30 March 2022: Clinical Research
Follow-Up of 108 Patients with Chronic Hepatitis B Virus Infection Treated with Polyethylene Glycol-Conjugated Derivatives of Interferon-Alpha and Monitoring of Off-Treatment Virological Relapse
Liyang Zhou1ABCDEF, Qin He1CEF, Xitao Liu2DEF, Xiaoan Yang3BC, Xueting Ou3EF, Bing Situ2DF, Yueping Li4DE, Xingfei Pan1DEFG*, Qihuan Xu3AEDOI: 10.12659/MSM.934785
Med Sci Monit 2022; 28:e934785
Abstract
BACKGROUND: This single center study, which enrolled 108 patients with chronic hepatitis B virus infection treated with pegylated interferon-alpha (PEG-IFN-α), aimed to follow up and monitor off-treatment responses, including virological relapse, and analyze predictors of long-term efficacy of the PEG-IFN-α regimen.
MATERIAL AND METHODS: In total, 108 hepatitis B e antigen (HBeAg)-positive patients with chronic hepatitis B who had completed the PEG-IFN-α regimen and achieved virological suppression were enrolled. The patients were followed up for 5 years to monitor off-treatment responses. Twenty-eight relevant factors, including the history of antiviral therapy and HBeAg seroconversion, were analyzed using the Cox proportional hazards regression model.
RESULTS: The cumulative rates of virological suppression were 75.70%, 68.68%, 65.25%, 63.91%, and 63.91% at 1, 2, 3, 4, and 5 years of the follow-up period, respectively. Compared with the rates of virological suppression, the cumulative rates of clinical suppression were 88.41%, 79.83%, 78.59%, 75.65%, and 75.65%, respectively, for the 5 years. Alanine aminotransferase (ALT) normalization at 24 weeks after off-therapy (relative risk [RR]=3.430, P=0.013) was a potential predictor for sustained virological suppression, and the history of anti-viral therapy (RR=0.164, P=0.004), quantitative value of hepatitis B virus surface antigen (HBsAg) at 48 weeks of anti-viral therapy (RR=2.697, P=0.039), and ALT normalization at 24 weeks after off-therapy (RR=5.467, P=0.004) were potential predictors for sustained clinical suppression.
CONCLUSIONS: Our results suggested that increased HBsAg levels at 48 weeks and normalization of ALT at 24 weeks after off-therapy might be predictive factors for long-term treatment efficacy.[color=red] [/color]
Keywords: Hepatitis B e Antigens, Hepatitis B, Chronic, peginterferon alfa-2a, Sustained Virologic Response, Antiviral Agents, Follow-Up Studies, Hepatitis B Surface Antigens, Hepatitis B virus, Humans, Interferon-alpha, Polyethylene Glycols, Recombinant Proteins, Recurrence
Background
Approximately 240 million people have chronic hepatitis B virus (HBV) infection, and 1 million deaths related to chronic HBV infection (CHB) occur every year worldwide [1,2]. CHB is still an important global public health problem because it can cause progression to cirrhosis, hepatocellular carcinoma (HCC), or other end-staged liver diseases with significant morbidity and mortality [3,4]. In China, 60% of patients with cirrhosis and 80% of patients with HCC have CHB [5]. Antiviral therapy can prevent patients with CHB from progressing to liver fibrosis, cirrhosis, and HCC [6]. Unfortunately, none of the available antiviral agents have cured CHB to date [7,8].
Currently, there are 2 main antiviral treatment options for active patients with CHB: treatment with a nucleos(t)ide analogue (NA) and treatment with pegylated interferon alpha (PEG-IFN-α), which are recommended by the European Association for the Study of Liver Disease (EASL) guidelines [1] and American Association for the Study of Liver Diseases (AASLD) guidelines [9]. PEG-IFN-α could have a weak direct antiviral effect but could also strongly regulate the immune response to a virus and alleviate the pathogenesis of liver fibrosis [10]. Furthermore, the most important advantages of PEG-IFN-α compared with NA are no risk of viral resistance development and the finite course of treatment (usually 48-week course of treatment) [1,9]. Recently, it was also reported that PEG-IFN-α therapy has considerable off-treatment sustainability in Chinese hepatitis B early antigen (HBeAg)-positive patients with CHB, with serological and complete responses [11]. In addition, the effect of PEG-IFN-α on HBsAg reduction or the seroconversion rate of HBsAg is relatively higher than that of NA [1,9]. Also, compared with the morbidity of patients with CHB treated with NA, the morbidity of cirrhosis or HCC is significantly lower in patients with CHB treated with PEG-IFN-α [12]. This evidence shows that PEG-IFN-α induces long-term immune control with a finite treatment. Meanwhile, PEG-IFN-α has some disadvantages, such as the relatively unfavorable safety profile, many contraindications, relatively high price, mode of subcutaneous injection administration, and, especially, the high variability of response [1,9,13]. In clinical practice, some patients with CHB need to receive the NA regimen again because hepatic flares could recur after the discontinuation of the PEG-IFN-α regimen [14].
According to the guidelines, patient selection before the PEG-IFN-α regimen and early treatment predictors could give an individual treatment strategy to patients with CHB [1,9,13]. However, the predictors to screen optimal patients with CHB with a potentially high chance of achieving good long-term efficacy after they complete the course of the PEG-IFN-α regimen are still uncertain.
Therefore, this single center study included 108 patients with HBeAg-positive CHB treated with PEG-IFN-α and aimed to follow up and monitor off-treatment responses, including virological relapse, and analyze predictors for the long-term efficacy of the PEG-IFN-α regimen.
Material and Methods
PATIENTS:
This study was approved by the Human Ethics Committee of the Third Affiliated Hospital of Sun Yat-sen University. Written informed consent was obtained from each participant. The diagnostic criteria of CHB followed the EASL guidelines, and all patients met the criteria of antiviral therapy recommended by the EASL guidelines [1].
We screened 320 patients with CHB who received the PEG-IFN-α-2a/α-2b regimen in the Department of Infectious Diseases of the Third Affiliated Hospital of Sun Yat-sen University from January 1, 2008, to December 31, 2013. A total of 268 patients were positive for HBeAg. However, only 108 who had completed the PEG-IFN-α regimen and achieved virological suppression were enrolled. All patients were treated with PEG-IFN-α-2a/α-2b and were regularly followed up for 5 years after completing the PEG-IFN-α regimen. The demographic information and clinical characteristics of the enrolled patients are summarized in Table 1.
The inclusion criteria were as follows: (a) history of HBV infection ≥6 months; (b) positive HBsAg and HBeAg; (c) completed course of PEG-IFN-α therapy; (d) achieved virological suppression during the regimen; (e) PEG-IFN-α was the only antiviral agent used during the course of treatment; and (f) good compliance.
The exclusion criteria were as follows: (a) HIV infection or tuberculosis; (b) other forms of chronic liver diseases; (c) decompensated cirrhosis, hypertension, diabetes, autoimmune diseases, or other underlying medical diseases; (d) psychiatric disorders; and (e) pregnant or lactating.
STUDY DESIGN:
Baseline data, including age, sex, and history of antiviral therapy, were collected. Routine blood tests, liver function, HBV serological markers (HBsAg, antibody to hepatitis B surface antigen (HBsAb), HBeAg, antibody to hepatitis B e antigen (HBeAb), and HBV deoxyribonucleic acid (DNA) levels were assayed every 4 weeks during the course of the PEG-IFN-α regimen. In the off-treatment state, patients were followed up every 12 weeks. Routine blood tests, liver function, HBV serological markers and HBV DNA load were assayed every 12 weeks. Alpha-fetoprotein and liver ultrasound were assayed every 24 weeks. All monitoring methods of patients treated with PEG-IFN-α were based on the guidelines’ suggestions [1,9].
