17 June 2020: Clinical Research
Comprehensive Decongestive Therapy as a Treatment for Secondary Lymphedema of the Lower Extremity and Quality of Life of Women After Gynecological Cancer Surgery
Lucia Kendrová1ABCDEFG*, Wioletta Mikuľáková1ABEG, Katarína Urbanová1BEF, Štefánia Andraščíková2CDG, Silvia Žultáková2EFG, Peter Takáč3CDE, Yuriy Peresta1DEFDOI: 10.12659/MSM.924071
Med Sci Monit 2020; 26:e924071
Abstract
BACKGROUND: Lymphedema is a clinical manifestation of lymphatic system failure, caused by an imbalance between the transport capacity of the lymphatic system and the volume of the produced lymph. Lymphedema is complication and significantly worsens quality of life (QoL).
MATERIAL AND METHODS: There were 50 patients diagnosed with secondary lymphedema of the lower extremities after gynecological cancer followed by radiotherapy included in this study. The average age was 57.76 years (standard deviation±10.08). Patients were treated at the Department of Physiotherapy, Balneology and Medical Rehabilitation, in hospital NsP in Bardejov. During therapy, we applied manual lymphatic drainage, instrumental lymphatic drainage, multilayer bandage, vascular gymnastics (with loaded external compression), hydrotherapy, and patient education on the adjustment necessary for a life-long regimen. The circumference of the limb was measured using the Kuhnkes disk method, QoL was assessed using the LYMQOL LEG questionnaire, and for assessment of pain the Visual Analogue Scale (VAS) was used.
RESULTS: After treatment, we found a reduction in lymphedema (P<0.001), an increase in QoL (P<0.001), and a reduction in pain (P<0.001). We found a significant relationship between QoL change and pain in the domains of symptoms, function, and overall QoL (P<0.005). The results showed that reduction of lymphedema was not a significant predictor of QoL (P>0.001).
CONCLUSIONS: We found a positive effect in the treatment of secondary lymphedema of the lower extremity on the QoL of women after uterine cancer surgery, and also found that reduction of lymphedema and age were not predictors of improvement in QoL.
Keywords: Gynecologic Surgical Procedures, lymphedema, Quality of Life, Compression Bandages, Genital Neoplasms, Female, Hydrotherapy, Manual Lymphatic Drainage, Pain Measurement, Patient Education as Topic, Physical Therapy Modalities, Postoperative Complications
Background
Oncological diseases are one of the main causes of mortality worldwide and are on an upward trend [1]. An increase in oncological diseases leads to an increase in possible complications after oncological treatment [1,2]. The fourth most common female cancer is endometrial cancer [3,4] and it is the most prevalent pelvic malignancy among gynecological cancers [3,5]. Lower limb lymphedema (LLL) is a chronic, progressive, and frequently incurable condition leading to disability that can affect women cured of gynecological cancer [3,6–9]. A study by Mirandola et al. [3] indicated that secondary LLL is the result of lymphatic system insufficiency and impaired lymph transport with increased limb volume related to cancer treatments, such as the surgical removal of pelvic and para-aortic lymph nodes and radiation therapy [3,10,11]. A late-diagnosed and inadequately treated lymphedema can have a significant negative impact on a patient’s somatic, social, and psychological state, which can lead to permanent disability [1,12]. For secondary lymphedema of the lower extremities in patients who have undergone lymph node surgery, radiation, or chemotherapy in the treatment of cancer, the prevalence is 20–27.2% of patients [3,7,13–16].
The high increase in oncological diseases leads to increased complications following oncological treatment [1,2]. The fourth most common type of cancer in women is endometrial cancer [3,4] and it is the most common pelvic malignancy among gynecological cancers [3,5]. Lower limb lymphedema (LLL) is a chronic, progressive, and incurable disability, particularly affecting women treated for gynecological cancer [3,6–9]. Mirandola et al. [3] report that secondary LLL is the result of lymphatic system deficiency resulting in disruption of lymph transport, resulting in increased limb volume associated with cancer treatment, such as surgical removal of pelvic and periaortic lymph nodes as well as radiation therapy [3,10,11]. A late-diagnosed and inadequately treated lymphedema has a negative impact on a patient’s somatic, social, and psychological state, which may lead to permanent disability [1,12]. In the case of secondary lower limb lymphedema, the incidence of lymphedema in patients who have undergone lymph node surgery followed by radiation or chemotherapy is 20–27.2% [3,7,13–16].
LLL has a negative impact on domestic work, physical activity, mobility, social activities, and psychological well-being [7,17,18]. If patients with LLL are not treated properly, LLL will gradually worsen, causing disability in daily life activities (ADL) and reducing QoL [7,17–21]. The key to LLL treatment is prevention, early diagnosis, and early treatment [7,14].
