02 December 2022: Review Articles
Narrative Review of Noninvasive Brain Stimulation in Stroke Rehabilitation
Qian-ru Shen1AEF, Meng-ting Hu1EF, Wei Feng1A, Kun-Peng Li1AEFG*, Wu WangDOI: 10.12659/MSM.938298
Med Sci Monit 2022; 28:e938298
Abstract
ABSTRACT: Stroke is a disease with a high incidence and disability rate, resulting in changes in neural network and corticoid-subcortical excitability and various functional disabilities. The aim of the present study was to discuss the current status of research and limitations and potential direction in the application of noninvasive brain stimulation (NIBS) on post-stroke patients. This literature review focused on clinical studies and reviews. Literature retrieval was conducted in PubMed, Cochrane, Scopus, and CNKI, using the following keywords: Repeated transcranial magnetic stimulation, Transcranial direct current stimulation, Transcranial alternating current stimulation, Transcranial alternating current stimulation, Transcranial focused ultrasound, Noninvasive vagus nerve stimulation, Stroke, and Rehabilitation. We selected 200 relevant publications from 1985 to 2022. An overview of recent research on the use of NIBS on post-stroke patients, including its mechanism, therapeutic parameters, effects, and safety, is presented. It was found that NIBS has positive therapeutic effects on dysfunctions of motor, sensory, cognitive, speech, swallowing, and depression after stroke, but standardized stimulus programs are still lacking. The literature suggests that rTMS and tDCS are more beneficial to post-stroke patients, while tFUS and tVNS are currently less studied for post-stroke rehabilitation, but are also potential interventions.
Keywords: Extracorporeal Shockwave Therapy, stroke rehabilitation, Transcranial Direct Current Stimulation, Transcranial Magnetic Stimulation, vagus nerve stimulation, Humans, Stroke
Background
Stroke, a common neurological disorder, occurs worldwide and has high mortality and disability rates [1,2]. Mortality rates have steadily declined due to recent advances in acute treatment and in primary and secondary prevention [3]. The irreversible brain damage caused by stroke leaves survivors with a permanent neurological deficit, causing serious dysfunction and affecting quality of life [2]. This causes a high demand for rehabilitation of stroke survivors.
Under normal circumstances, there is a functional balance between the 2 hemispheres of the brain, regulated by interhemispheric inhibition [4]. This equilibrium is affected after the stroke, with enhanced excitability in the contra-lesional hemisphere, while an abnormally increased interhemispheric inhibition occurs in the affected hemisphere [5,6]. Thus, it is of great importance to modify cortical excitability and adjust the balance between hemispheres for the rehabilitation of post-stroke patients [7]. In consequence, a potential strategy for rehabilitation is to modulate the plasticity by NIBS, including repeated transcranial magnetic stimulation (rTMS), transcranial direct current stimulation (tDCS), transcranial alternating current stimulation (tACS), transcranial focused ultrasound (tFUS), and noninvasive vagus nerve stimulation (nVNS), seeking to restore the normal activity pattern [8–12]. Therefore, a detailed overview of the literature summarizing the current state of NIBS research is essential and valuable. This article presents a narrative review of previous research on NIBS in the treatment of post-stroke patients, including the mechanism, therapeutic parameters, effect, and safety.
Material and Methods
The following electronic databases were searched: PubMed, Web of Science, Elsevier, The Cochrane Library, and CNKI. The search strategy was developed using the following subject heading associating with stroke: Ischemic stroke, Hemorrhagic stroke, Repeated transcranial magnetic stimulation (rTMS), Transcranial direct current stimulation (tCDS), Transcranial alternating current stimulation (tACS), Transcranial focused ultrasound (tFUS), and Noninvasive vagus nerve stimulation (nVNS). We included articles on the rehabilitation of stroke patients using NIBS and related literature on NIBS mechanism, while those not related to the research topic were excluded. In addition, the references of the selected literature were also considered.
Results
NIBS for Post-Stroke Patients
UTILIZATION OF RTMS: In the late 20th century, Barker et al [13] discovered that it was possible to use TMS to stimulate both nerves and the brain. Since then, TMS has been widely used clinically. TMS, a noninvasive form of neurostimulation, uses electromagnetic induction to generate an electrical current in the brain and affects the electrophysiological activity of nerves [14]. A sufficiently strong induced current causes the neurons to depolarize, creating an action potential induced by TMS [15]. rTMS is a series of pulses that can alter and modulate cortical activity after the stimulation period, which has shown promising therapeutic potential in multiple neurological conditions [16]. In addition, rTMS can significantly reduce blood–brain barrier (BBB) permeability, improve vascular structure and morphology, and regulate cerebral perfusion [17]. Angiogenesis can be promoted and apoptosis of vascular endothelial cells can be reduced by rTMS [18].
Cortical excitability can be modulated by rTMS in a frequency-dependent manner. Low-frequency rTMS (LF-rTMS, ≤1 Hz) decreased cortical excitability, while high-frequency rTMS (HF-rTMS, >1 Hz) increased it [19–22]. Thus, rTMS can be used in both the affected and unaffected hemispheres [7,23]. Clinically, LF-rTMS was applied to the contralateral hemisphere to inhibit it, while HF-rTMS was used to stimulate the affected hemisphere to increase cortical excitability, thereby correcting excessive mutual inhibition between cerebral hemispheres after stroke and improving the function of patients [24–27]. 1 Hz LF-rTMS and 10 Hz HF-rTMS are mostly used clinically, and both have been proven to be safe and well tolerated [28]. Various protocols were utilized in clinical trials, as shown in Table 1.
Regarding targeted regions, different dysfunctions correspond to different brain regions. The most common site of stimulation was the primary motor cortex (M1), which was used to treat motor dysfunction, spasm, and dysphagia [22,25,27]. rTMS has been applied to the contralateral dorsolateral prefrontal cortex (DLPFC) for post-stroke depression (PSD) and cognitive dysfunction [29]. For some post-stroke unilateral neglect patients, the parietal cortex was generally stimulated [30]. Stimulation of the Broca area was used to treat motor aphasia [31,32], and stimulation of the Wernicke area was applied for sensory aphasia [33]. However, large-scale alterations at the network level emerge after the plastic effects of rTMS, which contribute to local and long-term changes in the stimulated network and interactions between the stimulated and other functional networks [34,35].
UTILIZATION OF TDCS AND TACS: Appropriately 20 years ago, as a noninvasive modulation of motor cortex excitability, tDCS was applied in humans [36]. tDCS generates a constant and weak direct electrical current (1–2 mA) by 2 or more electrodes placed on the scalp, which promotes neuroplasticity and modulates cortical excitability via subthreshold alternation of neuronal membrane potentials [37,38]. Moreover, Nitsche et al [36] found that cortical excitability and activity altered by tDCS depends on the current flow direction through the target neurons. The anode depolarizes the resting potential of the cell membrane and increases excitability of the cortex, while the cathode hyper-polarizes the resting potential and inhibits excitability of the cortex [36,39]. In addition, its effect is not limited to the stimulated region, but also involves nearby brain regions, which can change the functional connections between large brain regions [40]. tDCS has also been shown to improve local cerebral blood flow, which helps alleviate inflammation and protect neurons in ischemic areas [41].
The treatment parameters and duration determine the effect of tDCS. Wen et al [42] found that tDCS had intensity and time dependence in dose effect, which means high-intensity and long-duration stimulation has a better effect. However, this is not a general rule. Increased intensity of tDCS does not necessarily increase the efficacy of the stimulus, but may also shift the direction of excitatory changes [43]. Moreover, Jamil et al [44] demonstrated that the post-excitability effect was not linearly correlated with the increase in direct current intensity. Most relevant studies used 1, 1.5, and 2mA, and the treatment duration was mostly 20 min, as shown in Table 2. The area stimulated is important; benefits have been shown using anodic tDCS of the ipsilateral primary motor cortex (M1) and cathodic tDCS of the contralateral M1, as well as bi-hemispheric stimulation for the rehabilitation of dyskinesia [11,45]. Studies [46] have shown that placing the anode on the DLPFC of the affected side and the cathode on the superior orbital margin of the contralateral side can effectively reduce fatigue after stroke. In addition, stimulation of the Broca and the Wernicke regions has been used in the treatment of aphasia after stroke, but its effectiveness remains uncertain [47,48].
Another transcutaneous electrical stimulation is tACS, with more than 10 years of application history. tACS affects cortical neurons by applying sinusoidal communication on the scalp, thus regulating brain oscillations to reshape internal brain rhythms and improve relevant brain functions [49]. As distinct from TMS, tDCS does not directly induce brain activity leading to a large-scale synchronous discharge of action potential, but rather alters the membrane potential and discharge threshold of neurons [50–52]. On the entrainment of endogenous neuronal oscillations, cortical excitability and activity-dependent are altered. By synchronizing the neural network, tACS alters the transmembrane potential, modulates the discharge frequency of neurons, and changes the oscillating rhythm of the brain [53,54]. Thus, tACS can specifically modulate oscillatory brain activity and selectively enhance oscillations at the applied stimulus frequency [55,56].
Stimulation conditions for tACS varied according to the frequency, site, and duration, as shown in Table 3. tACS at 70 Hz has been confirmed to temporarily enhance motor function, but whether it leads to long-term consolidation of motor learning remains to be studied [57]. It was previously demonstrated that 10 Hz and 20 Hz tACS improve motor sequence learning [58], but some studies have found that tACS has no effect on motor learning and can even be detrimental to motor consolidation [59]. The stimulation areas are also changed according to the lesion. Applying tACS to the primary motor cortex [58], the prefrontal cortex [60,61] and cerebellar cortex [62] can improve motor learning. Stimulation of the prefrontal cortex affects depression [63] and vision [64], and the parietal cortex is correlated with cognitive ability [65]. Wu et al [66] found that tACS over bilateral mastoids appears to improve functional recovery and cerebral hemodynamics in patients in the subacute phase of stroke. However, another study [67] of tACS stimulation for patients with hemianopia after chronic stroke found no significant improvement. There are few clinical studies [68] on tACS for post-stroke treatment, and new studies are needed.
