07 March 2022: Animal Study
Astragalus Flavone Induces Proliferation and Differentiation of Neural Stem Cells in a Cerebral Infarction Model
Han Gao1CEF, Ningjing Huang1BE, Weiwei Wang1BC, Lingling Zhang1CD, Li Cai1D, Min Chen1F, Wentao Li1AG*DOI: 10.12659/MSM.933830
Med Sci Monit 2022; 28:e933830
Abstract
BACKGROUND: Ischemic cerebrovascular disease leads to the activation and differentiation of neural stem cells (NSCs) into mature neurons and glia cells to repair nerve damage. Astragalus flavone (ASF) has shown its potential role in proliferation and differentiation into dopamine neurons of NSCs.
MATERIAL AND METHODS: Cerebral infarction models were constructed to determine the effects of ASF on NSCs in vivo and in vitro.
RESULTS: ASF therapy had the ability to reduce the neurologic function scores and the cerebral infarction volume of the cerebral infarction model. Moreover, ASF was able to increase BrdU-positive cells and promote the expression of Nestin, β-Tubulin III, and O4, while decreasing the expression of GFAP. qRT-PCR and western blot assays showed ASF promoted the expression of Mash1, Math1, and Ngn2 mRNA and protein in cerebral infarction rats. Meanwhile, ASF (20 μg/ml) was able to increase EdU-positive cells and promote the expression of Nestin, β-Tubulin III, and O4 of NSCs at day14 in vitro. In normoxia, ASF obviously promoted the expression of Mash1, Ngn1, and Ngn2 mRNA and proteins, but in hypoxia, ASF promoted the expression of Notch1 and Math1 mRNA and proteins and inhibited the expression of Ngn1 and Ngn2 mRNA and proteins.
CONCLUSIONS: ASF therapy can improve the neurologic functions and reduce the cerebral infarction volume in a cerebral infarction model. Moreover, ASF promoted the proliferation of NSCs and induced differentiation into neurons and oligodendrocytes, which might be involved in regulating factors in Notch signaling.
Keywords: Astragalus Plant, Cell Proliferation, cerebral infarction, Flavonoids, Neural Stem Cells, Animals, Astrocytes, Disease Models, Animal, Flavones, Male, neurogenesis, Rats, Signal Transduction
Background
Cerebral infarction is one of the leading causes of death and disability worldwide. The main treatment is thrombolytic in early stages and neuroprotection. However, reconstruction of nerve function is still a challenge. Thus, discovering effective therapies has become urgent. Neural stem cells (NSCs), with their capacity for self-renew, migrate and differentiate into mature neurons and glia cells, and are a promising cell resource commonly employed in treatment of nerve damage [1]. In adult mammals, there are 2 aggregation areas in the central nervous system: the subgranular zone (SGZ) of the hippocampal dentate gyrus and the subventricular zone (SVZ) [2]. When activated by traumatic injury or ischemia, NSCs proliferate, differentiate, and migrate to the site of injury and the cerebral cortex of the granular layer, ultimately integrating into the neural network to repair nerve damage [3]. However, the proliferative ability of endogenous NSCs after brain ischemia is insufficient to compensate for central nervous system self-repair [4]. Therefore, promoting endogenous NSCs proliferation and differentiation into functional neurons is beneficial for reconstructing neurological functions.
Notch signaling had the ability to regulate the balance between NSC maintenance and differentiation [5,6]. In the absence of this pathway, all neural progenitors prematurely differentiate and deplete the NSC pool, consequently reducing the supply of new neurons.
Material and Methods
PREPARATION OF DRUGS:
ASF was provided by the Yuanye Bio-company (batch number: B20563; Shanghai, China). ASF was dissolved in 0.9% normal saline in an in vivo experiment and dissolved in Neurobasal medium in an in vitro experiment, and then diluted to corresponding concentrations.
ANIMALS:
Male Wistar rats (200–240 g) were purchased from Shanghai SLAC Laboratory Animal Co., Ltd. A total of 88 rats were included in the experiment; 24 rats were selected as the normal group and the other 64 were used to establish the MCAO model. All efforts were made to minimize the number of rats used and their suffering. Animal experiments were performed in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals and were approved by the Committee of the Shanghai Municipal Hospital of Traditional Chinese Medicine (#2018SHL-KYYS).
INTRALUMINAL MIDDLE CEREBRAL ARTERY OCCLUSION (MCAO) MODEL ESTABLISHMENT AND TREATMENT IN VIVO:
Male Wistar rats were housed in clear cages in a room with controlled temperature of 23±2°C and humidity of 55±5% with a regular 12-h light-dark cycle. After acclimatization for 3 days, the modified Longa suture method was used to establish the MCAO model [11,12]. The rats were anesthetized with 2% pentobarbital sodium 0.3 mL/100 g body weight, and no peritonitis was observed in rats after injection of pentobarbital sodium. Then, we cut along the midline of the neck using scissors to expose the trachea and separating the right common carotid artery (CCA) and internal carotid artery (ICA). Next, the external carotid artery (ECA) and CCA were ligatured, and ICA was clipped by vascular clamp. An arteriotomy was performed between the proximal ligated suture and the vascular clamp on the CCA with micro-scissors. The nylon monofilament (Diameter: 0.34±0.02 mm, length: 45 mm, 2634–50, Cinontech Co. Ltd., Beijing, China) was introduced in CCA and advanced until it reached the applied vessel clamp in ICA, then the vessel clamp was loosened and the nylon monofilament was kept inserted at about 18.5±1.5 mm. At last, we ligatured the ICA and sutured the wounds, and the rats were placed on an electric blanket to keep warm until they awoke. After building MCAO models for 24 h, neurological deficits were evaluated according to Longa’s behavioral test [11], which was used to evaluate spontaneous contralateral circling and tumbling. Rats with Longa’s score of 1–3 were used in follow-up experiments. Animals with Longa’s score of 0 and 4 were sacrificed by overdose of chloral hydrate.
After MCAO surgery for 1 day, the rats were equally and randomly divided into the following 3 groups: 1) Normal group (N=24): no operation, with 0.9% normal saline intraperitoneal injection once a day; 2) Model group (N=24): MCAO models with 0.9% normal saline intraperitoneal injection once a day; 3) ASF group (N=40): MCAO models with 2.5mg/kg ASF (N=8), 5 mg/kg ASF (N=24), 7.5mg/kg (N=8) ASF intraperitoneal injection once a day. All rats were involved in the final analysis.
EVALUATION INDEXES OF NEUROLOGIC FUNCTION:
All rats underwent neurobehavioral tests to evaluate functional outcome. The neurobehavioral tests were assessed on a 25-point scale. All rats underwent behavioral testing before and after the MCAO procedure for 24 h (day 0 and day 1), and at days 3, 7, 10, and 14 after MCAO. The scoring was as follows: 1) the Berderson behavioral test [13] (0 to 3 points), and screen test (0 to 3 points) were used to measure the palsy myodynamia and dystonia of contralateral limbs (0 to 5 points); 2) the beam walking test [14] was employed to exam the ability to walk on a wood beam 80 cm in length and 2.5 cm in width (0 to 5 points); 3) the body swing test (EBST) [5] was conducted to evaluate asymmetric motor behavior (0 to 4 points); 4) the pole climbing test [15] was performed to study motor coordination (0 to 5 points). The higher the score, the more serious the neurological damage. The investigator performing the tests was blinded to the experimental condition.
MEASUREMENT OF CEREBRAL INFARCT VOLUME (CIV):
After being treated by ASF for 7 days and 14 days, rats in the normal group, model group, and ASF group (5 mg/kg) (N=8 per group) were sacrificed and the brains were removed, then 40-mm coronal sections were prepared and stained with a 2% solution of 2, 3,5-Triphenyltetrazolium chloride (TTC) (Sigma, USA) for 30 min at 37°C in the dark. Areas that were not stained red with TTC were considered injured, which were calculated by Image Pro Plus 6.0 (Media Cybernetics, Maryland, USA). The percentage of cerebral infarction volume in rats was calculated as follows: percentage of cerebral infarction volume (%)=(cerebral infarct area/bilateral hemisphere area) ×100% [12,16].
IMMUNOFLUORESCENCE DETECTED THE PROLIFERATION AND DIFFERENTIATION OF NSCS IN SVZ OF BRAINS:
After being treated by ASF for 7 days and 14 days, rats in the normal group, model group, and ASF group (5 mg/kg) (N=8 per group) were anesthetized and perfused from the heart with 4% paraformaldehyde solution (PFA). The rats were injected with 5 mg/Kg 5-bromodeoxyuridine (BrdU) (Sigma, USA) 3 times before heart perfusion. Then, immunofluorescence (IF) was used to detect the proliferation and differentiation of NSCs. Nestin was detected to confirm that the derived cells were NSCs. BrdU is a synthetic nucleoside that is an analog of thymidine and is commonly used in the detection of proliferating cells in living tissues. Simultaneously, neuron marker β-Tubulin III, oligodendrocyte marker O4 and astrocyte marker glial fibrillary acidic protein (GFAP) were detected to confirm that NSCs have the ability to differentiate into neurons, oligodendrocytes, and astrocytes. For IF, rats in each group were anesthetized with 10% chloral hydrate and perfused from the heart with 200 mL of normal saline and 200 mL of 4% PFA. The brains were removed and prefixed in 4% PFA for 48 h and dehydrated in gradient ethanol, transparentized by xylene, embedded in paraffin, and cut coronally in the SVZ into pieces of 5-μm thickness, then adhered to the glass slices. Then immunofluorescence was performed as described previously [18]. Anti-Nestin antibody produced in rabbit (1: 100, Boster, Wuhan, China), anti-β-Tubulin III antibody produced in rabbit (1: 70, Sigma, Missouri, USA), anti-O4 antibody produced in mouse (1: 50, Sigma, Missouri, USA), and anti-GFAP antibody produced in mouse (1: 500, CST, Danvers, USA) were used as primary antibodies, and FITC- (1: 50) and Cy3- (1: 70) were used as secondary antibodies (Boster, Wuhan, China). DAPI (blue) was used to counterstain nuclei.
