27 August 2020: Clinical Research
Correlation Between QTc Prolongation and Obstructive Sleep Apnea in Patients with Type 2 Diabetes Mellitus
Huan Shi1C, Xiaohong Jiang1A*DOI: 10.12659/MSM.926954
Med Sci Monit 2020; 26:e926954
Abstract
BACKGROUND: Obstructive sleep apnea (OSA) plays an important role in the progression of cardiovascular disease (CVD), and is a common symptom in patients with type 2 diabetes mellitus (T2DM). Prolongation of corrected QT interval (QTc) reflects ventricular arrhythmias and CVD. The aim of this study was to explore the relationship between OSA and QTc in T2DM patients and to evaluate the potential application of QTc in clinical practice.
MATERIAL AND METHODS: A total of 358 T2DM patients were involved in this study. OSA was diagnosed with apnea-hypopnea index ≥5 by full-night polysomnography and QTc was measured by a 12-lead electrocardiogram (ECG). Patients were grouped into 2 groups based on median QTc, and clinical data were studied. Logistic regression analysis was used to investigate the association between OSA and QTc with adjusted age, sex, body mass index (BMI), hypertension, total bilirubin (TBL), and smoking history.
RESULTS: Among 358 T2DM patients, 59.2% had OSA. Compared to those in the QTc <418 ms group, older patients, females, patients with higher BMI, and OSA patients in the QTc ≥418 ms group were more likely to have OSA (p<0.05). Correlation analysis suggested that OSA was associated with longer QTc (OR: 2.355, 95% CI: 1.529–3.626, p<0.001). For T2DM patients with QTc ≥418 ms, older patients (OR: 1.042, 95% CI: 1.042–1.064, p<0.001), females (OR: 2.36, 95% CI: 1.371–4.063, p<0.01), and patients with higher BMI (OR: 1.113, 95% CI: 1.037–1.195, p<0.01) were significantly more likely to have OSA.
CONCLUSIONS: In this cross-sectional study, we found that the presence and severity of OSA was associated with QTc prolongation in 358 patients with T2DM, and age, female sex, and BMI appear to be independent risk factors for OSA and CVD.
Keywords: Cardiovascular Diseases, Diabetes Mellitus, Type 2, Electrocardiography, Sleep Apnea Syndromes, Long QT Syndrome, Risk Factors, Sleep apnea, obstructive
Background
Obstructive sleep apnea (OSA) is repeated obstruction of the upper airway during sleep, which causes episodic hypoxia, arousal, sleep disturbance, and sleep-disordered breathing [1]. It is estimated that 9–38% of people experience OSA, particularly for men and people who are older and overweight or obese [2]. However, the reported prevalence of OSA in China is a very low 3–7%, which is mainly due to people ignoring the medical condition [3].
Previous studies have shown that OSA increases the risk of metabolic syndrome, cardiovascular disease, hypertension, and diabetic kidney disease [4–6]. Furthermore, evidence suggests that ~30% of people with OSA have type 2 diabetes mellitus (T2DM), and 60% of T2DM patients in China had newly diagnosed comorbid OSA [7]. The mechanism of OSA and T2DM is thought to be sleep fragmentation and chronic intermittent hypoxia, which activates the sympathoadrenal system, leads to oxidative stress and inflammatory responses, and causes abnormal alterations in adipokines [8–10]. Consequently, increased insulin resistance and pancreatic β-cell dysfunction occur during the progression of T2DM.
The QT interval is defined as the total time required for ventricular myocardial depolarization and repolarization. The prolonged heart rate-corrected QT interval (QTc interval) is closely associated with all-cause and CVD-related mortality in patients with T2DM [11–15]. A recent meta-analysis revealed that OSA is an independent risk factor for CVD and other cardiovascular consequences [16]. Few studies have investigated the relationship between QTc and OSA, and the results are conflicting. Shamsuzzman et al. and Cicek et al. reported that OSA was associated with prolonged QTc [17,18]. Rossi et al. found that the QTc interval was shorter in moderate-to-severe symptomatic OSA patients who had continuous positive airway pressure, and obvious prolongation of QTc was observed after withdrawal of airway pressure [19]. However, Barta et al. and Viigimae et al. suggested that nocturnal QTc did not differ from QTc in patients with OSA [20,21].
