11 December 2020: Review Articles
Preoperative Intranasal Decolonization with Topical Povidone-Iodine Antiseptic and the Incidence of Surgical Site Infection: A Review
Vidmi Taolam Martin1ABCDEF, Mohamed Abdullahi Abdi1ABEF, Jie Li1BCEF, Dongtai Li1BCDF, Zhaozhen Wang1BCDE, Xianliao Zhang1BDEF, Welera Haissou Elodie2BCEF, Bo Yu1ABCDEF*DOI: 10.12659/MSM.927052
Med Sci Monit 2020; 26:e927052
Abstract
ABSTRACT: Surgical site infection (SSI) occurs at the incisional site of a surgical procedure and usually involves the skin. The use of antibacterial courses to manage SSIs is still very challenging in clinical settings. When not used appropriately, antibacterial agents can lead to increased rates of adverse events. However, various antibacterial agents that can destroy the growth of bacteria are now available. This article aims to discuss the role of preoperative intranasal decolonization with topical povidone-iodine antiseptic in the incidence of SSI based on a review of the literature. Topical bactericidal agents can be administered intranasally before surgery to eliminate potentially harmful bacteria, including antibiotic-resistant strains of bacteria. Therefore, a few studies have recommended the use of intranasal povidone-iodine solution in the clinical setting; however, it also appears to be a promising antiseptic regimen for preoperative decontamination in patients planned to undergo surgery. Povidone-iodine is a commonly used medical antiseptic agent that is used by surgeons to promote wound healing and prevent postoperative bacterial infections. Chlorhexidine gluconate is both an antiseptic and a disinfectant, which is used to clean the skin and surgical instruments. Our review of the literature on studies on the effectiveness of intranasal povidone-iodine in the reduction of intranasal bacterial colonization and the prevention of SSI identified only 5 controlled clinical studies. One study, however, showed increased effectiveness in preventing SSI when topical intranasal povidone-iodine was combined with the use of chlorhexidine gluconate washcloths. Further large-scale controlled clinical studies are needed before proper guidelines can be made.
Keywords: Administration, Intranasal, Oral Surgical Procedures, Povidone-Iodine, Administration, Topical, Anti-Infective Agents, Local, Antibiotic Prophylaxis, Incidence, Surgical Wound Infection
Background
Surgical site infections (SSIs) are defined as infections that occur after surgery through the incisional surgical site and sometimes can involve the skin, soft tissues, or implanted material [1–3]. SSIs are complicated conditions for patients and surgeons and are associated with increased morbidity and mortality [3,4]. The term surgical site infection was introduced by the Surgical Wound Infection Task Force in 1992, when Horan et al. defined it as an infection affecting the wound and occurring within 30 days of surgery [5]. In 2013, the Centers for Disease Control and Prevention (CDC) modified the surveillance period to 30 days for superficial SSIs and 90 days for deep incisional or organ/space SSIs [6]. SSI is a common problem in the hospital setting. In 2009, the World Health Organization (WHO) reported that 23% of surgical patients worldwide developed an SSI [7]. Recently, the WHO reported that SSI is the most surveyed and common type of healthcare-associated infection in low-income and middle-income countries, affecting up to one-third of patients who have undergone a surgical procedure [8]. The impact of healthcare-associated infection is multifactorial and includes extended hospital stays, increased resistance of microorganisms to antimicrobials, and high healthcare system costs [9]. The prevention of SSI has become more important as the number of surgical procedures performed worldwide continues to grow. It is well known that there is an increasing need for evidence-based regimens for the prevention of SSI. Darouiche et al. suggested that since a patient’s skin is a reservoir of pathogens, improving skin antisepsis would reduce SSIs [1]. Antiseptics are chemical agents used to decrease the number of bacteria on the skin surface and surgical instruments [10]. However, the control of SSI remains challenging in the clinical setting. Notwithstanding the improvements that have been made in infection control systems, SSI continues to influence the quality and cost of health care [11].
