05 February 2022: Review Articles
A Review of Emerging Biomarkers for Immune Checkpoint Inhibitors in Tumors of the Gastrointestinal Tract
Xuqiang Liao1AE, Gao Li1B, Renzhong Cai1AF, Ru Chen1AG*DOI: 10.12659/MSM.935348
Med Sci Monit 2022; 28:e935348
Abstract
ABSTRACT: In recent years, immune checkpoint inhibition (ICI) therapy has made a tremendous improvement in the treatment of malignant tumors of gastrointestinal tract, especially for those with metastatic or recurrent lesions. However, while some patients benefit from ICI, others do not. In fact, predictive biomarkers can play a crucial role in screening patients who may benefit from a selected or targeted treatment, including immunotherapies such as programmed death-1/programmed death-1 ligand 1 (PD-1/PD-L1) inhibitors. A variety of techniques can be used to detect and quantify tumor biomarkers, each of which has a specific clinical application scenario and limitations. Cancer biomarkers in the gastrointestinal system involve an extremely complex network that requires careful interpretation and analysis. Different prognostic or predictive biomarkers are playing important roles in various tumor types, stages, and pathology/molecular subgroups, sometimes overlapping. Expression levels of biomarkers vary between different tumor types and even between the different lesions in the same tumor, depending on the heterogeneity of the patient, the tumor types, and the techniques of detection. The present systematic review comprehensively summarizes the potential biomarkers of immunotherapy, such as PD-1/PD-L1, total mutation burden (TMB), and tumor-infiltrating lymphocytes (TILs) in various gastrointestinal tumors, including tumors of the colon, stomach, esophagus, liver, and pancreas, to assist future application of immunotherapy and patient selection in clinical practice.
Keywords: Gastrointestinal Neoplasms, Immune Checkpoint Inhibitors, Immunotherapy, B7-H1 Antigen, Biomarkers, Tumor, Gastrointestinal tract, Humans, Lymphocytes, Tumor-Infiltrating, Stomach Neoplasms
Background
Immunotherapy is changing the treatment landscape for a variety of tumors, of which studies on immune checkpoint inhibitors (ICIs) has been at the forefront. In non-small cell lung cancer, immunological checkpoint inhibitors have become one of the standard first-line and sequential-line treatment options. In the standards gastrointestinal system, emerging research on immunological checkpoint inhibitors is also striking [1]. The programmed cell death-1/programmed cell death-ligand 1 (PD-1/PD-L1) pathway and the cytotoxic T-lymphocyte–associated antigen 4 (CTLA-4) pathway are the most well-known immunological checkpoint pathways, and their inhibitors, therefore, are the most widely used. To date, represented by PD-1/PD-L1 inhibitors, a series of immunological checkpoint inhibitors have been approved in various gastrointestinal tumors such as gastric cancer, liver cancer, and colorectal cancer [1,2].
Immunological checkpoint molecules play a key role in the process of cancer evasion, which helps tumor cells escape from immune surveillance, and the PD-1/PD-L1 pathway is one of the most critical for tumor cells to evade immune surveillance. PD-1 is widely expressed on the surface of tumor-infiltrating lymphocytes (TILs), B cells, natural killer cells, monocytes, and dendritic cells (DCs) [3,4]. As the major ligand of PD-1, PD-L1 mainly expresses on the surface of tumors in the tumor microenvironment, and can be detected in several types of tumors, such as lung cancer, gastric cancer, colorectal cancer, kidney cancer, and bladder cancer. PD-L1 expression in these tumor cells can be induced through multiple oncogene signaling pathways, or experiences an adaptive upregulation mediated by infiltrating inflammatory cytokines which are continuously secreted by immune cells [5]. The binding between PD-1 and its ligand PD-L1 inhibits the proliferation, activity, and survival of T cells in the tumor microenvironment, and reduces the expression of immune effector molecules such as interferon γ (IFN-γ), tumor necrosis factor α (TNF-α), and interleukin-2 (IL-2), which drive T cell dysfunction, reducing its anti-tumor effect [6].
Conversely, overexpression of PD-L1 also endows tumor cells with the ability of anti-apoptosis [7] and stimulates the proliferation of tumor stem cells [8]. Therefore, the PD-1/PD-L1 pathway plays an important role in weakening anti-tumor immunity as well as strengthening tumor proliferation. Inhibition of the PD-1/PD-L1 pathway restores the killing activity of T cells against tumors and controls tumor survival, growth, and invasion. Correspondingly, the CTLA-4 pathway acts mainly on the T cell-APC system, affecting the massive activation of T cells and the function of immune effector cells. When it binds to CD80 and CD86 on the surface of antigen-presenting cells (APC), it also inactivates T cells. CTLA-4 antibodies can block the signal by competitively inhibiting CTLA-4, allowing more T cells to resume activation, proliferation, and tumor microenvironment infiltration, reversing regulation of the T cell (Treg)-mediated immunosuppressive state, and enhancing the anti-tumor activity of T cells [9].
However, not all patients can benefit from treatment with immune checkpoint inhibitors. Although PD-1/PD-L1 and CTLA-4 antibodies can significantly improve the objective response rate (ORR) and long-term survival of patients, some patients do not respond to these treatments [2]. After patients respond to immunotherapy, their therapeutic effects usually seem to be durable, and their long-term survival outcomes are often significantly better than those of non-responders [10]. Therefore, determining which subgroup of patients can get the optimal benefit from treatment of immune checkpoint inhibitors is becoming an urgent problem in the clinical scenarios of such therapies. Researchers have begun to focus on biomarkers related to treatment response and effectiveness as predictive biomarkers (defined as a characteristic that is objectively measured and evaluated as an indicator of normal biological processes, pathogenic processes, or pharmacologic responses to a therapeutic intervention) [11], to provide guidance for clinical decision-making. How to accurately find biomarkers that can predict patients’ benefits from immunotherapy will become one of the important research directions in the future [12]. At present, a variety of biomarkers have been extensively studied or have been used clinically, such as PD-L1 expression, total mutation burden (TMB), DNA mismatch-repair defect (dMMR)/high microsatellite instability (MSI), TILs, T cell recognition of tumor-specific neo-antigens, immune transcriptomic signature, diversity of T cell repertoire, gene expression profile, peripheral blood PD-L1 expression, and other cytokines/chemokines [13,14].