Thus, in light of the limited data on the association between OSA and QTc in patients with T2DM, we conducted this study to investigate the risk factors for OSA and the relationship between OSA and QTc in patients with T2DM in China, as well as their potential applications in clinical practice.
Material and Methods
STUDY DESIGN AND PARTICIPANTS:
In this retrospective study, a total of 358 patients with T2DM were recruited from January 2017 to May 2019, who visited the Department of Endocrinology Inpatient Service, the 3rd Affiliated Hospital of Soochow University, Changzhou, China. T2DM patients was diagnosed according to the 1999 WHO criteria for diabetes (fasting plasma glucose (FPG) ≥7.0 mmol/l or 2-h plasma glucose (PG) ≥11.1 mmol/l) [22]. The exclusion criteria were: 1) previously treated for OSA; and 2) disease history, such as coronary artery disease, valvar heart disease, arrhythmia, cardiomyopathy, cerebrovascular disease, chronic lung or liver disease, acute or chronic kidney disease, malignancy, electrolyte disturbance, anemia, or other clinical conditions. This study was approved by the Ethics Committee of the Third Affiliated Hospital of Soochow University. Written informed consent was obtained from each participant before enrolment. Clinical data and life-style data were collected.
Demographic characteristics, including age, sex, weight, height, waist circumference, hip circumference, were recorded. Body mass index (BMI, weight (kg)/height2(m2)) was calculated and classified as overweight (24 kg/m2 ≤BMI ≤27.9 kg/m2) or obese (BMI ≥28 kg/m2) in Chinese adults. Patients were told not to eat or drink for 12 h before the general examinations. We assessed the following laboratory parameters using the AU5800 Series Chemistry System (Beckman, USA): ALT, AST, GGT, AKP, LDH, TBL, DBIL, TC, TG, HDL, LDL, CR, BUN, UA, and FPG. HbA1c values were measured using the D-10 Hemoglobin Testing System (Bio-Rab, USA). Patients taking anti-hypertensive drugs and those with blood pressure above 140/90 mmHg after sitting quietly for at least 5 min (OMRON model HEM-752 FUZZY, OMRON Company, Dalian, China) [23] were regarded as having hypertension. Liver ultrasound scanning (LOGIQ E9, GE, USA) was performed by an experienced radiologist who was blinded to the patients’ details. Diagnosis of fatty liver disease was made on the basis of characteristic ultrasonographic features: evidence of increased echogenicity of the liver parenchyma in comparison with right renal cortex, attenuation of the ultrasound beam, and poor visualization of intrahepatic structures [24].
OSA MEASUREMENT:
Full-night polysomnography was used to record sleep condition of each participant using the Voyager Digital Imaging E-series system (Compumedics, Melbourne, Australia), and OSA was assessed with the apnea-hypopnea index (AHI) ≥5 [25].
QTC MEASUREMENT:
A standard 12-lead ECG was recorded using the CardioDirect 12 system (Spacelabs Healthcare, Snoqualmie, WA, USA) or a standard red-paper machine. QT interval was measured as the mean of the QT intervals for all viable leads using Metasoft 3.9 software (CardioDirect 12 system) or CardioCaliper (standard red-paper machine). QTc was corrected for heart rate using Bazett’s formula [26].
STATISTICAL ANALYSIS:
SPSS 26.0 (IBM Corp., Armonk, NY, USA) was used for data analysis. The homeostasis model assessment 2 insulin resistance index (HOMA2-IR) was calculated using software downloaded from http://www.dtu.ox.ac.uk. For continuous data, normality and homogeneity of variance was first tested via Shapiro-Wilk test and Levene test, respectively, and results are presented as median (interquartile range), whereas categorical data are presented as number and percentage. The Mann-Whitney U test was performed to compare continuous data of 2 groups, and the chi-square test was used for comparison of categorical data. Logistic regression analyses were used to investigate the association between OSA and QTc. Here, we used median QTc interval (≥418 ms) to group the objectives in our study [27]. For logistic regression analysis, 3 models were used to demonstrate the relationship between OSA and QTc. In brief, the model for uncorrected analysis (Model 1), the model calibrated with age, sex, and BMI (Model 2), and Model 3 was substituted into Model 2 with age, sex, BMI, history of hypertension, smoke, and TBL. A forest plot was used for subgroup analysis with GraphPad prism 8 software. P value <0.05 was considered as statistically significant.