According to the surveillance of SSI in the National Health Service hospitals in the United Kingdom,
Antiseptic agents are classified into 3 levels, high, intermediate, and low, depending on the spectrum of microbicidal activity. High-level antiseptic agents are capable of killing all types of microorganisms, but their use is limited to disinfecting instruments. Povidone-iodine, which belongs to the intermediate-level, can be used for preoperative disinfection of the skin. Chlorohexidine gluconate, which is a low-level antibacterial agent, has a more limited microbicidal effect compared to povidone-iodine. Povidone-iodine and chlorohexidine gluconate are used worldwide because of their wide-spectrum antimicrobial activity and because bacteria resistance against these agents is rare. However, most staph infections (
In the past 20 years, mupirocin has been the most used regimen for preoperative decolonization. Perl et al. found that the risk of resistance to mupirocin, when mupirocin is used for preoperative decolonization, was low, at 0.6% [16]. However, mupirocin regimens are more expensive than intranasal povidone-iodine regimens. Povidone-iodine is a broad-spectrum antiseptic for topical application in the management and prevention of wound infection. It has long-lasting antiseptic effects, which are due to its slow absorption through soft tissue, making it the choice for longer operations [17]. Studies have shown that chlorhexidine gluconate provides superior prevention of SSI [18,19]. We believe that if the data support the relevant clinical effectiveness of a povidone-iodine strategy for SSI, this should be acknowledged. As highlighted by Liu et al., intranasal decolonization using antimicrobials or antiseptics is done to minimize the risk of SSI by preventing pathogens from the nasal cavity being transferred to the skin where a surgical incision will be performed [20]. They have also pointed out that the potential effectiveness of intranasal decolonization of
Observational Data
CURRENT APPROACHES TO THE PREOPERATIVE USE OF TOPICAL POVIDONE-IODINE AND CHLORHEXIDINE GLUCONATE IN THE PREVENTION OF SSI:
The outcomes of topical povidone-iodine and chlorhexidine use on local bacteria have been well investigated. Studies have reported that the inhibitory effects of chlorhexidine gluconate on bacteria were significantly stronger than those of topical povidone-iodine [26,27]. In contrast, a different study found that the effect of topical povidone-iodine was more persistent than that of chlorhexidine gluconate [28].
The instructions for the use of intranasal povidone-iodine and chlorhexidine gluconate are to apply chlorhexidine washcloths to the entire body of the patients, except the face, once the night before and once in the morning of the operation; and for the nasal povidone-iodine solution, a specialist nurse inserts a swab into both nostrils of patients, using a fresh swab each time and rotating for 15 s, once in the morning of the operation [4–7,9]. An amount of 15 mL of 0.12% chlorohexidine oral rinse is used as a mouthwash for 30 s and then spit out, once the night before and once in the morning of the operation. Following the mouthwash, patients are to wait at least 30 min before rinsing the mouth with water [4].
STUDIES SUPPORTING THE USE OF INTRANASAL POVIDONE-IODINE AND CHLORHEXIDINE GLUCONATE:
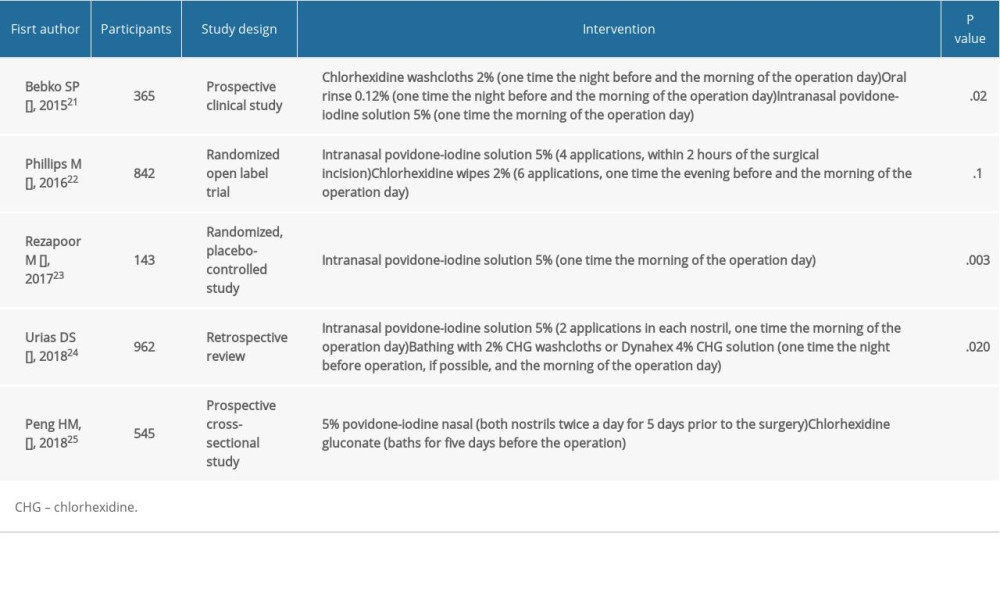
Bebko et al. [21] reported that the procedure including nasal povidone-iodine could likely improve antibiotic resistance, noting that the length of hospitalization was longer in patients receiving a mupirocin and chlorhexidine decolonization protocol than in patients receiving chlorhexidine washcloths, chlorohexidine oral rinse, and nasal povidone-iodine (5 days vs. 2 days). Also, the cost of decontamination was higher in the mupirocin and chlorhexidine group than in the chlorhexidine washcloths, chlorohexidine oral rinse, and nasal povidone-iodine group ($54 per patient vs. $35 per patient, respectively). This is consistent with results of previous studies that showed the handmade nasal povidone-iodine swab is a simple, inexpensive, and effective decolonization protocol [20,25].
