14 May 2022: Clinical Research
Case-Control Study to Investigate the Association Between Serum Apolipoprotein B/A1 Ratio and Atrial Fibrillation by Sex in 920 Patients from China
Xia Zhong1ACE, Huachen Jiao2DFG*, Dongsheng Zhao1BC, Jing Teng1AF, Mengqi YangDOI: 10.12659/MSM.936425
Med Sci Monit 2022; 28:e936425
Abstract
BACKGROUND: The serum apolipoprotein B/A1 ratio (APOB/APOA1) has been shown to predict cardiovascular events, whereas the effect of the APOB/APOA1 ratio on atrial fibrillation (AF) is less known. We investigated the association between the APOB/APOA1 ratio and AF by sex in 920 patients from China.
MATERIAL AND METHODS: We reviewed clinical data on 1840 hospitalized patients, including 920 patients with AF (male/female: 460/460, age: 68.62±10.36 years) and 920 age- and sex-matched patients without AF with sinus rhythm in China between January 2019 and September 2021. Pearson correlation analysis was performed to investigate the correlation between APOB/APOA1 ratio and AF-related metabolic factors. Logistic regression analysis was used to determine the odds ratios (ORs) and 95% confidence intervals (CIs).
RESULTS: Low serum APOB/APOA1 ratios in male and female patients were significantly associated with AF after adjusting for confounding factors (OR 0.159, 95% CI 0.058-0.432, P<0.05). Serum APOB/APOA1 ratio was positively correlated with triglyceride (TG) (r=0.146, P<0.05) and total cholesterol (TC) (r=0.227, P<0.05) and was negatively correlated with albumin (ALB) (r=-0.128, P<0.05) and prealbumin (PAB) (r=-0.107, P<0.05). There was no significant difference of APOB/APOA1 ratio in different subtypes, complications, and statin use in patients with AF (P>0.05).
CONCLUSIONS: A low serum APOB/APOA1 ratio in male and female patients from China was significantly related to AF. This finding implies that a low serum APOB/APOA1 ratio may be associated with the causes of AF. Further studies are needed to determine causalities.
Keywords: Apolipoprotein A-I, Apolipoprotein B (3304-3317), Atrial Fibrillation, Gender Identity, Inflammation, Aged, Apolipoproteins B, Case-Control Studies, China, Female, Humans, Male, Middle Aged, Triglycerides
Background
Atrial fibrillation (AF) is the most common and challenging clinically significant sustained cardiac arrhythmia, affecting almost 33 million people worldwide [1–3]. As a growing health threat, AF can contribute to an increased risk of heart failure, stroke, cognitive impairment, systemic embolism, and even death [4–8], with associated hospitalization rates, mortality, and a considerable healthcare burden [9]. Currently, antiarrhythmic drugs and catheter ablation are clinically recommended to control heart rate and rhythm in patients [10]. Although antiarrhythmic drugs and catheter ablation are recommended as effective treatments, the success rate of a single surgery has been reported to be only 60% to 70%, and the potential complications of the surgery cannot be ignored [11–14]. Risk factor management as an upstream noninvasive treatment has a potential beneficial effect on AF [15]. Exploring available blood biomarkers related to AF may contribute to understanding its underlying pathological mechanism and implementing corresponding prevention strategies.
Guidelines have been recommended for treating cardiovascular disease through the management of blood lipid profiles [16]. The nontraditional lipid biomarkers apolipoprotein A1 (APOA1) and apolipoprotein B (APOB) are the key protein moieties correlated with HDL cholesterol (HDL-C) and LDL cholesterol (LDL-C), respectively; they play a significant role in lipid metabolism and are both considered to be associated with the risk of cardiovascular disease [17,18]. Generally, APOA1 is atheroprotective and APOB is anti-atheroprotective [19,20]. In addition, several studies have reported that the ApoB/ApoA1 ratio is a convenient and accurate specific marker for cardiovascular risk events [21], having an even better predictive advantage than lipid adjuvant in coronary heart disease and ischemic events [22–24]. Some researchers have recently confirmed that APOA1 and APOB are closely related to inflammation and oxidative stress, which are important reaction chains for AF initiation and complexity [25–27]. Moreover, the effects of dyslipidemia on AF remain controversial.
In this study, we aimed to explore the association between the serum APOB/APOA1 ratio and AF by sex in 920 patients from China.
Material and Methods
STUDY DESIGN:
This study followed the principles of the Helsinki Declaration and passed the review of the Medical Research Ethics Committee of the Affiliated Hospital of Shandong University of Traditional Chinese Medicine. No informed consent was required because the data were anonymized. This study used a matched case-control design of 1840 hospitalized patients’ (male/female: 917/923, age: 68.35±10.92 years) electronic records from the Affiliated Hospital of Shandong University of Traditional Chinese Medicine. Inclusion criteria were as follows: Cases comprised patients aged 29 to 85 years diagnosed with AF between January 2019 and September 2021. Each patient with complete clinical data had age- and sex-matched controls. This study eventually involved 920 patients with AF and 920 controls. Meanwhile, we excluded patients with a history of cardiac surgery, heart failure, valvular disease, or malignancy, as well as patients with current liver or kidney dysfunction, hyperthyroidism, and infection and those taking diuretics. Patients with AF were diagnosed in a professional manner by their physicians, excluding self-described patients. We systematically screened clinical variables for all participants from the electronic medical record system, including baseline clinical data, laboratory data, subtypes of AF, and complications of AF. In addition, the data of patients with AF were stratified by sex and serum APOB/APOA1 ratio.
