15 July 2020: Clinical Research
Argatroban Increased the Basal Vein Drainage and Improved Outcomes in Acute Paraventricular Ischemic Stroke Patients
Shoufeng Liu12AE, Peipei Liu2BCDEF, Po Wang3BC, Fang Zhang4BD, Lijun Wang5BD, Yu Wang6BD, Hao Lu7B, Xiaofeng Ma4AG*DOI: 10.12659/MSM.924593
Med Sci Monit 2020; 26:e924593
Abstract
BACKGROUND: Since venous drainage in acute arterial ischemic stroke has not been thoroughly researched, we evaluate the effect of argatroban, a selective direct thrombin inhibitor, as a therapy to increase the rate of basal vein Rosenthal (BVR) drainage and improve patients’ post-stroke outcomes.
MATERIAL AND METHODS: In this multicenter clinical trial, 60 eligible patients at 4.5 to 48 hours after the stroke onset were recruited. After being randomly allocated into 2 groups, they were treated with standard therapy either alone or with argatroban.
RESULTS: Compared to the contralateral brain hemisphere, the mean flow velocity (MFV) in BVR drainage was significantly reduced in the stroke-afflicted ipsilateral hemisphere. After treatment with argatroban for 7 days, the MFV from BVR of the ipsilateral hemisphere in the argatroban treated group was significantly increased when compared to the control group. At 90 days after the onset of stroke, the MFV of BVR in the ipsilateral hemisphere was similar in both groups. Compared with controls, the argatroban-treated patients had smaller lesions from baseline to 7 days. Argatroban also improved National Institutes of Health Stroke Scale (NIHSS) scores on day 7 after the onset of stroke. Furthermore, the argatroban group’s neurological functions were superior to those of their untreated counterparts after 90 days. No difference was found in the incidence of adverse reactions between the 2 groups.
CONCLUSIONS: These observations indicate that vein drainage change may contribute to the acute phase of brain edema and the outcomes of ischemic stroke patients.
Keywords: Cerebral Veins, Drainage, Stroke, Ultrasonography, Doppler, Transcranial, Antithrombins, Arginine, Blood Flow Velocity, Brain Ischemia, Drug Therapy, Combination, ischemic stroke, Pipecolic Acids, Sulfonamides, Tissue Plasminogen Activator
Background
Ischemic stroke is a leading cause of disability and death [1]. Past studies focused on the arterial side of cerebral circulation, either thrombolysis or blood clot retrieval, have shown significant clinical improvement in stroke patients [2]. However, some recanalized patients failed to improve due to non-reflow, futile reperfusion, or other causes [3–5].
However, the cerebral venous system’s participation in acute arterial ischemic stroke has been rarely investigated, and the possibility that venous congestion aggravates brain injury remains to be explored [6]. The basal vein Rosenthal (BVR) drains large areas of the brain’s ventricular walls. Indeed, Stolz et al. reported that some patients with decreased flow velocity in the BVR suffered ipsilateral herniation after ischemic lesion [7].
Argatroban, a selective direct thrombin inhibitor, has been adopted to treat acute ischemic stroke [8]. Preclinical [9,10] and clinical [11,12] studies have indicated that argatroban is effective in treating acute ischemic stroke without increasing hemorrhage in the brain. In rat models, argatroban increased blood flow to the lesion area, reduced secondary microthrombi formation, and decreased neurologic deficit [13,14]. Because of its efficacy in lessening secondary microthrombi formation, we theorized that argatroban would increase venous drainage in ischemic periventricular stroke and alleviate its aftereffects.
Material and Methods
STUDY POPULATION:
This multicenter, open-label, randomized and evaluator-blinded study has been registered with
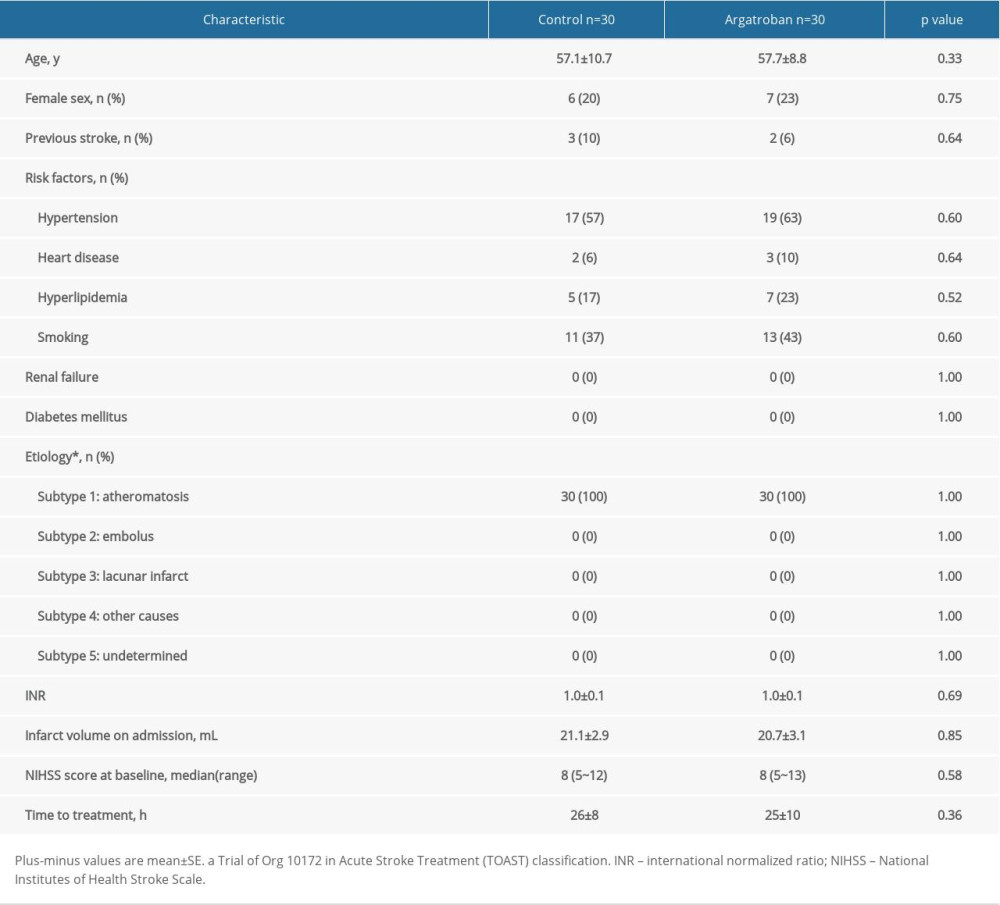
From the 526 patients with acute ischemic stroke, 60 patients with matched clinical characteristics, stroke etiology, lesion location and volume were selected and enrolled into this study in Tianjin Medical University General Hospital, Tianjin Huanhu Hospital, and Baotou Central Hospital, Bao tou, China.