DEFINITION OF VIROLOGICAL AND CLINICAL SUPPRESSION OR RELAPSE:
Virological suppression was defined as serum HBV DNA levels <2000 IU/mL during the course of the PEG-IFN-α regimen [1,9.] According to the guidelines [1,9], sustained off-therapy virological suppression was defined as serum HBV DNA levels <2000 IU/mL for at least 12 months after the end of therapy. Clinical suppression was defined as alanine aminotransferase (ALT) <2×upper limit of normal (ULN) during the course of the PEG-IFN-α regimen. Sustained clinical suppression was defined as ALT <2×ULN after the PEG-IFN-α regimen to the end of the follow-up period.
Virological relapse was defined as more than 2 times an HBV DNA level of greater than 2000 IU/mL (interval at least 1 month) after off-therapy. Clinical relapse was defined as ALT > 2×ULN on the basis of virological relapse.
DEFINITION OF THE OBSERVATION ENDPOINT:
For patients with virological relapse, the observation endpoint was the date of an HBV DNA level >2000 IU/mL. For patients with clinical relapse, the observation endpoint was the date of ALT >2×ULN on the basis of virological relapse. For patients who had been followed up for at least 6 months and showed no virological or clinical relapse, the observation endpoint was the date of loss to follow-up or December 31, 2015.
LABORATORY TESTS:
A routine automated analysis system (Beckman Coulter, Fullerton, CA, USA) was used to assay liver biochemistry. HBV serological markers were tested using a chemiluminescent microparticle enzyme immunoassay (Abbott, Chicago, IL, USA). HBV DNA levels were tested using the COBAS AmpliPrep/COBAS TaqMan HBV Test (v2.0). The detection limit was HBV DNA <20 IU/mL.
STATISTICAL ANALYSIS:
Data are expressed as the mean±standard deviation or median. The Cox proportional hazards regression model was used to analyze predictors for sustained virological suppression or clinical suppression. The Kaplan-Meier method was used to calculate the cumulative rate of virological suppression and the cumulative rate of clinical suppression. All analyses were performed using SPSS v22.0 statistical analysis software (IBM Corp, Armonk, NY, USA). Differences were considered statistically significant at a value of
Results
PATIENT CHARACTERISTICS:
A total of 320 patients were enrolled. Based on the inclusion and exclusion criteria, 108 patients (17 women, 91 men) were finally included in the present study (Figure 1). The characteristics of patients are shown in Table 1. The average age of patients was 28±8.16 years. The median course of the PEG-IFN-α regimen was 48 weeks (range 48–69 weeks). At the end of the course, the rate of ALT normalization was 60.19% (65/108), the rate of HBeAg seroconversion was 75.00% (81/108), and the rate of HBsAg clearance was 17.59% (19/108).
The median follow-up period was 245 weeks (range 16–360 weeks). At the end of the follow-up period, the rate of HBeAg seroconversion was 79.63% (86/108), and the rate of HBsAg clearance was 15.74% (17/108). Before the PEG-IFN-α regimen was initiated, 10 of the 108 patients had liver cirrhosis (compensation stage) as assessed by liver ultrasound. After the end of the follow-up period, 9 of the 10 patients with cirrhosis were still in the compensated stage of cirrhosis. Fortunately, 1 patient showed no cirrhosis as assessed by liver ultrasound. Furthermore, none of the enrolled patients had HCC at the end of the 5-year follow-up period.
RATES OF VIROLOGICAL RELAPSE AND CLINICAL RELAPSE AFTER OFF-THERAPY:
According to the definition of virological relapse, 38 of 108 patients (35.19%) had virological relapse during the follow-up period of 5 years. Furthermore, most of them (26/38, 68.42%) had virological relapse during the first 48 weeks of the follow-up period. The rates of virological relapse were 24.30%, 9.27%, 5.00%, 2.06%, and 0% at 1, 2, 3, 4, and 5 years of the follow-up period, respectively. The cumulative rates of virological suppression were 75.70%, 68.68%, 65.25%, 63.91%, and 63.91% at 1, 2, 3, 4, and 5 years of the follow-up period, respectively (Table 2).
According to the definition of clinical relapse, 23 of 108 patients (21.30%) experienced clinical relapse during the 5-year follow-up period. Twelve of the 23 patients (52.17%) showed clinical relapse during the first 48 weeks of the follow-up period. The rates of clinical relapse were 11.59%, 9.70%, 1.55%, 3.74%, and 0% at 1, 2, 3, 4, and 5 years of the follow-up period, respectively. The cumulative rates of clinical suppression were 88.41%, 79.83%, 78.59%, 75.65%, and 75.65% at 1, 2, 3, 4, and 5 years, respectively (Table 2).
PREDICTORS OF SUSTAINED VIROLOGICAL SUPPRESSION:
The Cox proportional hazards regression model was used to screen predictors of sustained virological suppression. Twenty-eight relevant factors, including age, sex, history of antiviral therapy, course of therapy, achievement of HBeAg seroconversion, ALT level, quantitative value of HBsAg, and HBV DNA level, were included and evaluated.
Univariate Cox regression analysis revealed that sustained virological suppression after off-therapy was associated with the quantitative value of HBsAg at 24 weeks (RR=1.000; 95% CI, 1.000-1.000; P=0.022), 36 weeks (RR=1.000; 95% CI, 1.000-1.000; P<0.001), and 48 weeks (RR=1.000; 95% CI, 1.000-1.000; P<0.001) of the PEG-IFN-α regimen; HBV DNA level at 36 weeks (RR=1.000; 95% CI, 1.000-1.000; P=0.028) and 48 weeks (RR=1.000; 95% CI, 1.000-1.000; P=0.025) of the PEG-IFN-α regimen; course of therapy (RR=0.961; 95% CI, 0.932–0.992; P=0.013); aspartate aminotransferase (AST) value at 24 weeks of the PEG-IFN-α regimen (RR=0.988; 95% CI, 0.978–0.999; P=0.029); and ALT normalization at 24 weeks after off-therapy (RR=5.027; 95% CI, 2.610–9.682; P<0.001) (Table 3).
Multivariate Cox regression analysis revealed that the ALT value at 24 weeks after off-therapy (RR=3.430, 95% CI 1.302–9.034, P=0.013) could be an independent predictor of sustained virological suppression after the PEG-IFN-α regimen was completed (Table 3).
Subsequently, the difference between the ALT normalization group and the ALT abnormality group at 24 weeks after off-therapy was compared by using the log-rank test. The cumulative rate of virological suppression in the ALT normalization group (Group A1) was significantly greater than that in the ALT abnormality group (Group A2) (χ2=30.645, P<0.001) (Figure 2).
PREDICTIVE FACTORS OF SUSTAINED CLINICAL SUPPRESSION:
Univariate Cox regression analysis revealed that sustained clinical suppression after off-therapy was associated with a history of antiviral therapy (RR=0.279; 95% CI, 0.098–0.794; P=0.017); ALT value at the initiation of the PEG-IFN-α regimen (RR=1.002; 95% CI, 1.000–1.004; P=0.041); quantitative value of HBsAg at 36 weeks (RR=1.000; 95% CI, 1.000–1.001; P<0.001) and 48 weeks (RR=1.000; 95% CI, 1.000-1.000; P<0.001) during the course of the PEG-IFN-α regimen; AST value at 24 weeks during the course of the PEG-IFN-α regimen (RR=0.982; 95% CI, 0.966–0.999; P=0.036), and ALT normalization at 24 weeks after off-therapy (RR=9.322; 95% CI, 4.042–21.499; P<0.001) (Table 4). Subsequently, multivariate Cox regression analysis revealed that a history of antiviral therapy (RR=0.147; 95% CI, 0.026–0.824; P=0.029), ALT normalization at 24 weeks after off-therapy (RR=4.131; 95% CI, 1.079–15.810; P=0.038), and the quantitative value of HBsAg at 48 weeks during the course of the PEG-IFN-α regimen (RR=1.000; 95% CI, 1.000–1.001; P=0.032) could be independent predictors of sustained clinical suppression after the PEG-IFN-α regimen was completed (Table 4). When the predictor “quantitative value of HBsAg at 48 weeks” was defined as measurement data in Cox regression analysis, the relative risk value was 1. It was necessary to convert measurement data into counting data. The quantitative value of HBsAg at 48 weeks was changed into a low quantitative value group of HBsAg (HBsAg ≤500 COI), middle quantitative value group of HBsAg (500–6000 COI HBsAg), and high quantitative value group of HBsAg (HBsAg >6000 COI). After the adjustment, the result of multivariate Cox regression analysis revealed that a history of antiviral therapy (RR=0.164; 95% CI, 0.048–0.558; P=0.004), ALT normalization at 24 weeks after off-therapy (RR=5.467; 95% CI, 1.705–17.524; P=0.004), and the quantitative value of HBsAg at 48 weeks during the PEG-IFN-α regimen (RR=2.697; 95% CI, 1.051–6.920; P=0.039) were still independent predictors of sustained clinical suppression after the PEG-IFN-α regimen was completed (Table 4).
