12 November 2020: Animal Study
Effects of Vitamin D3 on Intestinal Flora in a Mouse Model of Inflammatory Bowel Disease Treated with Rifaximin
Zijun Gu1BCDE, Mingxiu Duan2BCD, Yan Sun1BC, Tian Leng1BC, Ting Xu1BC, Yang Gu1ABE, Zejuan Gu13CD, Zheng Lin13AF*, Lu Yang1EF, Minghui Ji1ACEGDOI: 10.12659/MSM.925068
Med Sci Monit 2020; 26:e925068
Abstract
BACKGROUND: Rifaximin is an antimicrobial agent used to treat inflammatory bowel disease (IBD). Vitamin D3 can control IBD due to its effects on inflammatory cytokines. The purpose of this study was to assess the effect of vitamin D3 on the intestinal flora of a dextran sulfate sodium (DSS)-induced mouse model treated with rifaximin.
MATERIAL AND METHODS: The mouse model of IBD was developed using DSS (4%) administered via the drinking water. Twenty-four male C57BL6 mice were divided into the control group with a normal diet (N=6), the DSS group with a normal diet (N=6), the DSS group with a normal diet treated with rifaximin (N=6), and the DSS group with a normal diet treated with rifaximin and vitamin D3 (N=6). After 14 days, the colonic tissue was studied histologically. Serum levels of tumor necrosis factor-α (TNF-α) and interleukin-1β (IL-1β) and enzyme-linked immunosorbent assay (ELISA) were used to measure the level of IL-6 and P65, and phospho-p65 was measured by western blot. 16S rRNA gene sequencing was used to analyze fecal samples.
RESULTS: In the DSS mouse model of IBD, rifaximin reduced the inflammation severity of the colon and reduced the expression of phospho-p65, p65, TNF-α, and IL-6. In the DSS+rifaximin+vitamin D3 group, the therapeutic influences of rifaximin, in terms of weight loss and colonic disease activity, were significantly reduced, and the gut microbiota of the mice were completely changed in composition and diversity.
CONCLUSIONS: In a mouse model of IBD, treatment with vitamin D3 significantly increased the metabolism of rifaximin and reduced its therapeutic effects.
Keywords: Dextran Sulfate, Inflammatory Bowel Diseases, Microbiota, Animals, Cholecalciferol, Colon, Cytokines, Disease Models, Animal, Gastrointestinal Microbiome, inflammation, Male, Mice, Inbred C57BL, Phylogeny, rifaximin
Background
Ulcerative colitis (UC) and Crohn’s disease (CD) are 2 major types of inflammatory bowel diseases (IBD) [1, 2]. They result in inflammation and ulcers, with lesions usually located in the mucosa and submucosa of the colon [2]. Diarrhea, abdominal pain, bloody stool, and mucous pus are the main clinical symptoms of IBD [3]. Previous studies demonstrated that the dextran sulfate sodium (DSS)-induced murine colitis model could mimic human UC pathology. Recently, these findings have also been supported by several preclinical studies in which this model was used to evaluate the effects of anti-inflammatory agents [4]. Diarrhea and blood in the stool can cause malnutrition in UC patients. Therefore, nutrition powder, consisting of high levels of fat, carbohydrates, protein, minerals, and vitamin D3 (VD3), is widely used in clinical practice to improve patients’ nutrition status [5]. VD3 is a major ingredient of nutrition powder [6].
VD3 deficiency is reported to be a major factor in the increasing incidence of IBD [7]. Vitamin D receptor (VDR) has a high degree of expression in intestinal epithelial cells (IECs), suggesting that excessive apoptosis of IECs may partly explain the development of inflammation in the colon, ultimately leading to IBD [8]. In addition, VDR knockdown in mice can damage the mucosal barriers and increase the sensitivity to mucosal injury, which is likely to accelerate IBD progression [9].
Rifaximin has long been an important treatment for irritable bowel syndrome (IBS) [10]. Later, as a gut-specific human pregnane X receptor (PXR) agonist, rifaximin was also used to treat IBD patients. Some researchers have reported its ability to relieve colitis in humanized PXR mice [11]. Experimental results showed that rifaximin’s anti-inflammatory effects in colitis were attributed to PXR-dependent inhibition of NF kappa B-driven cytokine/chemokine production. Previous studies have demonstrated that intestinal flora imbalance is one of the primary causes of IBD [12]. Rifaximin was proved to have the potential to improve the imbalance of intestinal flora. In addition, another way that rifaximin can preserve intestinal flora balance by regulating relative abundance [13,14]. The abundance of Bifidobacteria and Faecalibacterium prausnitzii can also been influenced by rifaximin in CD patients, and can eventually alleviate the disease-related symptoms of patients [15].
In the human liver, CYP3A4 is a very important substance in the inactivation of 1,25(OH)2D3. It has also been confirmed in our previous research that by activating CYP3A4 activity and hydrolyzing midazolam, VD3 can ultimately be transformed into 1,25(OH)2D3 [16].
In the human liver it has been confirmed that microsomal CYP3A4 is the main cause of 1,25(OH)2D3 inactivation [19]. CYP3A4 is constitutively expressed in numerous tissues, especially in the liver and intestine. CYP3A4 is highly susceptible to modulation by endogenous and exogenous substances [20]. Several drugs, dietary substances, and environmental agents have been identified as CYP3A4 inhibitors and inducers [21].
In the present study, histological and macroscopic methods was used to verify the effect of VD3 on C57BL/6 mice with acute colitis induced by DSS. The evaluation of the levels of colonic infiltration of immune cells cytokine was carried out. In addition, VD3’s effect on intestinal microbes in mice treated with DSS was also studied (included in the research scope).
Material and Methods
ETHICS STATEMENT:
All animal experiments were performed in accordance with the requirements of the Animal Care and Use Committee of Nanjing Medical University (approval no. IACUC-1903045). It has been confirmed the study is in line with the national and international norms, in keeping with all procedures of ethical standards as well as the Helsinki Declaration.
ANIMAL:
Male C57BL/6 mice (age 6–8 weeks, body weight 18–20 g, Shanghai SLAC Laboratory Animal Co., Ltd., Shanghai, China) were housed with a 12 h/12 h light/dark cycle, room temperature 22°C, and humidity 55%. At first, 24 mice were placed in a plastic cage with free access to water and standard mouse food. Then, the mice were transferred and kept in the metabolic cage during the experiments. Mice were allowed 1 week to acclimate before the beginning of the study. Animal care and use conformed to animal welfare guidelines.