Fluorescent images were acquired under a Leica confocal microscope with LAS AF Lite software (QS00IW, LEICA, Wetzlar, Germany), and the positive cells was measured by Image Pro Plus 6.0 (Media Cybernetics, Maryland, USA). The effects of ASF on the NSCs proliferation and differentiation were evaluated by the ratio of positive cells/DAPI×100% in the same field of right SVZ.
WESTERN BLOTTING AND QUANTITATIVE REAL-TIME PCR (QRT-PCR) ANALYSIS IN SVZ OF BRAINS:
After being treated by ASF for 7 days and 14 days, rats in the normal group, model group, and ASF group (2.5 mg/kg, 5 mg/kg, and 7.5 mg/kg) (N=8 per group) were sacrificed and the brains were removed. Then, the cerebral subventricular zone of ischemic side of the brains were removed to detect the protein and mRNA levels of Notch1, Jagged1, Mash1, Math1, Ngn1, and Ngn2 by western blotting and qRT-PCR.
Western blotting was performed as described elsewhere [19]. In brief, the cerebral subventricular zone of ischemic side of rats were homogenized and cleaved with RIPA buffer (Yeasen Biotech Co., Ltd., Shanghai, China) including cocktail protease inhibitor PMSF (Boster, Wuhan, China) and centrifuged at 14 000 rpm for 15 min. The supernatant was collected and we quantified the protein concentration by BCA assay (Beyotime, Shanghai, China). Then, the supernatant was mixed with loading buffer (Yeasen Biotech Co., Ltd., Shanghai, China) and heated at 100°C for 10 min. Protein samples were separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to a poly-vinylidene fluoride membrane. The membrane was blocked with 5% BSA in Tris-buffered saline with Tween-20 (TBS-T) for 1 h and then incubated with anti-Notch1 (1: 1000, CST, Danvers, USA), anti-Jagged1 (1: 1000, CST, Danvers, USA), anti-Math1 (1: 1000, Abcam, Cambridge, UK), anti-Mash1 (1: 1000, Abcam, Cambridge, UK), anti-Ngn1 (1: 1000, Abcam, Cambridge, UK), anti-Ngn2 (1: 1000, Abcam, Cambridge, UK), and anti-β-actin (1: 1000, CST, Danvers, USA) antibodies at 4°C overnight. After being washed, the membranes were incubated with HRP-labeled goat IgG (H+L) (1: 1000, Beyotime, Shanghai, China) secondary antibody for 1 h at room temperature followed by washing and detection using an enhanced ECL system.
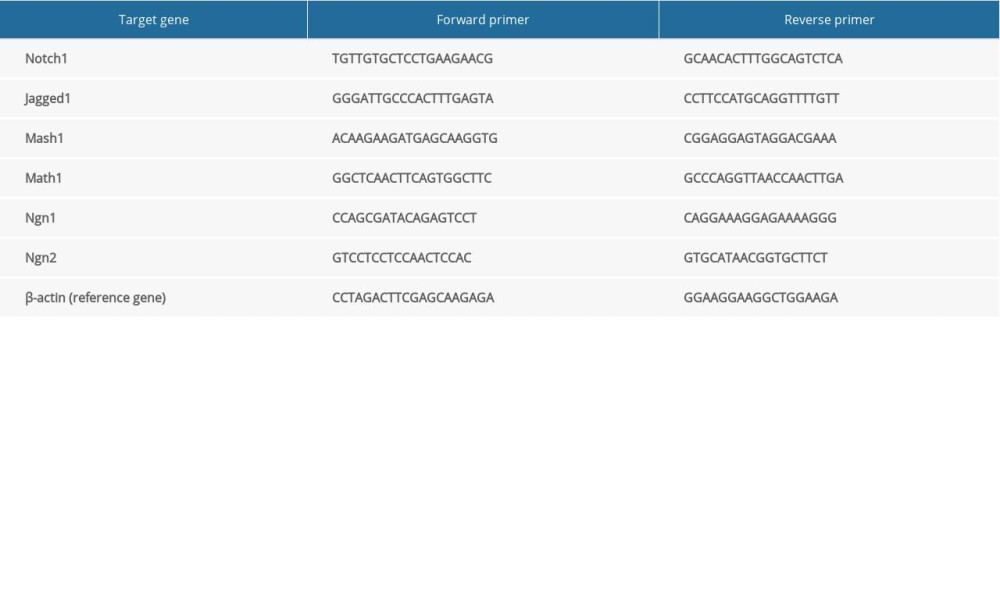
qRT-PCR was performed as described previously [20,21]. In brief, total RNA of cells was isolated by TRIZOL reagent (Invitrogen, New York, USA) and reverse-transcribed to synthesize cDNA using a PrimeScriptTM RT Master Mix kit (Perfect Real Time) (TaKaRa, Dalian, China). The SYBR Premix Ex TaqTM (Tli RNaseH Plus) kit (Takara, Dalian, China) was used to detect the expression of Notch1, Jagged1, Mash1, Math1, Ngn1, and Ngn2 mRNA. The primer sequences are listed in Table 1. According to the relative quantification 2−ΔΔCt method, the transcription expression of target genes could be determined when using β-actin as an internal reference.
PRIMARY NSCS CULTURE AND IDENTIFICATION:
At embryonic day 16.5 (E16.5), NSCs were isolated from mesencephalons of Wistar rat embryos (SLAC, Shanghai, China) using a method described previously [22]. We cultured 1×105 cells/mL in Neurobasal medium (Gibco, New York, USA) containing 2% B-27 without vitamin A (Gibco, New York, USA), 1% N2 (Invitrogen, New York, USA), Basic Fibroblast Growth Factor (bFGF, 10 ng/mL, Gibco, New York, USA), Epidermal Growth Factor (EGF, 20 ng/mL, PeproTech, Rocky Hill, USA), and penicillin (100 U/mL)/streptomycin (100 μg/mL) (Boster, Wuhan, China). NSCs were cultured in a 37°C incubator with 5% CO2. We replaced half of the medium every other day, and then passaged cells every 7 days. The expression of Nestin was detected to confirm that the derived cells were NSCs.
IMMUNOCYTOCHEMICAL DETECTED THE PROLIFERATION AND DIFFERENTIATION OF NSCS IN VITRO:
After passing for 2 generations, NSCs were treated with 0 μg/mL, 10 μg/mL, 20 μg/mL, and 40 μg/mL of ASF for 7 days and 14 days, and immunocytochemical (ICC) analysis was performed to identify the effect of ASF on the proliferation and differentiation of NSCs in vitro, and the optimal dose would be chosen. Cell proliferation was detected by 5-ethynyl-2′-deoxyuridine (EdU) staining. EdU is the analog of BrdU but requires no heat or acid treatment. After ASF intervention for 7 days and 14 days, neurospheres were seeded in 48-well plates coated with polylysine in a humidified 37°C incubator for 24 h, and then were fixed with paraformaldehyde. Before the cells were fixed with paraformaldehyde, they were pretreated with 10 mM EdU (Riobio, Guangzhou, China) for 5 days. After that, EdU was detected by ICC. The detection method was performed using a Cell-Light EdU Apollo 488 In Vitro Kit (Riobio, Guangzhou, China). In brief, excessive aldehyde groups were neutralized by glycine to ensure the staining reaction system. Then, cells were incubated in PBS containing 0.5% TritonX-100 for 10 min. After washing with PBS, cells were dyed with reaction solution by Apollo staining. The result was evaluated by the color intensity ratio of EdU-positive cells/DAPI×100% in the same field.
The NSCs marker Nestin was detected to confirm the expression of NSCs, and neuron marker β-Tubulin III, oligodendrocyte marker O4, and GFAP were detected to confirm the capacity of NSCs to differentiate into neurons, oligodendrocytes, and astrocytes by ICC. The antibodies were as in the previous experiment. The results were evaluated by the ratio of positive cells/DAPI×100% in the same field.
IDENTIFICATION OF THE RELEVANT MRNA AND PROTEIN IN NORMOXIA AND HYPOXIA:
NSCs were cultured as previously described. After passing for 2 generations, NSCs were plated into 6-well plates at a density of 1×105 cells/mL and cultured for 24 h. Then, NSCs were divided into 4 groups: 1) in the normoxia group, NSCs were cultured with complete medium in a 37°C incubator with 5% CO2; 2) in the normoxia+ASF group, NSCs were cultured with complete medium containing ASF (20 μg/mL) in a 37°C incubator with 5% CO2; 3) in the hypoxia group, NSCs were cultured with complete medium in a 37°C incubator with 5% CO2, 2.5% O2 and 92.5% N2 for 4 h, and then cultured in a 37°C incubator with 5% CO2; and 4) in the hypoxia+ASF group, NSCs were cultured with complete medium containing ASF in a 37°C incubator with 5% CO2, 2.5% O2, and 92.5% N2 for 4 h, and then cultured in a 37°C incubator with 5% CO2. After 5 days, β-Tubulin III was detected by western blotting to ensure the neurons’ expression, and the relevant factors such as Notch1, Jagged1, Mash1, Math1, Ngn1, and Ngn2 mRNA and proteins were detected by qRT-PCR and western blotting. The methods used were as previously described.
STATISTICAL ANALYSIS:
All statistical analyses were performed using SPSS 21.0 software (SPSS, Inc., Chicago, USA). Differences among groups were evaluated using one-way analysis of variance followed by least significant difference (LSD) tests. Differences in mean values were considered significant when the
Results
ASSESSMENT OF NEUROLOGIC FUNCTIONS AND CEREBRAL INFARCT VOLUME:
As shown in Figure 1A, the body weights of normal rats increased, while weights of MCAO rats declined in a time-dependent manner. The weights in the ASF group also declined, but the decrease in rate of weight loss slowed from day 3. For neurobehavioral testing, the neurobehavioral score before MCAO models were established was 0. The average score of the model group decreased in the first 7 days after surgery, then the score increased and kept stable from day 7. The average score of the ASF group also declined in the first 7 days, which was consistent with the model group. As time went on, the score of the ASF group was significantly decreased compared with the model group at days 10 and 14 (Figure 1B). These results showed ASF could improve neurologic functions of cerebral infarction model rats.
