26 September 2023: Clinical Research
Comparing the Effectiveness and Safety of MedAn with the Nishikawa Blade and UE Videolaryngoscopes for Left-Sided Double-Lumen Endobronchial Tube Intubation: A Randomized Controlled Trial
Yong Zhang1ACE, Wenwen Zhang1ACE, Shuai Wang1ACE, Hailing Yin1BC, Yajie XuDOI: 10.12659/MSM.940916
Med Sci Monit 2023; 29:e940916
Abstract
BACKGROUND: The purpose of this study was to compare the effectiveness and safety of the MedAn videolaryngoscope with the Nishikawa blade (MedAn) vs the UE videolaryngoscope (UE) for intubation with a left-sided double-lumen endobronchial tube (LDLT) in patients with normal airways.
MATERIAL AND METHODS: We randomly categorized 106 patients scheduled to undergo elective thoracic surgery with LDLT for one-lung ventilation into 2 groups: the UE group (Group UE) and the MedAn group (Group MedAn), using the MedAn or UE for LDLT intubation. The primary outcome was time to successful intubation. The Cormack-Lehane classification of laryngeal view was the key secondary outcome. Other secondary outcomes included first-attempt and overall intubation success rates, laryngoscopy time, LDLT placement time, operators’ subjective evaluation of videolaryngoscopes, hemodynamic changes during videolaryngoscopic intubation, and adverse outcomes.
RESULTS: The time to successful intubation and LDLT placement time of Group MedAn were 42.0 (32.35, 47.0) s and 23.0 (18.0, 26.0) s, and it was shorter than in Group UE (median, 42 s vs 49 s, 23 s vs 30 s, P<0.001). Group MedAn had a better laryngeal view (P=0.03) and less subglottic/tracheal mucosal injury (P<0.001) than Group UE. Moreover, the operators’ subjective grading of ease of laryngoscopy, quality of view, and ease of LDLT placement were higher in Group MedAn than in Group UE (P<0.05).
CONCLUSIONS: Compared with the UE, the MedAn could reduce the intubation time and provide a better laryngeal view and sufficient intubation space for safer LDLT intubation in patients with normal airways.
Keywords: Laryngoscopes, Intubation, Anesthesia, Endotracheal, Humans, Larynx, Elective Surgical Procedures, one-lung ventilation, Intubation, Intratracheal
Background
In thoracic surgery, left-sided double-lumen endobronchial tubes (LDLTs) are widely applied for one-lung ventilation [1]. Intubation with LDLTs is more challenging than with single-lumen tracheal tubes due to their longer catheter, larger diameter, and more complicated construction. Inappropriate intubation can cause upper-airway injury, postoperative sore throat, hoarseness, and even tracheobronchial rupture, which usually occurs in the lower part of the trachea or the membrane of the left main-stem bronchus [2,3]. Moreover, during LDLT intubation, stimulation of the oropharynx and tracheal mucosa can cause drastic hemodynamic changes that can lead to acute cardiovascular adverse events [4,5].
Although LDLTs intubation can be challenging, it is important to minimize intubation time and improve success rates, especially for patients at high risk of reflux aspiration and hypoxemia. Attempting to intubate a patient multiple times or spending too much time intubating them can lead to low pulse oximetry, increasing the risk of hypoxia. This can result in serious health issues such as arrhythmias, hypotension, and hypoxic brain damage. Studies have shown that multiple intubation attempts and rapid sequence induction durations longer than 180 s are associated with hypoxia. Therefore, finding a suitable intubation tool for LDLTs is crucial for effective airway management [6,7].
Videolaryngoscopy is preferred for difficult airway management, as it can help with glottic vision and increase the success rate of first-time intubations. Several types of videolaryngoscopes are effective for LDLT intubation [6–9]. The UE videolaryngoscope, with a similar blade shape to the Macintosh laryngoscope, is widely available in China. The distal portion (length, 3 cm) of the UE blade is relatively flat, which cannot prevent enlargement of the tongue base and hamper the blade tip from approaching the vallecula to lift the epiglottis sufficiently in patients with difficult laryngoscopy, causing difficulty in adjusting the direction of the tip of the bronchial tube due to the limited glottic opening and hypopharyngeal space [10] (Figure 1A). However, the MedAn videolaryngoscope with the Nishikawa blade, which is a new videolaryngoscope first commercially available in 2018, can better expose the glottis and create a larger pharyngeal space, reducing the incidence of unexpected difficult laryngoscopy by controlling the tongue base [11,12]. The S-shaped Nishikawa blade is superior to the Macintosh blade for displacing the tongue back to the oral cavity to increase intubation space at the hypopharynx, which is beneficial for adjusting the position and direction of a tracheal tube during laryngoscopy (Figure 1B). Moreover, the bifid tip of the Nishikawa blade makes it easy to approach the vallecula and exerts more pressure in the vallecula area to raise the epiglottis for ease of intubation [11]. Although the S-shaped Nishikawa blade can elevate the epiglottis to keep the glottis patent in patients with difficult laryngoscopy, the laryngeal view needed to be further improved. Therefore, 3 modifications of the S-shaped Nishikawa blade have been made for use with the MedAn to balance the dynamic and optical performances of the videolaryngoscope for ease of intubation. Despite differences in blade design, camera position, and video screens, no studies comparing the efficiency of these 2 videolaryngoscopes for LDLT intubation have been undertaken. Therefore, this study aimed to compare the clinical effectiveness between the UE and the MedAn for LDLT intubation.
Material and Methods
ETHICS AND REGISTRATION:
This single-blind, prospective, randomized controlled trial was approved by the Chinese Registered Clinical Trial Ethics Committee (ChiECRCT20210119) and registered with the Chinese Clinical Trial Center (ChiCTR2100049279) before patient enrollment. Every patient signed a written informed consent.