TREATMENT OF EXPERIMENTAL COLITIS:
All mice were randomly divided into 4 groups (n=6 per group): I, Control; II, DSS; III, DSS+Rifaximin; and IV, DSS+Rifaximin+VD3. Experimental acute colitis was induced by administration of dextran sulfate sodium (DSS, 36–50 kDa; MP Biomedicals Solon, OH, USA). Mice in the control group received sterile water for 14 days. In the DSS group, for the first 7 days, mice drank sterile water, and then drank 4% DSS water for 7 days. The DSS+Rifaximin group was given sterile water and DSS in exactly the same pattern as the DSS group, but 10 mg/mouse/day rifaximin (rifaximin purity ≥98.0%) was administrated intragastrically during the last 4 days. For the DSS+Rifaximin+VD3 group, sterile water and DSS were provided the same as in the other groups, but rifaximin (10 mg/mice/day) and VD3 (20 mg/mice/day) were intragastrically administrated for the last 7 days. During DSS treatment, mice were weighed every day. On Day14, after cardiac puncture, blood samples were collected and then coagulated for 2 h before centrifugation (3000 rpm, 4°C, 10 min) at room temperature. Serum was frozen at −80°C for further use. Colons were excised at necropsy and their lengths were measured between the anus and the ileocecal junction. As in previous research, the detailed disease activity index (DAI) was used to score the DSS-induced colitis. Briefly, the DAI was divided into 3 parts: the total scores of weight loss were 0 points for none; 1 point for 0–5%; 2 points for 5–10%; 3 points for 10–20%; and 4 points for >20%. The consistency change of feces was assessed as 0 points for none; 2 points for loose stool; and 4 points for diarrhea. Bleeding was scored as 0 points for none; 1 point for trace amount; 2 points for mild hemoccult; 3 points for obvious hemoccult; and 4 points for hemorrhage. DAI, which can reflect disease severity, was measured by the sum of weight loss, fecal consistency, and rectal bleeding level [22]. The DAI was assessed every day at the same time by the same person, who was unaware of group assignments.
HISTOLOGY ASSESSMENT:
The distal segment of colon sections was fixed with 10% neutral buffered formalin (1–2 cm from the anal verge), then embedded in paraffin and sliced and stained with hematoxylin and eosin for further assessment using with Panoramic digital slide scanners (Panoramic SCAN, 3DHISTECH Kft, Budapest, Hungary).
The distal colon section (1–2 cm from the anal edge) was fixed with 10% neutral buffered formalin, paraffin-embedded sections were stained with eosin and hematoxylin, and further study was conducted using a digital biopsy scanner (PANnooled scan, 3DHISTECH Kft, Budapest, Hungary).
WESTERN BLOT:
We discovered immunocomplexes by using enhanced chemiluminescence (ECL) (CST, Danvers, MA, USA) with specific antibodies: Anti-p-p65, Anti-p65, Anti-Lamin B (CST, Danvers, MA, USA). Protein bands on the blots were evaluated by densitometric analysis using Image J software [23].
PROFILING OF MULTIPLEX SERUM CYTOKINE:
The serum cytokine levels of IL-1β, TNF-α, and IL-6 in the DSS-treated mice were measured using ELISA kits according the protocols recommended by the manufacturer [22]. The cytokine concentration in the sample was calculated via a 5-parameter logic or spline curve fitting method.
MICROBIAL DNA EXTRACTION:
Fecal samples were collected every day until mice were killed on the 14th day. The whole stool of a single mouse was considered as 1 sample. Bacterial genomic DNA was extracted from stool samples (200 mg) using the QIA AMP RR DNA Fecal Mini-kit (Qiagen, Hilden, Germany) based on the manufacturer’s instructions.
BIOINFORMATICS:
We sequenced 16S rRNA of bacteria at the V3 hypervariable region using Illumina MiSeq (pe300). The QIIME toolkit was used to trim and classify these sequences. Using mothur software, the operational taxonomic units (OTUs) were clustered by high-quality reads. The effective data were clustered on the Meiji cloud platform, and the sequences were clustered into OTUs. Then, the abundance, diversity, similarity, and composition of bacteria were analyzed. With about 70% bootstrap grade, taxonomy-based analysis was used to classify the sequence, which was a part of the Naïve Bayesian Classifier item of the Ribosomal Database Project for Microbial Ecology at Michigan State University [24].
STATISTICAL ANALYSIS:
In SPSS20.0 (Chicago, IL, USA), single-factor analysis was used to calculate the variance of mouse body weight, colon length, and DAI score. Differences in fecal microbiota cytokines and taxonomy were evaluated by Mann-Whitney U test. A significant difference was defined as a
Results
VD3 REDUCED RIFAXIMIN’S EFFECT ON DISEASE ACTIVITY INDEX (DAI) OF DSS-INDUCED MICE:
On the 4th day, when colitis was induced by DSS, palpable diarrhea and rectal bleeding could be observed in all mice except for those in the control group. There was a significant decrease in weight after the 5-day DSS treatment. DAI continued to increase in the DSS group until the end of DSS treatment. Rifaximin treatment significantly attenuated body weight loss (Figure 1A), and also reduced diarrhea and rectal bleeding (Figure 1B). Colon length was measured during necropsy, as an indicator of severity of inflammation. Compared to the DSS group, colons of mice in the DSS+rifaximin group were longer. In the DSS group, mice showed more severe symptoms, such as severe diarrhea and a greater blood loss. In the treatment group, the symptoms of rectal bleeding and diarrhea were distinctly improved by administration of rifaximin (Figure 1C). Body weight and colon length (Figure 1D) in the DSS+rifaximin+VD3 group were not increased compared with the DSS+rifaximin group. Our results suggest that VD3 can lead to high expression of CYP3A4, and thus accelerate the metabolism of rifaximin, which may be the mechanism underlying the present results.
VD3 AFFECTED THE HISTOPATHOLOGICAL CHANGES OF MOUSE COLONS CAUSED BY DSS AND RIFAXIMIN:
Using HE staining, we performed histological evaluation of mouse colon tissues. Figure 2 shows results by group. Mice demonstrated an integrated crypt construction in the control group, but samples from DSS-treated mice showed more histological damage (e.g., the disappearance of histological structure, distinct epithelial disintegration, epithelial barrier destruction, and visible crypts decrease) compared to the control group. Furthermore, the results indicated that mononuclear and granulocytes cells had infiltrated into the mucosa and even submucosa in the colon tissues of mice with DSS-induced colitis (Figure 2). Macroscopic and histological examinations showed that rifaximin mitigated the severity of colon injuries. Compared with the DSS group, histological structure in rifaximin-treated mice was more complete and there was less epithelial disintegration. In the DSS+rifaximin+VD3 group, colon injuries were milder than in the DSS group but were more serious than in the DSS+rifaximin group, and their histological structure was not as complete as in the rifaximin-treated mice. In addition, in the DSS+rifaximin+VD3 group, granulocytes and mononuclear cells infiltrated into the mucosa and submucosa (Figure 2).