Results
CLINICAL CHARACTERISTICS OF PARTICIPANTS:
The clinical characteristics of the participants stratified by the median QTc are summarized in Table 1. The median age was 55 years (IQR: 48, 65) in group 2 (QTc ≥418 ms) and 49 years (IQR: 41, 58) in group 1 (QTc <418 ms) (p<0.001). In group 2, 39% of participants were females, compared to 20% in group 1. The median BMI of group 2 was 26.96 kg/cm2 (IQR: 25.21 kg/cm2, 30.08 kg/cm2), which was higher than that of group 1 (median: 25.78 kg/cm2, IQR: 23.19 kg/cm2, 28.29 kg/cm2) (p<0.001).
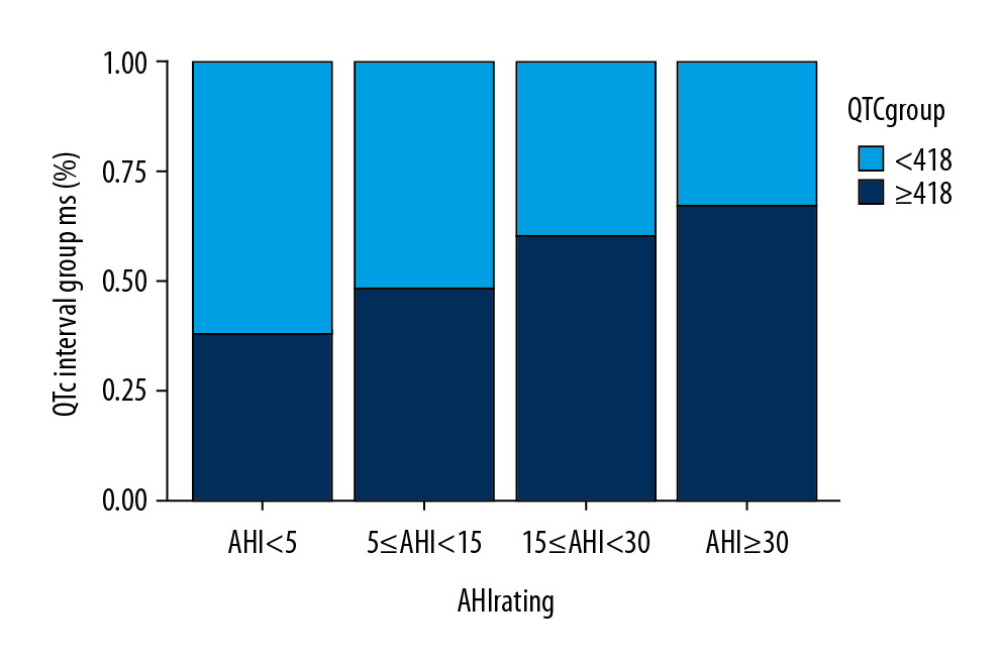
Group 2 had higher levels of AST, LDH, TG, HOMA2-IR, QTc, and AHI and lower TBL and HBA1C. In addition, the percentage of patients with OSA (AHI ≥5) in group 2 was higher than that in group 1 (69.23% vs. 48.86%). Further, when patients were grouped by OSA, the proportion of patients with QTc interval ≥418 ms was higher (Figure 1).
LOGISTIC REGRESSION ANALYSIS OF THE ASSOCIATION BETWEEN OSA AND QTC:
Logistic regression analysis was used to further investigate the association of OSA with QTc. Here, 3 models were generated: the model for uncorrected analysis (Model 1), the model calibrated with age, sex, and BMI (Model 2), and Model 3 was substituted into Model 2 with age, sex, BMI, history of hypertension, smoke, and TBL. The covariates were chosen as potential confounders based on their biological plausibility or statistical results with prolonged QTc in univariate analyses.
