17 September 2020: Database Analysis
Effects of Pulsatile Frequency of Left Ventricular Assist Device (LVAD) on Coronary Perfusion: A Numerical Simulation Study
Yuanfei Zhu1ABCE, Ming Yang1AEG*, Yan Zhang2BCF, Fan Meng1DEF, Tianyue Yang1AC, Zhiwei Fang1BFDOI: 10.12659/MSM.925367
Med Sci Monit 2020; 26:e925367
Abstract
BACKGROUND: Left ventricular assist devices (LVADs) with counter-pulsation mode have been widely used to support left ventricular function and improve coronary circulation. However, the frequency characteristics of the coronary system have not been considered. The aim of this study was to investigate the effects of pulsatile frequency of LVADs on coronary perfusion.
MATERIAL AND METHODS: First, a lumped parameter (LP) model incorporating coronary circulation, systemic circulation, left heart, and LVAD was established to simulate the cardiovascular system. Then, the frequency characteristics of the coronary system were analyzed and the calculation results showed that the pulsatile frequency of the LVAD has a substantial effect on coronary blood flow. To verify the accuracy of the theoretical analysis, the hemodynamic effects of the LVAD on the coronary artery were compared under 4 support modes: co-pulsation mode, and counter-pulsation modes in synchronization ratios of 1: 1, 2: 1, and 3: 1.
RESULTS: We found that the coronary flow increased by 5% when the working mode changed from co-pulsation to counter-pulsation in a synchronization ratio of 1: 1, and by an additional 6% when the working mode changed from counter-pulsation in a synchronization ratio of 1: 1 to counter-pulsation in a synchronization ratio of 3: 1.
CONCLUSIONS: This work provides a useful method to increase coronary perfusion and may be beneficial for improving myocardial function in patients with end-stage heart failure, especially those with ischemic cardiomyopathy (ICM).
Keywords: Coronary Vessels, Heart-Assist Devices, Myocardial Reperfusion, Pulsatile Flow, Computer Simulation, Models, Cardiovascular, Ventricular Function, Left
Background
The heart failure (HF) caused by ischemic cardiomyopathy (ICM) is common and is mainly related to insufficient coronary perfusion [1–4]. In recent years, continuous-flow left ventricular assist devices (CFLVADs) have been widely used in destination therapy for end-stage heart failure, with high reliability and durability [5–7]. However, several studies have shown decreased coronary flow under CFLVADs support [8,9]. The main reasons for decreased coronary perfusion are the decrease of left ventricular work and myocardial oxygen demand [9,10]. Moreover, under constant rotational speed, the mechanical characteristics of CFLVADs are also responsible for diminished coronary flow [11].
To increase coronary flow, Ando et al. [11] proposed a counter-pulsation mode for CFLVADs to improve myocardial perfusion. Lim et al. [12] also reported that the LVAD with counter-pulsation mode achieved better physiological coronary blood perfusion. Naito et al. [13] presented a rotational speed modulation system for CFLVADs to decrease the left ventricular (LV) load and increase coronary flow. However, under long-term support, CFLVADs may impair arterial contractility and myocardial microvascular structure, thus limiting coronary flow and causing myocardial ischemia [14,15]. Therefore, the flow profiles and pulsatility of LVADs still need to be optimized to improve destination therapy.
The cardiovascular system is a complicated impedance network [16,17]. The frequency characteristics of the cardiovascular system are closely related to blood perfusion. Wang et al. [18] reported that the arterial system had an optimal perfusion at its natural frequency. Gao et al. [19] showed that the heart beat generally matched the frequency characteristics of the arterial system, with low power consumption. Zhuang et al. [20] reported that when the pump speed was consistent with the resonance frequency of the arterial system, the perfusion of the arterial system was effectively improved. Rebholz et al. [21] analyzed the interaction between the high-frequency operating LVAD and the cardiovascular system to reduce the pump body size. However, in these studies, the coronary system was not included and the frequency characteristics of the coronary system were not investigated. Coronary flow is an important factor in determining cardiac function. However, the effects of pulsatile frequency of the LVAD on coronary perfusion have not been reported.
In this study we investigated the effects of pulsatile frequency of the LVAD on coronary perfusion. Firstly, we established a lumped parameter (LP) model of the cardiovascular system and analyzed the effects of coronary system frequency characteristics on blood perfusion. Then, the effectiveness of the established cardiovascular model was verified by comparing the present results with those of a previous study. Finally, the hemodynamic effects of the LVAD on the coronary artery were compared under 4 support modes: co-pulsation mode, and counter-pulsation modes in synchronization ratios of 1: 1, 2: 1, and 3: 1.
Material and Methods
MODEL OF CARDIOVASCULAR SYSTEM:
The cardiovascular system is modeled using a LP electric network, which includes 3 parts: left heart, systemic circulation, and coronary circulation. To analyze the frequency characteristics of the coronary system, the coronary circulation model was constructed from 3 main components: the coronary artery, the arteriolar resistance, and the myocardial vessels [24,25], as shown in Figure 1B. The coronary flow originates from the aorta root and flows into the vein [26]. The arteriolar resistance RAR is mainly responsible for hyperemia and baseline conditions. Because ventricular pressure acts upon intramyocardial (IMC) vessels, the IMC pressure is represented by an external pressure element (PIMC in Figure 1). The variable intramyocardial resistance RMA depends on PIMC and is modeled as RMA=0.9PIMC, PIMC=PLV [25], where PLV represents left ventricular pressure.
The left heart was modeled as a chamber pump with 2 valves that control the direction of ventricular blood flow. The ventricle was simulated with a commonly used time-varying elastance model [23,27], and the atrial compliance was characterized by a capacitor of electric analog [28]. The function CLV (t) of ventricle compliance was adapted from a previous model [29]. The function CLV (t)=1/E(t) can be calculated as:
where
The systemic circulation was modeled by the aorta and the typical four-element Windkessel model (W4S) [23], as shown in Figure 1C. The aorta compliance is represented by CA. In the W4S model, the model parameters are RA, LA, CS, and RS, which represent the aortic resistance, the aortic blood inertia, total arterial compliance, and total peripheral resistance, respectively. The parameters of the cardiovascular model were adapted from previous studies [22–29], as summarized in Table 1.
MODEL OF LVAD:
The LVAD model was established based on previous research [12,30]. The prototype of the LVAD is a pulsatile pneumatic ventricular assist device (the Hybrid Ventricular Assist Device; Korea Artificial Organ Center, Seoul, Korea) [30]. As shown in Figure 1A, the LVAD model consists of a flow generator (QLVAD), a blood cavity (CLVAD), and 2 valves (Di and Do). The flow profiles of the proposed pump model are simulated directly as sinusoidal waves for simplification, as the sinusoidal-wave-flow profiles parameterized with the stroke volume, the pulsatile frequency, and the phase shift [21]. The phase shift ϕ was introduced to represent the time delay between the start of the native cardiac cycle and the middle of the pump systole. The starting point of the cardiac cycle was defined as the middle of native heart systole. As shown in Figure 2, the phase shift ϕ was simulated as 50%, 25%, and 16.6% to characterize the counter-pulsation mode with the synchronization ratio of 1: 1, 2: 1, and 3: 1, respectively. To ensure comparable results for various operation modes, the stroke volume of the LVAD was further manually adjusted to keep mean aortic pressure and LVAD flow constant [12].
