21 March 2023: Clinical Research
Is There a Relationship Between Bell’s Palsy and White Matter Lesions?
Nurcan Yurtsever KumDOI: 10.12659/MSM.939723
Med Sci Monit 2023; 29:e939723
Abstract
BACKGROUND: The present study aimed to investigate the relationship between Bell’s palsy (BP) and cerebral white matter lesions (CWMLs) on brain gadolinium-enhanced magnetic resonance imaging (MRI).
MATERIAL AND METHODS: The study included 51 patients who were diagnosed with BP and a control group of 40 individuals who underwent brain MRI for investigation of headache etiology. The brain MRIs of the patients were reviewed with respect to CWMLs. CWMLs were compared between the 2 groups, and within the BP group, the correlation between CWML and House-Brackmann (HB) facial nerve palsy grade was investigated as well.
RESULTS: There were significantly more CWMLs in the BP group than in the control group (P=0.003). There was a statistically significant difference between the HB subgroups in terms of absence/presence of CWMLs (P=0.040). Within the BP patient group, there were significantly more CWMLs in the HB grade 5 and 6 groups compared with the HB grade 2 group (P=0.025 and P=0.042, respectively). Overall, the CWML scores correlated positively with HB grade. When absence/presence of CWMLs was compared between the BP and control groups in only patients aged <50 years, there was a statistically significant difference between the groups (P=0.008). No statistically significant difference in absence/presence of CWMLs, however, was found between the BP and control groups when only patients age ≥50 years were considered (P=0.809).
CONCLUSIONS: We showed that as the severity of Bell’s palsy increased, CWMLs increased and there was an association between CWMLs and BP. Microvascular ischemic pathologies may be among the most important factors in the etiopathogenesis of BP.
Keywords: Bell Palsy, Magnetic Resonance Imaging, white matter, Humans, Head, Vascular Diseases
Background
Idiopathic peripheral facial nerve paralysis has been designated as Bell’s palsy (BP). BP’s exact etiologic factors are still not clear, although atherosclerosis, viruses, microvascular injury, and neural ischemia have been implicated [1,2]. The ischemia hypothesis claims that microvascular damage, a coagulation disorder, or a vasospasm involving the vasa nervorum of the facial nerve results in BP [3]. Viral infections could lead to inflammation and could subsequently result in atherosclerosis [4]. Various diagnostic tests have been used in the assessment of BP, and magnetic resonance imaging (MRI) is widely used for the assessment of pathology involving the facial nerve, as well as for differential diagnosis [1,5–7].
Several diseases may affect the cerebral small vessels, which include small arterioles, arteries, capillaries, and venules of the brain. The pathologies that affect cerebral small vessels play crucial roles in central vascular events such as thromboembolism, deep or cortical hemorrhages, and lacunar cerebral infarction. Several studies have indicated that features of cerebral small vessel diseases visible via MRI include small subcortical infarcts and cerebral white matter lesions (CWMLs) [8–10].
CWMLs appear as hyperintense cerebral lesions in the periventricular white matter (PVWM) or cerebral deep white matter (DWM) on MRI. Numerous risk factors including hypertension, smoking, aging, obesity, metabolic syndrome, diabetes, obstructive sleep apnea syndrome, and stroke can worsen blood supply and tissue perfusion, and these factors have been implicated in the etiology of CWMLs [11,12].
The present study aimed to investigate the relationship between CWMLs and BP by using MRI.
Material and Methods
This study utilized a retrospective design, and included 118 BP patients admitted to a tertiary referral center between January 2012 and February 2020, and a control group consisting of 40 individuals who underwent brain MRIs for investigation of headache etiology. The individuals in the control group had no malignant tumors, hypertension, diabetes mellitus, hyperlipidemia, obstructive sleep apnea, stroke, cardiovascular disease, or facial palsy. A total of 67 patients were excluded from the study because they had disorders that might have caused CWMLs. These included 18 patients with hypertension, 16 patients with diabetes mellitus, 13 patients with angina pectoris and/or a history of myocardial infarction, 12 patients with hyperlipidemia, 5 patients with obstructive sleep apnea, and 3 patients with stroke.
House-Brackmann (HB) grading was used to determine the grade of facial paralysis in the BP patient group. The same therapeutic protocol was applied to all patients: 1 mg/kg/day prednisone, tapered and discontinued in 2 weeks. Cerebral and temporal MRIs were obtained within 7 days of BP onset.
