06 April 2023: Clinical Research
Evaluation of the Impact of Remote Monitoring Using the Sharesource Connectivity Platform on Adherence to Automated Peritoneal Dialysis in 51 Patients
Min-Yu Chang12AEF, Po-Jui Chi123ABCD, Hsi-Hao Wang12A, Yi-Che Lee12A, Li-Chun Ho12F, Ching-Fang Wu124CD, Ching-Yang Chen12DF, Hung-Hsiang Liou15AF, Shih-Yuan HungDOI: 10.12659/MSM.939523
Med Sci Monit 2023; 29:e939523
Abstract
BACKGROUND: This study from a single center in Taiwan aimed to evaluate the impact of remote patient monitoring (RPM) using the Sharesource connectivity platform on adherence to automated peritoneal dialysis (APD) in 51 patients.
MATERIAL AND METHODS: We analyzed data on 51 patients with end-stage renal disease (ESRD) under APD. They were treated with a traditional APD machine HomeChoice (phase 1), changed to new APD machine HomeChoice Claria for 12 weeks (phase 2), then connected to the Sharesource platform for another 12 weeks (phase 3), and were followed up for 1 year. The non-adherence rate was compared between the 3 phases. The secondary outcomes included peritonitis rate, hospitalization rate, and hospitalization days, 1 year before and after receiving a new APD machine. Patients were subdivided into good and poor adherence (>1 episode of non-adherence in phase 1) groups for further analysis.
RESULTS: The average non-adherence rates were 10.5%, 5.1%, and 4.9% in phases 1, 2, and 3, respectively, although differences were not significant. Serum potassium (P<0.0001) and C-reactive protein (CRP) (P=0.026) levels significantly decreased in phase 3. The 1-year peritonitis rate, hospitalization rate, and number of days of hospitalization showed no significant changes. Subgroup analysis revealed that the non-adherence rate in the poor adherence group decreased from 48.4% in phase 1 to 14.2% and 12.4% in phases 2 and 3, respectively (P=0.007).
CONCLUSIONS: Remoting monitoring using the Sharesource connectivity platform increased dialysis adherence in APD treatment, especially in patients with poor adherence. Serum potassium level and inflammation status were also improved by this system.
Keywords: Monitoring, Physiologic, Peritoneal Dialysis, Treatment Adherence and Compliance, Humans, Peritoneal Dialysis, Continuous Ambulatory, Kidney Failure, Chronic, peritonitis, Potassium
Background
Chronic kidney disease (CKD) is a global health burden with a high economic cost and is associated with increased cardiovascular mortality [1]. Dialysis is the main therapy for patients with CKD progression to end-stage renal disease (ESRD). Different from hemodialysis, peritoneal dialysis (PD) is the most common form of home-based dialysis [2] and approximately 11% of patients with ESRD worldwide are treated with PD [3]. PD is a treatment to remove uremic waste by infusion of dialysate into the abdominal cavity with the action of transmembrane ultrafiltration and osmosis across the peritoneum [4]. It can be performed either manually as in continuous ambulatory peritoneal dialysis (CAPD) or using mechanical devices as in automated PD (APD). In CAPD, the patient or caregiver must perform at least 3 to 5 exchanges every day; while in APD, the dialysate exchange is by a PD machine working at night for 8–10 hours during sleep time [5]. As the PD treatment is mainly autonomous without real-time monitoring, adherence to dialysis prescriptions is of great concern. Patients may skip the treatment, decrease the fill volume of dialysate, or shorten the treatment duration due to personal reasons. Non-adherence to dialysis has been shown to have major consequences, including an increased risk of mortality and hospitalization [6,7]. The overall non-adherence rates have been reported as 2.6–53% for PD prescriptions and 5–20% for APD prescriptions [7–9]. However, the non-adherence rate is usually self-reported by the patient, via a questionnaire or his/her healthcare practitioners, without real documentation. In addition, the measurement of adherence is complicated and time-consuming. Therefore, the accuracy of the non-adherence rate may be underestimated.
Remote patient monitoring (RPM) is a web-based system for monitoring patients at home by digital wireless technology [10]. The Sharesource software, launched by Baxter Healthcare Corporation in 2015, provides healthcare practitioners with real-time monitoring and recording of APD therapy. Briefly, it is based on a new APD machine, HomeChoice Claria, connected to a modem device that transfers data to a cloud-based platform. Healthcare providers can quickly read the information of dialysis performance from computer in the hospital. This remote monitoring can detect patients’ adherence to prescribed therapy [11], and also provide surveillance of critical conditions, allowing for identification and earlier intervention, including changing the patients’ prescription remotely [12]. According to previous reports, this RPM system provides better blood pressure control [8,13], increased patient survival, and decreased hospitalization rates and length of hospital stay [14], reduced the number of emergency visits and hospitalizations, especially in patients with higher comorbidity scores [15], and results in better patient satisfaction [16]. Recently, this RPM system was shown to improve patient-centered outcomes and reduced healthcare resource consumption in a randomized crossover-controlled trial [17].
