01 February 2021: Clinical Research
Functional Doxorubicin-Loaded Omega-3 Unsaturated Fatty Acids Nanoparticles in Reversing Hepatocellular Carcinoma Multidrug Resistance
Chunlei Wang12ABEFG, Xiaoyan Wei12BDF, Guoliang Shao12ACFG*DOI: 10.12659/MSM.927727
Med Sci Monit 2021; 27:e927727
Abstract
BACKGROUND: This study investigated a nanoparticle drug delivery system to reverse multidrug resistance (MDR) and assessed its anticancer efficacy in hepatocellular carcinoma (HCC).
MATERIAL AND METHODS: Docosahexaenoic acid (DHA) was used as the functional excipient and doxorubicin (DOX) as the chemotherapeutic drug to synthesize DOX nanoparticles (DOX-nano). The human HCC cell line HepG2 was used for experiments. HepG2/DOX, HepG2+DOX, HepG2+DOX-nano, HepG2/DOX+DOX, and HepG2/DOX+DOX-nano groups cells were treated with DOX or DOX-nano (5 μg/mL). Nude mice bearing a HepG2/DOX xenograft were divided into model, DOX, vector-nano, and DOX-nano groups and injected with saline, DOX reagent, vector-nano, and DOX-nano (2 mg/kg), respectively. Next, cytotoxicity, cellular uptake, cell apoptosis and migration, fluorescence imaging, TUNEL assay, and tumor inhibition effects were assessed in vitro and in vivo. Furthermore, expression of MDR-related proteins was also detected using western blotting.
RESULTS: Fluorescence imaging showed that the DOX uptake in the DOX-nano-treated group was the strongest in the HCC cells or tumors. Cell apoptosis was significantly increased in DOX-nano-treated HepG2/DOX cells and tumors, and cell migration was significantly inhibited in the DOX-nano-treated HepG2/DOX cells compared with the other groups. The tumor inhibitory rate in DOX-nano-injected tumors was also significantly higher than in other groups. The expression of breast cancer resistance protein, B-cell lymphoma 2, lung resistance protein, multidrug resistance protein, and protein kinase C alpha was significantly decreased in DOX-nano-treated HepG2/DOX cells and xenograft tumors. Significantly better antitumor and MDR-reversing effects were also observed in the HepG2+DOX group compared with the HepG2/DOX group.
CONCLUSIONS: This study revealed the potential efficacy of a DOX-nano drug delivery system for the treatment of HCC, using HepG2/DOX cells and nude mice bearing HepG2/DOX xenografts.
Keywords: Carcinoma, Hepatocellular, Docosahexaenoic Acids, Nanoparticles, Tuberculosis, Multidrug-Resistant, Antibiotics, Antineoplastic, Drug Carriers, Drug Delivery Systems, Drug Resistance, Multiple, Fatty Acids, Omega-3, Hep G2 Cells, Liver Neoplasms
Background
Liver cancer is one of the most common malignant tumors, and approximately 75–85% of the cases are hepatocellular carcinoma (HCC). Over the past few decades, morbidity and mortality related to liver cancer have sharply increased worldwide, representing a major public health problem. According to the Global Cancer Statistics, liver cancer was the sixth most commonly diagnosed cancer and the fourth leading cause of cancer death worldwide in 2018, with about 841,000 new cases and 782,000 deaths annually [1]. The currently available options for cancer treatment mainly rely on surgery, chemotherapy, and radiotherapy. Breakthroughs in chemotherapeutic drugs and regimens have contributed to the improved prognosis and extension of survival time for cancer patients, but multidrug-resistant cancer and adverse effects caused by chemotherapeutic agents such as doxorubicin (DOX) remain as nonnegligible obstacles in chemotherapy [2]. Multidrug resistance (MDR) is a self-protective response of tumor cells against drug attack, and it can significantly reduce or abolish the efficiency of cancer chemotherapy. MDR is a complex phenomenon caused by multiple resistance mechanisms in cancer cells, including apoptosis defects, drug uptake reduction, drug efflux increase, and inactivation by drug-metabolizing enzymes [3]. Activated multispecific drug efflux transporters, including MDR1, P-glycoprotein (P-GP), multidrug resistance protein (MRP), lung resistance protein (LRP), and breast cancer resistance protein (BCRP), also contribute to MDR [4]. They are located in the cell membrane and can intercept drugs, thus resulting in insufficient drug concentrations in cellular target structures and weakening the cytotoxicity.
Recently, nano-based strategies have attracted research attention for their ability to reverse MDR. The nanoparticle drug delivery system encapsulates drugs and thus bypasses MDR-related drug efflux transporters. This antagonizes and counteracts the effect of drug efflux in tumor cells and diminishes off-target toxicity to healthy tissues, while improving the bioavailability, delivery, selectivity, and efficacy of the chemotherapeutic drugs in target cancer cells [5]. Newly emerging technologies, including microfluidic platforms, are also used for nanoparticle preparation, with the advantages of miniaturation, stability, accuracy, improved polydispersity, and control of drug loading and release [6]. Accumulating studies also report the efficacy of nanomedicines in MDR reversal and antitumor effects in various cancers, including HCC [7,8]. Moreover, functional excipients with MDR-reversing effects combined with nanocarriers could enhance the reversal of MDR in cancer [9]. Therefore, for the use of nanoparticles in cancer chemotherapy, developing safe and functional excipients, particularly ones with a natural origin, is also important [10].
Docosahexaenoic acid (DHA) is a type of ω-3 unsaturated fatty acid that is abundant in deep-sea fish. This dietary fatty acid selectively concentrates in human retinal and neuronal cell membranes, which is consistent with its well-accepted role in neuroprotection [11,12]. In the absence of DHA, brain cell membranes fail to form and develop, and information transmission between the brain cells is impeded. Some studies have also shown a potential antitumor effect of DHA in cancer. A population-based prospective cohort study of 90,296 participants revealed that the consumption of fish rich in ω-3 fatty acids or n-3 fatty acids, particularly DHA, was inversely associated with HCC in a dose-dependent manner [13]. In addition, Gelsomino et al [14] found that DHA could chemosensitize multidrug-resistant colon cancer cells, which suggested an MDR-reversing role of DHA. Hence, in this study, we used DOX as the chemotherapeutic drug and DHA as a functional excipient to synthesize DOX-loaded DHA nanoparticles (DOX-nano), and we investigated the effect of these nanoparticles in reversing MDR and their anticancer efficacy in HCC. Both in vitro and in vivo experiments were performed to explore the potential of these nanoparticles for the effective treatment of multidrug-resistant HCC.
Material and Methods
CELL CULTURE AND INTERVENTION:
Human HCC cell line HepG2 cells were purchased from Shanghai YaJi Biological Ltd. (catalog number: iCell-h092, China), and multidrug-resistant cell line HepG2/DOX cells were purchased from ATCC (catalog number: YB-ATCC-8552, USA). Cells were cultured in Dulbecco’s modified Eagle’s medium containing 10% fetal bovine serum, 100 U/mL penicillin, and 100 μg/mL streptomycin in a humidified 5% CO2 incubator at 37°C.
PREPARATION OF DOX-LOADED NANOPARTICLES:
DOX-nano and drug-free vector nanoparticles were prepared using a high-pressure microfluidic technique [15]. Specially, the oil phase was composed of DHA (50 mg), Tween 80 (100 mg), octadecenic acid (10 mg), DOX (5 mg), and docosanoic acid (100 mg). Soybean phospholipids and 1,2-distearoyl-sn-glycero-3-phosphoethanolamine-N-[amino(polyethylene glycol)-2000 (DSPE-PEG2000) were added as surfactants, with DSPE-PEG2000-folate and glycerol as cosurfactants. The water phase was 10 mL of double-distilled water containing 4 mg of EDTA. The oil phase mixture was heated to dissolve, under high-speed shear mixing at 2000 rpm. The water phase was then added into the emulsion and sheared to mix evenly, and the emulsion underwent 8 cycles of a homogenizing regimen at 800 bar. Finally, the emulsion pH was adjusted to neutral, and filtered with a 0.45-μm cellulose acetate filter. The vector nanoparticles were prepared using the aforementioned processes without DOX being added. The resultant nanoparticles were kept under nitrogen-filled seal for subsequent experiments.
