23 September 2020: Clinical Research
A Study on the Predictors of Disease Severity of COVID-19
Liang Li1ABCDE, Wei Sun1ABCD, Mingfeng Han1BCD, Yunli Ying1CDEF, Quanzhi Wang2AFG*DOI: 10.12659/MSM.927167
Med Sci Monit 2020; 26:e927167
Abstract
BACKGROUND: Early and rapid identification of severe coronavirus disease 2019 (COVID-19) cases is important. The present study aimed to investigate the predictors of disease severity and thus determine the trends for disease progression early.
MATERIAL AND METHODS: Patients with COVID-19 were recruited from Fuyang Second People’s Hospital from January to February 2020. Patients’ demographic, epidemiological, and clinical data were collected, and the relationships between these variables and disease severity were analyzed.
RESULTS: A total of 158 cases were included according to COVID-19 diagnosis, and the treatment schemes were analyzed for identification of early indicators affecting COVID-19 progression. Severe cases accounted for 18.99% of the diagnosed cases. Analysis showed that patients’ age (χ²=10.640,=0.041); the time interval between onset and diagnosis (χ²=7.278, P=0.026); the source of cases (χ²=5.557, P=0.018); fever (χ²=5.676, P=0.014); dyspnea (χ²=113.085, P<0.001); muscle or joint pain (χ²=3.900, P=0.048); chest pain (χ²=13.446, P=0.006); the levels of lymphocytes (t=2.917, P=0.014), C-reactive protein (U=730.00, P<0.001), and aspartate aminotransferase (U=1235.00, P=0.002); damage in both lungs within 3 days of admission (χ²=7.632, P=0.003); and diabetes (χ²=6.675, P=0.010) were significantly correlated with the trend of intensification.
CONCLUSIONS: Older age, a long time interval from onset to diagnosis, imported cases from an affected area, dyspnea, muscle or joint pain, chest pain during the course of the disease, reduced lymphocytes, elevated C-reactive protein, computed tomography scan showing damage to both lungs within 3 days of admission, and diabetes mellitus are predictors for severe COVID-19.
Keywords: COVID-19, Epidemiology, SARS Virus, Adolescent, Age Factors, Betacoronavirus, C-Reactive Protein, COVID-19, Case-Control Studies, Comorbidity, Coronavirus Infections, delayed diagnosis, Pandemics, Pneumonia, Viral, Risk Factors, SARS-CoV-2, Socioeconomic Factors, Symptom Assessment, young adult
Background
In December 2019, coronavirus disease 2019 (COVID-19), caused by a novel coronavirus (severe acute respiratory syndrome coronavirus 2 [SARS-CoV-2]), emerged in Wuhan and subsequently spread rapidly throughout China and the world. COVID-19 was designated a global pandemic by the World Health Organization on March 1, 2020. As of July 15, 2020, a total of 13.11 million COVID-19 cases, including 573 752 deaths (case fatality rate 4.4%), have been reported worldwide [1]. A study on the clinical course and prognosis for severe COVID-19 showed that the fatality rate for these cases was higher than 50% [2]. It is estimated that severe disease occurs in approximately 20% of COVID-19 cases [3]. Current evidence suggests that severe COVID-19 rapidly progresses to acute respiratory distress syndrome over an average of 9 days from the time of symptom onset [4]. Severe COVID-19 cases have been the focus of intense investigation in public health, and early identification of the factors affecting the severity of COVID-19 could contribute to preventing adverse outcomes by allowing timely intervention. At present, many basic, clinical, and epidemiological studies have provided important direct guidance or reference for real-time field disease prevention, control, and treatment for COVID-19. However, early and rapid identification of patients with severe COVID-19 requires more attention.
In the present study, a detailed investigation was performed on the epidemiological and clinical characteristics of 158 patients with COVID-19 who were hospitalized in Fuyang Second People’s Hospital from January to February 2020. This investigation placed particular emphasis on the predictors of disease severity.
Material and Methods
STUDY PARTICIPANTS:
The Fuyang Second People’s Hospital, as the only designated treatment institution for COVID-19 patients in the city, is responsible for the centralized isolation treatment of COVID-19. All patients with COVID-19 admitted to Fuyang Second People’s Hospital from January to February 2020 were included as study subjects. The diagnostic criteria and clinical classification were based on the COVID-19 diagnosis and treatment scheme released by the National Health Commission of the People’s Republic of China [5].