VD3 DECREASED SERUM CYTOKINES IN DSS-INDUCED MICE VIA AFFECTING RIFAXIMIN:
To investigate whether the anti-inflammation effects of rifaximin on DSS-induced colitis would be affected by VD3, the levels of 3 cytokines (TNF-α, IL-1β, and IL-6) were measured in parallel (Figure 3). As shown in Figure 3, a significant increase of inflammatory factors (TNF-α, IL-6 and IL-1β) was found in DSS-treated mice. After treatment with rifaximin, the level of TNF-α and IL-6 in serum was distinctly reduced. However, in the DSS+rifaximin+VD3 group, the levels of TNF-α and IL-6 were dramatically higher than in the rifaximin-treated group. This shows that VD3 can interfere with the anti-inflammatory of rifaximin to some extent. Nonetheless, there were no obvious differences among the 3 groups in IL-1β serum level.
DSS-INDUCED INFLAMMATORY COLONIC INFILTRATION WAS ENERVATED BY RIFAXIMIN AND VD3 REVERSE ITS EFFECT:
NF-κB is a transcription factor that plays important roles in inflammatory processes. In addition, it has been proved that p65 phosphorylation is related to NF-κB activity. Western blot analysis showed that the high expression of p-P65 was influenced by DSS and was attenuated by rifaximin (Figure 4A). Compared with the DSS group, the protein levels of p-p65 and p65 were significantly lower in the rifaximin group (Figure 4B, P<0.05). More importantly, p-p65/p65 was significantly upregulated in the VD3 treatment group (DSS+Rif+VD3 group) compared to the DSS group (Figure 4B, P<0.05).
VD3 INFLUENCED THE EFFECT OF RIFAXIMIN ON MICROBIAL COMPOSITION IN DSS-INDUCED MICE:
Differences in the intestinal flora in the DSS+rifaximin group and the DSS+rifaximin+VD3 group show the effect of VD3 (Figure 5). Figure 5A shows the richness of intestinal flora at the phylum level, and it can be observed that the abundance of flora is not at the same level in the 4th group. Figure 5B shows differences in the proportion of Bacteroidetes and Firmicutes in each group and shows that the composition of bacteria in the DSS group has changed greatly compared with the other groups. In particular, there were significant differences in the composition of Firmicutes and Bacteroidetes between the DSS+rifaximin group and the DSS+rifaximin+VD3 group. These 2 flora can affect the release of energy in the intestinal tract of mice, and have an important impact on the nutritional status and weight of mice.
ANALYSIS OF INTESTINAL FLORA STRUCTURE AND COMPOSITION OF MICE IN EACH GROUP:
The species in the phylum levels are Bacteroidetes, Firmicutes, Proteobacteria, and Verrucomicrobia, according to the results of species analysis (Table 1). The dominant bacteria in each group of mice are consistent. It can be observed that the sequencing and composition of phylum horizontal dominant bacteria in the DSS group have significantly changed. Bacteroidetes significantly declined in the DSS group while Proteobacteria, Firmicutes, Tenericutes, Deferribacteres, and Verrucomicrobia were all improved to varying degrees compared with the control group. In addition, intestinal flora sequencing and composition in the DSS+rifaximin group are relatively close to that of the blank control group.
The annotation is further extended to the genus level to list the top 10 genera (Table 2). In the DSS group, the proportion of Helicobacter was the highest (16.70%), followed by S24-7-Bacteroidales (11.26%), Bacteroides (10.84%), Escherichia-Shigella (11.23%), Turicibacter (8.91%), and Mucispirillum (4.73%). After rifaximin treatment, the dominant bacteria in the intestinal tract of the mice were mostly probiotics, such as Bacteroidales_S24-7(25.17%), Morganella (11.40%), Erysipelatoclostridium (14.85%), Bacteroides (7.69%), Bacteroides (7.69%), and Escherichia-Shigella (4.37%). Furthermore, for the DSS+rifaximin+VD3 group, the dominant bacteria included Bacteroides (24.56%), Escherichia-Shigella (10.93%), and Erysipelato clostridium (6.05%).
Discussion
The gut microbiota exerts an important influence on the occurrence and development of IBD. Moreover, the inflammatory process typical of the disease can also be affected by the gut microbiota. Consequently, antimicrobial agents are used to reconstruct intestinal homeostasis in the course of treatment. Nevertheless, as the duration of treatment with drugs increases, there is increased risk of systemic adverse reactions due to antibiotics, which may even lead to antibiotic resistance. Rifaximin is a very safe antimicrobial drug that has very little absorption after oral administration. It is a local-acting antibiotic without serious systemic drug adverse effects. VD3 is a member of the Vitamin D family and is thought to affect IBD by playing a crucial role in the immune regulation of cytokines involved in the pathogenesis of IBD. However, how VD3 and rifaximin react together in IBD patients remains unclear. We therefore performed the present study to assess the effect of VD3 on the therapeutic effects of rifaximin for DSS-induced colitis as assessed by multiple aspects of fecal microbiota and colon inflammation.
We used a well-established model of DSS-induced murine colitis and ensured a safe, effective, and clinically relevant dose of rifaximin and VD3. Subsequently, rifaximin and VD3 were intragastric administrated to mice. We demonstrated that VD3 reduced the effect of rifaximin on the Disease Activity Index (DAI) of DSS-induced mice. Moreover, histological colon changes were also observed, in which DSS+rifaximin+VD3 group’s colon injuries were milder than in the DSS group but more serious than in the DSS+rifaximin group. Levels of TNF-α and IL-6 in the DSS+rifaximin+VD3 group were significantly higher than in the rifaximin-treated group. The western blotting results indicated that p-p65/p65 was downregulated in the rifaximin group and upregulated in the DSS+rifaximin+VD3 group. These findings suggest that VD3 attenuates the protective effects of rifaximin for DSS-induced colonic inflammatory cells infiltration. We further analyzed the intestinal flora structure and composition of mice in each group. Our findings suggest that VD3 altered rifaximin’s effects on microbial composition in DSS-induced mice.
The intestinal microbiota has a strong influence on chronic inflammatory diseases such as UC and CD [25,26]. A study of patients with IBD showed a significant correlation between microbiota composition and disease severity, and it is important to recognize the relationship between dysbiosis and IBD [27]. It is a complex and dynamic relationship rather than one of simple cause and effect [28]. Even within the same family, the causes of IBD can differ among family members, given that the gut microbiota composition of the family members differ. This suggests that microbial dysbiosis primarily pertains to the patients’ own disease state rather than genetic or environmental factors [26]. Faecalibacterium prausnitzii and Akkermansia are dramatically decreased in IBD patients [23]. Conversely, compared to healthy individuals, intestinal flora in the Proteobacteria phylum such as Escherichia coli and Enterobacteriaceae are generally more abundant in IBD patients [29,30].