At enrollment, written informed consents were obtained from all patients. The inclusion and exclusion criteria were presented as follows. Inclusion criteria were as follows: 1) age >40 years; 2) large artery atherosclerosis (LAA) defined by magnetic resonance angiography (MRA) and diffusion weighted imaging (DWI
Exclusion criteria were as follows: 1) hemorrhagic stroke; 2) other central nervous diseases; 3) diabetes; 4) tumor or hematological systemic diseases; 5) infection prior to stroke; and 6) antineoplastic or immune modulating therapies. Patients with diabetes were excluded, because their prothrombotic milieu with hyperreactive platelets and hypercoagulation abnormalities may contribute to a reduction in mean flow velocity (MFV) of BVR.
STUDY DESIGN:
Sixty patients with acute paraventricular ischemic stroke were randomly divided into 2 groups: a control group (standard treatment based on guidelines of American Heart Association) and an argatroban group (standard treatment combined with argatroban, TIRP Pharmaceutical Co., Ltd., Tianjin, China). Patients in the 2 groups had similar clinical characteristics when recruited (Table 1). The control group received standard treatment (antihypertensives, antiplatelet agents, and statins) following the guidelines of American Heart Association. Meanwhile, the patients in the argatroban group were additionally given intravenous infusion of 10 mg argatroban twice a day for 7 days consecutively, starting 1 hour after the baseline MRI and no later than 48 hours after symptom onset. The patients enrolled were allocated by computer in a 1: 1 ratio to either the control or the argatroban group in a random way. A centralized web-based randomization system was used for allocation concealment, with the identifier of the participant entered before the allocation. The treatment assignment was known only to the clinicians, but not to the evaluators (Figure 1).
CLINICAL ASSESSMENTS:
Each patient was clinically assessed upon enrollment (baseline) and at day 7 and day 90 after start of treatment, which was blinded to the evaluators (Figure 1). NIHSS was used to assess the neurologic deficit. The modified Rankin scale (mRS) was used to assess the 90-day outcomes. The modified Barthel index (mBI) was used to evaluate the limited ability to perform daily activities. A score of 0 to 1 represented a good outcome and a score of 2 to 5 depicted a poor outcome.
NEUROIMAGING:
At admission, MRIs (MRA, T2FLAIR, and DWI) were conducted using 3 Tesla GE and Siemens, following a comprehensive MRI protocol for acute stroke. Characteristically, ischemic stroke lesion locates in the ventricular walls of one brain hemisphere. In our study, 2 radiologists (blinded to the study design) conducted measurements independently with MIPAV software. By manual outlining and automatic calculation, the lesion area and slice thickness on each of the DWI and FLAIR slices were determined, and then lesion volume was calculated [15]. Another radiologist validated the lesion volume using the semiautomated technique (Cheshire; Perceptive Informatics, Waltham, MA, USA).
TRANSCRANIAL DOPPLER ULTRASONOGRAPHY (TCD):
We performed transcranial Doppler ultrasonography (TCD) measurements using the TC-2000 (Nicolet EME, Kleinostheim, Germany) with a handheld transducer in a range-gated, pulsed-wave mode with a frequency of 2 MHz [16]. TCD insonated the BVR in the ambient cistern, then the posterior cerebral artery cranially and medially. The Doppler gate depth was about 62 mm [17], and the probe was steered clear of the blood flowing direction in the vessel. Bilateral BVR were examined with a TCD device at admission, day 7 and day 90 after stroke onset. MFV in the BVR was calculated and displayed automatically by the TCD device in the ipsilateral and contralateral hemispheres.
STATISTICAL ANALYSES:
Data analyses were performed by SPSS for Windows version 17.0 software (SPSS, Inc., Chicago, IL, USA). Continuous variables (e.g., MFV) were calculated and presented as means±standard error (SE). Discontinuous or abnormally distributed variables were presented as a median (range) and compared by a Mann-Whitney U test. All continuous variables were compared with the
Results
BASELINE CHARACTERISTICS:
Sixty acute ischemic paraventricular stroke patients at 4.5 hours to 48 hours after symptom onset were enrolled in the present study (Figure 1). They were randomly placed in a treatment group or a control group. During the 90-day study, there were no losses to follow-up, dropouts, or fatalities. At the pretreatment baseline, no differences in age, stroke etiology, NIHSS score, or lesion volume were identified (Table 1). Similarly, at that time, no differences of MFV were found in ipsilateral or contralateral BVR between the 2 groups (9.76±1.95 versus 9.50±2.12, P=0.616, 11.26±1.79 versus 11.03±2.29, P=0.663) (Figure 2A). Standard treatment, with or without argatroban, ensued within 48 hours of diagnosis. Compared to these patients’ contralateral hemispheres, BVRs in their ipsilateral hemispheres manifested significant reductions in MFV in both control and argatroban-treated patients (9.50±2.12 versus 11.03±2.29, P=0.010, 9.76±1.95 versus 11.26±1.79, P=0.003) (Figure 2A).
ARGATROBAN INCREASED THE BVR DRAINAGE:
At day 7 after treatment, the argatroban group had a significantly higher MFV in BVR of the ipsilateral hemispheres than the control group (11.80±2.18 versus 10.46±2.14, P=0.020). However, at day 90, the MFV in BVR of the ipsilateral hemispheres no longer reached the level of statistical significance in comparisons between the two groups (11.60±1.79 versus 11.00±2.15, P=0.245) (Figure 2B). In contrast, despite the administration of argatroban, the MFV in the BVR of the contralateral hemisphere did not change at day 7 or day 90 after stroke onset (12.03±2.10 versus 12.13±2.45, P=0.866; 12.23±2.02 versus 11.96±2.09, P=0.618) (Figure 2C). However, the representative TCD in Figure 2D shows that argatroban improved venous drainage from day 1 to day 7 (Figure 2D).