There is no standard approach that is applicable to all patients, despite the numerous therapies available in the treatment of LLL [22,23]. Initially conservative measures were recommended for the treatment of lymphedema, while surgical management was generally considered palliative [25].
The International Society of Lympho Logs (ILS) Consensus Document suggests both operative treatment and non-operative treatment for lymphedema management. Various non-operative treatments such as complete decongestive therapy (CDT), compression garment, massage, intermittent pneumatic compression, aquatic therapy, drug therapy, and psychosocial rehabilitation are suggested [1,25]. However, there is still a lack of research on which of these methods are most effective and appropriate, and more research and a systematic review of studies is needed [25,26]. Today, the basis of successful treatment of lymphedema is CDT, which has 2 phases. Phase 1 is swelling reduction to maximize lymphedema volume loss and phase 2 is stabilization to sustain lymphedema loss in the long term [1]. The aim of our study was to determine whether reduction of lymphedema is a predictor of improvement in QoL and what effect CDT as treatment of secondary lymphedema of the lower extremity has on the QoL of women after gynecological cancer surgery.
Material and Methods
MEASUREMENTS:
The effect of physiotherapy on the reduction of lymphedema was monitored by measuring the circumference of both lower extremities using a centimeter marked type. We used the Kuhnkes disk method, such that the circumference of the extremity was measured from distal to proximal in 4-cm intervals [6]. A measuring board was used. We measured 16 identical sites on the lower extremities at the beginning and at the end of treatment [6,13,27].
The QoL of patients with LLL we evaluated using the standardized questionnaire LYMQOL LEG. The LYMQOL was developed by experienced healthcare professionals who worked with patients suffering from lymphedema. It was developed separately for lymphedema of the upper limb and lower limb [6,14,28]. The questions concern 4 domains (regions) [28]: symptoms (questions 1a–1f, 2, and 3), body image appearance (questions 4–10), function (questions 11–15), and mood (questions 16–21). Keeley et al. [28] and Wedin et al. [29] reported that each item in each domain is scored by a probability scale from 1 to 4: where 1=not at all, 2=a little; 3=quite a bit; and 4=a lot. Domain totals are calculated by adding individual scores and dividing the total by the number of questions answered for symptoms, body image/appearance, function, and mood. Overall QoL (Q22) is scored by a patient-scored score of 0 to 10, where 0 is the worst-rated QoL and 10 is the best-rated QoL [28,29].
Pain was scored according to the Visual Analogue Scale (VAS) ranging from 0 to 10, with 0 meaning no pain and 10 meaning maximum, unbearable pain. Woodforde and Merskey [30] first reported the use of VAS pain scale with descriptor extremes “no pain at all” and “my pain is bad as it could possibly be” in patients with a variety of pains [30].
STATISTICAL ANALYSIS:
The evaluation was performed by comparing before and after treatment (treatment lasted±16 days). Descriptive statistics, lymphedema reduction, pain, and QoL were evaluated using a paired
Results
The average age of patients was 57.76±10.08 years. After gynecological cancer patients received CDT. After treatment, we noted a reduction in LLL of 30.32 cm (4.19%) and PLL of 30.24 cm (4.27%) (Table 1).
We evaluated the QoL using the LYMQOL questionnaire before and after treatment. Using a paired
Using a Pearson correlation analysis between improvement in QoL domains after CDT in patients with lymphedema and baseline values of LLL and PLL reduction, no significant relationship was found (
The predicted predictors of improvement in QoL were the reduction of LLL and age. The results show that predictors are not significant for improving QoL (
Discussion
Lymphedema is a progressive chronic disease due to insufficient lymphatic system activity and impaired lymph transport [1]. Despite the numerous therapies available for the treatment of LLL, there is no standard approach for all patients [22]. Bakar and Tuğral. [6] indicated in their study that after pelvic lymph node dissection the incidence of lower limb lymphedema in patients with gynecologic malignancies ranges between 2.4% to 41%. Thus, the management of lower extremity lymphedema in patients after gynecologic cancer surgery is an important issue [6,15,31,32]. CDT is still the gold standard of lymphedema management [6,15,31,32]. Baumann et al. [33] conducted a systematic review examining the effects of physical exercise as part of rehabilitation. In 13 studies, they found that the studies involved conclude the positive effect of physiotherapy. Only a few studies have been conducted to determine the effects of manual lymphatic drainage (MLD) and compression therapy in the treatment of lymphedema [6,15,34–36]. Two controlled studies have shown that compression therapy with or without manual lymphatic drainage was equally effective for patients with lymphedema after cancer [36–39]. Andersen et al. [40] conducted a randomized, controlled study comparing manual lymphatic drainage and compression (n=20) with a control group that was only treated with compression therapy (n=20). Indeed, after 2 weeks, the control group had a greater percentage reduction in absolute swelling (60%) compared to the MLD group (48%). In both groups the symptoms of difficulty and swelling were reduced equally, but the QoL was also improved in the control group [15]. Singh et al. [41] in his study describes that patients with secondary lymphedema can safely participate in progressive, regular exercise without experiencing a worsening of lymphedema or related symptoms. There is insufficient evidence to support or refute the current clinical recommendation to wear compression garments during regular exercise [6,15,19].