UTILIZATION OF TFUS: While the modulation effects of rTMS, tDCS, and tACS are confined to the cortical surface, tFUS transmits ultrasound into deep brain regions [69,70]. tFUS can directly affect the ability of cells to discharge [71], and selectively stimulate and inhibit brain activity, thus modulating the excitability of neural tissues [72].
The commonly used tFUS includes high-intensity tFUS and low-intensity tFUS. High-intensity tFUS causes tissue damage through its thermal effects, thus blocking synaptic transmission and inhibiting the electrical activity of neurons [73,74]. However, mechanical or thermal damage to tissue is likely to occur [75]. As a result, high-intensity tFUS is rarely used clinically for neuromodulating effects. The amplitudes and conduction velocities of evoked nerve potentials are enhanced or suppressed by low-intensity tFUS through force effect and cavitation effect, thereby modulating neuronal activity [76–78]. Additionally, tFUS can improve blood supply around brain lesion tissues [12].
The effect of tFUS depends on 5 main parameters: frequency, peak intensity, duration, pulse repetition frequency, and duty cycle. Deng et al [79] treated mice with low-intensity low-frequency (0.5 MHz) tFUS with Isppa of 39 mW/cm2 for 10 min and found that it could reduce vasogenic edema after middle cerebral artery occlusion, as well as improve neural behavior and blood–brain barrier integrity. Wang et al [80] used tFUS with 120 mW/cm2 and 1000 Hz for 10 min for 7 consecutive days in mice and found that tFUS can promote microglia polarization, reduce the inflammatory response, and improve neuron repair and remodeling, thus promoting post-ischemic stroke recovery. Nevertheless, as shown in Table 4, few studies have been done on tFUS for stroke, and more studies are needed.
UTILIZATION OF TVNS: Unlike other noninvasive techniques, tVNS does not stimulate the brain. It delivers electrical stimulation to the brain through the vagus nerve, causing changes in brain electrical activity and neurotransmitters, thus modulating the functional activity of neurons [81]. tVNS includes transcutaneous auricular VNS (taVNS) and transcutaneous cervical VNS (tcVNS) [82]. taVNS stimulates the auricular branch of the vagus nerve by surface current electricity, and the best anatomical site for stimulation is the auricular plate [83]. tcVNS represents the stimulation of the vagus nerve in the cervical pulse sheath, which is usually applied on the anterolateral surface of the vagus nerve and can activate it through the skin and other biological barriers [84].
The effect of tVNS on cortical excitability was first discovered by Capone et al [85], who found that paired-pulse stimulation selectively and significantly increased intracortical inhibition. Moreover, another study [86] found that taVNS stimulation led to significant increases in neural activity in the right caudate nucleus, left prefrontal cortex, middle cingulate gyrus, and cerebellum. In addition, the mechanism of tVNS in stroke rehabilitation is also under investigation. The protective effect of tVNS on the blood–brain barrier has been proved in cerebral ischemia rats. It is achieved by reducing matrix metalloproteinase-mediated tight junction protein destruction [87]. Subsequently, other animal experiments were carried out, showing that tVNS can reduce cortical infarct volume [88], inhibit neuroinflammation, and relieve nerve injury [89], thus improving neural function.
The stimulation parameters of tVNS have a great influence on clinical efficacy, as summarized in Table 5. The stimulus current is usually set based on the subject’s sensitivity or pain threshold. The amplitude of the stimulus varied over a wide range due to the varying tolerance of different participants to the stimulus. Studies have shown that the vagus nerve could receive remarkable irreversible damage when applying VNS greater than or equal to 50 Hz [90]. Recent clinical studies have used stimulation frequencies between 20 and 30 Hz [91–96].
CLINICAL APPLICATIONS OF NIBS IN MOTOR RECOVERY AFTER STROKE: Improvement in motor function for post-stroke patients with rTMS has been confirmed in many clinical trials. In 2012, LF-rTMS was demonstrated to improve upper limb mobility and strength in chronic stroke patients [97]. Sasaki et al [98] found that rTMS also improves rehabilitation of upper extremity motor function for apoplexy patients at an early stage, and HF-rTMS was better than LF-rTMS. A year later, they [99] discovered that stimulation with bilateral rTMS was more effective in restoring upper limb function than stimulation with unilateral high-frequency rTMS. Recently, Wang et al [100] discovered that motor function improvement of the upper limb by high- and low-frequency rTMS was influenced by the integrity of the corticospinal tract in patients, but high-quality experiments are still needed to confirm this. Simultaneously, rTMS has been shown to promote the rehabilitation of lower limb motor function in stroke patients [26,101]. It was interesting to find that patients with acute stroke sustained motor function recovery for 1 month and upper limb function improvement for 1 year after 10 days of rTMS [27]. The impact of rTMS on improving walking ability after stroke has been convincing. Stroke patient’ gait and walking speed can be improved by rTMS [102]. rTMS can also improve hand function, including hand coordination, speed, grip strength, and fine movement, which has been shown in patients with acute, subacute, and chronic stroke [27,103–106].
tDCS is also commonly used in post-stroke patients with hemiplegia. Approximately 20 years ago, clinical trials of tDCS for motor rehabilitation after hemiplegia began [107,108]. The favorable effects of rTMS on recovery of upper extremity movement, flexibility, and hand grip strength in stroke patients have been demonstrated in acute, subacute, and chronic patients [11,109,110]. Moreover, some studies have found that the effect of bi-hemispheric tDCS is better than unilateral anodic tDCS in improving motor function [11,109]. For lower limb function and walking ability, rTMS also showed positive effects [111–113]. However, Aneksan [114] found that tDCS combined with task training was no more effective in improving gait and lower limb performance in patients with subacute stroke than training alone, but this was a small-sample study. Furthermore, rTMS can also help improve posture, exercise planning, preparation, and execution in patients in the chronic phase, and more studies are needed in patients at different stages [45]. It is now generally accepted that earlier rehabilitation after a stroke is associated with better functional outcome. Bornheim [115] demonstrated that tDCS improved functional activity in patients with acute stroke and maintained it for up to 1 year. In addition, rTMS combined with tDCS stimulation could be a preferable rehabilitative strategy for motor recovery in stroke patients [116,117]. Also, tACS is beneficial for gait changes after a stroke. Gait-specific plasticity can be induced when tACS is synchronized with the gait period frequency [118].
tVNS has been applied for the recovery of upper extremity function for stroke patients. Redgrave [92] and Wu’s [94] trial confirmed that tVNS is an effective and safe method for improvement of upper limb function in stroke, but the sample size was too small, requiring further confirmation by larger-sample studies. A recent study [96] demonstrated that taVNS was beneficial to both upper and lower extremity motor dysfunction in patients at the acute stage. The recent study mainly focused on upper limb function, and more high-quality studies are needed. Additionally, these trials have all used taVNS rather than tcVNS; therefore, it is hoped that taVNS will be used for post-stroke rehabilitation studies in the future.
CLINICAL APPLICATIONS OF NIBS IN SENSORY RECOVERY AFTER STROKE: There were several studies of rTMS in the rehabilitation of sensory dysfunction after stroke. Liang [119] demonstrated that HF-rTMS can improve upper extremity sensory function involving pain threshold and two-point discrimination in stroke patients. Pundik [120] also found that rTMS can improve the two-point discrimination ability of chronic stroke patients, but has no effect on improvement of vibration sense and motor sense. At present, there is no published phased trial of rTMS for the treatment of sensory disorders after stroke, and the evaluation method is relatively subjective, so it is hoped that there will be further studies.
tDCS also helps improve sensory function in stroke patients. In 2014, a study [121] found that anodic tDCS reduced thermal pain and thermal pain thresholds in stroke patients, and increased cold and cold pain thresholds, suggesting that tDCS improved sensory recognition in stroke patients with central pain after stroke. Subsequently, Koo et al [122] demonstrated that tDCS improved tactile sense, pain sense, and cortical sensibility in patients with subacute stroke. Moreover, somatosensory performance was also improved by tDCS in acute patients and could be maintained for up to 1 year [115]. Further trials are needed to identify the effects of tDCS with diverse stimulation times, stimulation methods, and longer follow-up periods.
tVNS has been demonstrated to improve stroke patients’ proprioception. Baig et al [93] treated ischemic stroke patients with taVNS for 6 weeks, and their proprioception and light touch improved after treatment. Furthermore, another study [96] has confirmed the taVNS has long-term effects on sensory dysfunction in acute stroke patients, but the measurement tools were not accurate, and specific sensory improvement needs to be studied.