ARGATROBAN REDUCED INFARCTION ENLARGEMENT:
Prior to the treatment, no significant difference was found in lesion volumes between the 2 groups (Table 1). At day 7 after treatment, the argatroban recipients had a significantly smaller lesion volume (day 7 volume versus baseline volume) than the controls (1.51±0.71 versus 4.67±1.29, P=0.002) (Figure 3A, 3B).Though, in the T2FLAIR image, the argatroban group had a smaller lesion volume at day 7 (P=0.87).
ARGATROBAN IMPROVED CLINICAL OUTCOMES:
As shown by the baseline and subsequent clinical evaluations (Figure 4), the argatroban group had only moderate neurological deficits, and most diminished in 7 days after treatment. Compared with the control group, patients who received argatroban exhibited lower NIHSS scores [0.75 (range, −3 to 4) versus 1.5 (range, 0 to 6); P=0.015] from baseline to day 7. The difference in NIHSS scores reached the level of statistical significance at 90 days after symptom onset [3.0 (range, 1 to 7) versus 5.0 (range, 2 to 10); P<0.01] (Figure 4A, 4B). Additionally, the argatroban group had significantly higher mBI scores (which reflected the ability to perform routine activities), than the control group at day 7 and day 90 (76.5±10.9 versus 70.5±11.6, P=0.045; 83.3±7.7 versus 75.3±10.1, P=0.001). And the mRS scores for clinical recovery of 0–1 at post-stroke day 90 were 69% in the argatroban group, compared to 51% in the control group (P=0.014; Figure 4C, 4D).
SAFETY:
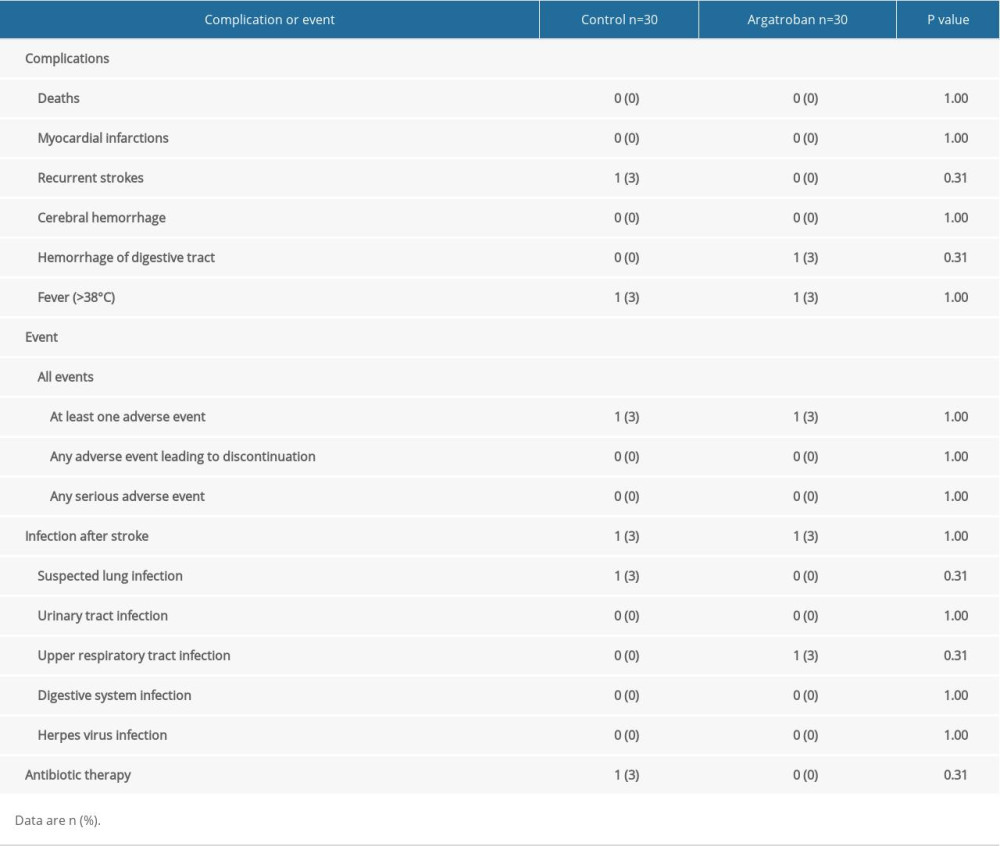
None of the argatroban recipients had recurrent strokes while 3% of patients in the control group had recurrent strokes. One patient in the control group had lung infection after stroke with temperatures >38°C and was administered antibiotics. One patient in the argatroban group had an upper respiratory tract infection. None of the patients in the argatroban group were given antibiotics. One patient had mild hemorrhage of digestive tract (Table 2).
Discussion
LIMITATIONS:
There were 2 limitations to our present study: First, the sample size of the present study was too small to draw a solid conclusion. Studies of larger scales are needed in the future to confirm the results of the present study. Second, there still exists a possibility that infarction reduction and neurological improvement might be directly related to improved BVR outflow in acute ischemic stroke after argatroban treatment. Although our observations in this study precluded us from drawing such a conclusion, this study is a first observation to show that argatroban increased flow velocity of the basal vein after acute ischemic paraventricular stroke for up to 7 days. This effect of argatroban on expanding venous blood flow was accompanied by a reduced infarction volume and improved clinical outcomes at day 7 and day 90 from the onset of stroke. Future confirmation directly linking argatroban to the reduction of venous microthrombi formation is well-warranted.
Conclusions
Vein drainage change may contribute to the acute phase of brain edema and the outcomes of ischemic stroke patients.