Gastrointestinal tumors with different primary lesions and different histopathological features also have different characteristics associated with their immune microenvironment and tumor microenvironment. Therefore, predictive biomarkers for immunotherapy may also vary between different types of gastrointestinal tumors. Systematic studies have been conducted on the predictive biomarkers of immunotherapy for gastrointestinal tumors, including the correlation, test consistency, and clinical significance, and it has been shown that there are still no perfect or well-established biomarkers in immunotherapy [12,15]. Therefore, the present systematic review comprehensively summarizes the potential biomarkers of immunotherapy, including PD-1/PD-L1, total mutation burden (TMB), and tumor-infiltrating lymphocytes (TILs), in different gastrointestinal tumors, such as tumors of the liver, pancreas, biliary tract, and colon, to provide help for the future application of immunotherapy and patient selection in clinical practice.
Pan-Tumor Biomarkers
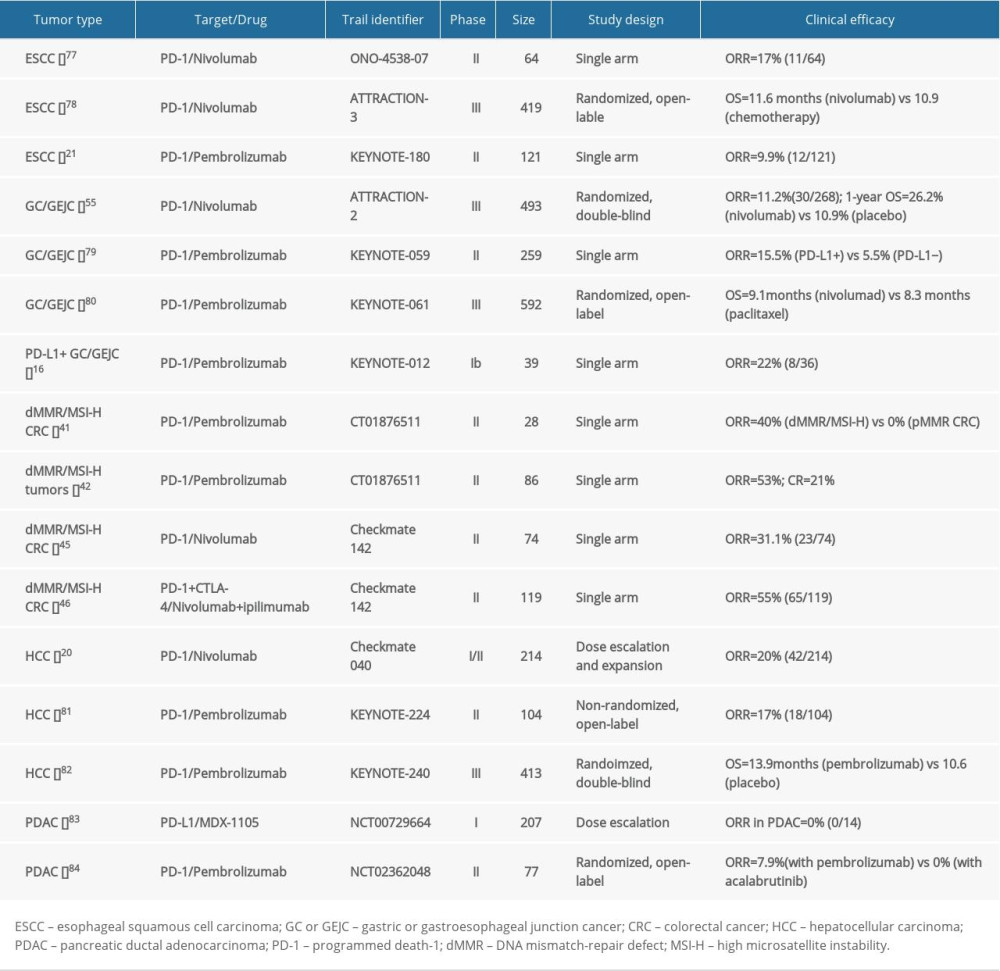
Early research on the use of immunological checkpoint inhibitors in gastrointestinal tumors has been emerging and has shown remarkable efficacy (Table 1). At present, immunotherapy-related pan-tumor predictive biomarkers that are of great interest to researchers include PD-L1 expression, TMB, and MSI-H/dMMR states.
