01 August 2020: Clinical Research
Contributes to the Development of Colorectal Cancer via Recruiting CD11bTLR-4 Cells
Qun Deng1ABCDEFG*, Changjian Wang2BC, Kailin Yu3DE, Yahui Wang1EF, Qinyan Yang2CD, Jingjing Zhang1FG, Xiaoping Xu4AGDOI: 10.12659/MSM.921886
Med Sci Monit 2020; 26:e921886
Abstract
BACKGROUND: An increasing number of studies have demonstrated that Streptococcus bovis and its concomitant inflammatory factors concentrate in the intestine in colorectal cancer (CRC). However, the molecular mechanism of S. bovis on colorectal tumorigenesis remains unclear. This study aimed to explore the role of S. bovis in carcinogenesis and its potential mechanism in CRC of mice orally pretreated with S. bovis.
MATERIAL AND METHODS: The colons of experimental mice were collected and evaluated for the extent of neoplasm. In addition, comparative feces DNA sequencing was adopted to verify the abundance change of S. bovis during the progression of CRC in patients.
RESULTS: The results of this study found that S. bovis is more likely to be present at higher levels in patients with progressive colorectal carcinoma compared to those adenoma patients and healthy volunteers (P<0.05). Pretreatment with S. bovis aggravated tumor formation in mice, resulting in more substantial and a higher number of tumor nodes (P<0.05). A cytokine expression pattern with increased levels of IL-6, Scyb1, Ptgs2, IL-1β, TNF, and Ccl2 was detected in S. bovis pretreated CRC mice (all P<0.05). Furthermore, S. bovis recruited myeloid cells, especially CD11b⁺TLR-4⁺ cells, which could promote pro-tumor immunity in the tumor microenvironment (P<0.05).
CONCLUSIONS: Collectively, our study indicates that S. bovis may induce a suppressive immunity that is conducive to CRC by recruiting tumor-infiltrating CD11b⁺TLR-4⁺ cells. In conclusion, S. bovis contributes to colorectal tumorigenesis via recruiting CD11b⁺TLR-4⁺ cells.
Keywords: Colorectal Neoplasms, Myeloid Cells, Streptococcus bovis, Adenoma, Bacterial Load, CD11b Antigen, carcinogenesis, Case-Control Studies, Chemokine CCL2, Chemokine CXCL1, Colonic Neoplasms, Cyclooxygenase 2, Feces, Interleukin-6, tumor microenvironment
Background
Colorectal cancer (CRC) remains the second highest cause of cancer death, according to GLOBOCAN statistics in 2018 [1]. An estimated 376 000 new cases of CRC are reported annually in China, with over 190 000 deaths [2]. While recent treatments restrict the growth of tumors, they also cause postoperative complications and excruciating side effects from radiotherapy and chemotherapy. Therefore, the exploration of a novel therapy for CRC based on a thorough understanding of its pathogenesis has received extensive attention.
Recently, the intestinal microflora has been found to be involved in multi-systemic diseases, including obesity, diabetes, and cancer [3]. Numerous studies suggest that bacterial over-colonization may be involved in the development of CRC [4]. Anaerobic bacteria are widely known to constitute the healthy colorectal microbiota, despite sizeable interpersonal variability [5]. Lumen microbiota usually over-colonize in the intestine, resulting in an increased risk of developing CRC [6]. Moreover,
Material and Methods
PATIENTS, FECES, AND TISSUES:
From December 2017 to March 2018, a total of 32 patients with cancer and 30 patients with adenoma underwent radical surgery in the Department of Surgery and Oncology of the Second Affiliated Hospital of Zhejiang University School of Medicine. We obtained the remaining feces from the Department of Clinical Laboratory. All patients had not received adjuvant chemotherapy, immunotherapy, or radiation therapy before surgery. At the same time, another 20 healthy volunteers were enrolled in this study and their corresponding feces were collected. The Ethics Committee of the Second Affiliated Hospital of Zhejiang University Medical College ethically approved the study. The written informed consent obtained from all participants. Feces were then stored in liquid nitrogen at −80°C for RNA examination.
BACTERIA AND CULTURE:
PREPARATION OF RNA AND QUANTITATIVE PCR:
According to the manufacturer’s instructions, Fecal Genomic DNA Extraction Kit (TianGen, Beijing, China) was used to isolate the DNA in the stool. Total RNA was extracted after elution with Tris-EDTA buffer (pH 8). Total RNA of tissues and cell lines was extracted using RNAiso Plus (TAKARA, Beijing, China) according to the instruction. The extracted RNA was synthesized to cDNA by the PrimeScript ™ RT reagent Kit (TAKARA, Beijing, China). Quantitative polymerase chain reaction (PCR) was done using SYBR® Green Realtime PCR Master Mix (TOYOBO, Shanghai, China) on the Applied Biosystems Veriti Thermal Cycler (Thermo Fisher Scientific, Waltham, MA, USA) following the manufacturer’s protocol. The quantitation of the target RNA expression was assessed using the endogenous control by the 2−ΔΔCt method (glyceraldehyde-phosphate dehydrogenase, GAPDH). NanoDrop 2000 spectrophotometer (Thermo Fisher Scientific, Waltham, MA, USA) was used to evaluate the quality of the prepared RNA, and cDNA was measured. All primers used are shown in Supplementary Table 1.