Subsequently, the difference between the ALT normalization group and the ALT abnormality group at 24 weeks after off-therapy was compared using the log-rank test. The cumulative rate of clinical suppression in the ALT normalization group (Group A1) was significantly greater than that in the ALT abnormality group (Group A2) (χ2=40.686, P<0.001) (Figure 3A).
The difference between the group without a history of antiviral therapy (Group B1) and the group with a history of antiviral therapy (Group B2) was also compared. The cumulative rate of clinical suppression in the group without a history of antiviral therapy was significantly greater than that of the group with a history of antiviral therapy (χ2=4.460, P=0.035) (Figure 3B).
Then, the differences among the low quantitative value group of HBsAg (HBsAg ≤500 COI, Group C1), middle quantitative value group of HBsAg (HBsAg 500–6000 COI, Group C2), and high quantitative value group of HBsAg (HBsAg >6000 COI, Group C3) were compared using the log-rank test. As shown in Figure 3C, the cumulative rate of clinical suppression in the low quantitative value group of HBsAg was significantly greater than that of the other 2 groups (χ2=15.016, P=0.001).
Discussion
In the present study, we found that 35.19% of patients (38/108) who had completed the PEG-IFN-α regimen and met the criteria of treatment withdrawal experienced virological relapse after the off-therapy period. Virological relapse (68.42%) often occurred during the first 48 weeks of the follow-up period. Furthermore, 21.30% of patients experienced clinical relapse (52.17%), which often occurred during the first 48 weeks of the follow-up period. The ALT value at 24 weeks of the follow-up period could represent a predictor for sustained virological suppression. The history of antiviral therapy, quantitative value of HBsAg at 48 weeks of the course of the PEG-IFN-α regimen, and ALT value at 24 weeks of the follow-up period could be predictors for sustained clinical suppression.
To date, none of the available antiviral agents can kill HBV, which resides in the liver. For patients with active CHB, PEG-IFN-α and NA are the first-line antiviral agents recommended by the EASL guidelines [1], AASLD guidelines [9], and Asian Pacific Association for the Study of the Liver guidelines [15]. The advantage of the NA regimen is that it has fewer adverse effects on patients and the level of viral suppression is unusually high, but the disadvantage is an indefinite course of treatment, perhaps lifelong. Furthermore, virological relapse, clinical relapse, and even liver failure could occur after the withdrawal of the NA regimen [16]. We previously found that although patients who had been treated with NA met the criteria of withdrawal from NA treatment advised by AASLD guidelines, the cumulative rates of virological recurrence at 1, 2, 3, and 4 years of the follow-up period were 72.5%, 77.5%, 80.0%, and 82.5%, respectively [17]. The relapse rate was 85.2% during the first 48 weeks after withdrawal from the NA regimen [17]. However, compared with our previous report, the cumulative rate of virological relapse in the present study was relatively lower. This difference might be caused by different pharmacological strategies that PEG-IFN-α aimed on the induction of a long-term immune control by finite treatment as well as NA aimed on controlling hepatitis flare and disease progression by inhibiting viral replication [1].
Covalently closed circular DNA (cccDNA) of HBV that persistently resides in the nucleus of hepatocytes could be related to virological recurrence after off-therapy [18]. The cccDNA level of HBV mildly decreases after patients with CHB are treated with NA [19]. NA inhibits de novo virus production by inhibiting the viral reverse transcriptase and reduces virus titers [20]. However, they cannot cure CHB infection because they do not directly target cccDNA [20]. As a result, patients with CHB treated with NA have a high risk of recurrence after the discontinuation of the NA regimen [21–23]. Meanwhile, the cccDNA level of HBV is dramatically reduced after patients with CHB are treated with PEG-IFN-α [24]. Interferons and tumor necrosis factor α as the key factors produced by immune cells can further promote non-cytolytic inhibition of HBV replication and result in cccDNA destabilization [25]. Furthermore, compared with cccDNA levels of patients with CHB treated with IFN-α, cccDNA levels of HBV are significantly decreased in patients treated with PEG-IFN-α [26]. These findings imply that PEG-IFN-α could clear cccDNA by mediating the immune response.
PEG-IFN-α could promote the proliferation of cytotoxic T cells and activate natural killer cells and macrophages, which could kill cells infected with virus and promote the proliferation and maturation of dendritic cells [27–31]. Interferons can also induce apolipoprotein B mRNA-editing enzyme catalytic polypeptide-like 3G (Apobec3G) protein expression in association with STAT3 activation [32]. Apobec3G can induce G to A hyper mutation in HBV DNA, which strongly inhibits replication and the HBV lifecycle by interacting with HBV core protein [32]. These mechanisms could be the reason for sustained virological response and high rates of HBsAg and HBeAg seroconversion when PEG-IFN-α is used to treat patients with CHB. Compared with the cumulative rate of virological suppression after the completion of the NA regimen [17], the cumulative rate of virological suppression after the completion of the PEG-IFN-α regimen was relatively higher in the present study. This result might be a benefit of the long-term immunological control [1]. Moreover, the cumulative rate of clinical suppression in the present study was relatively higher than the cumulative rate of virological suppression after off-therapy. Taken together, the advantages of the PEG-IFN-α regimen might be as follows: reducing cccDNA levels [21], mediating the immune response to pathogens, and decreasing the risk of relapse after off-therapy. Therefore, we believe that some patients with CHB should be recommended to take a PEG-IFN-α regimen if they desire a finite course of therapy.
For child-bearing women with active CHB, it might be an optimal choice to complete a finite course of this regimen before conception. As a result, the PEG-IFN-α regimen might be chosen to treat child-bearing women with active CHB. However, patients with CHB receiving the PEG-IFN-α regimen are recommended to become pregnant at least after 6 months of withdrawal of PEG-IFN-α therapy because of the possible teratogenicity of the medicine [1,9,15]. Could we have nothing to worry about after meeting the criteria of off-therapy and just recommend that patients wait 6 months to get pregnant? The answer is no. In the present study, 35.19% of patients (38/108) who met the criteria of off-therapy after completion of the course of PEG-IFN-α treatment showed virological relapse during the 5-year follow-up period. Most of these patients with virological relapse (68.42%) showed the relapse during the first 48 weeks of the follow-up period. Furthermore, 21.30% of patients showed clinical relapse. Most of these patients with clinical relapse (52.17%) also showed clinical relapse during the first 48 weeks of the follow-up period. Altered immunity in the state of pregnancy can increase the risk of HBV flares, which is reported to be about 6% to 14% in women with CHB during pregnancy [33,34]. If women suffered from virological relapse during pregnancy, they would run a risk of having high HBV DNA levels and severe flares because the rate of virological relapse after PEG-IFN-α off-therapy was relatively high in the first 48 weeks. As a result, these patients should receive an NA regimen to control the disease and decrease HBV DNA levels to prevent mother-to-child transmission of HBV during late pregnancy. Taken together, child-bearing women who had already completed the PEG-IFN-α regimen should plan to become pregnant at least after 48 weeks of the discontinuation of PEG-IFN-α therapy. Fortunately, the rate of clinical relapse was relatively low after PEG-IFN-α therapy in the present study. Therefore, these patients might not need to receive the NA regimen (tenofovir [TDF] or telbivudine [LDT]) during early pregnancy, which could have a potential adverse effect on the embryo.