Lymphedema can have adverse effects on psychological and social wellbeing and limit mobility and ability to perform daily activities [6,15,19,43]. To change in the clinical measured of lymphedema more emphasis in evaluating the effect of treatment as the results themselves QoL [6,15,19,43]. Considering that treatment for lymphedema involves a significant and ongoing commitment from patients, it is essential to determine whether the benefits to patients outweigh the burden associated with treatment [6,19]. We evaluated the QoL of patients with lymphedema of the lower extremities using the standardized questionnaire LYMQOL LEG. LYMQOL has good reliability and validity and builds on international research [44,45].
New interdisciplinary guidelines from the Netherlands appear to help improve the quality of day-to-day care for patients with lymphedema, as reported by Van de Pas [45]. In particular, this refers to a new approach to chronic diseases in which patients play a central role by actively participating in their treatment. Van de Pas [45] also points to the important role of the ICF (International Classification of Functioning, Disability and Health), which aims to use proper assessment, therapy, education and assistance for patients with specific diseases with respect to specific aspects of human body function [45–47].
The pain VAS originated from continuous visual analog scales developed in the field of psychology to measure well-being [47,48]. Smoot et al. [49] included 3 studies reporting VAS scores in their meta-analysis. The reported results were statistically homogeneous, Q=1.4 (
Similarly, the study by Micha et al. [24] suggests that conservative CDT lymphedema is effective and has a beneficial effect on limb volume reduction, which is strongly related to improving the QoL (pain and depression relief) of lymphedema patients. Despite this, we also encountered patients whose lymphatic drainage disorder worsened despite well-managed conservative therapy. Surgical therapy for lymphedema may be considered in these patients.
The study by Klernäs et al. [42] reported that lymphedema had an effect on the QoL in 20% of patients. QoL assessment tended to decrease with age, with a significant correlation in the psychosocial area (rs=0.194,
Regular exercise reduces the symptoms of lymphedema by reducing local swelling [51,52]. Exercise and physical activity are essential for treatment as they increase physical and emotional fitness, vitality, and quality of life [52–56]. What is important is the level and intensity that must be considered [3,51]. Studies have shown that strength training must be adaptable to patients [52,54]. The recommended exercises for post mastectomy women are walking, dancing, gymnastics, cycling, swimming, and Nordic walking [51]. Puszczalowska-Lizis et al. [51] investigated the quality of life (WHOQOL-BREF) of active and inactive women after postmastectomy. Women who performed physical activity rated their quality of life better in each domain than women who did not exercise [51].
There is scant research that has determined the level of physical activity in LLL patients during the treatment of cancer [15,57].
The need to learn all the rules to facilitate disease management related to the lifestyle of patients with chronic lymphedema has been described by Biglia et al. [31] and Gloviczki et al. [36]. These authors reported that after treatment of gynecological carcinoma, this chronic condition responds poorly to treatment interventions and thus has a significant impact on the QoL. They further state that the difference in secondary lymphedema incidence values is due to inconsistencies in the diagnostic evaluation system. The correct management after diagnosis and the real state of occurrence is unknown. The primary cause of LLL is lymphadenectomy whose side effects may become worse due to other most common risk factors [31,36].
Lymphedema affects millions of people. Its prevalence and morbidity have been underestimated, often shifted from the disease category to the “cosmetic error” category. Lymphedema is a progressive disease and requires lifelong treatment. Lymphedema can be significantly improved using contemporary traditional (conservative) and surgical treatment methods [36]. Conservative treatment always involves CDT, compression pumps and clothing. Pharmacotherapy and surgical treatment are optional. Surgery may be reconstructive or excision. Although non-surgical intervention has been effective in reducing lower extremity edema, other effects, such as pain and heaviness. have been measured differently for each study, and with a limited expectation of a definite effect. However, non-invasive therapies have shown positive effects, including improving the QoL and lowering extremity edema in cancer patients.
Conclusions
We found a positive effect in the treatment of secondary lymphedema of the lower extremity on the QoL of women after uterine cancer surgery. We also found a reduction in the volume of the lower limbs in these patients. However, despite positive changes in these parameters, our research did not find that reduction of lymphedema and age were predictors of improvement in QoL.