IN VITRO CYTOTOXICITY ASSAY OF DOX-NANO ON HEPG2/DOX CELLS:
The MTT assay was used to evaluate the cytotoxicity of DOX-nano on HepG2/DOX cells. HepG2/DOX cells (5×104/mL) were seeded in a 96-well plate and cultured for 24 h. The cells were then treated with 100 μL of DOX-nano reagent at different concentrations (1, 2, 4, 8, and 16 μg/mL) for 4 h. HepG2/DOX cells without DOX-nano administration were used for the control. Four hours later, 20 μL of MTT solution (5 mg/mL) was added to each well for another 4 h of co-incubation. The absorbance was measured using a microplate reader at 450 nm, and the IC50 value was calculated to choose the appropriate concentration for subsequent in vitro experiments.
EXPERIMENTAL GROUPS AND CELLULAR UPTAKE OF DOX-NANO:
For the in vitro experiments, HepG2 or HepG2/DOX cells were divided into 5 groups: HepG2/DOX group (HepG2/DOX cells without any treatment), HepG2+DOX group (HepG2 cells incubated with 5 μg/mL DOX), HepG2+DOX-nano group (HepG2 cells incubated with 5 μg/mL DOX-nano), HepG2/DOX+DOX group (HepG2/DOX cells incubated with 5 μg/mL DOX), and HepG2/DOX+DOX-nano group (HepG2/DOX cells incubated with 5 μg/mL DOX-nano). Briefly, these HepG2 or HepG2/DOX cells were seeded in 12-well plates and treated with the corresponding DOX reagent or DOX-nano at a concentration of 5 μg/mL. After incubation at 2, 8, or 24 h, cells lysed and the fluorescence intensity of DOX was detected using a fluorescence microscope (AE2000, Motic, China).
CELL APOPTOSIS ASSAY:
Cell apoptosis was detected after annexin V and fluorescein isothiocyanate/propidium iodide (FITC/PI) double staining. After the cells were co-incubated with DOX or DOX-nano for 24 h, they were centrifuged and resuspended in binding buffer. Then, 10 μL of annexin V-FITC and 5 μL PI reagent were added to the suspension, and cells were stained in the dark for 15 min. Cell apoptosis was analyzed using flow cytometry (Beckman Coulter, Fullerton, CA, USA), and the cell apoptosis rate was calculated using ImageJ software.
WOUND-HEALING ASSAY:
Cell migration was determined using the wound-healing assay. Briefly, HepG2 cells (1×105/mL) and HepG2/DOX cells (5×104/mL) were seeded in 6-well plates and then treated with the indicated reagent or nanoparticle reagent for 4 h. Subsequently, a line was scratched in the middle of the dish with a small pipette tip, and cells were incubated for 24 h. Pictures of the scratches were photographed at 0 and 24 h, respectively. Then, the relative cell migration distances were analyzed using ImageJ software.
HEPG2/DOX TUMOR XENOGRAFT ANIMAL MODEL:
Four-week-old BALB/c nude mice weighing 18±2 g were purchased from the Shanghai SIPPR-BK Laboratory Animal Co. Ltd. (certificate no. SCXK (Hu)2018-0006; Shanghai, China). The animal experiments were approved by the Ethics Committee of Zhejiang Traditional Chinese Medicine University (certificate no. SYXK (Zhe)2013-0184; Hangzhou, China). All animal procedures were in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals. One hundred microliters of phosphate-buffered saline containing 5×106 HepG2/DOX cells was injected into the right flank of the mice to induce a multidrug-resistant tumor xenograft animal model. After 1 week, the mice were randomly divided into 4 groups, with 6 mice in each group. Model group mice received saline, DOX group mice were injected with DOX reagent at 2 mg/kg, vector-nano group mice were injected with vector nanoparticles at 2 mg/kg, and DOX-nano group mice were injected with DOX-nano at 2 mg/kg, respectively. Each group received an injection via the tail vein every 3 days for 10 days. The body weight and tumor growth of the nude mice were recorded. Tumor volume was calculated according to the formula V=L×W2, with L and W representing the longest and shortest dimensions, respectively.
IN VIVO FLUORESCENCE IMAGING:
In vivo fluorescence imaging was performed to investigate the biodistribution of nanoparticles in HepG2/DOX tumor-bearing mice. After the tumor reached about 400 mm3, mice were injected intravenously with the near-infrared fluorescence probe-cy5.5-labeled nanoparticles. The fluorescence was recorded with a small animal in vivo fluorescence imaging system (In-Vivo Imaging System FX Pro, Carestream Health, Inc., New Haven, CT, USA) at the excitation wavelength of 670 nm.
TUNEL ASSAY:
During the 10 days of tail vein injections, the body weight and tumor growth of the nude mice were recorded. After the 10-day period, all mice were killed, and the tumors were removed, weighed, and photographed. The tumor inhibitory rate was calculated according to the following formula: tumor inhibitory rate=100%×(tumor weightmodel group–tumor weighttreatment group)/tumor weightmodel group. Furthermore, cell apoptosis in the tumors was detected by the terminal deoxynucleotidyl transferase dUTP end labeling (TUNEL) assay using an in situ cell death detection kit (Sangon Biotech, Shanghai, China). Tumor tissue sections were fixed, deparaffinized, rehydrated, and further stained with the TUNEL mixture for 1 h in a dark humidified chamber at 37°C, following the manufacturer’s instructions. Positive cells were observed under a light microscope (Leica, Germany).
WESTERN BLOTTING:
The relative protein expressions of MRP, LRP, BCRP, Bcl2, and PKC-α in HepG2 and HepG2/DOX cells and tumor tissues were detected by western blotting. Total proteins in cells or tumor tissues were extracted by RIPA buffer (Beyotime Biotechnology, Shanghai, China), and then the protein concentration was detected using a bicinchoninic acid (BCA) assay kit (Solarbio, USA). After 20-μg protein samples were separated on a 10% SDS-PAGE gel and transferred onto polyvinylidene fluoride (PVDF) membranes, the membranes were blocked with 5% nonfat milk and washed with Tris-buffered saline containing Tween 20 (TBST). The membranes were incubated with the primary antibodies against MRP (DF8801, Affinity), LRP1 (ab92544, Abcam), BCRP (ab207732, Abcam), Bcl2 (ab196495, Abcam), PKC-α (ab32376, Abcam), and β-actin (ab8226, Abcam) overnight at 4°C. The membranes were then washed with TBST and further incubated with the horseradish peroxidase-conjugated secondary antibody for 1.5 h at room temperature. Finally, the protein bands were exposed to the chemiluminescent reagent (enhanced chemiluminescence) for 3 min and captured with the Chemi Capture software.
STATISTICAL ANALYSIS:
Data are expressed as mean±standard deviation, and statistical analysis was performed using SPSS version 18.0 software. Comparisons between the 2 groups were analyzed with the 2-tailed
Results
EFFECT OF DOX-NANO ON HEPG2/DOX CELL CYTOTOXICITY:
The cytotoxicity of DOX-nano was assessed by MTT assay in HepG2/DOX cells. The results shown in Figure 1 suggest that DOX-nano was cytotoxic in a dose-dependent manner. Cytotoxicity was very low at 1 and 2 μg/mL of DOX-nano, but it progressively increased with concentrations of 4, 8, and 16 μg/mL. The IC50 concentration of DOX-nano in HepG2/DOX cells was 6.03 μg/mL, and thus 5 μg/mL was the dose chosen for the subsequent experiment.
CELLULAR UPTAKE OF DOX IN HEPG2 OR HEPG2/DOX CELLS:
The in vitro uptake of DOX in HepG2 or HepG2/DOX cells was evaluated after 2, 8, and 24 h of incubation (Figure 2). The fluorescence intensities of DOX were stronger in DOX-nano-treated groups cells (HepG2+DOX-nano, HepG2/DOX+DOX-nano) than in DOX reagent-treated group cells (HepG2+DOX, HepG2/DOX+DOX), indicating a stronger cellular uptake of DOX-nano compared with DOX only. The release of DOX was also gradually increased with the time of treatment from 2 h to 8 h and 24 h, and a longer treatment time was associated with stronger fluorescence intensities of DOX.