Treatment with rifaximin can attenuated the imbalance of these microbiota [31]. Rifaximin can restore the unbalance of fecal microbiota in DSS-induced colitis mice [32,33]. In addition, rifaximin is a poorly absorbed semi-synthetic rifamycin antibiotic and gut-specific human PXR agonist [34]. It can benefit intestinal health by stimulating beneficial bacteria and inhibiting pathogenic bacteria, which helps maintain a balance of enteric microorganisms [35,36]. It has been previously recognized that probiotics and antibiotics can clinically improve acute DSS-induced colitis because they modulate the commensal microbiota [37]. The intestinal mucosa could be considered as a physical and metabolic barrier. Once damaged, the submucosa is exposed to many luminal antigens such as bacteria or food, which can stimulate the innate immune response to produce a vast pool of cytokines [38].
Furthermore, it has been observed in many studies that intestinal barrier dysfunction is reversed in experimental colitis. In mice treated with rifaximin, the barrier restoration of the colon was enhanced and it had no connection with inflammatory status [39]. Furthermore, in another study using a rat model to investigate the intestinal hyperalgesia, the mucosal inflammation was obviously ameliorated by rifaximin. It also reconstructed the intestinal barrier function and relieved the visceral hyperalgesia originated from chronic stress [40]. Additionally, studies suggest that rifaximin, an PXR agonist, has therapeutic efficacy in treatment of inflammatory intestinal pathologies by enhancing intestinal epithelial metabolic capacity and attenuating NF kappa B-dependent inflammatory responses [41,42]. Our results show that rifaximin inhibits inflammation by suppressing NF kappa B-mediated p-P65 and p65 expression. These findings suggest that PXR signaling plays a key role in intestinal homeostasis. It was also demonstrated that PXR signaling plays a protective role in preventing the occurrence of IBD. Similarly, other studies suggested that the PXR activation caused by rifaximin can suppress the inflammatory response in IECs by instantly inhibiting NF kappa B-dependent cytokine/chemokine production [43]. Additionally, it has been reported that the number of Bacteroidetes in patients with active IBD was much higher than that of patients in IBD remission [44,45]. The rifaximin treatment group had a significantly lower proportion of Bacteroidetes, but VD3 even caused more severe intestinal disorders. The lack of difference in Bacteroidetes between the control group and the DSS group may be because this analysis was based only on the genus level of Bacteroidetes and did not analyze its quantity and composition, which can be further explored in future studies.
VD3 deficiency is very common in patients with IBD [44,45]. In IBD patients, there are several factors contributing to VD3 deficiency, some of which are closely associated with the underlying inflammatory disease, while the others are the same as in people without IBD. For most IBD patients, to a large extent, the increased risk of surgery in CD, and hospitalizations in CD and UC patients, all can lead directly to VD3 deficiency [46]. It appears that VD3 has an important effect on all aspects of the body [47]. In addition, activation of the small intestine epithelium and/or osteoblasts can maintain the homoeostasis of calcium in the patients’ body, which can greatly benefit their health. [48]. Furthermore, VD3 has been verified to play a crucial role in innate immunity and adaptive immunity, [49], and it can also influence the severity of inflammation in IBD [50].
Other questions have also emerged from our study. First, how other gut flora are influenced since the composition of intestinal flora in the DSS+rifaximin+Vitamin D3 group has not been explored. Moreover, further investigation for the metabolism of rifaximin and vitamin D3 is needed to determine the safe interval between administration of the 2 drugs in IBD patients.
Collectively, our study provide evidence that VD3 reverses the therapeutic effects of rifaximin on IBD mice and regulation of intestinal flora. However, its clinical application requires further research. VD3 should not be administered with rifaximin as combination therapy for inflammatory bowel diseases, but further research is needed on combination therapies with favorable cellular profiles.
Conclusions
In conclusion, dietary rifaximin was found to ameliorate tissue damage and disease activity in DSS-induced acute colitis. Although it can be used as an antibiotic and a PXR agonist, it appears to have little effect on the treatment of IBD. VD3 significantly reduces its therapeutic effect by accelerating the metabolism of rifaximin.
Figures
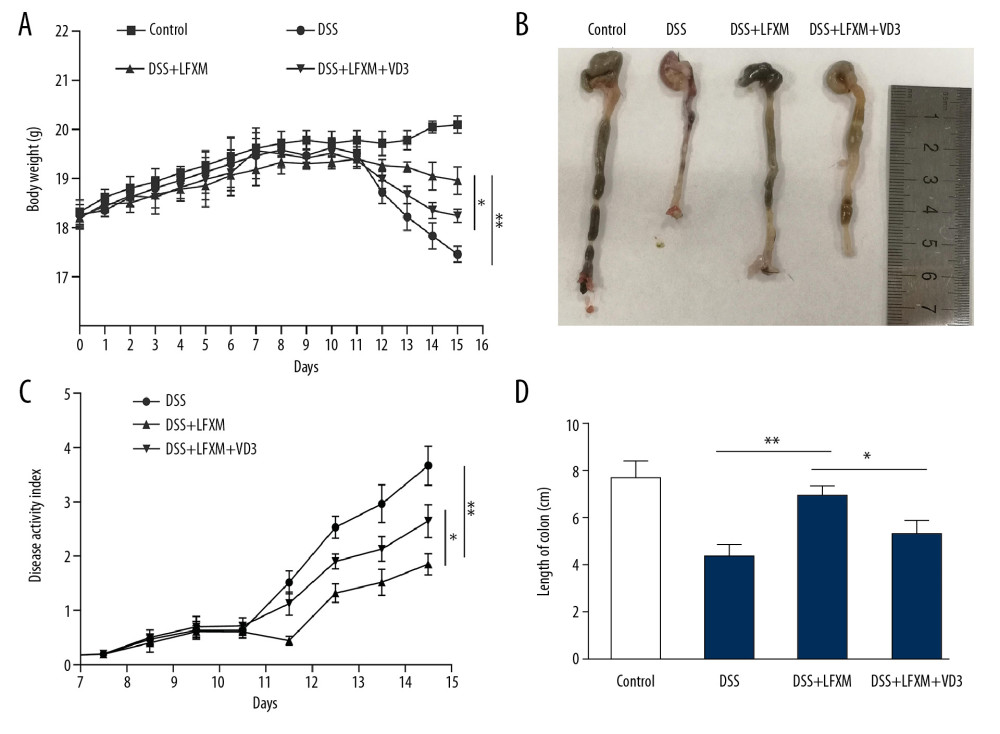 Figure 1. The effects of rifaximin on disease activity index (DAI) in DSS-induced mice was reduced by VD3. (A) Weight changes are shown as the mean change from the starting body weight; (B) DAI was assessed as the average of score of clinical parameters; (C) Colon length. (D) Colon length n=6 mice for each group. Data are presented as the mean±SD. * P<0.05, ** P<0.01, following one-way analysis of variance (ANOVA).