However, few studies have assessed the adherence rate of this RPM program. Bunch et al reported an 85% adherence rate in patients using APD with RPM [13] and a monthly increase in adherence rate after RPM initiation [18]. The adherence rate was up to 90.1% after 1 year use of the RPM system in another study [19]. Both studies were from Colombia.
Taiwan had the highest incidence and prevalence of treated ESRD in the world in 2020 according to the annual data report from the United States Renal Data System (USRDS) [20]. Among the 7 reporting countries or regions with highest incidence, 6 were in Asia.
Therefore, this study from a single center in Taiwan aimed to evaluate the impact of remote monitoring using the Sharesource connectivity platform on adherence to automated peritoneal dialysis in 51 patients.
Material and Methods
STUDY DESIGN:
This single-center prospective clinical study was conducted in the PD department of E-Da Hospital between March 5, 2020 and April 10, 2021. This study was registered in the Clinical Trial Registry (http://www.clinicaltrials.gov; trial number: NCT04157764). The study protocol is shown in Figure 1. All the patients enrolled in our study were first kept on a traditional APD machine (HomeChoice; Baxter Healthcare Corporation, Deerfield, IL, USA) for at least 1 week (phase 1) and were then changed to HomeChoice Claria without Sharesource platform (HomeChoice Claria, Baxter Healthcare Corporation, Deerfield, IL, USA) for 12 weeks (phase 2), and the machines were connected to the Sharesource platform for another 12 weeks (phase 3). Patients were followed up for 1 year (total 52 weeks from time 0). All participants provided informed consent. This study was approved by the Institutional Review Board for the Protection of Human Subjects at E-Da Hospital and was conducted in compliance with the Declaration of Helsinki (EMRP-108-057).
PARTICIPANTS:
A total of 83 patients who were regularly followed up at our PD department were evaluated for eligibility. Patients aged >18 years who were treated with APD using HomeChoice Cycler equipment for >1 year and were willing to participate in the study were included and provided informed consent. The exclusion criteria were active infection, inflammation, malignancy, acute hospitalization events due to acute coronary syndrome, stroke, heart failure, liver cirrhosis, systemic infection within 1 month, life expectancy <1 year, and PD prescriptions scheduled or expected to change within 3 months. After exclusion, 57 patients were enrolled in this study. Following dropouts (n=6) and terminations (n=1) for specific reasons, 51 patients were analyzed for the primary outcome and 50 patients were analyzed for secondary outcomes (Figure 2).
DATA RECORD, SAMPLE COLLECTION, AND LABORATORY PARAMETERS:
Patient characteristics, including age, sex, history of diabetes mellitus, hypertension, primary etiology of ESRD, and PD vintage, were recorded. Serum levels of blood urea nitrogen (BUN), creatinine, sodium, potassium, and C-reactive protein (CRP) were measured at weeks 0, 12, and 24 (Figure 1). After fasting for 8 h, blood samples were collected between 7: 00 AM and 10: 00 AM. The tests for dialysis adequacy included total weekly fractional clearance index for urea (presented as total Kt/V per week) for both dialysate (PD Kt/V per week) and urine (renal Kt/V per week). Residual renal function was assessed by residual urine volume and residual renal creatinine clearance (CCR). These tests were performed during the last week of each phase.
All records in the new APD machine, including the fill and drain volumes, fill and drain times, treatment duration, and number of exchanges, were collected from the RPM system through wireless connection by the computer. However, data from the traditional APD machine could only be read by technician from the machine and only 1 week of records could be downloaded. The dialysis program was conducted according to doctor’s prescription. Patients who had technical or therapeutic problems could use the telephone to ask a caregiver for assistance. There was no active intervention of the treatment program from the care team during the study period unless the prescription was changed by doctors.
STUDY OUTCOMES:
The primary outcome was the non-adherence rate. The definition of non-adherence was “not following doctor’s prescription in dialysis regimen (either using wrong duration, frequency, or filling volume).” Each occurrence of non-adherence episode was recorded in the RPM system and was analyzed. The non-adherence rate was calculated as days of occurrence of non-adherence divided by the number of treatment days in each phase. Participants who completed the course until 24 weeks were analyzed for this outcome.
The secondary outcomes were 1-year peritonitis rate (number of peritonitis episodes per patient-year), hospitalization rate (number of hospital admissions per person-year), and length of hospitalization per patient-year. Participants who completed the follow-up period of up to 52 weeks were analyzed for the outcomes. A retrospective analysis comparing the rates of peritonitis and hospitalization between the 2 periods (52 weeks before entry into phase 2 and 52 weeks after entry into phase 2) was performed.
SUBGROUP ANALYSIS:
All patients were subdivided into good and poor adherence groups. Patients who had more than 1 episode of non-adherence in phase 1 were placed in the poor adherence group, while the others were included in the good adherence group. The non-adherence rate, biochemical data, and dialysis parameters were compared between the groups.
