12 April 2021: Clinical Research
Epidemiological and Clinical Characteristics of 217 Cases of COVID-19 in Jiangsu Province, China
Jun Zhou1E, Bing Chen2C, Yali Wang1D, Feng Wu1AG*DOI: 10.12659/MSM.930853
Med Sci Monit 2021; 27:e930853
Abstract
BACKGROUND: COVID-19 has become a worldwide epidemic disease and is a public health crisis. We aim to provide evidence for clinical diagnosis and assessment of severity by analyzing patients’ clinical data and early laboratory results and exploring the correlation between laboratory results and clinical classification.
MATERIAL AND METHODS: We enrolled 283 cases of suspected and diagnosed COVID-19 from 16 hospitals in Jiangsu Province from January to April 2020. The routine laboratory blood examinations, T lymphocyte subsets, and biochemical and coagulation function among different populations were contrasted by t test and chi-square (χ²) test.
RESULTS: Cough, fever, and dyspnea could be helpful to diagnose COVID-19 infection (P<0.05). Patients who were older or had comorbidities tended to become severe and critical cases. Among all the patients, the most obvious abnormal laboratory results were higher neutrophil count, CRP, total bilirubin, BUN, CRE, APTT, PT, and D-dimer, and lower blood platelet and lymphocyte count. CD3⁺ T cell, CD4⁺ T cell, and CD8⁺ T cell counts gradually decreased with exacerbation of the disease (P<0.05).
CONCLUSIONS: Cough and fever were the most common symptom. Patients with comorbidities were in more serious condition. The detection of inflammatory indexes, coagulation function, lymphocyte subsets, and renal function can help diagnose and assess the severity of COVID-19.
Keywords: COVID-19, Diagnosis, Epidemiology, Aged, 80 and over, Blood Coagulation, COVID-19, Comorbidity, Cough, Fever, Risk Factors, SARS-CoV-2, Severity of Illness Index, T-Lymphocyte Subsets, young adult
Background
In December 2019, the outbreak of a novel fatal zoonotic disease, Coronavirus Disease 2019 (COVID-19), appeared in Wuhan, Hubei Province, China [1–3]. The disease spread all over the world and rapidly caused a pandemic. As of 13 December 2020, 73 235 212 COVID-19 cases with 1 625 160 deaths have been verified worldwide. COVID-19 can cause many kinds of respiratory symptoms, including fever, cough, fatigue, dyspnea, and coma [4–8]. Some severe patients with COVID-19 develop multifunctional organ damage, including kidney [9], liver [10], and acute cardiac damage [11]. Because COVID-19 can spread among people quickly, it has now become a huge threat to the world and the latest reported mortality rate is 2.3% [12]. Because the symptoms of COVID patients are nonspecific, early detection is very important and can reduce mortality. With the effective control by the government and other sectors, the epidemic was quickly controlled in China. However, there are still many new cases in the world every day, so it is imperative and significant to show Chinese data and experience to help other countries.
We retrospectively analyzed the clinical data of laboratory-diagnosed COVID-19 cases at 16 hospitals in Jiangsu Province from January to April 2020 in our study. Then, we aimed to determine the role of laboratory indexes in diagnosis and the relationship between laboratory indicators and disease severity to help diagnose COVID-19 infection early and evaluate the severity of disease.
Material and Methods
PATIENTS AND DATA COLLECTION:
This was a retrospective, multi-center study based on laboratory data from patients diagnosed with COVID-19. The data for this retrospective observational study were obtained by the Health Information Center of Jiangsu Provincial Health Commission through a private health network. All the patients with COVID-19 infection were admitted from 1 January 2020 to 30 April 2020 in 16 Jiangsu provincial hospitals.
All COVID-19 patients were defined according to the diagnostic and treatment guideline for SARS-CoV2 issued by the National Health Commission of China (version 7) [13]. All confirmed COVID-19 patients were diagnosed by a positive result for real-time polymerase chain reaction (RT-PCR) assay from a throat swab. Nasopharyngeal swab specimens collected from all cases were assessed by RT-PCR for diagnosis.