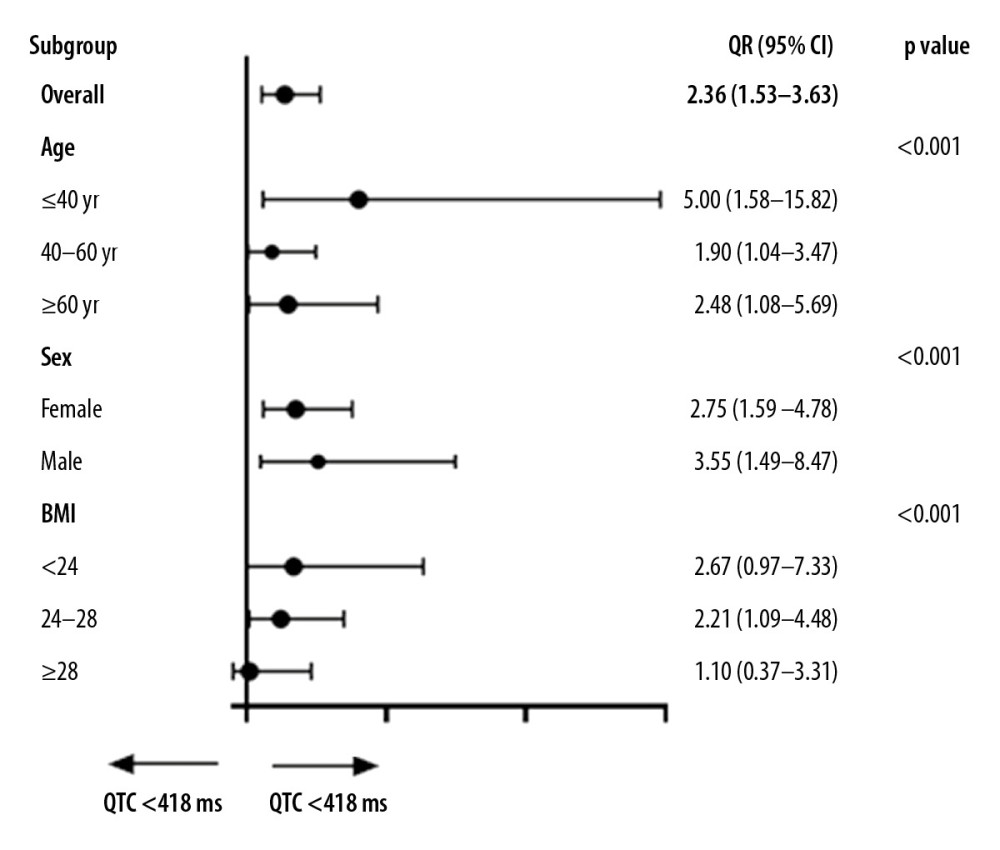
Table 2 shows the effects of adjustment for potential confounders on the relationship between OSA and QTc. In Model 1, OSA was associated with increased odds of QTc ≥418 ms (OR: 2.355; 95% CI: 1.529–3.626; <0.001). In Model 2 and Model 3, the association between OSA and QTc was not weakened after adjustment for potential confounding factors. In Model 3, independent predictors of QTc ≥418 ms were older age (OR: 1.866, 95% CI: 1.065–3.272; p<0.001), female sex (OR: 2.360; 95% CI: 1.371–4.063; p<0.01), and higher BMI (OR: 1.113; 95% CI: 1.037–1.195; p<0.01). Subgroup analysis was analyzed by using sex, age, and BMI for the adjustment of all factor’s models. Overweight patients with OSA tended to have longer QTc (OR, 2.21; 95% CI, 1.09–4.48; P<0.001), as shown in Figure 2.
Discussion
We found that T2DM patients with OSA were associated with QTc prolongation, which is consistent with previous results [17–19]. The potential risk factors were age, higher BMI, sex (female), hypertension history, and fatty liver disease, as well as higher levels of AST, LDH, TBL, TG, and HOMA2-IR. For the analysis of relationship between OSA and QTc intervals, we found that there were more patients with OSA in the QTc ≥418 ms group than that in the QTc <418 ms group, and the proportion of patients with QTc interval ≥418 ms increased according to the progression of OSA. Also, in logistic regression analysis, we revealed that OSA was significantly associated with QTc ≥418 ms, even after adjustment for multiple potential confounding factors. Independent risk factors for QTc ≥418 ms were older age, female sex, and higher BMI [14,25]. Thus, our results suggest that the presence and severity of OSA is associated with longer QTc in patients with T2DM, indicating that OSA-related QTc prolongation may be an additional predictor of increased CVD risk in patients with T2DM and showing the importance of evaluating global cardiovascular and arrhythmia risk [17].