Figures
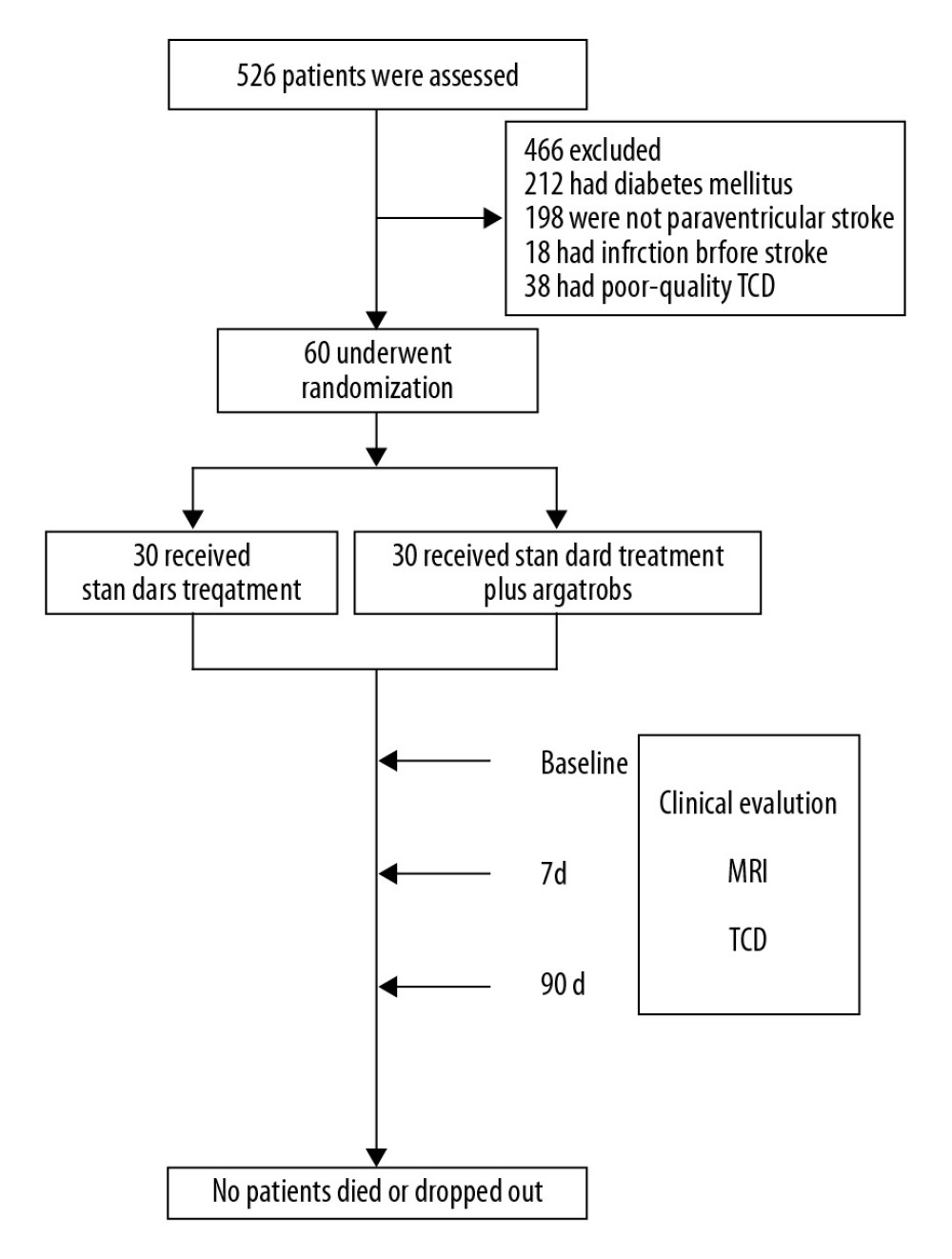 Figure 1. Effect of argatroban on BVR drainage in acute ischemic stroke: trial profile. Sixty patients with acute paraventricular ischemic stroke and matched clinical characteristics, who were beyond the 4.5-hour window for alteplase when recruited, were randomized into 2 groups. Both groups received standard stroke therapy, and one group (n=30) also received 10 mg intravenous argatroban (twice daily for 7 days consecutively). Treatment began between 1 hour after a baseline MRI and 48 hours after symptom onset. NIHSS, mBI, and mRS were conducted. Lesion volume was measured by MRI on admission. MFV in the BVR was measured by TCD at baseline and on day 7 and day 90. BVR – basal vein Rosenthal; MRI – magnetic resonance imaging; NIHSS – National Institutes of Health Stroke Scale; MFV – mean flow velocity; TCD – transcranial Doppler ultrasonography.