SCREENED VARIABLES:
The baseline data of patients were selected and included sex, age, types and complications of AF, use of statins, CCBs, β-blockers, and ACEI/ARB, as well as laboratory indicators, including serum APOB levels (0.75–1.55g/L), serum APOA1 levels (1.10–1.19 g/L), serum APOB/APOA1 ratio, blood lipid profiles (TC [3.0–5.7 mmol/L], TG [0.4–1.7 mmol/L], LDL-C [<3.63 mmol/L], HDL-C [>1.04 mmol/L]), alanine aminotransferase (ALT; 7–40 U/L), aspartate aminotransferase (AST; 13–35 U/L), lipoprotein (a) (Lp[a]; 0–30 mg/dL), serum uric acid (SUA; 2.6–6.0 mg/dL), serum creatinine (SCr; 41–81 μmoI/L), serum albumin (ALB; 40–55 U/L), and prealbumin (PAB; 18–35 g/L). All laboratory measurements were taken in strict accordance with hospital standards. The turbidimetric inhibition immunoassay was used to determine APOB and APOA1. Serum APOB/APOA1 ratios were divided into 3 tertiles by sex (male: ≤0.61, 0.61–0.83, ≥0.83; female: ≤0.56, 0.56–0.77, ≥0.77).
STATISTICAL ANALYSIS:
Data analysis was conducted using SPSS software (version 26.0; IBM Corp, Armonk, NY, USA) or GraphPad Prism software (version 9.0.0). Continuous data were presented as mean±standard deviation and compared using the
Results
CLINICAL CHARACTERISTICS:
As shown in Table 1, 920 patients with AF (male/female: 460/460, 68.62±10.36 years) and 920 age- and sex-matched patients without AF with sinus rhythm (male/female: 450/463, 68.08±11.46 years) were included. Patients with AF were more likely than controls to have hypertension, coronary heart disease, and diabetes (P<0.05) and were more likely to use statins, CCBs, β-blockers, and ACEI/ARB (P<0.05). Patients with AF also had significantly lower serum levels of APOAl, APOB, APOB/APOA1 ratio, TC, TG, LDL-C, HDL-C, ALB, and PAB (P<0.05) than controls and had significantly higher serum levels of AST, SUA, and SCr (P<0.05) than controls.
CORRELATION BETWEEN SERUM APOB/APOA1 RATIO AND AF:
As shown in Table 2, after adjusting for hypertension, CHD, diabetes, β-blockers, CCB, ACEI/ARB, and statins, the serum APOB/APOA1 ratio was related to AF (OR 0.312, 95% CI 0.192–0.508, P<0.05). After adjusting for TG, TC, LDL-C, HDL-C, AST, SUA, Scr, PAB, and ALB, the serum APOB/APOA1 ratio was still associated with AF (OR 0.055, 95% CI 0.024–0.126, P<0.05). Further, after adjusting for all confounding factors, the serum APOB/APOA1 ratio remained independently negatively correlated with AF (OR 0.159, 95% CI 0.058–0.432, P<0.05). Moreover, the serum APOB/APOA1 ratio was negatively correlated with AF in men and women (P<0.05).
DIFFERENCES OF SERUM APOB/APOA1 RATIO IN MALE AND FEMALE PATIENTS WITH AF:
As shown in Table 3, there was no significant difference in serum APOB/APOA1 ratio in men and women with AF, regardless of AF subtypes, complications, and whether or not statins were used (P>0.05).
DIFFERENCES OF SERUM APOB/APOA1 RATIO IN PATIENTS WITH AF BY DIFFERENT SUBTYPES, COMPLICATIONS, AND STATIN USE:
As shown in Table 4, the results showed there was no significant difference in serum APOB/APOA1 ratio in patients with AF by different subtypes, complications, and statin use (P>0.05).
CORRELATION BETWEEN SERUM APOB/APOA1 RATIO AND AF-RELATED METABOLIC FACTORS:
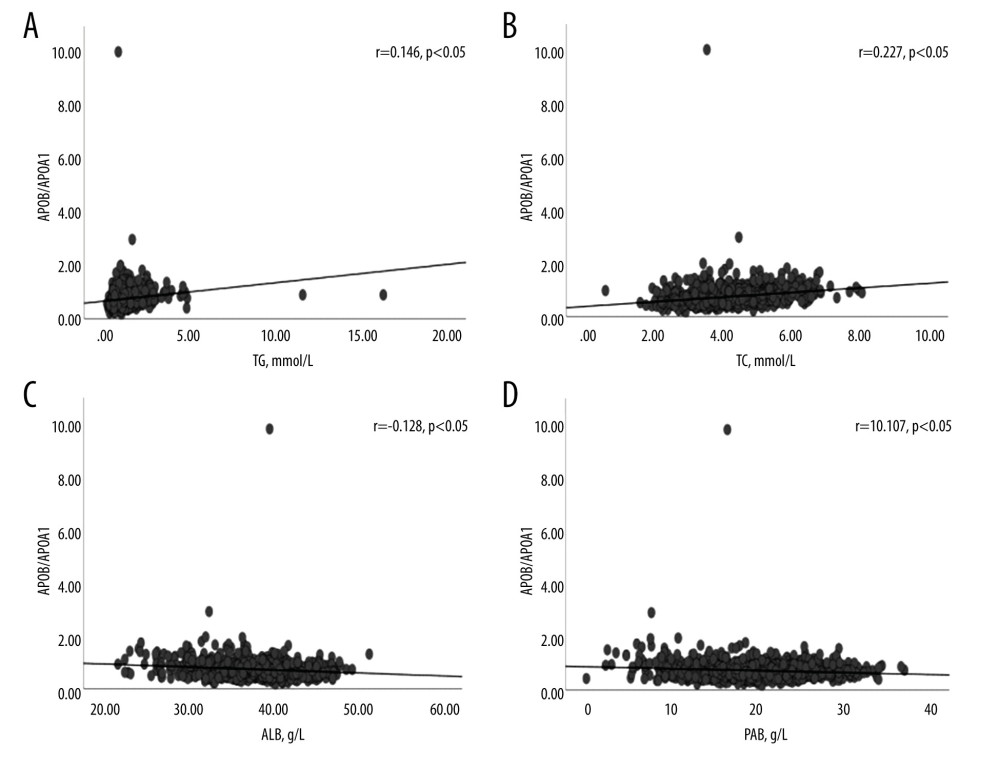
Figure 1 shows the serum APOB/APOA1 ratio and AF-related metabolic factors. Specifically, serum APOB/APOA1 ratio was positively correlated with TG (r=0.146, P<0.05; Figure 1A), TC (r=0.227, P<0.05; Figure 1B), ALB (r=−0.128, P<0.05; Figure 1C), and PAB (r=−0.107, P<0.05; Figure 1D).