Patients who spent more than 2 h in the operating room were more likely to get an SSI 4 weeks after surgery than patients who spent less time in the operating room (odds ratio, 4.59 [95% confidence interval, 1.67–12.65]; P=.003) [21]. Rezapoor et al. [23] suggested conducting surgery within 12 h of applying the nasal povidone-iodine to reduce the amount of S. aureus in the nostrils at the time of surgery. Daniel et al. [24] reported that povidone-iodine was the better option for their patients who had surgery planned within 24 h. Phillips et al. [22] reported that the use of nasal povidone-iodine by the patient care team just before surgery may ensure greater treatment compliance. Bebko et al. reported that a hospital stay longer than 24 h is a significant risk factor for SSI and the authors noted a significant reduction in overall SSI rates among orthopedic patients after the use of a decontamination protocol [21]. However, the current WHO guidelines for the prevention of SSI mention that it is good clinical practice for patients to bathe or shower before surgery, and their panel has suggested that patients undergoing cardiothoracic and orthopedic surgery, who have been identified to have nasal carriage of S. aureus, should be given preoperative 2% intranasal mupirocin ointment with or without a chlorhexidine gluconate body wash [8,15].
THE USE OF SELECTIVE ANTIBIOTIC THERAPY FOR SSI:
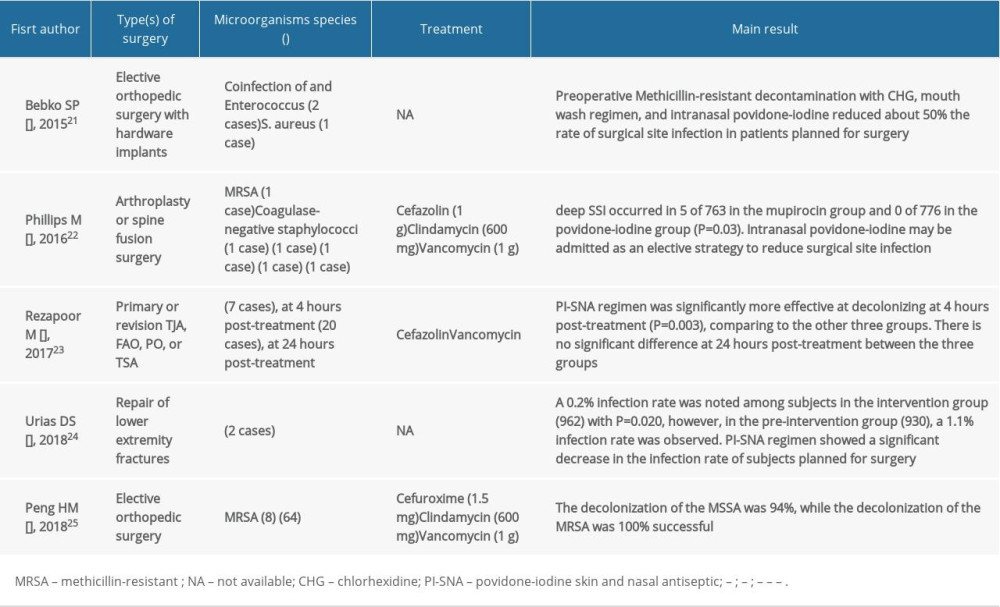
We identified 3 studies using either first generation cephalosporin [22,23], second generation cephalosporin [25], and glycopeptide [22,23,25] classes of antibiotics for the postoperative management of SSI (Table 2).