PEG-IFN-α has only a moderate level of viral suppression, while NA usually has a high level [1,35]. The viral suppression rate was just 7% to 14% after PEG-IFN-α off-treatment [10,36]. Thus, there are many studies available to find the pre-treatment predictive factors for choosing the suitable patients for a favorable virological response [37,38]. Low viral load, high serum ALT levels, HBV genotype, and high activity scores on liver biopsy are the acknowledged pretreatment predictors of favorable virological response [1,35]. Although patients already had the virological response and met the criteria of treatment, the rate of virological relapse after PEG-IFN-α off-therapy was relatively high in the present study. Therefore, it is urgent to find predictors for long-term efficacy of the PEG-IFN-α regimen.
Guidelines suggest that serum HBsAg levels are the most important on-treatment predictor of response to PEG-IFN-α [1]. Treatment failure can be predicted based on the quantitative level of HBsAg during therapy since no decline in HBsAg levels at week 12 is associated with a negative predictive value of 97% for a sustained viral response and 100% negative predictive value for HBsAg loss [39]. However, it might not be reliable in predicting treatment success because the best positive predictive values for sustained HBeAg seroconversion are 57% and 54%, which occur when HBsAg levels are under 1500 IU/mL at 12 and 24 weeks of therapy, respectively [40]. Furthermore, the corresponding negative predictive values are 72% and 76%, respectively [41]. These studies all focused on the response during treatment. Predictors for the long-term efficacy of the PEG-IFN-α regimen after meeting the criteria are still unknown.
In the present study, we also found that the quantitative values of HBsAg at 24, 36, and 48 weeks of the therapy could be predictors of sustained response, according to the univariate Cox regression analysis. The results were similar to studies mentioned previously [39–41]. The quantitative value of HBsAg at 48 weeks during the PEG-IFN-α regimen as a predictor was especially significant for sustained clinical suppression. This could be because the decrease in cccDNA level was accompanied by a decrease in the quantitative value of HBsAg [42–44].
According to the multivariate Cox regression analysis results, we found that ALT normalization at 24 weeks of the follow-up period might be a predictor for both sustained virological and clinical suppression. The guidelines advised that a minimum follow-up of at least 1 year after treatment with ALT determinations at least every 3 months be required to confirm sustained off-treatment biochemical response because ALT activity often fluctuates over time [1,9,15]. Elevation in serum ALT can be caused by a hepatitis flare or immune-mediated transitions to inactive disease, even clearance of infection [45]. Most ALT flares resolved within a period of 1 to 2 months, and on-treatment ALT flares have been observed most commonly after interferon treatment [45]. ALT flare during interferon therapy is a favorable indicator of immune reconstitution that can lead to virologic response through upregulation of NK cell and T-cell activity [46,47]. However, ALT normalization at 24 weeks after the PEG-IFN-α regimen could mean a biochemical response, which always depended on the virological response, as our study showed. In other research, there was a trend that an ALT flare to >10×ULN was associated with ALT normalization in HBeAg-negative patients at 6 months after PEG-IFN off-treatment [48], also showing ALT normalization at 24 weeks after PEG-IFN off-treatment could be a favorable predictor for the long-term efficacy of the PEG-IFN-α regimen.
This study had some limitations. First, this was a retrospective study and lacked an effective control group. Second, this was a single-center study. Finally, the sample size was relatively small. To clarify the predictors of long-term PEG-IFN-α response, more randomized and controlled prospective studies are required in the future.
Conclusions
We believe that for patients receiving the PEG-IFN-α regimen, the quantitative value of HBsAg at 48 weeks during the course of the PEG-IFN-α regimen and ALT normalization at 24 weeks after off-therapy represent potential predictors of the long-term efficacy of the PEG-IFN-α regimen. For patients with active CHB who desire a finite course of antiviral therapy, the PEG-IFN-α regimen might represent a good choice. Furthermore, it is necessary to regularly monitor ALT and HBsAg, especially in the first 48 weeks after treatment.
Figures
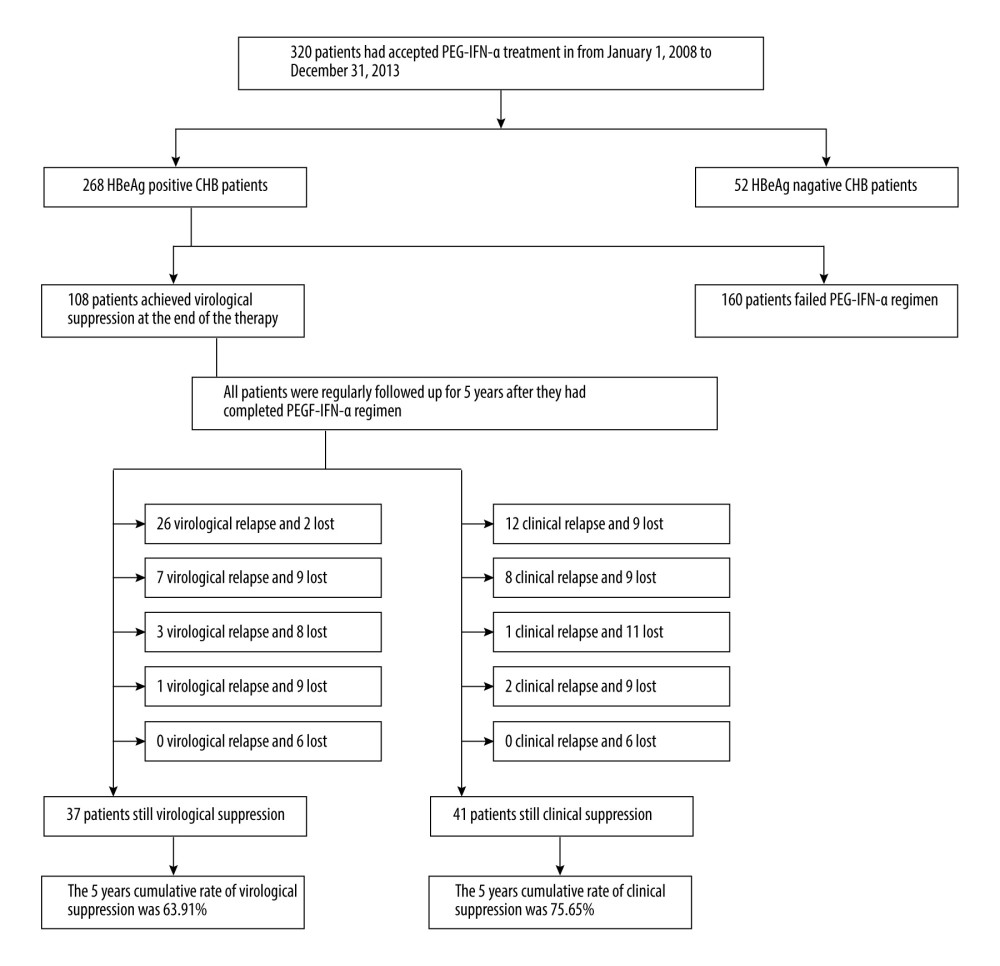 Figure 1. Enrollment and follow-up of the study cohorts. PEG-IFNα – pegylated interferon alfa; HBeAg – hepatitis B e antigen; CHB – chronic hepatitis B.