STUDY PARTICIPANTS:
This study included patients who were scheduled for elective thoracic surgery using LDLT for one-lung ventilation with an American Society of Anesthesiologists (ASA) physical status Class I–III, body mass index (BMI) <30 kg/m2, and age between 18 and 80 years. Exclusion criteria were patients with risk of aspiration and/or known or possible difficult airways, meeting 1 or more of the following conditions: thyromental distance <6.0 cm, Mallampati classification score >III, inter-incisor distance <3.0 cm, possibility of difficult laryngoscopy exposure or mask ventilation, and history of tracheal intubation while awake.
RANDOMIZATION AND BLINDING:
The videolaryngoscope for LDLT intubation included the UE videolaryngoscope with the TDC-3 blade (Youyi Medical Equipment Co., Ltd., Zhejiang, China) (Figure 1C) and the MedAn videolaryngoscope (MedAn Medical Corp., Tianjin, China) with the Nishikawa blade (ZH-LS) (Figure 1D).
Using randomization computer software (SPSS version 24.0; SPSS, Inc., Chicago, IL, USA), each patient was randomly assigned to the UE (Group UE) or the MedAn (Group MedAn) group in a 1: 1 ratio. The grouping assignments were kept in a sealed opaque envelope that was only accessed by the researchers before the induction of anesthesia. A blinded research team member conducted postoperative interviews with patients. The group allocation was hidden from all patients, perioperative follow-up assessors, and statisticians. Intubation was performed by one of the anesthetist-authors, who has performed over 1000 intubations, with 8 years of work experience [13]. The anesthesiologist used both manikins and patients to practice on until he felt competent with each type of video laryngoscope during intubation [14]. In addition, another airway specialist also rated him as being familiar with the video laryngoscope intubation process.
INTERVENTIONS AND ANESTHESIA:
After entering the operating room, the patients were instructed to lie on the operating bed in the sniffing position with a pillow (height, 7 cm). Electrocardiogram parameters, heart rate (HR), mean arterial pressure (MAP), pulse oxygen saturation (SpO2), bispectral index (BIS), and end-tidal carbon dioxide were monitored. Anesthesiologists chose the appropriate LDLT (28F, 32F, 35F, 37F, or 39F) according to the left bronchial diameter, as measured on chest computed tomography or based on clinical experience. After 5 min of pre-oxygenation, general anesthesia was induced with midazolam (0.03 mg/kg), propofol (1.5–2.5 mg/kg), sufentanil (0.3–0.6 μg/kg), rocuronium (0.6 mg/kg), and remifentanil (0.2–0.3 μg/kg/min). After the cessation of spontaneous breathing, assisted ventilation was provided. When the train-of-four count value was 0 and the BIS value was below 60, a videolaryngoscope (either UE or MedAn, depending on the allotted group) was inserted from the right side of the mouth and the tongue was subsequently moved to the left side. After confirming the epiglottis on the screen, the videolaryngoscope blade tip was advanced into the vallecula to lift the epiglottis. If necessary, backward upward rightward pressure was used to achieve an optimal laryngeal view. The stylet was removed after the bronchial cuff passed the glottis and LDLT was turned 90° to the left. Using the method reported by Park et al, under the guidance of a fiberoptic bronchoscope (FOB), the LDLT was advanced into the left main bronchus [15]. To determine the correct position of LDLT, the tracheal lumen had to allow visualization of the bronchial cuff immediately below the carina and the bronchial lumen has to allow viewing of the left upper- and lower-lobe bronchi [16].
We defined successful intubation as the insertion of the bronchial cuff of LDLT through the glottis within 180 s, which was finally confirmed by the end-tidal carbon dioxide waveform or direct view of the tracheal ring from the FOB. If there were 2 failed intubation attempts, airway damage, videolaryngoscope technology failure, or SpO2 drop below 90%, the patient was excluded from the analysis. Two attempts at LDLT intubation with the same videolaryngoscope were permitted. After 2 failed intubation attempts, the anesthesiologist followed the 2015 British Difficult Airway Society guidelines for the management of unanticipated difficult intubation in adults [17].
OUTCOMES AND DATA COLLECTION:
Time to successful intubation was the primary outcome parameter, defined as the time from insertion of the videolaryngoscope into the mouth until the bronchial cuff of LDLT passed through the glottis, as confirmed by the end-tidal carbon dioxide waveform or direct view of the tracheal ring from the FOB.
The Cormack-Lehane (C/L) classification grade was the secondary outcome. Although the C/L classification is used for assessing the laryngeal view during direct laryngoscopy, to the best of our knowledge, there is no more rational assessment for the laryngeal view during video laryngoscopy in clinical practice now. Most published papers on video laryngoscopy still use the C/L classification. The other secondary outcomes included the following parameters: 1) laryngoscopy time, defined as the time from the videolaryngoscope inserted into the mouth until the appropriate laryngeal view was obtained; 2) LDLT placement time, defined as the time from LDLT placed into the mouth until the bronchial cuff passed through the glottis, as confirmed by the end-tidal carbon dioxide waveform or direct view of the tracheal ring from the FOB; 3) overall intubation success rate and first-attempt intubation success rate; 4) rate of adverse events (eg, subglottic/tracheal mucosal injury, oropharyngeal soft-tissue injury, hoarseness, sore throat, dysphagia, nausea, and vomiting); and 5) causes of failed intubation. Additionally, subjective evaluations of anesthesiologists included the ease of laryngoscopy, quality of view, and ease of tube advancement on a subjective scale (excellent, good, fair, and poor). Changes in MAP and HR were recorded before anesthesia induction (T0), 1 min before intubation (T1), and 1 min (T2), 2 min (T3), and 3 min (T4) after intubation.