During the COVID-19 outbreak in January to February 2020, all patients with fever, cough, and other symptoms in the city were treated at the fever clinic of the local county hospital or Fuyang Second People’s Hospital. Upper respiratory tract samples (nose swab, throat swab), lower respiratory tract samples (deep cough sputum), and anticoagulant blood samples were collected from patients with suspected COVID-19 and sent to Fuyang Center for Disease Control and Prevention for testing for the novel coronavirus nucleic acid. All patients with confirmed COVID-19 were immediately sent to the Infection Department of Fuyang Second People’s Hospital for isolation and treatment.
DATA COLLECTION AND MANAGEMENT:
A case-control study design was applied in the present study. A structured questionnaire was used to obtain necessary data for each participant. After admission, the resident physicians collected participants’ sociodemographic, epidemiological, and clinical data and provided examinations and treatment. The patients were evaluated and clinically classified according to their disease progression. Patients with severe and critical illness were admitted to the intensive care unit, while those with mild and moderate illness were treated in the general ward. Furthermore, the following data were also collected at the time of discharge: clinical, laboratory, and imaging data as well as information on disease progression. Severe and nonsevere cases were determined upon discharge from the hospital, with patients with severe and critical illness being regarded as the case group, while those with mild and moderate illness were classified as the control group.
In imported cases, the patient had a clear history of travel and residence in the epidemic area (Wuhan) within 14 days before the onset of the disease, was infected in the epidemic area, and became ill after returning to the local area.
STATISTICAL ANALYSIS:
Data were entered using EpiData 3.1 and proofed for entry errors. Statistical analysis was done using SPSS 20.0 statistical software. The collected data are described in terms of frequency, percentage (%), mean, and standard deviation. Categorical variables were compared using χ2 tests, with a
ETHICAL STATEMENT:
The study was approved by the local Medical Ethics Committee of the Fuyang Second People’s Hospital. All subjects gave informed consent to the purpose and methods of the survey and agreed to the use of their biological specimens for the study of prevention, control, and treatment of COVID-19.
Results
EPIDEMIOLOGICAL CHARACTERISTICS OF ALL CASES:
A total of 158 cases were selected in the present study, with 128 (81.01%) mild/ordinary cases in the control group and 30 (18.99%) severe/critical cases in the case group. The sex ratio of males (89 cases) to females (69 cases) was 1.29: 1. The median age of the study population was 43 years, with a range from 1 to 82 years. The interval time from onset to diagnosis varied from 0 to 21 days, with an average of 6.5±3.7 days. The composition of the age group and the interval time from onset to diagnosis are shown in Table 1. Among all cases, imported cases accounted for 50.6% and 49.4% were local infections.
GENERAL CLINICAL CHARACTERISTICS OF ALL CASES:
The top 3 initial symptoms were fever (118, 74.7%), cough (23, 14.6%), and fatigue (7, 4.4%). The most common symptoms in the course of the COVID-19 were fever (86.7%) and cough (81.6%). Other common symptoms included diarrhea/vomiting (39.2%), fatigue (34.8%), dyspnea (21.5%), and sore throat (17.7%). Data from laboratory tests showed that 28.5% of patients had a white cell count below the normal range and 25.9% had a lower than normal lymphocyte count. Additionally, 20.3% had abnormally elevated liver enzymes, and 56.3% had increased concentrations of C-reactive protein (CRP).
All patients had a chest computed tomography (CT) scan. The majority of patients (94.9%) had obvious imaging changes in the lungs. Furthermore, participants with damage in only one lung accounted for 12.0%, whereas those with damage in both lungs accounted for 82.9%.
SOCIODEMOGRAPHIC AND EPIDEMIOLOGICAL CHARACTERISTICS OF PATIENTS:
To detect differences in sociodemographic and epidemiological characteristics between severe and nonsevere cases, we conducted a further analysis, and the findings are presented in Table 2. The results showed that age, the time from onset to diagnosis, and the source of the cases were associated with the intensification of COVID-19, with severe cases being more likely to be among older patients, to be imported, and to have a longer time interval from onset. Considering the possible sociodemographic differences between the imported cases and the local infection cases, we further analyzed the cases from the 2 sources and found a statistical difference in sex ratio between the 2 sources, but no statistical difference for age and time from onset to diagnosis (Table 3).