SUBGROUP ANALYSIS OF THE RELATIONSHIP BETWEEN APOB/APOA1 RATIO AND METABOLIC FACTORS IN PATIENTS WITH AF:
Table 5 shows the subgroup analysis of the relationship between APOB/APOA1 ratio and metabolic factors in patients with AF. The results showed that male and female patients with lower serum APOB/APOA1 ratios also had lower TG, TC, HDL-C, and APOB had higher LDL-C, APOA1, and PAB (P<0.05). In addition, men with a lower serum APOB/APOA1 ratio had higher ALB (P<0.05), and women with a lower APOB/APOA1 ratio had lower SUA (P<0.05).
Discussion
We used a retrospective matched case-control study design to investigate the relationship between the serum APOB/APOA1 ratio and AF by sex in the Chinese population. The present results showed that a low serum APOB/APOA1 ratio in men and women was significantly related to AF. Further results indicated that serum APOB/APOA1 ratio was positively correlated with TG, TC, and APOB and was negatively correlated with ALB and PAB. These significant findings imply that a low serum APOB/APOA1 ratio may be associated with causes of AF.
Many studies have indicated that the serum APOB/APOA1 ratio is a significant marker of risk for cardiovascular events [28], but the relationship between the serum APOB/APOA1 ratio and the risk of AF has not been consistently shown. In this study, the serum ApoB/ApoA1 ratio of patients with AF was decreased due to the decrease in APOB production. Studies have shown that there are low serum levels of APOB in patients with AF. A cohort study showed that low serum APOB was a dominant factor in the occurrence of AF in men and women [29]. Nevertheless, it has been reported that the APOB/APOA1 ratio is significantly elevated in people with inflammation and atherosclerosis [30–33], which is inconsistent with our findings. According to recent studies, there are several possible reasons for this difference. First, sleep quality in the study population was a significant factor. Ren et al reported that a long sleep duration was associated with a low APOB/APOA1 ratio [34]. Second, the patients used certain drugs. Hamedi-Kalajahi et al reported that oral L-carnitine supplementation caused a decrease in the APOB/APOA1 ratio [35]. Additionally, dietary habits, physical exercise, and family history were also possible factors.
Our main finding in this study was that a low serum APOB/APOA1 ratio in men and women was associated with AF in a Chinese population. Specifically, there were several potential mechanisms that could explain this finding. Increasing recognition that inflammation and oxidative stress contribute significantly to AF has elucidated their mechanistic links [36–38]. Inflammation, in particular, is thought to be closely associated with oxidative stress, apoptosis, and fibrosis that promote the formation of AF substrates [39], thereby increasing the vulnerability to AF. There is also evidence to support the relationship between blood lipid profiles and AF, based on the mechanisms of inflammation and oxidative stress [40,41]. Serum APOA1, a key protein element of HDL, is a main initiator and promoter of cholesterol reverse transport [42,43]. It has strong anti-inflammatory and anti-oxidant properties and anti-atherogenic effects, which can promote the production of nitric oxide and the release of prostacyclin [44,45]. Research has shown that serum APOA1 exerts anti-inflammatory effects mainly by inhibiting cytokine production by monocytes/macrophages [46–48]. Previous studies also reported that serum APOA1 was reduced in acute inflammation and may induce the production of TNF-α and IL-1β [46,48]. Serum APOB, the LDL-matching protein [49], has also been shown to be associated with inflammation. Faraj et al reported that serum APOB is a strongly correlated predictor of inflammatory markers, such as interleukin-6 and CRP, and results from further studies suggested that reduced serum APOB may contribute to inflammation [26,50]. Additionally, the relationship between atherosclerosis and inflammation has been increasingly recognized [51,52]. It can be said that atherosclerosis has been included in the category of chronic inflammatory diseases [53]. The serum APOB/APOA1 ratio has been shown to be an effective indicator to evaluate the balance between pro-atherogenic LDL particles and anti-atherogenic HDL particles [54]. Generally speaking, the higher the serum APOB/APOA1 ratio is, the more likely it is that atherosclerosis will occur [55]. However, our results suggested that a lower APOB/APOA1 ratio was more likely to contribute to inflammation, thereby increasing the likelihood of AF. We hypothesized that the reduction of HDL-C accompanied by the loss of anti-inflammatory, anti-oxidant, and anti-atherosclerosis effects was a possible factor in the increase of AF matrix formation [56–58]. Certainly, other potential confounding factors, such as lifestyle, regional environment, and aging, may also influence the results. Further studies should be conducted to explore these potential correlations and mechanisms.
In addition, the relationship between the serum APOB/APOA1 ratio and metabolic syndrome by sex has been confirmed. A study showed that there was a positive linear correlation between the number of metabolic syndrome components and the APOB/APOA1 ratio in men [59]. Another study reported that the serum APOB/APOA1 ratio was also correlated with metabolic syndrome and its components in women, and further suggested that this relationship was independent of patient sex [60]. Our present results were consistent with those of earlier studies and confirmed the relationship between the serum APOB/APOA1 ratio and metabolic factors in patients with AF. Specifically, the serum APOB/APOA1 ratio was positively correlated with TG, TC, HDL-C, and APOB and was negatively correlated with LDL-C, APOA1, ALB, and PAB. Also, we further investigated the sex distribution of this association. We found that patients with a lower serum APOB/APOA1 ratio had lower TG, TC, HDL-C, and APOB and higher LDL-C, APOA1, and PAB in both sexes, higher ALB in men, and lower SUA in women. These findings may help explain the relationship between APOB/APOA1 and AF-related metabolic factors, and it is essential to further study its internal mechanism.