Figure 1. Effect of argatroban on BVR drainage in acute ischemic stroke: trial profile. Sixty patients with acute paraventricular ischemic stroke and matched clinical characteristics, who were beyond the 4.5-hour window for alteplase when recruited, were randomized into 2 groups. Both groups received standard stroke therapy, and one group (n=30) also received 10 mg intravenous argatroban (twice daily for 7 days consecutively). Treatment began between 1 hour after a baseline MRI and 48 hours after symptom onset. NIHSS, mBI, and mRS were conducted. Lesion volume was measured by MRI on admission. MFV in the BVR was measured by TCD at baseline and on day 7 and day 90. BVR – basal vein Rosenthal; MRI – magnetic resonance imaging; NIHSS – National Institutes of Health Stroke Scale; MFV – mean flow velocity; TCD – transcranial Doppler ultrasonography. 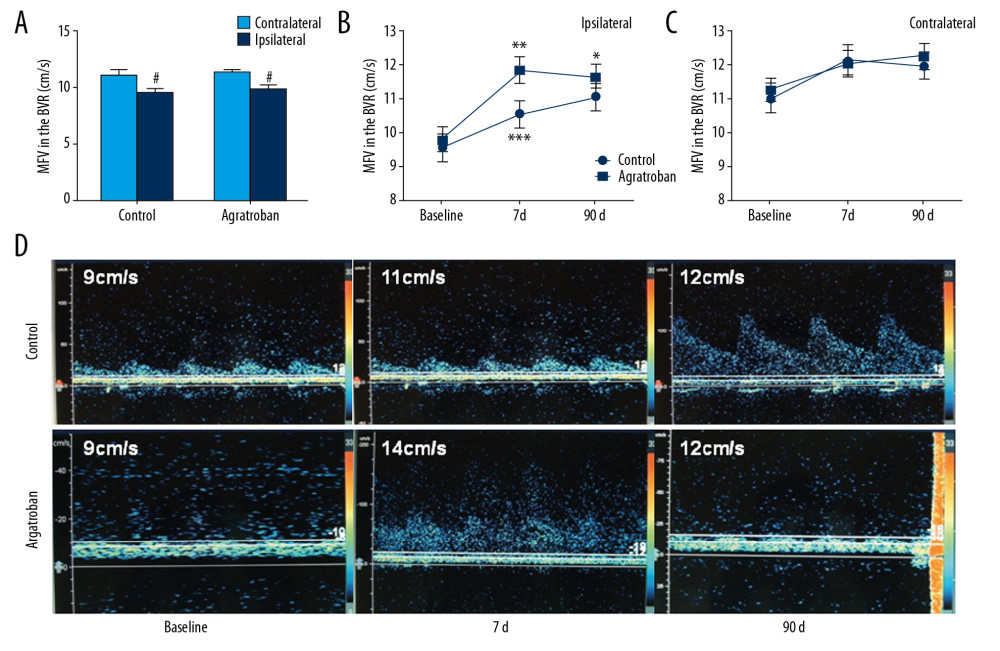 Figure 2. MFV in bilateral BVR of the patients with acute paraventricular ischemic stroke at baseline and after argatroban therapy. (A) Comparison of MFV in ipsilateral versus contralateral BVR between control and argatroban-treated patients. (B) Impact of argatroban on venous flow in the BVR of patients’ ipsilateral hemisphere. (C) Impact of argatroban on venous flow in BVR of patients’ contralateral hemisphere. (D) Representative TCD illustrates the MFV in BVRs of ipsilateral hemispheres in patients at the baseline and at day 7 and day 90 after argatroban treatment. # P<0.05 versus contralateral hemisphere at baseline, * P<0.05, ** P<0.05 compared to baseline of argatroban group, *** P<0.05 compared to control at the same time point. BVR – basal vein Rosenthal; MFV – mean flow velocity; TCD – transcranial Doppler ultrasonography.
Figure 2. MFV in bilateral BVR of the patients with acute paraventricular ischemic stroke at baseline and after argatroban therapy. (A) Comparison of MFV in ipsilateral versus contralateral BVR between control and argatroban-treated patients. (B) Impact of argatroban on venous flow in the BVR of patients’ ipsilateral hemisphere. (C) Impact of argatroban on venous flow in BVR of patients’ contralateral hemisphere. (D) Representative TCD illustrates the MFV in BVRs of ipsilateral hemispheres in patients at the baseline and at day 7 and day 90 after argatroban treatment. # P<0.05 versus contralateral hemisphere at baseline, * P<0.05, ** P<0.05 compared to baseline of argatroban group, *** P<0.05 compared to control at the same time point. BVR – basal vein Rosenthal; MFV – mean flow velocity; TCD – transcranial Doppler ultrasonography. 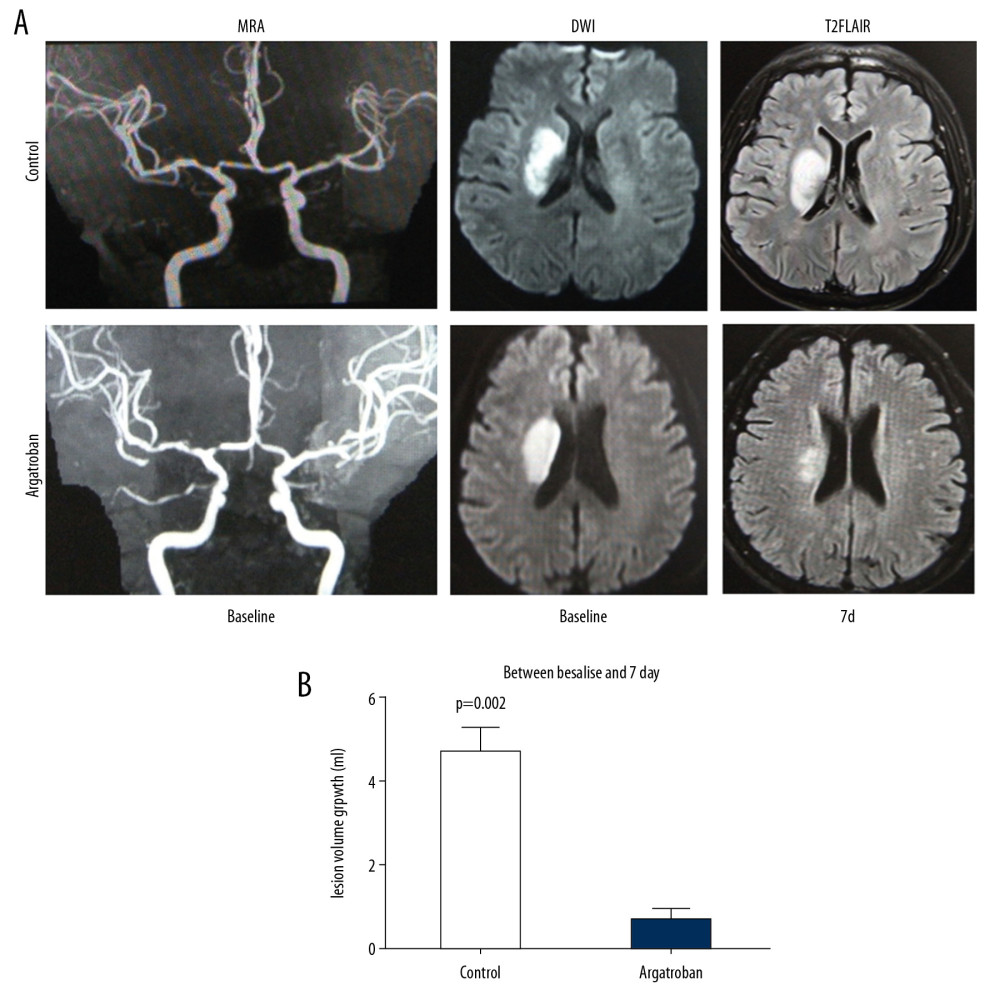 Figure 3. Impact of argatroban on lesion volume growth in patients. (A) MRI images show acute right hemisphere infarct with middle cerebral artery stenosis in a control patient (top) and acute right hemisphere infarct with middle cerebral artery stenosis in an argatroban-treated patient (bottom). Lesion volumes were measured on a DWI (baseline) and T2FLAIR (day 7). (B) Growth of lesion volume from baseline to day 7. Lesion growth equals lesion volume measured on T2FLAIR at day 7 minus that measured on DWI at baseline. Data are presented as mean±SD. An independent t-test was conducted for each comparison. MRI – magnetic resonance imaging; DWI – diffusion weighted imaging; SD – standard deviation.