STATISTICAL ANALYSIS:
Continuous data were evaluated for normality prior to statistical testing. Continuous parametric data were expressed as the mean±standard deviation, while nonparametric data were expressed as the median and 25th to 75th interquartile range (25–75). Categorical data were expressed as percentages. Comparisons among the phases were analyzed using repeated measures one-way analysis of variance (ANOVA) for parametric data and Friedman’s ANOVA for nonparametric data. The groups were compared using the independent
Results
GENERAL CHARACTERISTIC OF PATIENTS:
The average age of the 51 patients was 51.5±12.6 years; 60.8% of them were men, and 27.5% of the patients had diabetes mellitus. The mean duration of PD treatment was 69.5±39.9 months. Regarding the clinical diagnostic etiology of chronic kidney disease, glomerular disease (28.8%) and chronic interstitial nephritis (23.1%) were the most prevalent (Table 1).
PRIMARY END-POINT:
The non-adherence rate was 10.5±26% in phase 1 and it gradually decreased to 5.1±12.7% and 4.9±13.2% in phases 2 and 3, respectively, although this decrease was not significant (P=0.808) (Table 1).
SERIAL CHANGES OF LABORATORY PROFILE IN PATIENTS:
Serum potassium (3.63±0.72 vs 4.02±0.68, P<0.001) and CRP (2.1 (0.9–7.6) vs 3.1 (0.7–11.9), P=0.026) levels were significantly decreased in phase 3 compared to phase 1 (Table 1). Residual urine volume and residual CCR were significantly decreased, whereas BUN levels were significantly increased throughout the 3 phases because of worsening residual renal function. There were no significant differences in serum sodium levels, total Kt/V per week, and PD Kt/V per week among the 3 phases (Table 1).
COMPARISON BETWEEN THE GOOD ADHERENCE AND POOR ADHERENCE GROUPS:
There were more men in the poor adherence group than in the good adherence group (100% vs 50%, P=0.003) (Table 2). Patients in the poor adherence group tended to be younger and have longer PD vintages. The non-adherence rate showed a significant gradual reduction in the poor adherence group after the introduction of HomeChoice Claria and connection of Sharesource (12.4±17.3 in phase 3 vs 14.2±16.3 in phase 2 vs 48.4±36.9 in phase 1, P=0.007) (Table 2). However, there was no significant difference before and after the connection of Sharesource. Both groups showed a decrease in serum potassium levels throughout the 3 phases. Serum CRP levels were much higher in the poor adherence group (6.71 (2.22–24.52) vs 2.82 (0.47–10.94), P=0.044). The residual CCR significantly decreased throughout the study period in both groups; however, the total KT/V per week decreased only in the poor adherence group (1.81 (1.75–2) vs 1.89 (1.79–2.27), P=0.035).
SECONDARY END-POINTS:
The 1-year peritonitis rate, hospitalization rate, and days of hospitalization before and after the initiation of HomeChoice Claria did not differ significantly (Table 3).
Discussion
Our study revealed a significant improvement of dialysis adherence to APD therapy, especially in poor-adherent patients by RPM system.
In a systematic review published in 2014, the rate of non-adherence to dialysis prescription in PD therapy varied from 2.6% to 53% according to different studies [9]. However, most adherence-related studies on PD therapy were conducted in North America, and only 6 studies were conducted in Asia, when there was no RPM system. Moreover, few studies have discussed different non-adherence rates in different PD modalities. It was reported that non-adherence to APD treatment ranged from 5% to 20%, which was lower than that to CAPD (10–47%) [7,9,20,21]. RPM system must be able to provide a more correct evaluation of adherence by the real-time monitoring. After the introduction of the RPM system for APD therapy, the non-adherence rate was found to be between 10% and 15% reported from Colombia [13,19], while there were no reports from Asia.
We found a 10.5% non-adherence rate when the traditional APD machine HomeChoice was used. The rate decreased to 5.1% after the treatment was transferred to the new APD machine HomeChoice Claria, and further decreased to 4.9% when the new machines were connected to the Sharesource connectivity platform (Table 1). Currently, there is only 1 report, from Singapore in 2012, which shows a 5% non-adherence rate in 15 Asian APD patients by traditional APD machines without RPM [22]. Our study is the first to demonstrate the change of non-adherence rate of APD therapy in Asian patients, demonstrating the APD device revolution, especially when the RPM system was introduced.
APD patients are required to adhere to long overnight dialysis exchange by a APD machine setting according to doctor’s prescription. Self-adjusting the machine to reduce exchanges, shortening the durations, or decreasing filling volumes during dialysis treatment are considered as non-adherence. The reasons for non-adherence included socio-demographic conditions (eg, age, employment status, education level), medical/treatment-related factors (eg, duration of dialysis, number of comorbidities), and psychological and personality aspects [9]. Our study particularly shows the benefit of RMP system in improving APD adherence, especially in patients with poor adherence. Table 2 shows a significant reduction in the non-adherence rate when new APD machines were introduced and adherence was further improved after the RPM system was connected. Patients were believed to be more cautious and obeyed orders because of the real-time surveillance of the system. As a result, awareness of the monitoring system may increase adherence to treatment. This further shows that using the RPM system can be of great help to poorly-adherent patients.