TTC staining was used to demonstrate the effects of ASF on cerebral infarction area. The ischemic area was distinguished as a pale zone while the normal area was distinguished as a red zone (Figure 1C). In the model group, at day 7, the cerebral infarct volume was 23.95±3.09%, and at day 14, the volume was declined to 15.24±1.89%. Notably, at 7 days after ASF treatment, the cerebral infarct volume was 15.39±0.21%, which was significantly decreased compared with the model group at day 7. After ASF treatment for 14 days, the cerebral infarction volume had declined to 11.99±1.48% (Figure 1D). These results indicated that ASF was effective in reducing the cerebral infarction area.
EFFECT OF ASF ON THE PROLIFERATION AND DIFFERENTIATION OF NSCS IN CEREBRAL INFARCTION RATS:
Further, the effect of ASF on the proliferation and differentiation of NSCs in SVZ of cerebral infarction models were determined using ICC (Figure 2A). At day 7, the expression levels of BrdU and Nestin in the model group were significantly increased while β-Tubulin III levels were inhibited compared with the normal group. After ASF treatment, the expression levels of Nestin and β-Tubulin III were increased compared to the model group. At day 14, BrdU, Nestin, and β-Tubulin III levels in the model group were similar to those at day 7. As expected, they were obviously increased after treament with ASF at day 14 (Figure 2B).
Results from Figure 3A, 3B indicated that the levels of GFAP (an astrocyte marker) in MCAO model rats were significantly enhanced and O4 levels (an oligodendrocyte marker) were reduced at days 7 and 14 compared with the normal group. ASF could effectively upregulate O4 level and downregulate GFAP level compared with the model group (Figure 3B). These results indicated that ASF could promote NSC proliferation and differentiation into neurons and oligodendrocytes and inhibit NSCs differentiation into astrocytes, which was beneficial for repairing brain cell injury induced by cerebral infarction.
ASF REGULATED RELATED FACTORS IN CEREBRAL INFARCTION RATS:
To explore the potential mechanism by which ASF promoted the proliferation and differentiation of NSCs in cerebral infarction rats, we further analyzed related mRNA and protein levels in the Notch pathway by qRT-PCR and western blot (Figure 4). We divide the drug into 3 concentrations: 2.5 mg/mL, 5 mg/mL, and 7.5 mg/mL. In our preliminary experiment, we verified that ASF in 5 mg/mL had a stable effect on the proliferation and differentiation of NSCs, so we selected 5 mg/mL ASF for analysis. Seven days after building MCAO models, the expression of Mash1 and Ngn2 were higher while Math1 was lower. Over time, the expression of Mash1 was reduced while Math1 was increased at day 14. After ASF treatment, the expression of Mash1 was increased while Ngn2 was decreased at day 7, and the expression of Mash1, Math1, and Ngn2 were significantly higher at day14 compared with the model group.
ASF PROMOTED THE PROLIFERATION AND DIFFERENTIATION OF NSCS IN VITRO:
Neurospheres were formed after culturing NSCs for 3 days, and the positive expression of Nestin confirmed that these derived cells were NSCs (Figure 5A). In terms of NSCs proliferation, the expression of EdU was clearly different compared with the control after treatment with 20 μg/mL and 40 μg/mL ASF at day 7 and 40 μg/mL ASF at day 14 (Figure 5B, 5C). Accordingly, the expression of Nestin was obviously different compared with control after treament with 40 μg/mL ASF at day 7 and 20 μg/mL and 40 μg/mL ASF at day 14 (Figure 6A, 6B). In addition, within a certain range, the higher the dose, the higher the expressions of EdU and Nestin. In terms of differentiation, compared with the control group, the β-Tubulin III and O4 levels obviously increased after 10 μg/mL, 20 μg/mL, and 40 μg/mL ASF treatment for 7 days, and 10 μg/mL and 20 μg/mL ASF treatment for 14 days. Simultaneously, the O4 level was increased after 40 μg/mL ASF treatment for 7 days compared with the control group (Figure 6A, 6B).
Conclusively, these results demonstrated that ASF was able to promote NSCs proliferation and differentiation into neurons and oligodendrocytes. ASF in middle and high doses (20 μg/mL and 40 μg/mL) had obvious effects on the proliferation of NSCs, and in low and middle doses (10 μg/mL and 20 μg/mL) it had obvious effects on the differentiation of NSCs in vitro. Therefore, in the middle dose (20 μg/mL) of ASF had a stable effect on the proliferation and differentiation of NSCs.
ASF REGULATED RELATED MRNA AND PROTEIN EXPRESSION IN HYPOXIA CONDITION:
As in the previous experiment, ASF (20 μg/mL) induced the proliferation and differentiation of NSCs. The result of western blot also showed ASF promoted the expression of β-Tubulin III (Figure 7C). However, in hypoxia condition, NSCs over-differentiated as the stress response (Figure 7A, 7C), and the expression of β-Tubulin III was significantly increased. After ASF treatment in hypoxia condition, the expression of β-Tubulin III was lower than in the hypoxia model. To further explore the mechanism of NSCs differentiation after ASF treatment in normoxia and hypoxia conditions, relative mRNA and proteins were detected. As displayed in Figure 7B and 7C, in normoxia, ASF obviously promoted the expression of Mash1, Ngn1, and Ngn2 both at RNA and protein levels compared with the normal group. The expression of Notch1 and Jagged1 were slightly increased at the RNA level after ASF treatment, but the difference was not significant, while at the protein level, they showed a significant difference. In hypoxia, the mRNA and protein levels of Notch1, Jagged1, Mash1, Math1, Ngn1, and Ngn2 mRNA and proteins were all overexpressed as the stress response compared with normoxia. When treated with ASF, the expression of Notch1 and Math1 mRNA and proteins were increased while Ngn1 and Ngn2 mRNA and proteins were decreased.
Discussion
Cerebral infarction is a major challenge to human health. With the increasing demand for functional recovery after stroke, it is necessary to find new strategies. NSCs participate in nerve regeneration, and cerebral ischemic injury activate NSCs and promote the proliferation and differentiation of NSCs thus repair the brain function. However, the proliferative and differentiative ability of endogenous NSCs is limited. Therefore, promoting NSCs proliferation and differentiation into functional neurons is beneficial for reconstructing neurological functions. ASF has been demonstrated its effect on the proliferation of NSCs in cerebral infarction rats in our previous study, and this study aimed to further examine the effects of ASF on neural functional recovery of cerebral infarction rats and the proliferation and differentiation of NSCs in vivo and in vitro.
Hemiplegia occurred in the cerebral infarction rats, and the activity of the rats decreased significantly, and the feeding capacity also decreased, which lead to the weight of the rats declined. After treating with ASF, the activity of the rats was obviously enhanced. Meanwhile, we found that the weights of the cerebral infarction rats were significantly higher than model rats. On the one hand, the increasing weight of rats in ASF group indirectly showed their better ability to take food than model group. On the other hand, astragalus has been widely used as a tonic to enhance the body’s defenses in Traditional Chinese medicine [23]. Bioactive substances of astragalus include polysaccharides, triterpene glycosides, and flavonoids. Flavonoids are a class of polyphenolic secondary metabolites, which have been demonstrated to have the effects of immune enhancement, antioxidant, antihypertensive, antiaging, antifatigue, and anti-inflammatory activities, thus reduced energy consumption and provided energy support, which may be related to the weight gain of rats [24].
Then, we further studied the effect of ASF on cerebral infarct volume. After cerebral infarction, brain cells were necrotic and nerve function was damaged due to cerebral ischemia and hypoxia. At 7 days after cerebral infarction, the areas of cerebral infarction in ASF group were significantly smaller than in the model group, which proved that ASF alleviated cerebral necrosis and promoted recovery of nerve function. At 14 days after cerebral infarction, the areas of cerebral infarction began to repair due to the establishment of cerebral collateral circulation, and the volume of cerebral infarction was smaller than at 7 days. Although the cerebral infarction area of the model group was significantly reduced at 14 days, the neurological function was obviously impaired. These indicated that ASF was an effective therapy in promoting the recovery of nerve cells, reducing the cerebral infarction area, and improving neurologic functions, thus improving the prognosis and reducing the disability rate of cerebral infarction. Because nerve cells are sensitive to ischemia and hypoxia, and early intervention can reduce brain cell death, ASF treatment is an important early treatment of cerebral infarction.
After brain injury, neurons and oligodendrocytes die, and astrocytes proliferate in the damaged area to form a glial “scar”. This proliferation is accompanied by increased expression of glial fibrillary acidic protein (GFAP). Therefore, increased GFAP in tissue is a signal of central nervous system response to injury. Meanwhile, NSCs are activated and proliferate to repair the injured cells. However, the endogenous NSCs is insufficient to achieve self-repair. After treatment with ASF, the expression of NSCs, neurons, and oligodendrocytes was increased and expression of astrocytes was reduced compared with the cerebral infarction model. These may be involved in ASF promoting the proliferation of NSCs and differentiation into neurons and oligodendrocytes, and reduced the death of neurons and oligodendrocytes, thus protecting the brain from ischemia and hypoxia. The cells experiment also showed that ASF had an obvious effect on the proliferation and differentiation into neurons and oligodendrocytes of NSCs, while at a high dose, ASF played a major role in promoting the proliferation of NSCs.