FREQUENCY CHARACTERISTICS OF THE CORONARY SYSTEM:
The main factors affecting the coronary blood flow are blood pressure within the aortic root and vascular resistance. Generally, the coronary blood flow is about 250 ml/min and predominantly occurs during diastole [31]. Hence, the frequency characteristics of the coronary system were analyzed during diastole in our model. The diastolic ventricle pressure is very small and is close to zero. Therefore, the IMC pressure (PIMC) and intramyocardial resistance (RMA) can be ignored in the diastole. The relationship between the input impedance Z(jω) and frequency f can be expressed as:
where
The magnitude of input impedance can be obtained by the above equations, as shown in Figure 3, showing that the magnitude of input impedance initially decreases and then increases with increasing frequency. The magnitude of impedance reaches the smallest value near 20 Hz, which is the smallest impedance frequency point. Further, the coronary impedance seriously restricts the coronary flow. Hence, the pulsatile frequency of LVAD can have a substantial influence on the coronary flow.
However, since the high pulsatile frequency requires more reliable and accurate control of the LVAD, the smallest impedance frequency is difficult to realize in clinical applications. Therefore, our study mainly investigated the effects of increased pulsatile frequency on coronary flow. In addition, when the pulsatile frequency increases by 2 Hz, the magnitude of coronary impedance decreases by about 4%. In other words, the coronary blood flow can increase by about 4% (10 ml/min) when the pulsatile frequency is 3 Hz.
To further evaluate the effectiveness of coronary perfusion, the total input work (TIW) [20] and energy equivalent pressure (EEP) [32,33] were introduced. The TIW is an important index of hemodynamic power and is calculated as:
where
where
Results
MODEL VERIFICATION:
The simulated pressure and flow waveforms of the cardiovascular system are shown in Figure 4 and the key hemodynamic results for simulated normal and severe HF conditions are summarized in Table 2. In normal conditions, the peak systolic pressure of LV was about 125 mmHg and the cardiac output was approximately 5.4 L/min. However, the peak systolic pressure of LV declined to 83 mmHg and the cardiac output was about 3.5 L/min in the HF condition. We also found that the coronary flow of the HF condition had a 33% decrease compared with the normal condition, which was approximately consistent with previous reports [34].
HEMODYNAMIC EFFECTS UNDER VARIOUS PULSATILE FREQUENCIES OF LVAD:
Four different operation modes of LVAD were simulated: co-pulsation mode, counter-pulsation mode, and counter-pulsation modes in synchronization ratios of 2: 1 and 3: 1. The hemodynamic waveforms of the various operation modes are shown in Figure 5. The cardiac cycle starting point was set to the start of systole. To compare the effects of pulsatile frequency of LVAD on coronary perfusion, the LVAD flow was set as 5.4 L/min and the mean aortic pressure was maintained at 100 mmHg in all operation modes of LVAD. The arterial pressure exceeded the peak LV pressure, as shown in Figure 5D, 5G, and 5J, respectively. Therefore, the aortic valve was not open and the native cardiac output did not generate any flow in the counter-pulsation modes. We also found that the peak LV pressure gradually decreased with increasing pulsatile frequency, which suggests that elevated pulsatile frequency can result in lower LV pressure.
The changes in the aortic flow for various LVAD operation modes are shown in Figure 5B, 5E, 5H, and 5K, respectively. With the increase of pulsatile frequency, several similar peaks were formed in the aortic flow. Similarly, the number of coronary flow wave peaks was consistent with frequency, as shown in Figure 5C, 5F, 5I, and 5L, respectively. The simulation results are summarized in Table 3, showing that the pump stroke volume in a synchronization ratio of 3: 1 declined to 35.8% of that in the synchronization ratio of 1: 1. Therefore, this result suggests that the pump volume may be reduced with increasing pulsatile frequency.
Coronary perfusion was effectively improved with increasing pulsatile frequency because the magnitude of coronary impedance decreases with increasing pulsatile frequency, as shown in Table 3 and Figure 6. Under the same mean aortic pressure, the mean coronary flow was enhanced. The EEP of the coronary artery was almost constant under LVAD support, which can be attributed to unchanged mean aortic pressure. The TIW of the coronary artery under the HF condition was 0.0228 J/s, which is only 44% of that under the normal condition. However, under LVAD support, the maximum TIW of coronary artery increased to 0.0583 J/s. Moreover, the TIW of the coronary artery had the same trend as the coronary flow under various pulsatile frequencies. These results indicate that increasing LVAD pulsatile frequency can effectively improve coronary perfusion by reducing the coronary impedance. As shown in Figure 7, the coronary flow increased with increasing mean aortic pressure in the counter-pulsation mode with a synchronization ratio of 3: 1. Therefore, normal aortic pressure is also crucial for coronary perfusion.
Discussion
STUDY LIMITATIONS:
The cardiovascular system model used in this study has some limitations, similar to other zero-dimensional LP models. Some researchers have shown that pulsatile flow is directly related to endothelial production of nitric oxide, which can increase bradykinin-dependent vascular relaxation and decrease vascular oxidative stress [41,42]. Therefore, the corresponding nervous and hormonal regulation system should be considered in clinical applications. In addition, some peripheral organs, such as pulmonary and kidney tissue, should also be considered. The aortic valve is assumed to be closed during the cardiac cycle, which may result in aortic insufficiency [43]. Therefore, the LVAD operating mode still needs to be improved to protect the aortic valve in clinical applications. Moreover, further in vivo experiments are needed to evaluate the effects of LVAD pulsatile frequency on coronary perfusion.
Conclusions
We investigated the effects of pulsatile frequency of LVAD on coronary perfusion. First, we simulated the cardiovascular system using the LP model, which incorporates left heart, LVAD, coronary circulation, and systemic circulation. Then, the effects of pulsatile frequency on the coronary impedance were analyzed. The present theoretical analysis suggests that increased pulsatile frequency can effectively decrease the coronary impedance when the pulsatile frequency is not higher than the resonance frequency. Therefore, the 4 different operation modes of LVAD (co-pulsation and counter-pulsation modes in synchronization ratio of 1: 1, 2: 1, and 3: 1) were simulated to compare the hemodynamic effects. The simulation results show that the counter-pulsation mode with high pulsatile frequency not only effectively improves the coronary flow, but also helps unload the LV. Although the pulsatile LVAD was used in the study, changing the pulsatile frequency of CFLVADs by the speed control algorithms to improve myocardial function in patients with ICM appears to be promising.
