29 September 2021: Clinical Research
Identification of Risk Factors for Intraoperative Acquired Pressure Injury in Patients Undergoing Neurosurgery: A Retrospective Single-Center Study
Ying Wu1AD, Zhiqun Jiang2AF, Shuzhen Huang1BCD, Bin Shi1BCD, Chan Wang1BCD, Yu Zeng1BEG*DOI: 10.12659/MSM.932340
Med Sci Monit 2021; 27:e932340
Abstract
BACKGROUND: Risk factors for intraoperative acquired pressure injury (IAPI) in patients undergoing various surgical procedures have been described in previous studies. However, whether risk factors for IAPI in patients undergoing neurosurgery differ remains unknown. The aim of the present study was to explore independent risk factors for IAPI in patients undergoing neurosurgery.
MATERIAL AND METHODS: Data from 465 patients who underwent neurosurgery between October 2017and December 2018 and who were at high risk of IAPIs were retrospectively analyzed. Independent risk factors for IAPI were evaluated using univariate and multivariate logistic regression models.
RESULTS: Sixty-nine IAPIs (14.8%) in 465 patients undergoing neurosurgery were assessed. Multivariate logistic regression analyses showed that being overweight (odds ratio [OR] 2.685; 95% confidence interval [CI] 1.206-5.975; P=0.016), prone position (OR 7.502, 95% CI 2.470-22.787. P<0.001), lateral position (OR 15.301, 95% CI 4.903-47.753, P<0.001), use of a head frame (OR 3.716, 95% CI 1.431-9.653, P=0.007), surgical times of 4 to 8 h (OR 7.276, 95% CI 2.249-23.542, P<0.001), and surgical times ≥8 h (OR 173.248, 95% CI 32.629-919.896, P<0.001) all were associated with an increased risk of IAPI in patients undergoing neurosurgery. The factors associated with reduced risk of IAPI were high serum albumin levels (OR 0.099, 95% CI 0.016-0.608, P=0.013) and use of memory sponge pads (OR 0.064, 95% CI 0.020-0.202, P<0.001).
CONCLUSIONS: The present study indicates that being overweight, prone and lateral positioning, use of a head frame, and longer surgical times are associated with an increased risk of IAPI in patients undergoing neurosurgery. Prospective studies should be conducted to verify these findings and consideration should be given to use of these factors in clinical practice to identify high-risk patients.
Keywords: Intraoperative Care, Neurosurgical Procedures, Risk Factors, Brain Injuries, Female, Humans, Immobilization, Intraoperative Complications, operative time, overweight, Patient Positioning, Posture, Pressure, Serum Albumin
Background
Patients undergoing surgery can develop intraoperative acquired pressure injuries (IAPIs) in healthy skin and deeper tissue. Studies indicate that the prevalence of pressure injuries varies, the occurrence is higher in the operating room, and these injuries are associated with increased morbidity, longer hospital stays, and higher healthcare costs [1–3]. Direct and indirect costs of hospital-acquired pressure injuries were nearly $11 billion in 2009 and increased by 44% in 2016 [4–6]. Long surgeries are associated with an increased risk of IAPIs because of the increased risk of ischemia and perfusion-reperfusion injury [7]. Risk factors for IAPIs have already been described in several studies [8–11], but no research has focused on patients undergoing neurosurgery. These individuals have a high risk of IAPIs because their procedures are longer, continuous irrigation with saline is used during these surgeries, and craniotomy requires use of air and electric drills, which results in vibration.
Several intrinsic and extrinsic risk factors for IAPIs already have been identified in patients undergoing surgery [7] and they differ by procedure [12,13]. In patients undergoing urologic surgery, Connor et al found that anesthesia duration (odds ratio [OR] 1.005) and total time of diastolic blood pressure <50 mmHg (OR: 1.007) were significantly associated with IAPI [14]. In patients undergoing cardiovascular surgery, Lu et al found that risk of IAPI was significantly affected by disease category (valvular disease of the heart [OR 6.427], coronary artery disease [OR 8.800], and thoracic aortic aneurysms [OR 5.736]), and surgery length (OR 1.005) [15]. However, no study has focused on risk factors for IAPI in patients undergoing neurosurgery. Therefore, we conducted a retrospective study to identify independent risk factors for IAPI in patients undergoing neurosurgery.