In the process of proliferation and differentiation of NSCs, the Notch signal pathway plays a crucial role in embryonic and adult brains. The components of the pathway were widely expressed, including Notch receptor, Notch ligand (such as Jagged1 and 2), and proneural genes (such as Mash1, Math1, Ngn1, and Ngn2) [25]. After activating the Notch signal pathway, the transmembrane protein Notch releases the Notch intracellular domain (NICD), then moves into the nucleus and forms a NICD-RBPJ complex, which inhibits expression of bHLH family factors, which represses the expression of Notch ligand genes and proneural genes, thereby remaining as neural progenitor cells. The factors Mash1, Math1, Ngn1, and Ngn2 all belong to the early-expressed sub-group of bHLH factors. These factors may play a role in the types of neurons derived from progenitor populations [26]. At embryonic stages, in the spinal neural tube, Math1 is the most dorsally expressed gene, Ngn1 and Ngn2 are adjacent to this domain, and Mash1 is directly adjacent to the ventral boundary of the Ngn1 domain. Expression of these genes define 3 distinct, non-overlapping populations of neural progenitor cells. These factors may play a role in the types of neurons derived from these progenitor populations. Gain-of-function studies in multiple systems provided evidence that Math1 can induce neuronal differentiation [27]. Mash1, also known as Ascl1, is a key gene for the early development of mammalian neurons from pluripotent cells to neural progenitor cells to neurons. A study demonstrated that Mash1 mutant mice presented a severe loss of progenitors, particularly of neuronal precursors in the subventricular zone of the medial ganglionic eminence [28]. Mash1 was shown to convert mouse embryonic and postnatal fibroblasts into induced neurons [29]. During development, Mash1 directs neurogenesis and oligodendrogenesis. A study showed Mash1 lineage cells contribute to stroke-induced neurogenesis and oligodendrogenesis in the ipsilateral SVZ, striatum, and corpus callosum of the adult ischemic brain [30,31]. Another study found that actively proliferating SVZ cells expressing Ascl1 and Ascl1 lineage cells are fated to become neurons in the olfactory bulb and oligodendrocytes in the corpus callosum, but not astrocytes [32,33]. Ngn1 and Ngn2 encode bHLH transcription factors expressed in the developing neocortex. In the developing mammalian cerebral cortex, Ngn1 and Ngn2 are expressed only in the areas that have neuroepithelial precursor cells, such as the cortex and ventricle, and are expressed only during neuronal development [26]. Furthermore, related studies indicated that mRNA and protein levels of Ngn1 were increased during neurogenesis but dramatically decreased during astrogliogenesis. Both neurons and astrocytes originate from the same group of stem cells, which first differentiate into neurons and then switch to astrocytes [34]. Ngn1 induced neurogenesis and restrained the formation of astrocytes by regulating particular genes [35] and suppressed gliogenesis by sequestering CREB-binding protein/p300 and Smad1 away from the signal complexes that are required for the expression of glial fibrillary acidic protein [36,37]. Ngn2 was most closely related to Ngn1 and partially overlaps with Ngn1 and Mash1 [38].
An in vitro experiment, in the condition of hypoxia, showed that NSCs were sharply differentiated into neurons, and the related factors Notch1, Jagged1, Math1, Mash1, Ngn1, and Ngn2 were all significantly increased in vitro, perhaps because NSCs are quickly exhausted. After ASF treatment, the neurons were decreased under hypoxia and increased under normoxia. Meanwhile, Notch1, Math1 and Mash1 were increased while Ngn1 and Ngn2 were reduced after ASF treatment in hypoxia. ASF may protect NSCs from over-differentiating into neurons and consuming NSCs to balance the proliferation and differentiation in hypoxia. The long-term efficacy of ASF in NSCs in hypoxia needs further study. Our in vivo experiment showed that, after cerebral infarction, Math1 and Ngn2 increased while Notch, Jagged1 and Mash1 decreased. Over4 time, Notch1, Math1, Ngn1, and Ngn2 all decreased, suggesting a reduced ability os NSCs to differentiate. After treated with ASF, the expression of Math1, Mash1, and Ngn2 were significantly increased compared with the model group at day 14, which corresponds with the expression of neurons and oligodendroglia cells. This also means ASF had long-term efficacy in promoting NSCs to differentiate into neurons and oligodendroglia cells. Due to the complexity of the organic environment, the results were somewhat different between in vivo and in vitro experiments. In the treatment of cerebral infarction with ASF, there may be other factors involved in the proliferation and differentiation that need further study.
Conclusions
In conclusion, ASF therapy had the ability to improve the neurologic functions and reduce the cerebral infarction volume in a cerebral infarction model. Moreover, ASF was able to promote the proliferation of NSCs and induce differentiation into neurons and oligodendrocytes, which might be related with regulating factors in Notch signaling. There may be other factors involved in the proliferation and differentiation that need further study.
Figures
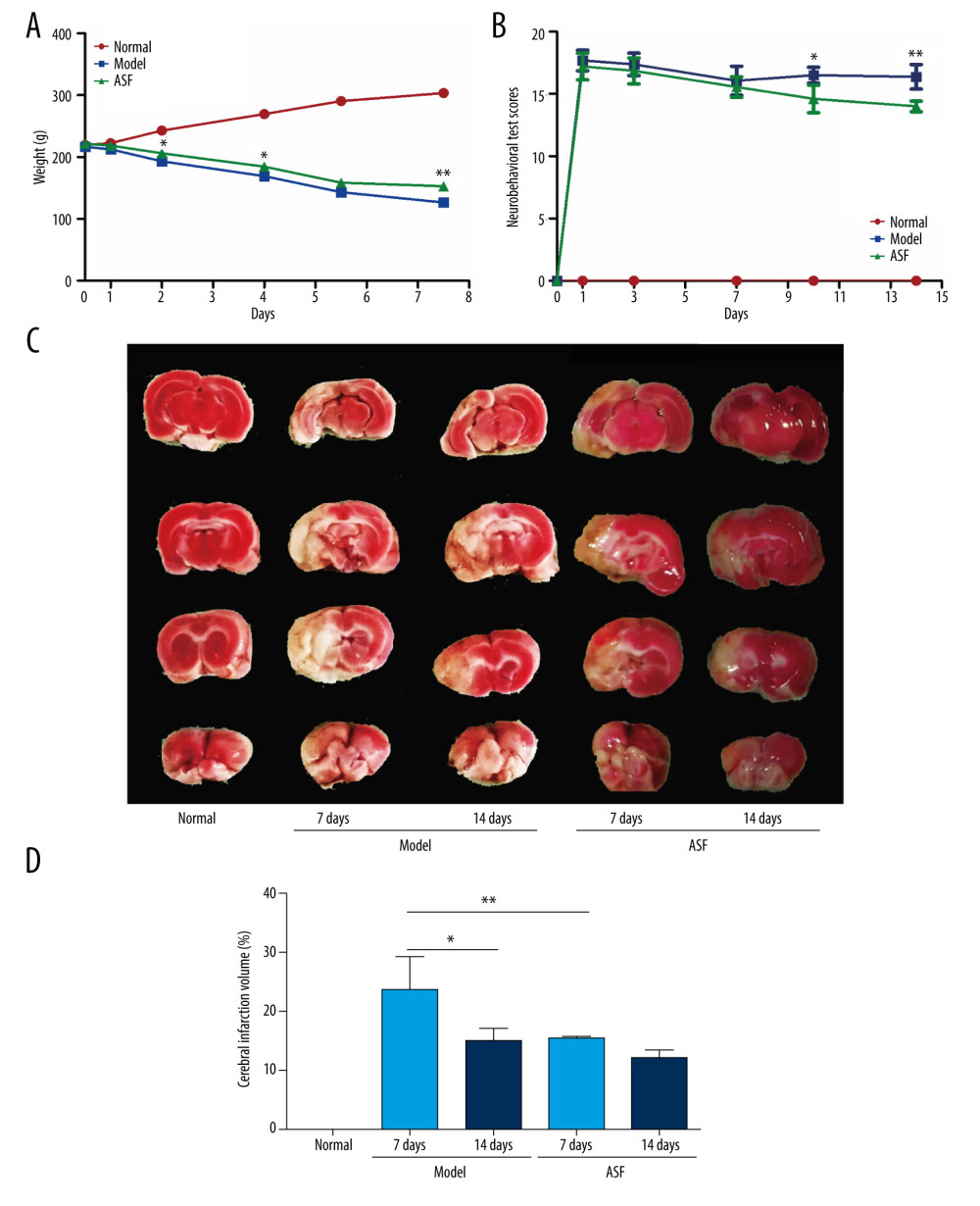 Figure 1. The average weight, neurobehavioral score, and cerebral infarction volume in each group (A) The average rat weight in normal, model, and ASF groups at days 0, 1, 3, 7, 10, and 14 (n=8 per group), * P<0.05, ** P<0.01. (B) The score of neurobehavioral tests in normal, model, and ASF groups at days 0, 3, 7, 10, and 14 (n=8 per group), * P<0.05, ** P<0.01. (C) Brains stained by TTC in normal, model (days 7 and 14), and ASF (days 7 and 14) groups. (D) Statistic analysis of cerebral infarction volume. Data are presented as mean±SEM, * P<0.05, ** P<0.01.