To minimize the risk of SSI, the WHO guidelines recommend that surgical antibiotic prophylaxis should be given before the surgical incision, when indicated by the type of operation. The panel recommended the administration of surgical antibiotic prophylaxis within 2 h before the incision, while considering the half-life of the antibiotic [8,15].
FUTURE DIRECTIONS OF TOPICAL ANTISEPTIC AGENTS AND THE PREVENTION OF SSI:
Preventive protocols with topical povidone-iodine alone or in combination with chlorhexidine gluconate are now in use. The application of nasal povidone-iodine may be considered as an option to reduce SSI [21,22]. In addition to an intranasal decolonization protocol, it could be important to include other sites (throat, axilla, groin, and/or rectum) in a study with a much larger number of patients with long-term follow-up, which also includes surgical antibiotic prophylaxis [23,25]. In brief, an additional large-scale, robust study is needed to determine the effectiveness of these antiseptic agents in decreasing the rate of SSI in the clinical setting before evidence-based conventional infection treatment guidelines can be established.
Discussion
The present review included a search of the literature to identify studies on the effectiveness of preoperative intranasal decolonization with topical antiseptic agents in the prevention of SSI. Few controlled studies have been published, and most studies have involved the use of topical povidone-iodine [1,18,21–23,25,29–31]. One study, however, showed increased effectiveness when topical intranasal povidone-iodine was combined with the use of chlorhexidine gluconate-impregnated washcloths [21]. Data on the identified pathogens were available in all reported cases [21–25]. The most commonly reported microorganism species were MRSA and
Similar to how the mouth and throat cavities of healthy persons are known to be colonized by a multitude of diverse microorganisms which affect the health of the mouth and body [35], SSIs are caused by contamination of an incision with microorganisms from the patient’s own body during surgery. Unfortunately, these microorganisms may develop biofilms, which are resistant to antibiotic therapy. And that is why the rate of resistant microorganisms has increased in clinical settings over the past years, leading to longer hospital stays. Urban et al. [36] reported that the costs per SSI caused by prolonged hospitalization, supplementary analyses, antibiotic therapy, and reoperation, range from $400 (USD) for superficial SSIs to more than $30 000 for serious infections. This indicates there is a need for antiseptic agents with broad-spectrum activity that ensure oral, oropharyngeal, and whole-body antiseptic coverage. Therefore, according to data from the literature, regimens using chlorohexidine and povidone-iodine can help patients and surgeons overcome the challenges of SSIs by minimizing
The intranasal povidone-iodine regimen was effective in the studies we reviewed. In the most comprehensive study, conducted by Daniel et al. [24], an intervention of nasal povidone-iodine and bathing with 2% chlorhexidine gluconate washcloths or Dynahex 4% chlorohexidine gluconate solution was compared with pre-intervention in trauma patients experiencing urgent repair of lower extremity fractures. Results showed better outcomes in the intervention group than in the pre-intervention group, including having no other risk factors correlated with the increase of SSI.
The timing of the intranasal povidone-iodine preoperative decontamination in the studies we reviewed varied, including once in the morning of the operation day, within 2 h of the surgical incision, and twice per day for 5 days before the operation [21–25]. The advantages of these antibacterial agent antisepsis regimens include a rapid bactericidal action against a broad spectrum of microorganisms, significant reduction in the number of microorganisms on intact skin, continued antimicrobial activity for up to 6 h after application, lack of contribution to antibiotic resistance, and lower cost than alternative treatments [4,5,7,12,14,21,23–25,31,36]. In other studies [29,30], the authors reported that povidone-iodine use resulted in hemostatic and anti-inflammatory outcomes during minor oral surgery.
The effectiveness of intranasal povidone-iodine solution does not indicate that this regimen is the correct choice for all patients undergoing surgery. Nevertheless, these data validate the use of intranasal povidone-iodine solution for suitable patients.
Based on available data and an evaluation of clinical suitability, intranasal povidone-iodine combined with chlorhexidine washcloths for patients with SSI should be considered if these patients are clinically stable with no immediate signs of postoperative infection, the application of intranasal povidone-iodine has completely cleared their bacteremia, and there are no concerns about the inhalation of nasal therapy into the upper respiratory system. More robust data on intranasal povidone-iodine combined with other antiseptics for the preoperative decontamination of MRSA are required to validate the effectiveness of this critical contamination.