Diagnosis of 2019-nCoV pneumonia was based on presence of any 1 of 3 epidemiology criteria and any 2 of 3 clinical manifestations. Epidemiological history criteria were: (1)14 days before the onset in the Wuhan area or other areas with local cases of continuous transmission or travel history or residence history; (2) previous contact with patients with fever or respiratory symptoms from Wuhan or other areas with persistent transmission of local cases within 14 days before onset; and (3) a cluster of disease or an epidemiological association with 2019-nCoV infection. Clinical manifestations were: (1) fever; (2) with the above imaging characteristics of pneumonia; and (3) total number of white blood cells was normal or decreased in the early stage of disease, or the lymphocyte count was reduced.
Confirmed COVID-19 patients had: (1) 2019-nCoV pneumonia suspected case of pneumonia; (2) Real-time fluorescence RT-PCR test results of nasopharyngeal swabs show 2019-nCoV nucleic acid positive. Patients meeting the above 2 conditions can be diagnosed with 2019-nCoV pneumonia. Real-time fluorescence RT-PCR test method was carried out in accredited laboratories and in accordance with the plan set by the World Health Organization.
According to the guideline criteria, all the patients were divided into 4 types: mild, moderate, severe, and critical illness. Because there were fewer patients with severe and critical illness, we put them into a single category to facilitate analysis. The control group consisted of suspected cases who had a history of close contact, but had a negative result for real-time fluorescence RT-PCR assay of nasopharyngeal swabs.
This study was approved by the Ethics Committees of the 16 hospitals, but written informed consent was not obtained because of the urgent conditions. Data on age, sex, time of admission, time of diagnosis, discharge time, basic diseases, first symptoms, signs, and laboratory tests were collected for all cases. Due to the obvious fluctuation of various indicators in the course of disease, the respiratory specimens, the basic data, and laboratory tests of all patients included in this study were the results of the examination on the first day after admission.