CLINICAL APPLICATION OF NIBS TO RECOVERY FROM POST-STROKE APHASIA: In 2011, the first randomized and sham-stimulation controlled trial of rTMS in stroke patients at the subacute stage was reported by Weiduschat et al [123]. They found that rTMS is an effective, safe, and viable complementary treatment for post-stroke aphasia. LF-rTMS has been shown to improve spontaneous speech, naming, auditory comprehension, and functional communication in stroke patients with aphasia, especially subacute patients, for at least 3 months [124–127]. Hu [128] found that, compared with HF-rTMS, LF-rTMS performed better in improving auditory comprehension, spontaneous speech, and aphasia quotients (AQ) in stroke patients. The therapeutic effect of rTMS may be related to the stimulus site. After applying LF-rTMS in the right posterior superior temporal gyrus, auditory comprehension and repetition improved significantly, while inhibition in the posterior inferior frontal gyrus contributed to a significant enhancement of spontaneous speech and repetition [129]. Recently, a randomized sham-controlled blinded study [32] first found that subacute aphasia patients received a better effect by 1 Hz subthreshold rTMS over the pars triangularis of the right inferior frontal gyrus compared with patients in the chronic phase. Now, more research is focusing on the effects of rTMS on functional communication, with the ultimate goal of restoring communication in everyday life. The effectiveness of tDCS in improving the accuracy and speed of image naming in people who lose their speech after a stroke, especially in the chronic phase, has been confirmed by several experiments [130–134]. Branscheidt [135] demonstrated that the overall accuracy in a lexical decision task was improved when the motor cortex of the dominant hemisphere received the anodal stimulation. Moreover, a randomized sham-controlled study [136] showed that applying anodal tDCS over the left M1 can recover patients’ speech function, and can also excite and recruit more areas in the motor speech network. However, some recent studies [137,138] have shown that tDCS does not improve the disturbance of speech in post-stroke patients with aphasia and may not be as effective as adjuvant treatment for post-stroke aphasia.
Although tDCS and rTMS have been shown to increase the chances of reasonable recovery of language after strokes in clinical trials, these treatments locally activate glutamate and GABA neurons and interfere with circuits that regulate motor function [139], whereas tVNS only increases neuromodulation and does not interfere with ongoing nerve impulses [140]. There are no clinical studies on tVNS in treating post-stroke dysphagia, but tVNS has potential as a means to promote the recovery of speech impairment after stroke [141].
CLINICAL APPLICATIONS OF NIBS IN THE REHABILITATION OF DYSPHAGIA AFTER STROKE: In 2013, Park [142] observed a reduction in the prevalence of aspiration and pharyngeal residuals in stroke patients after treatment with 5 Hz rTMS. Studies using rTMS in dysphagia after acute and chronic stroke have been carried out. Most studies have reported beneficial effects [143–145], but some trials have found that rTMS also improves dysphagia [146,147]. The site of stimulation affects bilateral or contralateral stimulation, but not ipsilateral stimulation [148]. Compared with 10 Hz, 5 Hz, and 1 Hz, Du [145] found that swallowing function improved after treatment with 10 Hz rTMS. In general, rTMS appears to be a promising therapy for post-stroke dysphagia, although the standard stimulus parameters still need to be investigated.
Many trials have proven that tDCS enhances cortical input, supports swallowing network reorganization, and may be beneficial for dysphagia recovery [149–153]. Suntrup-Krueger et al [153] used clinical assessment and the validated Fiberoptic Endoscopic Dysphagia Severity Scale (FEDSS) to assess deglutition function, and found greater improvement in FEDSS with each hour of treatment starting earlier. A recent study [154] focused on patients’ diets and found that dietary intake was improved but the risk of aspiration was not reduced by tDCS in patients at the early stage. This study’s sample size was small, and large-scale trials are needed to validate it.
CLINICAL APPLICATIONS OF NIBS IN THE REHABILITATION OF PSD: rTMS has been widely used as a complementary therapy for depression, and research on its use in PSD is also ongoing. It has been shown that the use of rTMS over the left DLPFC can alleviate PSD in the chronic phase [155]. A meta-analysis [156] also suggested an active effect of rTMS on PSD, but heterogeneity and potential biases in such studies should be carefully examined. Recent studies [29,157] have demonstrated the efficacy of HF-rTMS for PSD and found changes in functional brain connectivity in patients before and after treatment.
Studies on tDCS to treat depression after stroke are still in the preliminary stage. Valiengo [158] found that 2mA of tDCS applied to patients’ left and right DLPFC can effectively improve depressive symptoms. Bornheim [115] also found that tDCS improved PSD and the effect lasted for 1 year. It seems that tDCS is a safe and effective therapy for PSD, but trials with larger samples, longer follow-ups, and more accurate outcome measures are needed.
Currently, there are no clinical trials of tVNS in the treatment of PSD, but tVNS is commonly used in the treatment of depression, which can cause some changes in brain functional connectivity in PSD patients and significantly relieve depressive symptoms [159]. In addition, tVNS is convenient to use and conducive to clinical use. Therefore, tVNS can be tried as a treatment for PSD.
CLINICAL APPLICATION OF NIBS TO REHABILITATION OF POST-STROKE UNILATERAL NEGLECT: Many experiments[25,160,161] have found that 1 Hz repetitive rTMS of the unaffected hemispheric parietal cortex improves unilateral neglect after stroke. Yang [30] and Zhang [162] combined rTMS with sensory cues and found that the combination was more effective for unilateral neglect after stroke than rTMS alone. However, the effectiveness of 1 Hz rTMS applied in the angular gyrus in combination with visual scanning training in patients with unilateral spatial neglect (USN) in the subacute phase has not been determined [163].
In 2008, Ko [164] first reported that the polarization of the parietal cortex direct current could improve the visual scanning in USN patients. Subsequently, many researchers [165,166] have found that the excitatory effect of tDCS anode on the right posterior parietal cortex and the inhibitory effect of tDCS cathode on the opposite improved unilateral neglect symptoms, while bilateral tDCS is more effective than unilateral stimulation. In severely impaired patients in the acute phase, tDCS combined with optokinetic drift relieved egocentric neglect but not allocentric neglect, Turgut et al found [167]. Recent studies [168] focused on the feasibility of a trial in patients and found it was poor, as too many patients had to be excluded due to serious complications. Thus, the basis for recruitment, the criteria of eligibility, and the parameters and location of interventions should be carefully considered in future studies on using tDCS in unilateral neglect.
CLINICAL APPLICATIONS OF NIBS IN TREATING POST-STROKE COGNITIVE IMPAIRMENT (PSCI): Only a few small-scale studies have explored the impact of rTMS on cognitive function in stroke patients. In 2010, Kim et al [169] performed rTMS at 10 Hz and 1 Hz in the dorsolateral prefrontal region for 2 weeks and found no significant improvement in cognitive function. Five years later, Lu et al [170] found that 4 weeks of rTMS at 1 Hz enhanced cognitive function and memory function after stroke. Subsequently, many studies [171,172] have found that HF-rTMS can also improve overall cognition, attention, and memory function after stroke, but there is currently a lack of large-scale, long-term follow-up trials investigating the impact of rTMS on cognitive function after stroke. Recently, Li et al [173] discovered that serum triiodothyronine, free triiodothyronine, and thyroid-stimulating hormone levels of stroke patients were positively correlated with MoCA score, and rTMS increased these thyroid hormone levels to enhance patients’ cognitive function.
tDCS also contributes to the recovery of cognitive impairment. About 2 decades ago, studies found that anodic tDCS acting on the dorsal prefrontal lobe improved working memory [174] and attention [175,176] in patients. Moreover, Shaker et al [177] demonstrated that tDCS is a safe and effective neurorehabilitation model that improves cognitive function in several areas, including attention and attention, graphic memory, logical reasoning, and reactive behavior. Recently, a study discovered that tDCS combined with cognitive training significantly enhanced motivation, valuation, and decision-making abilities in stroke patients.
There are several studies on cognitive impairment after stroke with tFUS. Wang and colleagues[178] found that transcranial ultrasound together with routine cognitive training significantly improved cognitive function after stroke. Currently, there are no clinical trials of tVNS in the treatment of PSCI, but animal experiments [179] have found that neuronal stimulation can improve spatial and fear memory in rats with middle cerebral artery occlusion, and the clinical efficacy needs to be further explored.
SAFETY OF NIBS:
rTMS is well tolerated in clinical use and is a relatively safe method, but can cause adverse effects such as seizures, headache, neck pain, and transient hearing abnormalities. In Kakuda’s study [180], 1725 patients were stimulated with rTMS, and a total of 22 patients reported temporary but not severe adverse effects, including mild dizziness, mild headache, and discomfort at the site of the stimulation. Seizure is the most serious adverse effect of rTMS and is more common with the use of HF-rTMS. Although many patients are receiving rTMS in clinical trials, few cases have been reported, so it appears the risk of rTMS inducing seizure is relatively low [181]. Several factors may increase the risk of rTMS-induced seizures, such as sleep deprivation, stress, depression, and excessive alcohol consumption [182]. Care should be taken to avoid these factors in clinical application, and preparation for the possibility of seizures is necessary. Headache and regional pain at the site of stimulation are common adverse effects of rTMS. The incidence of these adverse effects may depend on the intensity, location, and frequency of stimulation. Therefore, clinical use of rTMS should follow the safety recommendations of relevant guidelines [183] and exclude patients with contraindications. More high-quality evidence is needed to explore the relationship between stimulus parameters and adverse reactions. In addition, the localization of rTMS is relatively inaccurate in clinical use, and inaccurate position and direction of the coil may reduce the response rate of patients while affecting irrelevant brain regions [184], so neuroimaging such as functional magnetic resonance imaging can be applied to help localization [185].