Material and Methods
POPULATION:
A total of 465 patients screened using the Scott Triggers Preoperation Evaluation Scale [16] who were undergoing neurosurgery between October 2017 and December 2018 were enrolled in the present study. Patients with 2 or more of the following characteristics were considered to be at high risk for IAPI: age ≥62.0 years, serum albumin level <35.0 g/L, body mass index (BMI) <19.0 or >40.0 kg/m2, estimated surgery length ≥3.0 h, and American Society of Anesthesiologists (ASA) score ≥3. All patients undergoing neurosurgery for cerebrovascular disease, brain tumors, spinal cord disease, craniocerebral trauma, or congenital malformations also were considered to be at high risk. The exclusion criteria for the present study were: (1) presence of pressure injuries before admission; (2) damage to the skin other than at the incision site; (3) presence of a skin disease that made it difficult to monitor the condition of the skin; (4) treatment with a second surgery after admission, transfer from another hospital to our hospital, or death during the study; and (5) mental illness.
DATA COLLECTION:
Guidelines, systematic reviews, original articles, and existing evaluation scales related to pressure injuries were screened on the basis of level of evidence for them. Factors in the Braden, Norton, Waterlow, and Munro scales related to IAPI were extracted to construct a questionnaire for patients undergoing neurosurgery. The tool was used to collect data on: (1) general patient characteristics (age, sex, BMI, smoking status, hypertension, and diabetes); (2) preoperative characteristics (history of surgery, physical activity, and hemoglobin and serum albumin levels); and (3) intraoperative characteristics (intraoperative position, type of position pad, use of a head frame, blood transfusion, use of vasoactive agents, ASA score, surgery length and type, and amount of blood loss).
ENDPOINTS:
The primary outcome of the present study was the incidence of IAPI, which was assessed based on criteria from the National Pressure Ulcer Advisory Committee, which were updated in 2016. IAPI was considered stage 1 when the skin was intact at the bony protuberance with localized erythema that did not fade under pressure; in stage II, part of the dermis was missing; and stage III IAPI constituted a full-thickness skin defect.
ETHICAL CONSIDERATIONS:
The present study was reviewed and approved by the Ethics Committee of the First Affiliated Hospital of Nanchang University (2019-103). No informed consent was required because of the retrospective nature of the research, which required no patient contact.
STATISTICAL ANALYSIS:
Continuous data are presented as median and interquartile ranges, if the data were non-normally distributed. Categorical data are presented as event rates. The characteristics of patients with and without IAPI were compared using Kruskal-Wallis and chi-square tests for continuous and categorical data, respectively. Univariate logistic regression models then were applied to explore independent risk factors for IAPI in patients undergoing neurosurgery. Variables found to be statistically significant on the univariate analyses were entered into a stepwise logistic regression model for multivariate analysis (α=0.05; β=0.10). All reported
Results
PATIENT CHARACTERISTICS:
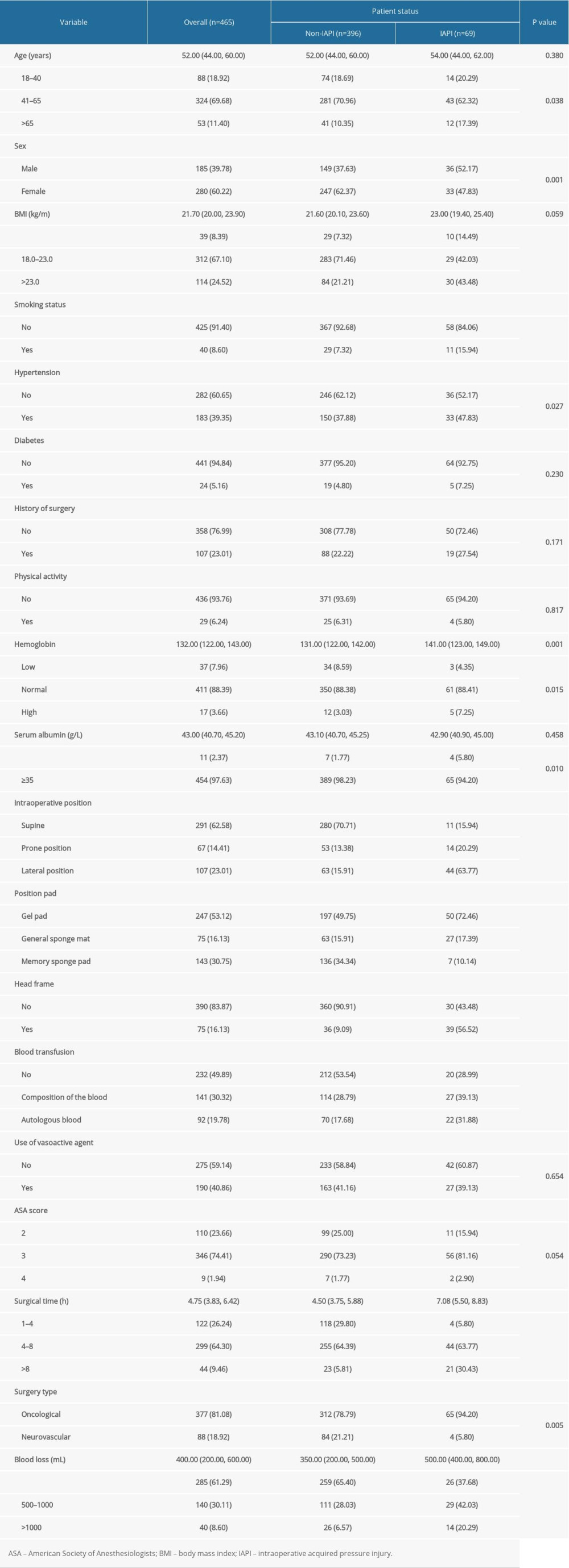
A total of 465 patients were enrolled, 69 of whom were diagnosed with IAPI (14.8%). Baseline characteristics of the participants are summarized in Table 1. The median age of the patients was 52.0 years and 185 of them (39.8%) were men. Prevalence of diabetes (P=0.230), history of surgery (P=0.171), physical activity level (P=0.817), use of vasoactive agents (P=0.654), and ASA scores (P=0.054) were not statistically different between the patients with and without IAPI. However, there were significant differences between the 2 groups in terms of age (P=0.038), sex (P=0.001), BMI (P<0.001), smoking status (P<0.001), hypertension (P=0.027), hemoglobin levels (P=0.015), serum albumin levels (P=0.010), intraoperative positioning (P<0.001), position pad use (P<0.001), head frame use (P<0.001), composition of blood transfusion (P<0.001), length of surgery (P<0.001), and amount of blood loss (P<0.001).
UNIVARIATE LOGISTIC REGRESSION:
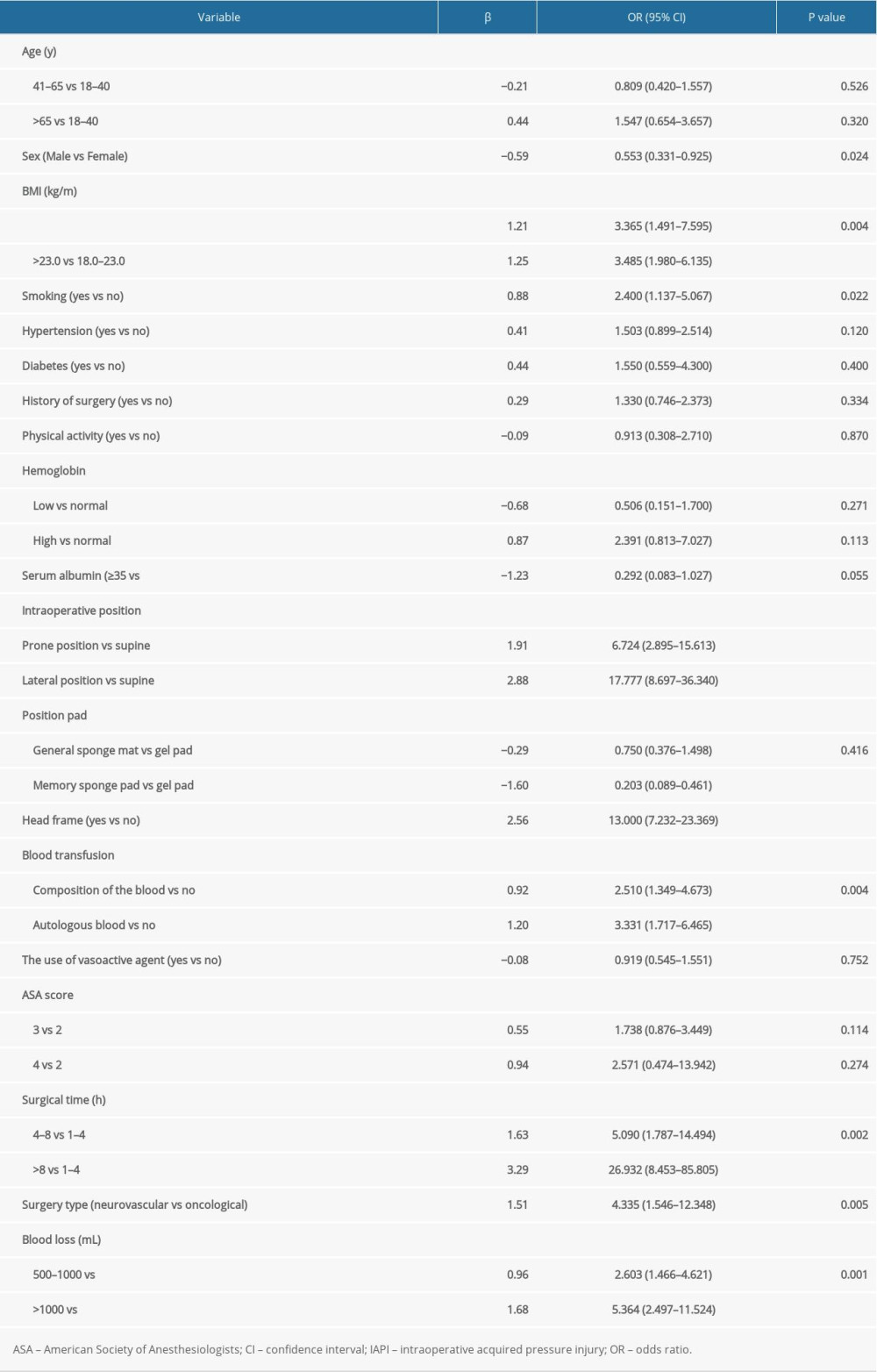
The univariate results for the potential risk factors of IAPI incidence in patients undergoing neurosurgery are summarized in Table 2. We noted that being underweight (OR 3.365, 95% confidence interval [CI] 1.491–7.595, P=0.004) or overweight (OR 3.485, 95% CI 1.980–6.135, P<0.001), a current smoker (OR: 2.400; 95% CI 1.137–5.067, P=0.022), prone positioning (OR 6.724, 95% CI 2.895–15.613, P<0.001), lateral positioning (OR 17.777, 95% CI 8.697–36.340, P<0.001), use of a head frame (OR 13.000, 95% CI 7.232–23.369, P<0.001), composition of blood transfusion (OR 2.510, 95% CI 1.349–4.673, P=0.004), autologous blood transfusion (OR 3.331, 95% CI 1.717–6.465, P<0.001), surgical time of 4 to 8 h (OR 5.090, 95% CI 1.787–14.494, P=0.002), surgical time ≥8 h (OR 26.932, 95% CI 8.453–85.805, P<0.001), treatment for neurovascular disease (OR 4.335, 95% CI 1.546–12.348, P=0.005), blood loss of 500 to 1000 mL (OR 2.603, 95% CI 1.466–4.621, P=0.001), and blood loss >1000 mL (OR 5.364, 95% CI 2.497–11.524, P<0.001) were associated with an increased risk of IAPI in patients undergoing neurosurgery. Conversely, being male (OR 0.553, 95% CI 0.331–0.925, P=0.024) and the use of a memory sponge pad (OR 0.203, 95% CI 0.089–0.461, P<0.001) were associated with a reduced risk of IAPI in patients undergoing neurosurgery. However, age, hypertension, diabetes, history of surgery, physical activity, hemoglobin level, serum albumin level, use of a general sponge mat, use of vasoactive agents, and ASA score were not associated with the risk of IAPI in patients undergoing neurosurgery (P>0.05).