Figure 1. The average weight, neurobehavioral score, and cerebral infarction volume in each group (A) The average rat weight in normal, model, and ASF groups at days 0, 1, 3, 7, 10, and 14 (n=8 per group), * P<0.05, ** P<0.01. (B) The score of neurobehavioral tests in normal, model, and ASF groups at days 0, 3, 7, 10, and 14 (n=8 per group), * P<0.05, ** P<0.01. (C) Brains stained by TTC in normal, model (days 7 and 14), and ASF (days 7 and 14) groups. (D) Statistic analysis of cerebral infarction volume. Data are presented as mean±SEM, * P<0.05, ** P<0.01. 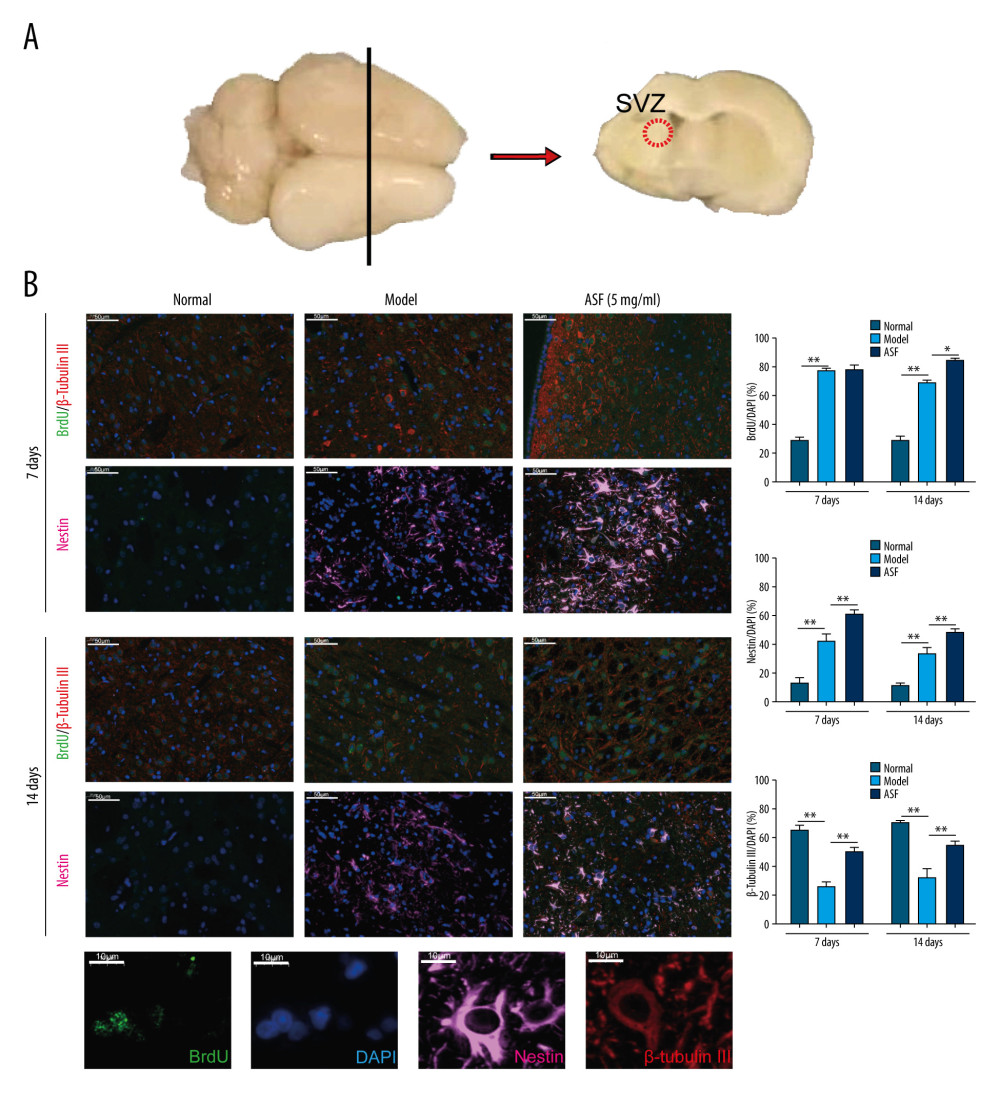 Figure 2. ASF induced NSC proliferation and differentiation into neurons in SVZ (A) The SVZ is shown in the coronal section of rat brain after heart perfusion. (B) Representative IF images of BrdU (green), Nestin (pink), β-Tubulin III (red), and DAPI (blue) in normal, model and ASF groups at days 7 and 14. Scale bar: 50 μm. The results are shown as bar graphs. Data are presented as mean±SEM, * P<0.05, ** P<0.01.
Figure 2. ASF induced NSC proliferation and differentiation into neurons in SVZ (A) The SVZ is shown in the coronal section of rat brain after heart perfusion. (B) Representative IF images of BrdU (green), Nestin (pink), β-Tubulin III (red), and DAPI (blue) in normal, model and ASF groups at days 7 and 14. Scale bar: 50 μm. The results are shown as bar graphs. Data are presented as mean±SEM, * P<0.05, ** P<0.01. 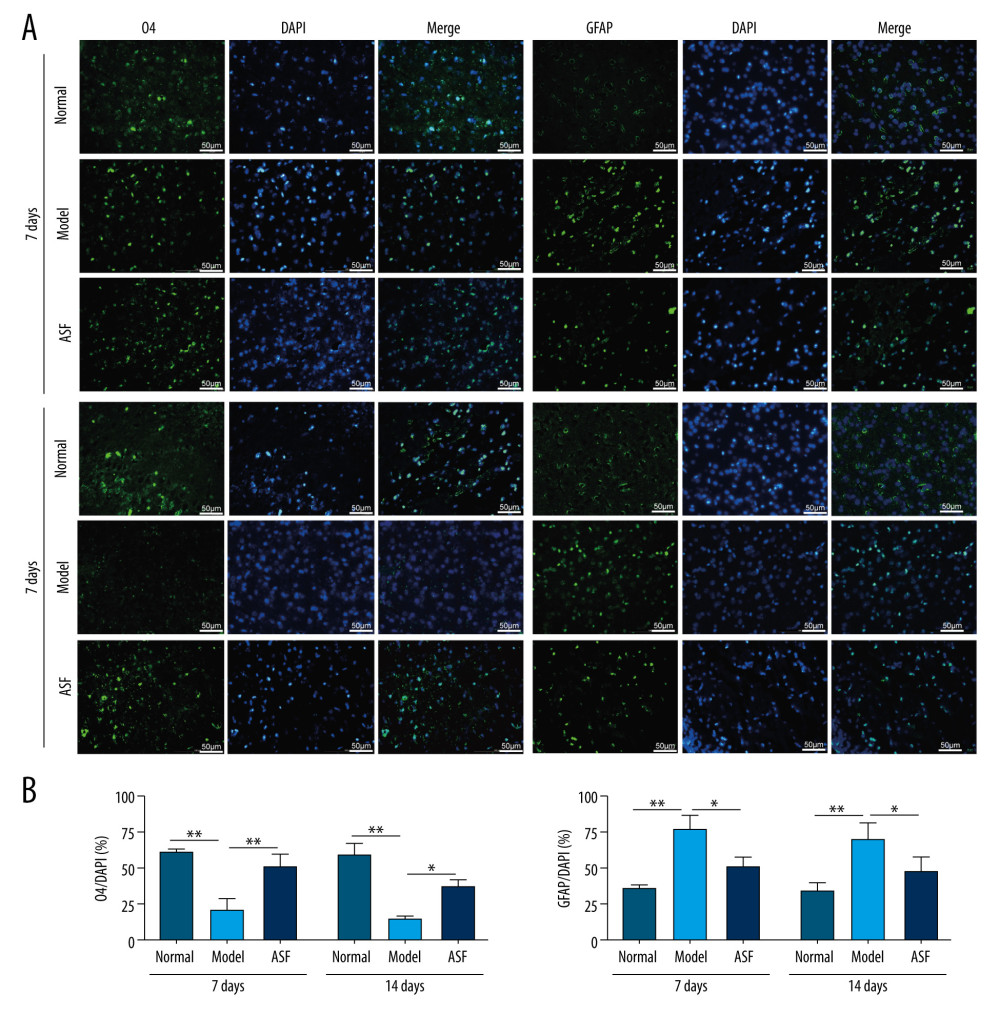 Figure 3. ASF induced NSC differentiation into oligodendrocytes and inhibited NSC differentiation into astrocytes in SVZ (A) Representative IF images of O4 (green), GFAP (green), and DAPI (blue) in normal, model and ASF groups at days 7 and 14. Scale bar: 50 μm. (B) The results were showed as the bar graphs. Data are presented as mean±SEM, * P<0.05, ** P<0.01.
Figure 3. ASF induced NSC differentiation into oligodendrocytes and inhibited NSC differentiation into astrocytes in SVZ (A) Representative IF images of O4 (green), GFAP (green), and DAPI (blue) in normal, model and ASF groups at days 7 and 14. Scale bar: 50 μm. (B) The results were showed as the bar graphs. Data are presented as mean±SEM, * P<0.05, ** P<0.01. 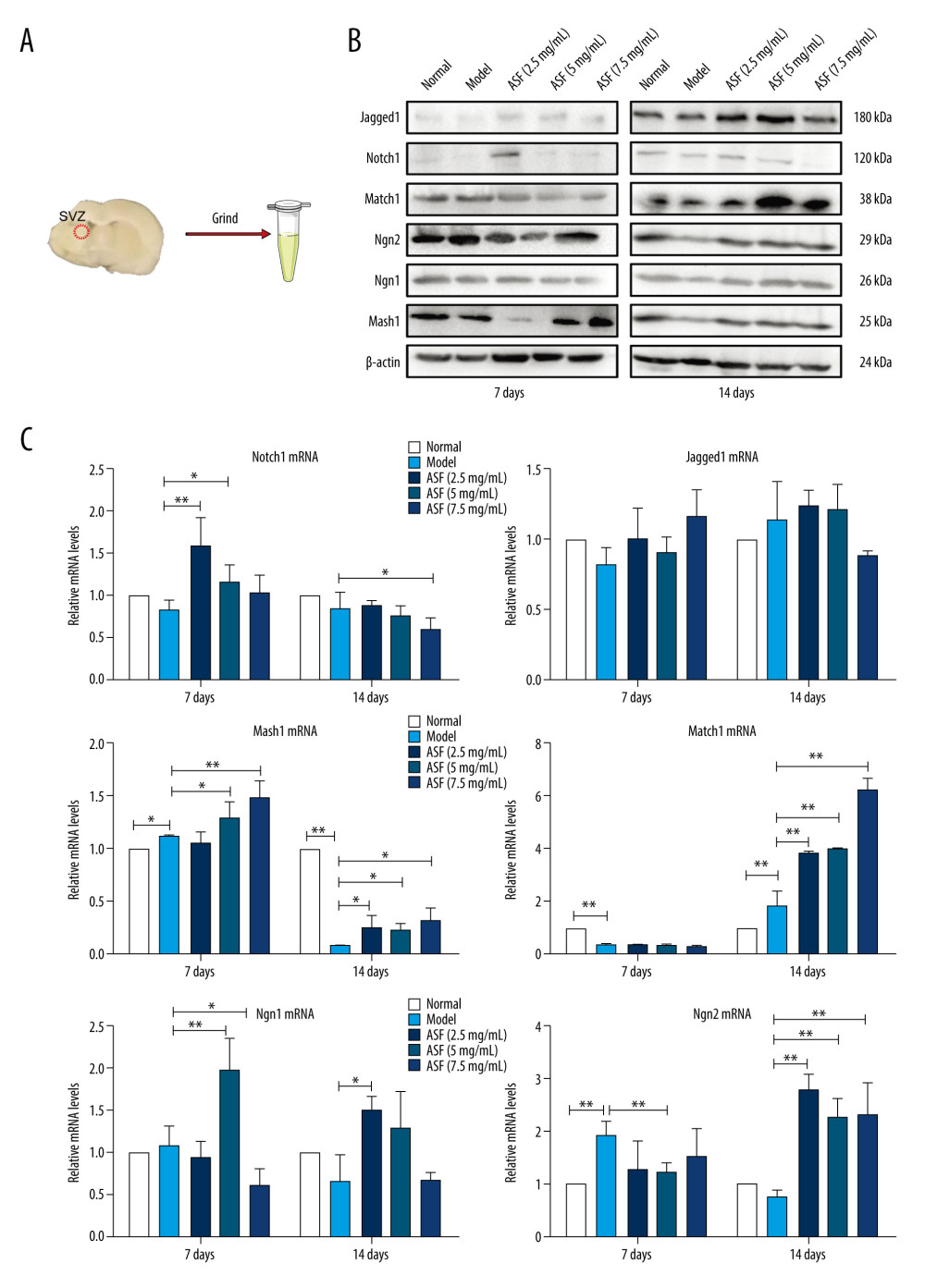 Figure 4. The expression of relevant mRNA and proteins in SVZ of brain (A) Tissues of rat brains in SVZ were taken and were used for western blotting and qRT-PCR. (B) The expression of Notch1, Jagged1, Math1, Mash1, Ngn1, and Ngn2 protein by western blotting. (C) The expression of Notch1, Jagged1, Math1, Mash1, Ngn1, and Ngn2 mRNA by qRT-PCR method. Data are presented as mean±SEM. * P<0.05, ** P<0.01.