Figure 1. Enrollment and follow-up of the study cohorts. PEG-IFNα – pegylated interferon alfa; HBeAg – hepatitis B e antigen; CHB – chronic hepatitis B. 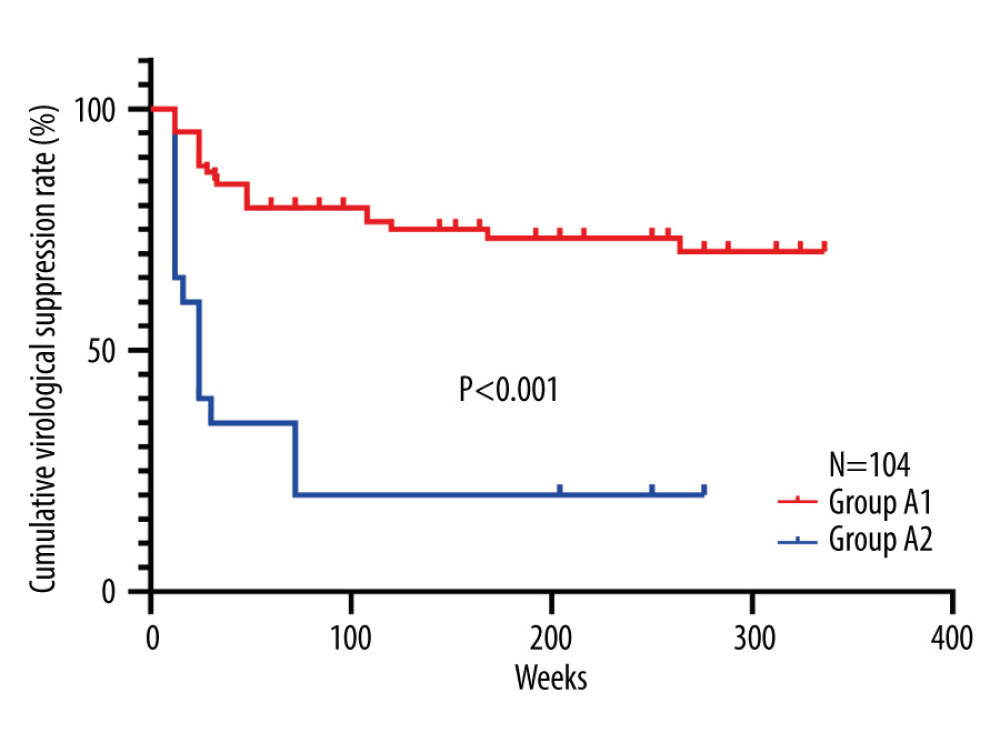 Figure 2. Comparison of the 5-year cumulative rate of virological suppression in different groups. Group A1, the normal alanine aminotransferase (ALT) value group at 24 weeks after off-therapy; Gropu A2, the abnormal ALT value group at 24 weeks after off-therapy.
Figure 2. Comparison of the 5-year cumulative rate of virological suppression in different groups. Group A1, the normal alanine aminotransferase (ALT) value group at 24 weeks after off-therapy; Gropu A2, the abnormal ALT value group at 24 weeks after off-therapy. 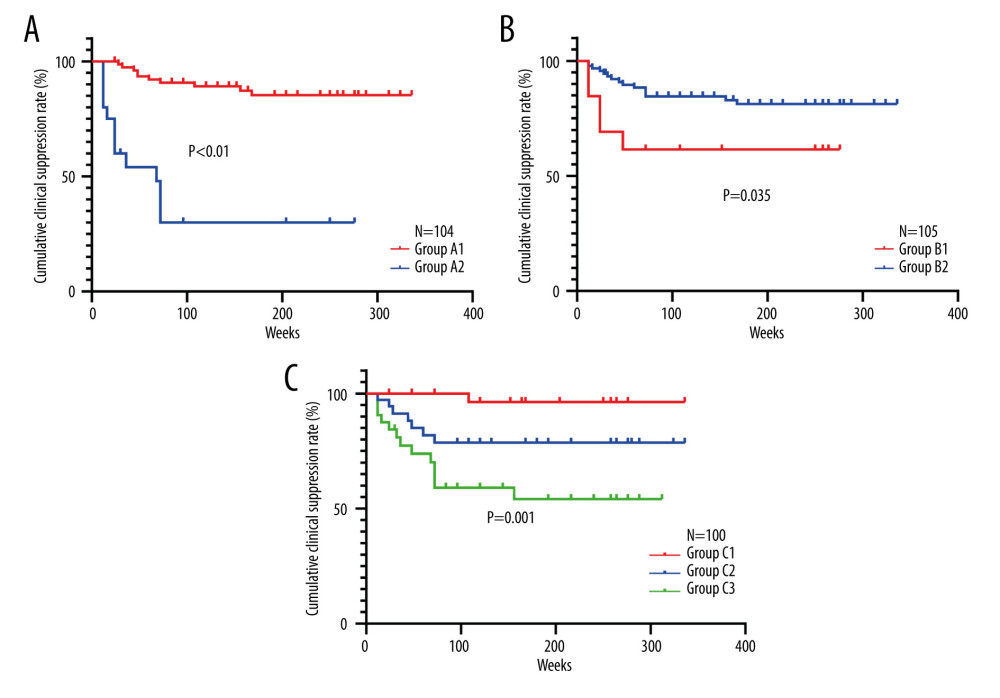 Figure 3. (A) Difference in the cumulative rate of clinical suppression between A1 and group A2 (P<0.01). Group A1, the normal alanine aminotransferase (ALT) value group at 24 weeks after off-therapy; Gropu A2, the abnormal ALT value group at 24 weeks after off-therapy. (B) Difference in the cumulative rate of clinical suppression between B1 and group B2 (P=0.035). Group B1, patients with a history of antirival treatment. Group B2, patients without a history of antirival treatment. (C) Difference in the cumulative rate of clinical suppression among group C1, C2 and C3 (P=0.001). Group C1, HBsAg ≤500 COI, the low HBsAg value group at 48 weeks of PEG-IFN-α treatment. Group C2, HBsAg 500–6000 COI, the middle HBsAg value group at 48 weeks of PEG-IFN-α treatment. Group C3, HBsAg >6000 COI, the high HBsAg value group at 48 weeks of PEG-IFN-α treatment. HBsAg – hepatitis B surface antigen; PEG-IFN-α – pegylated interferon-alpha.