The subglottic/tracheal mucosal injury was classified into 3 grades and scored as follows: 0 grade: normal mucosa without damage (0 points); 1 grade: mucosal hyperemia and/or mild submucosal hematoma (1 point); 2 grade: moderate submucosal hematoma (2 points); and 3 grade: mucosal defect and/or mucosal hemorrhage (3 points). Each mucosal injury from the glottis to the carina was graded and scored according to the above grading system.
Sample Size
STATISTICAL ANALYSIS:
The statistical analysis was carried out using SPSS software version 24.0 (SPSS, Inc., Chicago, IL, USA). The Shapiro-Wilk test was used to verify whether the data was normal, and Levene’s test was used to determine variance homogeneity. Normally distributed data were expressed as means±standard deviation, and the independent samples
Results
BASELINE PATIENT AND AIRWAY CHARACTERISTICS:
We evaluated date from 111 patients enrolled for elective thoracic surgery using LDLTs, and 5 of these were excluded according to the exclusion criteria. Finally, 106 patients were randomly assigned; 1 patient was intubated by UE unsuccessfully, 1 patient was intubated by MedAn unsuccessfully, and 1 patient required long-term mechanical ventilation in the intensive care unit. The flow diagram of the patient selection process is presented in Figure 2. The basic patient characteristics and airway assessment indicators did not differ statistically significantly between groups (Table 1).
INTUBATION TIME:
Group MedAn had significantly shorter time to successful intubation and LDLT placement times than Group UE (P<0.01). However, a significant difference in laryngoscopy time was not observed between groups (P>0.05; Table 2).
LARYNGEAL VIEW AND SUBJECTIVE EVALUATIONS OF ANESTHESIOLOGISTS:
Both groups had significantly different laryngeal views graded using the C/L classification (P=0.03; Table 3). The subjective evaluation of inserting the videolaryngoscope into the mouth, exposing the glottis, and placing the LDLT into the trachea was better in Group MedAn than in Group UE (P<0.05; Table 3).
INTUBATION SUCCESS RATE:
In Group MedAn, first-attempt and overall intubation success rates were 98.1% (52/53) and 98.1% (52/53), respectively. In Group UE, both rates were 96.2% (51/53) and 98.1% (52/53), respectively. The main reason for tracheal intubation failure on the first attempt was the inability to attain adequate glottic exposure in both groups.
COMPLICATIONS OF INTUBATION:
Group MedAn had a lower incidence of subglottic/tracheal mucosal injury than Group UE (median, 1 vs 2.5, P<0.001), and no statistically significant differences were found in the incidence of failed intubation, oropharyngeal soft-tissue injury, hoarseness, sore throat, dysphagia, nausea, or vomiting between both groups (Table 4).
COMPARISON OF HEMODYNAMIC INDICATORS AT DIFFERENT TIME POINTS:
The MAP and HR were not statistically significant between T0 and T4 (
Discussion
This study showed that MedAn, compared with UE, provided a better laryngeal view and reduced the intubation time by shortening the LDLT placement time and lowering the subglottic/tracheal mucosal injury incidence in patients with normal airways. MedAn also obtained a better subjective evaluation to facilitate glottic exposure and LDLT placement. We did not observe a significant difference in laryngoscopy time or overall and first-attempt intubation success rates between the 2 groups.
LDLT intubation includes 2 key steps: obtaining a laryngeal view and inserting the tip of the bronchial tube through the vocal cords [20]. Therefore, a clear laryngeal view with enough intubation space during laryngoscopy is crucial for LDLT placement.
The findings mentioned above were inconsistent with those of our previous study in which the MedAn shortened the tracheal intubation time in obese patients compared to UE [21]. However, this study showed no significant differences in laryngoscopy time between the both videolaryngoscopes, suggesting that both videolaryngoscopes could effectively control the tongue and lift the epiglottis to expose the glottis in patients with normal airways (BMI <30 kg/m2 without the abnormal enlargement of the tongue).
Group MedAn had a significantly better experience with device insertion, quality of view, and tube advancement than those in Group UE, as determined by the subjective evaluation by operators. Because the blade tip of MedAn is shown on the screen as visual guidance that enables the adjustment of the blade tip position precisely to obtain the optimal laryngeal view and avoid soft-tissue injury during videolaryngoscopy, Risse et al found that the most challenging steps of intubation using videolaryngoscopy were LDLT insertion and advancing it into the trachea [22]. In this study, the bronchial tip of LDLT was bent along the curvature of the videolaryngoscope blade (Figure 1). The level of laryngeal visualization and the need for external laryngeal manipulation had a significant impact on the occurrence of adverse outcomes [23]. MedAn can provide a better laryngeal view and larger intubation space to facilitate the insertion and rotation of LDLT, reducing the risk of oropharyngeal soft-tissue damage [24]. Therefore, the subjective evaluation of the quality of view and ease of tube advancement in Group MedAn was better than in Group UE.
The MAP and HR of patients in both groups increased when LDLT was placed in the correct position, which could be caused by the thick and hard LDLT stimulating the tracheal mucosa.
In the operator’s subjective evaluation, 78.4% of patients were rated “excellent” regarding glottic exposure, while only 68.6% of patients were rated “excellent” for LDLT placement, which is a common problem in videolaryngoscopic intubation; good glottic visualization does not always guarantee the easement of tracheal tube placement. Videolaryngoscopes offer a better view with less force by allowing one to functionally “look around the corner” due to the angle of the device and the presence of a camera distal to where the eye can see. However, it is important to note that the use of a videolaryngoscope may narrow the glottic inlet by reducing the angle between the epiglottis and the anterior wall of the larynx. The tip of the bronchial lumen tends to impinge upward on the anterior laryngeal wall or laryngotracheal migration, causing difficulties in LDLT insertion and rotation during LDLT placement. Moreover, videolaryngoscopes provide a two-dimensional view of the larynx on the screen separated from the intubation space and glottic opening, making LDLT intubation difficult.