Laboratory blood tests included hepatorenal function (alanine aminotransferase [ALT], aspartate aminotransferase [AST], total bilirubin [TBil], LDH, urea nitrogen [BUN] and creatinine [CRE]), coagulation function, myocardial enzyme, procalcitonin (PCT), and blood routine examination (white blood cell count [WBC], neutrophils [NEUT], lymphocyte [LYM], hemoglobin [HGB], thrombocyte [PLT], C-reactive protein [CRP]). An immunofluorescence dye quantitative detector was used to detect procalcitonin.
DETECTION OF LYMPHOCYTE SUBSETS:
A blood cytometer was used to count the total number of lymphocytes in the peripheral blood of patients when they were fasting. A DXFlex flow cytometer and CellQuest software (BD Bioscience) were used to analyze the percentage of lymphocyte subsets of patients. The absolute number of different lymphocyte subsets was calculated by multiplying the total number of lymphocytes by the percentage of lymphocyte subsets. Flow cytometry was used to determine the peripheral blood T lymphocyte subsets of immune typing, using counts of CD3+ T cell, CD4+ T cell, CD8+ T cell, and CD4+/CD8+.
STATISTICAL ANALYSIS:
Statistical analysis was performed using SPSS version 21 software (IBM, Inc., Armonk, NY, USA). Measurement data conforming to normal distribution were described as mean±standard deviation, and the independent-sample
Results
LABORATORY RISK FACTORS FOR COVID-19 INFECTION:
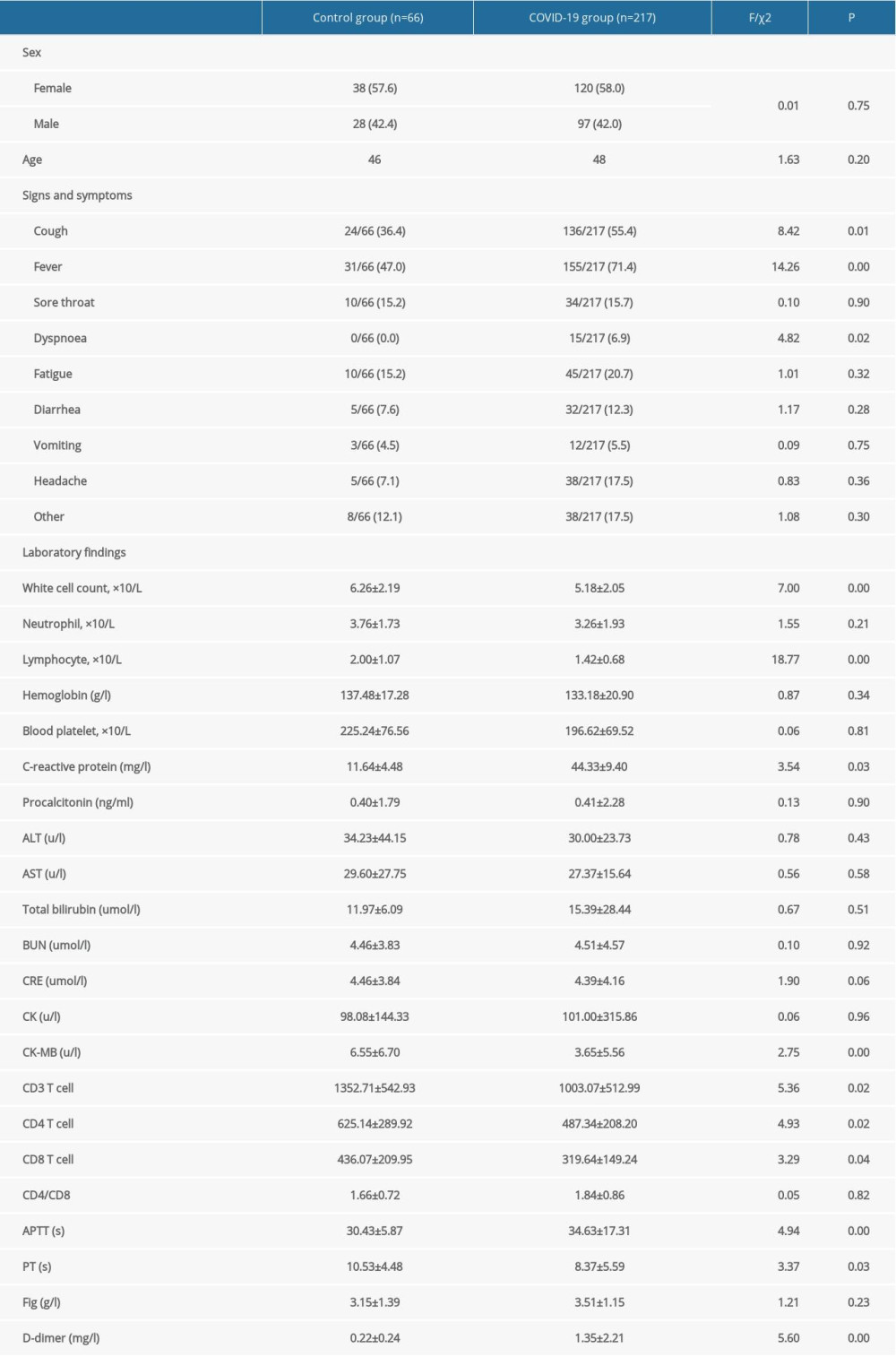
The laboratory results of the COVID-19 patients and healthy subjects on admission are presented in Table 1. The results showed that COVID-19 patients had lower white cell count, lymphocyte count, C-reactive protein, CK-MB, CD3+ T cell, CD4+ T cell, CD8+ T cell, and PT, but higher APTT and D-dimer levels. Cough, fever, and dyspnea could be helpful to diagnose COVID-19 infection. These results differ significantly between the COVID-19 group and control group.