Figures
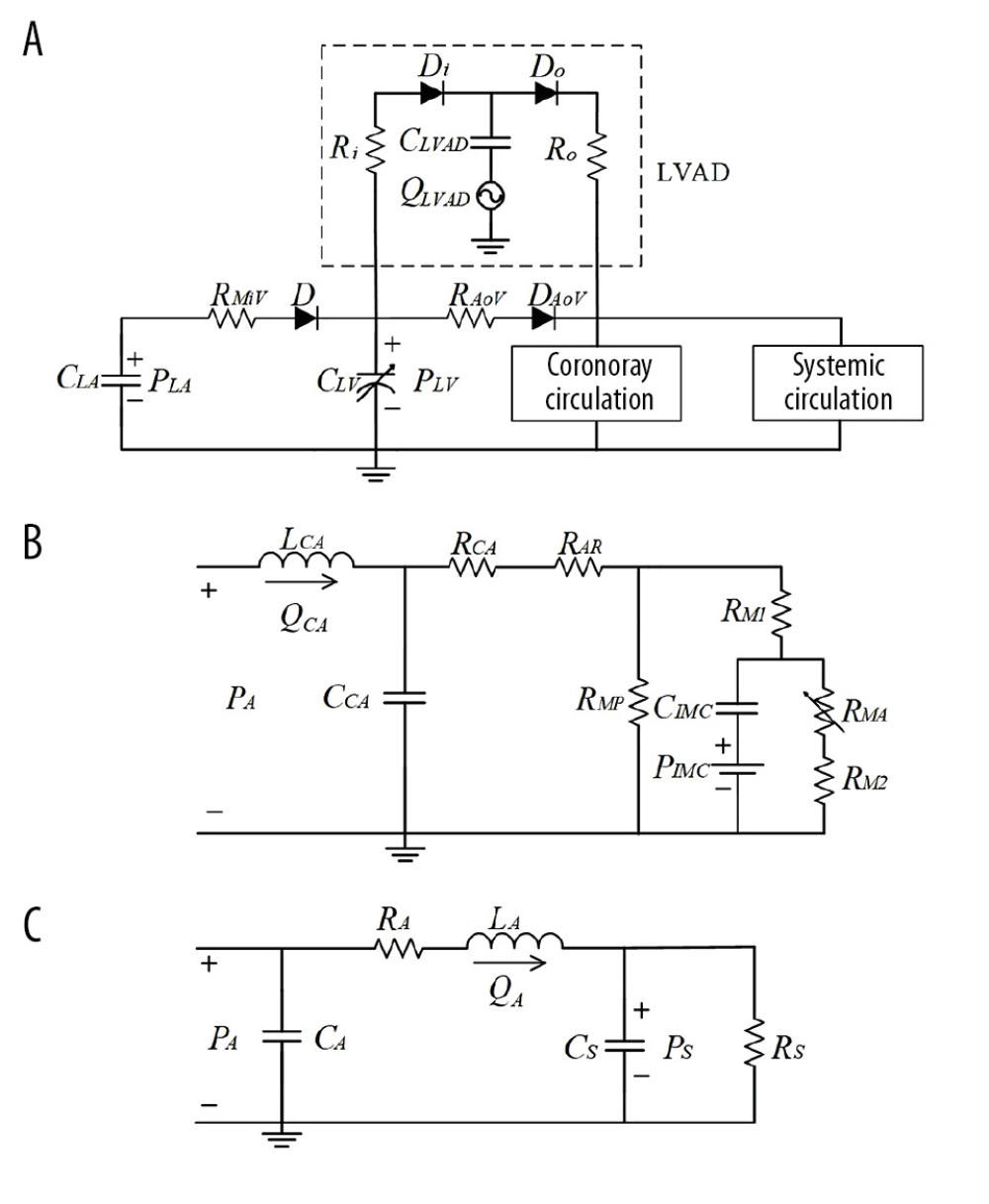 Figure 1. Circuit diagram of the cardiovascular system model. (A) Cardiovascular system and left ventricular assist device (LVAD), (B) Coronary circulation, (C) Systemic circulation. R – vascular resistance; C – vascular compliance; L – blood inertia; D – valve; P – pressure; Q – blood flow; LV – left ventricle; LA – left atrium; CA – coronary artery; A – aorta; S – total peripheral artery; MiV – mitral valve; AoV – aortic valve; AR – arteriolar resistance; IMC – intramyocardial vessels; RM1, RMA, RM2 and RMP – the myocardial resistances.
Figure 1. Circuit diagram of the cardiovascular system model. (A) Cardiovascular system and left ventricular assist device (LVAD), (B) Coronary circulation, (C) Systemic circulation. R – vascular resistance; C – vascular compliance; L – blood inertia; D – valve; P – pressure; Q – blood flow; LV – left ventricle; LA – left atrium; CA – coronary artery; A – aorta; S – total peripheral artery; MiV – mitral valve; AoV – aortic valve; AR – arteriolar resistance; IMC – intramyocardial vessels; RM1, RMA, RM2 and RMP – the myocardial resistances. 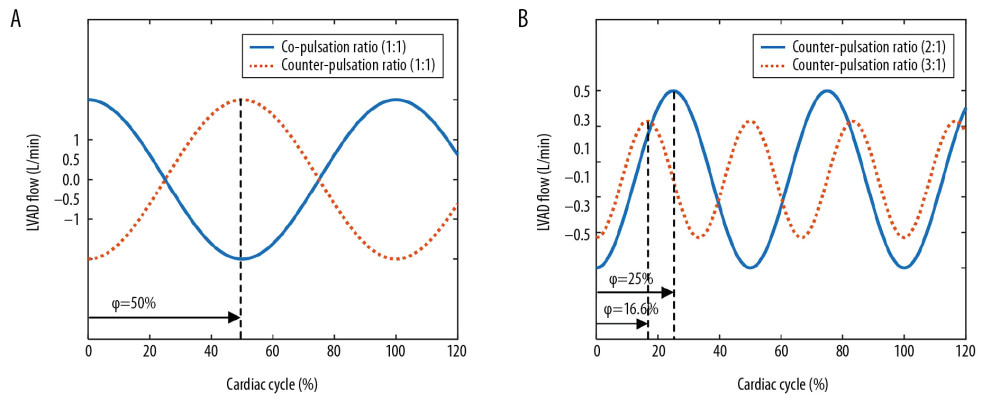 Figure 2. Flow waveforms of the LVAD in the co-pulsation mode and the counter-pulsation modes. The negative flows represent the pump’s diastole and positive flows represent the pump’s systole.