The overall intubation success rate was 98.1%, and the first-attempt success rates were not significantly different between Group UE (96.2%) and Group MedAn (98.1%). These results are consistent with those of a previous study [20]. In Group UE, intubation failed in 1 patient with a long U-shaped epiglottis because it was difficult to lift the epiglottis indirectly with UE (Figure 3A). Subsequently, the second-attempt intubation with MedAn was successful. Because the blade tip of MedAn was accurately placed beneath the epiglottis with the blade tip guidance on the screen, the epiglottis was directly lifted to expose the glottis (C/L grade 1) (Figure 3B). In Group MedAn, intubation failed in 1 patient with excessive sputum preoperatively, which obscured the camera lens of MedAn from obtaining an adequate laryngeal view during laryngoscopy. The patient was intubated successfully after sufficient suction. The indirect laryngeal view of the videolaryngoscope will be invisible due to excessive oral secretions; therefore, anti-fog and effective oral secretion suction devices are essential during videolaryngoscopic intubation.
This study has 2 limitations: (1) the performance of the 2 videolaryngoscopes could not be compared in a double-blind study; (2) this study involved patients with normal airways at a single center, and multicenter study to comprehensively assess the performance of MedAn for difficult airways (caused, for example, by limited cervical spine movement) should be performed because different types of videolaryngoscopes should be chosen depending on the type of difficult airway [25,26]; and (3) a comparative study was not conducted on other video laryngoscopes (eg, McGrath MAC X-Blade and C-MAC D-Blade), but only on these 2.
Conclusions
This study found that MedAn was effective and safe for LDLT intubation in patients with normal airways. MedAn can optimize glottic exposure and shorten LDLT placement time to reduce the total intubation time, improving the operator’s subjective evaluation of the videolaryngoscopic LDLT placement and reducing subglottic/tracheal mucosal damage when compared to UE.
Figures
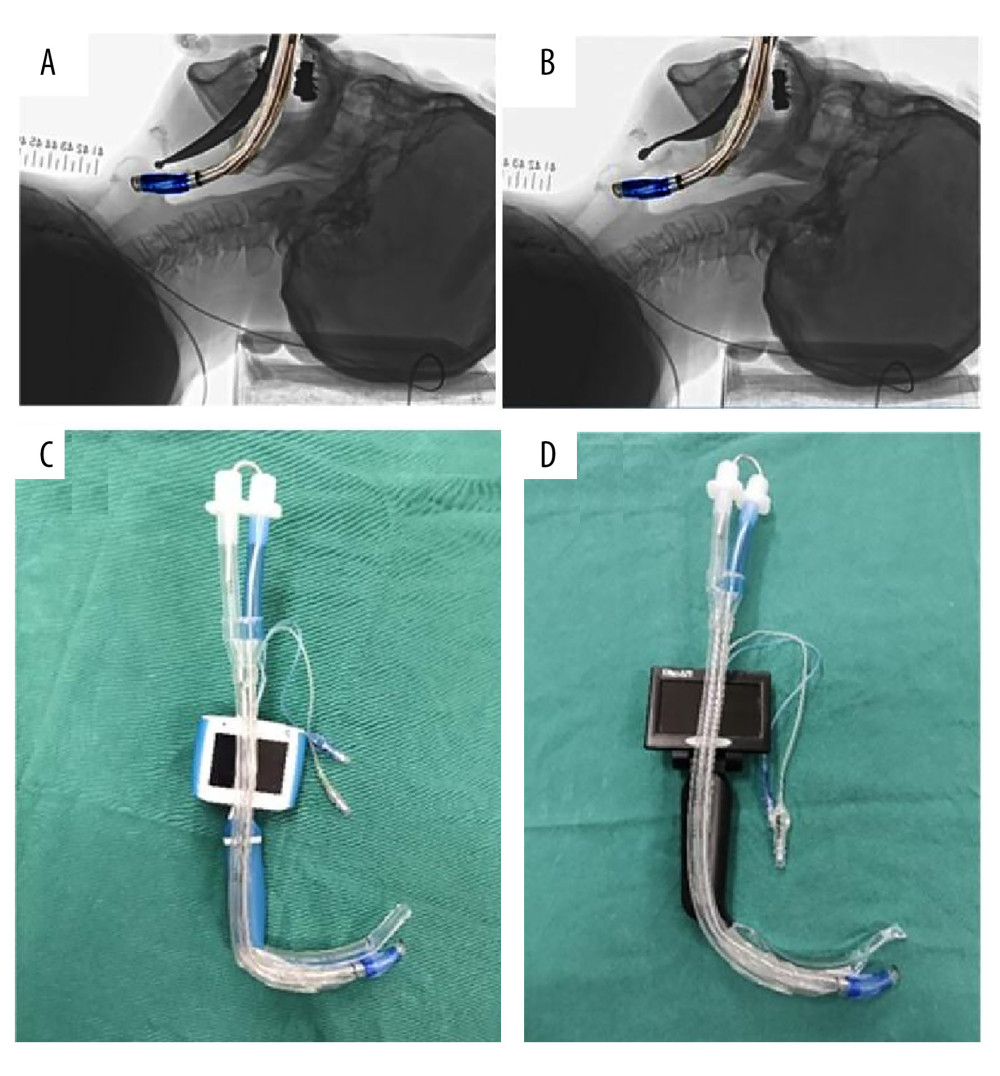 Figure 1. Two types of videolaryngoscopes and the left-sided double-lumen tube (LDLT). (A) Superimposing the image of the hockey stick-shaped LDLT on the radiograph laryngoscopy image with the Macintosh laryngoscope. (B) Superimposing the image of the hockey stick-shaped LDLT on the radiograph laryngoscopy image with the direct Nishikawa blade. (C) The UE videolaryngoscope and hockey stick-shaped LDLT. (D) The MedAn videolaryngoscope with the Nishikawa blade and LDLT.