Tables
Table 1. Reduction of lower limb volume.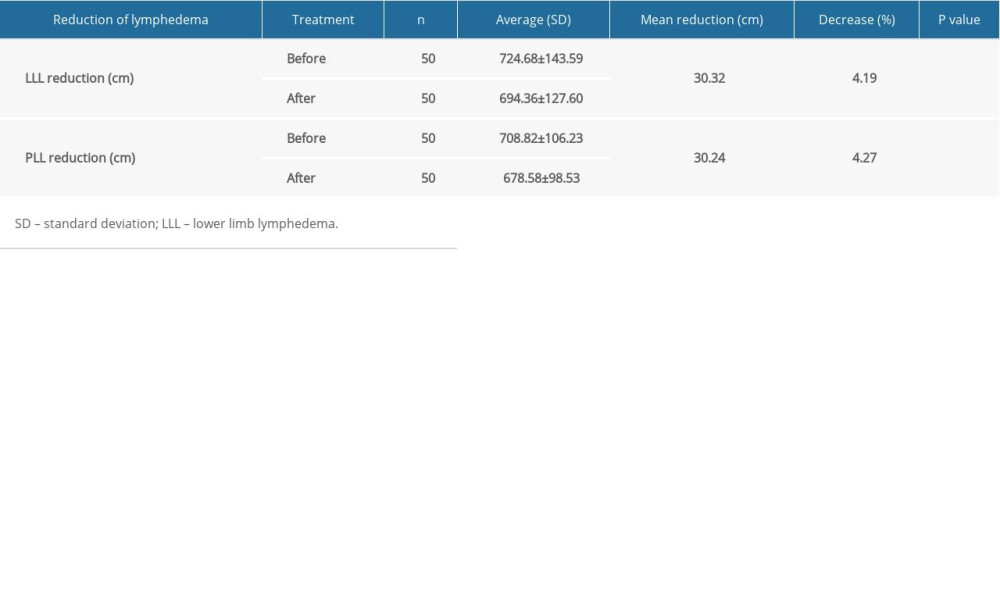 Table 2. Assessment of QoL with LYMQOL.
Table 2. Assessment of QoL with LYMQOL.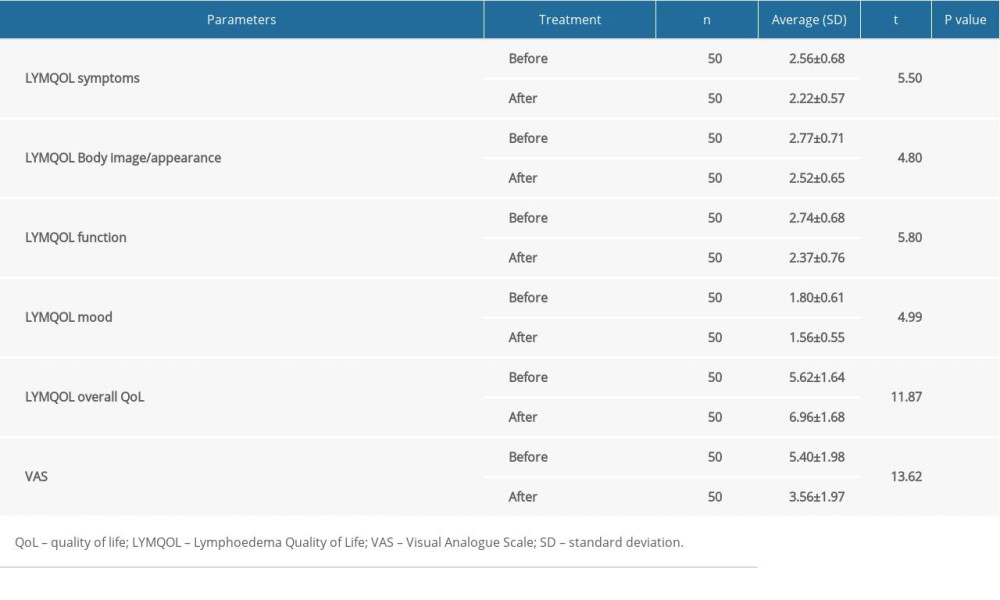 Table 3. Pearson correlation analysis between improvement in QoL domains after CDT and baseline values of reduction LLL, PLL, pain, and age.