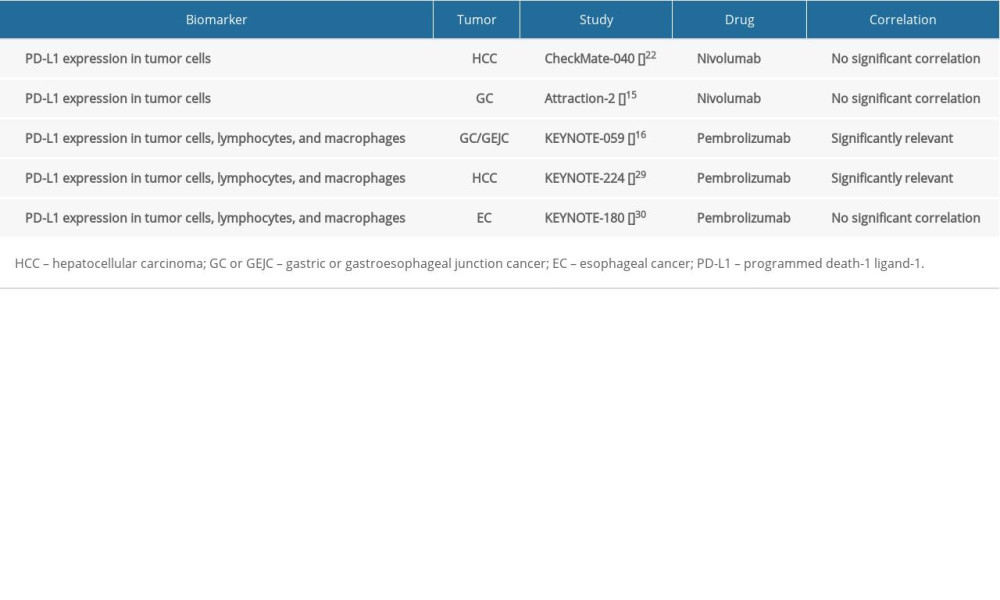
PD-L1 is the most widely studied predictive biomarker for immunotherapy and has mature application in several types of solid tumors such as non-small cell lung cancer [16]. In gastrointestinal tumors, the expression rate of PD-L1 is generally high, at about 64% in colorectal cancer [17], about 40–43.9% in gastric/esophageal cancer [18–20], and about 27% in hepatocellular carcinoma [21], but its predictive value is still controversial (Table 2). Some studies have shown that PD-L1 expression is associated with the efficacy of PD-1 inhibitors, but some studies have shown that regardless of PD-L1 expression levels, patients can benefit from treatment with PD-1 inhibitors [22,23]. However, PD-1/PD-L1 alone is insufficient to determine sensitivity to ICIs.
TMB is a quantitative tool for assessing the total number of mutant genes in the cancer genome. As tumors grow, somatic mutations accumulate, which do not exist in germline DNA [24]. TMB is measured by the non-synonym mutations of each mega base (Mb) in the coding region of the cancer genome [25]. The predictive value of TMB on the efficacy of immunotherapy has been widely verified in various tumor types, such as melanoma, non-small cell lung cancer, urothelial carcinoma, and small cell lung cancer [25]. A pooled analysis in 27 types of solid tumors showed a significant correlation between TMB and the ORR of PD-1 inhibitors (
MSI is a molecular phenotype caused by genomic hypermutation; the MMR system includes enzymes that correct DNA mismatches generated during DNA replication (eg, MLH1, MSH2, and MSH6), thereby preventing transient mutations in differentiated cells from becoming permanent. In contrast, the dMMR status results in persist mutations in the entire genome, especially in the microsatellite region, which in turn leads to a hypermutation phenotype of MSI-high (MSI-H) [28–30]. Therefore, the dMMR state is equal to the MSI-H state from a biological perspective [31]. In certain gastrointestinal tumors, including gastric cancer and colorectal cancer, a high proportion of MSI-H patients (indicating dMMR status) can be observed [32]. Recent studies have shown that MSI-H/dMMR status is associated with significantly better PD-1 inhibitor treatment response and survival outcome, so the Food and Drug Administration (FDA) has approved nivolumab for MSI-H/dMMR-positive patients with metastatic colorectal cancer that progressed after receiving a fluoropyrimidine, oxaliplatin, and irinotecan regimen [33], or pembrolizumab for the sequential-line treatment of solid tumors with MSI-H/dMMR phenotype, without tumor type limitation [34]. However, clinical research showed that MS-stable tumors with high TMB significantly benefit from immunotherapy [35].
Combination of the expression of PD-L1 and presence or absence of TILs can classify tumors into 4 types: type I (PD-L1 positive with TILs), type II (PD-L1 negative with no TILs), type III (PD-L1 positive with no TIL), and type IV (PD-L1 negative with TILs). Among then, type I indicates adaptive immune resistance. Type I tumors are most likely to benefit from single-agent anti-PD-1/L1 blockade, as these tumors have evidence of pre-existing intratumor T cells that are turned off by PD-L1 engagement [36]. A clinical analysis of surgically resected esophageal cancer showed that a stratification based on PD-L1 expression and TIL status was significantly associated with overall survival [37]. Although there are no published studies on these 4 immune types and sensitivity to immune checkpoint inhibitor in gastrointestinal tumor, the classification may be able to predict the response.
The status of other immune effect cells, such as tumor-associated macrophages (TAM) and natural killer (NK) cells, can also contribute to the treatment effects of immune checkpoint inhibitors. There are 2 types of TAM: M1 TAM has anti-tumor activity, while M2 TAM promotes the proliferation of tumors [38]. A study has showed that in the tumor microenvironment of melanoma, CTLA-4 inhibitor responders have higher CD68+CD16+ M1-like TAM ratios at baseline and show decreased Treg infiltration after treatment [39]. As for NK cells, a clinical trial showed that patients with melanoma treated with Nivolumab and patients with tumor infiltration of NK cells after treatment have better clinical response [40].
Colon Cancer
Based on gene expression data, colorectal cancer can be classified into 4 distinct groups, known as the Consensus Molecular Subtypes (CMS). Among them, CMS1 subtype, also referred to as microsatellite instability/immune subgroup, is hypermutated, microsatellite unstable, and has strong immune activated states; it is observed in about 14% of colorectal cancer patients and is immunogenic and hypermutated [41]. Because of the immunogenicity of these tumors, patients in this subgroup may be responsive to immunotherapy.