Figure 4. The expression of relevant mRNA and proteins in SVZ of brain (A) Tissues of rat brains in SVZ were taken and were used for western blotting and qRT-PCR. (B) The expression of Notch1, Jagged1, Math1, Mash1, Ngn1, and Ngn2 protein by western blotting. (C) The expression of Notch1, Jagged1, Math1, Mash1, Ngn1, and Ngn2 mRNA by qRT-PCR method. Data are presented as mean±SEM. * P<0.05, ** P<0.01. 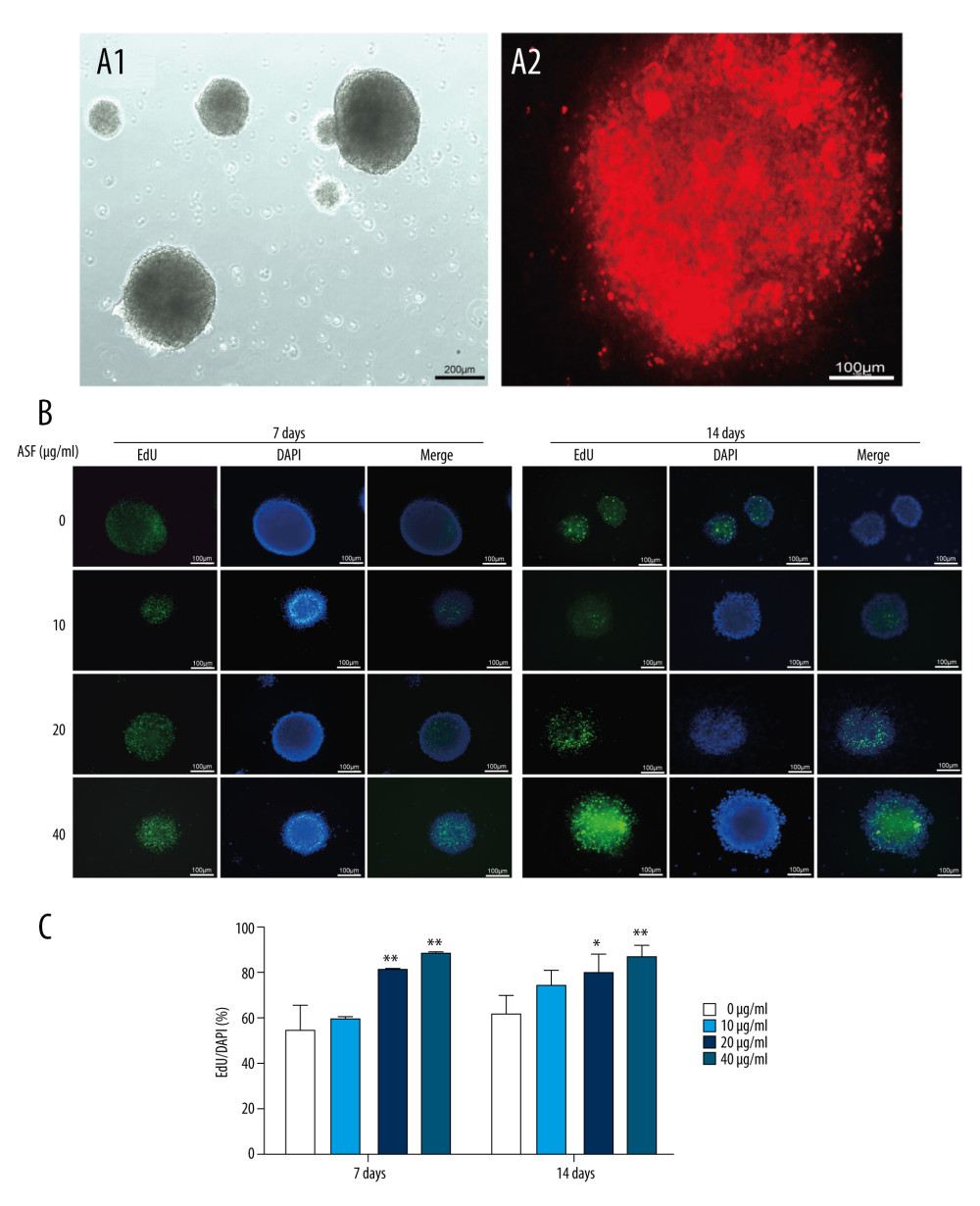 Figure 5. ASF induced NSCs proliferation in vitro (A) The bright field image of neurospheres after culturing for 3 days (A1, Scale bar: 200 μm), and the representative IF image of Nestin confirmed that these derived cells were NSCs (A2, Scale bar: 100 μm). (B) Representative IF images of EdU+ cells treated with ASF at concentrations of 0, 10, 20 and 40 μg/mL. DAPI was used to counterstain nuclei. Scale bar: 100μm. (C) The number of EdU+ cells. Data are presented as mean±SEM, * P<0.05, ** P<0.01.
Figure 5. ASF induced NSCs proliferation in vitro (A) The bright field image of neurospheres after culturing for 3 days (A1, Scale bar: 200 μm), and the representative IF image of Nestin confirmed that these derived cells were NSCs (A2, Scale bar: 100 μm). (B) Representative IF images of EdU+ cells treated with ASF at concentrations of 0, 10, 20 and 40 μg/mL. DAPI was used to counterstain nuclei. Scale bar: 100μm. (C) The number of EdU+ cells. Data are presented as mean±SEM, * P<0.05, ** P<0.01. 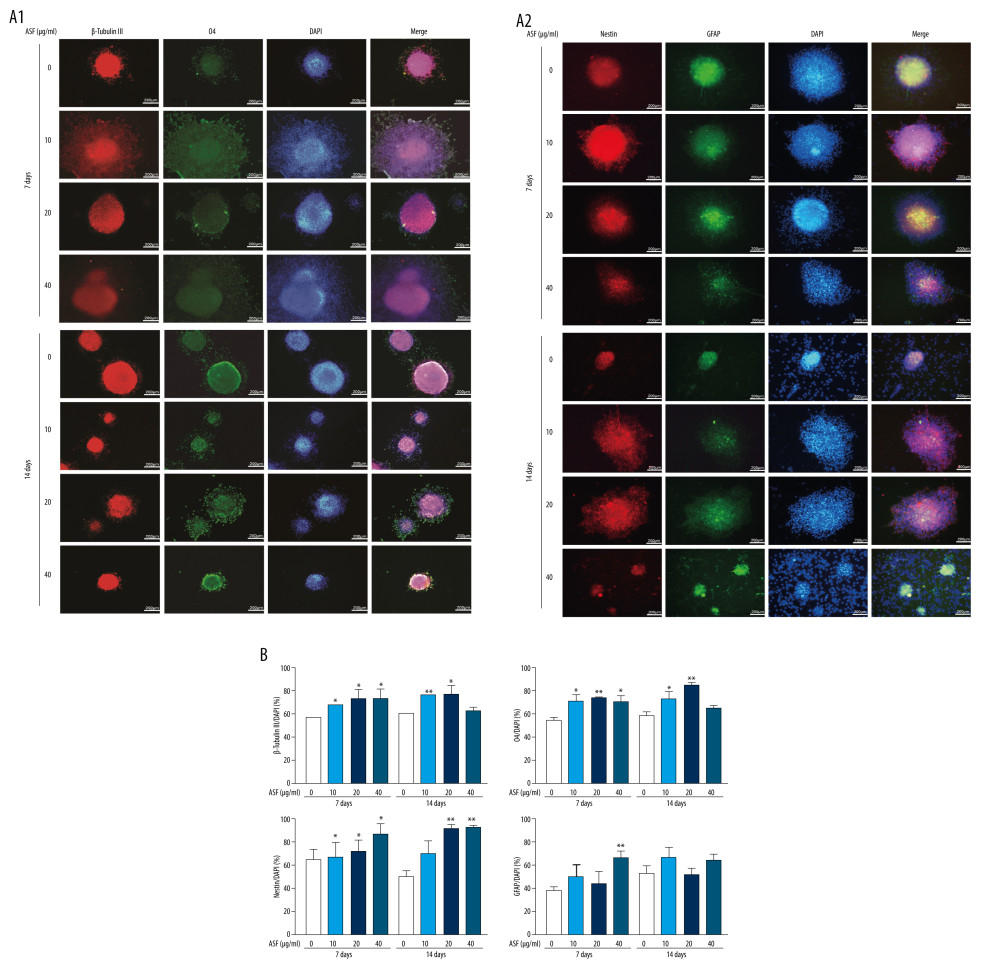 Figure 6. ASF induced differentiation of NSCs in vitro (A1, A2) Representative ICC images of β-Tubulin III (red), O4 (green), Nestin (red), GFAP (green), and DAPI (blue) at days 7 and 14. Scale bar: 200 μm. (B) The result of relativeβ-Tubulin III, O4, Nestin and GFAP positive cells. Data are represented as mean±SEM, * P<0.05, ** P<0.01.