Table 3. Pearson correlation analysis between improvement in QoL domains after CDT and baseline values of reduction LLL, PLL, pain, and age.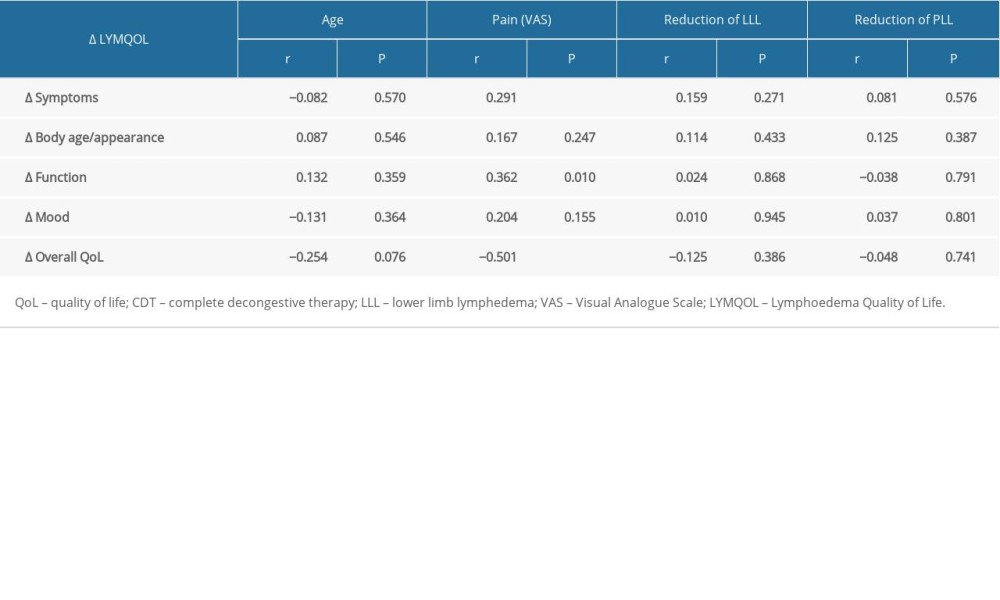 Table 4. Predictors of achieving a significant improvement in QoL (reduction of LLL).
Table 4. Predictors of achieving a significant improvement in QoL (reduction of LLL).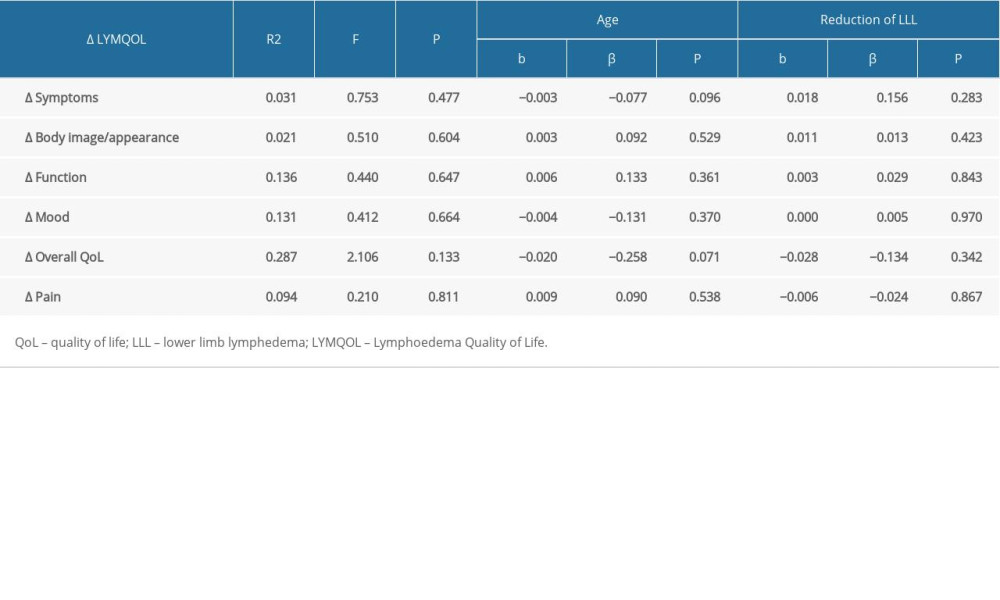 Table 5. Predictors of significant improvement in QoL (reduction of PLL).