This study had some limitations as a retrospective study from a single center, using data from patient medical records, which relied on accurate data input. First, the retrospective case-control study design cannot determine causality. Second, a smaller sample size, single-center design, and higher age distribution may have had a certain impact on our results. Third, this study did not involve clinical data on persistent AF; therefore, our results are not applicable to all patients with AF. Fourth, several potential confounding factors may also have been ignored, such as the markers of inflammation and oxidative stress. Finally, previous researchers have stated that patients should be evaluated for stenosis, prognosis, and ischemic heart disease; we did not evaluate these variables [61,62]. Nevertheless, this study did provide a new perspective on the pathology of AF. Further studies are encouraged to systematically assess the association between the serum APOB/APOA1 ratio and AF, as well as AF-related factors.
Conclusions
In conclusion, our findings show that a low serum APOB/APOA1 ratio in men and women from China was significantly related to AF. This finding suggests that a low serum APOB/APOA1 ratio may be related to the causes of AF. We recommend future prospective cohort investigations to confirm these results.
Tables
Table 1. Clinical characteristics.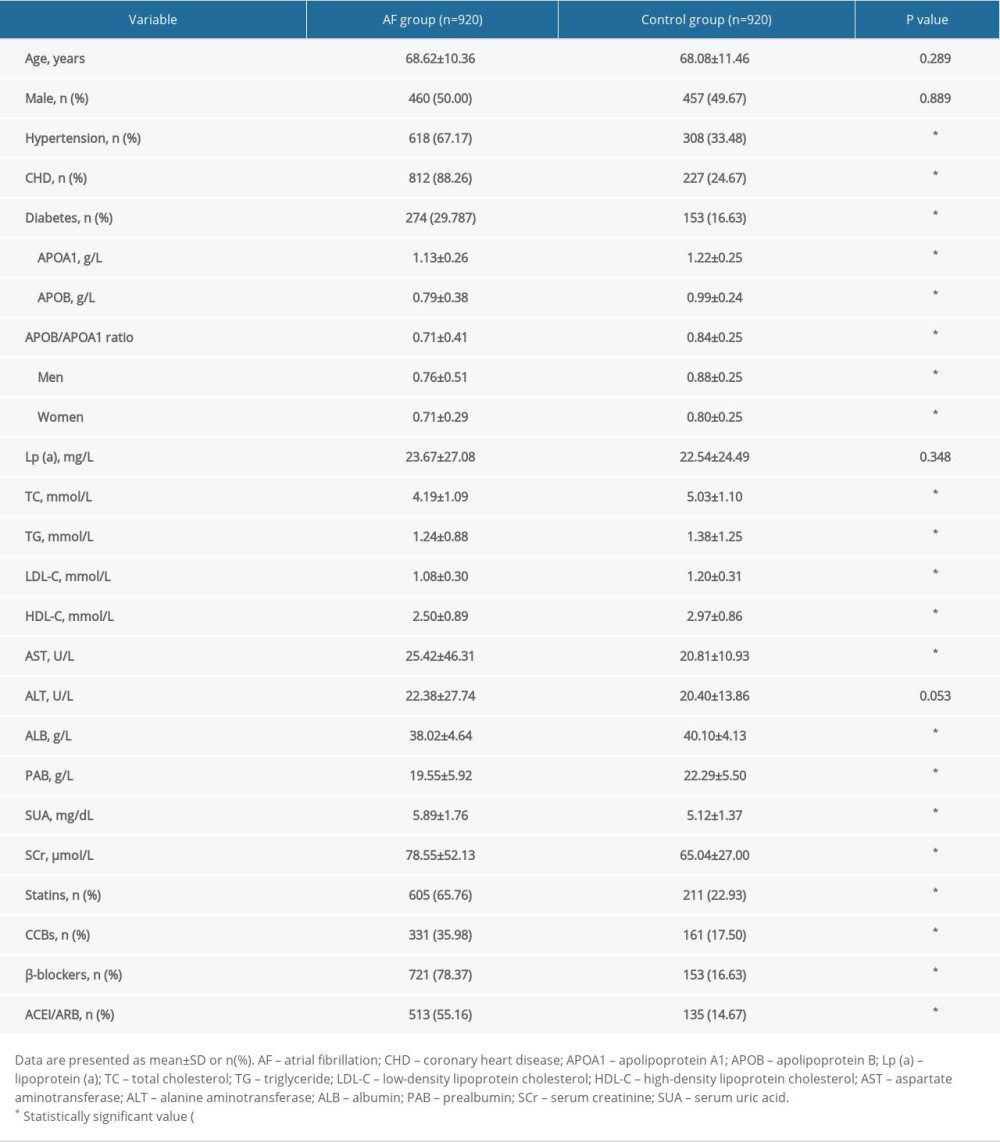 Table 2. Correlation between serum APOB/APOA1 ratio and atrial fibrillation.
Table 2. Correlation between serum APOB/APOA1 ratio and atrial fibrillation.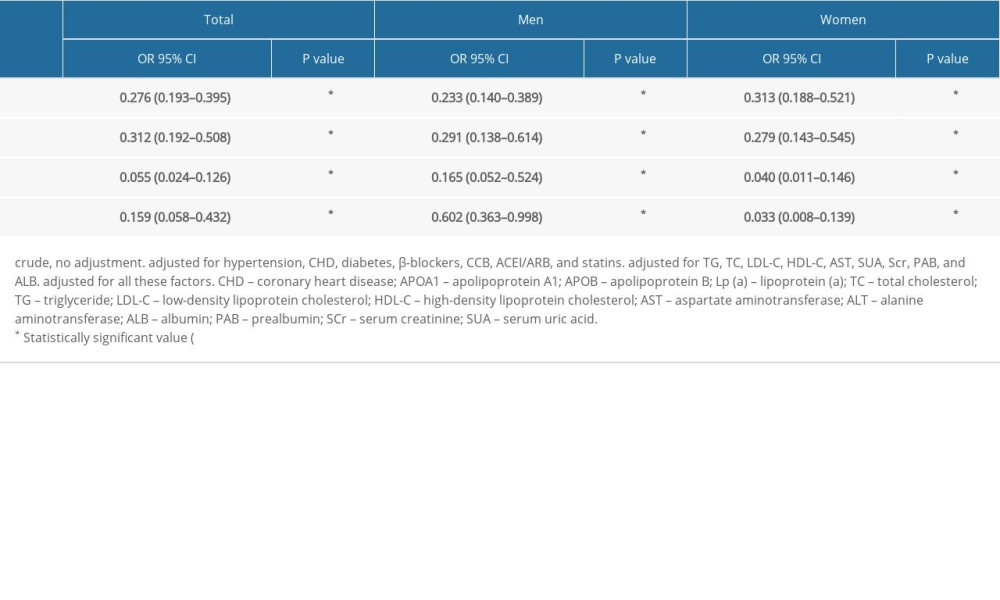 Table 3. Differences of serum APOB/APOA1 ratio in male and female patients with atrial fibrillation.