The mechanisms underlying the relationship between OSA and QTc prolongation are poorly investigated, but it has been suggested that repetitive episodes of apnea during sleep cause activation of the sympathoadrenal system, resulting in sustained elevation of sympathetic activity while awake [28,29]. Goyal et al. [25] found that the elevation in plasma catecholamine levels can initiate inflammatory cascades, causing injured tissues to express damage-associated molecular patterns that stimulate receptors on immune cells. Then, the release of inflammatory cytokines may induce the homing of inflammatory cells to vulnerable end organs, thereby aggravating end-organ damage and increasing the risk of hypertension and myocardial infarction. OSA also causes intermittent hypoxemia, which stimulates ventilation, leading to increased negative pressure in the chest, heart rate, and blood pressure. As the consequence, a greater left ventricular afterload can cause myocardial ischemia, QTc prolongation, and ventricular arrhythmias.
This study has some limitations. First, the study had a small sample drawn from individuals referred to a sleep clinic. Secondly, besides QT interval, other markers of cardiac repolarization abnormalities and sudden cardiac death [30], such as QT dispersion, were not evaluated. Thirdly, a possible effect played by medication to control QTC was not considered, which mainly involves patients with long-term use of diuretics. Lastly, follow-up is needed to assess health conditions of patients regarding the effects of QTc prolongation on outcomes such cardiovascular complications (e.g., arrythmia).
Conclusions
In conclusion, OSA, older age, female sex, and higher BMI were independently associated with QTc > 418 ms in patients with T2DM in China. The QTc prolongation in patients with T2DM raises the risk of OSA and adverse cardiac outcomes.
References
1. Maspero C, Giannini L, Galbiati G, Obstructive sleep apnea syndrome: A literature review: Minerva Stomatol, 2015; 64(2); 97-109
2. Senaratna CV, Perret JL, Lodge CJ, Prevalence of obstructive sleep apnea in the general population: A systematic review: Sleep Med Rev, 2017; 34; 70-81
3. Xu ZF, Luo X, Shi J, Lai Y, Quality analysis of smart phone sleep apps in China: Can apps be used to conveniently screen for obstructive sleep apnea at home?: BMC Med Inform Decis Mak, 2019; 19(1); 224
4. Leong WB, Jadhakhan F, Taheri S, The association between obstructive sleep apnea on diabetic kidney disease: A systematic review and meta-analysis: Sleep, 2016; 39(2); 301-8
5. Xu S, Wan Y, Xu M, The association between obstructive sleep apnea and metabolic syndrome: A systematic review and meta-analysis: BMC Pulm Med, 2015; 15; 105