PROCEDURE FOR MICE EXPERIMENTS:
Thirty-two C57BL/6 mice (weighted 20 g) were purchased from the Experimental Animal Research Center of Zhejiang University (Hangzhou, Zhejiang, China). All mice were housed at 22°C, 55% humidity, 12 hours circadian rhythm, and in pathogen-free conditions. CRC animal models were performed as described previously [10,11]: one injection of ethoxymethane at day 0 (AOM, 12.5 mg/kg; Sigma-Aldrich, St. Louis, MO, USA); 10 days after AOM injection, 2.5% dextran sulfate sodium sulfate (DSS, MW=36 000–50 000 LLC, MP Biomedicals, USA) drinking for 5 days; 7 days after the previous cycle DSS drinking, next cycle began. The mice were sacrificed on the day the third DSS drinking finished (day 53). For the S. bovis group (n=12), each day (10: 00 am), the mice were gavage with S. bovis (1.2×107 CFU/day per mouse in physiological saline) for 2 weeks. The model group (n=12) was only subjected to AOM/DSS processing. The control group was treated only with drinking water (n=8). At several time points in these 3 groups on 0.5, 1.5, 2.5, 3.5, 4.5, 5.5, 6.5, and 7.5 weeks, the abundance of S. bovis was tested to evaluate the efficacy of gavage pretreatment. The weight of the mice was evaluated every 2 weeks and expressed as a relative weight (body weight change per mouse: test weight/day 0 weight×100%). After the mice were killed on day 53, the intestines were removed for follow-up experiments. The tumor burden of each mouse is shown in Supplementary Table 2. The Animal Ethics Committee has approved all animal experiments following relevant ethical principles and guidelines set out in the Animal Welfare Act and the NIH Guidelines for the Care and Use of Laboratory Animals.
ISOLATION OF IMMUNE CELLS IN TUMOR NODULES AND FLOW CYTOMETRY ANALYSIS:
Intertumoral immune cells were isolated as described [12,13] with mild modification. Briefly, the tumor nodes were washed with Dulbecco’s phosphate-buffered saline (PBS; Thermo Fisher Scientific, Waltham, MA, USA). Then they were incubated (37°C) in Hank’s balanced salt solution containing mixed enzymes (0.1 mg/mL collagenase D, 50 U/mL DNase I, 50 μg/mg dispase, Thermo Fisher Scientific, Waltham, MA, USA) for 30 minutes. The cells mixture was lightly covered with a 70% Percoll solution (Sigma-Aldrich, St. Louis, MO, USA), and then the mixture was lightly covered with 40% Percoll. The solution was centrifuged at 1500 g for 30 minutes at room temperature. The cells between the interfaces were gently aspirated and washed three times with PBS. The single-cell suspension was resuspended, and flow cytometry was performed. After incubation with Fc receptors (BD Biosciences, USA) for a blockade of non-specific signals, single-cell suspensions were incubated with primary antibodies (FITC-anti-CD11b, catalog number 557396, clone M1/70, BD Bioscience, USA) (PE-anti-TLR-4, catalog number 558294, clone MTS510, BD Bioscience, USA) in darkness at 4°C. Samples were measured by Invitrogen Attune NxT Flow Cytometer (Thermo Fisher Scientific, Waltham, MA, USA), and Flowjio was used to Analyze (BD Biosciences, USA) management data.
STATISTICAL ANALYSIS:
Data are shown as mean±standard error of the mean (SEM) or median of interquartile range. One-way analysis of variance (ANOVA, for example, Figure 1) or Kruskal-Wallis test was used to analyze the comparison of multiple groups. A Mann-Whitney U test (for example, Figures 2A, 2C, 3A, 3C) with a Bonferroni-corrected post hoc test and a Student’s t-test were used for comparison between the 2 groups. The relationship between the level of S. bovis and the clinicopathological characteristics in Table 1 was evaluated by chi-square analysis. P<0.05 was considered statistically significant. All statistical analyses were performed using GraphPad 6.0.
Results
:
The results of this study found that S. bovis was more likely to be enriched in cancer patients than in adenoma patients or healthy volunteers at P<0.01 (Figure 1). Analyzing the relationship between S. bovis abundance and clinicopathological characteristics (Table 1), we found that S. bovis was more likely present at patients with larger tumor size (P=0.028) and advanced TNM (P=0.033). These data indicate that S. bovis is related to advance CRC, which indicates that it may promote the development of colorectal cancer (Table 1).