Figure 2. Flow waveforms of the LVAD in the co-pulsation mode and the counter-pulsation modes. The negative flows represent the pump’s diastole and positive flows represent the pump’s systole. 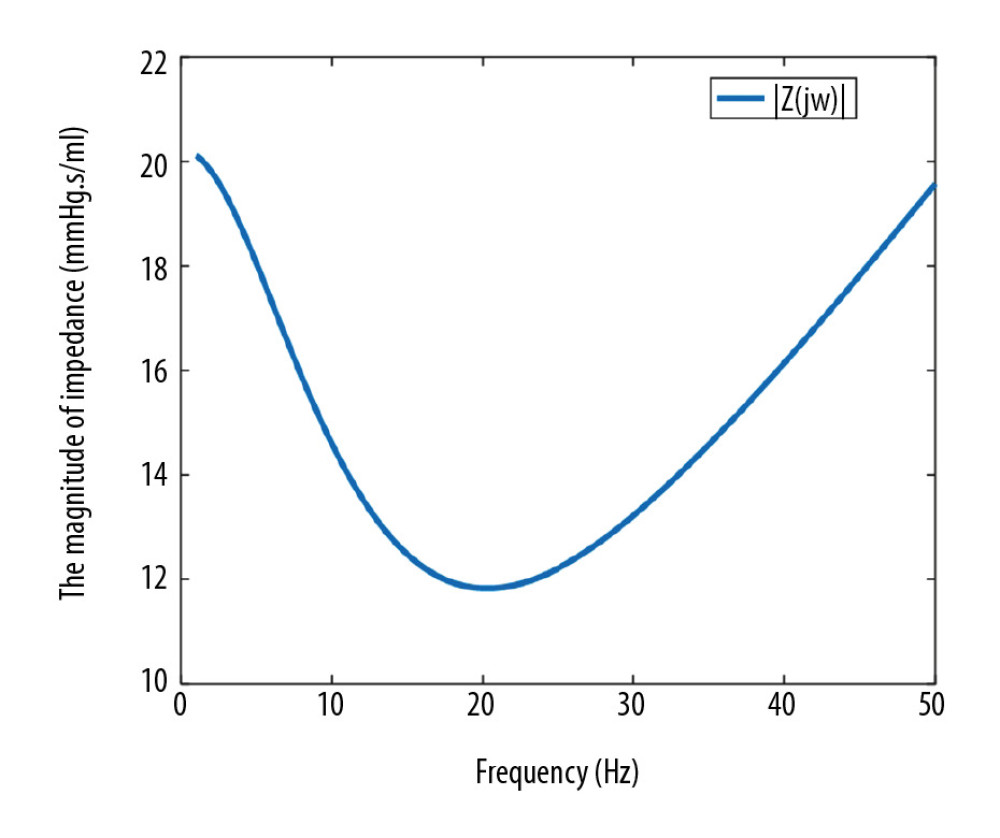 Figure 3. Frequency characteristics of the coronary system.
Figure 3. Frequency characteristics of the coronary system. 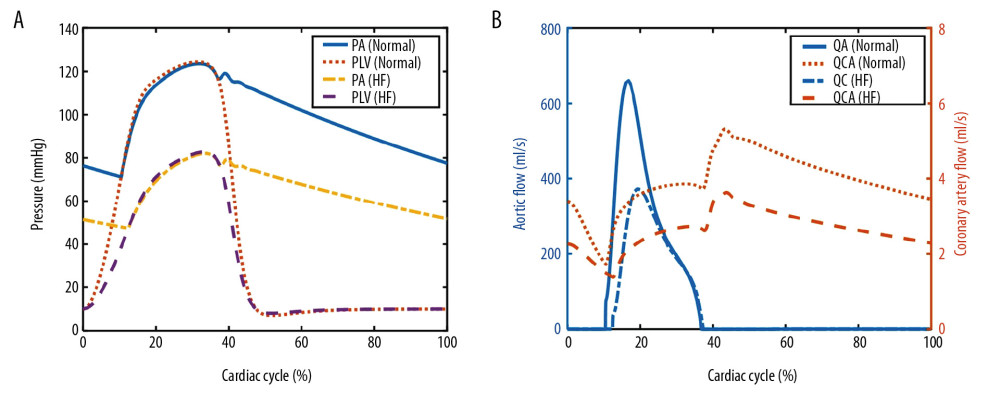 Figure 4. Simulated pressure waveforms (A) and flow waveforms (B) in the cardiovascular system. PA – aortic pressure; PLV – left ventricular pressure; QA – aortic flow; QCA – coronary artery flow.
Figure 4. Simulated pressure waveforms (A) and flow waveforms (B) in the cardiovascular system. PA – aortic pressure; PLV – left ventricular pressure; QA – aortic flow; QCA – coronary artery flow. 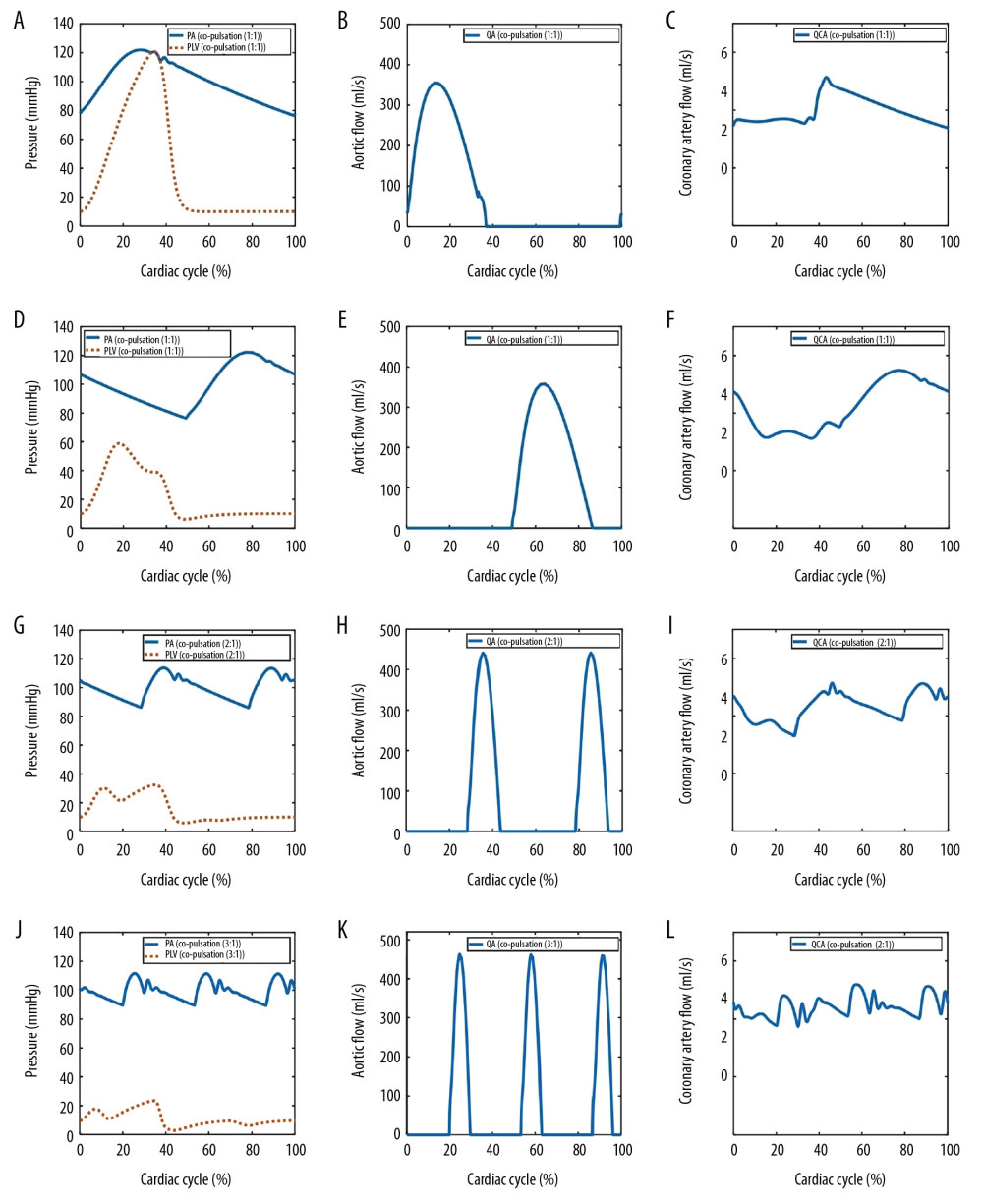 Figure 5. Simulated hemodynamic waveforms in the cardiovascular model with 4 different operation modes. (A–C) co-pulsation