Figure 6. ASF induced differentiation of NSCs in vitro (A1, A2) Representative ICC images of β-Tubulin III (red), O4 (green), Nestin (red), GFAP (green), and DAPI (blue) at days 7 and 14. Scale bar: 200 μm. (B) The result of relativeβ-Tubulin III, O4, Nestin and GFAP positive cells. Data are represented as mean±SEM, * P<0.05, ** P<0.01. 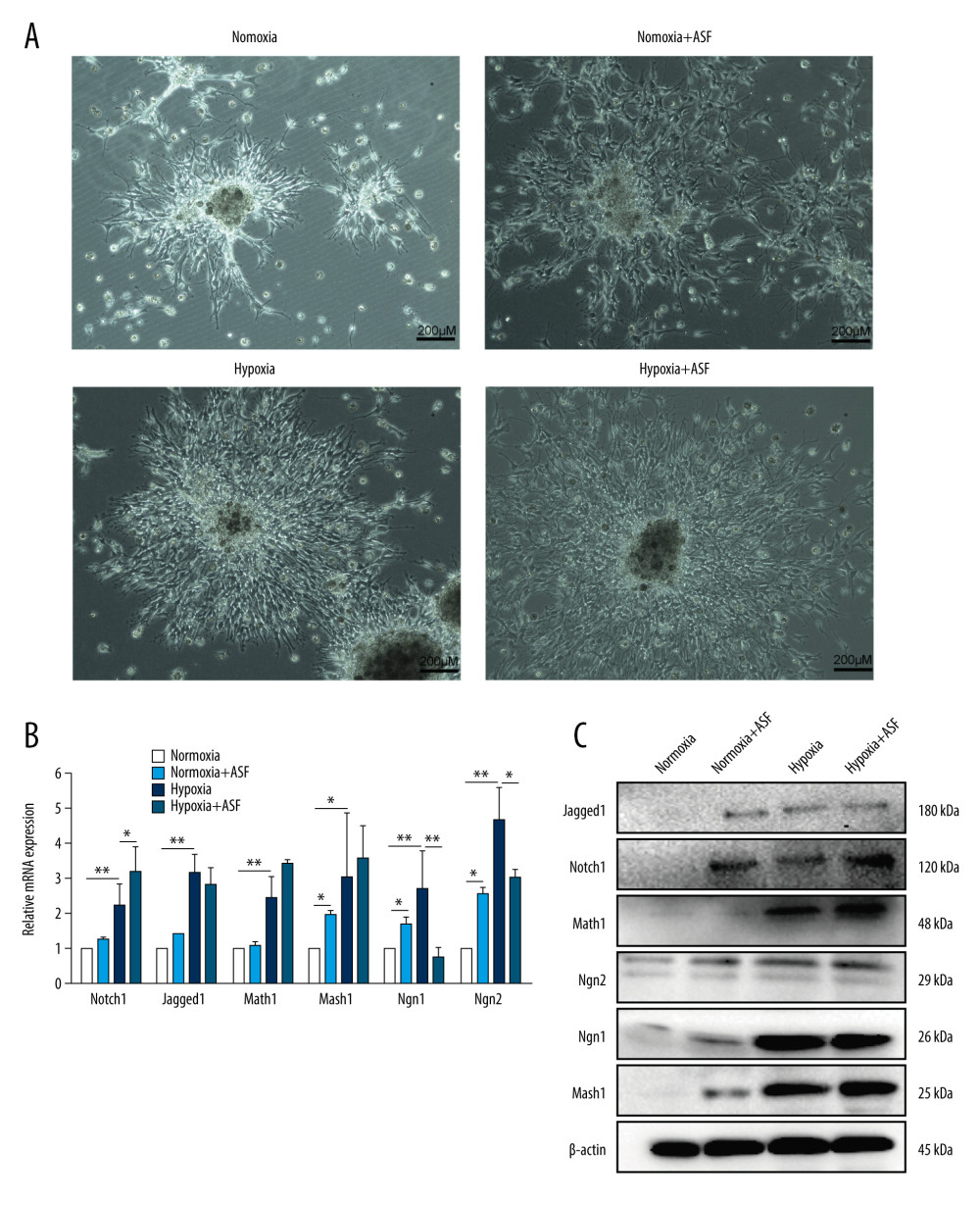 Figure 7. The expression of relevant mRNA and proteins in vitro (A) NSCs under the microscope under conditions of normoxia, hypoxia, and treated with or without ASF. Scale bar: 200 μm. (B) The expression of Notch1, Jagged1, Math1, Mash1, Ngn1, and Ngn2 mRNA by qRT-PCR method under conditions of normoxia, hypoxia, and treated with or without ASF. Data are presented as mean±SEM. * P<0.05, ** P<0.01. (C) The expression of Notch1, Jagged1, Math1, Mash1, Ngn1, and Ngn2 proteins by western blotting under conditions of normoxia, hypoxia, and treated with or without ASF.
Figure 7. The expression of relevant mRNA and proteins in vitro (A) NSCs under the microscope under conditions of normoxia, hypoxia, and treated with or without ASF. Scale bar: 200 μm. (B) The expression of Notch1, Jagged1, Math1, Mash1, Ngn1, and Ngn2 mRNA by qRT-PCR method under conditions of normoxia, hypoxia, and treated with or without ASF. Data are presented as mean±SEM. * P<0.05, ** P<0.01. (C) The expression of Notch1, Jagged1, Math1, Mash1, Ngn1, and Ngn2 proteins by western blotting under conditions of normoxia, hypoxia, and treated with or without ASF. References
1. Li J, Yan Q, Ma Y, Directional induction of dopaminergic neurons from neural stem cells using substantia nigra homogenates and basic fibroblast growth factor: Neural Regen Res, 2012; 7(7); 511-16
2. Park KI, Hack MA, Ourednik J, Acute injury directs the migration, proliferation, and differentiation of solid organ stem cells: Evidence from the effect of hypoxia-ischemia in the CNS on clonal “reporter” neural stem cells: Exp Neurol, 2006; 199(1); 156-78
3. Bürgers H, Schelshorn DW, Wagner W, Acute anoxia stimulates proliferation in adult neural stem cells from the rat brain: Exp Brain Res, 2008; 188(1); 33-43
4. Zheng P-D, Rajneesh M, Zhou H-J, Ginkgolide B promotes the proliferation and differentiation of neural stem cells following cerebral ischemia/reperfusion injury, both in vivo and in vitro: Neural Regen Res, 2018; 13(07); 86-93
5. Kageyama R, Ohtsuka T, Shimojo H, Imayoshi I, Dynamic Notch signaling in neural progenitor cells and a revised view of lateral inhibition: Nat Neurosci, 2008; 11(11); 1247-51
6. Sun F, Mao XO, Xie L, Notch1 signaling modulates neuronal progenitor activity in the subventricular zone in response to aging and focal ischemia: Aging Cell, 2013; 12(6); 978-87
7. Imayoshi I, Shimojo H, Sakamoto M, Genetic visualization of notch signaling in mammalian neurogenesis: Cell Mol Life Sci, 2013; 70(12); 2045-57
8. Huang XP, Tan H, Chen BY, Deng CQ, Astragalus extract alleviates nerve injury after cerebral ischemia by improving energy metabolism and inhibiting apoptosis: Biol Pharm Bull, 2012; 35(4); 449-54
9. Gao H, Dou L, Shan L, Proliferation and committed differentiation into dopamine neurons of neural stem cells induced by the active ingredients of radix astragali: Neuroreport, 2018; 29(7); 577-82
10. Gao H, Wentao LI, Encephalopathy DO, Effects of astragalus flavone on proliferation of neural stem cells in cerebral infarction rats: Academic Journal of Shanghai University of Traditional Chinese Medicine, 2018
11. Longa E, Reversible middle cerebral artery occlusion without craniotomy in rats: Stroke, 1989; 20(1); 84-91
12. Ansari S, Azari H, Mcconnell DJ, Intraluminal middle cerebral artery occlusion (MCAO) model for ischemic stroke with laser Doppler flowmetry guidance in mice: J Vis Exp, 2011(51); 2879
13. Bederson JB, Pitts LH, Tsuji M, Rat middle cerebral artery occlusion: Evaluation of the model and development of a neurologic examination: Stroke, 1986; 17(3); 472-76
14. Altumbabic M, Peeling J, Bigio M, Intracerebral hemorrhage in the rat: Effects of hematoma aspiration: Stroke, 1998; 29(9); 1917-22 discussion 1922–23
15. Ghazale H, Ramadan N, Mantash S, Docosahexaenoic acid (DHA) enhances the therapeutic potential of neonatal neural stem cell transplantation post traumatic brain injury: Behav Brain Res, 2017; 340; 1-13
16. Wang XM, Mao XO, Xie L, Involvement of Notch1 signaling in neurogenesis in the subventricular zone of normal and ischemic rat brain in vivo: J Cereb Blood Flow Metab, 2009; 29(10); 1644-54
17. Li W, Gao H, Li W, Effects of combined Bushen Zhichan recipe and levodopa in a rodent model of Parkinson disease: potential mechanisms: Med Sci Monit, 2020; 26; e922345
18. Cheng X, Yang YL, Li WH, Cerebral ischemia-reperfusion aggravated cerebral infarction injury and possible differential genes identified by RNA-Seq in rats: Brain Res Bull, 2019; 156; 33-42
19. Qi L, Tang Y, He W, Lithium chloride promotes neuronal differentiation of rat neural stem cells and enhances neural regeneration in Parkinson’s disease model: Cytotechnology, 2017; 69(2); 1-11
20. Sinha P, Chakrabarti N, Ghosh N, Alterations of thyroidal status in brain regions and hypothalamo-pituitary-blood-thyroid-axis associated with dopaminergic depletion in substantia nigra and ROS formation in different brain regions after MPTP treatment in adult male mice: Brain Res Bull, 2020; 156; 131-40
21. Li HL, Zhang H, Hua H, The effect of amino density on the attachment, migration, and differentiation of rat neural stem cells in vitro: Mol Cells, 2013; 35(5); 436-43
22. Ross SE, Greenberg ME, Stiles CD, Basic helix-loop-helix factors in cortical development: Neuron, 2003; 39(1); 13-25
23. Auyeung KK, Han QB, Ko JK, Astragalus membranaceus: A review of its protection against inflammation and gastrointestinal cancers: Am J Chin Med, 2016; 44(1); 1-22
24. Balakrishnan B, Liang Q, Fenix K, Combining the anticancer and immunomodulatory effects of Astragalus and Shiitake as an integrated therapeutic approach: Nutrients, 2021; 13(8); 2564
25. Mattar P, Britz O, Johannes C, A screen for downstream effectors of Neurogenin2 in the embryonic neocortex: Dev Biol, 2004; 273(2); 373-89
26. Gowan K, Helms AW, Hunsaker TL, Crossinhibitory activities of Ngn1 and Math1 allow specification of distinct dorsal interneurons: Neuron, 2001; 31(2); 219-32
27. Casarosa S, Fode C, Guillemot F, Mash1 regulates neurogenesis in the ventral telencephalon: Development, 1999; 126(3); 525-34
28. Vierbuchen T, Ostermeier A, Pang ZP, Direct conversion of fibroblasts to functional neurons by defined factors: Nature, 2010; 463; 1035-41
29. Parras CM, Hunt C, Sugimori M, The proneural gene Mash1 specifies an early population of telencephalic oligodendrocytes: J Neurosci, 2007; 27(16); 433-42
30. Rui LZ, Chopp M, Roberts C, Ascl1 lineage cells contribute to ischemia-induced neurogenesis and oligodendrogenesis: J Cereb Blood Flow Metab, 2011; 31(2); 614-25
31. Battiste J, Helms AW, Kim EJ, Ascl1 defines sequentially generated lineage-restricted neuronal and oligodendrocyte precursor cells in the spinal cord: Development, 2007; 134(2); 285-93
32. Kim EJ, Leung CT, Reed RR, Johnson JE, In vivo analysis of Ascl1 defined progenitors reveals distinct developmental dynamics during adult neurogenesis and gliogenesis: J Neurosci, 2007; 27(47); 12764-74
