 365, Microsoft Corporation, USA.
Figure 2. Age of pediatric patients hospitalized due to cerebral venous sinus thrombosis in Poland from 2013 to 2020 (n=52). Figure was made in Excel, Office 365, Microsoft Corporation, USA. Figure 3. Scatter diagram to illustrate the cost of hospitalization against the duration of hospitalization for cerebral venous sinus thrombosis in Poland from 2013 to 2020 (n=77 hospitalizations). PLN – Polish zloty. One hospitalization is not included in the figure due to the outlier cost of 19759 PLN and the duration of hospitalization of 29 days. The size of the bubbles corresponds to the number of patients. Figure was made in Excel, Office 365, Microsoft Corporation, USA.
Figure 3. Scatter diagram to illustrate the cost of hospitalization against the duration of hospitalization for cerebral venous sinus thrombosis in Poland from 2013 to 2020 (n=77 hospitalizations). PLN – Polish zloty. One hospitalization is not included in the figure due to the outlier cost of 19759 PLN and the duration of hospitalization of 29 days. The size of the bubbles corresponds to the number of patients. Figure was made in Excel, Office 365, Microsoft Corporation, USA. Tables
 Table 1. Region of residence and hospitalization of pediatric patients hospitalized for cerebral venous sinus thrombosis in Poland from 2013 to 2020.
Table 1. Region of residence and hospitalization of pediatric patients hospitalized for cerebral venous sinus thrombosis in Poland from 2013 to 2020. Table 2. Season of a year and month at the admission of pediatric patients hospitalized due to cerebral venous sinus thrombosis in Poland from 2013 to 2020 (n=78 hospitalizations).
Table 2. Season of a year and month at the admission of pediatric patients hospitalized due to cerebral venous sinus thrombosis in Poland from 2013 to 2020 (n=78 hospitalizations). Table 3. Admission procedures of pediatric patients hospitalized due to cerebral venous sinus thrombosis in Poland from 2013 to 2020 (n=78 hospitalizations).
Table 3. Admission procedures of pediatric patients hospitalized due to cerebral venous sinus thrombosis in Poland from 2013 to 2020 (n=78 hospitalizations). Table 4. Discharge procedures of pediatric patients hospitalized due to cerebral venous sinus thrombosis in Poland from 2013 to 2020 (n=78 hospitalizations).
Table 4. Discharge procedures of pediatric patients hospitalized due to cerebral venous sinus thrombosis in Poland from 2013 to 2020 (n=78 hospitalizations). Table 5. Duration and cost of hospitalization due to cerebral venous sinus thrombosis in Poland from 2013 to 2020 (n=78 hospitalizations).
Table 5. Duration and cost of hospitalization due to cerebral venous sinus thrombosis in Poland from 2013 to 2020 (n=78 hospitalizations). Table 1. Region of residence and hospitalization of pediatric patients hospitalized for cerebral venous sinus thrombosis in Poland from 2013 to 2020.
Table 1. Region of residence and hospitalization of pediatric patients hospitalized for cerebral venous sinus thrombosis in Poland from 2013 to 2020. Table 2. Season of a year and month at the admission of pediatric patients hospitalized due to cerebral venous sinus thrombosis in Poland from 2013 to 2020 (n=78 hospitalizations).
Table 2. Season of a year and month at the admission of pediatric patients hospitalized due to cerebral venous sinus thrombosis in Poland from 2013 to 2020 (n=78 hospitalizations). Table 3. Admission procedures of pediatric patients hospitalized due to cerebral venous sinus thrombosis in Poland from 2013 to 2020 (n=78 hospitalizations).
Table 3. Admission procedures of pediatric patients hospitalized due to cerebral venous sinus thrombosis in Poland from 2013 to 2020 (n=78 hospitalizations). Table 4. Discharge procedures of pediatric patients hospitalized due to cerebral venous sinus thrombosis in Poland from 2013 to 2020 (n=78 hospitalizations).
Table 4. Discharge procedures of pediatric patients hospitalized due to cerebral venous sinus thrombosis in Poland from 2013 to 2020 (n=78 hospitalizations). Table 5. Duration and cost of hospitalization due to cerebral venous sinus thrombosis in Poland from 2013 to 2020 (n=78 hospitalizations).
Table 5. Duration and cost of hospitalization due to cerebral venous sinus thrombosis in Poland from 2013 to 2020 (n=78 hospitalizations). In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