Table 3. Differences of serum APOB/APOA1 ratio in male and female patients with atrial fibrillation.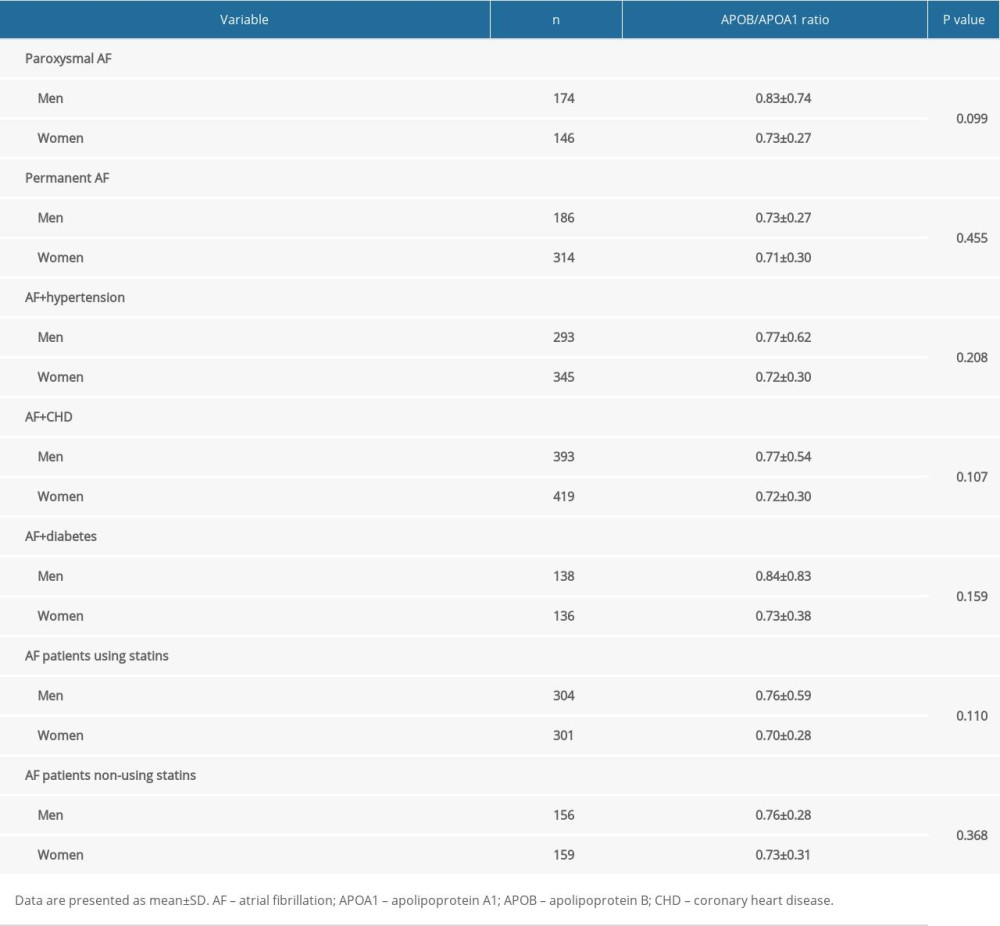 Table 4. Differences in serum APOB/APOA1 ratio by different subtypes, complications, and statins use in patients with atrial fibrillation.
Table 4. Differences in serum APOB/APOA1 ratio by different subtypes, complications, and statins use in patients with atrial fibrillation.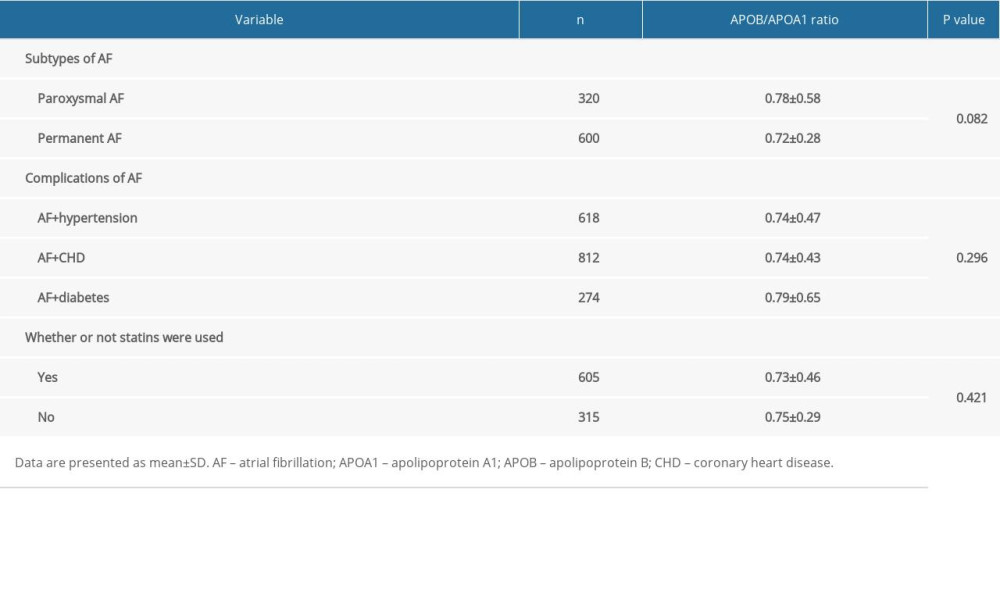 Table 5. Subgroup analysis of the relationship between APOB/APOA1 ratio and metabolic factors in patients with atrial fibrillation.