Colorectal cancer can be divided into 2 groups when taking mutation patterns into consideration: tumors with a dMMR-MSI-H signature (>12 mutation per 106 DNA base tumor burden) and tumors with a pMMR-MSI-L signature (<8.24 mutations per 106 DNA base) [42]. Because of defects in MMR proteins, such as MLH1, MSH2, and MSH6, replication errors of microsatellites cannot be corrected, and they accumulated continuously, which changes the sequence length or base composition of microsatellites, resulting in tumors with high-level microsatellite instability and generating an enlarged neoantigen repertoire for T cell priming [33]. In advanced colorectal cancer, MSI-H/dMMR status has been shown to predict the efficacy of the PD-1 inhibitor pembrolizumab [43]. In a recent study, MSI-H/dMMR status was shown to be related to significantly better PD-1 inhibitor response [44]. Pembrolizumab has been approved by the FDA for MSI-H/dMMR solid tumors, including colorectal cancer and other solid tumors.
Positive PD-L1 expression (with a cut-off value of 10%) is seen in approximately 53% of colorectal cancer patients [45]. High expression of PD-1 and PD-L1 has been shown to be associated with better prognosis [46]; however, in colorectal cancer, PD-L1 does not precisely predict patient response to PD-1/PD-L1 inhibitors [47,48]. In MSI-H/dMMR type colorectal cancer, PD-L1 failed to predict the efficacy of PD-1 inhibitor nivolumab monotherapy or combination therapy (with CTLA-4 inhibitor) [48], or the effectiveness of the PD-1 inhibitor pembrolizumab [43].
In right colon cancer and left colon cancer, the mean TMB/mega base (MB) was relatively high, reported as 11.6/MB and 9.9/MB, respectively. About 12% of patients with right colon cancer can be divided into TMB-high patient subgroups (3% in left colon cancer). A recent study showed that TMB appeared to be an important independent biomarker within MSI-H metastatic colorectal cancer to stratify patients for likelihood of response [49].
In addition to immune cells markers, cytokines receptors such as IL2RB have been identified to be extensively linked to immune-checkpoints in colorectal cancer [50]. Furthermore, it has been proven that the gut microbiome not only acts as a barrier to bacterial invasion but also plays an important role in cytotoxic T-lymphocyte-associated antigen 4 (CTLA-4) blockade, programmed cell death protein 1 (PD-L1) mediation, and T cell stimulation, suggesting that the gut microbiome may be a predictive factor for immunotherapy of colorectal cancer [51].
Stomach Cancer
According to their molecular characteristics, gastric cancer can be divided into 4 subgroups [52]: 1) chromosomal instability (CIN), accounting for about 50% of gastric cancer, which is characterized by intestinal histology and related to more frequent TP53 mutation and RTK-RAS activation; 2) Epstein-Barr virus (EBV), accounting for about 9% of gastric cancer, usually presents with more common genetic alterations including PIK3CA mutation, PD-L1/2 overexpression, EBV-CIMP and CDKN2A silencing, and abnormalities in immune cell signaling; 3) gene stability (GS), accounting for about 20% of gastric cancer, which is characterized by diffuse histology and related to CDH1, RHOA mutations, CLDN18-ARHGAP fusion, and functional abnormalities in cells adhesion pathway; and 4) microsatellite instability (MSI), accounting for about 22% of gastric cancer, with hypermutation and CpG island methylator phenotype (CIMP) status, whose genetic alterations are often involved in MLH1 silencing and mitotic pathways. In these 4 subgroups, EBV and MSI subgroups are considered to be more “immunogenic” and may be more sensitive to immunotherapies [53].
MSI-H/dMMR are present in approximately 8–37% of gastric cancer patients [52,54,55], and compared to MSI-stable/lower tumors, MSI-H type gastric cancer is associated with a better prognosis and is a prognostic biomarker for better survival outcome [54,56]. The appearance of MSI-H/dMMR status is mainly due to epigenetic gene silencing caused by hypermethylation of MLH1 gene promoter [57]. In general, MSI-H/dMMR status is a predictive biomarker for anti-PD-1/PD-L1 drugs in gastric cancer.
PD-L1 overexpression occurs in approximately 25–65% of gastric cancer patients [20,45,58]. In gastric cancer, increased PD-L1 expression is associated with lymph node metastasis, late stage of the disease, and poor prognosis [20]. Currently, PD-L1 expression has been accepted as one of the conditions for the use of certain immunotherapies (eg, pembrolizumab) in clinical treatment of gastric cancer and may guide the application of other therapeutic approaches. However, this conclusion is still controversial because patients with gastric cancer can benefit from some immune checkpoint inhibitors, especially nivolumab, regardless of PD-L1 levels [13].
The average TMB in gastric adenocarcinoma is relatively high (9/MB), and about 11% of patients with gastric adenocarcinoma can be divided into the TMB-high patient subgroup [59]. It was thought that there is no correlation between TMB status and PD-L1 expression level in gastric cancer, but it is highly correlated with MSI/dMMR status [59]. Moreover, a recent clinical trial showed that high TMB may be a predictive marker for OS of advanced gastric cancer patients receiving a new PD-1 antibody, toripalimab, as a single agent [60].
TILs, including T cells, B cells, and natural killer cells, are considered to be signs of host immune response against tumor cells [61,62]. In gastric cancer, a high density of TILs has been confirmed to be significantly associated with PD-L1 expression and MSI-H [55,56]. Different types of infiltrating lymphocytes have different prognostic and predictive implications and may be predictive of anti-PD-1/PD-L1 treatment response [61].
Virus-associated tumors have significant immunogenic features. In gastric cancer, EBV and MSI subtypes have more significant immunological characteristics; in contrast, CIN and GS subtypes have weaker immunological characteristics [52]. In virus-related tumors (such as EBV-related gastric cancer, HPV-related cervical cancer, and HBV-related liver cancer), immunotherapy is usually more effective. For example, 80% of Merkel cell carcinoma is related to the high load of MCV virus; correspondingly, the efficacy of pembrolizumab for Merkel cell carcinoma is ≥50% [63].