IN VITRO ANTICANCER EFFICACY OF DOX NANOPARTICLES:
The effect of DOX-nano on apoptosis in HepG2/DOX cells was detected by annexin V-FITC/PI staining. The results in Figure 3 show that, compared with HepG2/DOX cells, the cell apoptosis rates in the HepG2+DOX, HepG2+DOX-nano, HepG2/DOX+DOX, and HepG2/DOX+DOX-nano groups were significantly increased (P<0.01, P<0.01, P<0.05, P<0.01, respectively). Furthermore, the cell apoptosis rate in the HepG2/DOX+DOX-nano group was the highest among these 5 groups, and it was also significantly higher than the rate in the HepG2/DOX+DOX group (P<0.01). The effect of DOX-nano on HepG2/DOX cell migration was detected by the wound-healing assay. As indicated by the results in Figure 4, after 24 h, the relative cell migration distances in the HepG2+DOX, HepG2+DOX-nano, HepG2/DOX+DOX, and HepG2/DOX+DOX-nano groups were all significantly lower than the distance in the HepG2/DOX group (P<0.01, P<0.01, P<0.05, P<0.01, respectively). More importantly, the cell migration distance in the HepG2/DOX+DOX-nano group was significantly shorter than the distances in the HepG2+DOX-nano and HepG2/DOX+DOX groups (P<0.01), showing that DOX-nano had a significantly stronger antimigration effect compared with DOX in HepG2/DOX cells, and DOX-nano exhibited a better antimigration effect in HepG2/DOX cells than in HepG2 cells.
EXPRESSION OF MDR-RELATED PROTEINS IN HEPG2/DOX CELLS:
The expression of MDR-related proteins, including MRP, LRP, BCRP, Bcl-2, and PKC-α, in HepG2 or HepG2/DOX cells was detected using western blotting, and the results are shown in Figure 5. The relative protein expression of MRP, LRP, BCRP, Bcl-2, and PKC-α in the HepG2+DOX, HepG2+DOX-nano, HepG2/DOX+DOX, and HepG2/DOX+DOX-nano groups were significantly suppressed compared with the HepG2/DOX group (P<0.05 or P<0.01). Furthermore, the relative protein expression of MRP, LRP, BCRP, Bcl-2, and PKC-α in the HepG2/DOX+DOX-nano group was significantly higher compared with the HepG2+DOX-nano group (P<0.01). The relative protein expression of these drug efflux transporters was lowest in the HepG2+DOX-nano group, and it seemed that the normal HepG2 cells did not express MDR-related proteins.
DOX UPTAKE IN THE TUMOR OF NUDE MICE BEARING HEPG2/DOX XENOGRAFT:
In vivo fluorescence imaging was performed to evaluate the uptake of DOX in tumors of nude mice with HepG2/DOX xenografts (Figure 6A). At 10 days after injection, DOX was mainly accumulated in the tumors of the mice, and the fluorescence of the tumor sites maintained a higher intensity in the DOX-nano group mice, followed by the DOX group, the model group, and the vector-nano group with weak fluorescence in the tumor site.
IN VIVO ANTI-APOPTOTIC AND ANTITUMOR PROPERTIES OF DOX-NANO:
The TUNEL assay was performed to detect the effect of DOX on cell apoptosis in tumor tissues among the different treatment groups (Figure 6B). The results showed that compared with the weakly positive cells in the model and vector groups, the TUNEL-positive apoptotic cells were highly accumulated in the DOX group and DOX-nano group tumor tissues, indicating a promoter effect of DOX in tumor cell apoptosis. The anticancer efficacy of DOX-nano was also investigated in nude mice bearing HepG2 xenografts (Figure 7). The results showed that the weight of the tumor tissues in the DOX-nano group were significantly lighter than in the model group, the DOX group, and the vector-nano group (P<0.01, P<0.05, P<0.01, respectively). The tumor inhibitory rate in the DOX-nano group was also significantly higher than in the DOX group and the vector-nano group (P<0.05, P<0.01, respectively).
EXPRESSION OF MDR-RELATED PROTEINS IN TUMOR TISSUES:
The expression of MDR-related proteins, including MRP, LRP, BCRP, Bcl-2, and PKC-α, in tumor tissue was detected using western blot assay, and the results are shown in Figure 8. Among these MDR tumors, the relative protein expression of MRP, LRP, BCRP, Bcl-2, and PKC-α were highest in the model group. But the expression in the DOX group, vector-nano group, and DOX-nano group were significantly suppressed compared with the model group (P<0.05 or P<0.01). More importantly, the relative protein expression of MRP, LRP, BCRP, Bcl-2, and PKC-α in the DOX-nano group was significantly lower compared with the DOX group and the vector-nano group (P<0.05 or P<0.01).
Discussion
Chemotherapy is a mainstay treatment for HCC, but MDR has emerged as the most important reason for chemotherapy failure [16]. Recently, nano-delivery of anticancer agents has been developed to optimize the routes of chemotherapy drug administration. It bypasses multispecific drug efflux transporters and delivers agents to tumor tissues by passive or active tumor-targeting strategies, and it thus contributes to the reversal of MDR [17]. In this study, we used DHA, a natural product with a marine origin, as a functional excipient to synthesize DOX-nano. We then investigated the effect of DOX-nano in reversing MDR as well as its anticancer efficacy in HCC. Our in vitro and in vivo experiments in HepG2/DOX cells and nude mice bearing HepG2/DOX xenograft revealed that the nanoparticles have a strong potential for effectively treating HCC.
The complexity of MDR mechanisms requires synergistic drug use in reversing MDR. This concept has been extended to the synergistic use of pharmaceutical dosage forms, such as the nanoparticle drug delivery system, for the co-delivery of MDR reversal agents and chemotherapeutic agents [18]. Zhao et al [15] showed that DOX and curcumin co-delivery by lipid nanoparticles (DOX/Cur-NPs) could enhance cytotoxicity and decrease IC50 and the MDR index of HCC in mice. In addition, the expression of the MDR-related transporters MDR1, Bcl-2, HIF-1α, and P-GP were also decreased in mice treated with DOX/Cur-NPs, indicating that DOX/Cur-NPs could be used as a potential MDR-reversing and anticancer agent. DOX and pyrrolidinedithiocarbamate co-encapsulated nanoparticles could reverse MDR, improve intracellular drugs accumulation, and enhance tumor penetration in breast cancer [19]. A new combination of salinomycin (SL), which can selectively inhibit multidrug-resistant tumor cells, and the traditional broad-spectrum antitumor drug DOX were used to develop DOX+SL co-loaded nanomicelles. These nanomicelles were able to escape drug efflux and had an enhanced antiproliferative effect in doxorubicin-resistant MCF-7 cells. In addition, strong inhibition of tumor growth, recurrence, and metastasis was also observed [20].
In developing nanocarriers to load chemotherapeutic agents for cancer treatment, some functional excipients added to the nanoparticle drug delivery system have been found to be effective in overcoming MDR in cancers [21]. In developing functional excipients as potential MDR-reversing agents, the advantages of potent, selective, and relatively low toxicity make natural products as possible choices for the design of nanoparticle drug delivery systems in cancer chemotherapy [10]. Tubulin polyglutamylase complex subunit with suppression P-GP function was used as an excipient to synthesize plasminogen-like protein A nanoparticles that could reverse cancer MDR in vitro, and it was also found to improve the antitumor efficacy of the polyene paclitaxel in vivo [22]. In the current study, DHA was chosen as the functional MDR-reversing excipient, and the anticancer drug DOX was encapsulated to form DOX-DHA nanoparticles. The MTT assay confirmed that the IC50 of DOX-nano was 6.03 μg/mL, and thus the dosage of 5 μg/mL was used for further study. In a study focused on reversing P-GP-mediated MDR in MCF-7/ADR cells and HepG2/ADM cells [23], the IC50 value of DOX in HepG2/DOX cells was 102.14±14.34 μg/mL, which was higher than the IC50 value of DOX-DHA nano in the HepG2/DOX cells in our study. This indirectly suggests that the use of DOX-DHA nano could enhance the cytotoxicity of DOX to HepG2/DOX cells. Cellular uptake of the drug is an important index for MDR reversal because higher drug uptake indicates higher drug concentrations at cellular target structures and enhanced cytotoxicity. In vitro and in vivo fluorescence imaging in the present study showed that the drug uptake in DOX-nano-treated group cells and tumors was stronger than in other groups, which indicated that this nanoparticle drug delivery system could enhance drug uptake and inhibit drug efflux, thus overcoming MDR in HCC cells and tumors. The anticancer efficacy of DOX-nano in HepG2 cells and tumor was also assessed based on cell apoptosis, cell migration, and the tumor inhibitory rate.