Table 5. Subgroup analysis of the relationship between APOB/APOA1 ratio and metabolic factors in patients with atrial fibrillation.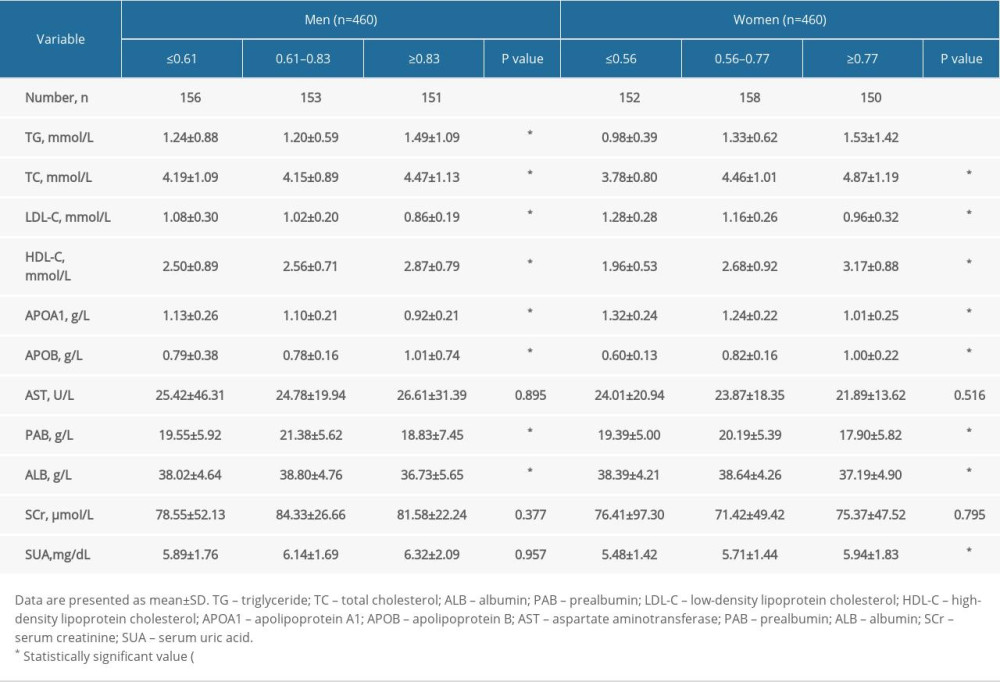
References
1. Zimetbaum P, Atrial Fibrillation [published correction appears in Ann Intern Med. 2017;166(12):920]: Ann Intern Med, 2017; 166(5); ITC33-48
2. Waldmann V, Jouven X, Narayanan K, Association between atrial fibrillation and sudden cardiac death: Pathophysiological and epidemiological insights: Circ Res, 2020; 127(2); 301-9
3. Chugh SS, Havmoeller R, Narayanan K, Worldwide epidemiology of atrial fibrillation: A Global Burden of Disease 2010 Study: Circulation, 2014; 129(8); 837-47
4. Santhanakrishnan R, Wang N, Larson MG, Atrial fibrillation begets heart failure and vice versa: Temporal associations and differences in preserved versus reduced ejection fraction: Circulation, 2016; 133(5); 484-92
5. Healey JS, Amit G, Field TS, Atrial fibrillation and stroke: How much atrial fibrillation is enough to cause a stroke?: Curr Opin Neurol, 2020; 33(1); 17-23
6. Diener HC, Hart RG, Koudstaal PJ, Atrial fibrillation and cognitive function: JACC review topic of the week: J Am Coll Cardiol, 2019; 73(5); 612-19
7. Berg DD, Ruff CT, Jarolim P, Performance of the ABC scores for assessing the risk of stroke or systemic embolism and bleeding in patients with atrial fibrillation in ENGAGE AF-TIMI 48: Circulation, 2019; 139(6); 760-71
8. Lopes RD, Rordorf R, De Ferrari GM, Digoxin and mortality in patients with atrial fibrillation: J Am Coll Cardiol, 2018; 71(10); 1063-74
9. Wong CX, Brooks AG, Leong DP, The increasing burden of atrial fibrillation compared with heart failure and myocardial infarction: A 15-year study of all hospitalizations in Australia: Arch Intern Med, 2012; 172(9); 739-41
10. Baman JR, Passman RS, Atrial fibrillation: JAMA, 2021; 325(21); 2218
11. January CT, Wann LS, Alpert JS, 2014 AHA/ACC/HRS guideline for the management of patients with atrial fibrillation: executive summary: A report of the American College of Cardiology/American Heart Association Task Force on practice guidelines and the Heart Rhythm Society: Circulation, 2014; 130; 2071-104
12. Gupta A, Perera T, Ganesan A, Complications of catheter ablation of atrial fibrillation: A systematic review: Circ Arrhythm Electrophysiol, 2013; 6; 1082-88
13. Link MS, Haïssaguerre M, Natale A, Ablation of atrial fibrillation: Patient selection, periprocedural anticoagulation, techniques, and preventive measures after ablation: Circulation, 2016; 134(4); 339-52
14. Verma A, Jiang CY, Betts TR, Approaches to catheter ablation for persistent atrial fibrillation: N Engl J Med, 2015; 372(19); 1812-22
15. Lau DH, Nattel S, Kalman JM, Sanders P, Modifiable risk factors and atrial fibrillation: Circulation, 2017; 136(6); 583-96
16. Grundy SM, Stone NJ, Bailey AL, 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA guideline on the management of blood cholesterol: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines [published correction appears in Circulation. 2019;139(25): e1182–86]: Circulation, 2019; 139(25); e1082-143
17. Chistiakov DA, Orekhov AN, Bobryshev YV, ApoA1 and ApoA1-specific self-antibodies in cardiovascular disease: Lab Invest, 2016; 96(7); 708-18
18. Benn M, Apolipoprotein B levels, APOB alleles, and risk of ischemic cardiovascular disease in the general population, a review: Atherosclerosis, 2009; 206(1); 17-30
19. Valanti EK, Dalakoura-Karagkouni K, Sanoudou D, Current and emerging reconstituted HDL-apoA-I and HDL-apoE approaches to treat atherosclerosis: J Pers Med, 2018; 8(4); 34
20. Lu X, Chen D, Endresz V, Immunization with a combination of ApoB and HSP60 epitopes significantly reduces early atherosclerotic lesion in Apobtm2SgyLdlrtm1Her/J mice: Atherosclerosis, 2010; 212(2); 472-80
21. Walldius G, Jungner I, The apoB/apoA-I ratio: A strong, new risk factor for cardiovascular disease and a target for lipid-lowering therapy – a review of the evidence: J Intern Med, 2006; 259; 493519
22. Walldius G, Jungner I, Holme I, High apolipoprotein B, low apolipoprotein A-I, and improvement in the prediction of fatal myocardial infarction (AMORIS study): A prospective study: Lancet, 2001; 358(9298); 2026-33
23. Tousoulis D, Biomarkers in atrial fibrillation; from pathophysiology to diagnosis and treatment: Curr Med Chem, 2019; 26(5); 762-64
24. Jiang Q, Wang K, Shi J, No association between alcohol consumption and risk of atrial fibrillation: A two-sample Mendelian randomization study: Nutr Metab Cardiovasc Dis, 2020; 30(8); 1389-96