Figure 1. Two types of videolaryngoscopes and the left-sided double-lumen tube (LDLT). (A) Superimposing the image of the hockey stick-shaped LDLT on the radiograph laryngoscopy image with the Macintosh laryngoscope. (B) Superimposing the image of the hockey stick-shaped LDLT on the radiograph laryngoscopy image with the direct Nishikawa blade. (C) The UE videolaryngoscope and hockey stick-shaped LDLT. (D) The MedAn videolaryngoscope with the Nishikawa blade and LDLT. 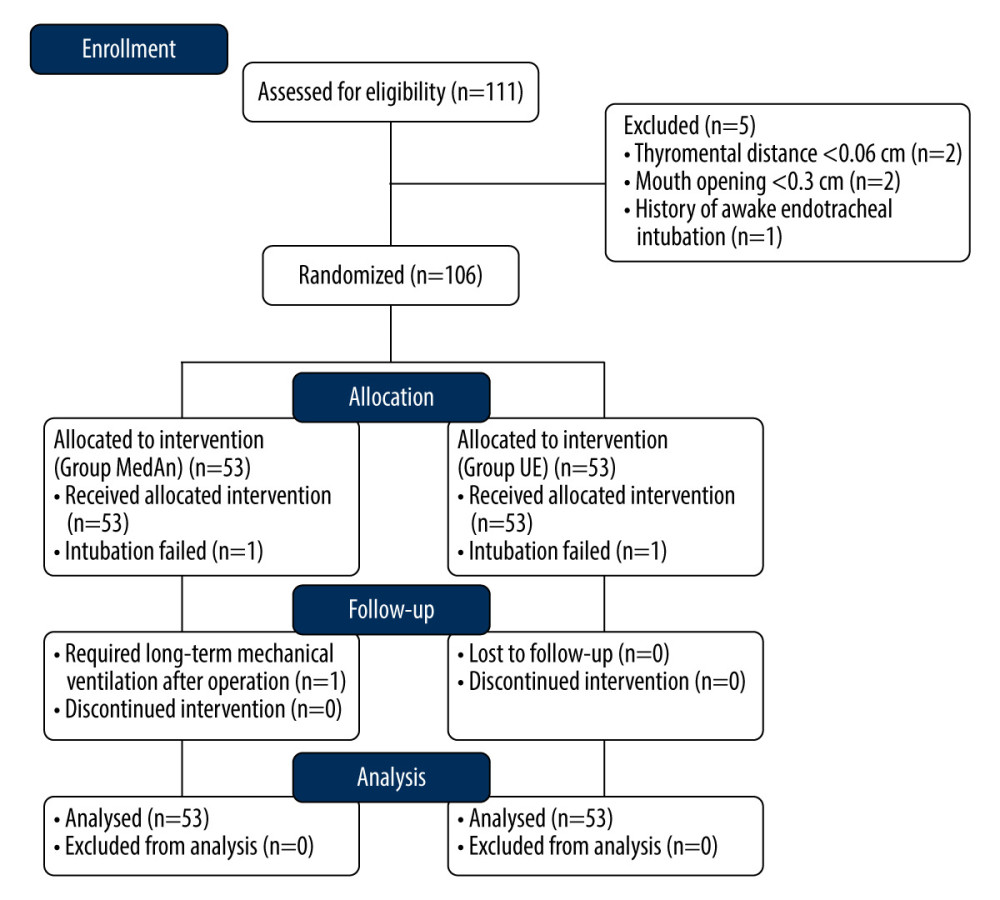 Figure 2. Flow diagram of patient enrollment.
Figure 2. Flow diagram of patient enrollment. 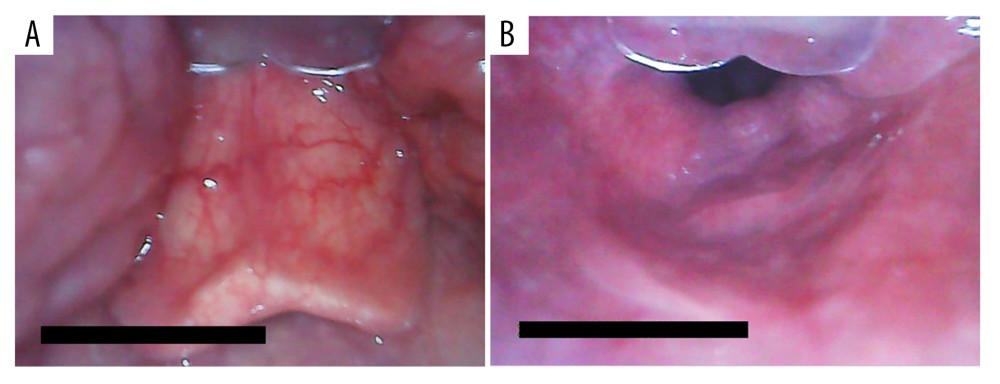 Figure 3. Glottic exposure with the MedAn videolaryngoscope. (A) The long U-shaped epiglottis observed with the MedAn videolaryngoscope by indirectly lifting the epiglottis. (B) The laryngeal view observed with the MedAn videolaryngoscope by directly lifting the epiglottis.