CLINICAL CHARACTERISTICS OF SEVERE AND NONSEVERE CASES:
Clinical characteristics, including fever, dyspnea, muscle or joint pain, and chest pain, were significantly different between severe and nonsevere cases. In addition, the CT results showed that lung damage within 3 days of admission was a predictor for severe cases (Table 4). With regard to laboratory parameters, the lymphocyte levels in the severe case group were significantly lower than those in the nonsevere case group within 3 days after admission, and the aspartate aminotransferase (AST) and CRP levels were significantly higher than those in the nonsevere case group (Table 5).
CHRONIC DISEASE IN SEVERE AND NONSEVERE CASES:
We further analyzed data on chronic diseases to explore whether any of these diseases represents a risk factor for COVID-19. The prevalence of diabetes mellitus differed significantly between 2 groups, but there was no significant difference for other chronic diseases. All details are presented in Table 6.
Discussion
In this study, we provided a relatively comprehensive estimation for the early identification indicators affecting COVID-19 disease status. In our study, all mild cases tended to have minor symptoms and a good prognosis; in addition, they mostly recovered within 1 to 2 weeks. However, the sudden deterioration of most severe cases appeared within 1 to 2 weeks after admission, with rapid disease progression and high fatality. Therefore, risk assessment at the early stage of disease is important for predicting intensification and enabling effective interventions to improve the cure rate and reduce case fatality rate.
The proportion of severe cases was 18.99% in the current study, which was similar to the national severe disease rate of 19.9% as of February 27, 2020, and lower than that of 22.3% in Chongqing [3]. Relevant studies have shown that older age is a risk factor for severe COVID-19, with 80% of deaths occurring in individuals over 60 years old [2,6]. This association is believed to be related to the weakened immune function in the elderly population [3,7]. However, we found that the rate of severe disease increased with age, suggesting that it was not only related to the weakened immune function of the elderly population, but also to variable susceptibility to the virus in different age groups. The age-related associations need to be further studied.
The time interval from onset to diagnosis is related to the trend in intensification. When COVID-19 is diagnosed early, patients can receive effective treatment as soon as possible, especially antiviral treatment, which can limit disease progression and significantly improve the prognosis [7]. The proportion of severe cases among imported cases was higher than among local cases. Further analysis showed that there were no statistically significant differences in age and time from onset to diagnosis between the 2 sources. These results suggest that the inherent virulence of the virus may change in the process of transmission.
Fever cannot be used as the basis for early identification of severe disease because it is one of the most common symptoms of COVID-19. Symptoms such as dyspnea and chest pain occurred less in the early stage of COVID-19 among control patients, but were more likely among patients with severe COVID-19. Studies have shown that 17% of COVID-19 patients develop acute respiratory distress syndrome, which is the leading cause of death [8]. Patients with severe COVID-19 may experience myocardial damage due to hypoxemia or virulence of the virus and develop symptoms of chest pain [9]. Therefore, great attention should be paid to COVID-19 patients with dyspnea, chest pain, and other symptoms in the early onset.
Lymphocytes are important immune cells because they determine the specificity of the immune response. The reason for the decrease in peripheral lymphocyte count in COVID-19 patients is the excessive consumption of immune cells caused by an overly strong immune response. However, a large number of lymphocytes typically migrate to an inflammatory site during the course of a disease. Studies have shown that SARS-CoV-2 may overconsume the immune cells and reduce the cellular immune function in patients, and the decrease of lymphocytes is an important indicator of the deterioration of COVID-19 patients [10]. Evidence from previous studies suggested that the rate of lymphocyte decline is more rapid in patients with severe disease than that in those with mild or moderate disease [8], which is consistent with the findings of our study. CRP is an important marker of systemic inflammation during infection or tissue damage. Of note, an elevated CRP level and the degree of elevation are closely related to the severity of the disease due to SARS-CoV-2 infection and can cause a systemic inflammatory response [11]. In addition, the CRP level is positively correlated with the severity of acute lung damage [12]. Previous literature has shown that there is no significant change in the level of liver enzymes in patients with severe COVID-19 [13,14]. In our study, although the AST level was higher in the case group than in the control group, it was still within the normal range. Therefore, it appeared to have little significance as a predictor of the trend in disease severity.