DIFFERENCES IN CLINICAL CHARACTERISTICS AND LABORATORY FINDINGS OF PATIENTS WITH COVID-19 ILLNESS:
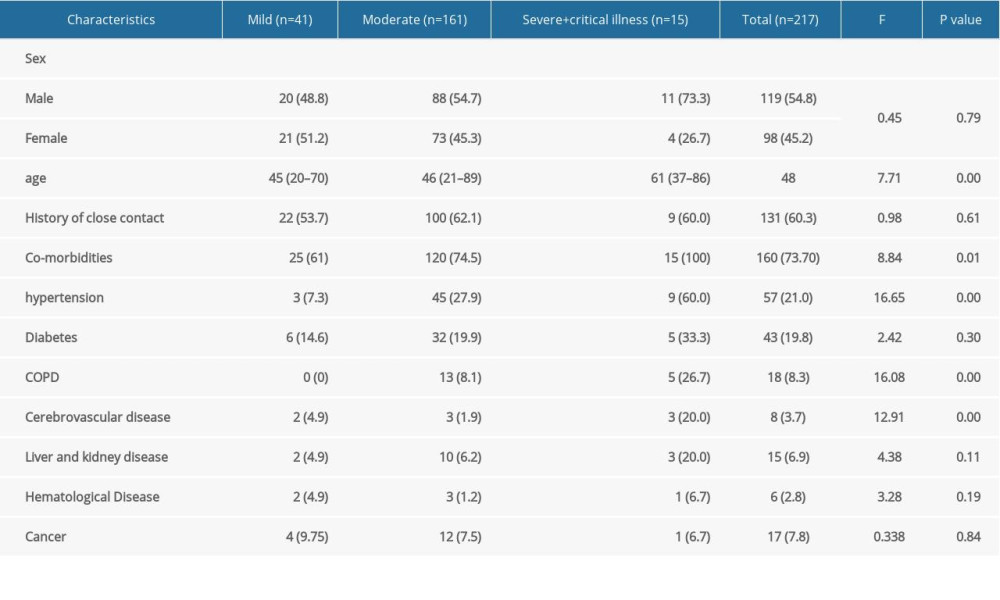
Table 2 shows the general characteristics of 217 patients with COVID-19 illness (54.8% male, 45.2% female, median age 48 years). Of all the COVID-19 patients, 18.9% had mild illness, 74.2% had moderate illness, and 6.9% had severe or critical illness. The median age of severe and critical COVID-19 patients was higher than that of the mild and moderate patients (61 years, 46 years, and 45 years, respectively, P<0.001). Among those with severe and critical illness, 73.3% were males. A history of close contact with COVID-19 cases was found in 63.1% of the COVID-19 patients, and chronic medical illness was present in 73.7% of patients. The most common chronic diseases were hypertension (21%), diabetes (19.8%), and COPD (8.3%). The incidence of these chronic diseases was higher among severely and critically ill patients than among patients with mild and moderate COVID-19 illness.
LABORATORY FINDINGS AT PRESENTATION AMONG DIFFERENT TYPES OF COVID-19 PATIENTS:
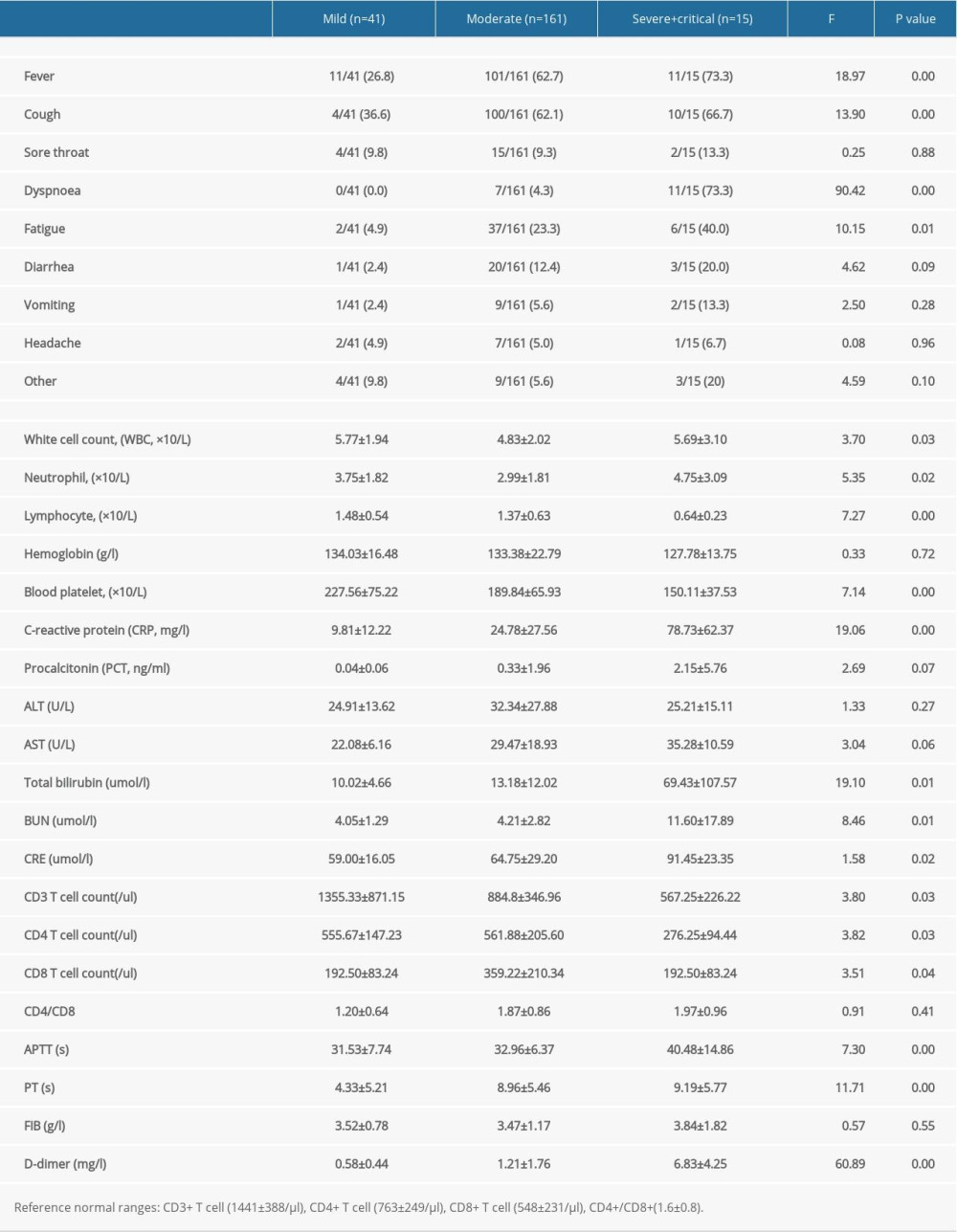
As shown in Table 3, among all the patients, the most obvious abnormal laboratory indexes were higher neutrophil count, CRP, total bilirubin, BUN, CRE, APTT, PT, and D-dimer, and lower blood platelet and lymphocyte count. The differences among the 3 types of patients were increasingly obvious with disease progression. CD3+ T cell, CD4+ T cell, and CD8+ T cell counts decreased gradually with exacerbation of the disease.
Discussion