:
The evaluation of the carcinogenic effect of S. bovis on colonic mucosa administered S. bovis gavage to mice with AOM/DSS treatment. Nucleic acid assays were used to evaluate the efficiency of gavage pretreatment (Supplementary Figure 1). After the second time of DSS drinking, a more severe weight loss was observed in the S. bovis group than in the model group (Figure 2A). As shown in Figure 2B, S. bovis pretreatment exacerbated the number of tumor nodules grown in the colorectum at P<0.01. Moreover, S. bovis pretreatment significantly increased the size of colorectal tumors (the average diameter of the S. bovis group was 2.26 mm, the model group was 1.83 mm, P<0.05, Figure 2C). In the control group, the colorectal mucosal glands were intact with occasional mild lymphocytic infiltration. In the model group, adenomas and highly differentiated adenocarcinomas could be seen everywhere. In the S. bovis group, poorly differentiated adenocarcinoma with disordered arrangement was the most significant change. Compared to the model group, the incidence of atypical hyperplasia, crypt abnormalities, and adenocarcinoma was highest in AOM/DSS mice fed with S. bovis (Figure 2D). Altogether, these data suggested that S. bovis can aggravate tumorigenesis in the colonic mucosa of AOM/DSS mice.
:
The evaluation expression pattern of cytokines associated with myeloid cells in the colon microenvironment (Figure 3A). Therefore, the results of this study showed interleukin (IL)-6 at P<0.01, Scyb1 (equivalent to human IL-8, P<0.05), Ptgs2 (equivalent to human COX-2, P<0.01), IL-1β (P<0.01), tumor necrosis factor (TNF) (P<0.05), and Ccl2 (P<0.01) gene expression increased significantly (Figure 3A). To investigate whether tumorigenesis of CRC can be attributed to myeloid cell recruitment caused by S. bovis. The measurement of the colon-infiltrated immune cells (Figure 3B) was done. Analysis shows that S. bovis-fed mice, tumor nodules TLR-4+ CD11b+ cells are more abundant than cells in model and control mice (Figure 3C). Supplementary Figure 2 shows a gated control of TLR-4+ cells. These results indicate that S. bovis may recruit TLR-4+ CD11b+ cells to promote the development of CRC.
Discussion
Distinct from the other types of cancer, CRC is thought to be caused mainly by colonization and inflammation in the colon [14]. The role of pathogenic bacteria in the development of CRC has gradually attracted scientific attention [15]. Based on the epidemiological investigation of
CD11b+ myeloid cells are heterogeneous in the tumor microenvironment, mainly consisting of macrophages and myeloid-derived suppressor cells (MDSCs). Intratumoral expression of IL-1β and IL-8, which contribute to tumorigenesis, has been shown to be derived from MDSCs [22]. A previous study reported that long noncoding RNAs could control the immune-suppressive function and differentiation of MDSCs via regulating the expression of Arg-1 and COX2 in MDSCs [23]. Increased TNF recruits MDSCs to the stroma, assisting tumor growth [24]. In addition, CCL2 has been deemed a strong chemokine to macrophages in the development of CRC [25]. Macrophages in the tumor microenvironment cannot only promote the epithelial-mesenchymal transition and stemness of cancer via TNF-α [26] but can also enhance invasion by secreting IL-6 and IL-8 [27]. Although the cytokine expression profile mentioned above was shown to be associated with
The concentration of intratumoral
There are some obvious limitations to our study. First, we did not obtain additional kinds of clinical samples (including serum and tumor tissues) apart from feces to investigate changes in
Conclusions
In conclusion, the results of this study show that
Figures
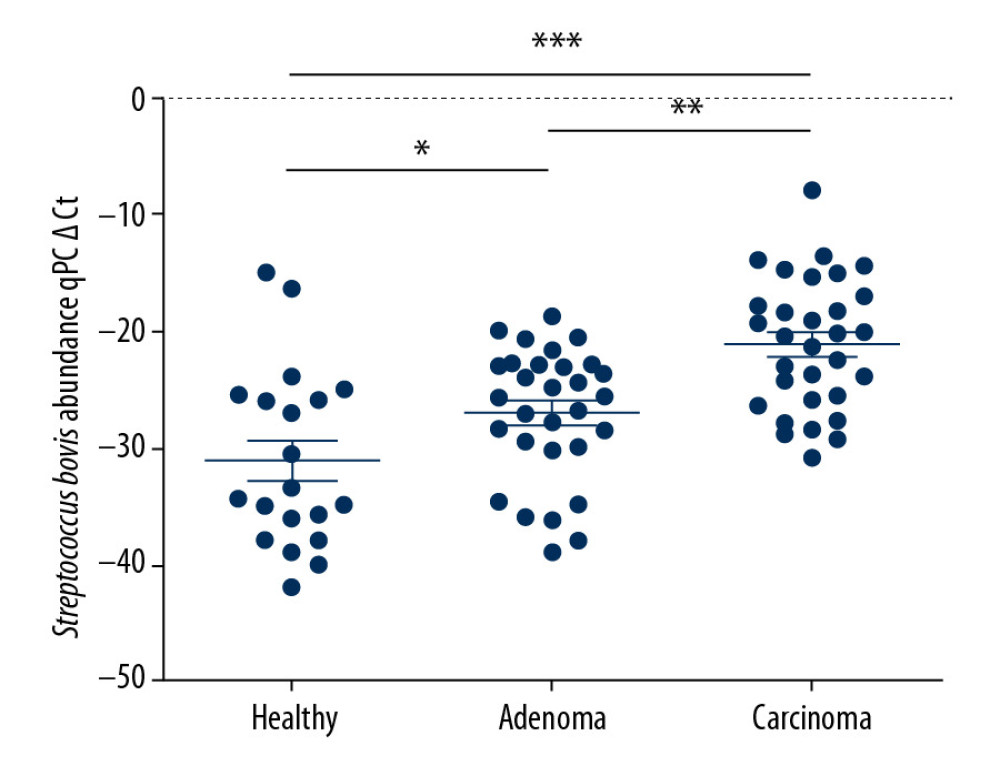 Figure 1. Streptococcus bovis are enriched in colorectal carcinoma patients. Abundance of S. bovis in feces from healthy subjects (n=20), patients with colorectal adenomas (n=30), and CRC (n=32). *** P<0.001.