Figure 3. (A) Difference in the cumulative rate of clinical suppression between A1 and group A2 (P<0.01). Group A1, the normal alanine aminotransferase (ALT) value group at 24 weeks after off-therapy; Gropu A2, the abnormal ALT value group at 24 weeks after off-therapy. (B) Difference in the cumulative rate of clinical suppression between B1 and group B2 (P=0.035). Group B1, patients with a history of antirival treatment. Group B2, patients without a history of antirival treatment. (C) Difference in the cumulative rate of clinical suppression among group C1, C2 and C3 (P=0.001). Group C1, HBsAg ≤500 COI, the low HBsAg value group at 48 weeks of PEG-IFN-α treatment. Group C2, HBsAg 500–6000 COI, the middle HBsAg value group at 48 weeks of PEG-IFN-α treatment. Group C3, HBsAg >6000 COI, the high HBsAg value group at 48 weeks of PEG-IFN-α treatment. HBsAg – hepatitis B surface antigen; PEG-IFN-α – pegylated interferon-alpha. Tables
Table 1. Demographic information and clinical characteristics of the enrolled patients.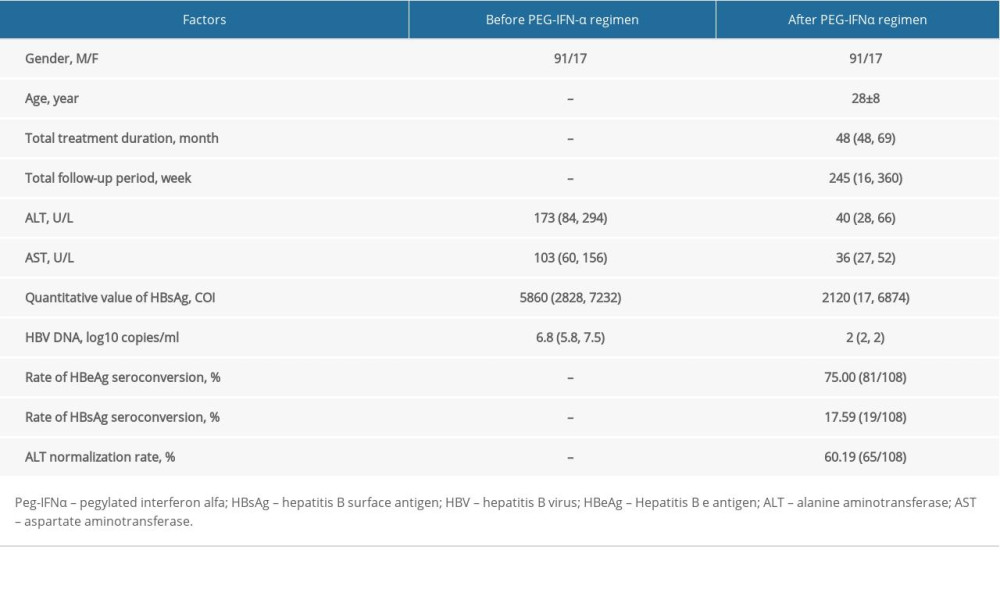 Table 2. Rate of virological/clinic relapse and cumulative rate of suppression after pegylated interferon-alpha regimen was completed.
Table 2. Rate of virological/clinic relapse and cumulative rate of suppression after pegylated interferon-alpha regimen was completed.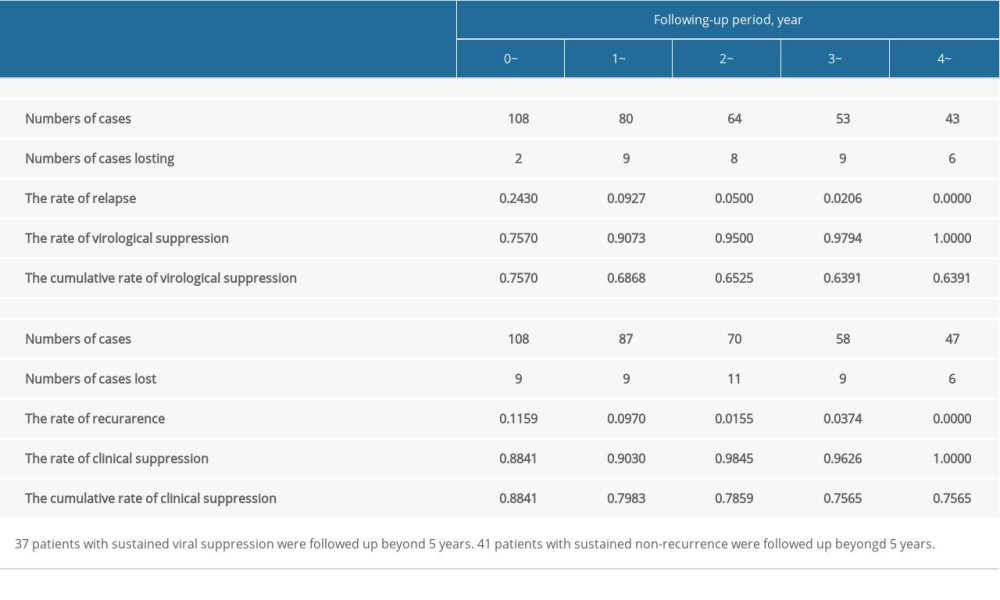 Table 3. Univariate and multivariate Cox analysis of factors associated with sustained virological suppression after pegylated interferon-alpha regimen was completed.
Table 3. Univariate and multivariate Cox analysis of factors associated with sustained virological suppression after pegylated interferon-alpha regimen was completed.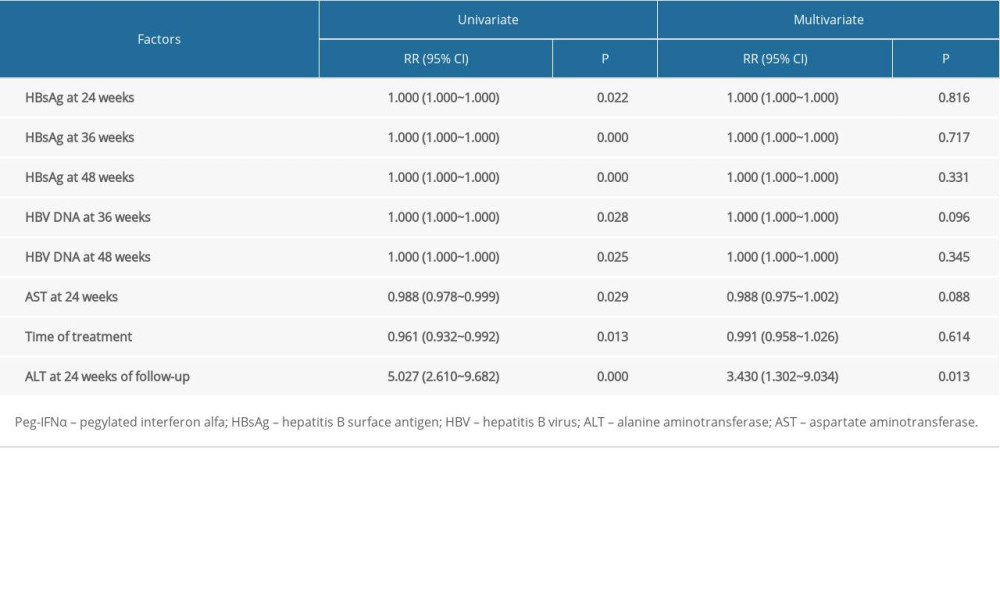 Table 4. Univariate and multivariate Cox analysis of factors associated with sustained clinical suppression after pegylated interferon-alpha regimen was completed.