A novel coronavirus, SARS-CoV-2, was isolated from Wuhan in December 2019 [4] and quickly spread to other Chinese cities and many foreign countries. On 12 January, WHO named it “2019 new coronavirus (COVID-19)”. It had 85% or more homology to bat SARS-like coronavirus (bat-SL-COVZC45) [14], which was transmitted rapidly from human to human by close contacts and was classified by the government as a B infectious disease [15]. Simultaneously, the disease progresses rapidly; about 30% of patients developed severe and critical illness, even those transferred to ICU treatment [16]. Therefore, in clinical practice, we are actively looking for meaningful laboratory examination results which can provide help for clinical diagnosis, treatment, and disease assessment as soon as possible.
The present multi-center, retrospective, observational study described the epidemiological and laboratory characteristics of COVID-19 patients and identified the risk factors for disease severity. These patients all received centralized treatment at various infectious disease hospitals or municipal-level COVID-19 treatment centers in Jiangsu Province. Most of the enrolled patients were residents infected by coming into close contact with imported cases or with a history of travel to or residence in Wuhan.
Previous studies showed the most common symptoms of patients with COVID-19 were fever and cough and had certain diagnostic significance, which was consistent with the results of our study. Our findings suggested dyspnea occurred in severe and critically ill patients, which indicated a serious condition of disease. Sore throat also has a certain reference significance for clinical diagnosis. Although there was no statistically significant difference in symptoms such as diarrhea, vomiting, fatigue, and headache between the control group and the COVID-19 group, it may be related to the inconsistent immune function of each patient and the above different clinical manifestations appeared in each patient.