Figure 1. Streptococcus bovis are enriched in colorectal carcinoma patients. Abundance of S. bovis in feces from healthy subjects (n=20), patients with colorectal adenomas (n=30), and CRC (n=32). *** P<0.001. 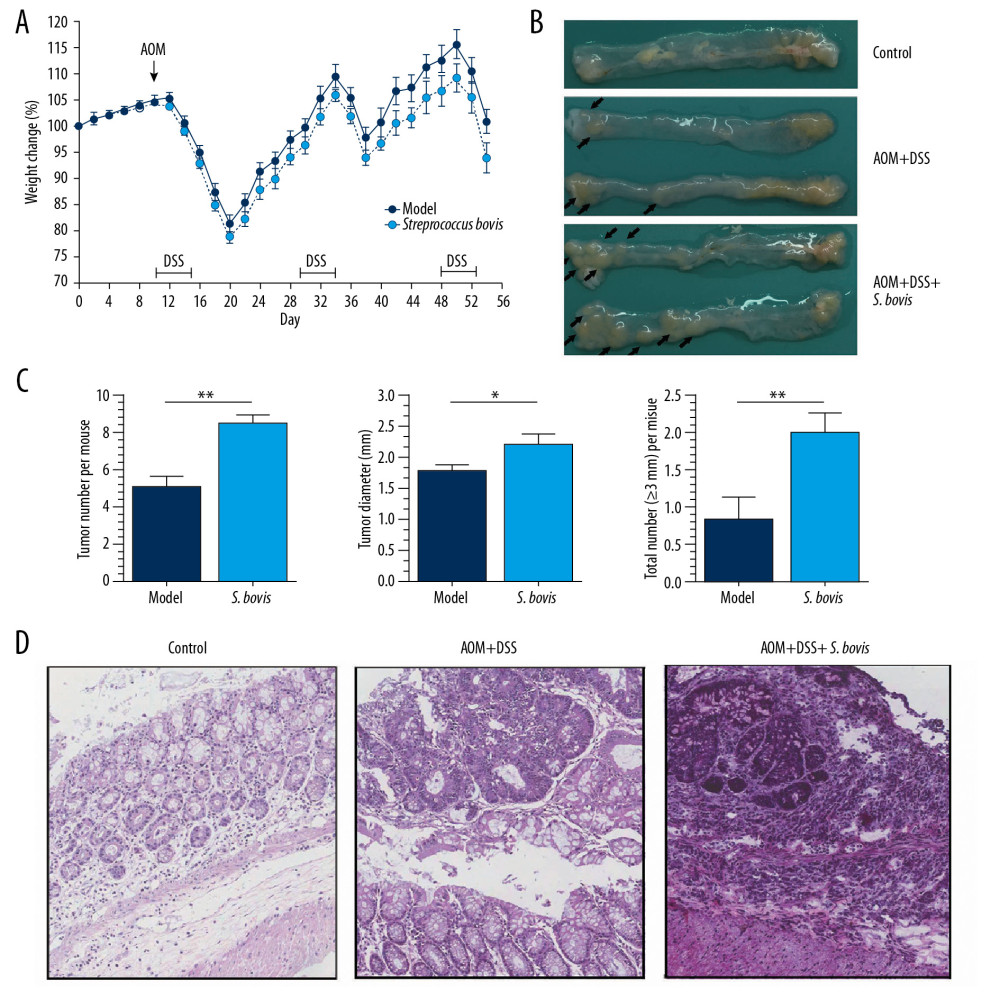 Figure 2. Streptococcus bovis accelerates tumor development of CRC in vivo. (A) Weight changes during the strains pretreatment. (B) Colorectal morphology for tumor nodes (black arrows indicate the diameter is larger than 1 mm). (C) Comparison between the Model group (n=12) and the S. bovis group (n=12). (D) Hematoxylin and eosin stained sections of colons in these 3 groups at the 53rd day. * P<0.05, ** P<0.01.