33. He F, Ge W, Martinowich K, A positive autoregulatory loop of Jak-STAT signaling controls the onset of astrogliogenesis: Nat Neurosci, 2005; 8(5); 616-25
34. Zhao J, Lin Q, Kim K, Ngn1 inhibits astrogliogenesis through induction of miR-9 during neuronal fate specification: eLife, 2015; 4; e06885
35. Yi S, Nadal-Vicens M, Misono S, Neurogenin promotes neurogenesis and inhibits glial differentiation by independent mechanisms: Cell, 2001; 104(3); 365-76
36. Kim SS, Yoo SW, Park TS, Neural induction with neurogenin1 increases the therapeutic effects of mesenchymal stem cells in the ischemic brain: Stem Cells, 2008; 26(9); 2217-28
37. Helms AW, Sequential roles for Mash1 and Ngn2 in the generation of dorsal spinal cord interneurons: Development, 2005; 132(12); 2709-19
Figures
 Figure 1. The average weight, neurobehavioral score, and cerebral infarction volume in each group (A) The average rat weight in normal, model, and ASF groups at days 0, 1, 3, 7, 10, and 14 (n=8 per group), * P<0.05, ** P<0.01. (B) The score of neurobehavioral tests in normal, model, and ASF groups at days 0, 3, 7, 10, and 14 (n=8 per group), * P<0.05, ** P<0.01. (C) Brains stained by TTC in normal, model (days 7 and 14), and ASF (days 7 and 14) groups. (D) Statistic analysis of cerebral infarction volume. Data are presented as mean±SEM, * P<0.05, ** P<0.01.
Figure 1. The average weight, neurobehavioral score, and cerebral infarction volume in each group (A) The average rat weight in normal, model, and ASF groups at days 0, 1, 3, 7, 10, and 14 (n=8 per group), * P<0.05, ** P<0.01. (B) The score of neurobehavioral tests in normal, model, and ASF groups at days 0, 3, 7, 10, and 14 (n=8 per group), * P<0.05, ** P<0.01. (C) Brains stained by TTC in normal, model (days 7 and 14), and ASF (days 7 and 14) groups. (D) Statistic analysis of cerebral infarction volume. Data are presented as mean±SEM, * P<0.05, ** P<0.01. Figure 2. ASF induced NSC proliferation and differentiation into neurons in SVZ (A) The SVZ is shown in the coronal section of rat brain after heart perfusion. (B) Representative IF images of BrdU (green), Nestin (pink), β-Tubulin III (red), and DAPI (blue) in normal, model and ASF groups at days 7 and 14. Scale bar: 50 μm. The results are shown as bar graphs. Data are presented as mean±SEM, * P<0.05, ** P<0.01.
Figure 2. ASF induced NSC proliferation and differentiation into neurons in SVZ (A) The SVZ is shown in the coronal section of rat brain after heart perfusion. (B) Representative IF images of BrdU (green), Nestin (pink), β-Tubulin III (red), and DAPI (blue) in normal, model and ASF groups at days 7 and 14. Scale bar: 50 μm. The results are shown as bar graphs. Data are presented as mean±SEM, * P<0.05, ** P<0.01. Figure 3. ASF induced NSC differentiation into oligodendrocytes and inhibited NSC differentiation into astrocytes in SVZ (A) Representative IF images of O4 (green), GFAP (green), and DAPI (blue) in normal, model and ASF groups at days 7 and 14. Scale bar: 50 μm. (B) The results were showed as the bar graphs. Data are presented as mean±SEM, * P<0.05, ** P<0.01.
Figure 3. ASF induced NSC differentiation into oligodendrocytes and inhibited NSC differentiation into astrocytes in SVZ (A) Representative IF images of O4 (green), GFAP (green), and DAPI (blue) in normal, model and ASF groups at days 7 and 14. Scale bar: 50 μm. (B) The results were showed as the bar graphs. Data are presented as mean±SEM, * P<0.05, ** P<0.01. Figure 4. The expression of relevant mRNA and proteins in SVZ of brain (A) Tissues of rat brains in SVZ were taken and were used for western blotting and qRT-PCR. (B) The expression of Notch1, Jagged1, Math1, Mash1, Ngn1, and Ngn2 protein by western blotting. (C) The expression of Notch1, Jagged1, Math1, Mash1, Ngn1, and Ngn2 mRNA by qRT-PCR method. Data are presented as mean±SEM. * P<0.05, ** P<0.01.
Figure 4. The expression of relevant mRNA and proteins in SVZ of brain (A) Tissues of rat brains in SVZ were taken and were used for western blotting and qRT-PCR. (B) The expression of Notch1, Jagged1, Math1, Mash1, Ngn1, and Ngn2 protein by western blotting. (C) The expression of Notch1, Jagged1, Math1, Mash1, Ngn1, and Ngn2 mRNA by qRT-PCR method. Data are presented as mean±SEM. * P<0.05, ** P<0.01. Figure 5. ASF induced NSCs proliferation in vitro (A) The bright field image of neurospheres after culturing for 3 days (A1, Scale bar: 200 μm), and the representative IF image of Nestin confirmed that these derived cells were NSCs (A2, Scale bar: 100 μm). (B) Representative IF images of EdU+ cells treated with ASF at concentrations of 0, 10, 20 and 40 μg/mL. DAPI was used to counterstain nuclei. Scale bar: 100μm. (C) The number of EdU+ cells. Data are presented as mean±SEM, * P<0.05, ** P<0.01.
Figure 5. ASF induced NSCs proliferation in vitro (A) The bright field image of neurospheres after culturing for 3 days (A1, Scale bar: 200 μm), and the representative IF image of Nestin confirmed that these derived cells were NSCs (A2, Scale bar: 100 μm). (B) Representative IF images of EdU+ cells treated with ASF at concentrations of 0, 10, 20 and 40 μg/mL. DAPI was used to counterstain nuclei. Scale bar: 100μm. (C) The number of EdU+ cells. Data are presented as mean±SEM, * P<0.05, ** P<0.01. Figure 6. ASF induced differentiation of NSCs in vitro (A1, A2) Representative ICC images of β-Tubulin III (red), O4 (green), Nestin (red), GFAP (green), and DAPI (blue) at days 7 and 14. Scale bar: 200 μm. (B) The result of relativeβ-Tubulin III, O4, Nestin and GFAP positive cells. Data are represented as mean±SEM, * P<0.05, ** P<0.01.
Figure 6. ASF induced differentiation of NSCs in vitro (A1, A2) Representative ICC images of β-Tubulin III (red), O4 (green), Nestin (red), GFAP (green), and DAPI (blue) at days 7 and 14. Scale bar: 200 μm. (B) The result of relativeβ-Tubulin III, O4, Nestin and GFAP positive cells. Data are represented as mean±SEM, * P<0.05, ** P<0.01. Figure 7. The expression of relevant mRNA and proteins in vitro (A) NSCs under the microscope under conditions of normoxia, hypoxia, and treated with or without ASF. Scale bar: 200 μm. (B) The expression of Notch1, Jagged1, Math1, Mash1, Ngn1, and Ngn2 mRNA by qRT-PCR method under conditions of normoxia, hypoxia, and treated with or without ASF. Data are presented as mean±SEM. * P<0.05, ** P<0.01. (C) The expression of Notch1, Jagged1, Math1, Mash1, Ngn1, and Ngn2 proteins by western blotting under conditions of normoxia, hypoxia, and treated with or without ASF.
Figure 7. The expression of relevant mRNA and proteins in vitro (A) NSCs under the microscope under conditions of normoxia, hypoxia, and treated with or without ASF. Scale bar: 200 μm. (B) The expression of Notch1, Jagged1, Math1, Mash1, Ngn1, and Ngn2 mRNA by qRT-PCR method under conditions of normoxia, hypoxia, and treated with or without ASF. Data are presented as mean±SEM. * P<0.05, ** P<0.01. (C) The expression of Notch1, Jagged1, Math1, Mash1, Ngn1, and Ngn2 proteins by western blotting under conditions of normoxia, hypoxia, and treated with or without ASF. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387