In addition to the potential for reversing MDR in combination with DOX, DHA itself exhibits antitumor effect in cancers, including HCC. DHA has been reported to selectively kill HCC cells and to reduce the growth of orthotopic liver tumors in rats, and it can also selectively disrupt the redox balance within the cancer cell and lead to tumor-specific necrosis [24]. The results from a study by Zajdel et al [25] showed that DHA could suppress cell viability and cell proliferation, enhance cell death, and induce the activation of caspase-3/7 in SKOV3 and OVCAR3 epithelial ovarian cancer cell lines. More importantly, DHA could significantly potentiate cisplatin cytotoxicity in SKOV3 and OVCAR3 cells. Another study found that DHA has efficacy in chemosensitizing multidrug-resistant colon cancer cells, and it could decrease the amount of P-GP and MRP1 in detergent resistant membranes, inhibit the transporters’ activity, and restore the antitumor effects of different chemotherapeutic drugs [14]. These studies indicate an MDR-reversing and chemosensitivity effect of DHA with chemotherapeutic drugs. In our present study, as an excipient in DOX-nano, DHA was also observed to exhibit antitumor and MDR-reversing effects in HCC HepG2 cells and tumor-bearing nude mice.
Abnormal expression of active drug efflux transporters, including P-GP, MRP, LRP, and BCRP, are also involved in the regulation of MDR [26,27]. The current study also investigated the expression of relative drug efflux transporters in HCC cells and tumors. The results showed that the expression levels of MRP, LRP, BCRP, Bcl-2, and PKC-α were the highest in HepG2/DOX cells among all groups, suggesting the existence of MDR in HepG2/DOX cells. However, in DOX-nano-treated HepG2/DOX cells, significantly decreased expression of MRP, LRP, BCRP, Bcl-2, and PKC-α was found compared with untreated HepG2/DOX cells, which indicated the MDR-reversing efficacy of DOX-nano drug delivery system. Furthermore, in the HepG2+DOX-nano group cells, expression of these proteins was the lowest, which may be due to the shortage of MDR in HepG2 cells and the administration of DOX-nano reversing MDR. In nude mice bearing HepG2/DOX xenografts, the expression of MRP, LRP, BCRP, Bcl-2, and PKC-α was the highest in model group tumors, which indicated that the injection of MDR cell line HepG2/DOX into the right flank of nude mice could induce MDR and activate the expression of drug efflux transporters. These results accorded with those in the HepG2/DOX cells
Conclusions
This study used DHA as a functional excipient and DOX as a chemotherapeutic agent to synthesize DOX-loaded nanoparticles and investigate the effect of this nanoparticle drug delivery system in HCC. In vitro and in vivo experiments revealed that this nanoparticle drug delivery system has strong potential for MDR reversal and anticancer efficacy for the treatment of HCC. Experiments in HepG2/DOX cells and nude mice bearing HepG2/DOX xenograft showed enhanced cytotoxicity, increased cell apoptosis, inhibition of migration, strong inhibition of tumor growth, and suppressed expression of drug efflux transporters. Further in-depth study with larger samples, more types of cell lines, and a systematic experimental design is needed to investigate the role of this DOX-loaded DHA nanoparticle drug delivery system in multidrug-resistant cancers.
Figures
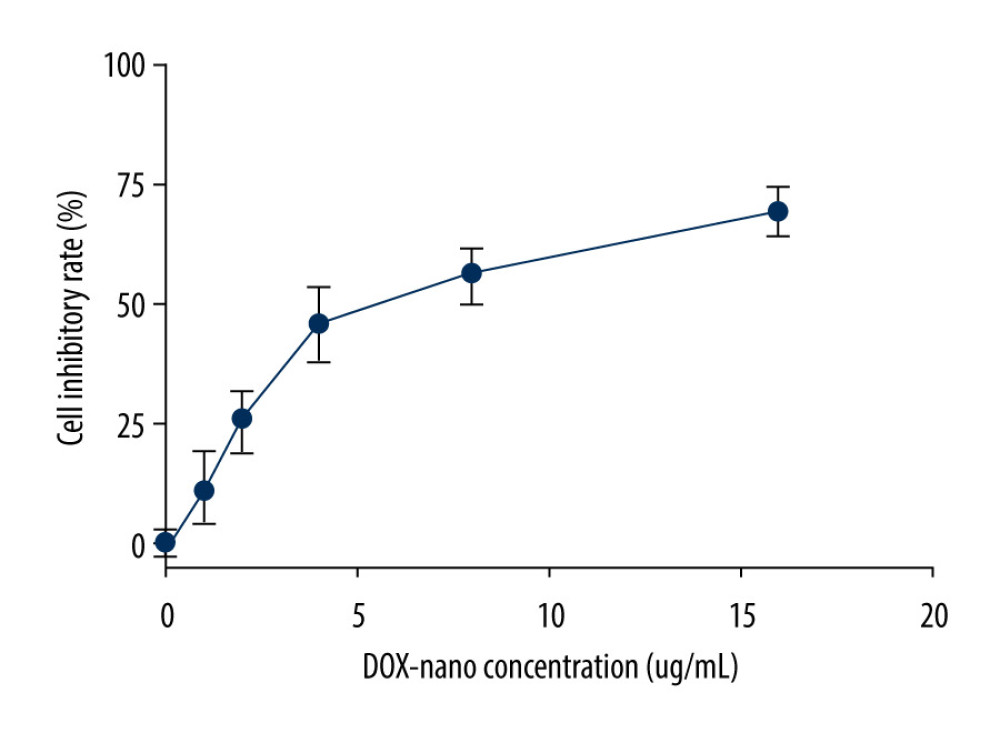 Figure 1. Cytotoxicities of the DOX-nano in HepG2/DOX cells. HepG2/DOX cells were treated with DOX-nano reagent at different dosages (1, 2, 4, 8, 16 μg/mL) for 4 h, and the MTT assay was used to detect the cytotoxicities and IC50 values. DOX – doxorubicin; DOX-nano – doxorubicin-loaded docosahexaenoic acid nanoparticles.