Figure 2. Streptococcus bovis accelerates tumor development of CRC in vivo. (A) Weight changes during the strains pretreatment. (B) Colorectal morphology for tumor nodes (black arrows indicate the diameter is larger than 1 mm). (C) Comparison between the Model group (n=12) and the S. bovis group (n=12). (D) Hematoxylin and eosin stained sections of colons in these 3 groups at the 53rd day. * P<0.05, ** P<0.01. 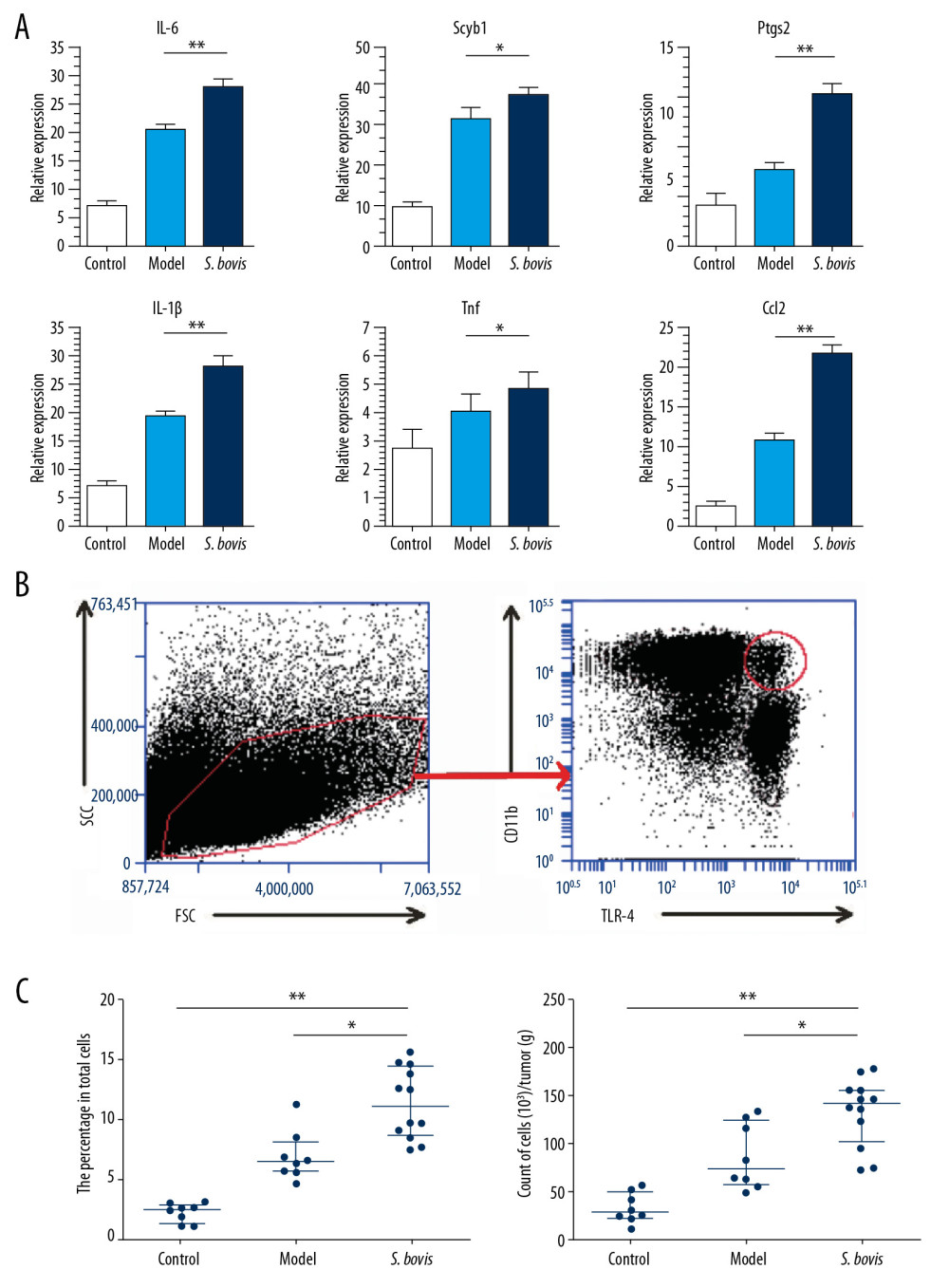 Figure 3. Streptococcus bovis contribute to the development of CRC by recruiting TLR-4+CD11b+ cells. (A) Relative mRNA expression of IL-6, Scyb, Ptgs2, IL-1β, TNF, and Ccl2 (Model group n=12; S. bovis group, n=12). (B) The gating methods with density plots for intratumoral myeloid cells. (C) Percentage and count of colonic TLR-4+CD11b+ cells (Control group, n=8; Model group, n=12; S. bovis group, n=12). * P<0.05, ** P<0.01.