Figure 1. The effects of rifaximin on disease activity index (DAI) in DSS-induced mice was reduced by VD3. (A) Weight changes are shown as the mean change from the starting body weight; (B) DAI was assessed as the average of score of clinical parameters; (C) Colon length. (D) Colon length n=6 mice for each group. Data are presented as the mean±SD. * P<0.05, ** P<0.01, following one-way analysis of variance (ANOVA). 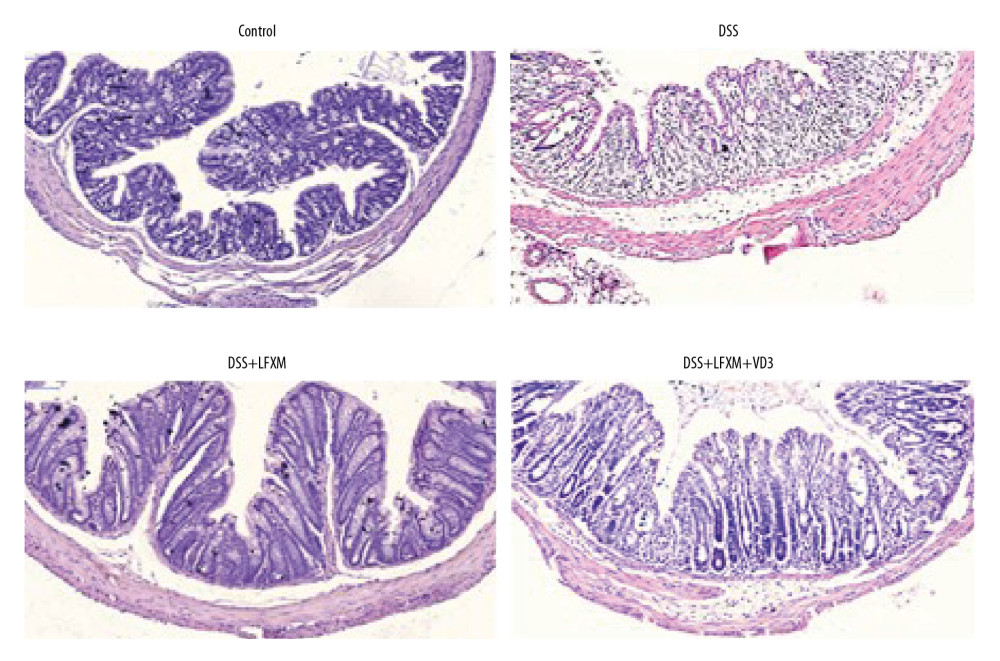 Figure 2. VD3 changed rifaximin’s effects on the histological characterization in DSS-colitis mice. Representative HE staining of the distal colonic tissues in the control group, DSS group, DSS+rifaximin group, and DSS+rifaximin+VD3 group. Five-μm cross-sections were fixed by formalin and embedded in paraffin, then stained with primary antibody. Scale bar: 100 μm.
Figure 2. VD3 changed rifaximin’s effects on the histological characterization in DSS-colitis mice. Representative HE staining of the distal colonic tissues in the control group, DSS group, DSS+rifaximin group, and DSS+rifaximin+VD3 group. Five-μm cross-sections were fixed by formalin and embedded in paraffin, then stained with primary antibody. Scale bar: 100 μm. 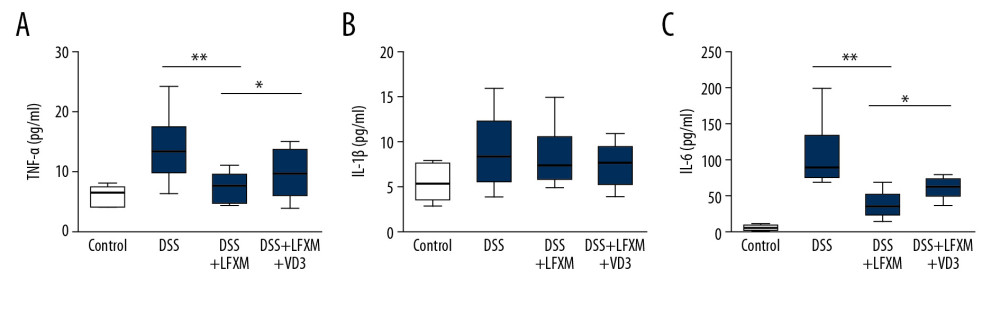 Figure 3. The effects of VD3 on serum cytokines in DSS-colitis mice treated with rifaximin. Serum concentration comparison (A) TNF-α; (B) IL-1β; (C) IL-6. The boxplot demonstrates the values of cytokines from minimum to maximum. n=6 mice. * P<0.05, ** P<0.01.
Figure 3. The effects of VD3 on serum cytokines in DSS-colitis mice treated with rifaximin. Serum concentration comparison (A) TNF-α; (B) IL-1β; (C) IL-6. The boxplot demonstrates the values of cytokines from minimum to maximum. n=6 mice. * P<0.05, ** P<0.01. 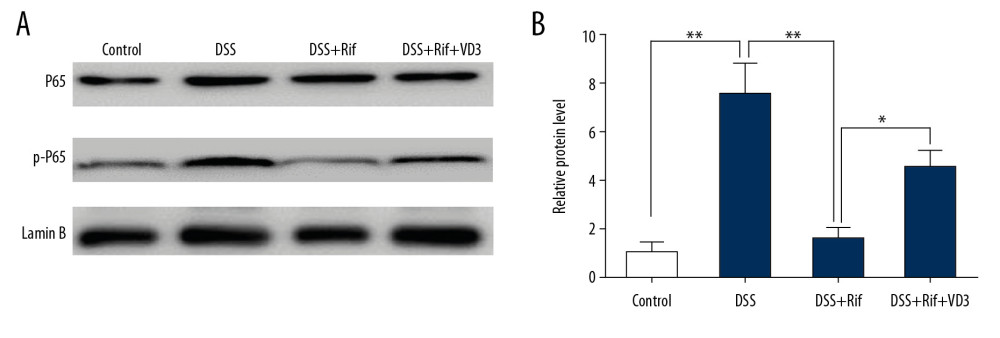 Figure 4. The effects VD3 on inflammatory cells colonic infiltration and NF-κB signaling in DSS-colitis mice treated with rifaximin. (A) Each protein (20 μg) was used for detection of phosphorylated p-P65 and p65. (B) ImageJ software was used to evaluate the integrated band intensity. Data are presented as relative values using corresponding p-P65, p65, and Lamin B as internal reference with the mean±SD. n=5 independent experiments from each mouse. * P<0.05, ** P<0.01.