The MRIs and medical files of 51 patients and 40 controls were reviewed using the hospital’s computerized patient data software. The MRIs were examined for CWML and graded using the Fazekas scale by a neuroradiologist who was blinded to the clinical data of the subjects. The Fazekas scale divides CWMLs into PVWM lesions and DWM lesions, and each lesion is given a rating from 1 to 3 based on the size and composition of the lesion [13].
The MRIs were performed using 1 of 2 different instruments; namely, the 1.5-T Optima 450 W or the 1.5-T Excite (General Electric Healthcare, Wisconsin, USA). For the brain MRI, fast-recovery fast spin echo T2 weighted images (repetition time (TR), 4240 ms; echo time (TE), 98.1 ms; slice thickness, 5 mm; interslice gap, 1.5 mm; field of view (FOV), 24×18 cm; matrix, 352×224; number of excitations (NEX), 2) and fluid-attenuated inversion recovery images were obtained before and after intravenous gadolinium (0.2 ml/kg) injection in all subjects.
The age, gender, MRI findings in terms of CWMLs, and clinical characteristics of the patients were analyzed. The BP group and control group were compared for CWMLs. In addition, correlations between CWML score and HB grade were investigated.
Our local ethics committee approved the study protocol (date: 04.02.2020, Project No: E1-20-336).
The statistical analyses were performed using IBM-SPSS for Windows v. 21.0 software (IBM Corporation, Armonk, NY, USA). The Kolmogorov-Smirnov test was employed to determine the normality of the data distribution. The descriptive data were presented as mean±standard deviation, or median and minimum-maximum. Numbers and percentages were used for categorical variables. Two-group comparisons of normally distributed quantitative data were carried out by
Results
COMPARISONS OF CWML SCORE WITHIN THE BP GROUP ACCORDING TO HB GRADE:
The CWML scores in the BP patient group and the severity of BP according to HB grade are presented in Table 2. The HB grade subgroups did not differ significantly in PVWM or DWM scores (P=0.123 and P=0.071, respectively). However, the subgroups were significantly different in terms of absence/presence of CWMLs (P=0.040). The groups responsible for the strongest differences were determined through paired comparisons, which revealed that, compared with the HB grade 2 group, there were significantly more CWMLs in the HB grade 5 and 6 groups (P=0.025 and P=0.042, respectively) (Table 2).
CORRELATION BETWEEN CWML SCORE AND HB GRADE:
The CWML scores (PVWM, DWM, and absence/presence of CWMLs) were positively correlated with HB grade (P=0.020, P=0.012, and P=0.003, respectively) (Table 3).
COMPARISONS OF CWML SCORES BETWEEN BP AND CONTROL GROUPS IN PATIENTS AGED ≥50 YEARS AND <50 YEARS:
When absence/presence of CWMLs was compared in patients aged <50 years, there was a statistically significant difference between the BP and control patients (P=0.008). However, no significant difference was seen between BP and control patients when only considering patients aged ≥50 years (P=0.809) (Figure 2).
Discussion
In the present study, we found increased CWMLs in patients with BP, especially in cases of severe facial palsy according to the HB grading system. Furthermore, there was a positive correlation between CWMLs and BP, which may be ischemic in etiology.
The underlying cause of acute peripheral facial nerve paresis or paralysis is not clear in Bell’s palsy. Although the etiology of BP has not yet been elucidated, viral infections, ischemia, and autoimmune disorders of the facial nerve have been implicated [14]. In the ischemia hypothesis, it has been claimed that an embolism or vasospasm in the vasa nervorum due to a bleeding disorder or dysfunction in the autonomic nervous system results in facial nerve damage [15]. The cerebral small vessels are essential to the maintenance of an adequate blood supply to the brain and related structures, which include small arteries, arterioles, capillaries, and postcapillary venules [16]. Omori et al showed high α-plasmin inhibitor-plasmin complex and thrombin-antithrombin-3 levels in patients with BP, and claimed that a coagulation and circulatory disorder played a role in its etiopathogenesis [15].