Esophageal Cancer
Esophageal adenocarcinomas (EAC) and esophageal squamous cell carcinomas (ESCC) have distinct histopathology, epidemiology, and molecular characteristics. A comprehensive molecular analysis including 164 patients with esophageal cancer showed that ESCC is more similar to squamous cell carcinoma located in other organs, whereas EAC is more similar to the CIN subtype of gastric cancer [64].
MSI-H/dMMR status occurs in <10% of patients with esophageal squamous cell carcinoma [65], which may be associated with rapid disease progression in standard cytotoxic treatment and significantly shorter PFS in first-line chemotherapy; however, distinct from chemotherapy that may result in endogenous resistance, MSI-H/dMMR status during immunotherapy may be associated with more durable remission [66].
Positive PD-L1 expression (with a cut-off value of 5%) is seen in approximately 20% of patients with esophageal cancer (approximately 44% of squamous cell carcinoma patients with a positive cut-off value of 10%) [45]. Increased PD-L1 expression is associated with lymph node metastasis, later disease stage, and poor prognosis [20]. For immunotherapy, positive PD-L1 expression defined as PD-L1 combined positive score (CPS) ≥10 seems to be associated with significantly better response and improved survival compared with chemotherapy in different treatment lines [67,68]. A combination of PD-L1 and TIL status may serve as predictive biomarkers in PD-1/PD-L1-targeting therapy for patients with surgically resected esophageal cancer [37].
The average TMB of patients with esophageal cancer is relatively low (6.7/MB for EAC and 6.4/MB for ESCC). About 2.0% of patients with EAC and 0% of those with ESCC can be divided into TMB-high subgroups [59]. To date, there has been no large-scale study demonstrating the relationship between TMB and immunotherapy in esophageal cancer.
Liver Cancer
Hepatocellular carcinomas with different histopathological characteristics and stages also have different molecular features [69]. In hepatocellular carcinoma, the incidence of MSI-H/dMMR is relatively low, varying from 0% to 18% [70]. Moreover, dMMR does not involve the pathogenesis mechanism of hepatocellular carcinoma [70]. Therefore, no clinical study has confirmed the clinical significance of MSI-H as a predictive biomarker for immunotherapy in hepatocellular carcinoma.
Positive PD-L1 expression (with a cut-off value of 5%) can be seen in approximately 15% of hepatocellular carcinoma patients [45]. In the Checkmate-040 study, the objective response rates for hepatocellular carcinoma patients with PD-L1 ≥1% and PD-L1 <1% were 27% and 12%, respectively [22].
In hepatocellular carcinoma, TMB has been shown to be associated with the expression of PD-L1 (
HBV and HCV infection increase the risk of hepatocellular carcinoma. Viral dynamics may have a predictive role for response to certain treatments. In patients with hepatocellular carcinoma receiving sorafenib and without prophylactic antiviral therapy, high HBV DNA levels have been shown to be an independent predictive factor for poor survival (
A better understanding of the etiology of hepatocellular carcinoma may contribute to the development of more practical and accurate biomarkers. Potential biomarkers include [72]: 1) miRNAs, such as miR-4147 that can allele-specifically regulate PD-1 expression through interaction with the 3′ UTR of
Pancreas Cancer
Pancreatic cancer with different histopathological types may have distinct molecular and immune characteristics and thus may affect treatment decisions [78]. In pancreatic cancer, the incidence of MSI-H/dMMR is 13–22%, which may be a predictive biomarker for response to anti-PD-1/PD-L1 drugs [79,80]. PD-L1 overexpression can be observed in approximately 19–40% of patients with pancreatic cancer and is associated with poor prognosis [45]. In pancreatic cancer, TMB has been demonstrated to be positively associated with PD-L1 expression (
Conclusions
Modern personalized medicine now includes the screening of tumor tissue for predictive biomarkers for targeted therapy. Immunotherapy that includes PD-1/PD-L1 inhibitors has a role in the management of patients with advanced gastrointestinal malignancy. Prognostic and predictive biomarkers also have important roles in identifying and targeting molecular subgroups of tumors and identifying tumor heterogeneity between primary and secondary tumors.
This review has highlighted how the identification of biomarkers for gastrointestinal tumors has affected treatment decisions. For example, MSI-H/dMMR status is required before implementing treatment with pembrolizumab and nivolumab, and tumor PD-L1 expression is required before implementing treatment with pembrolizumab and atezolizumab. Some emerging biomarkers such as TMB contribute to the efficacy of PD-1/PD-L1 inhibitors. Continuing advances and methods of detecting biomarkers expressed by tumors of the gastrointestinal tract will guide future targeted immunotherapy.