Figure 3. Streptococcus bovis contribute to the development of CRC by recruiting TLR-4+CD11b+ cells. (A) Relative mRNA expression of IL-6, Scyb, Ptgs2, IL-1β, TNF, and Ccl2 (Model group n=12; S. bovis group, n=12). (B) The gating methods with density plots for intratumoral myeloid cells. (C) Percentage and count of colonic TLR-4+CD11b+ cells (Control group, n=8; Model group, n=12; S. bovis group, n=12). * P<0.05, ** P<0.01. References
1. Bray F, Ferlay J, Soerjomataram I, Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries: Cancer J Clin, 2018; 68; 394-424
2. Chen W, Cancer statistics: Updated cancer burden in China: Chin J Cancer Res, 2015; 27; 1
3. Gholizadeh P, Mahallei M, Pormohammad A, Microbial balance in the intestinal microbiota and its association with diabetes, obesity and allergic disease: Microb Pathog, 2018; 127; 48-55
4. Kelly D, Yang L, Pei Z, Gut microbiota, fusobacteria, and colorectal cancer: Diseases, 2018; 6; 109
5. Zhu Q, Gao R, Wu W, Qin H, The role of gut microbiota in the pathogenesis of colorectal cancer: Tumour Biol, 2013; 34; 1285-300
6. Chen W, Liu F, Ling Z, Human intestinal lumen and mucosa-associated microbiota in patients with colorectal cancer: PLoS One, 20112; 7(6); e39743
7. Kostic AD, Chun E, Robertson L, Fusobacterium nucleatum potentiates intestinal tumorigenesis and modulates the tumor-immune microenvironment: Cell Host Microbe, 2013; 14; 207-15
8. Richard ML, Liguori G, Lamas B, Mucosa-associated microbiota dysbiosis in colitis associated cancer: Gut Microbes, 2018; 9; 131-42
9. Rezasoltani S, Asadzadeh Aghdaei H, Dabiri H, The association between fecal microbiota and different types of colorectal polyp as precursors of colorectal cancer: Microb Pathog, 2018; 124; 244-49
10. Okayasu I, Ohkusa T, Kajiura K, Promotion of colorectal neoplasia in experimental murine ulcerative colitis: Gut, 1996; 39; 87-92
11. Greten FR, Eckmann L, Greten TF, IKKbeta links inflammation and tumorigenesis in a mouse model of colitis-associated cancer: Cell, 2004; 118; 285-96
12. Davies MD, Parrott DM, Preparation and purification of lymphocytes from the epithelium and lamina propria of murine small intestine: Gut, 1981; 22; 481-88
13. Xu L, Yi HG, Wu Z, Activation of mucosal mast cells promotes inflammation-related colon cancer development through recruiting and modulating inflammatory CD11b(+)Gr1(+) cells: Cancer Lett, 2015; 364; 173-80
14. Park CH, Eun CS, Han DS, Intestinal microbiota, chronic inflammation, and colorectal cancer: Intest Res, 2018; 16; 338-45
15. Al-Jashamy K, Murad A, Zeehaida M: Asian Pac J Cancer Prev, 2010; 11; 1765-68
16. Corredoira J, Garcia-Pais MJ, Coira A: Eur J Clin Microbiol Infect Dis, 2015; 34; 1657-65
17. Chand G, Shamban L, Forman A, Sinha P: Case Rep Gastrointest Med, 2016; 2016 7815843
18. Ellmerich S, Djouder N, Scholler M, Klein JP: Cytokine, 2000; 12; 26-31
19. Abdulamir AS, Hafidh RR, Mahdi LK: BMC Cancer, 2009; 9; 403
20. Abdulamir AS, Hafidh RR, Bakar FA: Mol Cancer, 2010; 9; 249
21. Biarc J, Nguyen IS, Pini A: Carcinogenesis, 2004; 25; 1477-84
22. Najjar YG, Rayman P, Jia X, Myeloid-derived suppressor cell subset accumulation in renal cell carcinoma parenchyma is associated with intratumoral expression of IL1beta, IL8, CXCL5, and Mip-1alpha: Clin Cancer Res, 2017; 23; 2346-55
23. Gao Y, Sun W, Shang W, Lnc-C/EBPbeta negatively regulates the suppressive function of myeloid-derived suppressor cells: Cancer Immunol Res, 2018; 6; 1352-63
24. Atretkhany KS, Nosenko MA, Gogoleva VS, TNF neutralization results in the delay of transplantable tumor growth and reduced MDSC accumulation: Front Immunol, 2016; 7; 147
25. Nakatsumi H, Matsumoto M, Nakayama KI, Noncanonical pathway for regulation of CCL2 expression by an mTORC1-FOXK1 axis promotes recruitment of tumor-associated macrophages: Cell Rep, 2017; 21; 2471-86
26. Chen Y, Wen H, Zhou C, TNF-alpha derived from M2 tumor-associated macrophages promotes epithelial-mesenchymal transition and cancer stemness through the Wnt/beta-catenin pathway in SMMC-7721 hepatocellular carcinoma cells: Exp Cell Res, 2019; 378; 41-50
27. Xu H, Lai W, Zhang Y, Tumor-associated macrophage-derived IL-6 and IL-8 enhance invasive activity of LoVo cells induced by PRL-3 in a KCNN4 channel-dependent manner: BMC Cancer, 2014; 14; 330
28. Tu X, Hong D, Jiang Y, FH535 inhibits proliferation and migration of colorectal cancer cells by regulating CyclinA2 and Claudin1 gene expression: Gene, 2018; 690; 48-56
29. Wu M, Wu Y, Li J: Int J Mol Sci, 2018; 19(9); 2809
Figures
 Figure 1. Streptococcus bovis are enriched in colorectal carcinoma patients. Abundance of S. bovis in feces from healthy subjects (n=20), patients with colorectal adenomas (n=30), and CRC (n=32). *** P<0.001.