Cerebral small vessel disease (CSVD) is a degenerative vessel disorder that refers to intracranial vascular disease related to various pathological conditions linked to vascular injury, including aging, hypertension, and atherosclerosis, that result in different clinical manifestations and neuroimaging features [8,17]. CSVD lesions can manifest as various MRI findings; most commonly, CWMLs, lacunar infarcts, and enlargement of perivascular spaces [16]. Hachinski et al first described white matter lesions (WMLs) as “leukoaraiosis” or white matter hyperintensities, detected as hyperintense cerebral white matter signals on T2-weighted MRI [18]. The most widely assumed pathophysiological mechanism underlying WMLs is ischemia. The condition results when hypoxia-ischemia induces endothelial dysfunction leading to infiltration of inflammatory mediators or cytokines [11,12]. The prevalence of CWMLs has been reported in a very wide range in the literature (5.3% to 95%) [19]. The wide spectrum of those results may be due to differences in the study populations, including comorbidities, different inclusion and exclusion criteria, and different radiological grading methods [20]. CWMLs are associated with several clinical manifestations, such as memory impairment, cognitive dysfunction, depression, cerebrovascular disease, and stroke [21,22]. For these reasons, we investigated the MRI findings of patients with BP in terms of CWMLs to assess the ischemic theory of BP etiology.
Microcirculatory failure of the vasa nervosum impairs nerve metabolism and causes inflammation and venous stasis, resulting in BP [23]. Liou et al reported that BP patients had a significantly greater risk of peripheral arterial occlusive disease than subjects without BP [24].
Jonsson et al investigated MRI findings in patients with BP in terms of abnormal changes. The authors found high-intensity signals in the brain and/or cerebral white matter, and concluded that those lesions may be related to a microvascular or demyelinating disorder, indicating central nervous system involvement in BP [25].
Fusconi et al claimed that sudden sensorineural hearing loss (SSNHL)- and CSVD-related diseases have similar vascular etiologies. They investigated the relationship between SSNHL and CWMLs, and found a higher CSVD prevalence in SSNHL patients aged between 48 and 60 years compared with the general population [26]. However, others claimed that the CWML incidence was similar in SSNHL patients and the control group; suggesting that the presence of CWMLs may instead be correlated with a poorer hearing recovery rate in SSNHL patients [27]. Recently, Shin et al reported that severe CWMLs and concomitant cardiovascular risk factors may increase the possibility of a worse baseline level of hearing and poor treatment response in SSNHL, although CWML prevalence was not high in patients with SSNHL [28]. BP and SSNHL idiopathic cranial nerve pathologies both have similar etiologic factors. Therefore, these studies may offer evidence for a relationship between BP and CWMLs.
In our study, we found significant differences between the BP patients and the control patients for PVWM, DWM, and the absence/presence of CWMLs. Furthermore, we found a positive correlation between the CWML scores and HB grades, in which, as the severity of the HB grade increased, the CWML score increased. This may suggest that there is circulation failure or vascular damage in patients with BP, especially in patients with severe BP.
Cerebral white matter changes have frequently been reported on brain imaging, most frequently in the elderly. Aging is a major risk factor for CWMLs. These lesions are usually seen in patients aged 50–65 years [20,29]. For this reason, we compared the BP and control groups separately in groups divided according to age: the first group included patients <50 years old only and the second included patients ≥50 years old only. In our study, when comparing only patients under the age of 50, the absence/presence of CWMLs was higher in the BP patients compared with the control group. However, the BP and control groups were similar for absence/presence of CWMLs when only patients ≥50 years of age were compared. Although CWMLs have a higher incidence in the elderly population, our results offer evidence for a higher incidence of CWMLs in patients with BP compared with the general population when considering individuals under 50 years of age.
Furthermore, as we excluded patients with comorbidities related to CWMLs, such as hyperlipidemia, obstructive sleep apnea, diabetes, hypertension, and stroke, from our study, we believe that our results offer evidence for a correlation between CWMLs and BP.
Our study has some limitations: it is a retrospective single-center study, and the neurophysiological circumstances and recovery status of the patients were not analyzed. Prospective, controlled, multi-center studies with a more homogeneous and larger study population are required.
Conclusions
To our knowledge, ours is the first study that has investigated the role of CWMLs in BP. The number of CWMLs was higher in BP patients than in controls, and CWMLs were predominantly seen in BP of HB grade 5–6. Microvascular ischemic pathologies may be a major factor in the etiopathogenesis of BP. Consequently, comprehension of the risk factors for CSVD, and prevention and management of these factors, is critical.