mode; (D–F) counter-pulsation mode; (G–I) counter-pulsation mode in a synchronization ratio of 2: 1; (J–L) counter-pulsation mode in a synchronization ratio of 3: 1.
Figure 5. Simulated hemodynamic waveforms in the cardiovascular model with 4 different operation modes. (A–C) co-pulsation mode; (D–F) counter-pulsation mode; (G–I) counter-pulsation mode in a synchronization ratio of 2: 1; (J–L) counter-pulsation mode in a synchronization ratio of 3: 1. 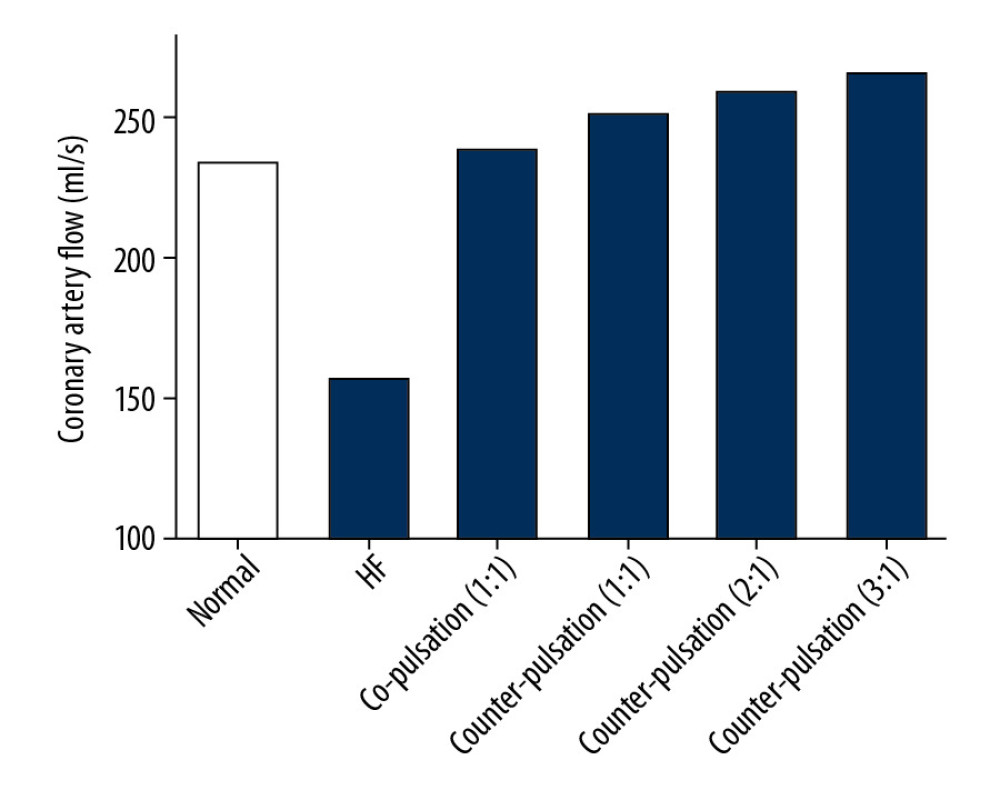 Figure 6. Coronary perfusion in the normal, HF, co-pulsation mode, and counter-pulsation mode in synchronization ratios of 1: 1, 2: 1, and 3: 1.
Figure 6. Coronary perfusion in the normal, HF, co-pulsation mode, and counter-pulsation mode in synchronization ratios of 1: 1, 2: 1, and 3: 1. 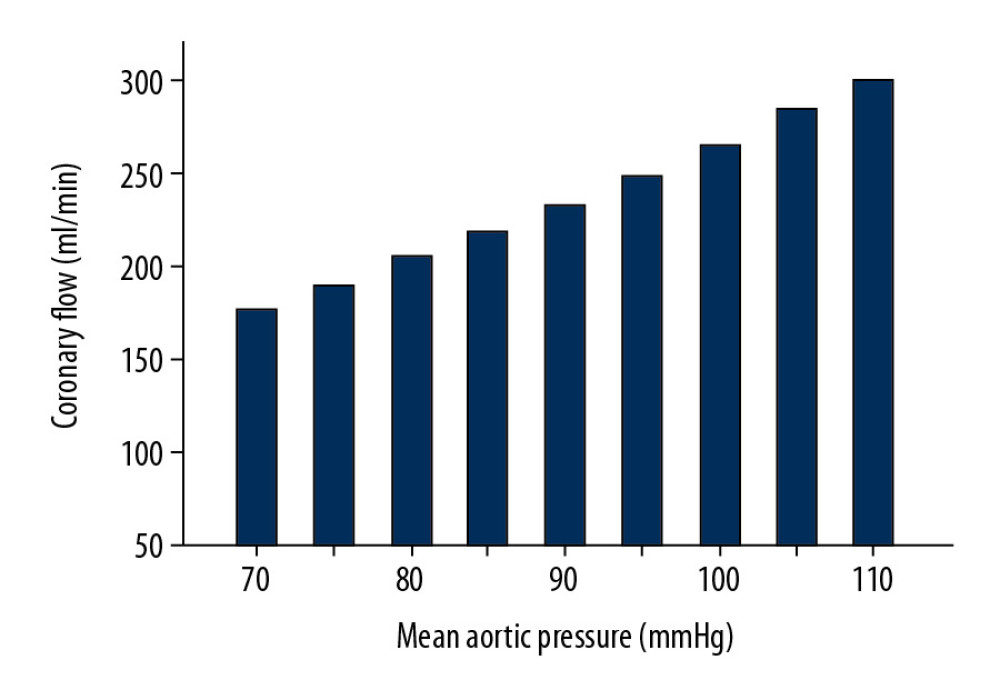 Figure 7. The effects of aortic pressure on coronary perfusion in counter-pulsation mode with a synchronization ratio of 3: 1.