Serum potassium and CRP levels significantly decreased after connection of the RPM system in all participants (Table 1). This implies that the serum potassium level and inflammation status were better controlled by the RPM system owing to better adherence. Although residual renal function gradually decreased throughout the study period, we speculated that dialytic clearance could be improved if adherence was improved. However, the PD Kt/V did not show an increasing trend in our analysis, possibly because the Kt/V was measured only in the last week of each phase, which did not represent the full period of the study phases.
In the poor adherence group, more patients were male, younger, and had a longer PD duration than those in the good adherence group. These findings are similar to those of previous studies [9]. The inflammation status was believed to be higher in this group because of the higher serum CRP levels, possibly due to poor adherence. The total KT/V was lower in this group than in the good adherence group, although the difference was not significant, at 1.89 (1.79–2.27) vs 2.05 (1.84–2.21),
Our study revealed that the PD non-adherence rate was improved, especially in the poor adherence group. However, there was no significant difference between patients with and without use of the RMP system. There are several possible explanations for this finding. First, the patients might have believed that there was real-time monitoring after changing to the new APD machines and had better compliance, although there was no connection to the RPM system. Second, the follow-up period was not sufficiently long. The fluctuation in non-adherence may limit the difference to only 3 months. Third, the non-adherence rate of our patients was obviously low and was not significantly different between the different time periods.
The 1-year peritonitis rate, hospitalization rate, and number of hospitalization days showed no differences before and after the introduction of the new APD machine. A possible reason may be the lower incidence of the above-mentioned episodes in our study group due to the small number of cases and inadequate observation time.
There are several limitations in this study. First, the number of patients was low due to the single-center design of the study and the statistical power may have been reduced accordingly. Second, the follow-up time was not adequate and the event rate was too low to reach a significant difference between observation periods. Third, there was lack of a control group during the same study period. A study with multi-center enrollment and longer observation time with a control group is needed to understand the effects of RPM on APD treatment.
Conclusions
The RPM system using the Sharesource connectivity platform improved dialysis adherence in APD treatment, especially in patients with poor adherence. In addition, it provided better potassium control and decreased inflammation status in patients with PD. Further investigations should be performed with healthcare practitioner intervention, which is more similar to real-world conditions. It is anticipated that a real-time and remote monitoring of home-based treatment could bring more promising results to patients undergoing APD.
Figures
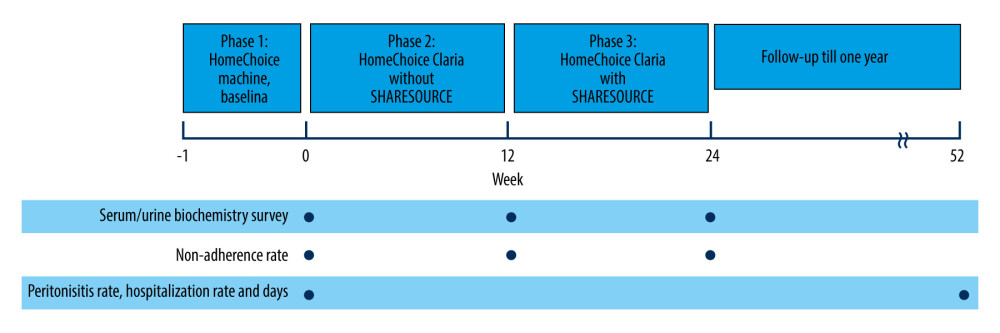 Figure 1. Study design. Patients were maintained on a traditional automated peritoneal dialysis machine (HomeChoice, Baxter Healthcare Corporation, Deerfield, IL, USA) for at least 1 week (phase 1), and then changed to HomeChoice Claria (Baxter Healthcare Corporation, Deerfield, IL, USA) without Sharesource for 12 weeks (phase 2). The machines were then connected to Sharesource for another 12 weeks (phase 3). Patients were followed up for 1 year. (Designed in Microsoft Word and converted to JPG).