MULTIVARIATE LOGISTIC REGRESSION:
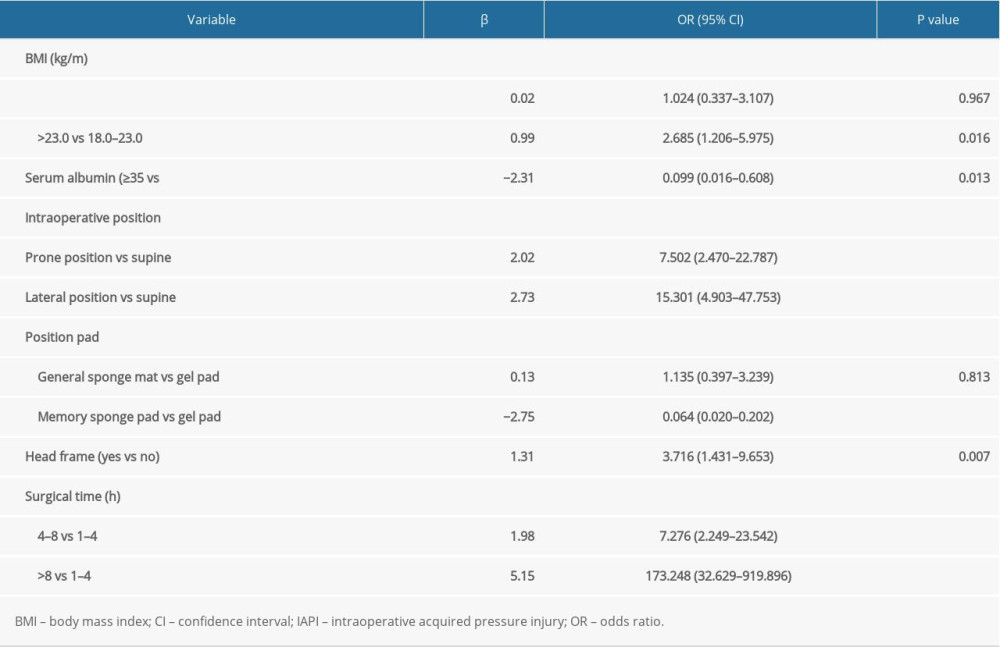
The results of multivariate logistic regression for potential risk factors for IAPI incidence in patients undergoing neurosurgery are summarized in Table 3. The identified independent risk factors for IAPI in patients undergoing neurosurgery included: being overweight (OR 2.685, 95% CI 1.206–5.975, P=0.016), prone positioning (OR 7.502, 95% CI 2.470–22.787, P<0.001), lateral positioning (OR 15.301, 95% CI 4.903–47.753, P<0.001), use of a head frame (OR 3.716, 95% CI 1.431–9.653, P=0.007), surgery length of 4 to 8 h (OR 7.276, 95% CI 2.249–23.542, P<0.001), and surgery length of ≥8 h (OR 173.248, 95% CI 32.629–919.896, P<0.001). Having a high serum albumin level (OR 0.099, 95% CI 0.016–0.608, P=0.013) and use of a memory sponge pad (OR 0.064, 95% CI 0.020–0.202, P<0.001) were associated with a reduced risk of IAPI in patients undergoing neurosurgery.
Discussion
Systematic exploration of the risk factors for IAPI in patients undergoing neurosurgery is important because these factors have not yet been defined. Data from a total of 465 patients undergoing neurosurgery with a wide range of characteristics were retrospectively analyzed in the present study. The results show that risk factors for IAPI in patients undergoing neurosurgery include being overweight, prone or lateral positioning, use of a head frame, and surgery duration of 4 to 8 or ≥8 h. Moreover, having a high serum albumin level and use of a memory sponge pad could lower the risk of IAPI in patients undergoing neurosurgery.