Table 4. Univariate and multivariate Cox analysis of factors associated with sustained clinical suppression after pegylated interferon-alpha regimen was completed.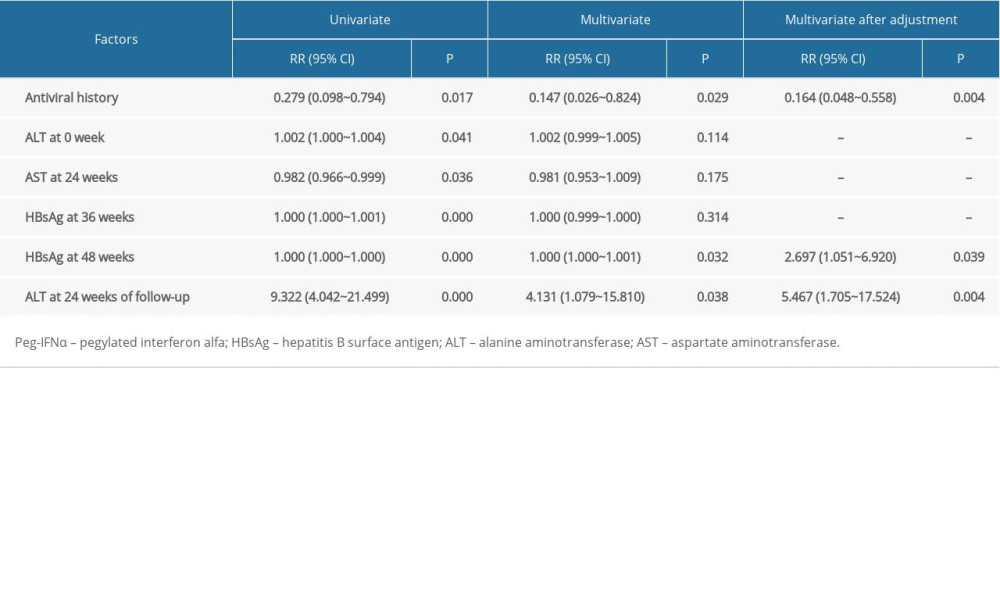
References
1. , EASL 2017 Clinical Practice Guidelines on the management of hepatitis B virus infection: J Hepatol, 2017; 67(2); 370-98
2. Martinez MG, Villeret F, Testoni B, Can we cure hepatitis B virus with novel direct-acting antivirals?: Liver Int, 2020; 40(Suppl 1); 27-34
3. Indolfi G, Easterbrook P, Dusheiko G, Hepatitis B virus infection in children and adolescents: Lancet Gastroenterol Hepatol, 2019; 4(6); 466-76
4. Funk AL, Lu Y, Yoshida K, Efficacy and safety of antiviral prophylaxis during pregnancy to prevent mother-to-child transmission of hepatitis B virus: A systematic review and meta-analysis: Lancet Infect Dis, 2021; 21(1); 70-84
5. Wang FS, Fan JG, Zhang Z, The global burden of liver disease: The major impact of China: Hepatology, 2014; 60(6); 2099-108
6. Sarin S K, Kumar M, Lau G K, Asian-Pacific clinical practice guidelines on the management of hepatitis B: A 2015 update: Hepatol Int, 2016; 10(1); 1-98
7. Yip TC, Wong VW, Chan HL, Tenofovir is associated with lower risk of hepatocellular carcinoma than entecavir in patients with chronic HBV infection in China: Gastroenterology, 2020; 158(1); 215-25.e6
8. Reijnders JG, Perquin MJ, Zhang N, Nucleos(t)ide analogues only induce temporary hepatitis B e antigen seroconversion in most patients with chronic hepatitis B: Gastroenterology, 2010; 139(2); 491-98
9. Terrault NA, Lok A, McMahon BJ, Update on prevention, diagnosis, and treatment of chronic hepatitis B: AASLD 2018 hepatitis B guidance: Hepatology, 2018; 67(4); 1560-99
10. Lau GK, Piratvisuth T, Luo KX, Peginterferon Alfa-2a, lamivudine, and the combination for HBeAg-positive chronic hepatitis B: N Engl J Med, 2005; 352(26); 2682-95
11. Sun J, Ding H, Chen G, Sustained serological and complete responses in HBeAg-positive patients treated with Peginterferon alfa-2b: A 6-year long-term follow-up of a multicenter, randomized, controlled trial in China: BMC Gastroenterol, 2019; 19(1); 65
12. Liang KH, Hsu CW, Chang ML, Peginterferon is superior to nucleos(t)ide analogues for prevention of hepatocellular carcinoma in chronic hepatitis B: J Infect Dis, 2016; 213(6); 966-74
13. Singal AK, Fontana RJ, Meta-analysis: Oral anti-viral agents in adults with decompensated hepatitis B virus cirrhosis: Aliment Pharmacol Ther, 2012; 35(6); 674-89
14. Luo XD, Chen XP, Chen XFOptimal treatment regimen for patients with HBeAg-positive chronic hepatitis B after suboptimal response to 24 weeks of Peg-IFN alpha-2a: Zhonghua Gan Zang Bing Za Zhi, 2017; 25(12); 896-901 [in Chinese]
15. Sarin SK, Kumar M, Lau GK, Asian-Pacific clinical practice guidelines on the management of hepatitis B: A 2015 update: Hepatol Int, 2016; 10(1); 1-98
16. van Nunen AB, Hansen BE, Suh DJ, Durability of HBeAg seroconversion following antiviral therapy for chronic hepatitis B: Relation to type of therapy and pretreatment serum hepatitis B virus DNA and alanine aminotransferase: Gut, 2003; 52(3); 420-24
17. Pan X, Zhang K, Yang X, Relapse rate and associated-factor of recurrence after stopping NUCs therapy with different prolonged consolidation therapy in HBeAg positive patients with CHB: PLoS One, 2013; 8(7); e68568
18. Allweiss L, Dandri M, The role of cccDNA in HBV maintenance: Viruses, 2017; 9(6); 156
19. Cheng PN, Liu WC, Tsai HW, Association of intrahepatic cccDNA reduction with the improvement of liver histology in chronic hepatitis B patients receiving oral antiviral agents: J Med Virol, 2011; 83(4); 602-7
20. Seeger C, Control of viral transcripts as a concept for future HBV therapies: Curr Opin Virol, 2018; 30; 18-23
21. Lai CL, Wong DK, Wong GT, Rebound of HBV DNA after cessation of nucleos/tide analogues in chronic hepatitis B patients with undetectable covalently closed: JHEP Rep, 2020; 2(3); 100112
22. Tanaka E, Matsumoto A, Guidelines for avoiding risks resulting from discontinuation of nucleoside/nucleotide analogs in patients with chronic hepatitis B: Hepatol Res, 2014; 44(1); 1-8
23. Seto WK, Hui AJ, Wong VW, Treatment cessation of entecavir in Asian patients with hepatitis B e antigen negative chronic hepatitis B: A multicentre prospective study: Gut, 2015; 64(4); 667-72
24. Micco L, Peppa D, Loggi E, Differential boosting of innate and adaptive antiviral responses during pegylated-interferon-alpha therapy of chronic hepatitis B: J Hepatol, 2013; 58(2); 225-33
25. Xia Y, Stadler D, Lucifora J, Interferon-γ and tumor necrosis factor-α produced by T cells reduce the HBV persistence form, cccDNA, without cytolysis: Gastroenterology, 2016; 150(1); 194-205
26. Liu S, Zheng H, Huang Y, The effect of peginterferon alpha-2a vs. interferon alpha-2a on intrahepatic covalently closed circular DNA in HBeAg-positive chronic hepatitis B patients: Clin Res Hepatol Gastroenterol, 2016; 40(3); 304-8
27. Aspord C, Bruder CJ, Jacob MC, Remodeling of B-cell subsets in blood during pegylated IFNalpha-2a therapy in patients with chronic hepatitis B infection: PLoS One, 2016; 11(6); e0156200
28. Micco L, Peppa D, Loggi E, Differential boosting of innate and adaptive antiviral responses during pegylated-interferon-alpha therapy of chronic hepatitis B: J Hepatol, 2013; 58(2); 225-33
29. Liu YZ, Hou FQ, Ding P, Pegylated interferon alpha enhances recovery of memory T cells in e antigen positive chronic hepatitis B patients: Virol J, 2012; 9; 274
30. Stelma F, de Niet A, Tempelmans PM, Natural killer cell characteristics in patients with chronic hepatitis B virus (HBV) infection are associated with HBV surface antigen clearance after combination treatment with pegylated interferon alfa-2a and adefovir: J Infect Dis, 2015; 212(7); 1042-51
31. Cao WH, Li MH, Pan CQ, Quantitation of plasmacytoid dendritic cells in chronic hepatitis B patients with HBeAg positivity during PEG-IFN and entecavir therapy: J Interferon Cytokine Res, 2018; 38(5); 197-205
32. Xu F, Song H, Li N, Tan G, HBsAg blocks TYPE I IFN induced up-regulation of A3G through inhibition of STAT3: Biochem Biophys Res Commun, 2016; 473(1); 219-23
33. Kushner T, Sarkar M, Chronic hepatitis B in pregnancy: Clin Liver Dis (Hoboken), 2018; 12(1); 24-28
34. Chilaka VN, Konje JC, Viral Hepatitis in pregnancy: Eur J Obstet Gynecol Reprod Biol, 2021; 256; 287-96
35. Woo A, Kwok R, Ahmed T, Alpha-interferon treatment in hepatitis B: Ann Transl Med, 2017; 5(7); 159
36. Janssen H L, van Zonneveld M, Senturk H, Pegylated interferon alfa-2b alone or in combination with lamivudine for HBeAg-positive chronic hepatitis B: A randomised trial: Lancet, 2005; 365(9454); 123-29
37. Rijckborst V, Sonneveld MJ, Janssen HL, Review article: Chronic hepatitis B – anti-viral or immunomodulatory therapy?: Aliment Pharmacol Ther, 2011; 33(5); 501-13
38. Buster EH, Hansen BE, Lau GK, Factors that predict response of patients with hepatitis B e antigen-positive chronic hepatitis B to peginterferon-alfa: Gastroenterology, 2009; 137(6); 2002-9