Figure 1. Study design. Patients were maintained on a traditional automated peritoneal dialysis machine (HomeChoice, Baxter Healthcare Corporation, Deerfield, IL, USA) for at least 1 week (phase 1), and then changed to HomeChoice Claria (Baxter Healthcare Corporation, Deerfield, IL, USA) without Sharesource for 12 weeks (phase 2). The machines were then connected to Sharesource for another 12 weeks (phase 3). Patients were followed up for 1 year. (Designed in Microsoft Word and converted to JPG). 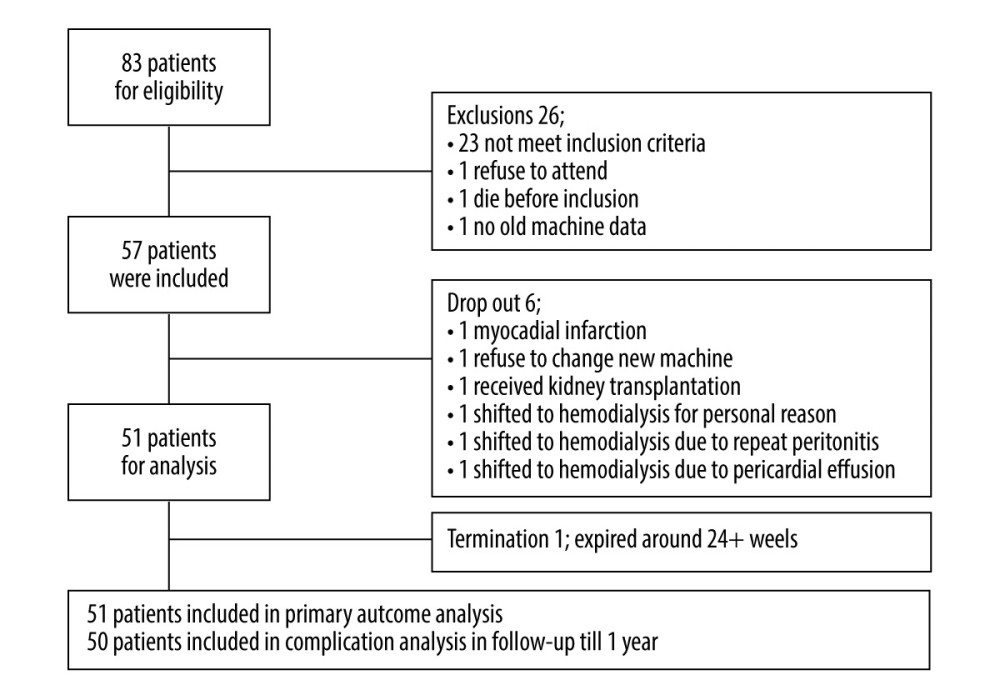 Figure 2. Trial profile. A total of 83 patients were evaluated for eligibility in this study, and after exclusion, 57 patients were enrolled. After exclusion of dropout (n=6) and termination (n=1) for specific reasons, 51 patients were analyzed for the primary outcome and 50 patients were analyzed for the secondary outcomes. (Designed in Microsoft Word and converted to JPG).
Figure 2. Trial profile. A total of 83 patients were evaluated for eligibility in this study, and after exclusion, 57 patients were enrolled. After exclusion of dropout (n=6) and termination (n=1) for specific reasons, 51 patients were analyzed for the primary outcome and 50 patients were analyzed for the secondary outcomes. (Designed in Microsoft Word and converted to JPG). Tables
Table 1. Clinical and laboratory parameters at baseline, after HomeChoice Claria machine and after connection with remote patient monitoring (RPM) system (n=51).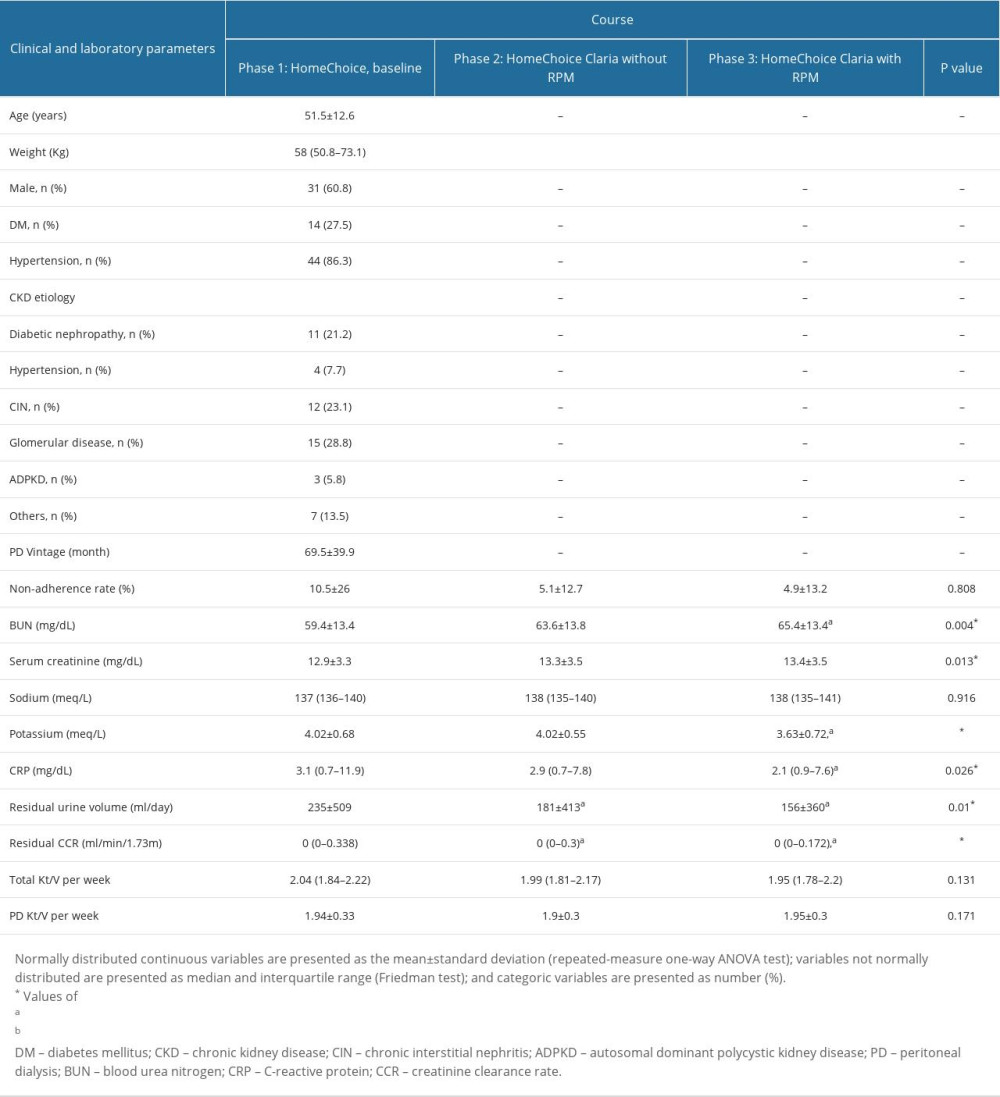 Table 2. Subgroup analysis at baseline, after HomeChoice Claria machine and after connection with remote patient monitoring (RPM) system.