Figure 7. The effects of aortic pressure on coronary perfusion in counter-pulsation mode with a synchronization ratio of 3: 1. Tables
Table 1. Parameter values of the lumped parameter model.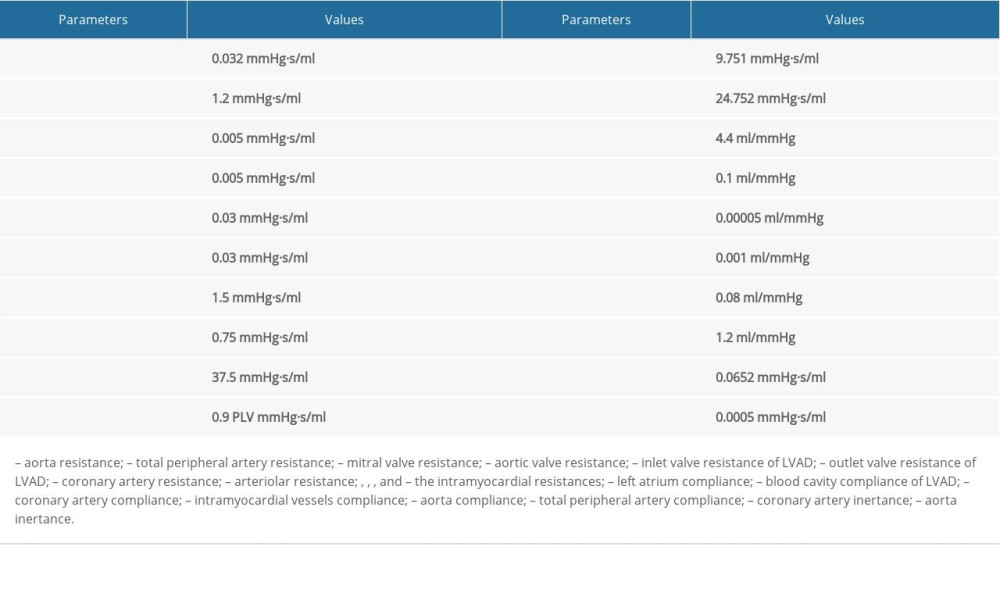 Table 2. Key hemodynamic results for simulated normal and severe HF conditions (normal and HF-III, respectively).
Table 2. Key hemodynamic results for simulated normal and severe HF conditions (normal and HF-III, respectively).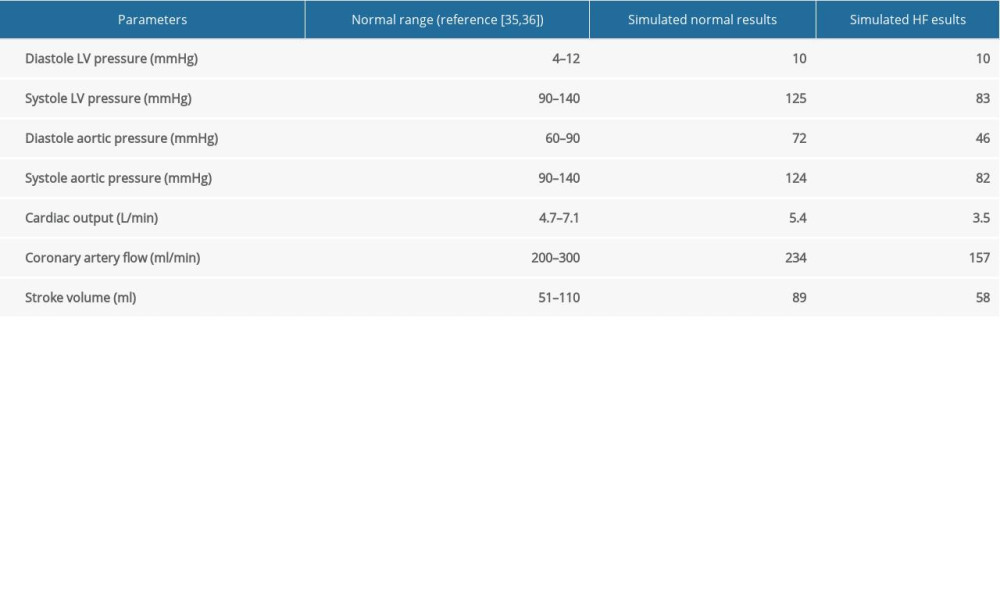 Table 3. Simulated hemodynamic responses of the normal, severe HF, and HF with different modes of LVAD support (TIW – total input work; EEP – energy equivalent pressure).