Most of the COVID-19 patients had significant changes in the lungs based on chest CT scans, indicating that SARS-CoV-2 infection was strongly associated with lower respiratory tract disease. The range of pulmonary lesions determined the severity of the disease and affected prognosis. COVID-19 patients with multiple lesions in both lungs are more likely to be severe cases, and the changes in the lungs on chest CT scans are more pronounced in patients with severe disease than in those with mild disease [10].
Previous studies have shown that diabetes is an important risk factor for acute exacerbation of lower respiratory tract infection [15]. For COVID-19, around 75% of deaths have one or more chronic diseases, mainly diabetes and hypertension [16,17]. The exact underlying mechanisms between blood glucose levels and infectious diseases are not yet fully understood. It is worth noting that abnormal blood glucose levels can disrupt immunity in diabetic patients, resulting in abnormal immune regulation and a decreased immune response.
Several potential limitations in the current study should be acknowledged. First, the small number of severe cases in this study limits further multifactor analysis, such as multivariate logistic regression analysis, and controlling for confounding factors may not be adequate. Second, the investigation of each case was time intensive and was not completed by the same individual. Consequently, there is a risk of bias in the results.
Conclusions
We provided a relatively comprehensive estimation for the early identification indicators that are closely associated with COVID-19 disease status. We found several predictors for severe COVID-19 cases, including older age, long time interval from onset to diagnosis, imported cases from an affected area, dyspnea, muscle or joint pain, chest pain, reduced lymphocyte, elevated C-reactive protein, damage in both lungs within 3 days of admission, and diabetes. The patients with severe COVID-19 tend to have a high mortality rate, so it is very important to identify the early predictors of severe disease, assess the trend in disease progression, and to take intervention measures.
Tables
Table 1. Basic characteristics of 158 COVID-19 patients.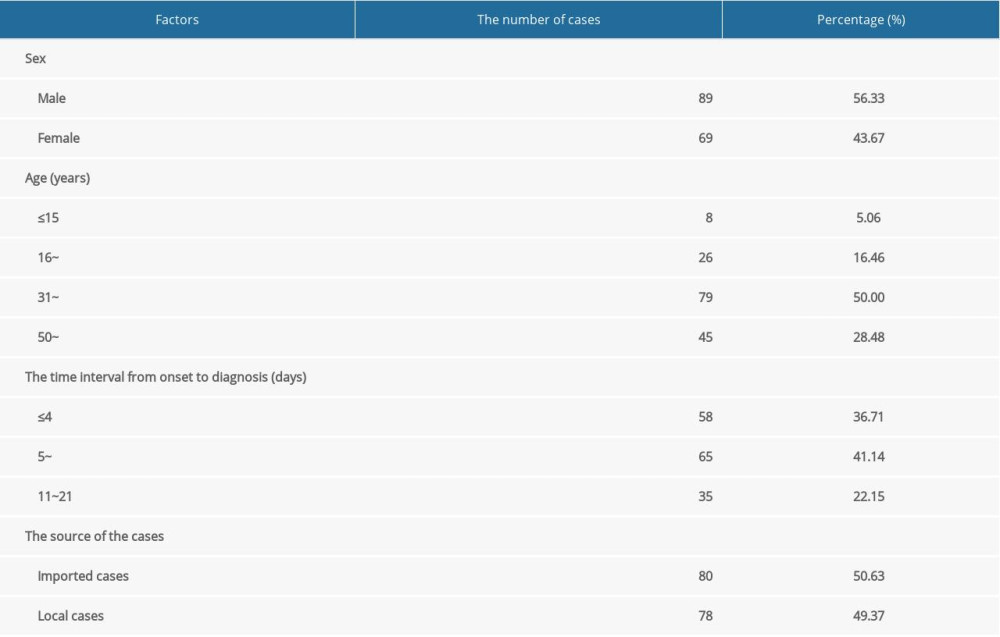 Table 2. The sociodemographic and epidemiological characteristics of severe cases and nonsevere cases.