Figure 4. The effects VD3 on inflammatory cells colonic infiltration and NF-κB signaling in DSS-colitis mice treated with rifaximin. (A) Each protein (20 μg) was used for detection of phosphorylated p-P65 and p65. (B) ImageJ software was used to evaluate the integrated band intensity. Data are presented as relative values using corresponding p-P65, p65, and Lamin B as internal reference with the mean±SD. n=5 independent experiments from each mouse. * P<0.05, ** P<0.01. 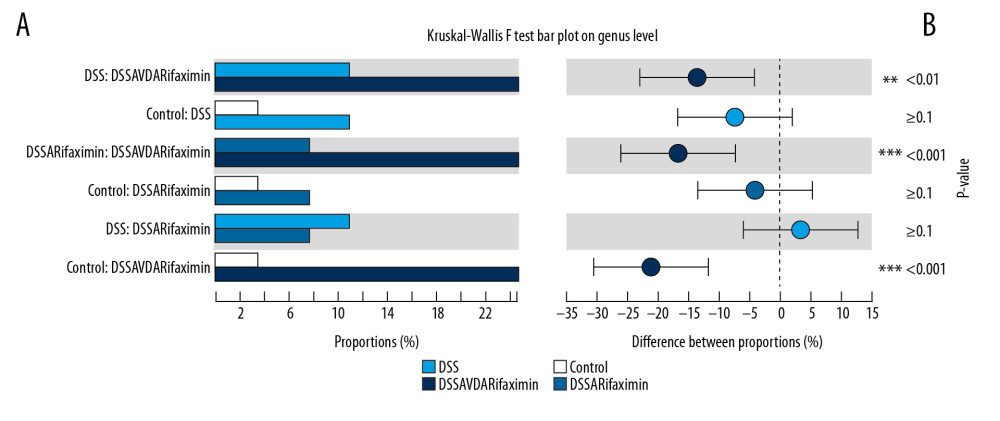 Figure 5. The effects VD3 on Bacteroidetes in DSS-colitis mice treated with rifaximin. (A) Relative abundance of Bacteroidetes phyla; (B) Ratio of Bacteroidetes.
Figure 5. The effects VD3 on Bacteroidetes in DSS-colitis mice treated with rifaximin. (A) Relative abundance of Bacteroidetes phyla; (B) Ratio of Bacteroidetes. References
1. Bernstein CN, Review article: Changes in the epidemiology of inflammatory bowel disease-clues for aetiology: Aliment Pharmacol Ther, 2017; 46(10); 911-19
2. Danese S, Fiocchi C, Ulcerative colitis: N Engl J Med, 2011; 365(18); 1713-25
3. Bannaga AS, Selinger CP, Inflammatory bowel disease and anxiety: Links, risks, and challenges faced: Clin Exp Gastroenterol, 2015; 8; 111-17
4. Runtsch MC, Hu R, Alexander M, MicroRNA-146a constrains multiple parameters of intestinal immunity and increases susceptibility to DSS colitis: Oncotarget, 2015; 6(30); 28556-72
5. Scaldaferri F, Pizzoferrato M, Lopetuso LR, Nutrition and IBD: Malnutrition and/or sarcopenia? A practical guide: Gastroenterol Res Pract, 2017; 2017 8646495
6. Limketkai BN, Wolf A, Parian AM, Nutritional interventions in the patient with inflammatory bowel disease: Gastroenterol Clin North Am, 2018; 47(1); 155-77
7. White JH, Vitamin D deficiency and the pathogenesis of Crohn’s disease: J Steroid Biochem Mol Biol, 2018; 175; 23-28
8. Zhu T, Liu TJ, Shi YY, Zhao Q, Vitamin D/VDR signaling pathway ameliorates 2,4,6-trinitrobenzene sulfonic acid-induced colitis by inhibiting intestinal epithelial apoptosis: Int J Mol Med, 2015; 35(5); 1213-18
9. Zhao H, Zhang H, Wu H, Protective role of 1,25(OH)2 vitamin D3 in the mucosal injury and epithelial barrier disruption in DSS-induced acute colitis in mice: BMC Gastroenterol, 2012; 12; 57
10. Fodor AA, Pimentel M, Chey WD, Rifaximin is associated with modest, transient decreases in multiple taxa in the gut microbiota of patients with diarrhoea-predominant irritable bowel syndrome: Gut Microbes, 2019; 10(1); 22-33
11. Sartor RB, Review article: the potential mechanisms of action of rifaximin in the management of inflammatory bowel diseases: Aliment Pharmacol Ther, 2016; 43(Suppl 1); 27-36
12. Alipour M, Zaidi D, Valcheva R, Mucosal barrier depletion and loss of bacterial diversity are primary abnormalities in paediatric ulcerative colitis: J Crohns Colitis, 2016; 10(4); 462-71
13. Jin Y, Ren X, Li G, Beneficial effects of Rifaximin in post-infectious irritable bowel syndrome mouse model beyond gut microbiota: J Gastroenterol Hepatol, 2018; 33(2); 443-52
14. Kang DJ, Kakiyama G, Betrapally NS, Rifaximin exerts beneficial effects independent of its ability to alter microbiota composition: Clin Transl Gastroenterol, 2016; 7(8); e187
15. Prantera C, Lochs H, Campieri M, Antibiotic treatment of Crohn’s disease: Results of a multicentre, double blind, randomized, placebo-controlled trial with rifaximin: Aliment Pharmacol Ther, 2006; 23(8); 1117-25
16. Deb S, Chin MY, Adomat H, Guns ES: J Steroid Biochem Mol Biol, 2014; 144 Pt A; 50-58
17. Zhu H, Wu R, Gu Z, Vitamin D3 is transformed into 1,25(OH)2D3 by triggering CYP3A11(CYP3A4) activity and hydrolyzing midazolam: Med Sci Monit, 2019; 25; 9159-66
18. Trapnell CB, Connolly M, Pentikis H, Absence of effect of oral rifaximin on the pharmacokinetics of ethinyl estradiol/norgestimate in healthy females: Ann Pharmacother, 2007; 41(2); 222-28
19. Deb S, Pandey M, Adomat H, Guns ES, Cytochrome P450 3A-mediated microsomal biotransformation of 1α,25-dihydroxyvitamin D3 in mouse and human liver: Drug-related induction and inhibition of catabolism: Drug Metab Dispos, 2012; 40(5); 907-18
20. Thummel KE, Wilkinson GR: Annu Rev Pharmacol Toxicol, 1998; 38; 389-430
21. Zhou SF, Drugs behave as substrates, inhibitors and inducers of human cytochrome P450 3A4: Curr Drug Metab, 2008; 9(4); 310-22
22. Zhang Z, Wu X, Cao S, Caffeic acid ameliorates colitis in association with increased Akkermansia population in the gut microbiota of mice: Oncotarget, 2016; 7(22); 31790-99
23. Zhang Z, Wu X, Cao S, Chlorogenic acid ameliorates experimental colitis by promoting growth of akkermansia in mice: Nutrients, 2017; 9(7); 677
24. Wang Q, Garrity GM, Tiedje JM, Cole JR, Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy: Appl Environ Microbiol, 2007; 73(16); 5261-67
25. Ohkusa T, Yoshida T, Sato N, Commensal bacteria can enter colonic epithelial cells and induce proinflammatory cytokine secretion: A possible pathogenic mechanism of ulcerative colitis: J Med Microbiol, 2009; 58(Pt 5); 535-45
26. Torres J, Mehandru S, Colombel JF, Peyrin-Biroulet L, Crohn’s disease: Lancet, 2017; 389(10080); 1741-55
27. Kolho KL, Korpela K, Jaakkola T, Fecal microbiota in pediatric inflammatory bowel disease and its relation to inflammation: Am J Gastroenterol, 2015; 110(6); 921-30
28. Ni J, Wu GD, Albenberg L, Tomov VT, Gut microbiota and IBD: Causation or correlation?: Nat Rev Gastroenterol Hepatol, 2017; 14(10); 573-84
29. Lewis JD, Chen EZ, Baldassano RN, Inflammation, antibiotics, and diet as environmental stressors of the gut microbiome in pediatric Crohn’s disease [published correction appears in Cell Host Microbe, 2017; 22(2): 247]: Cell Host Microbe, 2015; 18(4); 489-500