Figure 3. Glottic exposure with the MedAn videolaryngoscope. (A) The long U-shaped epiglottis observed with the MedAn videolaryngoscope by indirectly lifting the epiglottis. (B) The laryngeal view observed with the MedAn videolaryngoscope by directly lifting the epiglottis. References
1. Lu Y, Dai W, Zong Z, Bronchial blocker versus left double-lumen endotracheal tube for one-lung ventilation in right video-assisted thoracoscopic surgery: J Cardiothorac Vasc Anesth, 2018; 32; 297-301
2. Liu S, Mao Y, Qiu P, Airway rupture caused by double-lumen tubes: A review of 187 cases: Anesth Analg, 2020; 131(5); 1485-90
3. Qiu Y, Huang C, Zhang Z, Incidence and risk factors of vocal cord injuries after intubation of double-lumen endobronchial tubes under UEScopes: A prospective observational cohort study: J Clin Anesth, 2020; 60; 62-63
4. Mathew A, Chandy J, Punnoose J, A randomized controlled study comparing CMAC video laryngoscope and Macintosh laryngoscope for insertion of double lumen tube in patients undergoing elective thoracotomy: J Anaesthesiol Clin Pharmacol, 2021; 37(2); 266-71
5. Kim TK, Hong DM, Lee SH, Effect-site concentration of remifentanil required to blunt haemodynamic responses during tracheal intubation: A randomized comparison between single- and double-lumen tubes: J Int Med Res, 2018; 46(1); 430-39
6. Gopinath B, Sachdeva S, Kumar A, Kumar G, Advancing emergency airway management by reducing intubation time at a high-volume academic emergency department: BMJ Open Qual, 2021; 10(Suppl 1); e001448
7. Liu TT, Li L, Wan L, Zhang CH, Yao WL, Videolaryngoscopy vs. Macintosh laryngoscopy for double-lumen tube intubation in thoracic surgery: A systematic review and meta-analysis: Anaesthesia, 2018; 73(8); 997-1007
8. Defosse J, Kleinschmidt J, Schmutz A, Dental strain on maxillary incisors during tracheal intubation with double-lumen tubes and different laryngoscopy techniques – a blinded mannequin study: J Cardiothorac Vasc Anesth, 2022; 36(8 Pt B); 3021-27
9. Lewis SR, Butler AR, Parker J, Videolaryngoscopy versus direct laryngoscopy for adult patients requiring tracheal intubation: A Cochrane systematic review: Br J Anaesth, 2017; 119; 369-83
10. Xue FS, Yang BQ, Liu YY, Current evidences for the use of UEscope in airway management: Chin Med J (Engl), 2017; 130; 1867-75
11. Nishikawa K, Yamada K, Sakamoto A, A new curved laryngoscope blade for routine and difficult tracheal intubation: Anesth Analg, 2008; 107; 1248-52
12. Wang M, Huang H, Luo C, Nishikawa K, Initial clinical experience of the MedAn videolaryngoscope with the Nishikawa blade: Trends in Anaesthesia and Critical Care, 2022; 44; 36-42
13. Anandraja R, Karthekeyan BR, A comparative study of haemodynamic effects of single-blinded orotracheal intubations with intubating laryngeal mask airway, Macintosh and McGrath video laryngoscopes: Anaesthesiol Intensive Ther, 2021; 53(1); 30-36
14. Kleine-Brueggeney M, Greif R, Schoettker P, Evaluation of six videolaryngoscopes in 720 patients with a simulated difficult airway: A multicentre randomized controlled trial: Br J Anaesth, 2016; 116(5); 670-79
15. Park JW, Jo JH, Park JH, Comparison of conventional and fibreoptic-guided advance of left-sided double-lumen tube during endobronchial intubation: A randomised controlled trial: Eur J Anaesthesiol, 2020; 37(6); 466-73
16. Essandoh M, Andritsos M, Double-Lumen endotracheal tube placement: Knowing depth of insertion firsthand may make a difference: J Cardiothorac Vasc Anesth, 2018; 32(2); 860-62
17. Frerk C, Mitchell VS, McNarry AF, Difficult Airway Society 2015 guidelines for management of unanticipated difficult intubation in adults: Br J Anaesth, 2015; 115; 827-48
18. Zhao JP, Xiong T, Fan H, Zhang HComparison of HC video laryngoscope and Macintosh laryngoscope in VivaSight double-lumen bronchial intubation: Chinese Physician Journal, 2018; 20(10); 1579-81 [in Chinese]
19. Hsu HT, Kuo YW, Ma CW: BMC Anesthesiol, 2022; 22(1); 260
20. Ozdemirkan A, Onal O, Ozcan IG, Comparison of the intubation success rate between the intubating catheter and videolaryngoscope in difficult airways: A prospective randomized trial: Braz J Anesthesiol, 2022; 72(1); 55-62
21. Zhang WW, Xu YJ, Cao YYEffect of S-type videolaryngoscope-guided tracheal intubation in obese patients: Zhonghua Ma Zui Xue Za Zhi, 2021; 41; 504-5 [in Chinese]
22. Risse J, Schubert AK, Wiesmann T, Videolaryngoscopy versus direct laryngoscopy for double-lumen endotracheal tube intubation in thoracic surgery – a randomised controlled clinical trial: BMC Anesthesiol, 2020; 20; 150
23. Karczewska K, Bialka S, Smereka J, Efficacy and safety of video-laryngoscopy versus direct laryngoscopy for double-lumen endotracheal intubation: A systematic review and meta-analysis: J Clin Med, 2021; 10; 5524
24. Ajimi J, Nishikawa K, Niwa Y, Suzuki T, Successful tracheal intubation in a patient with a large epiglottic cyst using the MedAn video laryngoscope with Nishikawa blade: J Clin Anesth, 2021; 71; 110232
25. Chandy J, Pillai R, Mathew A, A randomized clinical trial comparing the King Vision (channeled blade) and the CMAC (D blade) videolaryngoscopes in patients with cervical spine immobilization: J Anaesthesiol Clin Pharmacol, 2021; 37(4); 604-9
26. Kido H, Komasawa N, Matsunami S, Comparison of McGRATH MAC and Macintosh laryngoscopes for double-lumen endotracheal tube intubation by anesthesia residents: A prospective randomized clinical trial: J Clin Anesth, 2015; 27; 476-80
Figures
 Figure 1. Two types of videolaryngoscopes and the left-sided double-lumen tube (LDLT). (A) Superimposing the image of the hockey stick-shaped LDLT on the radiograph laryngoscopy image with the Macintosh laryngoscope. (B) Superimposing the image of the hockey stick-shaped LDLT on the radiograph laryngoscopy image with the direct Nishikawa blade. (C) The UE videolaryngoscope and hockey stick-shaped LDLT. (D) The MedAn videolaryngoscope with the Nishikawa blade and LDLT.