Table 5. Predictors of significant improvement in QoL (reduction of PLL).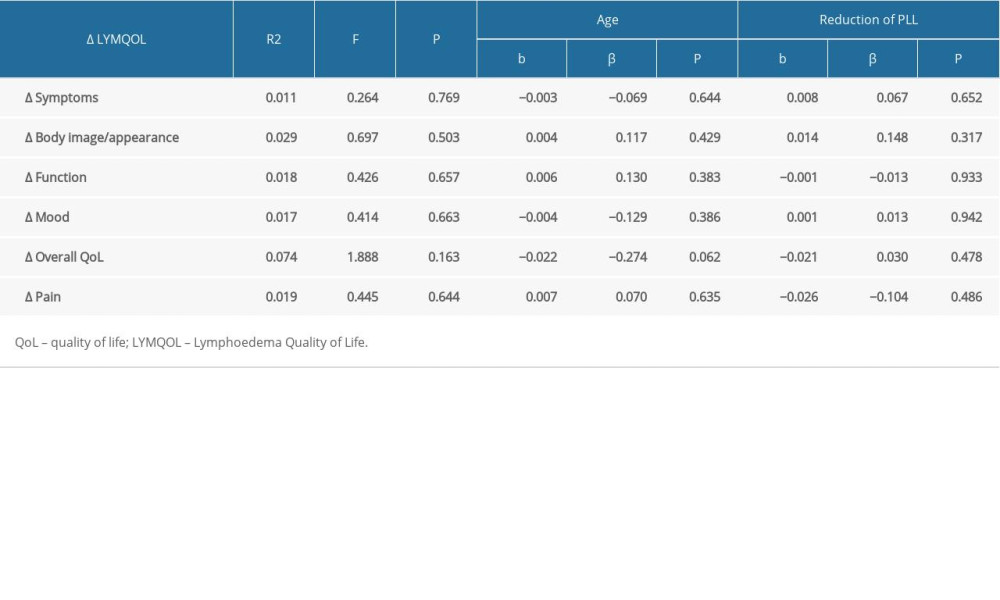
References
1. International Society of Lymphology, The diagnosis and treatment of peripheral lymphedema: 2013 Consensus document of the International Society of Lymphology: Lymhology, 2013; 46(1); 1-11
2. Hayes AC, Janda M, Ward LC, Lymphedema following gynecological cancer: Results from a prospective, longitudinal cohort study on prevalence, incidence and risk factors: Gynecol Oncol, 2017; 146(3); 623-29
3. Mirandola D, Muraca MG, Sgambati E, Role of a structured physical activity pathway in improving functional disability, pain and quality of life in a case of breast and gynecological cancer survivorship: In J Clin Med, 2019; 8(4); 53
4. Martin-Dunlap TM, Wachtel MS, Margenthaler JA, Outcomes for patients who are diagnosed with breast and endometrial cancer: Oncol Lett, 2013; 6(4); 1103-7
5. Gil KM, Von Gruenigen VE, Physical activity and gynecologic cancer survivorship: Recent Results Cancer Res, 2011; 186; 305-15
6. Bakar Y, Tuğral A, Lower extremity lymphedema management after gynecologic cancer surgery: A review of current management strategies: Ann Vasc Surg, 2017; 44; 442-50
7. Fukushima T, Tsuji T, Sano Y, Immediate effects of active exercise with compression therapy on lower-limb lymphedema: Support Care Cancer, 2017; 25(8); 2603-10
8. Cemal Y, Jewell S, Albornoz CR, Systematic review of quality of life and patient reported outcomes in patients with oncologic related lower extremity lymphedema: Lymphat Res Biol, 2013; 11(1); 14-19
9. Stolldorf DP, Dietrich MS, Ridner SH, Symptom frequency, intensity, and distress in patients with lower limb lymphedema: Lymphat Res Biol, 2016; 14(2); 78-87
10. Lindqvist E, Wedin M, Fredrikson M, Kjølhede P, Lymphedema after treatment for endometrial cancer – a review of prevalence and risk factors: Eur J Obstet Gynecol Reprod Biol, 2017; 211; 112-21
11. Greene A, Meskell P, The impact of lower limb chronic oedema on patients’ quality of life: Int Wound J, 2017; 14(3); 561-68
12. Tessa CG, Hoda ES, Cheryl LB, Breast cancer-related lymphedema: Risk factors, precautionary measures, and treatments: Gland Surg, 2018; 7(4); 379-403
13. Ezzo J, Manheimer E, McNeely ML, Manual lymphatic drainage for lymphedema following breast cancer treatment: Cochrane Database Syst Rev, 2015; 5; CD003475
14. Brorson H, Liposuction in lymphedema treatment: J Reconstr Microsurg, 2016; 32(1); 56-65
15. Katz E, Dugan NL, Cohn JC, Weight lifting in patients with lower-extremity lymphedema secondary to cancer: a pilot and feasibility study: Arch Phys Med Rehabil, 2010; 91(7); 1070-76
16. Ohba Y, Todo Y, Kobayashi N, Risk factors for lower-limb lymphedema after surgery for cervical cancer: Int J Clin Oncol, 2011; 16(3); 238-43
17. Dunberger G, Lindquist H, Waldenström AC, Lower limb lymphedema in gynecological cancer survivors – effect on daily life functioning: Support Care Cancer, 2013; 21(11); 3063-70
18. Brown JC, Lin LL, Segal S, Physical activity, daily walking, and lower limb lymphedema associate with physical function among uterine cancer survivors: Support Care Cancer, 2014; 22(11); 3017-25
19. Finnane A, Hayes SC, Obermair A, Janda M, Quality of life of women with lower-limb lymphedema following gynecological cancer: Expert Rev Pharmacoecon Outcomes Res, 2011; 11(3); 287-97
20. Farrell R, Gebski V, Hacker NF, Quality of life after complete lymphadenectomy for vulvar cancer: Do women prefer sentinel lymph node biopsy?: Int J Gynecol Cancer, 2014; 24(4); 813-19
21. Ferrandina G, Petrillo M, Mantegna G, Evaluation of quality of life and emotional distress in endometrial cancer patients: A 2-year prospective, longitudinal study: Gynecol Oncol, 2014; 133(3); 518-25
22. Boyages J, Kastanias K, Koelmeyer LA, Liposuction for advanced lymphedema: A multidisciplinary approach for complete reduction of arm and leg swelling: Ann Surg Oncol, 2015; 22; 1263-70