30. Sartor RB, Therapeutic correction of bacterial dysbiosis discovered by molecular techniques: Proc Natl Acad Sci USA, 2008; 105(43); 16413-14
31. Weber D, Oefner PJ, Dettmer K, Rifaximin preserves intestinal microbiota balance in patients undergoing allogeneic stem cell transplantation: Bone Marrow Transplant, 2016; 51(8); 1087-92
32. Machiels K, Joossens M, Sabino J: Gut, 2014; 63(8); 1275-83
33. Yang L, Liu B, Zheng J, Rifaximin alters intestinal microbiota and prevents progression of ankylosing spondylitis in mice: Front Cell Infect Microbiol, 2019; 9; 44
34. Ma X, Shah YM, Guo GL, Rifaximin is a gut-specific human pregnane X receptor activator: J Pharmacol Exp Ther, 2007; 322(1); 391-98
35. Khan KJ, Ullman TA, Ford AC, Antibiotic therapy in inflammatory bowel disease: A systematic review and meta-analysis [published correction appears in Am J Gastroenterol, 2011; 106(5): 1014]: Am J Gastroenterol, 2011; 106(4); 661-73
36. Dueñas M, Muñoz-González I, Cueva C, A survey of modulation of gut microbiota by dietary polyphenols: Biomed Res Int, 2015; 2015 850902
37. Dai C, Zheng CQ, Meng FJ, VSL#3 probiotics exerts the anti-inflammatory activity via PI3k/Akt and NF-κB pathway in rat model of DSS-induced colitis: Mol Cell Biochem, 2013; 374(1–2); 1-11
38. Han F, Zhang H, Xia X, Porcine β-defensin 2 attenuates inflammation and mucosal lesions in dextran sodium sulfate-induced colitis: J Immunol, 2015; 194(4); 1882-93
39. Terc J, Hansen A, Alston L, Hirota SA, Pregnane X receptor agonists enhance intestinal epithelial wound healing and repair of the intestinal barrier following the induction of experimental colitis: Eur J Pharm Sci, 2014; 55; 12-19
40. Xu D, Gao J, Gillilland M, Rifaximin alters intestinal bacteria and prevents stress-induced gut inflammation and visceral hyperalgesia in rats: Gastroenterology, 2014; 146(2); 484-96.e4
41. Mencarelli A, Migliorati M, Barbanti M, Pregnane-X-receptor mediates the anti-inflammatory activities of rifaximin on detoxification pathways in intestinal epithelial cells: Biochem Pharmacol, 2010; 80(11); 1700-7
42. Mencarelli A, Renga B, Palladino G, Inhibition of NF-κB by a PXR-dependent pathway mediates counter-regulatory activities of rifaximin on innate immunity in intestinal epithelial cells: Eur J Pharmacol, 2011; 668(1–2); 317-24
43. Cheng J, Shah YM, Ma X, Therapeutic role of rifaximin in inflammatory bowel disease: clinical implication of human pregnane X receptor activation: J Pharmacol Exp Ther, 2010; 335(1); 32-41
44. Levin AD, Wadhera V, Leach ST, Vitamin D deficiency in children with inflammatory bowel disease: Dig Dis Sci, 2011; 56(3); 830-36
45. Ulitsky A, Ananthakrishnan AN, Naik A, Vitamin D deficiency in patients with inflammatory bowel disease: Association with disease activity and quality of life: J Parenter Enteral Nutr, 2011; 35(3); 308-16
46. Ananthakrishnan AN, Cagan A, Gainer VS, Normalization of plasma 25-hydroxy vitamin D is associated with reduced risk of surgery in Crohn’s disease: Inflamm Bowel Dis, 2013; 19(9); 1921-27
47. Mouli VP, Ananthakrishnan AN, Review article: vitamin D and inflammatory bowel diseases: Aliment Pharmacol Ther, 2014; 39(2); 125-36
48. Yasuda YH, Shima N, Nakagawa N, Osteoclast differentiation factor is a ligand for osteoprotegerin/osteoclastogenesis-inhibitory factor and is identical to TRANCE/RANKL: Proc Natl Acad Sci USA, 1998; 95(7); 3597-602
49. Cantorna MT, Mahon BD, D-hormone and the immune system: J Rheumatol Suppl, 2005; 76; 11-20
50. Cantorna MT, Munsick C, Bemiss C, Mahon BD, 1,25-Dihydroxycholecalciferol prevents and ameliorates symptoms of experimental murine inflammatory bowel disease: J Nutr, 2000; 130; 2648-52
Figures
 Figure 1. The effects of rifaximin on disease activity index (DAI) in DSS-induced mice was reduced by VD3. (A) Weight changes are shown as the mean change from the starting body weight; (B) DAI was assessed as the average of score of clinical parameters; (C) Colon length. (D) Colon length n=6 mice for each group. Data are presented as the mean±SD. * P<0.05, ** P<0.01, following one-way analysis of variance (ANOVA).
Figure 1. The effects of rifaximin on disease activity index (DAI) in DSS-induced mice was reduced by VD3. (A) Weight changes are shown as the mean change from the starting body weight; (B) DAI was assessed as the average of score of clinical parameters; (C) Colon length. (D) Colon length n=6 mice for each group. Data are presented as the mean±SD. * P<0.05, ** P<0.01, following one-way analysis of variance (ANOVA). Figure 2. VD3 changed rifaximin’s effects on the histological characterization in DSS-colitis mice. Representative HE staining of the distal colonic tissues in the control group, DSS group, DSS+rifaximin group, and DSS+rifaximin+VD3 group. Five-μm cross-sections were fixed by formalin and embedded in paraffin, then stained with primary antibody. Scale bar: 100 μm.
Figure 2. VD3 changed rifaximin’s effects on the histological characterization in DSS-colitis mice. Representative HE staining of the distal colonic tissues in the control group, DSS group, DSS+rifaximin group, and DSS+rifaximin+VD3 group. Five-μm cross-sections were fixed by formalin and embedded in paraffin, then stained with primary antibody. Scale bar: 100 μm. Figure 3. The effects of VD3 on serum cytokines in DSS-colitis mice treated with rifaximin. Serum concentration comparison (A) TNF-α; (B) IL-1β; (C) IL-6. The boxplot demonstrates the values of cytokines from minimum to maximum. n=6 mice. * P<0.05, ** P<0.01.