Figure 1. Cytotoxicities of the DOX-nano in HepG2/DOX cells. HepG2/DOX cells were treated with DOX-nano reagent at different dosages (1, 2, 4, 8, 16 μg/mL) for 4 h, and the MTT assay was used to detect the cytotoxicities and IC50 values. DOX – doxorubicin; DOX-nano – doxorubicin-loaded docosahexaenoic acid nanoparticles. 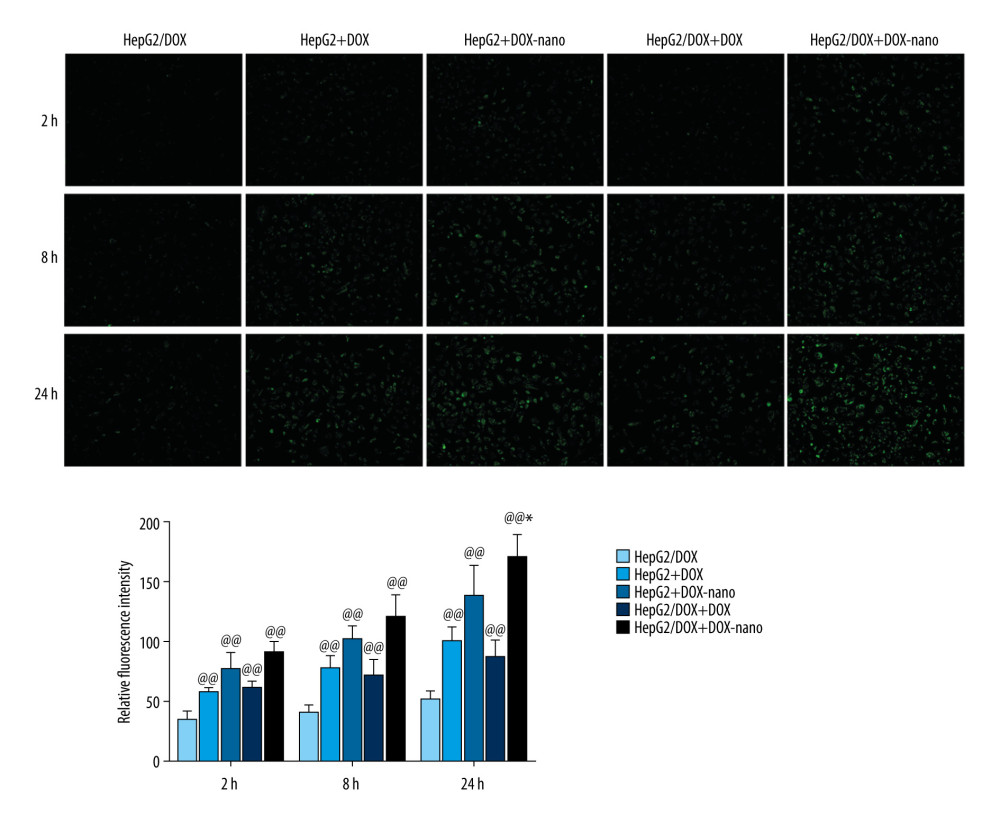 Figure 2. Cellular uptake of DOX-nano. Cells in the HepG2+DOX, HepG2+DOX-nano, HepG2/DOX+DOX, and HepG2/ADM+DOX-nano groups were treated with DOX reagent (5 μg/mL) or DOX-nano (5 μg/mL) for 2, 8, and 24 h after incubation, and fluorescence intensity of DOX was detected. DOX – doxorubicin; DOX-nano – doxorubicin-loaded docosahexaenoic acid nanoparticles.
Figure 2. Cellular uptake of DOX-nano. Cells in the HepG2+DOX, HepG2+DOX-nano, HepG2/DOX+DOX, and HepG2/ADM+DOX-nano groups were treated with DOX reagent (5 μg/mL) or DOX-nano (5 μg/mL) for 2, 8, and 24 h after incubation, and fluorescence intensity of DOX was detected. DOX – doxorubicin; DOX-nano – doxorubicin-loaded docosahexaenoic acid nanoparticles. 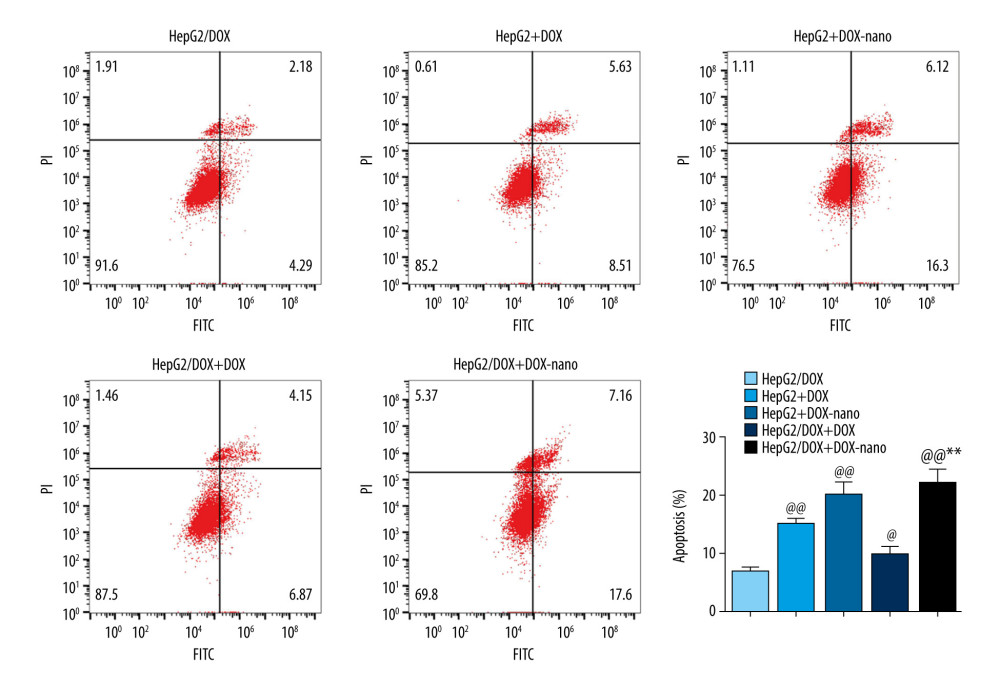 Figure 3. Analysis of apoptotic HepG2 or HepG2/DOX cells using annexin V-fluorescein isothiocyanate/propidium iodide flow cytometry assay. Compared with the HepG2/DOX group, @ P<0.05, @@ P<0.01; compared with the HepG2+DOX-nano group, # P<0.05, ## P<0.01; compared with the HepG2/DOX+DOX group, * P<0.05, ** P<0.01. DOX – doxorubicin; DOX-nano – doxorubicin-loaded docosahexaenoic acid nanoparticles.
Figure 3. Analysis of apoptotic HepG2 or HepG2/DOX cells using annexin V-fluorescein isothiocyanate/propidium iodide flow cytometry assay. Compared with the HepG2/DOX group, @ P<0.05, @@ P<0.01; compared with the HepG2+DOX-nano group, # P<0.05, ## P<0.01; compared with the HepG2/DOX+DOX group, * P<0.05, ** P<0.01. DOX – doxorubicin; DOX-nano – doxorubicin-loaded docosahexaenoic acid nanoparticles.  Figure 4. (A, B) Analysis of HepG2 or HepG2/DOX cells migration. Pictures of the scratches were photographed at 0 and 24 h. Compared with the HepG2/DOX group, @ P<0.05, @@ P<0.01; compared with the HepG2+DOX-nano group, # P<0.05, ## P<0.01; compared with the HepG2/DOX+DOX group, * P<0.05, ** P<0.01. DOX – doxorubicin; DOX-nano – doxorubicin-loaded docosahexaenoic acid nanoparticles.
Figure 4. (A, B) Analysis of HepG2 or HepG2/DOX cells migration. Pictures of the scratches were photographed at 0 and 24 h. Compared with the HepG2/DOX group, @ P<0.05, @@ P<0.01; compared with the HepG2+DOX-nano group, # P<0.05, ## P<0.01; compared with the HepG2/DOX+DOX group, * P<0.05, ** P<0.01. DOX – doxorubicin; DOX-nano – doxorubicin-loaded docosahexaenoic acid nanoparticles. 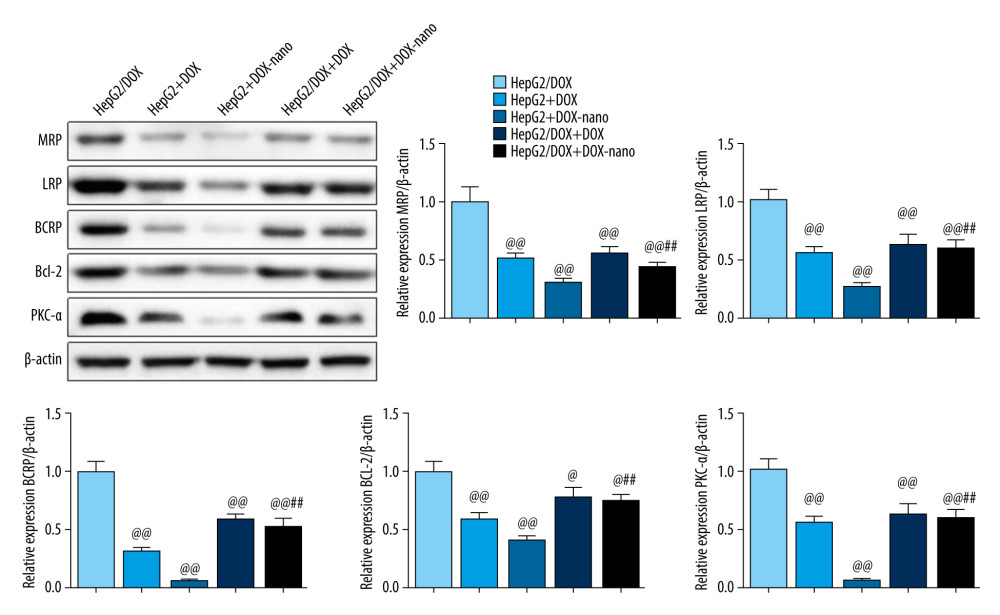 Figure 5. Protein expressions of drug efflux transporters, MRP, LRP, BCRP, Bcl-2, and PKC-α in HepG2 or HepG2/DOX cells. The protein expression was detected using western blotting, and β-actin was used as the internal control. Compared with the HepG2/DOX group, @ P<0.05, @@ P<0.01; compared with the HepG2+DOX-nano group, # P<0.05, ## P<0.01; compared with the HepG2/DOX+DOX group, * P<0.05, ** P<0.01. BCRP – breast cancer resistance protein; Bcl-2 – B-cell lymphoma 2; DOX – doxorubicin; DOX-nano – doxorubicin-loaded docosahexaenoic acid nanoparticles; LRP – lung resistance protein; MRP – multidrug resistance protein; PKC-α – protein kinase C alpha.