25. Trieb M, Kornej J, Knuplez E, Atrial fibrillation is associated with alterations in HDL function, metabolism, and particle number: Basic Res Cardiol, 2019; 114(4); 27
26. Faraj M, Lavoie ME, Messier L, Reduction in serum apoB is associated with reduced inflammation and insulin resistance in post-menopausal women: A MONET study: Atherosclerosis, 2010; 211(2); 682-88
27. Li JY, He Y, Ke HH, Plasma oxidative stress and inflammatory biomarkers are associated with the sizes of the left atrium and pulmonary vein in atrial fibrillation patients: Clin Cardiol, 2017; 40(2); 89-94
28. Sierra-Johnson J, Fisher RM, Romero-Corral A, Concentration of apolipoprotein B is comparable with the apolipoprotein B/apolipoprotein A-I ratio and better than routine clinical lipid measurements in predicting coronary heart disease mortality: Findings from a multi-ethnic US population: Eur Heart J, 2009; 30(6); 710-17
29. Şimşek B, Altay S, Özbilgin N, Onat A, Autoimmune activation as a determinant of atrial fibrillation among Turks: A prospective evaluation: Medicine (Baltimore), 2018; 97(31); e11779
30. Xu W, Li R, Zhang S, The relationship between high-sensitivity C-reactive protein and ApoB, ApoB/ApoA1 ratio in general population of China: Endocrine, 2012; 42(1); 132-38
31. Wang B, Deng H, Hu Y, The difference of lipid profiles between psoriasis with arthritis and psoriasis without arthritis and sex-specific downregulation of methotrexate on the apolipoprotein B/apolipoprotein A-1 ratio: Arthritis Res Ther, 2022; 24(1); 17
32. Song X, Tian SP, Ju HY, Predictive value of apolipoprotein for coronary atherosclerosis in asymptomatic non-diabetic population: Zhongguo Yi Xue Ke Xue Yuan Xue Bao, 2015; 37(1); 55-60
33. Kaneva AM, Potolitsyna NN, Bojko ER, Odland JØ, The apolipoprotein B/apolipoprotein A-I ratio as a potential marker of plasma atherogenicity: Dis Markers, 2015; 2015; 591454
34. Ren H, Liu Z, Zhou X, Yuan G, Association of sleep duration with apolipoproteins and the apolipoprotein B/A1 ratio: The China health and nutrition survey: Nutr Metab (Lond), 2018; 15; 1
35. Hamedi-Kalajahi F, Zarezadeh M, Mojtahedi SY, Effect of L-carnitine supplementation on lipid profile and apolipoproteins in children on hemodialysis: A randomized placebo-controlled clinical trial: Pediatr Nephrol, 2021; 36(11); 3741-47
36. Harada M, Van Wagoner DR, Nattel S, Role of inflammation in atrial fibrillation pathophysiology and management: Circ J, 2015; 79(3); 495-502
37. Gutierrez A, Van Wagoner DR, Oxidant and inflammatory mechanisms and targeted therapy in atrial fibrillation: An update: J Cardiovasc Pharmacol, 2015; 66(6); 523-29
38. da Silva RM, Influence of inflammation and atherosclerosis in atrial fibrillation: Curr Atheroscler Rep, 2017; 19(1); 2
39. Harada M, Van Wagoner DR, Nattel S, Role of inflammation in atrial fibrillation pathophysiology and management: Circ J, 2015; 79(3); 495-502
40. Guan B, Li X, Xue W, Blood lipid profiles and risk of atrial fibrillation: A systematic review and meta-analysis of cohort studies: J Clin Lipidol, 2020; 14(1); 133-142e3
41. Allan V, Honarbakhsh S, Casas J, Are cardiovascular risk factors also associated with the incidence of atrial fibrillation? A systematic review and field synopsis of 23 factors in 32 population-based cohorts of 20 million participants: Thromb Haemost, 2017; 117(5); 837-50
42. Florvall G, Basu S, Larsson A, Apolipoprotein A1 is a stronger prognostic marker than are HDL and LDL cholesterol for cardiovascular disease and mortality in elderly men: J Gerontol A Biol Sci Med Sci, 2006; 61(12); 1262-66
43. Ajees AA, Anantharamaiah GM, Mishra VK, Crystal structure of human apolipoprotein A-I: insights into its protective effect against cardiovascular diseases [retracted in: Proc Natl Acad Sci USA. 2018;115(29): E6966]: Proc Natl Acad Sci USA, 2006; 103(7); 2126-31
44. Lu M, Lu Q, Zhang Y, Tian G, ApoB/apoA1 is an effective predictor of coronary heart disease risk in overweight and obesity: J Biomed Res, 2011; 25(4); 266-73
45. Van Linthout S, Spillmann F, Riad A, Human apolipoprotein A-I gene transfer reduces the development of experimental diabetic cardiomyopathy: Circulation, 2008; 117(12); 1563-73
46. Öhman M, Öhman ML, Wållberg-Jonsson S, The apoB/apoA1 ratio predicts future cardiovascular events in patients with rheumatoid arthritis: Scand J Rheumatol, 2014; 43(4); 259-64
47. Burger D, Dayer JM, High-density lipoprotein-associated apolipoprotein A-I: The missing link between infection and chronic inflammation?: Autoimmun Rev, 2002; 1(1–2); 111-17