References
1. Grierson P, Lim KH, Amin M, Immunotherapy in gastrointestinal cancers: J Gastrointest Oncol, 2017; 8(3); 474-84
2. Tannapfel A, Reinacher-Schick A, Hacker U, Immunotherapy: New options in gastrointestinal cancers?: Visc Med, 2019; 35(1); 47-51
3. Pardoll DM, The blockade of immune checkpoints in cancer immunotherapy: Nat Rev Cancer, 2012; 12(4); 252-64
4. Iwai Y, Ishida M, Tanaka Y, Involvement of PD-L1 on tumor cells in the escape from host immune system and tumor immunotherapy by PD-L1 blockade: Proc Natl Acad Sci USA, 2002; 99(19); 12293-97
5. Ji M, Liu Y, Li Q, PD-1/PD-L1 pathway in non-small-cell lung cancer and its relation with EGFR mutation: J Transl Med, 2015; 13; 5
6. Buchbinder EI, Desai A, CTLA-4 and PD-1 pathways: Similarities, differences, and implications of their inhibition: Am J Clin Oncol, 2016; 39(1); 98-106
7. Azuma T, Yao S, Zhu G, B7-H1 is a ubiquitous antiapoptotic receptor on cancer cells: Blood, 2008; 111(7); 3635-43
8. Yang Y, Wu KE, Zhao E, B7-H1 enhances proliferation ability of gastric cancer stem-like cells as a receptor: Oncol Lett, 2015; 9(4); 1833-38
9. Mahoney KM, Rennert PD, Freeman GJ, Combination cancer immunotherapy and new immunomodulatory targets: Nat Rev Drug Discov, 2015; 14(8); 561-84
10. Gettinger S, Horn L, Jackman D, Five-year follow-up of nivolumab in previously treated advanced non-small-cell lung cancer: Results from the CA209-003 Study: J Clin Oncol, 2018; 36(17); 1675-84
11. Wagner JA, Atkinson AJ, Measuring biomarker progress: Clin Pharmacol Ther, 2015; 98(1); 2-5
12. Hazama S, Tamada K, Yamaguchi Y, Current status of immunotherapy against gastrointestinal cancers and its biomarkers: Perspective for precision immunotherapy: Ann Gastroenterol Surg, 2018; 2(4); 289-303
13. Ma W, Gilligan BM, Yuan J, Li T, Current status and perspectives in translational biomarker research for PD-1/PD-L1 immune checkpoint blockade therapy: J Hematol Oncol, 2016; 9(1); 47
14. Manson G, Norwood J, Marabelle A, Biomarkers associated with checkpoint inhibitors: Ann Oncol, 2016; 27(7); 1199-206
15. Zeng Z, Yang B, Liao Z, Biomarkers in immunotherapy-based precision treatments of digestive system tumors: Front Oncol, 2021; 11; 650481
16. Diggs LP, Hsueh EC, Utility of PD-L1 immunohistochemistry assays for predicting PD-1/PD-L1 inhibitor response: Biomark Res, 2017; 5; 12
17. Dong H, Strome SE, Salomao DR, Tumor-associated B7-H1 promotes T-cell apoptosis: A potential mechanism of immune evasion: Nat Med, 2002; 8(8); 793-800
18. Muro K, Chung HC, Shankaran V, Pembrolizumab for patients with PD-L1-positive advanced gastric cancer (KEYNOTE-012): A multicentre, open-label, phase 1b trial: Lancet Oncol, 2016; 17(6); 717-26
19. Wu P, Wu D, Li L, PD-L1 and survival in solid tumors: A meta-analysis: PLoS One, 2015; 10(6); e131403
20. Raufi AG, Klempner SJ, Immunotherapy for advanced gastric and esophageal cancer: Preclinical rationale and ongoing clinical investigations: J Gastrointest Oncol, 2015; 6(5); 561-69
21. Jung HI, Jeong D, Ji S, Overexpression of PD-L1 and PD-L2 is associated with poor prognosis in patients with hepatocellular carcinoma: Cancer Res Treat, 2017; 49(1); 246-54
22. El-Khoueiry AB, Sangro B, Yau T, Nivolumab in patients with advanced hepatocellular carcinoma (CheckMate 040): An open-label, non-comparative, phase 1/2 dose escalation and expansion trial: Lancet, 2017; 389(10088); 2492-502
23. Shah MA, Kojima T, Hochhauser D, Efficacy and safety of pembrolizumab for heavily pretreated patients with advanced, metastatic adenocarcinoma or squamous cell carcinoma of the esophagus: The phase 2 KEYNOTE-180 study: JAMA Oncol, 2019; 5(4); 546-50
24. Stratton MR, Campbell PJ, Futreal PA, The cancer genome: Nature, 2009; 458(7239); 719-24
25. Samstein RM, Lee CH, Shoushtari AN, Tumor mutational load predicts survival after immunotherapy across multiple cancer types: Nat Genet, 2019; 51(2); 202-6
26. Yarchoan M, Hopkins A, Jaffee EM, Tumor mutational burden and response rate to PD-1 inhibition: N Engl J Med, 2017; 377(25); 2500-1
27. Goodman AM, Kato S, Bazhenova L, Tumor mutational burden as an independent predictor of response to immunotherapy in diverse cancers: Mol Cancer Ther, 2017; 16(11); 2598-608
28. Li GM, Mechanisms and functions of DNA mismatch repair: Cell Res, 2008; 18(1); 85-98
29. Kawakami H, Zaanan A, Sinicrope FA, Microsatellite instability testing and its role in the management of colorectal cancer: Curr Treat Options Oncol, 2015; 16(7); 30
30. Jalal S, Earley JN, Turchi JJ, DNA repair: From genome maintenance to biomarker and therapeutic target: Clin Cancer Res, 2011; 17(22); 6973-84
31. Richman S, Deficient mismatch repair: Read all about it (Review): Int J Oncol, 2015; 47(4); 1189-202
32. Lee V, Murphy A, Le DT, Diaz LJ, Mismatch repair deficiency and response to immune checkpoint blockade: Oncologist, 2016; 21(10); 1200-11
33. Ganesh K, Stadler ZK, Cercek A, Immunotherapy in colorectal cancer: Rationale, challenges and potential: Nat Rev Gastroenterol Hepatol, 2019; 16(6); 361-75