Figures
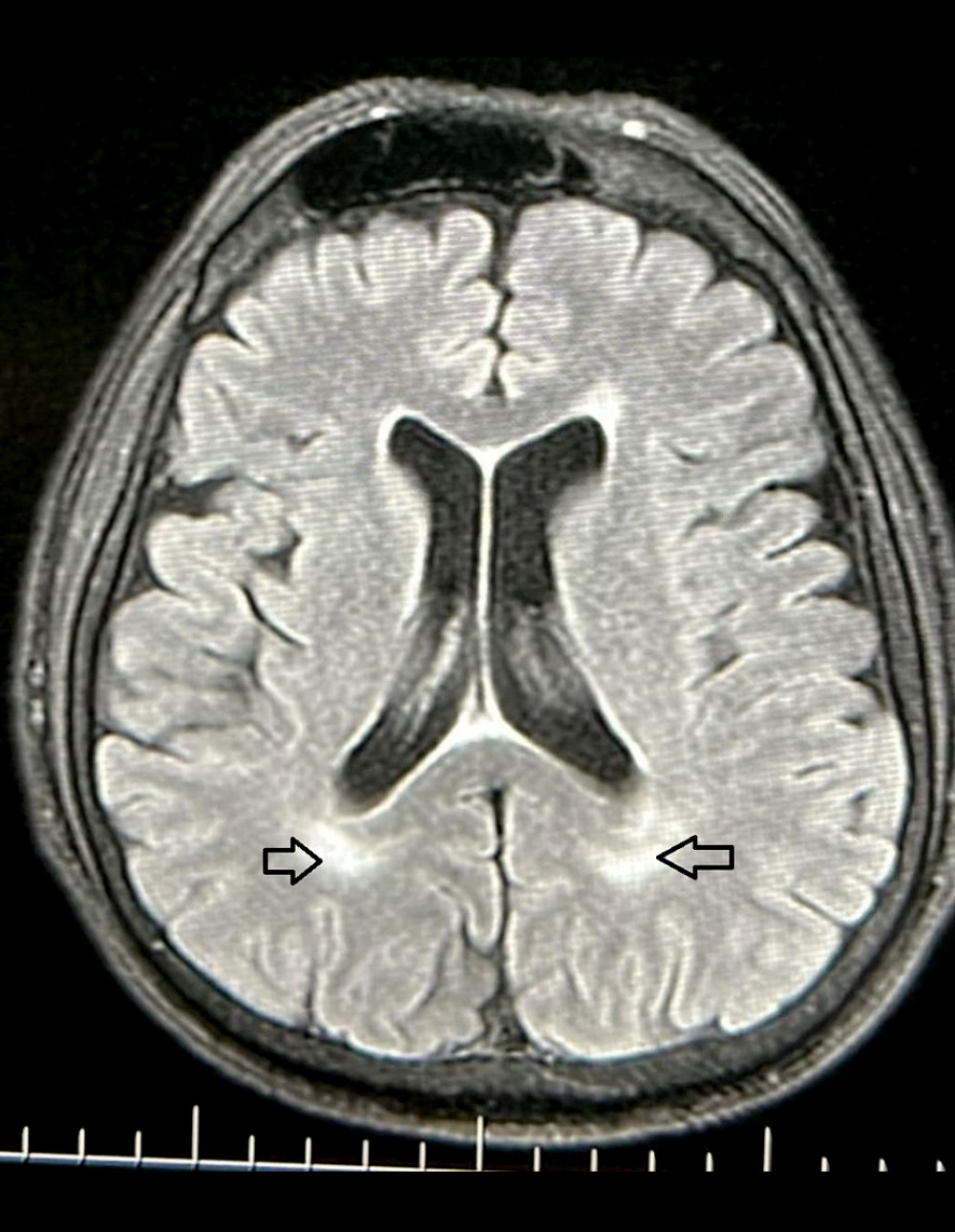 Figure 1. Axial section of periventricular white matter lesions in a T2 weighted magnetic resonance image. The lesions in cerebral white matter are shown (arrows) in a patient with House-Brackmann grade 5 Bell’s palsy. This figure was created using PowerPoint 2013 (Microsoft).