Conclusions
Our review of the literature on the effectiveness of preoperative intranasal decolonization with topical antiseptic agents and the prevention of SSI has shown there is a lack of controlled studies. Although the use of topical intranasal povidone-iodine has been supported in the literature, and one study showed increased effectiveness when also using chlorhexidine gluconate washcloths, additional large-scale controlled clinical studies are needed before further guidelines or recommendations can be made for their routine use in the prevention of SSI.
References
1. Darouiche RO, Wall MJ, Itani KM, Chlorhexidine-alcohol versus povidone-iodine for surgical-site antisepsis: N Engl J Med, 2010; 362(1); 18-26
2. Korol E, Johnston K, Waser N, A systematic review of risk factors associated with surgical site infections among surgical patients: PLoS One, 2013; 8(12); e83743
3. Owens CD, Stoessel K, Surgical site infections: Epidemiology, microbiology and prevention: J Hosp Infect, 2008; 70(Suppl 2); 3-10
4. Garner BH, Anderson DJ, Surgical site infections: an update: Infect Dis Clin North Am, 2016; 30(4); 909-29
5. Horan TC, Gaynes RP, Martone WJ, CDC definitions of nosocomial surgical site infections, 1992: A modification of CDC definitions of surgical wound infections: Infect Control Hosp Epidemiol, 1992; 13; 606-8
6. Centers for Disease Control and Prevention: Highlighted NHSN January 2013 Patient Safety Component (PSC) Manual Updates https://www.cdc.gov/nhsn/PDFs/Newsletters/January-2013-PSC-Updates.pdf
7. World Health Organization: First global patient safety challenge clean care is safer care, 2009 whqlibdoc.who.int/publications/2009/9789241597906_eng.pdf
8. Allegranzi B, Zayed B, Bischoff P, New WHO recommendations on intraoperative and postoperative measures for surgical site infection prevention: An evidence-based global perspective: Lancet Infect Dis, 2016; 16(12); e288-303
9. Famakinwa TT, Bellow BG, Oyeniran YA, Knowledge and practice of post-operative wound infection prevention among nurses in the surgical unit of teaching hospital in Nigeria: International Journal of Basic, Applied and Innovative Research, 2014; 3(1); 23-28
10. Echols K, Graves M, LeBlanc KG, Role of antiseptics in the prevention of surgical site infections: Dermatol Surg, 2015; 41(6); 667-76
11. Zimlichman E, Henderson D, Tamir O, Health care-associated infections: JAMA Intern Med, 2013; 173(22); 2039-46
12. Health Protection Agency: Surveillance of surgical site infections in NHS hospitals in England. 2010/2012, 2012 https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/364329/SSI_in_orthopaedic_surgery_Apr_2010_to_Mar_2012.pdf
13. Balm M, Andrew Lover A, Salmon S, Progression from new methicillin-resistant Staphylococcus aureus colonisation to infection: An observational study in hospital cohort: MBC Infect Dis, 2013; 13; 491
14. Zywiel MG, Daley JA, Delanois RE, Advance pre-operative chlorhexidine reduces the incidence of surgical site infections in knee arthroplasty: Int Orthop, 2011; 35(7); 1001-6
15. World Health Organization, Global guidelines for the prevention of surgical site infection: Executive Summary, 2018, Geneva https://www.ncbi.nlm.nih.gov/books/NBK536430/
16. Perl TM, Cullen JJ, Wenzel RP, Intranasal mupirocin to prevent postoperative Staphylococcus aureus infections: N Engl J Med, 2002; 346(24); 1871-77
17. World Health Organization: Global guidelines for the prevention of surgical site infection. 4, Evidence-based recommendations on measures for the prevention of surgical site infection, 2016, Geneva https://www.ncbi.nlm.nih.gov/books/NBK401151/
18. Wade RG, Burr NE, McCauley G, The comparative efficacy of chlorhexidine gluconate and povidone-iodine antiseptics for the prevention of infection in clean surgery: A systematic review and network meta-analysis: Ann Surg, 2020 [Online ahead of print]