Table 2. The sociodemographic and epidemiological characteristics of severe cases and nonsevere cases.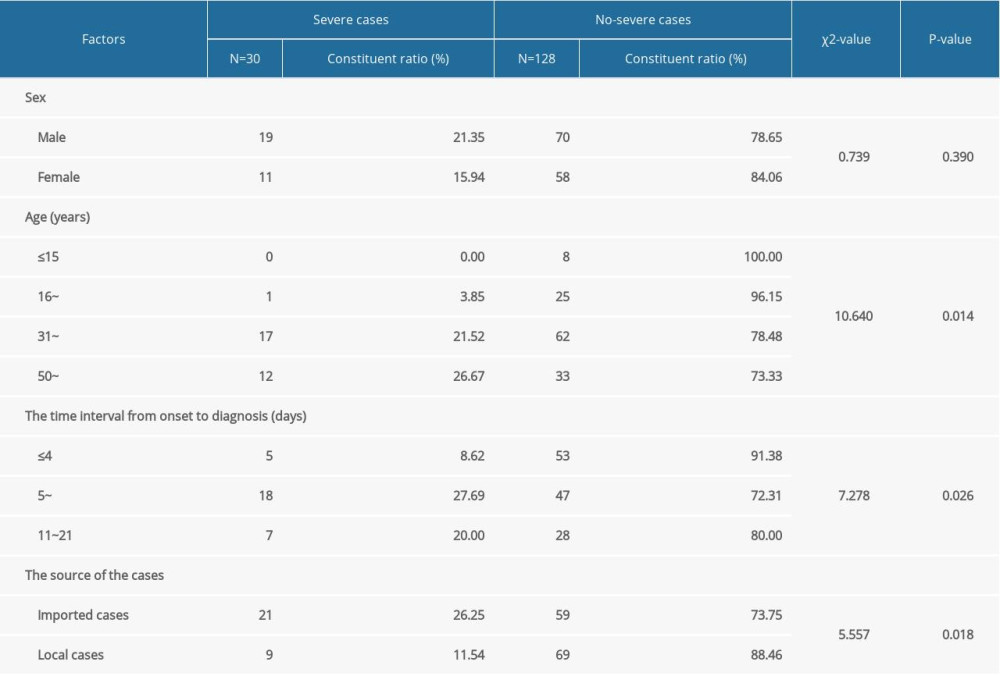 Table 3. The sociodemographic and epidemiological characteristics of imported cases and local cases.
Table 3. The sociodemographic and epidemiological characteristics of imported cases and local cases.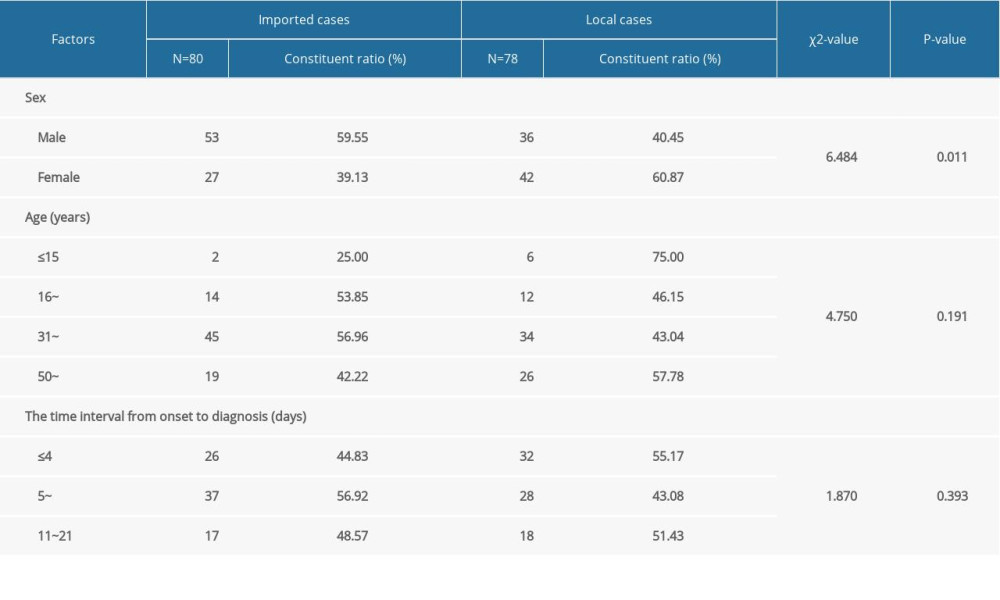 Table 4. The clinical characteristics of severe cases and nonsevere cases.