Figure 1. Axial section of periventricular white matter lesions in a T2 weighted magnetic resonance image. The lesions in cerebral white matter are shown (arrows) in a patient with House-Brackmann grade 5 Bell’s palsy. This figure was created using PowerPoint 2013 (Microsoft). 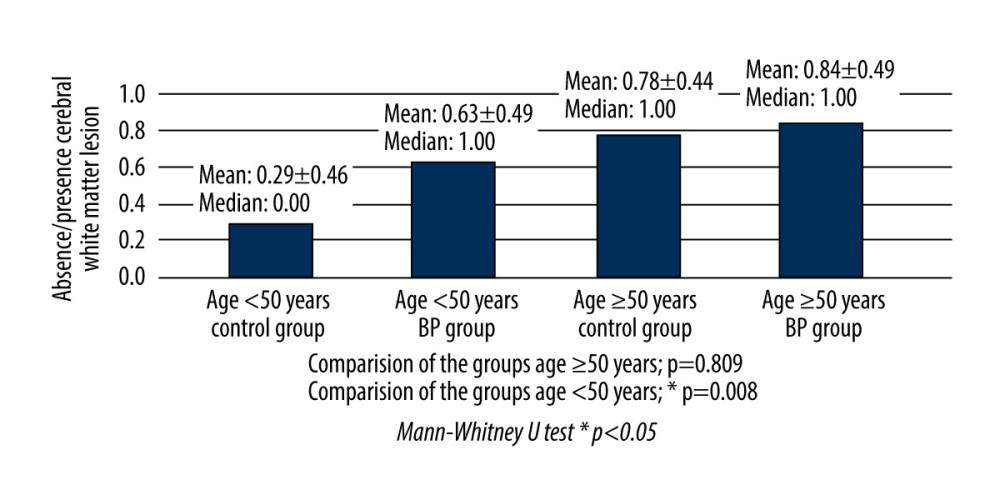 Figure 2. Comparisons of cerebral white matter lesions in Bell’s palsy and control patients, in patients grouped according to age: ≥50 years and <50 years.
Figure 2. Comparisons of cerebral white matter lesions in Bell’s palsy and control patients, in patients grouped according to age: ≥50 years and <50 years. Tables
Table 1. The demographic characteristics and the variables studied in Bell’s palsy patients and the control group patients.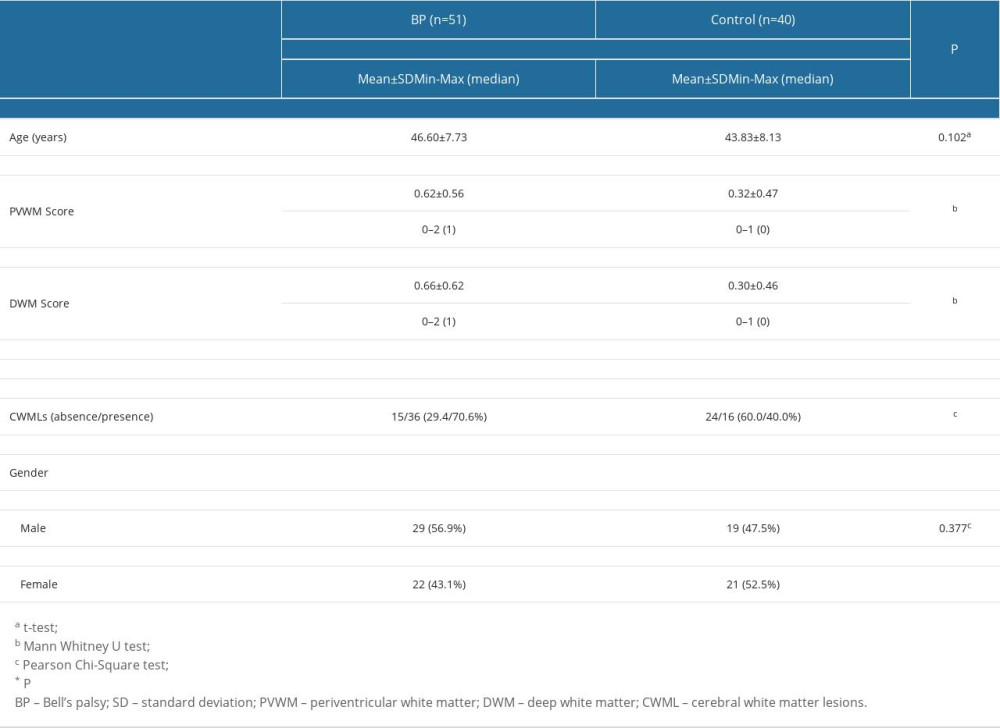 Table 2. Comparisons of cerebral white matter lesions within the Bell’s palsy group according to House-Brackmann grade.