Figure 1. Two types of videolaryngoscopes and the left-sided double-lumen tube (LDLT). (A) Superimposing the image of the hockey stick-shaped LDLT on the radiograph laryngoscopy image with the Macintosh laryngoscope. (B) Superimposing the image of the hockey stick-shaped LDLT on the radiograph laryngoscopy image with the direct Nishikawa blade. (C) The UE videolaryngoscope and hockey stick-shaped LDLT. (D) The MedAn videolaryngoscope with the Nishikawa blade and LDLT. Figure 2. Flow diagram of patient enrollment.
Figure 2. Flow diagram of patient enrollment. Figure 3. Glottic exposure with the MedAn videolaryngoscope. (A) The long U-shaped epiglottis observed with the MedAn videolaryngoscope by indirectly lifting the epiglottis. (B) The laryngeal view observed with the MedAn videolaryngoscope by directly lifting the epiglottis.
Figure 3. Glottic exposure with the MedAn videolaryngoscope. (A) The long U-shaped epiglottis observed with the MedAn videolaryngoscope by indirectly lifting the epiglottis. (B) The laryngeal view observed with the MedAn videolaryngoscope by directly lifting the epiglottis. Tables
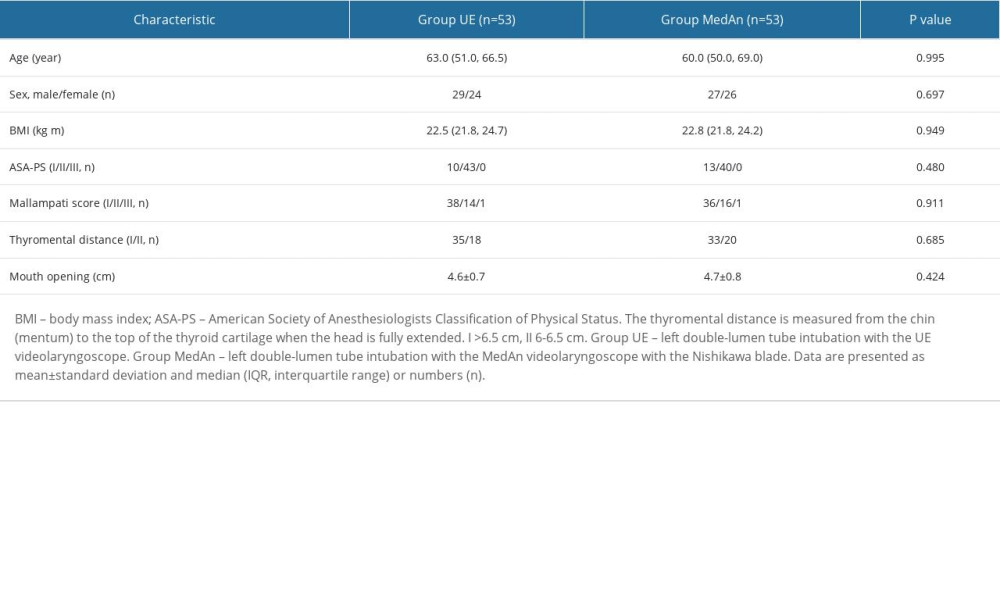 Table 1. Baseline patient and airway characteristics.
Table 1. Baseline patient and airway characteristics.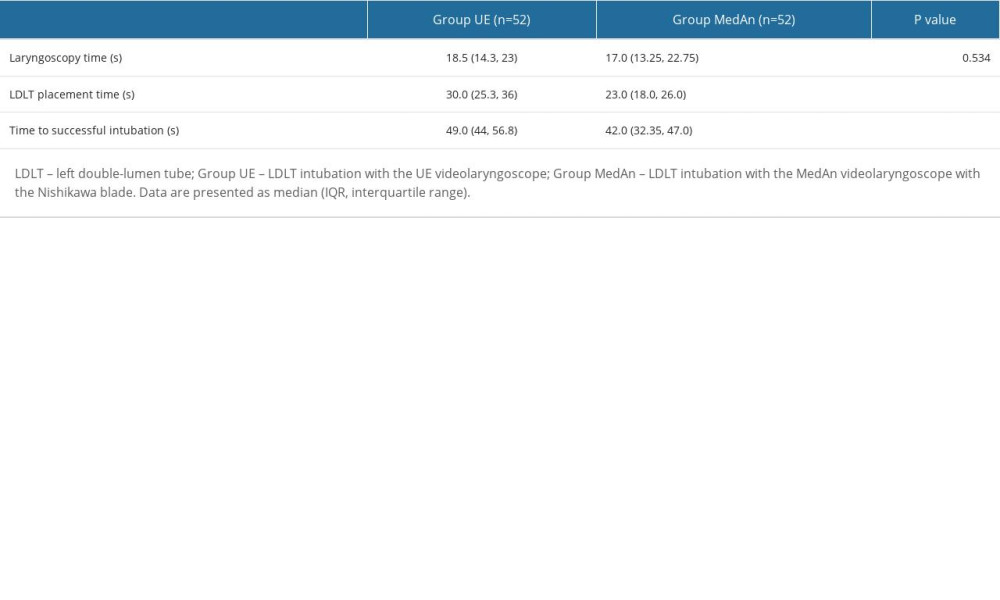 Table 2. Intubation time of LDLT.