According to the latest guide on COVID-19 diagnosis from the National Health Commission of China [17] and severity degree, new COVID-19 pneumonia was divided into 4 categories: mild, moderate, severe, and critical. Because there were fewer critical cases, we classified critical patients and severe patients into a single category. Previous studies have reported that older patients are more likely to get COVID-19 pneumonia, and their prognosis is worse [18]. In addition, our study showed older patients with chronic comorbidity had more severe condition, especially those with hypertension, COPD, and heart and brain diseases. Therefore, age and comorbidities are risk factors for COVID-19, which is consistent with previous research [19]. Based on the clinical manifestations and laboratory test results of the patients upon admission, we found that the symptoms of cough, fever, dyspnea, and fatigue in the 3 groups of patients became more obvious and more serious. The laboratory results of this study showed that with the aggravation of the disease, WBC count and neutrophils increased, and lymphocyte count and blood platelets decreased, suggesting that the cellular immune system can be inhibited by COVID-19, and the results showed that critical and severe patients were more susceptible to bacterial infections. Therefore, the decreased lymphocyte count and increased WBC, neutrophils, and CRP may be related to the infection of patients, but the change in PCT was not obvious. COVID-19 can cause damage to organs such as the liver, kidneys, and myocardium, and then develop into multifunction organ failure [20]. Our study also showed that the levels of total bilirubin, urea nitrogen, and creatinine were significantly different among the 3 groups, and gradually increased with progression of the disease, which is consistent with previous research results [21]. However, there was no significant difference in the results of alanine aminotransferase (ALT) and glutamic oxaloacetic transaminase (AST) among the 3 groups. The renal function damage occurred in the severe and critical patients group, which in extrapulmonary organs indicates the severity of the disease.
The majority of COVID-19 cases may be due to failure of the body to clear SARS-CoV-2 in time, the persistent immune cell damage caused by the over-active immune response, and then the falling CD4+ T and CD8+ T cell counts [22]. Our study showed that the levels of CD3+ T cells, CD4+ T cells, and CD8+ T cells were significantly different between the patients with COVID-19 and the control group. Moreover, the lower the expression level was, the more severe the disease was, which has a certain reference value for clinical classification. These results were in accordance with the patients in Wuxi Hospital, which were reported by doctor Chen’s team [23]. The decrease of T lymphocyte count suggests that SARS-CoV-2 consumes a large number of immune cells and inhibits the cellular immune function of the body, which is a major factor in the pathogenesis of COVID-19 [24]. Therefore, T lymphocyte subsets can be used as an index to evaluate the condition of COVID-19 patients. Our study also found that an important reason for the critical condition of patients with COVID-19 was the deterioration of the patient’s condition, coagulation dysfunction, and even DIC, which eventually leads to death [25]. Elevated levels of D-dimer indicate hypercoagulability and secondary hyperfibrinolysis, which is of great significance in the diagnosis, efficacy evaluation, and prognosis prediction of thrombotic diseases. The results of this study suggested that the expression levels of D-dimer, partial prothrombin time, and prothrombin time were significantly different between the COVID-19 patient group and the control group. The expression levels of D-dimer, partial prothrombin time, and prothrombin time indicated the aggravation of the disease (
Conclusions
Our study highlighted the clinical features of patients with COVID-19 of various degrees of severity. COVID-19 was more likely to occur in a population with comorbidities and in older patients, especially in severe and critical patients. The most obvious differences in indexes were lower lymphocyte count, blood platelet, higher PT, APTT, D-dimer, CRE, and CRP among the 3 groups. Other indexes, such as lower CD3+ T cell, CD4+ T cell and CD8+ T cell, and higher total bilirubin, were also important. Some clinical manifestations and specific laboratory indicators are helpful to diagnose and evaluate COVID-19.