Table 2. Subgroup analysis at baseline, after HomeChoice Claria machine and after connection with remote patient monitoring (RPM) system.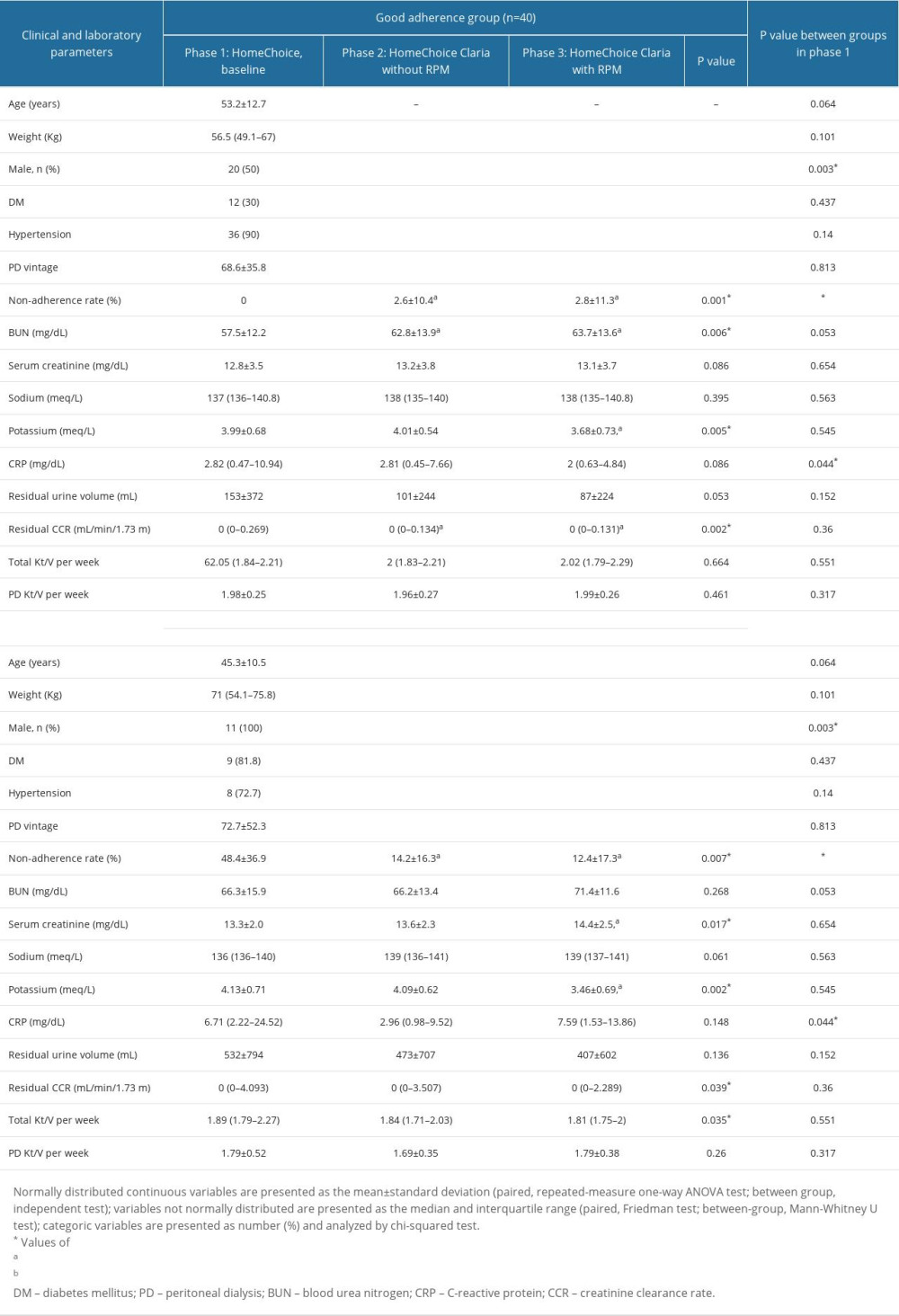 Table 3. Peritonitis and hospitalization incidence before and after use of HomeChoice Claria.