Compared to rTMS, tDCS has a better clinical safety profile, with no serious adverse effects [186]. However, there are still minor adverse reactions in the treatment process, such as skin redness, slight tingling, dizziness, and fatigue, which are safe, short-lived, and can be tolerated by most patients. The efficacy and safety of tDCS are dose-dependent, with higher current intensities providing better efficacy and potentially more brain damage. Currently, a single dose of 4 mA tDCS for 30 min is considered tolerable and safe in stroke subjects in clinical studies [187], and tDCS with current intensity ≤2 mA is mostly used for treatment [188]. Moreover, the charge density is more comprehensive than the current density in judging the safety of tDCS, since the charge density parameter takes into account the duration of the stimulus, such as 1 mA for 10 min, 2 mA for 5 min, and 10 mA for 1 min which have similar charges. Therefore, during the clinical application of tDCS, the relationship between dose and efficacy should be considered and the most appropriate parameters for the patient are effective and safe.
tFUS is used for neuromodulation in humans, without any reports of serious related symptoms; moderate and mild symptoms may occur, including neck pain, inattention, muscle twitching, and anxiety [189]. A study [190] found that symptoms generally appeared after the stimulation ended, none of these symptoms persisted after 1 month of follow-up, and no new symptoms were reported. Animal studies [191] have found microbleeds in the primary visual cortex of sheep after 600 treatments with tFUS of 6.6 W/cm2 Isppa, but the Ispta value of 3.3 W/cm2 exceeds the physical therapy US limit of 3 W/cm2. FDA guidelines [192] define safety thresholds for an ultrasound for head ultrasound diagnosis and neural regulation in adults that are Isspa ≤190 W/cm2, Ispta ≤94 mW/cm2, together with mechanical index ≤1.9, which need to be appropriately evaluated in future studies to select the safety parameters of tFUS for neurological rehabilitation.
So far, the clinical use of tVNS has been relatively safe and well tolerated. The most common adverse effects are local skin irritation, headache, and dizziness, but these are short-lived and may resolve during treatment [193,194]. Palpitations, arrhythmias, hypotension, and bradycardia were also reported in a small number of patients, but their association with nVNS has yet to be examined [195,196].
Discussion
The current clinical status [2,3] showed that stroke is extremely harmful, the incidence is increasing, and the motor, sensory, speech, swallowing, and other functions of survivors are seriously affected. Therefore, how to improve the functions of stroke survivors is the key and difficult point in clinical practice. Our extensive literature review found that the application of NIBS in treating stroke is an important research focus. The recent literature shows that NIBS, including rTMS, tDCS, tACS, tFUS, and tVNS, can improve functions in stroke patients. In particular, in terms of motor function, the studies found that function of patients with stroke can be significantly improved with rTMS [102] or tDCS [11] combined with traditional rehabilitation treatment. Additionally, tVNS, which sends electrical stimulation to the brain via the vagus nerve, has also been verified to improve motor and sensory functions in stroke patients [93]. However, there have been few trials on the use of tACS and tFUS in stroke rehabilitation. One study [67] has shown that tACS has no benefit for curing hemianopia after stroke, but tACS and tFUS have been used to treat the dysfunction caused by other clinical diseases, showing their potential in stroke rehabilitation in the future. Some studies [12,41] have found that NIBS can improve the vascular structure, morphology, and cerebral blood flow in stroke patients, which also indicates that NIBS is beneficial for stroke patients. The present article reviewed the status of research of the mechanism of action, therapeutic parameters, efficacy, and safety of NIBS in patients after stroke, and discussed its future development trend, to support the clinical application of NIBS in stroke rehabilitation. We look forward to publication of more high-quality evidence to verify the role of NIBS in stroke rehabilitation in the future.
Conclusions
NIBS has been shown to effectively improve motor, sensory, speech, swallowing, cognitive impairment, and depression after stroke. However, due to the lack of standardized stimulus protocols and single-use efficacy validation, the potential clinical utility of NIBS needs to be confirmed by higher-quality evidence-based medicine results.
Tables
Table 1. Summary of clinical studies on post-stroke patients using rTMS.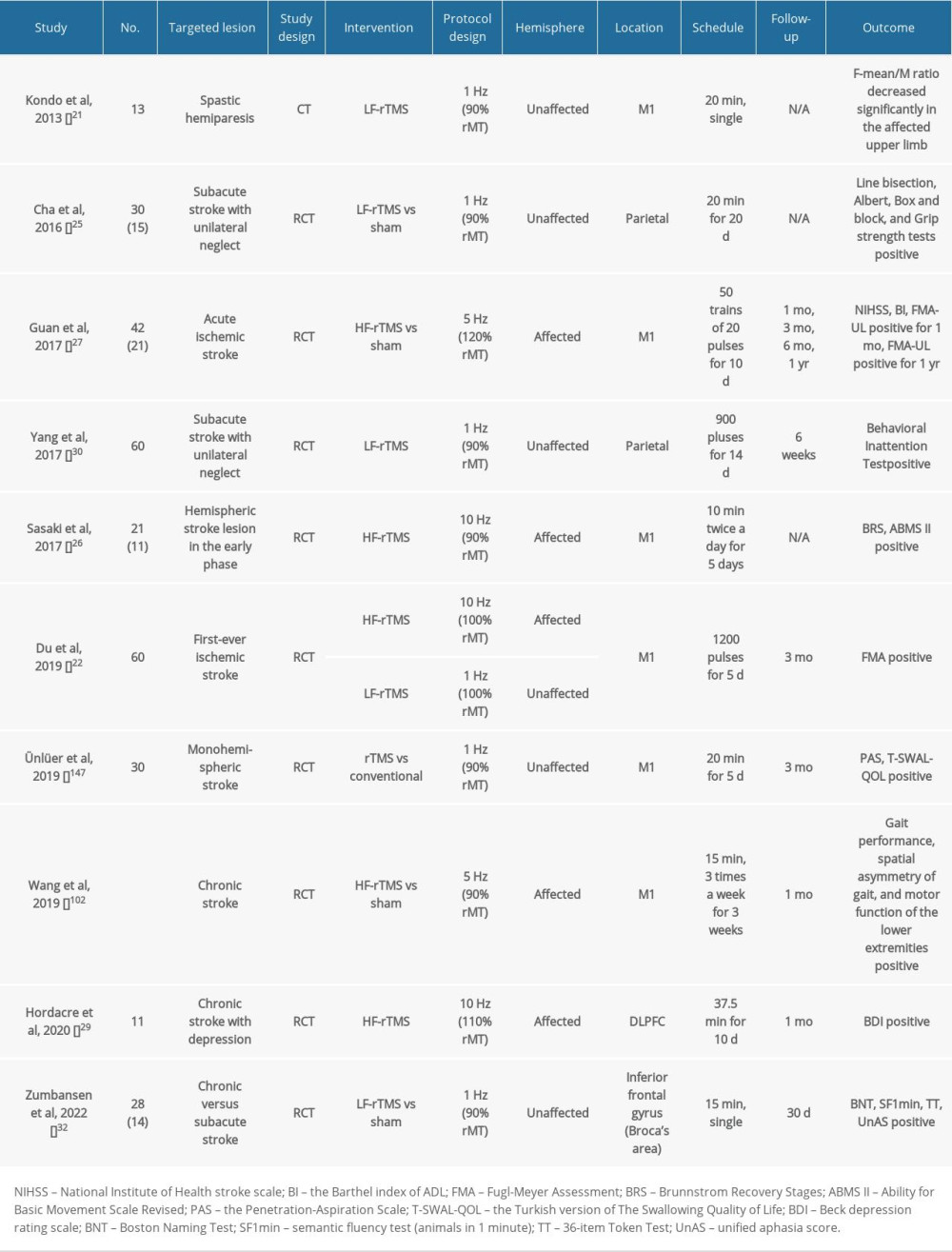 Table 2. Summary of clinical studies on post-stroke patients using tDCS.
Table 2. Summary of clinical studies on post-stroke patients using tDCS.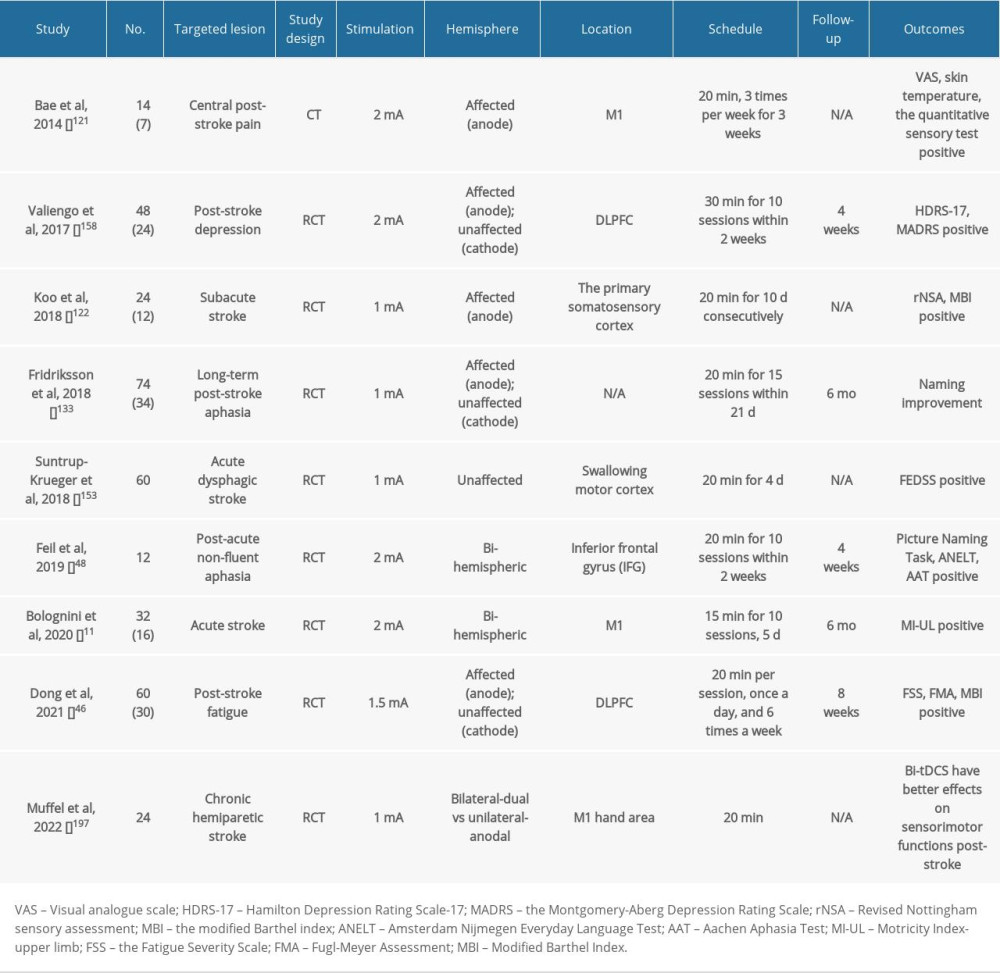 Table 3. Summary of clinical studies on post-stroke patients using tACS.