Table 2. Comparisons of cerebral white matter lesions within the Bell’s palsy group according to House-Brackmann grade.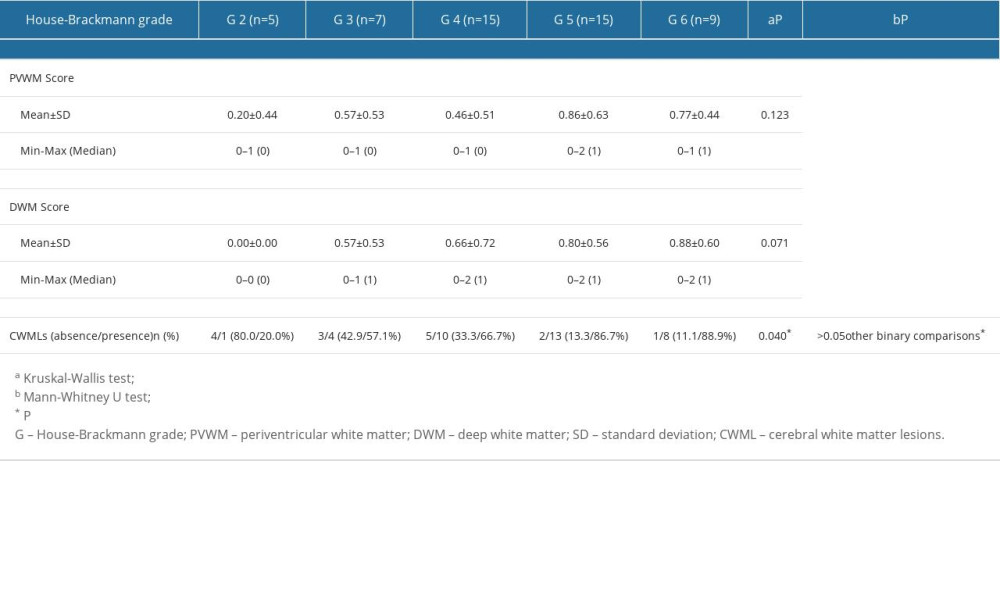 Table 3. Correlation between cerebral white matter lesions and House-Brackmann grade.
Table 3. Correlation between cerebral white matter lesions and House-Brackmann grade.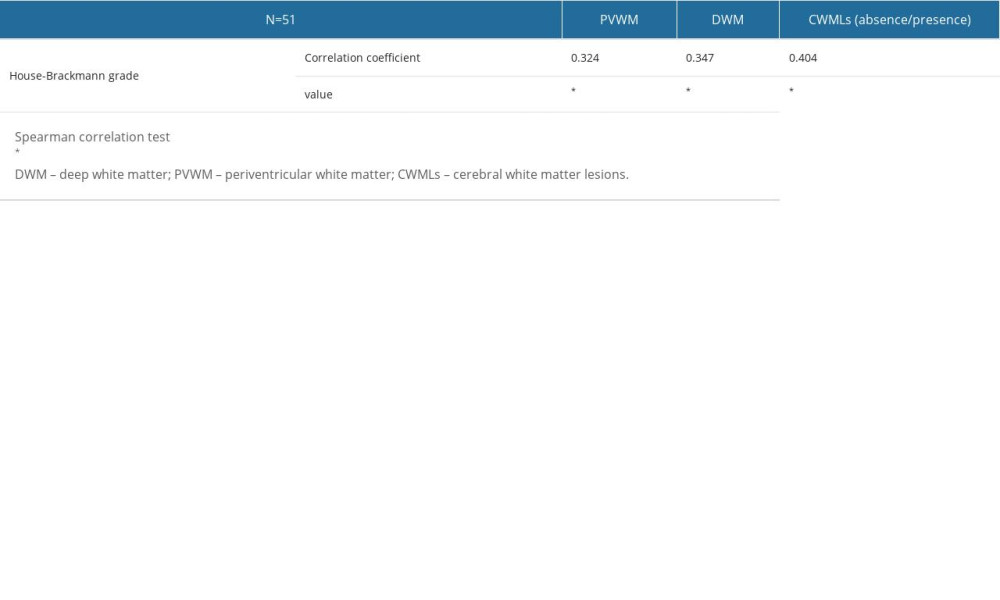
References
1. Singh A, Deshmukh P, Bell’s palsy: A review: Cureus, 2022; 14(10); e30186
2. Zhao Y, Feng G, Gao Z, Advances in diagnosis and non-surgical treatment of Bell’s palsy: J Otol, 2015; 10(1); 7-12
3. Omori H, Ikeda M, Kukimoto N, Activation of intravascular coagulation in Bell’s palsy: Eur Arch Otorhinolaryngol, 1994; S471-72
4. Elkind MS, Infectious burden: A new risk factor and treatment target for atherosclerosis: Infect Disord Drug Targets, 2010; 10(2); 84-90
5. Sittel C, Stennert E, Prognostic value of electromyography in acute peripheral facial nerve palsy: Otol Neurotol, 2001; 22(1); 100-4
6. Kum RO, Yurtsever Kum N, Ozcan M, Elevated neutrophil-to-lymphocyte ratio in Bell’s palsy and its correlation with facial nerve enhancement on MRI: Otolaryngol Head Neck Surg, 2015; 152(1); 130-35
7. Hong HS, Yi BH, Cha JG, Enhancement pattern of the normal facial nerve at 3.0 T temporal MRI: Br J Radiol, 2010; 83(986); 118-21
8. Li Q, Yang Y, Reis C, Cerebral small vessel disease: Cell Transplant, 2018; 27(12); 1711-22
9. Shi Y, Li S, Li W, MRI lesion load of cerebral small vessel disease and cognitive impairment in patients with CADASIL: Front Neurol, 2018; 9; 862
10. Zhu H, Li Z, Lv J, Zhao R, Effects of cerebral small vessel disease on the outcome of patients with ischemic stroke caused by large artery atherosclerosis: Neurol Res, 2018; 40(5); 381-90
11. Doerner J, Kupczyk PA, Wilsing M, Cerebral white matter lesion burden is associated with the degree of aortic valve calcification and predicts peri-procedural cerebrovascular events in patients undergoing transcatheter aortic valve implantation (TAVI): Catheter Cardiovasc Interv, 2018; 91(4); 774-82
12. Kum RO, Baklaci D, Ozcan M, Increased risk of cerebral white matter lesions in obstructive sleep apnea syndrome: Sleep and Biological Rhythms, 2017; 15(1); 49-55
13. Fazekas F, Chawluk JB, Alavi A, MR signal abnormalities at 1.5 T in Alzheimer’s dementia and normal aging: Am J Roentgenol, 1987; 149(2); 351-56
14. Baugh RF, Basura GJ, Ishii LE, Clinical practice guideline: Bell’s palsy: Otolaryngol Head Neck Surg, 2013; 149(3 Suppl); S1-27
15. Omori H, Ikeda M, Intravascular hypercoagulability in patients with recent Bell’s palsy: Eur Arch Otorhinolaryngol, 1994; 251(5); 278-82