Figure 3. The effects of VD3 on serum cytokines in DSS-colitis mice treated with rifaximin. Serum concentration comparison (A) TNF-α; (B) IL-1β; (C) IL-6. The boxplot demonstrates the values of cytokines from minimum to maximum. n=6 mice. * P<0.05, ** P<0.01. Figure 4. The effects VD3 on inflammatory cells colonic infiltration and NF-κB signaling in DSS-colitis mice treated with rifaximin. (A) Each protein (20 μg) was used for detection of phosphorylated p-P65 and p65. (B) ImageJ software was used to evaluate the integrated band intensity. Data are presented as relative values using corresponding p-P65, p65, and Lamin B as internal reference with the mean±SD. n=5 independent experiments from each mouse. * P<0.05, ** P<0.01.
Figure 4. The effects VD3 on inflammatory cells colonic infiltration and NF-κB signaling in DSS-colitis mice treated with rifaximin. (A) Each protein (20 μg) was used for detection of phosphorylated p-P65 and p65. (B) ImageJ software was used to evaluate the integrated band intensity. Data are presented as relative values using corresponding p-P65, p65, and Lamin B as internal reference with the mean±SD. n=5 independent experiments from each mouse. * P<0.05, ** P<0.01. Figure 5. The effects VD3 on Bacteroidetes in DSS-colitis mice treated with rifaximin. (A) Relative abundance of Bacteroidetes phyla; (B) Ratio of Bacteroidetes.
Figure 5. The effects VD3 on Bacteroidetes in DSS-colitis mice treated with rifaximin. (A) Relative abundance of Bacteroidetes phyla; (B) Ratio of Bacteroidetes. Tables
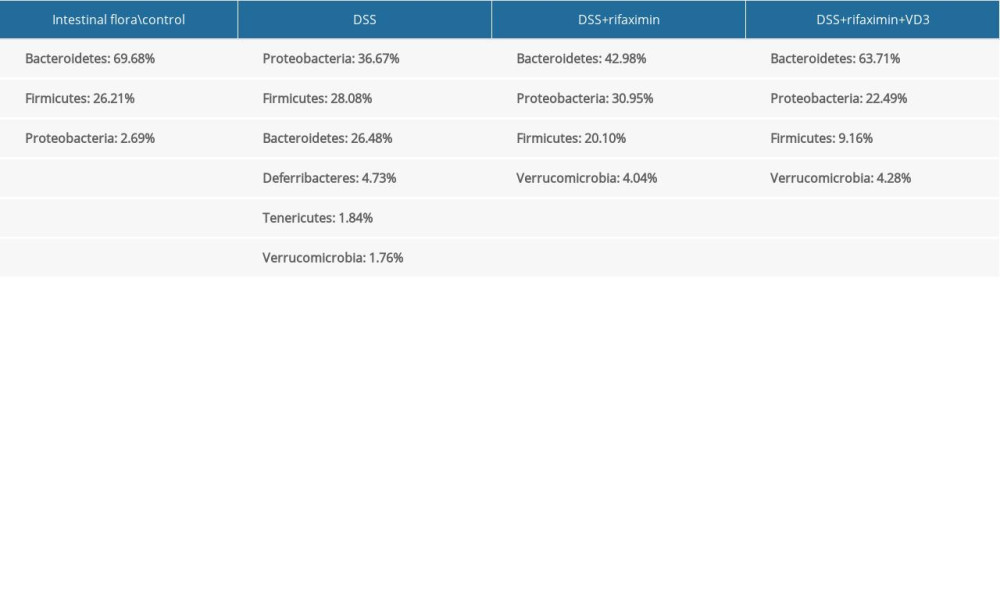 Table 1. Community analysis pie-plot on phylum level. DSS: Dextran sulfate sodium; VD3: Vitamin D3.
Table 1. Community analysis pie-plot on phylum level. DSS: Dextran sulfate sodium; VD3: Vitamin D3.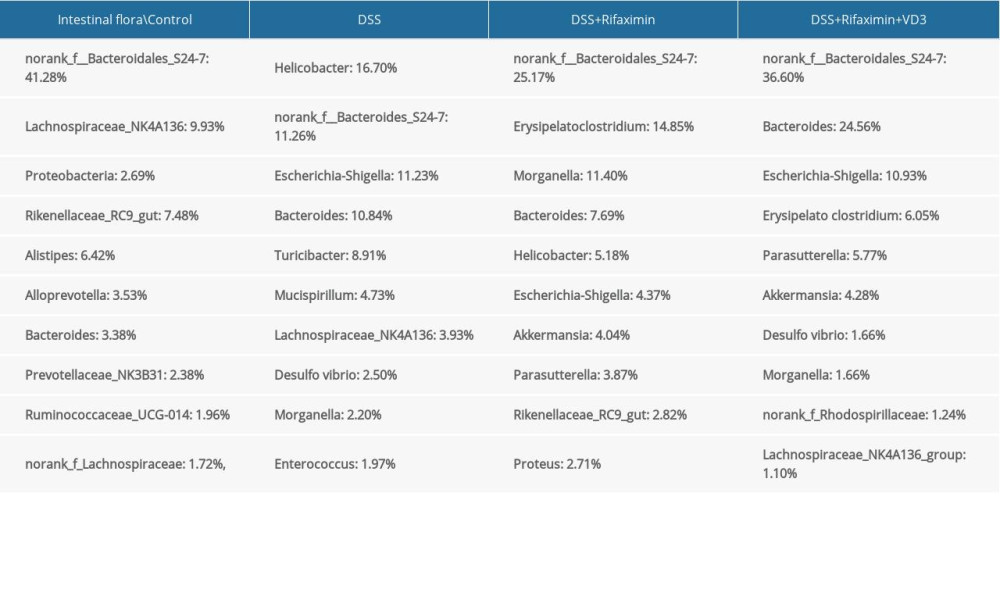 Table 2. Community analysis pie-plot on genus level. DSS: Dextran sulfate sodium; VD3: Vitamin D3.
Table 2. Community analysis pie-plot on genus level. DSS: Dextran sulfate sodium; VD3: Vitamin D3. Table 1. Community analysis pie-plot on phylum level. DSS: Dextran sulfate sodium; VD3: Vitamin D3.
Table 1. Community analysis pie-plot on phylum level. DSS: Dextran sulfate sodium; VD3: Vitamin D3. Table 2. Community analysis pie-plot on genus level. DSS: Dextran sulfate sodium; VD3: Vitamin D3.
Table 2. Community analysis pie-plot on genus level. DSS: Dextran sulfate sodium; VD3: Vitamin D3. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