Figure 1. Streptococcus bovis are enriched in colorectal carcinoma patients. Abundance of S. bovis in feces from healthy subjects (n=20), patients with colorectal adenomas (n=30), and CRC (n=32). *** P<0.001. Figure 2. Streptococcus bovis accelerates tumor development of CRC in vivo. (A) Weight changes during the strains pretreatment. (B) Colorectal morphology for tumor nodes (black arrows indicate the diameter is larger than 1 mm). (C) Comparison between the Model group (n=12) and the S. bovis group (n=12). (D) Hematoxylin and eosin stained sections of colons in these 3 groups at the 53rd day. * P<0.05, ** P<0.01.
Figure 2. Streptococcus bovis accelerates tumor development of CRC in vivo. (A) Weight changes during the strains pretreatment. (B) Colorectal morphology for tumor nodes (black arrows indicate the diameter is larger than 1 mm). (C) Comparison between the Model group (n=12) and the S. bovis group (n=12). (D) Hematoxylin and eosin stained sections of colons in these 3 groups at the 53rd day. * P<0.05, ** P<0.01. Figure 3. Streptococcus bovis contribute to the development of CRC by recruiting TLR-4+CD11b+ cells. (A) Relative mRNA expression of IL-6, Scyb, Ptgs2, IL-1β, TNF, and Ccl2 (Model group n=12; S. bovis group, n=12). (B) The gating methods with density plots for intratumoral myeloid cells. (C) Percentage and count of colonic TLR-4+CD11b+ cells (Control group, n=8; Model group, n=12; S. bovis group, n=12). * P<0.05, ** P<0.01.
Figure 3. Streptococcus bovis contribute to the development of CRC by recruiting TLR-4+CD11b+ cells. (A) Relative mRNA expression of IL-6, Scyb, Ptgs2, IL-1β, TNF, and Ccl2 (Model group n=12; S. bovis group, n=12). (B) The gating methods with density plots for intratumoral myeloid cells. (C) Percentage and count of colonic TLR-4+CD11b+ cells (Control group, n=8; Model group, n=12; S. bovis group, n=12). * P<0.05, ** P<0.01. Tables
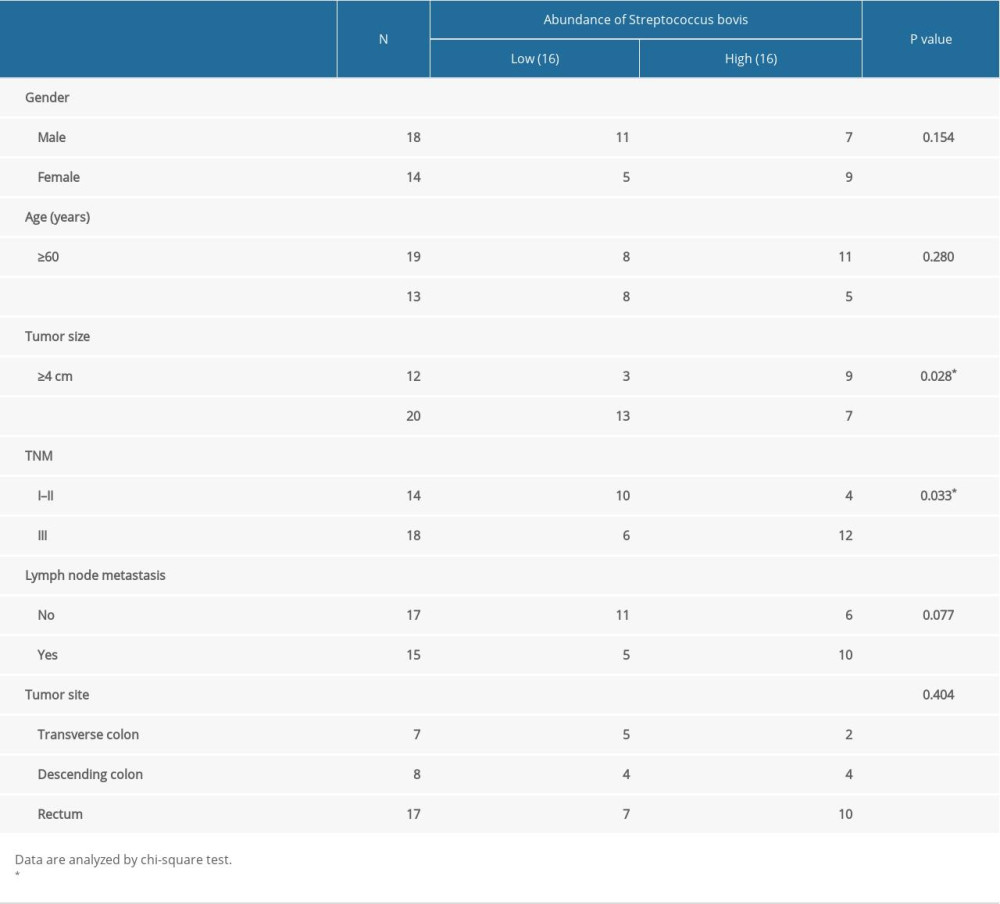 Table 1. Relationship between abundance of Streptococcus bovis and clinicopathological characteristics of CRC patients.