23. Narahari SR, Aggithaya MG, Moffatt C, Future research priorities for morbidity control of lymphedema: Indian J Dermatol, 2017; 62; 33-40
24. Micha JP, Nguyen H, Goldsteina HB, Successful management of persistent lower extremity lymphedema with suction-assisted lipectomy: Gynecol Oncol Rep, 2017; 23; 13-15
25. : Lymphoedema Framework: Best practice for the management of lymphoedema. international consensus, 2006, London, MEP Ltd https://www.lympho.org/wp-content/uploads/2016/03/Best_practice.pdf
26. Jung LY, Yeun JS, A systematic review of non-surgical interventions applied to cancer patients with low limb lymphedema in international information institute (Tokyo): Information; Koganei, 2019; 22(3); 263-72
27. Williams AF, Vadgama A, Franks PJ, Mortimer PS, A randomized controlled crossover study of manual lymphatic drainage therapy in women with breast cancer-related lymphoedema: Eur J Cancer Care (Engl), 2002; 11(4); 254-61
28. Hilliam R, Riches K, Veigas D, A quality of life measure for limb lymphoedema (LYMQOL): Journal of Lymphoedema, 2010; 5(1); 26-37
29. Wedin M, Fredrikson M, Ahlner E, Validation of the Lymphoedema Quality of Life Questionnaire (LYMQOL) in Swedish cancer patients: Acta Oncol, 2020; 59(3); 365-71
30. Woodforde JM, Merskey H, Some relationships between subjective measures of pain: J Psychosom Res, 1972; 16(3); 173-78
31. Biglia N, Zanfagnin V, Daniele A, Lower body lymphedema in patients with gynecologic cancer: Anticancer Res, 2017; 37(8); 4005-15
32. Kunitake T, Kakuma T, Ushijima K, Risk factors for lower limb lymphedema in gynecologic cancer patients after initial treatment: Int J Clin Oncol, 2020 [Epub ahead of print]
33. Baumann FT, Reike A, Reimer V, Effects of physiotherapy on breast cancer related secondary lymphedema: A systematic review: Breast Cancer Res Treat, 2018; 170(1); 1-13
34. Müller M, Klingberg K, Wertli MM, Carreira H, Manual lymphatic drainage and quality of life in patients with lymphoedema and mixed oedema: A systematic review of randomised controlled trials: Qual Life Res, 2018; 27(6); 1403-14
35. Zasadzka E, Trzmiel T, Kleczewska M, Pawlaczyk M, Comparison of the effectiveness of complex decongestive therapy and compression bandaging as a method of treatment of lymphedema in the elderly: Clin Interv Aging, 2018; 13; 929-34
36. Gloviczki ML, Gloviczki P, Advances and controversies in the contemporary management of chronic lymphedema: Indian J Vasc Endovasc Surg, 2018; 5(4); 219-26
37. Smile TD, Tendulkar R, Schwarz G, A review of treatment for breast cancer-related lymphedema: paradigms for clinical practice: Am J Clin Oncol, 2018; 41(2); 178-90
38. Damstra JR, Upper limb lymphedema: lymphedema, 2018; 537-46 https://link.springer.com/content/pdf/10.1007%2F978-3-319-52423-8_42.pdf
39. Huang TW, Tseng SH, Lin CC, Effects of manual lymphatic drainage on breast cancer-related lymphedema: A systematic review and meta-analysis of randomized controlled trials: World J Surg Oncol, 2013; 11; 15
40. Andersen L, Hojris I, Erlandsen M, Andersen J, Treatment of breast-cancer-related lymphedema with or without manual lymphatic drainage. A randomized study: Acta Oncol, 2000; 39(3); 399-405
41. Singh B, Disipio T, Peake J, Hayes CS, Systematic review and meta-analysis of the effects of exercise for those with cancer-related lymphedema: Arch Phys Med Rehabil, 2016; 97(2); 302-15.e13
42. Klernäs P, Johnsson A, Horstmann V, Johansson K, Health-related quality of life in patients with lymphoedema – a cross-sectional study: Scand J Caring Sci, 2017; 32(2); 634-44
43. Loudon L, Petrek J, Lymphedema in women treated for breast cancer: Cancer Pract, 2000; 8(2); 65-71
44. Borman P, Yaman A, Denizli M, Karahan S, The reliability and validity of Lymphedema Quality of Life Questionnaire-Leg in Turkish patients with lower limb lymphedema: Lymphat Res Biol, 2020; 18(1); 42-48
45. Van de Pas CB, Biemans AA, Boonen RS, Validation of the lymphoedema Quality-of-Life Questionnaire (LYMQOL) in Dutch Patients diagnosed with lymphoedema of the lower limbs: Phlebology, 2016; 31(4); 257-63