19. Schmalz G, Galler KM, Biocompatibility of biomaterials – Lessons learned and considerations for the design of novel materials: Dent Mater, 2017; 33(4); 382-93
20. Chen AF, Heyl AE, Xu PZ, Preoperative decolonization was effective at reducing staphylococcal colonization in total joint arthroplasty patients: J Arthroplasty, 2013; 28; 18-20
21. Bebko SP, Green DM, Awad SS, Effect of a preoperative decontamination protocol on surgical site infections in patients undergoing elective orthopedic surgery with hardware implantation: JAMA Surg, 2015; 150(5); 390-95
22. Phillips M, Rosenberg A, Shopsin B, Preventing surgical site infections: A randomized, open-label trial of nasal mupirocin ointment and nasal povidone-iodine solution: Infect Control Hosp Epidemiol, 2014; 35(7); 826-32
23. Rezapoor M, Nicholson T, Tabatabaee RM, Povidone-iodine-based solutions for decolonization of nasal staphylococcus aureus: A randomized, prospective, placebo-controlled study: J Arthroplasty, 2017; 32(9); 2815-19
24. Urias DS, Varghese M, Simunich T, Preoperative decolonization to reduce infections in urgent lower extremity repairs: Eur J Trauma Emerg Surg, 2018; 44(5); 787-93
25. Peng HM, Wang LC, Zhai JL, Effectiveness of preoperative decolonization with nasal povidone iodine in Chinese patients undergoing elective orthopedic surgery: A prospective cross-sectional study: Braz J Med Biol Res, 2017; 51(2); e6736
26. Chen S, Chen JW, Guo B, Preoperative antisepsis with chlorhexidine versus povidone-iodine for the prevention of surgical site infection: A systematic review and meta-analysis: World J Surg, 2020; 44(5); 1412-24
27. Guo S, Li L, Comparison of the effects of chlorhexidine gluconate and povidone iodine on preoperative skin preparation: Chin J Disinfect, 2017; 34(07); 684-86
28. Kulkarni AP, Awode RM, A prospective randomised trial to compare the efficacy of povidone-iodine 10% and chlorhexidine 2% for skin disinfection: Indian J Anaesth, 2013; 57; 270-75
29. Kumar SK, Reddy GV, Naidu G, Role of povidone iodine in periapical surgeries: Hemostyptic and anti-inflammatory?: Ann Maxillofac Surg, 2011; 1; 107-11
30. Arakeri G, Brennan PA, Povidone-iodine: An anti-oedematous agent?: Int J Oral Maxillofac Surg, 2011; 40; 173-76
31. Brown L, Shelly M, Greene LR, The effect of universal intranasal povidone iodine antisepsis on total joint replacement surgical site infections: Am J Infect Control, 2014; 42(6); S81-82
32. Gorwitz RJ, Kruszon-Moran D, McAllister SK, Changes in the prevalence of nasal colonization with Staphylococcus aureus in the United States, 2001–2004: J Infect Dis, 2008; 197; 1226-34
33. Anderson DJ, Kaye KS, Classen D, Strategies to prevent surgical site infections in acute care hospitals: Infect Control Hosp Epidemiol, 2008; 29(Suppl 1); S51-61
34. Gupta K, Strymish J, Abi-Haidar Y: Infect Control Hosp Epidemiol, 2011; 32; 791-96
35. Zarco MF, Vess TJ, Ginsburg GS, The oral microbiome in health and disease and the potential impact on personalised dental medicine: Oral Dis, 2012; 18; 109-20
36. Urban JA, Cost analysis of surgical site infections: Surg Infect (Larchmt), 2006; 7(Suppl 1); S19-22
In Press
07 Mar 2024 : Clinical Research
Knowledge of and Attitudes Toward Clinical Trials: A Questionnaire-Based Study of 179 Male Third- and Fourt...Med Sci Monit In Press; DOI: 10.12659/MSM.943468
08 Mar 2024 : Animal Research
Modification of Experimental Model of Necrotizing Enterocolitis (NEC) in Rat Pups by Single Exposure to Hyp...Med Sci Monit In Press; DOI: 10.12659/MSM.943443
18 Apr 2024 : Clinical Research
Comparative Analysis of Open and Closed Sphincterotomy for the Treatment of Chronic Anal Fissure: Safety an...Med Sci Monit In Press; DOI: 10.12659/MSM.944127
08 Mar 2024 : Laboratory Research
Evaluation of Retentive Strength of 50 Endodontically-Treated Single-Rooted Mandibular Second Premolars Res...Med Sci Monit In Press; DOI: 10.12659/MSM.944110
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952