Table 1. Relationship between abundance of Streptococcus bovis and clinicopathological characteristics of CRC patients. Table 1. Relationship between abundance of Streptococcus bovis and clinicopathological characteristics of CRC patients.
Table 1. Relationship between abundance of Streptococcus bovis and clinicopathological characteristics of CRC patients.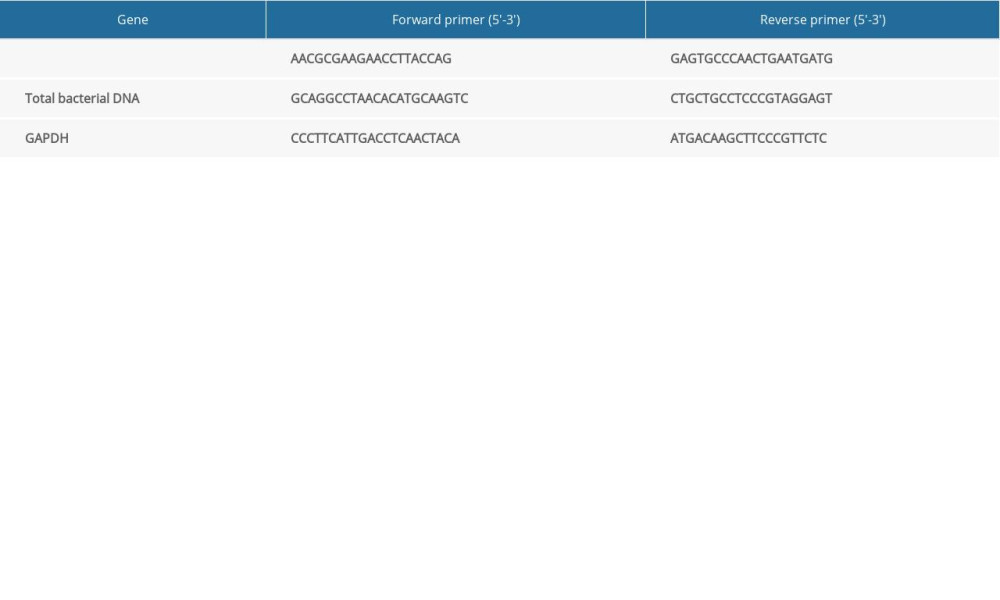 Supplementary Table 1. The primers for genes detected by real-time polymerase chain reaction.
Supplementary Table 1. The primers for genes detected by real-time polymerase chain reaction.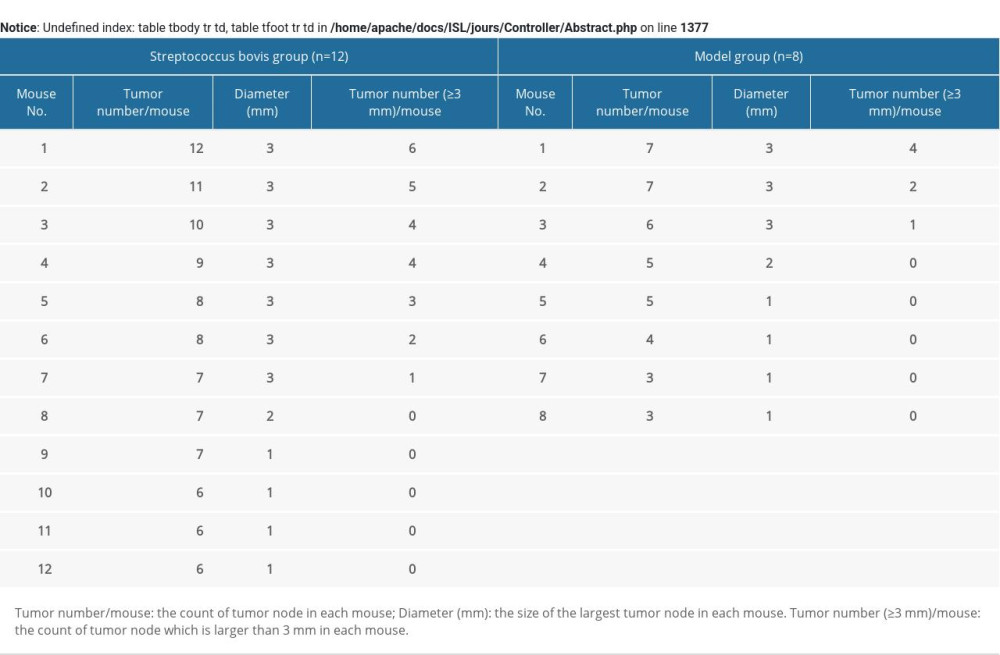 Supplementary Table 2. The tumor burden of each animal in Figure 2.
Supplementary Table 2. The tumor burden of each animal in Figure 2. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