Table 3. Simulated hemodynamic responses of the normal, severe HF, and HF with different modes of LVAD support (TIW – total input work; EEP – energy equivalent pressure).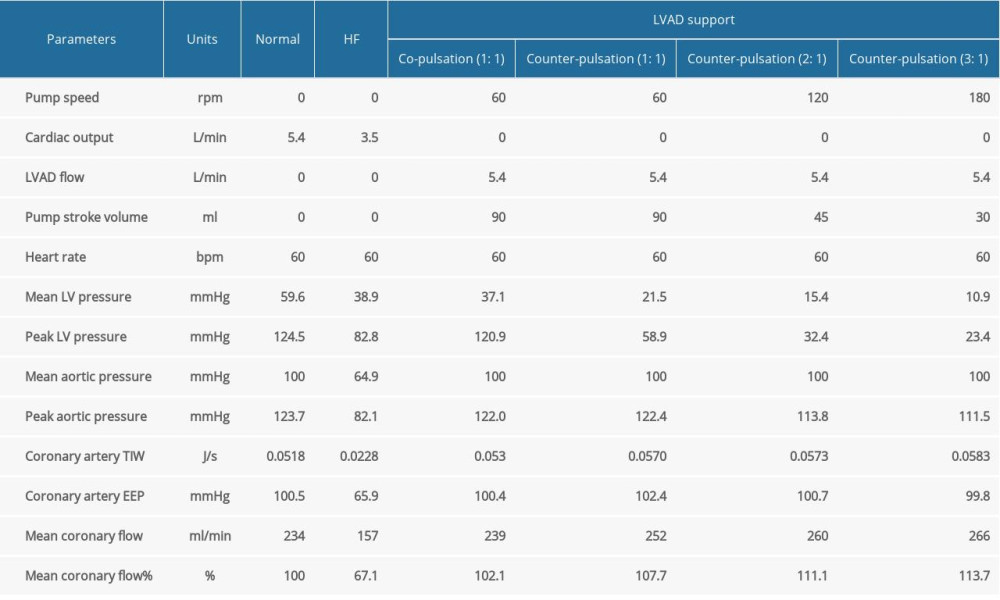
References
1. Gallone G, Palmisano A, Baldetti L, Improved myocardial function with coronary sinus reducer in a patient with refractory angina and heart failure with reduced ejection fraction: Can J Cardiol, 2020; 36(4); 589.e1-4
2. Chow SCY, Wong RHL, Yu PSY, 10-year outcomes post coronary artery bypass grafting in Asian patients with ischemic cardiomyopathy: A comprehensive analysis of survival and cardiac performance: J Thorac Dis, 2020; 12(3); 803-12
3. Wilson RF, Ischemic cardiomyopathy: Congestive heart failure and cardiac transplantation: Clinical, pathology, imaging and molecular profiles, 2017; 119-33, Cham, Springer International Publishing
4. Wang L, Lu M-J, Feng L, Relationship of myocardial hibernation, scar, and angiographic collateral flow in ischemic cardiomyopathy with coronary chronic total occlusion: J Nucl Cardiol, 2019; 26(5); 1720-30
5. Kirklin JK, Naftel DC, Pagani FD, Seventh INTERMACS annual report: 15,000 patients and counting: J Heart Lung Transplant, 2015; 34(12); 1495-504
6. Cheng A, Williamitis CA, Slaughter MS, Comparison of continuous-flow and pulsatile-flow left ventricular assist devices: Is there an advantage to pulsatility?: Ann Cardiothorac Surg, 2014; 3(6); 573-81
7. Wever-Pinzon O, Selzman CH, Drakos SG, Pulsatility and the risk of nonsurgical bleeding in patients supported with the continuous-flow left ventricular assist device HeartMate II: Circ Heart Fail, 2013; 6(3); 517-26
8. Ootaki Y, Kamohara K, Akiyama M, Phasic coronary blood flow pattern during a continuous flow left ventricular assist support: Eur J Cardiorthorac Surg, 2005; 28(5); 711-16
9. Tuzun E, Eya K, Chee HK, Myocardial hemodynamics, physiology, and perfusion with an axial flow left ventricular assist device in the calf: ASAIO J, 2004; 50(1); 47-53
10. Noda H, Takano H, Taenaka Y, Regulation of coronary circulation during left ventricular assist: ASAIO Trans, 1989; 35(3); 445-47
11. Ando M, Takewa Y, Nishimura T, A novel counterpulsation mode of rotary left ventricular assist devices can enhance myocardial perfusion: J Artif Organs, 2011; 14(3); 185-91
12. Lim KM, Kim IS, Choi SW, Computational analysis of the effect of the type of LVAD flow on coronary perfusion and ventricular afterload: J Physiol Sci, 2009; 59(4); 307-16
13. Naito N, Nishimura T, Iizuka K, Novel rotational speed modulation system used with venoarterial extracorporeal membrane oxygenation: Ann Thorac Surg, 2017; 104(5); 1488-95
14. Ambardekar AV, Weiser-Evans MCM, Li M, Coronary artery remodeling and fibrosis with continuous-flow left ventricular assist device support: Circ Heart Fail, 2018; 11(5); e004491
15. Saito T, Miyagawa S, Toda K, Effect of continuous-flow mechanical circulatory support on microvasculature remodeling in the failing heart: Artif Organs, 2019; 43(4); 350-62
16. Frank O, Die grundform des arteriellen pulses: Mathematische analyse: Zentralbl Biol, 1899; 37; 483-526 [in German]
17. Wang Y-YL, Sze W-K, Lin C-C, Examining the response pressure along a fluid-filled elastic tube to comprehend Frank’s arterial resonance model: J Biomech, 2015; 48(6); 907-10
18. Wang YYL, Jan MY, Shyu CS, The natural frequencies of the arterial system and their relation to the heart rate: IEEE Trans Biom Eng, 2004; 51(1); 193-95
19. Gao B, Nie LY, Chang Y, Zeng Y, Physiological control of intraaorta pump based on heart rate: ASAIO J, 2011; 57(3); 152-57
20. Zhuang X, Yang M, Xu L, Pumping rate study of a left ventricular assist device in a mock circulatory system: ASAIO J, 2016; 62(4); 410-20
21. Rebholz M, Amacher R, Petrou A, High-frequency operation of a pulsatile VAD – a simulation study: Biomed Tech (Berl), 2017; 62(2); 161-70
22. Simaan MA, Ferreira A, Chen S, A dynamical state space representation and performance analysis of a feedback-controlled rotary left ventricular assist device: IEEE Trans Control Syst Technol, 2009; 17(1); 15-28
23. Huang H, Yang M, Zang W: IEEE Trans Biomed Eng, 2011; 58(9); 2672-80
24. Bajaj M, Ibrahim S, The microcirculation and the lymphatic system: Capillary fluid exchange, interstitial fluid, and lymph flow: Textbook of Medical Physiology, 2003; 252-72
25. Geven MCF, Bohte VN, Aarnoudse WH: Physiol Meas, 2004; 25(4); 891-904
26. Querzoli G, Fortini S, Espa S, Melchionna S, A laboratory model of the aortic root flow including the coronary arteries: Exp Fluids, 2016; 57(8); ID134
27. Suga H, Sagawa K, Shoukas AA, Load independence of the instantaneous pressure-volume ratio of the canine left ventricle and effects of epinephrine and heart rate on the ratio: Circ Res, 1973; 32(3); 314-22
28. Song Z, Gu K, Gao B, Hemodynamic effects of various support modes of continuous flow LVADs on the cardiovascular system: A numerical study: Med Sci Monit, 2014; 20; 733-41
29. Stergiopulos N, Meister JJ, Westerhof N, Determinants of stroke volume and systolic and diastolic aortic pressure: Am J Physiol, 1996; 270(6); H2050-59
30. Hwang , Changmo , Won K, Compact biventricular assist device with pneumatic actuation mechanism: ASAIO J, 2006; 52(2); 37A
31. Tsuruta H, Sato T, Ikeda N, Mathematical model of cardiovascular mechanics for diagnostic analysis and treatment of heart failure: Part 2. Analysis of vasodilator therapy and planning of optimal drug therapy: Med Biol Eng Comput, 1994; 32(1); 12-18
32. Undar A, Rosenberg G, Myers JL, Major factors in the controversy of pulsatile versus nonpulsatile flow during acute and chronic cardiac support: ASAIO J, 2005; 51(3); 173-75
33. Guan Y, Karkhanis T, Wang S, Physiologic benefits of pulsatile perfusion during mechanical circulatory support for the treatment of acute and chronic heart failure in adults: Artif Organs, 2010; 34(7); 529-36