Table 3. Summary of clinical studies on post-stroke patients using tACS.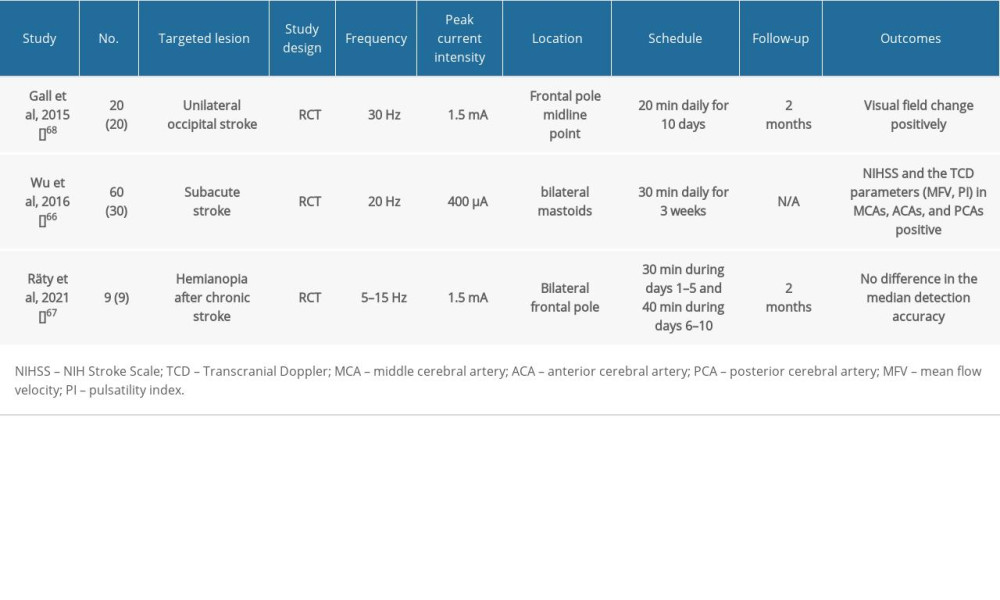 Table 4. Summary of clinical studies on post-stroke patients using tFUS.
Table 4. Summary of clinical studies on post-stroke patients using tFUS.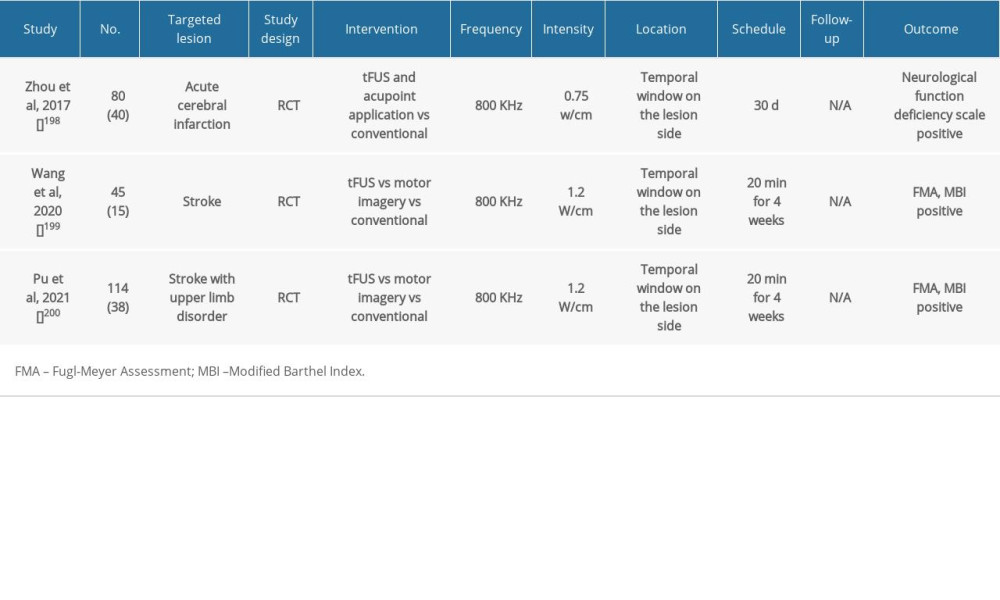 Table 5. Summary of clinical studies on post-stroke patients using tVNS.
Table 5. Summary of clinical studies on post-stroke patients using tVNS.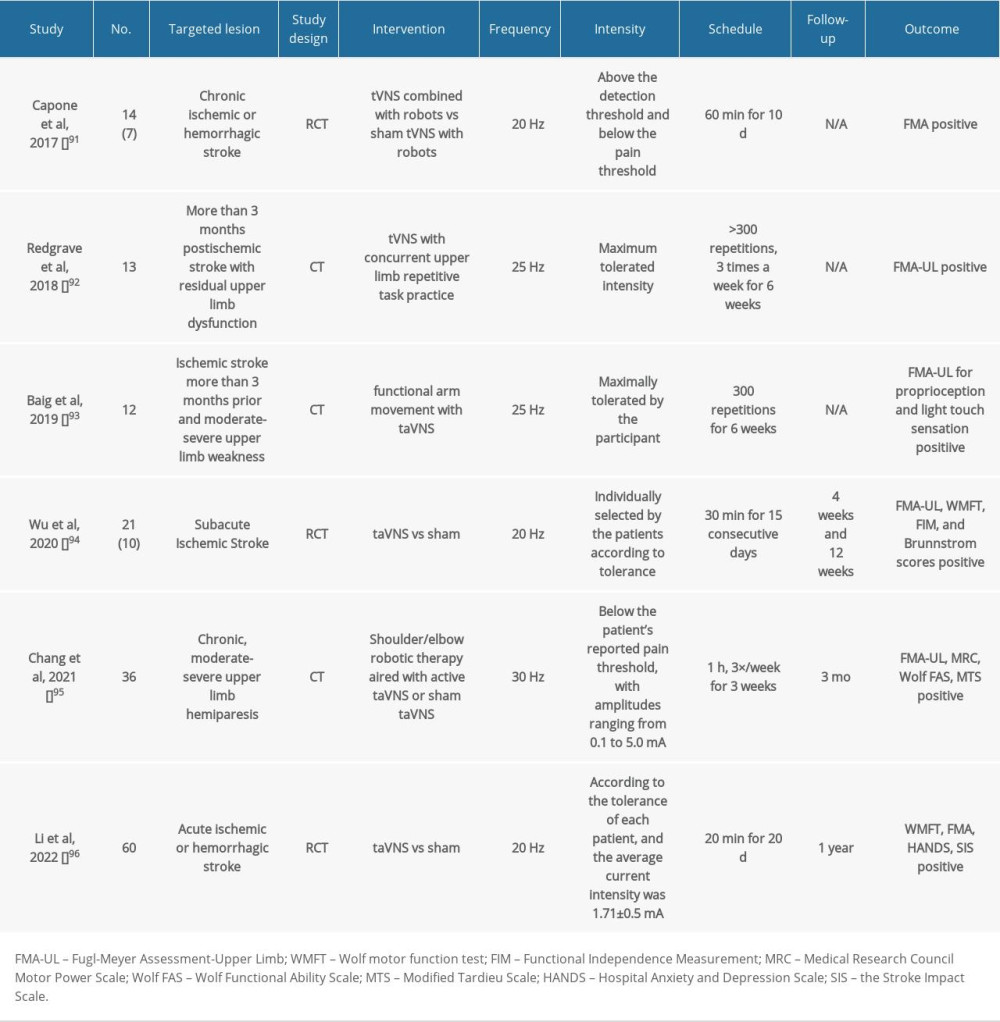
References
1. , Global, regional, and national burden of neurological disorders, 1990-2016: A systematic analysis for the Global Burden of Disease Study 2016: Lancet Neurol, 2019; 18(5); 459-80
2. , Global burden of 369 diseases and injuries in 204 countries and territories, 1990-2019: A systematic analysis for the Global Burden of Disease Study 2019: Lancet (London, England), 2020; 396(10258); 1204-22
3. Wang YJ, Li ZX, Gu HQ, China Stroke Statistics: An update on the 2019 report from the National Center for Healthcare Quality Management in Neurological Diseases, China National Clinical Research Center for Neurological Diseases, the Chinese Stroke Association, National Center for Chronic and Non-communicable Disease Control and Prevention, Chinese Center for Disease Control and Prevention and Institute for Global Neuroscience and Stroke Collaborations: Stroke Vasc Neurol, 2022; 7(5); 415-50
4. Meyer BU, Röricht S, Gräfin von Einsiedel H, Inhibitory and excitatory interhemispheric transfers between motor cortical areas in normal humans and patients with abnormalities of the corpus callosum: Brain, 1995; 118(Pt 2); 429-40
5. Bertolucci F, Chisari C, Fregni F, The potential dual role of transcallosal inhibition in post-stroke motor recovery: Restor Neurol Neurosci, 2018; 36(1); 83-97
6. Murase N, Duque J, Mazzocchio R, Cohen LG, Influence of interhemispheric interactions on motor function in chronic stroke: Ann Neurol, 2004; 55(3); 400-9
7. Nowak DA, Grefkes C, Ameli M, Fink GR, Interhemispheric competition after stroke: Brain stimulation to enhance recovery of function of the affected hand: Neurorehabil Neural Repair, 2009; 23(7); 641-56
8. Engelhardt M, Picht T, 1 Hz repetitive transcranial magnetic stimulation of the primary motor cortex: Impact on excitability and task performance in healthy subjects: J Neurol Surgery A Cent Eur Neurosurg, 2020; 81(2); 147-54
9. Klomjai W, Lackmy-Vallée A, Roche N, Pradat-Diehl P, Repetitive transcranial magnetic stimulation and transcranial direct current stimulation in motor rehabilitation after stroke: An update: Ann Phys Rehabil Med, 2015; 58(4); 220-24
10. Han T, Xu Z, Liu C, Simultaneously applying cathodal tDCS with low frequency rTMS at the motor cortex boosts inhibitory aftereffects: J Neurosci Methods, 2019; 324; 108308
11. Bolognini N, Russo C, Souza Carneiro MI, Bi-hemispheric transcranial direct current stimulation for upper-limb hemiparesis in acute stroke: A randomized, double-blind, sham-controlled trial: Eur J Neurol, 2020; 27(12); 2473-82
12. Kim E, Anguluan E, Youn S, Non-invasive measurement of hemodynamic change during 8 MHz transcranial focused ultrasound stimulation using near-infrared spectroscopy: BMC Neurosci 18, 2019; 20(1); 12
13. Barker AT, Jalinous R, Freeston IL, Non-invasive magnetic stimulation of human motor cortex: Lancet (London, England), 1985; 1(8437); 1106-7
14. Hallett M, Transcranial magnetic stimulation and the human brain: Nature, 2000; 406(6792); 147-50
15. Thut G, Northoff G, Ives JR, Effects of single-pulse transcranial magnetic stimulation (TMS) on functional brain activity: A combined event-related TMS and evoked potential study: Clin Neurophysiol, 2003; 114(11); 2071-80
16. Rossini PM, Rossi S, Transcranial magnetic stimulation: diagnostic, therapeutic, and research potential: Neurology, 2007; 68(7); 484-88
17. Sallustio F, Di Legge S, Rizzato B, Changes in cerebrovascular reactivity following low-frequency repetitive transcranial magnetic stimulation: J Neurol Sci, 2010; 295(1–2); 58-61
18. Zong X, Li Y, Liu C, Theta-burst transcranial magnetic stimulation promotes stroke recovery by vascular protection and neovascularization: Theranostics, 2020; 10(26); 12090-110
19. Chen R, Classen J, Gerloff C, Depression of motor cortex excitability by low-frequency transcranial magnetic stimulation: Neurology, 1997; 48(5); 1398-403
20. de Jesus DR, Favalli GPS, Hoppenbrouwers SS, Determining optimal rTMS parameters through changes in cortical inhibition: Clin Neurophysiol, 2014; 125(4); 755-62
21. Kondo T, Kakuda W, Yamada N, Effect of low-frequency rTMS on motor neuron excitability after stroke: Acta Neurol Scand, 2013; 127(1); 26-30
22. Du J, Yang F, Hu J, Effects of high- and low-frequency repetitive transcranial magnetic stimulation on motor recovery in early stroke patients: Evidence from a randomized controlled trial with clinical, neurophysiological and functional imaging assessments: Neuroimage Clin, 2019; 21; 101620
23. Hoyer EH, Celnik PA, Understanding and enhancing motor recovery after stroke using transcranial magnetic stimulation: Restor Neur neuroscience, 2011; 29(6); 395-409
24. Guo Z, Jin Y, Bai X, Distinction of high- and low-frequency repetitive transcranial magnetic stimulation on the functional reorganization of the motor network in stroke patients: Neural Plast, 2021; 2021; 8873221
25. Cha HG, Kim MK, Effects of repetitive transcranial magnetic stimulation on arm function and decreasing unilateral spatial neglect in subacute stroke: A randomized controlled trial: Clin Rehabil, 2016; 30(7); 649-56
26. Sasaki N, Abo M, Hara T, High-frequency rTMS on leg motor area in the early phase of stroke: Acta Neurol Belg, 2017; 117(1); 189-94
27. Guan YZ, Li J, Zhang XW, Effectiveness of repetitive transcranial magnetic stimulation (rTMS) after acute stroke: A one-year longitudinal randomized trial: CNS Neurosci Ther, 2017; 23(12); 940-46
28. Lefaucheur JP, Aleman A, Baeken C, Evidence-based guidelines on the therapeutic use of repetitive transcranial magnetic stimulation (rTMS): An update (2014–2018): Clin Neurophysiol, 2020; 131(2); 474-528
29. Hordacre B, Comacchio K, Williams L, Hillier S, Repetitive transcranial magnetic stimulation for post-stroke depression: A randomised trial with neurophysiological insight: J Neurol, 2021; 268(4); 1474-84
30. Yang NY, Fong KN, Li-Tsang CW, Zhou D, Effects of repetitive transcranial magnetic stimulation combined with sensory cueing on unilateral neglect in subacute patients with right hemispheric stroke: A randomized controlled study: Clin Rehabil, 2017; 31(9); 1154-63
31. Fahmy EM, Elshebawy HM, Effect of high frequency transcranial magnetic stimulation on recovery of chronic post-stroke aphasia: J Stroke Cerebrovasc Dis, 2021; 30(8); 105855
32. Zumbansen A, Kneifel H, Lazzouni L, Differential effects of speech and language therapy and rTMS in chronic versus subacute post-stroke aphasia: Results of the NORTHSTAR-CA trial: Neurorehabil Neural Repair, 2022; 36(4–5); 306-16