Table 3. Peritonitis and hospitalization incidence before and after use of HomeChoice Claria.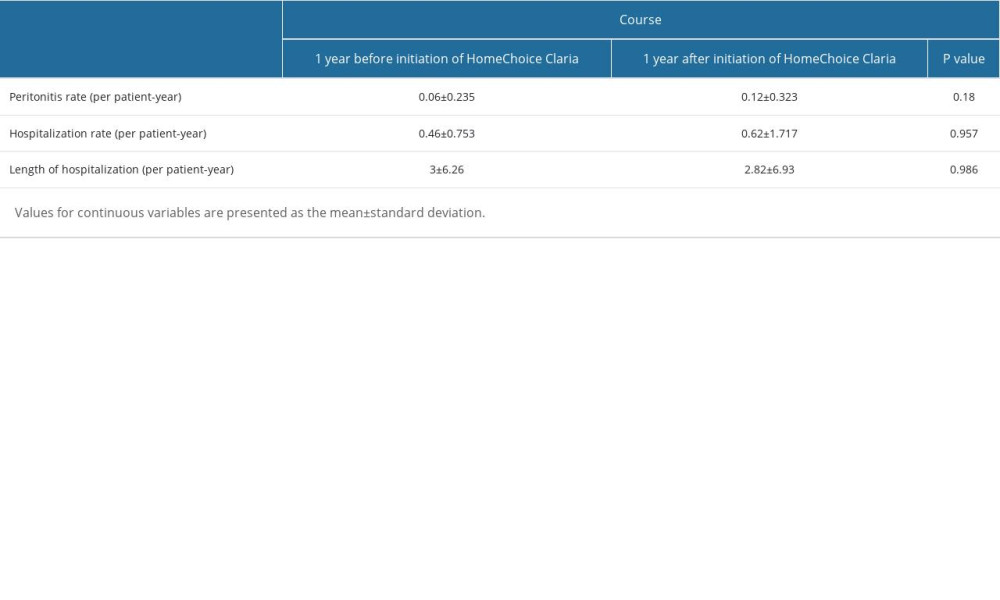
References
1. Hill NR, Fatoba ST, Oke JL, Global prevalence of chronic kidney disease – a systematic review and meta-analysis: PLoS One, 2016; 11(7); e0158765
2. Saran R, Robinson B, Abbott KC, US renal data system 2018 annual data report: Epidemiology of kidney disease in the United States: Am J Kidney Dis, 2019; 73(3); A7-A8
3. Cho Y, Bello AK, Levin A, Peritoneal dialysis use and practice patterns: An international survey study: Am J Kidney Dis, 2021; 77; 315-25
4. Teitelbaum I, Peritoneal dialysis: N Engl J Med, 2021; 385(19); 1786-95
5. Roumeliotis A, Roumeliotis S, Leivaditis K, APD or CAPD: One glove does not fit all: Int Urol Nephrol, 2021; 53(6); 1149-60
6. Denhaerynck K, Manhaeve D, Dobbels F, Prevalence and consequences of nonadherence to hemodialysis regimens: Am J Crit Care, 2007; 16(3); 222-35 quiz 236
7. Bernardini J, Piraino B, Compliance in CAPD and CCPD patients as measured by supply inventories during home visits: Am J Kidney Dis, 1998; 31(1); 101-7
8. Yeter HH, Akcay OF, Ronco C, Derici U, Automated remote monitoring for peritoneal dialysis and its impact on blood pressure: Cardiorenal Med, 2020; 10(3); 198-208
9. Griva K, Lai AY, Lim HA, Nonadherence in patients on peritoneal dialysis: A systematic review: PLoS One, 2014; 9(2); e89001
10. Whitlow M, Wallace E, Remote patient monitoring: an important tool in advancing home dialysis: Kidney Med, 2019; 1(6); 327-28
11. Milan Manani S, Crepaldi C, Giuliani A, Remote monitoring of automated peritoneal dialysis improves personalization of dialytic prescription and Patient’s independence: Blood Purif, 2018; 46(2); 111-17
12. Uchiyama K, Washida N, Yube N, The impact of a remote monitoring system of healthcare resource consumption in patients on automated peritoneal dialysis (APD): A simulation study: Clin Nephrol, 2018; 90(5); 334-40
13. Bunch A, Vesga JI, Camargo DO, Remote automated peritoneal dialysis management in Colombia: Kidney Int Rep, 2019; 4(6); 873-76
14. Sanabria M, Buitrago G, Lindholm B, Remote patient monitoring program in automated peritoneal dialysis: Impact on hospitalizations: Perit Dial Int, 2019; 39(5); 472-78
15. Milan Manani S, Baretta M, Giuliani A, Remote monitoring in peritoneal dialysis: benefits on clinical outcomes and on quality of life: J Nephrol, 2020; 33(6); 1301-8
16. Jung HY, Jeon Y, Kim YS, Outcomes of remote patient monitoring for automated peritoneal dialysis: A randomized controlled trial: Nephron, 2021; 145(6); 702-10
17. Uchiyama K, Morimoto K, Washida N, Effects of a remote patient monitoring system for patients on automated peritoneal dialysis: A randomized crossover controlled trial: Int Urol Nephrol, 2022; 54(10); 2673-81
18. Bunch A, Ardila F, Castaño R, Through the storm: Automated peritoneal dialysis with remote patient monitoring during COVID-19 pandemic: Blood Purif, 2021; 50(3); 279-82
19. Sanabria M, Rosner M, Vesga J, A remote management program in automated peritoneal dialysis patients in Colombia: Nefrol Latinoam, 2018; 15(2); 48-51
20. : United States Renal Data System 2022 USRDS Annual Data Report: Epidemiology of kidney disease in the United States National Institutes of Health, 2022, Bethesda, MD, National Institute of Diabetes and Digestive and Kidney Diseases
21. Bernardini J, Piraino B, Measuring compliance with prescribed exchanges in CAPD and CCPD patients: Perit Dial Int, 1997; 17(4); 338-42
22. Yu ZL, Yeoh LY, Seow YY, Evaluation of adherence and depression among patients on peritoneal dialysis: Singapore Med J, 2012; 53(7); 474-80
Figures
 Figure 1. Study design. Patients were maintained on a traditional automated peritoneal dialysis machine (HomeChoice, Baxter Healthcare Corporation, Deerfield, IL, USA) for at least 1 week (phase 1), and then changed to HomeChoice Claria (Baxter Healthcare Corporation, Deerfield, IL, USA) without Sharesource for 12 weeks (phase 2). The machines were then connected to Sharesource for another 12 weeks (phase 3). Patients were followed up for 1 year. (Designed in Microsoft Word and converted to JPG).