34. Greenberg DM, Lejemtel TH, Grose KR, Strain AJ, Systemic and coronary effects of intravenous milrinone and dobutamine in congestive heart failure: J Am Soc Cardiol, 1986; 7(5); 1107-13
35. Schampaert S, Rutten MCM, van ‘t Veer M, Modeling the interaction between the intra-aortic balloon pump and the cardiovascular system: The effect of timing: ASAIO J, 2013; 59(1); 30-36
36. Heldt T, Shim EB, Kamm RD, Mark RG, Computational modeling of cardiovascular response to orthostatic stress: J Appl Physiol, 2002; 92(3); 1239-54
37. Naidu SS, Novel percutaneous cardiac assist devices the science of and indications for hemodynamic support: Circulation, 2011; 123(5); 533-43
38. Mebazaa A, Karpati P, Renaud E, Algotsson L, Acute right ventricular failure-from pathophysiology to new treatments: Intensive Care Med, 2004; 30(2); 185-96
39. Jannati M, Attar A, Intra-aortic balloon pump postcardiac surgery: A literature review: J Res Med Sci, 2019; 24; 6
40. Wang Y-YL, Wang W-K, A hemodynamics model to study the collective behavior of the ventricular-arterial system: J Appl Phys, 2013; 113(2); 024702
41. Nakano T, Tominaga R, Morita S, Impacts of pulsatile systemic circulation on endothelium-derived nitric oxide release in anesthetized dogs: Ann Thorac Surg, 2001; 72(1); 156-62
42. Thacher T, Gambillara V, da Silva RF: Cardiovasc Pathol, 2010; 19(4); e91-98
43. John R, Mantz K, Eckman P, Aortic valve pathophysiology during left ventricular assist device support: J Heart Lung Transplant, 2010; 29(12); 1321-29
Figures
 Figure 1. Circuit diagram of the cardiovascular system model. (A) Cardiovascular system and left ventricular assist device (LVAD), (B) Coronary circulation, (C) Systemic circulation. R – vascular resistance; C – vascular compliance; L – blood inertia; D – valve; P – pressure; Q – blood flow; LV – left ventricle; LA – left atrium; CA – coronary artery; A – aorta; S – total peripheral artery; MiV – mitral valve; AoV – aortic valve; AR – arteriolar resistance; IMC – intramyocardial vessels; RM1, RMA, RM2 and RMP – the myocardial resistances.
Figure 1. Circuit diagram of the cardiovascular system model. (A) Cardiovascular system and left ventricular assist device (LVAD), (B) Coronary circulation, (C) Systemic circulation. R – vascular resistance; C – vascular compliance; L – blood inertia; D – valve; P – pressure; Q – blood flow; LV – left ventricle; LA – left atrium; CA – coronary artery; A – aorta; S – total peripheral artery; MiV – mitral valve; AoV – aortic valve; AR – arteriolar resistance; IMC – intramyocardial vessels; RM1, RMA, RM2 and RMP – the myocardial resistances. Figure 2. Flow waveforms of the LVAD in the co-pulsation mode and the counter-pulsation modes. The negative flows represent the pump’s diastole and positive flows represent the pump’s systole.
Figure 2. Flow waveforms of the LVAD in the co-pulsation mode and the counter-pulsation modes. The negative flows represent the pump’s diastole and positive flows represent the pump’s systole. Figure 3. Frequency characteristics of the coronary system.
Figure 3. Frequency characteristics of the coronary system. Figure 4. Simulated pressure waveforms (A) and flow waveforms (B) in the cardiovascular system. PA – aortic pressure; PLV – left ventricular pressure; QA – aortic flow; QCA – coronary artery flow.
Figure 4. Simulated pressure waveforms (A) and flow waveforms (B) in the cardiovascular system. PA – aortic pressure; PLV – left ventricular pressure; QA – aortic flow; QCA – coronary artery flow. Figure 5. Simulated hemodynamic waveforms in the cardiovascular model with 4 different operation modes. (A–C) co-pulsation mode; (D–F) counter-pulsation mode; (G–I) counter-pulsation mode in a synchronization ratio of 2: 1; (J–L) counter-pulsation mode in a synchronization ratio of 3: 1.
Figure 5. Simulated hemodynamic waveforms in the cardiovascular model with 4 different operation modes. (A–C) co-pulsation mode; (D–F) counter-pulsation mode; (G–I) counter-pulsation mode in a synchronization ratio of 2: 1; (J–L) counter-pulsation mode in a synchronization ratio of 3: 1. Figure 6. Coronary perfusion in the normal, HF, co-pulsation mode, and counter-pulsation mode in synchronization ratios of 1: 1, 2: 1, and 3: 1.
Figure 6. Coronary perfusion in the normal, HF, co-pulsation mode, and counter-pulsation mode in synchronization ratios of 1: 1, 2: 1, and 3: 1. Figure 7. The effects of aortic pressure on coronary perfusion in counter-pulsation mode with a synchronization ratio of 3: 1.
Figure 7. The effects of aortic pressure on coronary perfusion in counter-pulsation mode with a synchronization ratio of 3: 1. Tables
 Table 1. Parameter values of the lumped parameter model.
Table 1. Parameter values of the lumped parameter model. Table 2. Key hemodynamic results for simulated normal and severe HF conditions (normal and HF-III, respectively).
Table 2. Key hemodynamic results for simulated normal and severe HF conditions (normal and HF-III, respectively). Table 3. Simulated hemodynamic responses of the normal, severe HF, and HF with different modes of LVAD support (TIW – total input work; EEP – energy equivalent pressure).
Table 3. Simulated hemodynamic responses of the normal, severe HF, and HF with different modes of LVAD support (TIW – total input work; EEP – energy equivalent pressure). Table 1. Parameter values of the lumped parameter model.
Table 1. Parameter values of the lumped parameter model. Table 2. Key hemodynamic results for simulated normal and severe HF conditions (normal and HF-III, respectively).
Table 2. Key hemodynamic results for simulated normal and severe HF conditions (normal and HF-III, respectively). Table 3. Simulated hemodynamic responses of the normal, severe HF, and HF with different modes of LVAD support (TIW – total input work; EEP – energy equivalent pressure).
Table 3. Simulated hemodynamic responses of the normal, severe HF, and HF with different modes of LVAD support (TIW – total input work; EEP – energy equivalent pressure). In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