Several studies have already identified risk factors for IAPI in patients undergoing various types of surgery [8–11]. Celik et al recruited 151 patients undergoing elective procedures and found that intraoperative vasopressor usage, skin turgor, and diastolic blood pressure ≤60 mmHg were linked with excess risk of pressure injuries [8]. However, these results were based on univariate analyses, and after adjusting for potential confounders, the authors could not detect any significant risk factors. Luo et al conducted a retrospective study and enrolled 3834 patients undergoing spinal surgery. After adjusting for potential confounders, they found that risk factors for IAPI included: age >60.0 years, being underweight (BMI <18.0 kg/m2), cumulative Braden Scale score ≤13.0, and prolonged preoperative and postoperative times [9]. Xiong et al conducted a retrospective study of 5136 patients undergoing gastrointestinal surgery. They found that preoperative skin compression, physical activity, surgical positioning, and extra intraoperative pressure were independent risk factors for IAPI [10]. A retrospective study conducted by Chen et al of 803 patients undergoing major hepatobiliary surgery found that pancreaticoduodenectomy, open surgery, surgery length, and intraoperative hypotensive episodes were associated with an increased risk of IAPI [11]. No previous studies have investigated risk factors for IAPI in patients undergoing neurosurgery. Therefore, we conducted a retrospective study to explore potential independent risk factors for IAPI in patients undergoing neurosurgery.
The present study showed that risk factors for IAPI associated with neurosurgery included being overweight, prone or lateral positioning, use of a head frame, and surgery length of 4 to 8 h and ≥8 h. The potential reasons are as follows. The influence of BMI on the risk of IAPI may be U-shaped because the exposure to internal soft tissue loads under bony prominences was increased in patients with high or low BMI [17,18]. The main pressure points for patients in the supine position were the shoulder, sacral tail, and heel; for patients in the prone position, they were the 2 shoulder peaks on the side, both sides of the rib cage, iliac spine, and knees; and for patients in the lateral position, they were the temporal head, shoulder, axillary, iliac spine, interior and exterior of the knee, and areas such as the external ankle. The stress point areas for patients in the prone and lateral positions were small while the pressure was great, which was associated with an increased risk of IAPI [19]. The intraoperative position could be restricted by using a head frame, which can increase the risk of IAPI. Hypoperfusion or ischemia of locally compressed tissues lasted longer in patients whose surgeries were longer. Moreover, the epidermal temperature at the compression site was lower in patients who had longer surgeries, which was associated with an increased risk of pressure injuries [20].
The present study also showed that high serum albumin levels and use of a memory sponge pad could protect against IAPI in patients undergoing neurosurgery. Albumin levels are considered an important indicator of nutritional status and malnutrition is significantly associated with risk of pressure injuries [21,22]. Moreover, sponge mats used during surgery are light and soft, but can be easily deformed, which can result in uneven force. Gel pads have good elasticity and they are soft and can relieve pressure, but they can be heavy and have poor air permeability. Memory sponge pads have a slow rebound and are soft, comfortable, and skin-friendly. Their use, therefore, was associated with a reduced risk of IAPI, and it should be recommended in clinical practice.
Several limitations of the present study should be acknowledged. First, it was retrospective and the results could be affected by selection and recall biases. Second, the severity of IAPI was not assessed because the number of patients with stage II IAPI (6 of 465) was smaller than expected and the power was insufficient to detect potentially significant associations. Third, the specific areas of injury were not addressed and they should be explored in a future study. Fourth, intraoperative variables may modify the risk of IAPI and further studies are needed to assess their potential role. Finally, analyses stratified according to patient characteristics were not conducted because our analysis was based on comprehensive risk profiles.
Conclusions
In conclusion, the present study found that being overweight, use of prone or lateral positioning, use of a head frame, and longer surgery increased the risk of IAPI in patients undergoing neurosurgery. High serum albumin levels and use of a memory sponge pad were found to protect against IAPI. Prospective studies should be conducted to verify the findings from our study.