34. Marcus L, Lemery SJ, Keegan P, Pazdur R, FDA approval summary: Pembrolizumab for the treatment of microsatellite instability-high solid tumors: Clin Cancer Res, 2019; 25(13); 3753-58
35. Goodman AM, Sokol ES, Frampton GM, Microsatellite-stable tumors with high mutational burden benefit from immunotherapy: Cancer Immunol Res, 2019; 7(10); 1570-73
36. Teng MW, Ngiow SF, Ribas A, Smyth MJ, Classifying cancers based on T-cell infiltration and PD-L1: Cancer Res, 2015; 75(11); 2139-45
37. Yagi T, Baba Y, Ishimoto T, PD-L1 expression, tumor-infiltrating lymphocytes, and clinical outcome in patients with surgically resected esophageal cancer: Ann Surg, 2019; 269(3); 471-78
38. Ino Y, Yamazaki-Itoh R, Shimada K, Immune cell infiltration as an indicator of the immune microenvironment of pancreatic cancer: Br J Cancer, 2013; 108(4); 914-23
39. Romano E, Kusio-Kobialka M, Foukas PG, Ipilimumab-dependent cell-mediated cytotoxicity of regulatory T cells ex vivo by nonclassical monocytes in melanoma patients: Proc Natl Acad Sci USA, 2015; 112(19); 6140-45
40. Riaz N, Havel JJ, Makarov V, Tumor and microenvironment evolution during immunotherapy with nivolumab: CELL, 2017; 171(4); 934-49
41. Thanki K, Nicholls ME, Gajjar A, Consensus molecular subtypes of colorectal cancer and their clinical implications: Int Biol Biomed J, 2017; 3(3); 105-11
42. Cancer Genome Atlas Network, Comprehensive molecular characterization of human colon and rectal cancer: Nature, 2012; 487(7407); 330-37
43. Le DT, Uram JN, Wang H, PD-1 blockade in tumors with mismatch-repair deficiency: N Engl J Med, 2015; 372(26); 2509-20
44. Le DT, Durham JN, Smith KN, Mismatch repair deficiency predicts response of solid tumors to PD-1 blockade: Science, 2017; 357(6349); 409-13
45. Patel SP, Kurzrock R, PD-L1 Expression as a predictive biomarker in cancer immunotherapy: Mol Cancer Ther, 2015; 14(4); 847-56
46. Li Y, Liang L, Dai W, Prognostic impact of programed cell death-1 (PD-1) and PD-ligand 1 (PD-L1) expression in cancer cells and tumor infiltrating lymphocytes in colorectal cancer: Mol Cancer, 2016; 15(1); 55
47. Overman MJ, McDermott R, Leach JL, Nivolumab in patients with metastatic DNA mismatch repair-deficient or microsatellite instability-high colorectal cancer (CheckMate 142): An open-label, multicentre, phase 2 study: Lancet Oncol, 2017; 18(9); 1182-91
48. Overman MJ, Lonardi S, Wong K, Durable clinical benefit with nivolumab plus ipilimumab in DNA mismatch repair-deficient/microsatellite instability-high metastatic colorectal cancer: J Clin Oncol, 2018; 36(8); 773-79
49. Schrock AB, Ouyang C, Sandhu J, Tumor mutational burden is predictive of response to immune checkpoint inhibitors in MSI-high metastatic colorectal cancer: Ann Oncol, 2019; 30(7); 1096-103
50. Alderdice M, Craig SG, Humphries MP, Evolutionary genetic algorithm identifies IL2RB as a potential predictive biomarker for immune-checkpoint therapy in colorectal cancer: NAR Genom Bioinform, 2021; 3(2); lqab016
51. Temraz S, Nassar F, Nasr R, Gut microbiome: A promising biomarker for immunotherapy in colorectal cancer: Int J Mol Sci, 2019; 20(17); 4155
52. Cancer Genome Atlas Research Network, Comprehensive molecular characterization of gastric adenocarcinoma: Nature, 2014; 513(7517); 202-9
53. Wang M, Busuttil RA, Pattison S, Immunological battlefield in gastric cancer and role of immunotherapies: World J Gastroenterol, 2016; 22(28); 6373-84
54. Zhu L, Li Z, Wang Y, Microsatellite instability and survival in gastric cancer: A systematic review and meta-analysis: Mol Clin Oncol, 2015; 3(3); 699-705
55. Kawazoe A, Kuwata T, Kuboki Y, Clinicopathological features of programmed death ligand 1 expression with tumor-infiltrating lymphocyte, mismatch repair, and Epstein-Barr virus status in a large cohort of gastric cancer patients: Gastric Cancer, 2017; 20(3); 407-15
56. Jin Z, Yoon HH, The promise of PD-1 inhibitors in gastro-esophageal cancers: Microsatellite instability vs. PD-L1: J Gastrointest Oncol, 2016; 7(5); 771-88
57. Baniak N, Senger JL, Ahmed S, Gastric biomarkers: A global review: World J Surg Oncol, 2016; 14(1); 212
58. Kang YK, Boku N, Satoh T, Nivolumab in patients with advanced gastric or gastro-oesophageal junction cancer refractory to, or intolerant of, at least two previous chemotherapy regimens (ONO-4538-12, ATTRACTION-2): A randomised, double-blind, placebo-controlled, phase 3 trial: Lancet, 2017; 390(10111); 2461-71
59. Salem ME, Xiu J, Weinberg BA, Characterization of tumor mutation burden (TMB) in gastrointestinal (GI) cancers: J Clin Oncol, 2017; 35(4 Suppl); 530
60. Wang F, Wei XL, Wang FH, Safety, efficacy and tumor mutational burden as a biomarker of overall survival benefit in chemo-refractory gastric cancer treated with toripalimab, a PD-1 antibody in phase Ib/II clinical trial NCT02915432: Ann Oncol, 2019; 30(9); 1479-86
61. Kang BW, Kim JG, Lee IH, Clinical significance of tumor-infiltrating lymphocytes for gastric cancer in the era of immunology: World J Gastrointest Oncol, 2017; 9(7); 293-99