Figure 3. Impact of argatroban on lesion volume growth in patients. (A) MRI images show acute right hemisphere infarct with middle cerebral artery stenosis in a control patient (top) and acute right hemisphere infarct with middle cerebral artery stenosis in an argatroban-treated patient (bottom). Lesion volumes were measured on a DWI (baseline) and T2FLAIR (day 7). (B) Growth of lesion volume from baseline to day 7. Lesion growth equals lesion volume measured on T2FLAIR at day 7 minus that measured on DWI at baseline. Data are presented as mean±SD. An independent t-test was conducted for each comparison. MRI – magnetic resonance imaging; DWI – diffusion weighted imaging; SD – standard deviation. 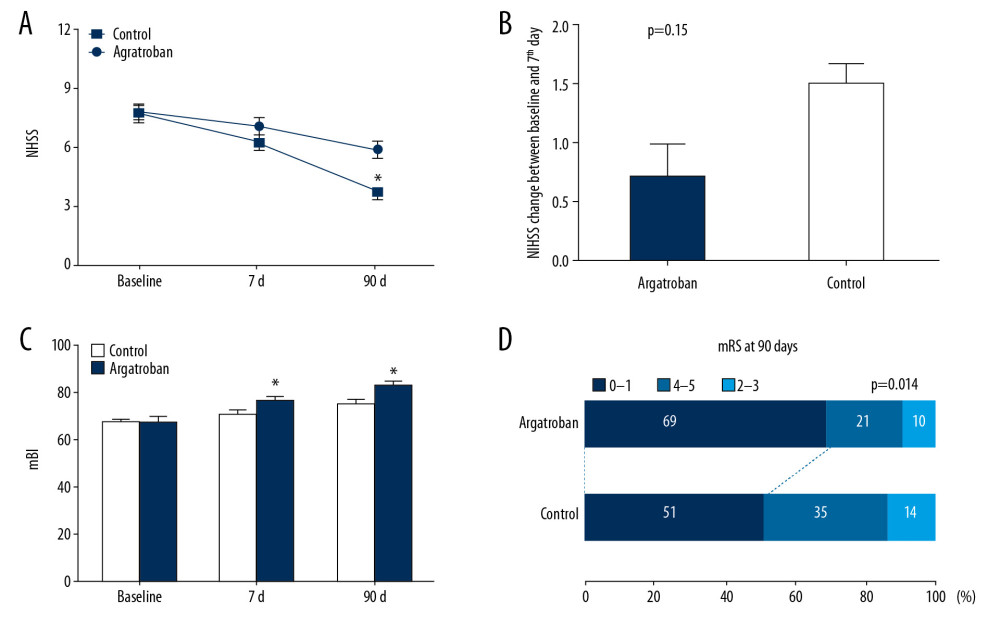 Figure 4. Impact of argatroban on clinical outcomes in the argatroban group compared to the control group. (A) Trends of NIHSS scores from argatroban-treated and control patients. (B) NIHSS values for argatroban-treated and control patients in the 7 days (NIHSS change=baseline – day 7). (C) Comparison of mBI between groups. (D) mRS difference between groups at day 90. * P<0.05 compared to the control at the same time point. NIHSS – National Institutes of Health Stroke Scale; mBI – modified Barthel Index; mRS – modified Rankin Scale.
Figure 4. Impact of argatroban on clinical outcomes in the argatroban group compared to the control group. (A) Trends of NIHSS scores from argatroban-treated and control patients. (B) NIHSS values for argatroban-treated and control patients in the 7 days (NIHSS change=baseline – day 7). (C) Comparison of mBI between groups. (D) mRS difference between groups at day 90. * P<0.05 compared to the control at the same time point. NIHSS – National Institutes of Health Stroke Scale; mBI – modified Barthel Index; mRS – modified Rankin Scale. References
1. Chwojnicki K, Ryglewicz D, Wojtyniak B, Acute ischemic stroke hospital admissions, treatment, and outcomes in Poland in 2009–2013: Front Neurol, 2018; 9; 134
2. Zhang JH, Obenaus A, Liebeskind DS, Recanalization, reperfusion, and recirculation in stroke: J Cereb Blood Flow Metab, 2017; 37(12); 3818-23
3. Manning NW, Warne CD, Meyers PM, Reperfusion and clinical outcomes in acute ischemic stroke: systematic review and meta-analysis of the Stent-Retriever-Based, Early Window Endovascular Stroke Trials: Front Neurol, 2018; 9; 301
4. Iglesias-Rey R, Rodríguez-Yáñez M, Rodríguez-Castro E, Worse outcome in stroke patients treated with rt-PA without early reperfusion: Associated factors: Transl Stroke Res, 2018; 9(4); 347-55
5. Puetz V, Gerber JC, Krüger P, Cerebral venous drainage in patients with space-occupying middle cerebral artery infarction: Effects on functional outcome after hemicraniectomy: Front Neurol, 2018; 9; 876
6. Li Q, Khatibi N, Zhang JH, Vascular neural network: The importance of vein drainage in stroke: Transl Stroke Res, 2014; 5(2); 163-66
7. Stolz E, Gerriets T, Babacan SS, Intracranial venous hemodynamics in patients with midline dislocation due to postischemic brain edema: Stroke, 2002; 33(2); 479-85
8. Escolar G, Bozzo J, Maragall S, Argatroban: A direct thrombin inhibitor with reliable and predictable anticoagulant actions: Drugs Today (Barc), 2006; 42(4); 223-36
9. LaMonte MP, Nash ML, Wang DZ, Argatroban anticoagulation in patients with acute ischemic stroke (ARGIS-1): A randomized, placebo-controlled safety study: Stroke, 2004; 35(7); 1677-82
10. Sugg RM, Pary JK, Uchino K, Argatroban tPA stroke study: Study design and results in the first treated cohort: Arch Neurol, 2006; 63(8); 1057-62
11. Fujimori Y, Wakui M, Katagiri H, Evaluation of anticoagulant effects of direct thrombin inhibitors, dabigatran and argatroban, based on the Lineweaver-Burk plot applied to the Clauss assay: J Clin Pathol, 2016; 69(4); 370-72
12. Park JS, Park SS, Koh EJ, Treatment for patients with acute ischemic stroke presenting beyond six hours of ischemic symptom onset: Effectiveness of intravenous direct thrombin inhibitor, argatroban: J Korean Neurosurg Soc, 2010; 47(4); 258-64
13. LaMonte MP, Brown PM, Hursting MJ, Alternative parenteral anticoagulation with argatroban, a direct thrombin inhibitor: Expert Rev Cardiovasc Ther, 2005; 3(1); 31-41
14. Kawai H, Umemura K, Nakashima M, Effect of argatroban on microthrombi formation and brain damage in the rat middle cerebral artery thrombosis model: Jap J Pharmacol, 1995; 69(2); 143-48
15. Zhang F, Yan C, Wei C, Vinpocetine inhibits NF-κB-dependent inflammation in acute ischemic stroke patients: Transl Stroke Res, 2018; 9(2); 174-84
16. Gao S, Lam WW, Chan YL, Optimal values of flow velocity on transcranial Doppler in grading middle cerebral artery stenosis in comparison with magnetic resonance angiography: J Neuroimaging, 2002; 12(3); 213-18
17. Stolz E, Kaps M, Kern A, Transcranial color-coded duplex sonography of intracranial veins and sinuses in adults. Reference data from 130 volunteers: Stroke, 1999; 30(5); 1070-75