Figure 5. Protein expressions of drug efflux transporters, MRP, LRP, BCRP, Bcl-2, and PKC-α in HepG2 or HepG2/DOX cells. The protein expression was detected using western blotting, and β-actin was used as the internal control. Compared with the HepG2/DOX group, @ P<0.05, @@ P<0.01; compared with the HepG2+DOX-nano group, # P<0.05, ## P<0.01; compared with the HepG2/DOX+DOX group, * P<0.05, ** P<0.01. BCRP – breast cancer resistance protein; Bcl-2 – B-cell lymphoma 2; DOX – doxorubicin; DOX-nano – doxorubicin-loaded docosahexaenoic acid nanoparticles; LRP – lung resistance protein; MRP – multidrug resistance protein; PKC-α – protein kinase C alpha. 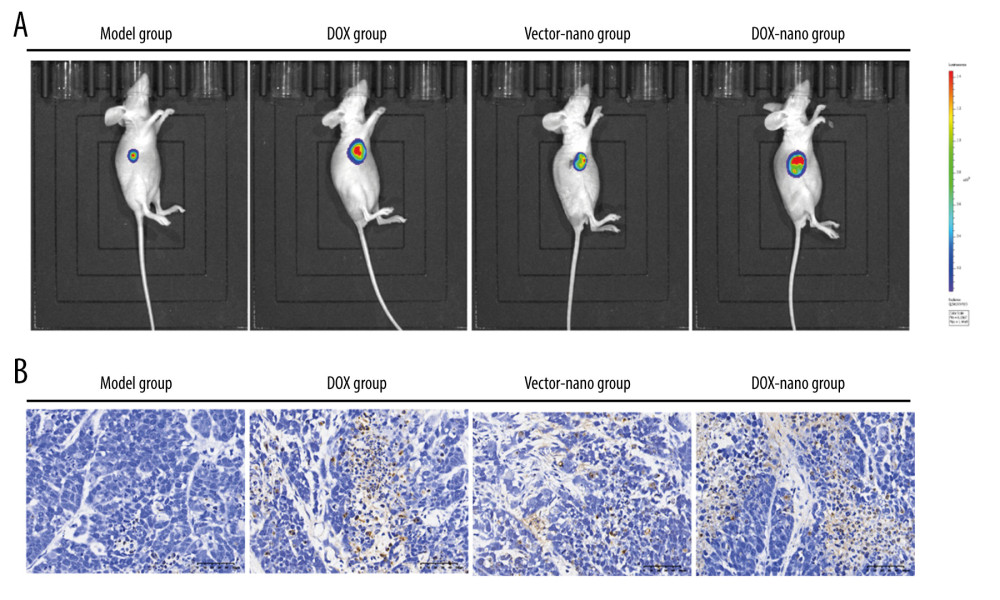 Figure 6. In vivo fluorescence imaging (A) and TUNEL assay (B). In vivo fluorescence imaging showing tumor accumulation of DOX-nano after intravenous injection. TUNEL assay observed cell apoptosis in tumor tissues with DOX-nano injection. DOX-nano – doxorubicin-loaded docosahexaenoic acid nanoparticles.
Figure 6. In vivo fluorescence imaging (A) and TUNEL assay (B). In vivo fluorescence imaging showing tumor accumulation of DOX-nano after intravenous injection. TUNEL assay observed cell apoptosis in tumor tissues with DOX-nano injection. DOX-nano – doxorubicin-loaded docosahexaenoic acid nanoparticles. 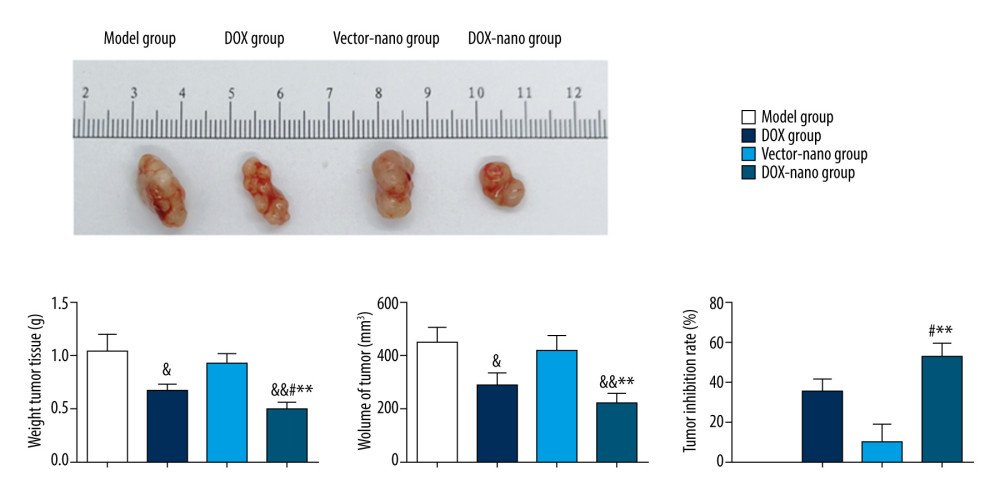 Figure 7. In vivo anticancer efficacy of DOX-nano in nude mice bearing HepG2/DOX xenograft. Compared with the model group, & P<0.05, && P<0.01; compared with the DOX group, # P<0.05, ## P < 0.01; compared with the vector-nano group, * P<0.05, ** P<0.01. DOX – doxorubicin; DOX-nano – doxorubicin-loaded docosahexaenoic acid nanoparticles.