48. Hyka N, Dayer JM, Modoux C, Apolipoprotein A-I inhibits the production of interleukin-1beta and tumor necrosis factor-alpha by blocking contact-mediated activation of monocytes by T lymphocytes: Blood, 2001; 97(8); 2381-89
49. Elovson J, Chatterton JE, Bell GT, Plasma very low density lipoproteins contain a single molecule of apolipoprotein B: J Lipid Res, 1988; 29(11); 1461-73
50. Faraj M, Messier L, Bastard JP, Apolipoprotein B: A predictor of inflammatory status in postmenopausal overweight and obese women: Diabetologia, 2006; 49(7); 1637-46
51. Zhu Y, Xian X, Wang Z, Research progress on the relationship between atherosclerosis and inflammation: Biomolecules, 2018; 8(3); 80
52. Geovanini GR, Libby P, Atherosclerosis and inflammation: overview and updates: Clin Sci (Lond), 2018; 132(12); 1243-52
53. Wolf D, Ley K, Immunity and inflammation in atherosclerosis: Circ Res, 2019; 124(2); 315-27
54. Walldius G, Jungner I, Aastveit AH, The apoB/apoA-I ratio is better than the cholesterol ratios to estimate the balance between plasma proatherogenic and antiatherogenic lipoproteins and to predict coronary risk: Clin Chem Lab Med, 2004; 42(12); 1355-63
55. Holme I, Aastveit AH, Hammar N, Inflammatory markers, lipoprotein components and risk of major cardiovascular events in 65,005 men and women in the Apolipoprotein MOrtality RISk study (AMORIS): Atherosclerosis, 2010; 213(1); 299-305
56. Guo Y, Lip GY, Apostolakis S, Inflammation in atrial fibrillation: J Am Coll Cardiol, 2012; 60(22); 2263-70
57. Yang KC, Dudley SC, Oxidative stress and atrial fibrillation: Finding a missing piece to the puzzle: Circulation, 2013; 128(16); 1724-26
58. Korantzopoulos P, Letsas KP, Tse G, Inflammation and atrial fibrillation: A comprehensive review: J Arrhythm, 2018; 34(4); 394-401
59. Wallenfeldt K, Bokemark L, Wikstrand J, Apolipoprotein B/apolipoprotein A-I in relation to the metabolic syndrome and change in carotid artery intima-media thickness during 3 years in middle-aged men [published correction appears in Stroke. 2005;36(2): 415]: Stroke, 2004; 35(10); 2248-52
60. Zhong L, Li Q, Jiang Y, The ApoB/ApoA1 ratio is associated with metabolic syndrome and its components in a Chinese population: Inflammation, 2010; 33(6); 353-58
61. Ivert T, Hammar N, Talbäck M, Elevated apolipoprotein B/A-1 ratio is associated with an increased risk of aortic stenosis: Experience from the AMORIS cohort: Heart Lung Circ, 2021; 30(7); 1050-57
62. Walldius G, Jungner I, The apoB/apoA-I ratio: A strong, new risk factor for cardiovascular disease and a target for lipid-lowering therapy – a review of the evidence: J Intern Med, 2006; 259(5); 493-519
Tables
 Table 1. Clinical characteristics.
Table 1. Clinical characteristics. Table 2. Correlation between serum APOB/APOA1 ratio and atrial fibrillation.
Table 2. Correlation between serum APOB/APOA1 ratio and atrial fibrillation. Table 3. Differences of serum APOB/APOA1 ratio in male and female patients with atrial fibrillation.
Table 3. Differences of serum APOB/APOA1 ratio in male and female patients with atrial fibrillation. Table 4. Differences in serum APOB/APOA1 ratio by different subtypes, complications, and statins use in patients with atrial fibrillation.
Table 4. Differences in serum APOB/APOA1 ratio by different subtypes, complications, and statins use in patients with atrial fibrillation. Table 5. Subgroup analysis of the relationship between APOB/APOA1 ratio and metabolic factors in patients with atrial fibrillation.
Table 5. Subgroup analysis of the relationship between APOB/APOA1 ratio and metabolic factors in patients with atrial fibrillation. Table 1. Clinical characteristics.
Table 1. Clinical characteristics. Table 2. Correlation between serum APOB/APOA1 ratio and atrial fibrillation.
Table 2. Correlation between serum APOB/APOA1 ratio and atrial fibrillation. Table 3. Differences of serum APOB/APOA1 ratio in male and female patients with atrial fibrillation.
Table 3. Differences of serum APOB/APOA1 ratio in male and female patients with atrial fibrillation. Table 4. Differences in serum APOB/APOA1 ratio by different subtypes, complications, and statins use in patients with atrial fibrillation.
Table 4. Differences in serum APOB/APOA1 ratio by different subtypes, complications, and statins use in patients with atrial fibrillation. Table 5. Subgroup analysis of the relationship between APOB/APOA1 ratio and metabolic factors in patients with atrial fibrillation.
Table 5. Subgroup analysis of the relationship between APOB/APOA1 ratio and metabolic factors in patients with atrial fibrillation. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387