Table 4. The clinical characteristics of severe cases and nonsevere cases.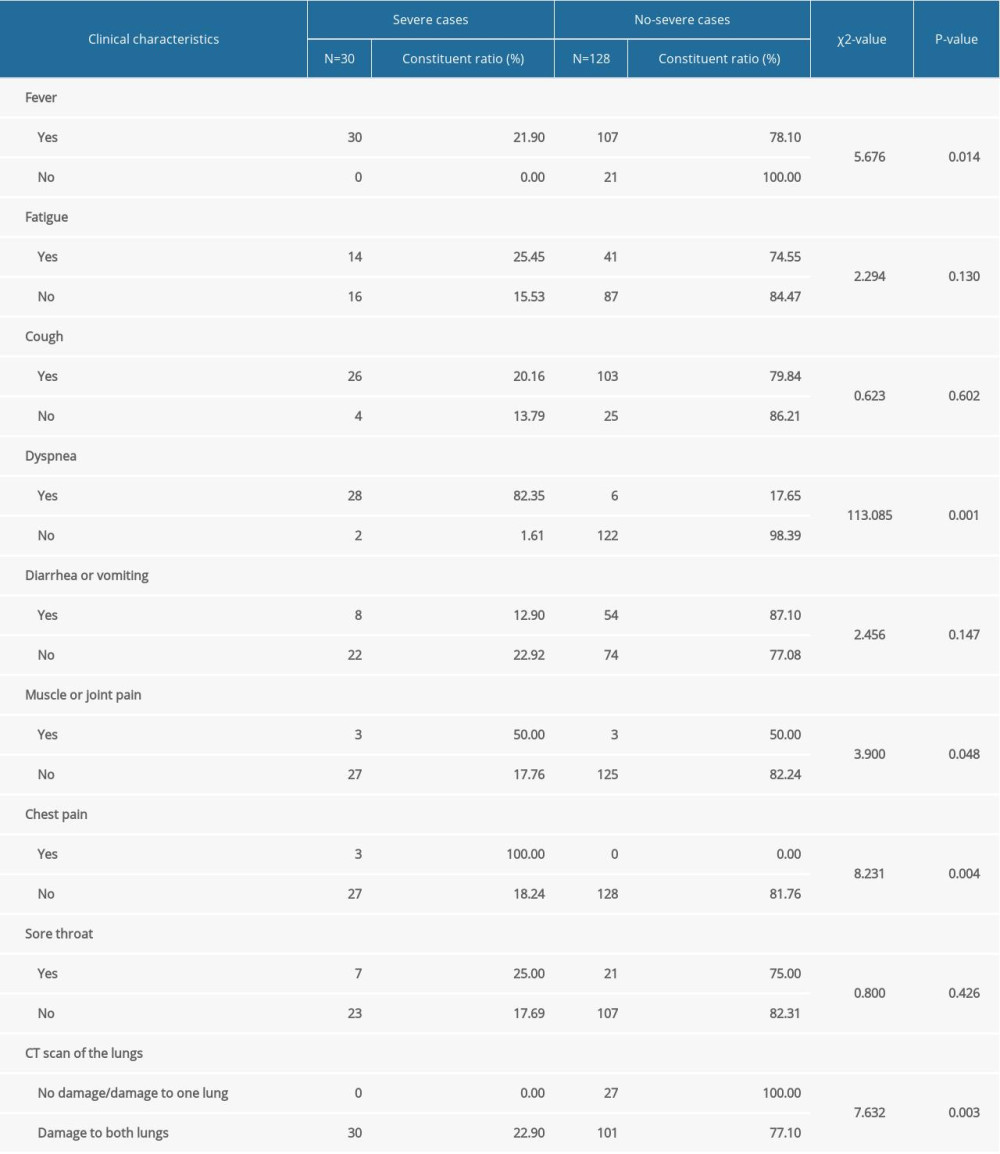 Table 5. Laboratory indices of COVID-19 patients in the severe group and nonsevere group.