6. Wang X, Bi Y, Zhang Q, Obstructive sleep apnoea and the risk of type 2 diabetes: a meta-analysis of prospective cohort studies: Respirology, 2013; 18(1); 140-46
7. Zhang P, Zhang R, Zhao F, The prevalence and characteristics of obstructive sleep apnea in hospitalized patients with type 2 diabetes in China: J Sleep Res, 2016; 25(1); 39-46
8. Rajan P, Greenberg H, Obstructive sleep apnea as a risk factor for type 2 diabetes mellitus: Nat Sci Sleep, 2015; 7; 113-25
9. Greco C, Spallone V, Obstructive sleep apnea syndrome and diabetes. Fortuitous association or interaction?: Curr Diabetes Rev, 2015; 12(2); 129-55
10. Veasey SC, Rosen IM, Obstructive sleep apnea in adults: N Engl J Med, 2019; 380(15); 1442-49
11. Kuzu F, The effect of type 2 diabetes on electrocardiographic markers of significant cardiac events: Pak J Med Sci, 2018; 34(3); 626-32
12. Dörr M, Ruppert J, Robinson DM, The relation of thyroid function and ventricular repolarization: decreased serum thyrotropin levels are associated with short rate-adjusted QT intervals: J Clin Endocrinol Metab, 2006; 91(12); 4938-42
13. Ma Q, Li Z, Guo X, Prevalence and risk factors of prolonged corrected QT interval in general Chinese population: BMC Cardiovasc Disord, 2019; 19(1); 276
14. Cox AJ, Azeem A, Yeboah J, Heart rate-corrected QT interval is an independent predictor of all-cause and cardiovascular mortality in individuals with type 2 diabetes: The Diabetes Heart Study: Diabetes Care, 2014; 37(5); 1454-61
15. Cardoso CR, Salles GF, Deccache W, Prognostic value of QT interval parameters in type 2 diabetes mellitus: Results of a long-term follow-up prospective study: J Diabetes Complications, 2003; 17(4); 169-78
16. Xie W, Zheng F, Song X, Obstructive sleep apnea and serious adverse outcomes in patients with cardiovascular or cerebrovascular disease: A PRISMA-compliant systematic review and meta-analysis: Medicine (Baltimore), 2014; 93(29); e336
17. Shamsuzzaman A, Amin RS, van der Walt C, Daytime cardiac repolarization in patients with obstructive sleep apnea: Sleep Breath, 2015; 19(4); 1135-40
18. Çiçek D, Lakadamyali H, Gökay S, Effect of obstructive sleep apnea on heart rate, heart rate recovery and QTc and P-wave dispersion in newly diagnosed untreated patients: Am J Med Sci, 2012; 344(3); 180-85
19. Rossi VA, Stoewhas AC, Camen G, The effects of continuous positive airway pressure therapy withdrawal on cardiac repolarization: data from a randomized controlled trial: Eur Heart J, 2012; 33(17); 2206-12
20. Barta K, Szabó Z, Kun C, The effect of sleep apnea on QT interval, QT dispersion, and arrhythmias: Clin Cardiol, 2010; 33(6); E35-39