33. Abo M, Kakuda W, Watanabe M, Effectiveness of low-frequency rTMS and intensive speech therapy in poststroke patients with aphasia: A pilot study based on evaluation by fMRI in relation to type of aphasia: Eur Neurol, 2012; 68(4); 199-208
34. Shafi MM, Westover MB, Fox MD, Pascual-Leone A, Exploration and modulation of brain network interactions with noninvasive brain stimulation in combination with neuroimaging: Eur J Neurol, 2012; 35(6); 805-25
35. Hartwigsen G, Volz LJ, Probing rapid network reorganization of motor and language functions via neuromodulation and neuroimaging: Neuroimage, 2021; 224; 117449
36. Nitsche MA, Paulus W, Excitability changes induced in the human motor cortex by weak transcranial direct current stimulation: J Physiol, 2000; 527(Pt 3); 633-39
37. Woods AJ, Antal A, Bikson M, A technical guide to tDCS, and related non-invasive brain stimulation tools: Clin Neurophysiol, 2016; 127(2); 1031-48
38. Jackson MP, Rahman A, Lafon B, Animal models of transcranial direct current stimulation: Methods and mechanisms: Clin Neurophysiol, 2016; 127(11); 3425-54
39. Nitsche MA, Paulus W, Sustained excitability elevations induced by transcranial DC motor cortex stimulation in humans: Neurology, 2001; 57(10); 1899-901
40. Kunze T, Hunold A, Haueisen J, Jirsa V, Spiegler A, Transcranial direct current stimulation changes resting state functional connectivity: A large-scale brain network modeling study: Neuroimage, 2016; 140; 174-87
41. Zheng X, Alsop DC, Schlaug G, Effects of transcranial direct current stimulation (tDCS) on human regional cerebral blood flow: Neuroimage, 2011; 58(1); 26-33
42. Wen HZ, Gao SH, Zhao YD, Parameter optimization analysis of prolonged analgesia effect of tDCS on neuropathic pain rats: Front Behav Neurosci, 2017; 11; 115
43. Batsikadze G, Moliadze V, Paulus W, Partially non-linear stimulation intensity-dependent effects of direct current stimulation on motor cortex excitability in humans: J Physiol, 2013; 591(7); 1987-2000
44. Jamil A, Batsikadze G, Kuo HI, Systematic evaluation of the impact of stimulation intensity on neuroplastic after-effects induced by transcranial direct current stimulation: J Physiol, 2017; 595(4); 1273-88
45. Yang CL, Gad A, Creath RA, Effects of transcranial direct current stimulation (tDCS) on posture, movement planning, and execution during standing voluntary reach following stroke: J Neuroeng Rehabil, 2021; 18(1); 5
46. Dong XL, Sun X, Sun WM, A randomized controlled trial to explore the efficacy and safety of transcranial direct current stimulation on patients with post-stroke fatigue: Medicine (Baltimore), 2021; 100(41); e27504
47. Elsner B, Kugler J, Pohl M, Mehrholz J, Transcranial direct current stimulation (tDCS) for improving aphasia in adults with aphasia after stroke: Cochrane Database Syst Rev, 2019; 5(5); CD009760
48. Feil S, Eisenhut P, Strakeljahn F, Left shifting of language related activity induced by bihemispheric tDCS in postacute aphasia following stroke: Front Neurosci, 2019; 13; 295
49. Elyamany O, Leicht G, Herrmann CS, Mulert C, Transcranial alternating current stimulation (tACS): From basic mechanisms towards first applications in psychiatry: Eur Arch Psychiatry Clin Neurosci, 2021; 271(1); 135-56
50. Bikson M, Inoue M, Akiyama H, Effects of uniform extracellular DC electric fields on excitability in rat hippocampal slices in vitro: J Physiol, 2004; 557(Pt 1); 175-90
51. Fritsch B, Reis J, Martinowich K, Direct current stimulation promotes BDNF-dependent synaptic plasticity: Potential implications for motor learning: Neuron, 2010; 66(2); 198-204
52. Krause MR, Vieira PG, Csorba BA, Transcranial alternating current stimulation entrains single-neuron activity in the primate brain: Proc Natl Acad Sci USA, 2019; 116(12); 5747-55
53. Helfrich RF, Schneider TR, Rach S, Entrainment of brain oscillations by transcranial alternating current stimulation: Curr Biol, 2014; 24(3); 333-39
54. Tavakoli AV, Yun K, Transcranial alternating current stimulation (tACS) mechanisms and protocols: Front Cell Neurosci, 2017; 11; 214
55. Zaehle T, Rach S, Herrmann CS, Transcranial alternating current stimulation enhances individual alpha activity in human EEG: PLoS One, 2010; 5(11); e13766
56. Ali MM, Sellers KK, Fröhlich F, Transcranial alternating current stimulation modulates large-scale cortical network activity by network resonance: J Neurosci, 2013; 33(27); 11262-75
57. Sugata H, Yagi K, Yazawa S, Modulation of motor learning capacity by transcranial alternating current stimulation: Neuroscience, 2018; 391; 131-39
58. Pollok B, Boysen AC, Krause V, The effect of transcranial alternating current stimulation (tACS) at alpha and beta frequency on motor learning: Behav Brain Res, 2015; 293; 234-40
59. Rumpf JJ, Barbu A, Fricke C, Posttraining alpha transcranial alternating current stimulation impairs motor consolidation in elderly people: Neural plasticity, 2019; 2019; 2689790
60. Hsu WY, Zanto TP, Gazzaley A, Parametric effects of transcranial alternating current stimulation on multitasking performance: Brain Stimul, 2019; 12(1); 73-83
61. Berntsen MB, Cooper NR, Hughes G, Romei V, Prefrontal transcranial alternating current stimulation improves motor sequence reproduction: Behav Brain Res, 2019; 361; 39-49
62. Wessel MJ, Draaisma LR, de Boer AFW, Cerebellar transcranial alternating current stimulation in the gamma range applied during the acquisition of a novel motor skill: Sci Rep, 2020; 10(1); 11217
63. Haller N, Senner F, Brunoni AR, Gamma transcranial alternating current stimulation improves mood and cognition in patients with major depression: J Psychiatr Res, 2020; 130; 31-34
64. Gall C, Schmidt S, Schittkowski MP, Alternating current stimulation for vision restoration after optic nerve damage: A randomized clinical trial: PLoS One, 2016; 11(6); e0156134
65. Clayton MS, Yeung N, Cohen Kadosh R, Electrical stimulation of alpha oscillations stabilizes performance on visual attention tasks: J Exp Psychol Gen, 2019; 148(2); 203-20
66. Wu JF, Wang HJ, Wu Y, Efficacy of transcranial alternating current stimulation over bilateral mastoids (tACS(bm)) on enhancing recovery of subacute post-stroke patients: Top Stroke Rehabil, 2016; 23(6); 420-29
67. Räty S, Borrmann C, Granata G, Non-invasive electrical brain stimulation for vision restoration after stroke: An exploratory randomized trial (REVIS): Restor Neurol Neurosci, 2021; 39(3); 221-35
68. Gall C, Silvennoinen K, Granata G, Non-invasive electric current stimulation for restoration of vision after unilateral occipital stroke: Contemp Clin Trials, 2015; 43; 231-36
69. Yoo SS, Kim H, Min BK, Transcranial focused ultrasound to the thalamus alters anesthesia time in rats: Neuroreport, 2011; 22(15); 783-87
70. Folloni D, Verhagen L, Mars RB, Manipulation of subcortical and deep cortical activity in the primate brain using transcranial focused ultrasound stimulation: Neuron, 2019; 101(6); 1109-16e5