References
1. VanGilder C, Lachenbruch C, Algrim-Boyle C, The International Pressure Ulcer Prevalence survey: 2006–2015: A 10-year pressure injury prevalence and demographic trend analysis by care setting: J Wound Ostomy Continence Nurs, 2017; 44; 20-28
2. Bauer K, Rock K, Nazzal M, Pressure ulcers in the United States’ inpatient population from 2008 to 2012: Results of a retrospective nationwide study: Ostomy Wound Manage, 2016; 62; 30-38
3. VanGilder C, Amlung S, Harrison P, Results of the 2008–2009 International Pressure Ulcer Prevalence Survey and a 3-year, acute care, unit-specific analysis: Ostomy Wound Manage, 2009; 55; 39-45
4. Russo CA, Steiner C, Spector W, Hospitalizations related to pressure ulcers among adults 18 years and older, 2006: Statistical Brief #64: Healthcare Cost and Utilization Project (HCUP) Statistical Briefs, 2006, Rockville, MD, Agency for Healthcare Research and Quality
5. Spector WD, Limcangco R, Owens PL, Marginal hospital cost of surgery-related hospital-acquired pressure ulcers: Med Care, 2016; 54; 845-51
6. Chicano SG, Drolshagen C, Reducing hospital-acquired pressure ulcers: J Wound Ostomy Continence Nurs, 2009; 36; 45-50
7. Engels D, Austin M, McNichol L, Pressure ulcers: Factors contributing to their development in the OR: AORN J, 2016; 103; 271-81
8. Celik B, Karayurt Ö, Ogce F, The Effect of selected risk factors on perioperative pressure injury development: AORN J, 2019; 110; 29-38
9. Luo M, Long XH, Wu JL, Incidence and risk factors of pressure injuries in surgical spinal patients: A retrospective study: J Wound Ostomy Continence Nurs, 2019; 46; 397-400
10. Xiong C, Gao X, Ma Q, Risk factors for intraoperative pressure injuries in patients undergoing digestive surgery: A retrospective study: J Clin Nurs, 2019; 28; 1148-55
11. Chen Y, He L, Qu W, Predictors of intraoperative pressure injury in patients undergoing major hepatobiliary surgery: J Wound Ostomy Continence Nurs, 2017; 44; 445-49
12. Price MC, Whitney JD, King CA, Development of a risk assessment tool for intraoperative pressure ulcers: J Wound Ostomy Continence Nurs, 2005; 32; 19-30
13. Lumbley JL, Ali SA, Tchokouani LS, Retrospective review of predisposing factors for intraoperative pressure ulcer development: J Clin Anesth, 2014; 26; 368-74
14. Connor T, Sledge JA, Bryant-Wiersema L, Identification of pre-operative and intra-operative variables predictive of pressure ulcer development in patients undergoing urologic surgical procedures: Urol Nurs, 2010; 30; 289-95
15. Lu CX, Chen HL, Shen WQ, A new nomogram score for predicting surgery-related pressure ulcers in cardiovascular surgical patients: Int Wound J, 2017; 14; 226-32
16. Scott SM, Progress and challenges in perioperative pressure ulcer prevention: J Wound Ostomy Continence Nurs, 2015; 42; 480-85
17. Kayser SA, VanGilder CA, Lachenbruch C, Predictors of superficial and severe hospital-acquired pressure injuries: A cross-sectional study using the international pressure ulcer prevalence™ survey: Int J Nurs Stud, 2019; 89; 46-52
18. Sopher R, Nixon J, Gorecki C, Exposure to internal muscle tissue loads under the ischial tuberosities during sitting is elevated at abnormally high or low body mass indices: J Biomech, 2010; 43; 280-86
19. Linder-Ganz E, Gefen A, Mechanical compression-induced pressure sores in rat hindlimb: Muscle stiffness, histology, and computational models: J Applied Physiology, 2004; 96; 2034-49
20. Schoonhoven L, Defloor T, Grypdonck MH, Incidence of pressure ulcers due to surgery: J Clin Nurs, 2002; 11; 479-87
21. Iizaka S, Okuwa M, Sugama J, The impact of malnutrition and nutrition-related factors on the development and severity of pressure ulcers in older patients receiving home care: Clin Nutr, 2010; 29; 47-53
22. Taylor C, Importance of nutrition in preventing and treating pressure ulcers: Nurs Older People, 2017; 29; 33-39
In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387