Figure 7. In vivo anticancer efficacy of DOX-nano in nude mice bearing HepG2/DOX xenograft. Compared with the model group, & P<0.05, && P<0.01; compared with the DOX group, # P<0.05, ## P < 0.01; compared with the vector-nano group, * P<0.05, ** P<0.01. DOX – doxorubicin; DOX-nano – doxorubicin-loaded docosahexaenoic acid nanoparticles. 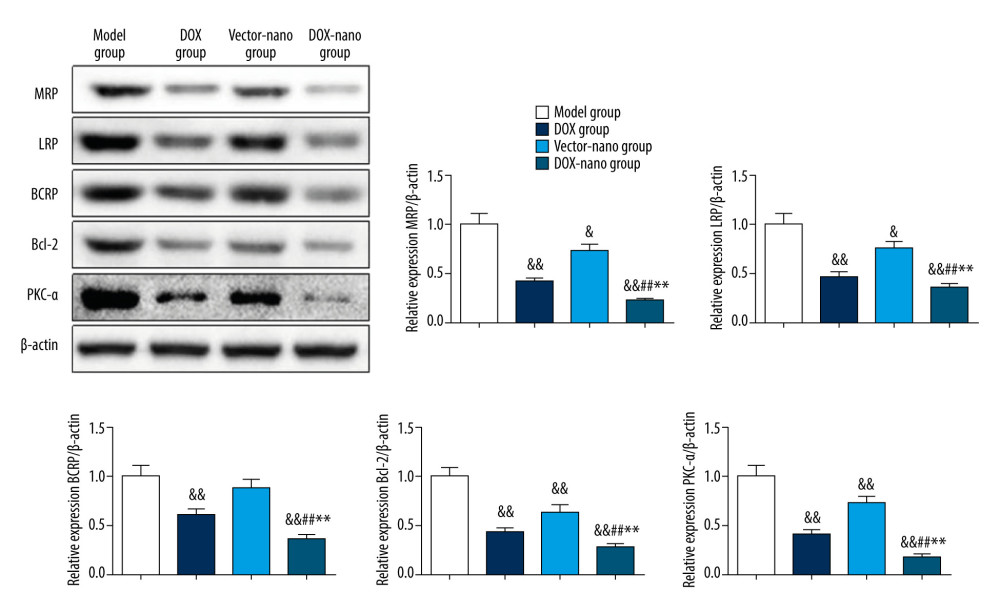 Figure 8. Protein expressions of drug efflux transporters, MRP, LRP, BCRP, Bcl-2, and PKC-α in tumor tissues. The protein expressions were detected using western blotting, and β-actin was used as the internal control. Compared with the model group, & P<0.05, && P<0.01; compared with the DOX group, # P<0.05, ## P<0.01; compared with the vector-nano group, * P<0.05, ** P<0.01. BCRP – breast cancer resistance protein; Bcl-2 – B-cell lymphoma 2; LRP – lung resistance protein; MRP – multidrug resistance protein; PKC-α – protein kinase C alpha.
Figure 8. Protein expressions of drug efflux transporters, MRP, LRP, BCRP, Bcl-2, and PKC-α in tumor tissues. The protein expressions were detected using western blotting, and β-actin was used as the internal control. Compared with the model group, & P<0.05, && P<0.01; compared with the DOX group, # P<0.05, ## P<0.01; compared with the vector-nano group, * P<0.05, ** P<0.01. BCRP – breast cancer resistance protein; Bcl-2 – B-cell lymphoma 2; LRP – lung resistance protein; MRP – multidrug resistance protein; PKC-α – protein kinase C alpha. References
1. Bray F, Ferlay J, Soerjomataram I, Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries: Cancer J Clin, 2018; 68(6); 394-424
2. Nooter K, Stoter G, Molecular mechanisms of multidrug resistance in cancer chemotherapy: Pathol Res Pract, 1996; 192(7); 768-80
3. Chistiakov DA, Myasoedova VA, Orekhov AN, Nanocarriers in improving chemotherapy of multidrug resistant tumors: Key developments and perspectives: Curr Pharm Des, 2017; 23(22); 3301-8
4. Wu CP, Calcagno AM, Ambudkar SV, Reversal of ABC drug transporter-mediated multidrug resistance in cancer cells: Evaluation of current strategies: Curr Mol Pharmacol, 2008; 1(2); 93-105
5. Lepeltier E, Rijo P, Rizzolio F, Nanomedicine to target multidrug resistant tumors: Drug Resist Updat, 2020; 52; 100704
6. Karnik R, Gu F, Basto P, Microfluidic platform for controlled synthesis of polymeric nanoparticles: Nano Lett, 2008; 8(9); 2906-12
7. Liu HY, Zhang ZJ, Chi XQ, Arsenite-loaded nanoparticles inhibit PARP-1 to overcome multidrug resistance in hepatocellular carcinoma cells: Sci Rep, 2016; 6; 31009
8. Lei M, Ma GL, Sha SJ, Dual-functionalized liposome by co-delivery of paclitaxel with sorafenib for synergistic antitumor efficacy and reversion of multidrug resistance: Drug Deliv, 2019; 26(1); 262-72
9. Zhu H, Chen H, Zeng X, Co-delivery of chemotherapeutic drugs with vitamin E TPGS by porous PLGA nanoparticles for enhanced chemotherapy against multi-drug resistance: Biomaterials, 2014; 35; 2391-400
10. Cort A, Ozben T, Natural product modulators to overcome multidrug resistance in cancer: Nutr Cancer, 2015; 67; 411-23
11. Cardoso C, Afonso C, Bandarra NM, Dietary DHA and health: Cognitive function ageing: Nutr Res Rev, 2016; 29(2); 281-94
12. Cardoso C, Afonso C, Bandarra NM, Dietary DHA, bioaccessibility, and neurobehavioral development in children: Crit Rev Food Sci Nutr, 2018; 58(15); 2617-31
13. Sawada N, Inoue M, Iwasaki M, Consumption of n-3 fatty acids and fish reduces risk of hepatocellular carcinoma: Gastroenterology, 2012; 142(7); 1468-75
14. Gelsomino G, Corsetto PA, Campia L, Omega 3 fatty acids chemosensitize multidrug resistant colon cancer cells by down-regulating cholesterol synthesis and altering detergent resistant membranes composition: Mol Cancer, 2013; 12; 137
15. Zhao XJ, Chen Q, Li YS, Doxorubicin and curcumin co-delivery by lipid nanoparticles for enhanced treatment of diethylnitrosamine-induced hepatocellular carcinoma in mice: Eur J Pharm Biopharm, 2015; 93; 27-36
16. Gillet JP, Andersen JB, Madigan JP, A gene expression signature associated with overall survival in patients with hepatocellular carcinoma suggests a new treatment strategy: Mol Pharmacol, 2016; 89(2); 263-72
17. Yuan Y, Cai T, Xia X, Nanoparticle delivery of anticancer drugs overcomes multidrug resistance in breast cancer: Drug Deliv, 2016; 23(9); 3350-57
18. Jabr-Milane LS, van Vlerken LE, Yadav S, Amiji MM, Multi-functional nanocarriers to overcome tumor drug resistance: Cancer Treat Rev, 2008; 34; 592-602
19. Cheng X, Li D, Sun M, Co-delivery of DOX and PDTC by pH-sensitive nanoparticles to overcome multidrug resistance in breast cancer: Colloids Surf B Biointerfaces, 2019; 181; 185-97
20. Cui Y, Yang Y, Ma M, Reductive responsive micelle overcoming multidrug resistance of breast cancer by co-delivery of DOX and specific antibiotic: J Mater Chem B Mater Biol Med, 2019; 7(40); 6075-86
21. Kirtane AR, Kalscheuer SM, Panyam J, Exploiting nanotechnology to overcome tumor drug resistance: Challenges and opportunities: Adv Drug Deliv Rev, 2013; 65(13–14); 1731-47
22. Zhu H, Chen H, Zeng X, Co-delivery of chemotherapeutic drugs with vitamin E TPGS by porous PLGA nanoparticles for enhanced chemotherapy against multi-drug resistance: Biomaterials, 2014; 35; 2391-400
23. Ye RP, Chen ZD, Saikosaponin A, an active glycoside from Radix bupleuri, reverses P-glycoprotein-mediated multidrug resistance in MCF-7/ADR cells and HepG2/ADM cells: Xenobiotica, 2017; 47(2); 176-84
24. Wen XD, Reynolds L, Mulik RS, Hepatic arterial infusion of low-density lipoprotein docosahexaenoic acid nanoparticles selectively disrupts redox balance in hepatoma cells and reduces growth of orthotopic liver tumors in rats: Gastroenterology, 2016; 150(2); 488-98
25. Zajdel A, Kałucka M, Chodurek E, Wilczok A, DHA but not AA enhances cisplatin cytotoxicity in ovarian cancer cells: Nutr Cancer, 2018; 70(7); 1118-25
26. Teodori E, Braconi L, Bua S, A new strategy to reverse the P-gp mediated multidrug resistance (MDR) in cancer cells: Molecules, 2020; 25(7); 1748
27. Li GP, Chen XP, Wang Q, The roles of four multi-drug resistance proteins in hepatocellular carcinoma multidrug: J Huazhong Univ Sci Technol Med Sci, 2007; 27(2); 173-75
Figures
 Figure 1. Cytotoxicities of the DOX-nano in HepG2/DOX cells. HepG2/DOX cells were treated with DOX-nano reagent at different dosages (1, 2, 4, 8, 16 μg/mL) for 4 h, and the MTT assay was used to detect the cytotoxicities and IC50 values. DOX – doxorubicin; DOX-nano – doxorubicin-loaded docosahexaenoic acid nanoparticles.