71. Tufail Y, Matyushov A, Baldwin N, Transcranial pulsed ultrasound stimulates intact brain circuits: Neuron, 2010; 66(5); 681-94
72. Yoo SS, Bystritsky A, Lee JH, Focused ultrasound modulates region-specific brain activity: Neuroimage, 2011; 56(3); 1267-75
73. Tsui PH, Wang SH, Huang CC, In vitro effects of ultrasound with different energies on the conduction properties of neural tissue: Ultrasonics, 2005; 43(7); 560-65
74. Colucci V, Strichartz G, Jolesz F, Focused ultrasound effects on nerve action potential in vitro: Ultrasound Med Biol, 2009; 35(10); 1737-47
75. O’Brien WD, Ultrasound-biophysics mechanisms: Prog Biophys Mol Biol, 2007; 93(1–3); 212-55
76. Tyler WJ, Tufail Y, Finsterwald M, Remote excitation of neuronal circuits using low-intensity, low-frequency ultrasound: PLoS One, 2008; 3(10); e3511
77. King RL, Brown JR, Newsome WT, Pauly KB, Effective parameters for ultrasound-induced in vivo neurostimulation: Ultrasound Med Biol, 2013; 39(2); 312-31
78. Kubanek J, Shukla P, Das A, Ultrasound elicits behavioral responses through mechanical effects on neurons and ion channels in a simple nervous system: J Neurosci, 2018; 38(12); 3081-91
79. Deng LD, Qi L, Suo Q, Transcranial focused ultrasound stimulation reduces vasogenic edema after middle cerebral artery occlusion in mice: Neural Regen Res, 2022; 17(9); 2058-63
80. Wang J, Li G, Deng L, Transcranial focused ultrasound stimulation improves neurorehabilitation after middle cerebral artery occlusion in mice: Aging Dis, 2021; 12(1); 50-60
81. Fallgatter AJ, Neuhauser B, Herrmann MJ, Far field potentials from the brain stem after transcutaneous vagus nerve stimulation: J Neural Transm (Vienna), 2003; 110(12); 1437-43
82. Ben-Menachem E, Revesz D, Simon BJ, Silberstein S, Surgically implanted and non-invasive vagus nerve stimulation: A review of efficacy, safety and tolerability: Eur J Neurol, 2015; 22(9); 1260-68
83. Yakunina N, Kim SS, Nam EC, Optimization of transcutaneous vagus nerve stimulation using functional MRI: Neuromodulation, 2017; 20(3); 290-300
84. Yuan H, Silberstein SD, Vagus nerve and vagus nerve stimulation, a comprehensive review: Part II: Headache, 2016; 56(2); 259-66
85. Capone F, Assenza G, Di Pino G, The effect of transcutaneous vagus nerve stimulation on cortical excitability: J Neural Transm (Vienna), 2015; 122(5); 679-85
86. Badran BW, Dowdle LT, Mithoefer OJ, Neurophysiologic effects of transcutaneous auricular vagus nerve stimulation (taVNS) via electrical stimulation of the tragus: A concurrent taVNS/fMRI study and review: Brain Stimul, 2018; 11(3); 492-500
87. Yang Y, Yang LY, Orban L, Non-invasive vagus nerve stimulation reduces blood-brain barrier disruption in a rat model of ischemic stroke: Brain Stimul, 2018; 11(4); 689-98
88. Lindemann J, Rakers C, Matuskova H, Vagus nerve stimulation reduces spreading depolarization burden and cortical infarct volume in a rat model of stroke: PLoS One, 2020; 15(7); e0236444
89. Zhao JJ, Wang ZH, Zhang YJ, The mechanisms through which auricular vagus nerve stimulation protects against cerebral ischemia/reperfusion injury: Neural Regen Res, 2022; 17(3); 594-600
90. Agnew WF, McCreery DB, Considerations for safety with chronically implanted nerve electrodes: Epilepsia, 1990; 31(Suppl 2); S27-32
91. Capone F, Miccinilli S, Pellegrino G, Transcutaneous vagus nerve stimulation combined with robotic rehabilitation improves upper limb function after stroke: Neural Plast, 2017; 2017; 7876507
92. Redgrave JN, Moore L, Oyekunle T, Transcutaneous auricular vagus nerve stimulation with concurrent upper limb repetitive task practice for poststroke motor recovery: A pilot study: J Stroke Cerebrovasc Dis, 2018; 27(7); 1998-2005
93. Baig SS, Falidas K, Laud PJ, Transcutaneous auricular vagus nerve stimulation with upper limb repetitive task practice may improve sensory recovery in chronic stroke: J Stroke Cerebrovasc Dis, 2019; 28(12); 104348
94. Wu D, Ma J, Zhang L, Effect and safety of transcutaneous auricular vagus nerve stimulation on recovery of upper limb motor function in subacute ischemic stroke patients: A randomized pilot study: Neural Plast, 2020; 2020; 8841752
95. Chang JL, Coggins AN, Saul M, Transcutaneous auricular vagus nerve stimulation (tAVNS) delivered during upper limb interactive robotic training demonstrates novel antagonist control for reaching movements following stroke: Front Neurosci, 2021; 15; 767302
96. Li JN, Xie CC, Li CQ, Efficacy and safety of transcutaneous auricular vagus nerve stimulation combined with conventional rehabilitation training in acute stroke patients: A randomized controlled trial conducted for 1 year involving 60 patients: Neural Regen Res, 2022; 17(8); 1809-13
97. Avenanti A, Coccia M, Ladavas E, Low-frequency rTMS promotes use-dependent motor plasticity in chronic stroke: A randomized trial: Neurology, 2012; 78(4); 256-64
98. Sasaki N, Mizutani S, Kakuda W, Abo M, Comparison of the effects of high- and low-frequency repetitive transcranial magnetic stimulation on upper limb hemiparesis in the early phase of stroke: J Stroke Cerebrovasc Dis, 2013; 22(4); 413-18
99. Sasaki N, Kakuda W, Abo M, Bilateral high- and low-frequency rTMS in acute stroke patients with hemiparesis: A comparative study with unilateral high-frequency rTMS: Brain Inj, 2014; 28(13–14); 1682-86
100. Wang L, Zhu QX, Zhong MH, Effects of corticospinal tract integrity on upper limb motor function recovery in stroke patients treated with repetitive transcranial magnetic stimulation: J Integr Neurosci, 2022; 21(2); 50
101. Rastgoo M, Naghdi S, Nakhostin Ansari N, Effects of repetitive transcranial magnetic stimulation on lower extremity spasticity and motor function in stroke patients: Disabil Rehabil, 2016; 38(19); 1918-26
102. Wang RY, Wang FY, Huang SF, Yang YR, High-frequency repetitive transcranial magnetic stimulation enhanced treadmill training effects on gait performance in individuals with chronic stroke: A double-blinded randomized controlled pilot trial: Gait Posture, 2019; 68; 382-87
103. Kim WS, Kwon BS, Seo HG, Low-frequency repetitive transcranial magnetic stimulation over contralesional motor cortex for motor recovery in subacute ischemic stroke: A randomized sham-controlled trial: Neurorehabilitation and neural repair, 2020; 34(9); 856-67
104. Lüdemann-Podubecká J, Bösl K, Nowak DA, Inhibition of the contralesional dorsal premotor cortex improves motor function of the affected hand following stroke: Eur J Neurol, 2016; 23(4); 823-30
105. Gomes-Osman J, Field-Fote EC, Improvements in hand function in adults with chronic tetraplegia following a multiday 10-Hz repetitive transcranial magnetic stimulation intervention combined with repetitive task practice: J Neurol Phys Ther, 2015; 39(1); 23-30
106. Yang Y, Pan H, Pan W, Repetitive transcranial magnetic stimulation on the affected hemisphere enhances hand functional recovery in subacute adult stroke patients: A randomized trial: Front Aging Neurosci, 2021; 13; 636184