39. Sonneveld MJ, Rijckborst V, Boucher CA, Prediction of sustained response to peginterferon alfa-2b for hepatitis B e antigen-positive chronic hepatitis B using on-treatment hepatitis B surface antigen decline: Hepatology, 2010; 52(4); 1251-57
40. Gane E, Jia J, Han K, 69 Neptune study: On-treatment HBsAg level analysis confirms prediction of response observed in phase 3 study of peginterferon alfa-2a in HBeAg-positive patients: J Hepatol, 2011; 54(1); S31
41. Piratvisuth T, Marcellin P, Popescu M, Hepatitis B surface antigen: Association with sustained response to peginterferon alfa-2a in hepatitis B e antigen-positive patients: Hepatol Int, 2013; 7(2); 429-36
42. Wong DK, Seto WK, Fung J, Reduction of hepatitis B surface antigen and covalently closed circular DNA by nucleos(t)ide analogues of different potency: Clin Gastroenterol Hepatol, 2013; 11(8); 1004-10.e1
43. Zhang C, Yang Z, Wang Z, HBV DNA and HBsAg: Early prediction of response to peginterferon α-2a in HBeAg-negative chronic hepatitis B: Int J Med Sci, 2020; 17(3); 383-89
44. Yeh ML, Huang JF, Yu ML, Chuang WL, Hepatitis b infection: Progress in identifying patients most likely to respond to peginterferon alfa: Expert Rev Gastroenterol Hepatol, 2021; 15(4); 427-35
45. Ghany MG, Feld JJ, Chang KM, Serum alanine aminotransferase flares in chronic hepatitis B infection: the good and the bad: Lancet Gastroenterol Hepatol, 2020; 5(4); 406-17
46. Chan H, Ahn SH, Chang TT, Peginterferon lambda for the treatment of HBeAg-positive chronic hepatitis B: A randomized phase 2b study (LIRA-B): J Hepatol, 2016; 64(5); 1011-19
47. Reesink HW, Chan H, Ahn SH, Treatment and HBeAg-status differentiate clinical outcomes following ALT flares – analysis of tenofovir disoproxil fumarate (TDF) plus peginterferon (PEG) combination study for chronic hepatitis B (CHB): Hepatology, 2015; 62(Suppl 1); 333A
48. Marcellin P, Lau GK, Bonino F, Peginterferon alfa-2a alone, lamivudine alone, and the two in combination in patients with HBeAg-negative chronic hepatitis B: N Engl J Med, 2004; 351(12); 1206-17
Figures
 Figure 1. Enrollment and follow-up of the study cohorts. PEG-IFNα – pegylated interferon alfa; HBeAg – hepatitis B e antigen; CHB – chronic hepatitis B.
Figure 1. Enrollment and follow-up of the study cohorts. PEG-IFNα – pegylated interferon alfa; HBeAg – hepatitis B e antigen; CHB – chronic hepatitis B. Figure 2. Comparison of the 5-year cumulative rate of virological suppression in different groups. Group A1, the normal alanine aminotransferase (ALT) value group at 24 weeks after off-therapy; Gropu A2, the abnormal ALT value group at 24 weeks after off-therapy.
Figure 2. Comparison of the 5-year cumulative rate of virological suppression in different groups. Group A1, the normal alanine aminotransferase (ALT) value group at 24 weeks after off-therapy; Gropu A2, the abnormal ALT value group at 24 weeks after off-therapy. Figure 3. (A) Difference in the cumulative rate of clinical suppression between A1 and group A2 (P<0.01). Group A1, the normal alanine aminotransferase (ALT) value group at 24 weeks after off-therapy; Gropu A2, the abnormal ALT value group at 24 weeks after off-therapy. (B) Difference in the cumulative rate of clinical suppression between B1 and group B2 (P=0.035). Group B1, patients with a history of antirival treatment. Group B2, patients without a history of antirival treatment. (C) Difference in the cumulative rate of clinical suppression among group C1, C2 and C3 (P=0.001). Group C1, HBsAg ≤500 COI, the low HBsAg value group at 48 weeks of PEG-IFN-α treatment. Group C2, HBsAg 500–6000 COI, the middle HBsAg value group at 48 weeks of PEG-IFN-α treatment. Group C3, HBsAg >6000 COI, the high HBsAg value group at 48 weeks of PEG-IFN-α treatment. HBsAg – hepatitis B surface antigen; PEG-IFN-α – pegylated interferon-alpha.
Figure 3. (A) Difference in the cumulative rate of clinical suppression between A1 and group A2 (P<0.01). Group A1, the normal alanine aminotransferase (ALT) value group at 24 weeks after off-therapy; Gropu A2, the abnormal ALT value group at 24 weeks after off-therapy. (B) Difference in the cumulative rate of clinical suppression between B1 and group B2 (P=0.035). Group B1, patients with a history of antirival treatment. Group B2, patients without a history of antirival treatment. (C) Difference in the cumulative rate of clinical suppression among group C1, C2 and C3 (P=0.001). Group C1, HBsAg ≤500 COI, the low HBsAg value group at 48 weeks of PEG-IFN-α treatment. Group C2, HBsAg 500–6000 COI, the middle HBsAg value group at 48 weeks of PEG-IFN-α treatment. Group C3, HBsAg >6000 COI, the high HBsAg value group at 48 weeks of PEG-IFN-α treatment. HBsAg – hepatitis B surface antigen; PEG-IFN-α – pegylated interferon-alpha. Tables
 Table 1. Demographic information and clinical characteristics of the enrolled patients.
Table 1. Demographic information and clinical characteristics of the enrolled patients. Table 2. Rate of virological/clinic relapse and cumulative rate of suppression after pegylated interferon-alpha regimen was completed.
Table 2. Rate of virological/clinic relapse and cumulative rate of suppression after pegylated interferon-alpha regimen was completed. Table 3. Univariate and multivariate Cox analysis of factors associated with sustained virological suppression after pegylated interferon-alpha regimen was completed.
Table 3. Univariate and multivariate Cox analysis of factors associated with sustained virological suppression after pegylated interferon-alpha regimen was completed. Table 4. Univariate and multivariate Cox analysis of factors associated with sustained clinical suppression after pegylated interferon-alpha regimen was completed.
Table 4. Univariate and multivariate Cox analysis of factors associated with sustained clinical suppression after pegylated interferon-alpha regimen was completed. Table 1. Demographic information and clinical characteristics of the enrolled patients.
Table 1. Demographic information and clinical characteristics of the enrolled patients. Table 2. Rate of virological/clinic relapse and cumulative rate of suppression after pegylated interferon-alpha regimen was completed.
Table 2. Rate of virological/clinic relapse and cumulative rate of suppression after pegylated interferon-alpha regimen was completed. Table 3. Univariate and multivariate Cox analysis of factors associated with sustained virological suppression after pegylated interferon-alpha regimen was completed.
Table 3. Univariate and multivariate Cox analysis of factors associated with sustained virological suppression after pegylated interferon-alpha regimen was completed. Table 4. Univariate and multivariate Cox analysis of factors associated with sustained clinical suppression after pegylated interferon-alpha regimen was completed.
Table 4. Univariate and multivariate Cox analysis of factors associated with sustained clinical suppression after pegylated interferon-alpha regimen was completed. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