Table 5. Laboratory indices of COVID-19 patients in the severe group and nonsevere group.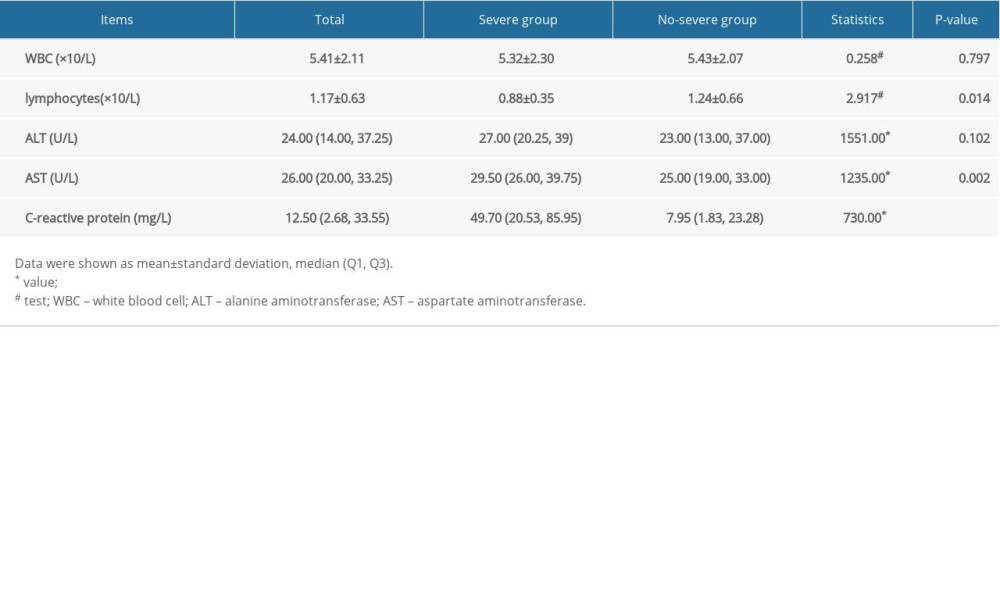 Table 6. The chronic disease of severe cases and nonsevere cases.
Table 6. The chronic disease of severe cases and nonsevere cases.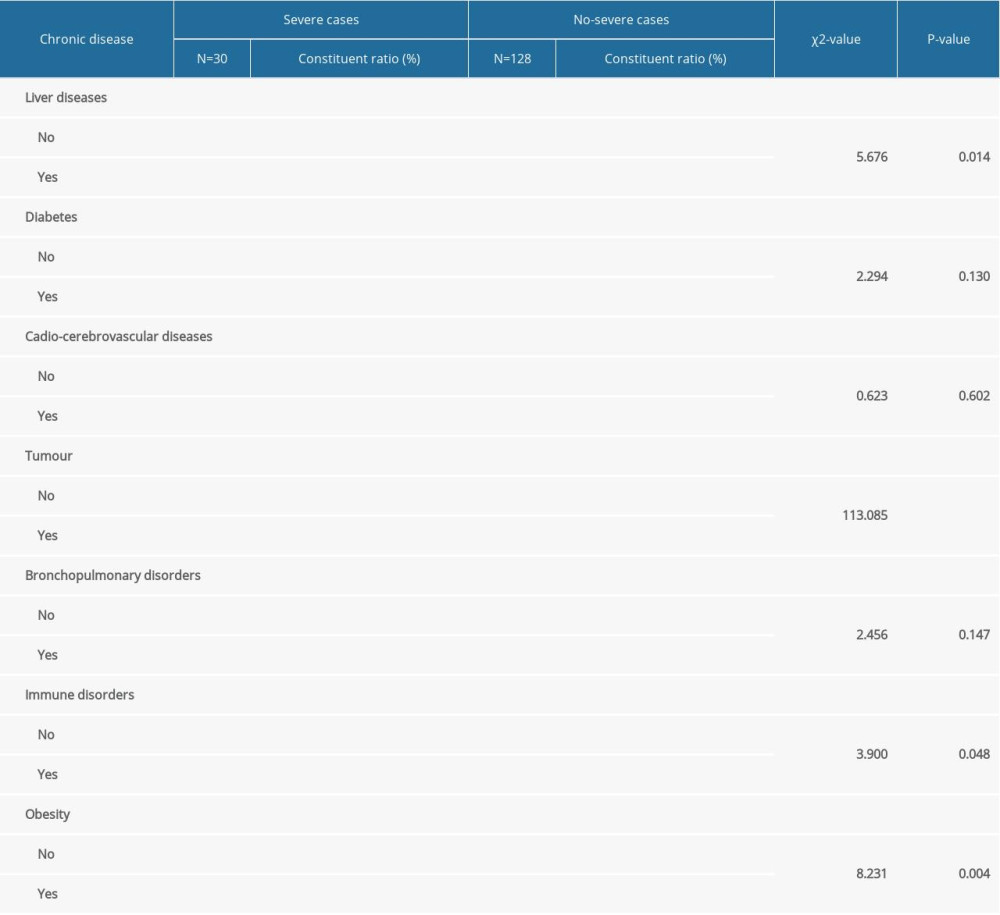
References
1. China News: WHO: More than 13.11 million confirmed cases of new coronary pneumonia worldwide July 15, 2020 http://news.sina.com.cn/w/2020-07-15/doc-iivhvpwx5582121.shtml
2. Yang X, Yu Y, Xu J, Clinical course and outcome of critically ill patients with SARS-CoV-pneumonia in Wuhan, China: A single-centered, retrospective, observational study: Lancet Respir Med, 2020; 8; 475-81
3. Chen X, Tong J, Xiang JHThe epidemiological characteristics of 139 COVID-19 patients on the impact of severe illness: A retrospective study: Chong Qing Medicine, 2020 [in Chinese]
4. Huang C, Wang Y, Li X, Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China: Lancet, 2020; 395(10223); 497-506
5. National Health Commission of the People’ Republic of China, National Administration of Traditional Chinese Medicine, COVID-19 diagnosis and treatment scheme (trial seventh edition): Clinical Education of General Practice, 2020; 18(2); 100-5
6. Analysis of epidemiological characteristics of COVID-19: Chinese J Epidemiol, 2020; 21(2); 145-51 [in Chinese]
7. Wu J, Li W, Shi XW, Early antiviral treatment contributes to alleviate the severity and improve the prognosis of patients with novel coronavirus disease (COVID-19): J Intern Med, 2020; 288(1); 128-38
8. Chen N, Zhou M, Dong X, Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: A descriptive study: Lancet, 2020; 395; 507-13
9. Zhang JG, Ding DYClinical analysis of changes in myocardial enzyme spectrum for severe cases of COVID-19: Journal of Jiangsu University (Medicine Edition), 2020; 30(2); 1-3 [in Chinese]
10. Yan L, Li YS, Identification and management strategies for severe patients of COVID-19: J New Med, 2020; 51(3); 161-65 [in Chinese]
11. Gao L, Liu X, Zhang D, Early diagnosis of bacterial infection in patients with septicopyemia by laboratory analysis of PCT, CRP and IL-6: Exp Ther Med, 2017; 13(6); 3479-83