18. Ahnstedt H, Sweet J, Cruden P, Effects of early post-ischemic reperfusion and tPA on cerebrovascular function and nitrosative stress in female rats: Transl Stroke Res, 2016; 7(3); 228-38
19. Lu Y, Wang J, Huang R, Microbubble-mediated sonothrombolysis improves outcome after thrombotic microembolism-induced acute ischemic stroke: Stroke, 2016; 47(5); 1344-53
20. Nagai N, Zhao BQ, Suzuki Y, Tissue-type plasminogen activator has paradoxical roles in focal cerebral ischemic injury by thrombotic middle cerebral artery occlusion with mild or severe photochemical damage in mice: J Cereb Blood Flow Metab, 2002; 22(6); 648-51
21. Siler DA, Gonzalez JA, Wang RK, Intracisternal administration of tissue plasminogen activator improves cerebrospinal fluid flow and cortical perfusion after subarachnoid hemorrhage in mice: Transl Stroke Res, 2014; 5(2); 227-37
22. Mandava P, Martini SR, Munoz M, Hyperglycemia worsens outcome after rt-PA primarily in the large-vessel occlusive stroke subtype: Transl Stroke Res, 2014; 5(4); 519-25
23. Paris C, Derex L, Intravenous thrombolysis in ischemic stroke: Therapeutic perspectives: Rev Neurol, 2015; 171(12); 866-75
24. Asadi H, Yan B, Dowling R, Advances in medical revascularisation treatments in acute ischemic stroke: Thrombosis, 2014; 2014 714218
25. Aaslid R, Markwalder TM, Nornes H, Noninvasive transcranial Doppler ultrasound recording of flow velocity in basal cerebral arteries: J Neurosurg, 1982; 57(6); 769-74
26. Valdueza JM, Schmierer K, Mehraein S, Assessment of normal flow velocity in basal cerebral veins. A transcranial doppler ultrasound study: Stroke, 1996; 27(7); 1221-25
27. Aaslid R, Newell DW, Stooss R, Assessment of cerebral autoregulation dynamics from simultaneous arterial and venous transcranial Doppler recordings in humans: Stroke, 1991; 22(9); 1148-54
28. Ono M, Rhoton AL, Peace D, Microsurgical anatomy of the deep venous system of the brain: Neurosurgery, 1984; 15(5); 621-57
29. Karatas A, Cakir V, Sevin E, Angiographic assessment of variants of basal vein of Rosenthal in idiopathic subarachnoid hemorrhage: Neurol Neurochir Pol, 2015; 49(4); 207-11
30. Baumgartner RW, Nirkko AC, Muri RM, Transoccipital power-based color-coded duplex sonography of cerebral sinuses and veins: Stroke, 1997; 28(7); 1319-23
31. Schreiber SJ, Stolz E, Valdueza JM, Transcranial ultrasonography of cerebral veins and sinuses: Eur J Ultrasound, 2002; 16(1–2); 59-72
32. Ostergaard L, Aamand R, Karabegovic S, The role of the microcirculation in delayed cerebral ischemia and chronic degenerative changes after subarachnoid hemorrhage: J Cereb Blood Flow Metab, 2013; 33(12); 1825-37
33. Chen S, Chen Y, Xu L, Venous system in acute brain injury: Mechanisms of pathophysiological change and function: Exp Neurol, 2015; 272; 4-10
34. Desilles JP, Loyau S, Syvannarath V, Alteplase reduces downstream microvascular thrombosis and improves the benefit of large artery recanalization in stroke: Stroke, 2015; 46(11); 3241-48
35. Wong KS, Is the measurement of cerebral microembolic signals a good surrogate marker for evaluating the efficacy of antiplatelet agents in the prevention of stroke?: Eur Neurol, 2005; 53(3); 132-39
36. Gao S, Wong KS, Characteristics of microembolic signals detected near their origins in middle cerebral artery stenoses: J Neuroimaging, 2003; 13(2); 124-32
37. Lyden P, Pereira B, Chen B, Direct thrombin inhibitor argatroban reduces stroke damage in 2 different models: Stroke, 2014; 45(3); 896-99
38. Hosomi N, Naya T, Kohno M, Efficacy of anti-coagulant treatment with argatroban on cardioembolic stroke: J Neurol, 2007; 254(5); 605-12
39. Kobayashi S, Tazaki Y, Effect of the thrombin inhibitor argatroban in acute cerebral thrombosis: Semin Thromb Hemost, 1997; 23(6); 531-34
40. Barreto AD, Alexandrov AV, Lyden P, The argatroban and tissue-type plasminogen activator stroke study: Final results of a pilot safety study: Stroke, 2012; 43(3); 770-75
Figures
 Figure 1. Effect of argatroban on BVR drainage in acute ischemic stroke: trial profile. Sixty patients with acute paraventricular ischemic stroke and matched clinical characteristics, who were beyond the 4.5-hour window for alteplase when recruited, were randomized into 2 groups. Both groups received standard stroke therapy, and one group (n=30) also received 10 mg intravenous argatroban (twice daily for 7 days consecutively). Treatment began between 1 hour after a baseline MRI and 48 hours after symptom onset. NIHSS, mBI, and mRS were conducted. Lesion volume was measured by MRI on admission. MFV in the BVR was measured by TCD at baseline and on day 7 and day 90. BVR – basal vein Rosenthal; MRI – magnetic resonance imaging; NIHSS – National Institutes of Health Stroke Scale; MFV – mean flow velocity; TCD – transcranial Doppler ultrasonography.