16. Pantoni L, Cerebral small vessel disease: From pathogenesis and clinical characteristics to therapeutic challenges: Lancet Neurol, 2010; 9(7); 689-701
17. Vermeer SE, Den Heijer T, Koudstaal PJ, Incidence and risk factors of silent brain infarcts in the population-based Rotterdam Scan Study: Stroke, 2003; 34(2); 392-96
18. Hachinski VC, Potter P, Merskey H, Leuko-araiosis: An ancient term for a new problem: Can J Neurol Sci, 1986; 13(4 Suppl); 533-34
19. de Leeuw FE, de Groot JC, Achten E, Prevalence of cerebral white matter lesions in elderly people: aA population based magnetic resonance imaging study. The Rotterdam Scan Study: J Neurol Neurosurg Psychiatry, 2001; 70(1); 9-14
20. Grueter BE, Schulz UG, Age-related cerebral white matter disease (leukoaraiosis): A review: Postgrad Med J, 2012; 88(1036); 79-87
21. Enzinger C, Fazekas F, Ropele S, Schmidt R, Progression of cerebral white matter lesions – clinical and radiological considerations: J Neurol Sci, 2007; 257(1–2); 5-10
22. Thomas AJ, O’Brien JT, Davis S, Ischemic basis for deep white matter hyperintensities in major depression: A neuropathological study: Arch Gen Psychiatry, 2002; 59(9); 785-92
23. Riga M, Kefalidis G, Danielides V, The role of diabetes mellitus in the clinical presentation and prognosis of Bell palsy: J Am Board Fam Med, 2012; 25(6); 819-26
24. Liou LS, Chang CY, Chen HJ, Increased risk of peripheral arterial occlusive disease in patients with Bell’s palsy using population data: PLoS One, 2017; 12(12); e0188982
25. Jonsson L, Hemmingsson A, Thomander L, Magnetic resonance imaging in patients with Bell’s palsy: Acta Otolaryngol Suppl, 1989; 468; 403-5
26. Fusconi M, Attanasio G, Capitani F, Is there a relation between sudden sensorineural hearing loss and white matter lesions? Eur Arch Otorhinolaryngol, 2019; 276(11); 3043-49
27. Ciorba A, Bianchini C, Crema L, White matter lesions and sudden sensorineural hearing loss: J Clin Neurosci, 2019; 65; 6-10
28. Shin SH, Wan Byun S, Jin Kim S, Clinical significance of white matter lesions in idiopathic sudden sensorineural hearing loss: J Int Adv Otol, 2022; 18(2); 100-5
29. Hopkins RO, Beck CJ, Burnett DL, Prevalence of white matter hyperintensities in a young healthy population: J Neuroimaging, 2006; 16(3); 243-51
Figures
 Figure 1. Axial section of periventricular white matter lesions in a T2 weighted magnetic resonance image. The lesions in cerebral white matter are shown (arrows) in a patient with House-Brackmann grade 5 Bell’s palsy. This figure was created using PowerPoint 2013 (Microsoft).