12. Liu YX, Yang Y, Zhang CClinical and biochemical indicators of lung damage in patients infected with novel coronavirus (2019-ncov): Sci China Life Sci, 2020; 50(3); 258-69 [in Chinese]
13. Klekotka RB, Mizgała E, Król W, The etiology of lower respiratory tract infections in people with diabetes: Pneumonol Alergol Pol, 2015; 83(5); 401-8
14. MacLaren G, Fisher D, Brodie D, Preparing for the most critically ill patients with COVID-19: the potential role of extracorporeal membrane oxygenation: JAMA, 2020 [Online ahead of print]
15. Tong ZD, Tang A, Li KF, Potential presymptomatic transmission of SARS-CoV-2, Zhejiang Province, China: Emerg Infect Dis, 2020; 26(5); 1052-54
16. Liu Y, Yang Y, Zhang C, Clinical and biochemical indexes from 2019-nCoV infected patients linked to viral loads and lung injury: Sci China Life Sci, 2020; 63; 364-74
17. Liu W, Tao ZW, Wang L, Analysis of factors associated with disease outcomes in hospitalized patients with 2019 novel coronavirus disease: Chin Med J, 2020; 133(9); 1032-38
Tables
 Table 1. Basic characteristics of 158 COVID-19 patients.
Table 1. Basic characteristics of 158 COVID-19 patients. Table 2. The sociodemographic and epidemiological characteristics of severe cases and nonsevere cases.
Table 2. The sociodemographic and epidemiological characteristics of severe cases and nonsevere cases. Table 3. The sociodemographic and epidemiological characteristics of imported cases and local cases.
Table 3. The sociodemographic and epidemiological characteristics of imported cases and local cases. Table 4. The clinical characteristics of severe cases and nonsevere cases.
Table 4. The clinical characteristics of severe cases and nonsevere cases. Table 5. Laboratory indices of COVID-19 patients in the severe group and nonsevere group.
Table 5. Laboratory indices of COVID-19 patients in the severe group and nonsevere group. Table 6. The chronic disease of severe cases and nonsevere cases.
Table 6. The chronic disease of severe cases and nonsevere cases. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