Figure 1. Effect of argatroban on BVR drainage in acute ischemic stroke: trial profile. Sixty patients with acute paraventricular ischemic stroke and matched clinical characteristics, who were beyond the 4.5-hour window for alteplase when recruited, were randomized into 2 groups. Both groups received standard stroke therapy, and one group (n=30) also received 10 mg intravenous argatroban (twice daily for 7 days consecutively). Treatment began between 1 hour after a baseline MRI and 48 hours after symptom onset. NIHSS, mBI, and mRS were conducted. Lesion volume was measured by MRI on admission. MFV in the BVR was measured by TCD at baseline and on day 7 and day 90. BVR – basal vein Rosenthal; MRI – magnetic resonance imaging; NIHSS – National Institutes of Health Stroke Scale; MFV – mean flow velocity; TCD – transcranial Doppler ultrasonography. Figure 2. MFV in bilateral BVR of the patients with acute paraventricular ischemic stroke at baseline and after argatroban therapy. (A) Comparison of MFV in ipsilateral versus contralateral BVR between control and argatroban-treated patients. (B) Impact of argatroban on venous flow in the BVR of patients’ ipsilateral hemisphere. (C) Impact of argatroban on venous flow in BVR of patients’ contralateral hemisphere. (D) Representative TCD illustrates the MFV in BVRs of ipsilateral hemispheres in patients at the baseline and at day 7 and day 90 after argatroban treatment. # P<0.05 versus contralateral hemisphere at baseline, * P<0.05, ** P<0.05 compared to baseline of argatroban group, *** P<0.05 compared to control at the same time point. BVR – basal vein Rosenthal; MFV – mean flow velocity; TCD – transcranial Doppler ultrasonography.
Figure 2. MFV in bilateral BVR of the patients with acute paraventricular ischemic stroke at baseline and after argatroban therapy. (A) Comparison of MFV in ipsilateral versus contralateral BVR between control and argatroban-treated patients. (B) Impact of argatroban on venous flow in the BVR of patients’ ipsilateral hemisphere. (C) Impact of argatroban on venous flow in BVR of patients’ contralateral hemisphere. (D) Representative TCD illustrates the MFV in BVRs of ipsilateral hemispheres in patients at the baseline and at day 7 and day 90 after argatroban treatment. # P<0.05 versus contralateral hemisphere at baseline, * P<0.05, ** P<0.05 compared to baseline of argatroban group, *** P<0.05 compared to control at the same time point. BVR – basal vein Rosenthal; MFV – mean flow velocity; TCD – transcranial Doppler ultrasonography. Figure 3. Impact of argatroban on lesion volume growth in patients. (A) MRI images show acute right hemisphere infarct with middle cerebral artery stenosis in a control patient (top) and acute right hemisphere infarct with middle cerebral artery stenosis in an argatroban-treated patient (bottom). Lesion volumes were measured on a DWI (baseline) and T2FLAIR (day 7). (B) Growth of lesion volume from baseline to day 7. Lesion growth equals lesion volume measured on T2FLAIR at day 7 minus that measured on DWI at baseline. Data are presented as mean±SD. An independent t-test was conducted for each comparison. MRI – magnetic resonance imaging; DWI – diffusion weighted imaging; SD – standard deviation.
Figure 3. Impact of argatroban on lesion volume growth in patients. (A) MRI images show acute right hemisphere infarct with middle cerebral artery stenosis in a control patient (top) and acute right hemisphere infarct with middle cerebral artery stenosis in an argatroban-treated patient (bottom). Lesion volumes were measured on a DWI (baseline) and T2FLAIR (day 7). (B) Growth of lesion volume from baseline to day 7. Lesion growth equals lesion volume measured on T2FLAIR at day 7 minus that measured on DWI at baseline. Data are presented as mean±SD. An independent t-test was conducted for each comparison. MRI – magnetic resonance imaging; DWI – diffusion weighted imaging; SD – standard deviation. Figure 4. Impact of argatroban on clinical outcomes in the argatroban group compared to the control group. (A) Trends of NIHSS scores from argatroban-treated and control patients. (B) NIHSS values for argatroban-treated and control patients in the 7 days (NIHSS change=baseline – day 7). (C) Comparison of mBI between groups. (D) mRS difference between groups at day 90. * P<0.05 compared to the control at the same time point. NIHSS – National Institutes of Health Stroke Scale; mBI – modified Barthel Index; mRS – modified Rankin Scale.
Figure 4. Impact of argatroban on clinical outcomes in the argatroban group compared to the control group. (A) Trends of NIHSS scores from argatroban-treated and control patients. (B) NIHSS values for argatroban-treated and control patients in the 7 days (NIHSS change=baseline – day 7). (C) Comparison of mBI between groups. (D) mRS difference between groups at day 90. * P<0.05 compared to the control at the same time point. NIHSS – National Institutes of Health Stroke Scale; mBI – modified Barthel Index; mRS – modified Rankin Scale. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387