Figure 1. Study design. Patients were maintained on a traditional automated peritoneal dialysis machine (HomeChoice, Baxter Healthcare Corporation, Deerfield, IL, USA) for at least 1 week (phase 1), and then changed to HomeChoice Claria (Baxter Healthcare Corporation, Deerfield, IL, USA) without Sharesource for 12 weeks (phase 2). The machines were then connected to Sharesource for another 12 weeks (phase 3). Patients were followed up for 1 year. (Designed in Microsoft Word and converted to JPG). Figure 2. Trial profile. A total of 83 patients were evaluated for eligibility in this study, and after exclusion, 57 patients were enrolled. After exclusion of dropout (n=6) and termination (n=1) for specific reasons, 51 patients were analyzed for the primary outcome and 50 patients were analyzed for the secondary outcomes. (Designed in Microsoft Word and converted to JPG).
Figure 2. Trial profile. A total of 83 patients were evaluated for eligibility in this study, and after exclusion, 57 patients were enrolled. After exclusion of dropout (n=6) and termination (n=1) for specific reasons, 51 patients were analyzed for the primary outcome and 50 patients were analyzed for the secondary outcomes. (Designed in Microsoft Word and converted to JPG). Tables
 Table 1. Clinical and laboratory parameters at baseline, after HomeChoice Claria machine and after connection with remote patient monitoring (RPM) system (n=51).
Table 1. Clinical and laboratory parameters at baseline, after HomeChoice Claria machine and after connection with remote patient monitoring (RPM) system (n=51). Table 2. Subgroup analysis at baseline, after HomeChoice Claria machine and after connection with remote patient monitoring (RPM) system.
Table 2. Subgroup analysis at baseline, after HomeChoice Claria machine and after connection with remote patient monitoring (RPM) system. Table 3. Peritonitis and hospitalization incidence before and after use of HomeChoice Claria.
Table 3. Peritonitis and hospitalization incidence before and after use of HomeChoice Claria. Table 1. Clinical and laboratory parameters at baseline, after HomeChoice Claria machine and after connection with remote patient monitoring (RPM) system (n=51).
Table 1. Clinical and laboratory parameters at baseline, after HomeChoice Claria machine and after connection with remote patient monitoring (RPM) system (n=51). Table 2. Subgroup analysis at baseline, after HomeChoice Claria machine and after connection with remote patient monitoring (RPM) system.
Table 2. Subgroup analysis at baseline, after HomeChoice Claria machine and after connection with remote patient monitoring (RPM) system. Table 3. Peritonitis and hospitalization incidence before and after use of HomeChoice Claria.
Table 3. Peritonitis and hospitalization incidence before and after use of HomeChoice Claria. In Press
07 Mar 2024 : Clinical Research
Knowledge of and Attitudes Toward Clinical Trials: A Questionnaire-Based Study of 179 Male Third- and Fourt...Med Sci Monit In Press; DOI: 10.12659/MSM.943468
08 Mar 2024 : Animal Research
Modification of Experimental Model of Necrotizing Enterocolitis (NEC) in Rat Pups by Single Exposure to Hyp...Med Sci Monit In Press; DOI: 10.12659/MSM.943443
18 Apr 2024 : Clinical Research
Comparative Analysis of Open and Closed Sphincterotomy for the Treatment of Chronic Anal Fissure: Safety an...Med Sci Monit In Press; DOI: 10.12659/MSM.944127
08 Mar 2024 : Laboratory Research
Evaluation of Retentive Strength of 50 Endodontically-Treated Single-Rooted Mandibular Second Premolars Res...Med Sci Monit In Press; DOI: 10.12659/MSM.944110
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952