21. Viigimae M, Karai D, Pilt K, QT interval variability index and QT interval duration during different sleep stages in patients with obstructive sleep apnea: Sleep Med, 2017; 37; 160-67
22. Bellentani S, Saccoccio G, Masutti F, Prevalence of and risk factors for hepatic steatosis in Northern Italy: Ann Intern Med, 2000; 132(2); 112-17
23. National guideline for hypertension management in China (2019): Zhonghua Xin Xue Guan Bing Za Zhi, 2020; 48(1); 10-46
24. Gabir MM, Roumain Janine, Hanson RL, The 1997 American Diabetes Association and 1999 World Health Organization Criteria for hyperglycemia in the diagnosis and prediction of diabetes: Diabetes Care, 2000; 23(8); 1108-12
25. Goyal M, Johnson J, Obstructive sleep apnea diagnosis and management: Mo Med, 2017; 114(2); 120-24
26. Luo S, Michler K, Johnston P, Macfarlane PW, A comparison of commonly used QT correctio formulae: The effect of heart rate on the QTc of normal ECGs: J Electrocardiol, 2004; 37(Suppl); 81-90
27. Targher G, Valbusa F, Bonapace S, Association of nonalcoholic fatty liver disease with QTc interval in patients with type 2 diabetes: Nutr Metab Cardiovasc Dis, 2014; 24(6); 663-69
28. Whitsel EA, Boyko EJ, Siscovick DS, Reassessing the role of QTc in the diagnosis of autonomic failure among patients with diabetes: A meta-analysis: Diabetes Care, 2000; 23(2); 241-47
29. Abboud F, Kumar R, Obstructive sleep apnea and insight into mechanisms of sympathetic overactivity: J Clin Invest, 2014; 124(4); 1454-57
30. Bazoukis G, Yeung C, Ho WH, Association of QT dispersion with mortality and arrhythmic events-A meta-analysis of observational studies: J Arrhythm, 2020; 36(1); 105-15
Figures
Tables
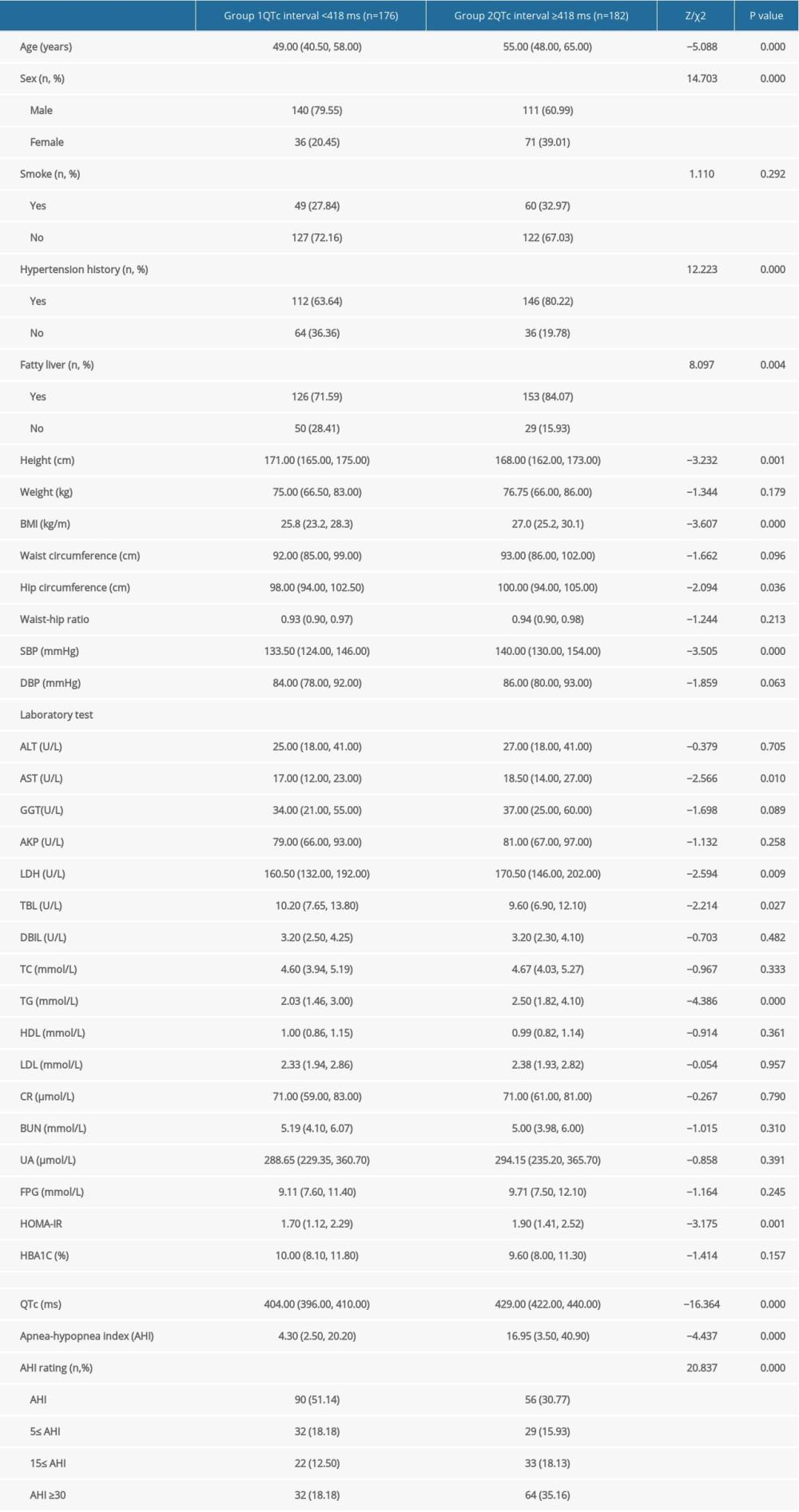 Table 1. Clinical characteristics of participants stratified according to median corrected QT interval.
Table 1. Clinical characteristics of participants stratified according to median corrected QT interval. Table 2. Logistic regression analysis of the association between obstructive sleep apnea and corrected QT interval ≥418 ms.
Table 2. Logistic regression analysis of the association between obstructive sleep apnea and corrected QT interval ≥418 ms. Table 1. Clinical characteristics of participants stratified according to median corrected QT interval.
Table 1. Clinical characteristics of participants stratified according to median corrected QT interval. Table 2. Logistic regression analysis of the association between obstructive sleep apnea and corrected QT interval ≥418 ms.
Table 2. Logistic regression analysis of the association between obstructive sleep apnea and corrected QT interval ≥418 ms. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387