Figure 1. Axial section of periventricular white matter lesions in a T2 weighted magnetic resonance image. The lesions in cerebral white matter are shown (arrows) in a patient with House-Brackmann grade 5 Bell’s palsy. This figure was created using PowerPoint 2013 (Microsoft).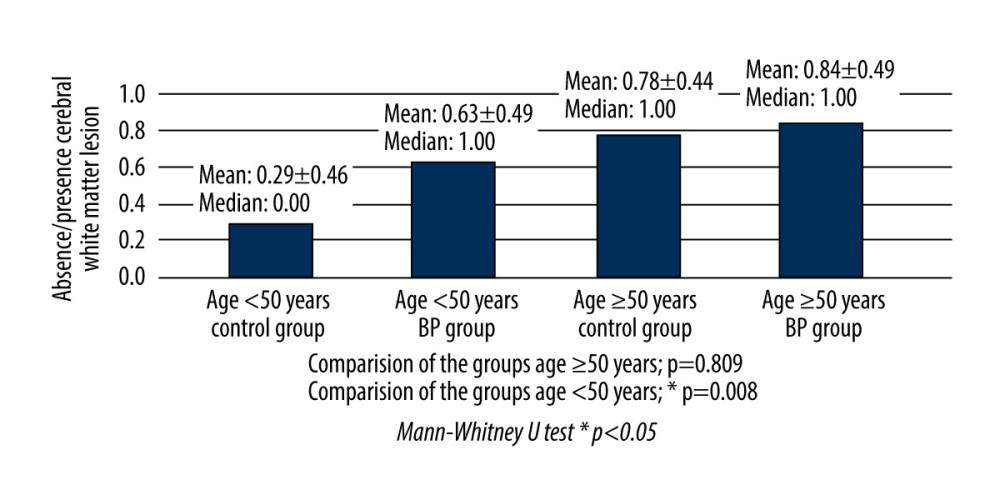 Figure 2. Comparisons of cerebral white matter lesions in Bell’s palsy and control patients, in patients grouped according to age: ≥50 years and <50 years.
Figure 2. Comparisons of cerebral white matter lesions in Bell’s palsy and control patients, in patients grouped according to age: ≥50 years and <50 years. Tables
 Table 1. The demographic characteristics and the variables studied in Bell’s palsy patients and the control group patients.
Table 1. The demographic characteristics and the variables studied in Bell’s palsy patients and the control group patients. Table 2. Comparisons of cerebral white matter lesions within the Bell’s palsy group according to House-Brackmann grade.
Table 2. Comparisons of cerebral white matter lesions within the Bell’s palsy group according to House-Brackmann grade. Table 3. Correlation between cerebral white matter lesions and House-Brackmann grade.
Table 3. Correlation between cerebral white matter lesions and House-Brackmann grade. Table 1. The demographic characteristics and the variables studied in Bell’s palsy patients and the control group patients.
Table 1. The demographic characteristics and the variables studied in Bell’s palsy patients and the control group patients. Table 2. Comparisons of cerebral white matter lesions within the Bell’s palsy group according to House-Brackmann grade.
Table 2. Comparisons of cerebral white matter lesions within the Bell’s palsy group according to House-Brackmann grade. Table 3. Correlation between cerebral white matter lesions and House-Brackmann grade.
Table 3. Correlation between cerebral white matter lesions and House-Brackmann grade. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