62. Wein L, Savas P, Luen SJ, Clinical validity and utility of tumor-infiltrating lymphocytes in routine clinical practice for breast cancer patients: Current and future directions: Front Oncol, 2017; 7; 156
63. Topalian SL, Taube JM, Anders RA, Pardoll DM, Mechanism-driven biomarkers to guide immune checkpoint blockade in cancer therapy: Nat Rev Cancer, 2016; 16(5); 275-87
64. Cancer Genome Atlas Research Network, Integrated genomic characterization of oesophageal carcinoma: Nature, 2017; 541(7636); 169-75
65. Kubo N, Yashiro M, Ohira M, Frequent microsatellite instability in primary esophageal carcinoma associated with extraesophageal primary carcinoma: Int J Cancer, 2005; 114(2); 166-73
66. Janjigian YY, Sanchez-Vega F, Jonsson P, Genetic predictors of response to systemic therapy in esophagogastric cancer: Cancer Discov, 2018; 8(1); 49-58
67. Kato K, Shah MA, Enzinger P, KEYNOTE-590: Phase III study of first-line chemotherapy with or without pembrolizumab for advanced esophageal cancer: Future Oncol, 2019; 15(10); 1057-66
68. Kojima T, Muro K, Francois E, Hsu C, Pembrolizumab versus chemotherapy as second-line therapy for advanced esophageal cancer: Phase III KEYNOTE-181 study: J clin oncol, 2019; 37(4 Suppl); 2
69. Khemlina G, Ikeda S, Kurzrock R, The biology of Hepatocellular carcinoma: Implications for genomic and immune therapies: Mol Cancer, 2017; 16(1); 149
70. Goumard C, Desbois-Mouthon C, Wendum D, Low levels of microsatellite instability at simple repeated sequences commonly occur in human hepatocellular carcinoma: Cancer Genomics Proteomics, 2017; 14(5); 329-39
71. Lim S, Han J, Kim GM, Hepatitis B viral load predicts survival in hepatocellular carcinoma patients treated with sorafenib: J Gastroenterol Hepatol, 2015; 30(6); 1024-31
72. Mathew S, Ali A, Abdel-Hafiz H, Biomarkers for virus-induced hepatocellular carcinoma (HCC): Infect Genet Evol, 2014; 26; 327-39
73. Zhang G, Li N, Li Z, microRNA-4717 differentially interacts with its polymorphic target in the PD1 3′ untranslated region: A mechanism for regulating PD-1 expression and function in HBV-associated liver diseases: Oncotarget, 2015; 6(22); 18933-44
74. Jiang C, Liu X, Wang M, Lv G, Wang G, High blood miR-802 is associated with poor prognosis in HCC patients by regulating DNA damage response 1 (REDD1)-mediated function of T cells: Oncol Res, 2019; 27(9); 1025-34
75. Riener MO, Stenner F, Liewen H, Golgi phosphoprotein 2 (GOLPH2) expression in liver tumors and its value as a serum marker in hepatocellular carcinomas: Hepatology, 2009; 49(5); 1602-9
76. Wang M, Zhai X, Li J, The role of cytokines in predicting the response and adverse events related to immune checkpoint inhibitors: Front Immunol, 2021; 12; 670391
77. Chen J, Gingold JA, Su X, Immunomodulatory TGF-beta signaling in hepatocellular carcinoma: Trends Mol Med, 2019; 25(11); 1010-23
78. Ghaneh P, Costello E, Neoptolemos JP, Biology and management of pancreatic cancer: Gut, 2007; 56(8); 1134-52
79. Nakata B, Wang YQ, Yashiro M, Prognostic value of microsatellite instability in resectable pancreatic cancer: Clin Cancer Res, 2002; 8(8); 2536-40
80. Yamamoto H, Itoh F, Nakamura H, Genetic and clinical features of human pancreatic ductal adenocarcinomas with widespread microsatellite instability: Cancer Res, 2001; 61(7); 3139-44
81. Ammannagari N, Atasoy A, Current status of immunotherapy and immune biomarkers in gastro-esophageal cancers: J Gastrointest Oncol, 2018; 9(1); 196-207
82. Hopkins AC, Yarchoan M, Durham JN, T cell receptor repertoire features associated with survival in immunotherapy-treated pancreatic ductal adenocarcinoma: JCI Insight, 2018; 3(13); e122092
83. Kudo T, Hamamoto Y, Kato K, Nivolumab treatment for oesophageal squamous-cell carcinoma: An open-label, multicentre, phase 2 trial: Lancet Oncol, 2017; 18(5); 631-39
84. Kato K, Cho BC, Takahashi M, Nivolumab versus chemotherapy in patients with advanced oesophageal squamous cell carcinoma refractory or intolerant to previous chemotherapy (ATTRACTION-3): A multicentre, randomised, open-label, phase 3 trial: Lancet Oncol, 2019; 20(11); 1506-17
In Press
21 Mar 2024 : Meta-Analysis
Economic Evaluation of COVID-19 Screening Tests and Surveillance Strategies in Low-Income, Middle-Income, a...Med Sci Monit In Press; DOI: 10.12659/MSM.943863
10 Apr 2024 : Clinical Research
Predicting Acute Cardiovascular Complications in COVID-19: Insights from a Specialized Cardiac Referral Dep...Med Sci Monit In Press; DOI: 10.12659/MSM.942612
06 Mar 2024 : Clinical Research
Enhanced Surgical Outcomes of Popliteal Cyst Excision: A Retrospective Study Comparing Arthroscopic Debride...Med Sci Monit In Press; DOI: 10.12659/MSM.941102
06 Mar 2024 : Clinical Research
Prevalence and Variation of Medical Comorbidities in Oral Surgery Patients: A Retrospective Study at Jazan ...Med Sci Monit In Press; DOI: 10.12659/MSM.943884
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952