References
1. Wu F, Zhao S, Yu B, A new coronavirus associated with human respiratory disease in China: Nature, 2020; 579(7798); 265-69
2. Zhu N, Zhang D, Wang W, A novel coronavirus from patients with pneumonia in China, 2019: N Engl J Med, 2020; 382(8); 727-33
3. Guan WJ, Ni ZY, Hu Y, Clinical characteristics of coronavirus disease 2019 in China: N Engl J Med, 2020; 382(18); 1708-20
4. Lu H, Stratton CW, Tang YW, Outbreak of pneumonia of unknown etiology in Wuhan, China: The mystery and the miracle: J Med Virol, 2020; 92(4); 401-2
5. Hui DS, I Azhar E, Madani TA, The continuing 2019-nCoV epidemic threat of novel coronaviruses to global health-The latest 2019 novel coronavirus outbreak in Wuhan, China: Int J Infect Dis, 2020; 91; 264-66
6. Ludvigsson JF, Systematic review of COVID-19 in children shows milder cases and a better prognosis than adults: Acta Paediatr, 2020; 109(6); 1088-95
7. Molloy EJ, Bearer CF, COVID-19 in children and altered inflammatory responses: Pediatr Res, 2020; 88(3); 340-41
8. Wölfel R, Corman VM, Guggemos W, Virological assessment of hospitalized patients with COVID-2019: Nature, 2020; 581(7809); 465-69
9. Ronco C, Reis T, Husain-Syed F, Management of acute kidney injury in patients with COVID-19: Lancet Respir Med, 2020; 8(7); 738-42
10. Jothimani D, Venugopal R, Abedin MF, COVID-19 and the liver: J Hepatol, 2020; 73(5); 1231-40
11. Zheng YY, Ma YT, Zhang JY, COVID-19 and the cardiovascular system: Nat Rev Cardiol, 2020; 17(5); 259-60
12. Wu Z, McGoogan JM, Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: Summary of a report of 72314 cases from the Chinese Center for Disease Control and Prevention: JAMA, 2020; 323(13); 1239-42
13. , Diagnosis and treatment protocol for novel coronavirus pneumonia (Trial Version 7): China NHCOTPSRO (2020-03-29) http://en.nhc.gov.cn/2020-03/29/c_78469.htm
14. Cui J, Li F, Shi ZL, Origin and evolution of pathogenic coronaviruses: Nat Rev Microbiol, 2019; 17(3); 181-92
15. Li Q, Guan X, Wu P, Early transmission dynamics in Wuhan, China, of novel coronavirus-infected pneumonia: N Engl J Med, 2020; 382(13); 1199-1207
16. Chen N, Zhou M, Dong X, Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: A descriptive study: Lancet, 2020; 395(10223); 507-13
17. Jin YH, Cai L, Cheng ZS, A rapid advice guideline for the diagnosis and treatment of 2019 novel coronavirus (2019-nCoV) infected pneumonia (Standard version): Mil Med Res, 2020; 7(1); 4
18. Dang JZ, Zhu GY, Yang YJ, Clinical characteristics of coronavirus disease 2019 in patients aged 80 years and older: J Integr Med, 2020; 18(5); 395-400
19. Hu X, Hu C, Yang Y, Clinical characteristics and risk factors for severity of COVID-19 outside Wuhan: A double-center retrospective cohort study of 213 cases in Hunan, China: Ther Adv Respir Dis, 2020; 14; 1753466620963035
20. Rodriguez-Morales AJ, Bonilla-Aldana DK, Balbin-Ramon GJ, History is repeating itself: Probable zoonotic spillover as the cause of the 2019 novel Coronavirus Epidemic: Infez Med, 2020; 28(1); 3-5
21. Lins PRG, de Albuquerque CCC, Assis CF, Cov-hep study: heparin in standard anticoagulation based on citrate for continuous veno-venous hemodialysis in patients with COVID-19: A structured summary of a study protocol for a randomized controlled trial: Trials, 2020; 21(1); 920
22. Gan J, Li J, Li S, Leucocyte subsets effectively predict the clinical outcome of patients with COVID-19 pneumonia: A retrospective case-control study: Front Public Health, 2020; 8; 299
23. Chu M, Bian L, Huang J, Clinical characteristics and outcomes of 48 patients hospitalized for COVID-19 infection in Wuxi: A retrospective cohort study: Medicine (Baltimore), 2021; 1005(5); e2399
24. Chen N, Zhou M, Dong X, Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study: Lancet, 2020; 395(10223); 507-13
25. Guan WJ, Ni ZY, Hu Y, Clinical characteristics of coronavirus disease 2019 in China: N Engl J Med, 2020; 382(18); 1708-20
26. Huang C, Wang Y, Li X, Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China: Lancet, 2020; 395(10223); 497-506
27. Zhou F, Yu T, Du R, Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study: Lancet, 2020; 395(10229); 1054-62
In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387