46. Devoogdt N, De Groef A, Hendrickx A, Lymphoedema functioning, disability and health questionnaire for lower limb lymphoedema (Lymph-ICF-LL): Reliability and validity: Phys Ther, 2014; 94(5); 705-21
47. Aitken RC, Measurement of feelings using visual analogue scales: Proc R Soc Med, 1969; 62(10); 989-93
48. Hawker GA, Mian S, Kendzerska T, French M, Measures of adult pain Visual Analog Scale for Pain (VAS Pain), Numeric Rating Scale for Pain (NRS Pain), McGill Pain Questionnaire (MPQ), Short-Form McGill Pain Questionnaire (SF-MPQ), Chronic Pain Grade Scale (CPGS), Short Form-36 Bodily Pain Scale (SF-36 BPS), and Measure of Intermittent and Constant Osteoarthritis Pain (ICOAP): Arthritis Care Res (Hoboken), 2011; 63(11); 240-52
49. Smoot B, Chiavola-Larson L, Lee J, Effect of low-level laser therapy on pain and swelling in women with breast cancer-related lymphedema: A systematic review and meta-analysis: J Cancer Surviv, 2015; 9(2); 287-304
50. Teo I, Novy MD, Chang WD, Examining pain, body image, and depressive symptoms in patients with lymphedema secondary to breast cancer: Psychooncology, 2015; 24(11); 1377-83
51. Puszczalowska-Lizis E, Flak K, Biskup M, Zak M, Physical activity of women after radical unilateral mastectomy and its impact on overall quality of life: Cancer Control, 2020; 7 1073274819900407
52. Borman P, Lymphedema diagnosis, treatment, and follow-up from the viewpoint of physical medicine and rehabilitation specialists: Turk J Phys Med Rehabil, 2018; 64(3); 179-97
53. Özdemir K, Haspolat M, Keser I, The adverse impact of strenuous exercise on breast cancer-related lymphoedema: A case report: Journal of Lymphoedema, 2017; 12(1); 49-51
54. Vignes S, Greene AK, Slavin SA, Brorson H: Lymphedema presentation, diagnosis and treatment, 2015, Switzerland, Springer
55. Zuther JE, Norton S: Lymphedema management: The comprehensive guide for practitioners, 2013; 165-342, Sttuttgard, Thieme Verlag KG
56. Lasinski BB, McKillip Thrift K, Squire D, A systematic review of the evidence for complete decongestive therapy in the treatment of lymphedema from 2004 to 2011: PM R, 2012; 4; 580-601
57. Iyer NS, Cartmel B, Friedman L, Lymphedema in ovarian cancer survivors: Assessing diagnostic methods and the effects of physical activity: Cancer, 2018; 124(9); 1929-37
Tables
 Table 1. Reduction of lower limb volume.
Table 1. Reduction of lower limb volume. Table 2. Assessment of QoL with LYMQOL.
Table 2. Assessment of QoL with LYMQOL. Table 3. Pearson correlation analysis between improvement in QoL domains after CDT and baseline values of reduction LLL, PLL, pain, and age.
Table 3. Pearson correlation analysis between improvement in QoL domains after CDT and baseline values of reduction LLL, PLL, pain, and age. Table 4. Predictors of achieving a significant improvement in QoL (reduction of LLL).
Table 4. Predictors of achieving a significant improvement in QoL (reduction of LLL). Table 5. Predictors of significant improvement in QoL (reduction of PLL).
Table 5. Predictors of significant improvement in QoL (reduction of PLL). In Press
07 Mar 2024 : Clinical Research
Knowledge of and Attitudes Toward Clinical Trials: A Questionnaire-Based Study of 179 Male Third- and Fourt...Med Sci Monit In Press; DOI: 10.12659/MSM.943468
08 Mar 2024 : Animal Research
Modification of Experimental Model of Necrotizing Enterocolitis (NEC) in Rat Pups by Single Exposure to Hyp...Med Sci Monit In Press; DOI: 10.12659/MSM.943443
18 Apr 2024 : Clinical Research
Comparative Analysis of Open and Closed Sphincterotomy for the Treatment of Chronic Anal Fissure: Safety an...Med Sci Monit In Press; DOI: 10.12659/MSM.944127
08 Mar 2024 : Laboratory Research
Evaluation of Retentive Strength of 50 Endodontically-Treated Single-Rooted Mandibular Second Premolars Res...Med Sci Monit In Press; DOI: 10.12659/MSM.944110
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952