Table 2. Intubation time of LDLT.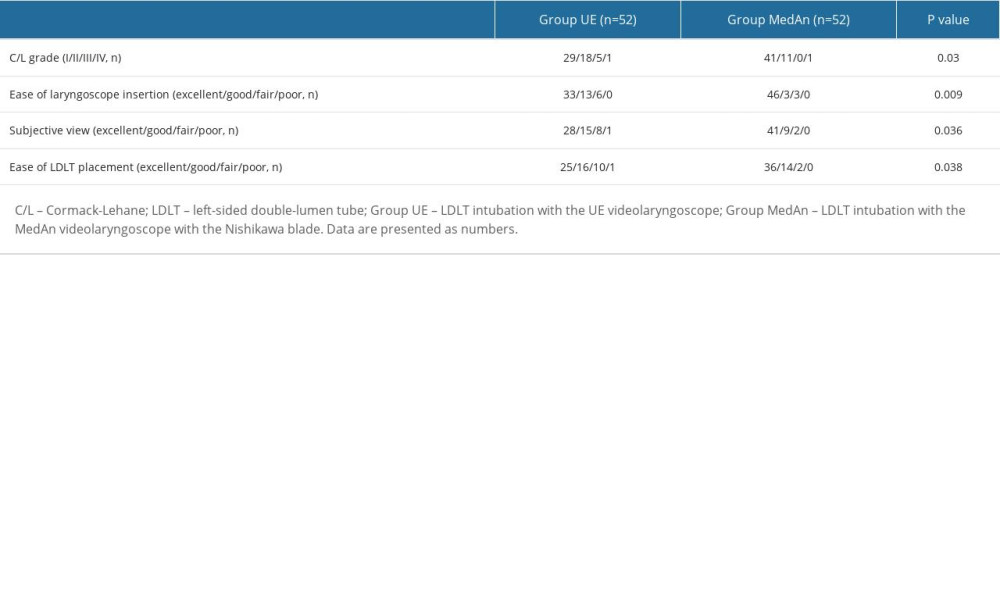 Table 3. Laryngeal view and subjective evaluations of anesthesiologists.
Table 3. Laryngeal view and subjective evaluations of anesthesiologists.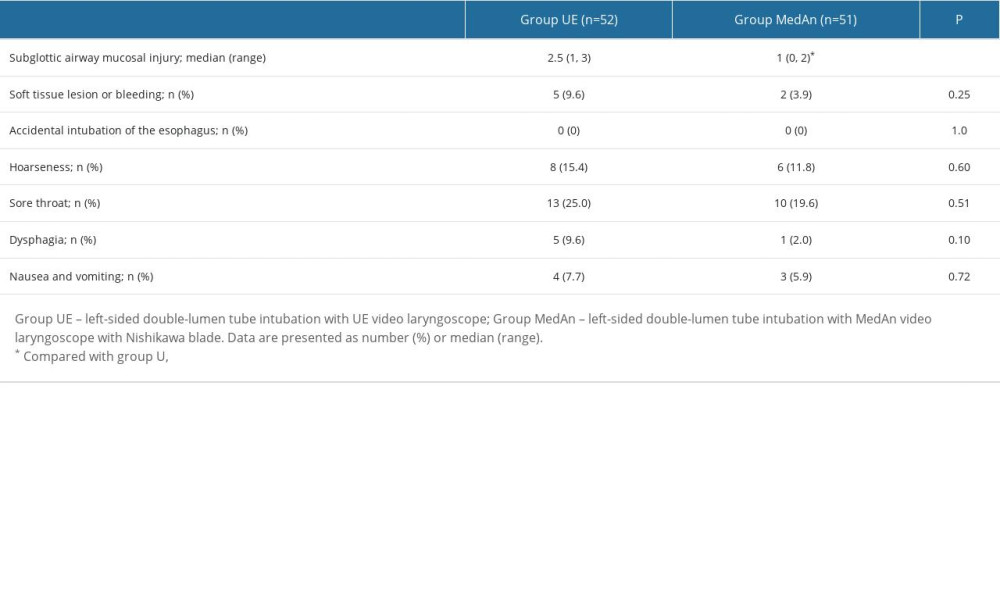 Table 4. Comparison of adverse events after endotracheal intubation.
Table 4. Comparison of adverse events after endotracheal intubation. Table 1. Baseline patient and airway characteristics.
Table 1. Baseline patient and airway characteristics. Table 2. Intubation time of LDLT.
Table 2. Intubation time of LDLT. Table 3. Laryngeal view and subjective evaluations of anesthesiologists.
Table 3. Laryngeal view and subjective evaluations of anesthesiologists. Table 4. Comparison of adverse events after endotracheal intubation.
Table 4. Comparison of adverse events after endotracheal intubation. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