Figure 1. Cytotoxicities of the DOX-nano in HepG2/DOX cells. HepG2/DOX cells were treated with DOX-nano reagent at different dosages (1, 2, 4, 8, 16 μg/mL) for 4 h, and the MTT assay was used to detect the cytotoxicities and IC50 values. DOX – doxorubicin; DOX-nano – doxorubicin-loaded docosahexaenoic acid nanoparticles. Figure 2. Cellular uptake of DOX-nano. Cells in the HepG2+DOX, HepG2+DOX-nano, HepG2/DOX+DOX, and HepG2/ADM+DOX-nano groups were treated with DOX reagent (5 μg/mL) or DOX-nano (5 μg/mL) for 2, 8, and 24 h after incubation, and fluorescence intensity of DOX was detected. DOX – doxorubicin; DOX-nano – doxorubicin-loaded docosahexaenoic acid nanoparticles.
Figure 2. Cellular uptake of DOX-nano. Cells in the HepG2+DOX, HepG2+DOX-nano, HepG2/DOX+DOX, and HepG2/ADM+DOX-nano groups were treated with DOX reagent (5 μg/mL) or DOX-nano (5 μg/mL) for 2, 8, and 24 h after incubation, and fluorescence intensity of DOX was detected. DOX – doxorubicin; DOX-nano – doxorubicin-loaded docosahexaenoic acid nanoparticles. Figure 3. Analysis of apoptotic HepG2 or HepG2/DOX cells using annexin V-fluorescein isothiocyanate/propidium iodide flow cytometry assay. Compared with the HepG2/DOX group, @ P<0.05, @@ P<0.01; compared with the HepG2+DOX-nano group, # P<0.05, ## P<0.01; compared with the HepG2/DOX+DOX group, * P<0.05, ** P<0.01. DOX – doxorubicin; DOX-nano – doxorubicin-loaded docosahexaenoic acid nanoparticles.
Figure 3. Analysis of apoptotic HepG2 or HepG2/DOX cells using annexin V-fluorescein isothiocyanate/propidium iodide flow cytometry assay. Compared with the HepG2/DOX group, @ P<0.05, @@ P<0.01; compared with the HepG2+DOX-nano group, # P<0.05, ## P<0.01; compared with the HepG2/DOX+DOX group, * P<0.05, ** P<0.01. DOX – doxorubicin; DOX-nano – doxorubicin-loaded docosahexaenoic acid nanoparticles. Figure 4. (A, B) Analysis of HepG2 or HepG2/DOX cells migration. Pictures of the scratches were photographed at 0 and 24 h. Compared with the HepG2/DOX group, @ P<0.05, @@ P<0.01; compared with the HepG2+DOX-nano group, # P<0.05, ## P<0.01; compared with the HepG2/DOX+DOX group, * P<0.05, ** P<0.01. DOX – doxorubicin; DOX-nano – doxorubicin-loaded docosahexaenoic acid nanoparticles.
Figure 4. (A, B) Analysis of HepG2 or HepG2/DOX cells migration. Pictures of the scratches were photographed at 0 and 24 h. Compared with the HepG2/DOX group, @ P<0.05, @@ P<0.01; compared with the HepG2+DOX-nano group, # P<0.05, ## P<0.01; compared with the HepG2/DOX+DOX group, * P<0.05, ** P<0.01. DOX – doxorubicin; DOX-nano – doxorubicin-loaded docosahexaenoic acid nanoparticles. Figure 5. Protein expressions of drug efflux transporters, MRP, LRP, BCRP, Bcl-2, and PKC-α in HepG2 or HepG2/DOX cells. The protein expression was detected using western blotting, and β-actin was used as the internal control. Compared with the HepG2/DOX group, @ P<0.05, @@ P<0.01; compared with the HepG2+DOX-nano group, # P<0.05, ## P<0.01; compared with the HepG2/DOX+DOX group, * P<0.05, ** P<0.01. BCRP – breast cancer resistance protein; Bcl-2 – B-cell lymphoma 2; DOX – doxorubicin; DOX-nano – doxorubicin-loaded docosahexaenoic acid nanoparticles; LRP – lung resistance protein; MRP – multidrug resistance protein; PKC-α – protein kinase C alpha.
Figure 5. Protein expressions of drug efflux transporters, MRP, LRP, BCRP, Bcl-2, and PKC-α in HepG2 or HepG2/DOX cells. The protein expression was detected using western blotting, and β-actin was used as the internal control. Compared with the HepG2/DOX group, @ P<0.05, @@ P<0.01; compared with the HepG2+DOX-nano group, # P<0.05, ## P<0.01; compared with the HepG2/DOX+DOX group, * P<0.05, ** P<0.01. BCRP – breast cancer resistance protein; Bcl-2 – B-cell lymphoma 2; DOX – doxorubicin; DOX-nano – doxorubicin-loaded docosahexaenoic acid nanoparticles; LRP – lung resistance protein; MRP – multidrug resistance protein; PKC-α – protein kinase C alpha. Figure 6. In vivo fluorescence imaging (A) and TUNEL assay (B). In vivo fluorescence imaging showing tumor accumulation of DOX-nano after intravenous injection. TUNEL assay observed cell apoptosis in tumor tissues with DOX-nano injection. DOX-nano – doxorubicin-loaded docosahexaenoic acid nanoparticles.
Figure 6. In vivo fluorescence imaging (A) and TUNEL assay (B). In vivo fluorescence imaging showing tumor accumulation of DOX-nano after intravenous injection. TUNEL assay observed cell apoptosis in tumor tissues with DOX-nano injection. DOX-nano – doxorubicin-loaded docosahexaenoic acid nanoparticles. Figure 7. In vivo anticancer efficacy of DOX-nano in nude mice bearing HepG2/DOX xenograft. Compared with the model group, & P<0.05, && P<0.01; compared with the DOX group, # P<0.05, ## P < 0.01; compared with the vector-nano group, * P<0.05, ** P<0.01. DOX – doxorubicin; DOX-nano – doxorubicin-loaded docosahexaenoic acid nanoparticles.
Figure 7. In vivo anticancer efficacy of DOX-nano in nude mice bearing HepG2/DOX xenograft. Compared with the model group, & P<0.05, && P<0.01; compared with the DOX group, # P<0.05, ## P < 0.01; compared with the vector-nano group, * P<0.05, ** P<0.01. DOX – doxorubicin; DOX-nano – doxorubicin-loaded docosahexaenoic acid nanoparticles. Figure 8. Protein expressions of drug efflux transporters, MRP, LRP, BCRP, Bcl-2, and PKC-α in tumor tissues. The protein expressions were detected using western blotting, and β-actin was used as the internal control. Compared with the model group, & P<0.05, && P<0.01; compared with the DOX group, # P<0.05, ## P<0.01; compared with the vector-nano group, * P<0.05, ** P<0.01. BCRP – breast cancer resistance protein; Bcl-2 – B-cell lymphoma 2; LRP – lung resistance protein; MRP – multidrug resistance protein; PKC-α – protein kinase C alpha.
Figure 8. Protein expressions of drug efflux transporters, MRP, LRP, BCRP, Bcl-2, and PKC-α in tumor tissues. The protein expressions were detected using western blotting, and β-actin was used as the internal control. Compared with the model group, & P<0.05, && P<0.01; compared with the DOX group, # P<0.05, ## P<0.01; compared with the vector-nano group, * P<0.05, ** P<0.01. BCRP – breast cancer resistance protein; Bcl-2 – B-cell lymphoma 2; LRP – lung resistance protein; MRP – multidrug resistance protein; PKC-α – protein kinase C alpha. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