107. Kim DY, Ohn SH, Yang EJ, Enhancing motor performance by anodal transcranial direct current stimulation in subacute stroke patients: Am J Phys Med Rehabil, 2009; 88(10); 829-36
108. Kim DY, Lim JY, Kang EK, Effect of transcranial direct current stimulation on motor recovery in patients with subacute stroke: Am J Phys Med Rehabil, 2010; 89(11); 879-86
109. Kuo IJ, Tang CW, Tsai YA, Neurophysiological signatures of hand motor response to dual-transcranial direct current stimulation in subacute stroke: A TMS and MEG study: J Neuroeng Rehabil, 2020; 17(1); 72
110. Rocha S, Silva E, Foerster Á, The impact of transcranial direct current stimulation (tDCS) combined with modified constraint-induced movement therapy (mCIMT) on upper limb function in chronic stroke: A double-blind randomized controlled trial: Disabil Rehabil, 2016; 38(7); 653-60
111. Ojardias E, Azé OD, Luneau D, The effects of anodal transcranial direct current stimulation on the walking performance of chronic hemiplegic patients: Neuromodulation, 2020; 23(3); 373-79
112. Tanaka S, Takeda K, Otaka Y, Single session of transcranial direct current stimulation transiently increases knee extensor force in patients with hemiparetic stroke: Neurorehabil Neural Repair, 2011; 25(6); 565-69
113. Tahtis V, Kaski D, Seemungal BM, The effect of single session bi-cephalic transcranial direct current stimulation on gait performance in sub-acute stroke: A pilot study: Restor Neurol Neurosci, 2014; 32(4); 527-32
114. Aneksan B, Sawatdipan M, Bovonsunthonchai S, Five-session dual-transcranial direct current stimulation with task-specific training does not improve gait and lower limb performance over training alone in subacute stroke: A pilot randomized controlled trial: Neuromodulation, 2022; 25(4); 558-68
115. Bornheim S, Croisier JL, Maquet P, Kaux JF, Transcranial direct current stimulation associated with physical-therapy in acute stroke patients – a randomized, triple blind, sham-controlled study: Brain Stimul, 2020; 13(2); 329-36
116. Kwon TG, Park E, Kang C, The effects of combined repetitive transcranial magnetic stimulation and transcranial direct current stimulation on motor function in patients with stroke: Restor Neurol Neurosci, 2016; 34(6); 915-23
117. Gong Y, Long XM, Xu Y, Effects of repetitive transcranial magnetic stimulation combined with transcranial direct current stimulation on motor function and cortex excitability in subacute stroke patients: A randomized controlled trial: Clin Rhabil, 2021; 35(5); 718-27
118. Kitatani R, Koganemaru S, Maeda A, Gait-synchronized oscillatory brain stimulation modulates common neural drives to ankle muscles in patients after stroke: A pilot study: Neurosci Res, 2020; 156; 256-64
119. Qiting L, Yantao Z, Xiaogeng S, Effect of high-frequency repetitive transcranial magnetic stimulation on motor and sensory function of upper limbs in ischemic stroke patients: Chinese Journal of Geriatric Heart Brain and Vessel Diseases, 2018; 20(11); 1187-90
120. Pundik S, Skelly M, McCabe J, Does rTMS targeting contralesional S1 enhance upper limb somatosensory function in chronic stroke? A proof-of-principle study: Neurorehabil Neural Repair, 2021; 35(3); 233-46
121. Bae SH, Kim GD, Kim KY, Analgesic effect of transcranial direct current stimulation on central post-stroke pain: Tohoku J Exp Med, 2014; 234(3); 189-95
122. Koo WR, Jang BH, Kim CR, Effects of anodal transcranial direct current stimulation on somatosensory recovery after stroke: A randomized controlled trial: Am J Phys Med Rehabil, 2018; 97(7); 507-13
123. Weiduschat N, Thiel A, Rubi-Fessen I, Effects of repetitive transcranial magnetic stimulation in aphasic stroke: A randomized controlled pilot study: Stroke, 2011; 42(2); 409-15
124. Tsai PY, Wang CP, Ko JS, The persistent and broadly modulating effect of inhibitory rTMS in nonfluent aphasic patients: A sham-controlled, double-blind study: Neurorehabil Neural Repair, 2014; 28(8); 779-87
125. Wang CP, Hsieh CY, Tsai PY, Efficacy of synchronous verbal training during repetitive transcranial magnetic stimulation in patients with chronic aphasia: Stroke, 2014; 45(12); 3656-62
126. Rubi-Fessen I, Hartmann A, Huber W, Add-on effects of repetitive transcranial magnetic stimulation on subacute aphasia therapy: Enhanced improvement of functional communication and basic linguistic skills. A randomized controlled study: Arch Phys Med Rehabil, 2015; 96(11); 1935-44e2
127. Haghighi M, Mazdeh M, Ranjbar N, Seifrabie MA, Further evidence of the positive influence of repetitive transcranial magnetic stimulation on speech and language in patients with aphasia after stroke: Results from a double-blind intervention with sham condition: Neuropsychobiology, 2017; 75(4); 185-92
128. Hu XY, Zhang T, Rajah GB, Effects of different frequencies of repetitive transcranial magnetic stimulation in stroke patients with non-fluent aphasia: A randomized, sham-controlled study: Neurol Res, 2018; 40(6); 459-65
129. Ren C, Zhang G, Xu X, The effect of rTMS over the different targets on language recovery in stroke patients with global aphasia: A randomized sham-controlled study: Biomed Res Int, 2019; 2019; 4589056
130. Baker JM, Rorden C, Fridriksson J, Using transcranial direct-current stimulation to treat stroke patients with aphasia: Stroke, 2010; 41(6); 1229-36
131. Fridriksson J, Richardson JD, Baker JM, Rorden C, Transcranial direct current stimulation improves naming reaction time in fluent aphasia: A double-blind, sham-controlled study: Stroke, 2011; 42(3); 819-21
132. Meinzer M, Darkow R, Lindenberg R, Flöel A, Electrical stimulation of the motor cortex enhances treatment outcome in post-stroke aphasia: Brain, 2016; 139(Pt 4); 1152-63
133. Fridriksson J, Rorden C, Elm J, Transcranial direct current stimulation vs sham stimulation to treat aphasia after stroke: A randomized clinical trial: JAMA Neurol, 2018; 75(12); 1470-76
134. Pestalozzi MI, Di Pietro M, Martins Gaytanidis C, Effects of prefrontal transcranial direct current stimulation on lexical access in chronic poststroke aphasia: Neurorehabil Neural Repair, 2018; 32(10); 913-23
135. Branscheidt M, Hoppe J, Zwitserlood P, Liuzzi G, tDCS over the motor cortex improves lexical retrieval of action words in poststroke aphasia: J Neurophysiol, 2018; 119(2); 621-30
136. Wang J, Wu D, Cheng Y, Effects of transcranial direct current stimulation on apraxia of speech and cortical activation in patients with stroke: A randomized sham-controlled study: American journal of speech-language pathology, 2019; 28(4); 1625-37
137. Spielmann K, van de Sandt-Koenderman WME, Heijenbrok-Kal MH, Ribbers GM, Transcranial direct current stimulation does not improve language outcome in subacute poststroke aphasia: Stroke, 2018; 49(4); 1018-20
138. Guillouët E, Cogné M, Saverot E, Impact of combined transcranial direct current stimulation and speech-language therapy on spontaneous speech in aphasia: A randomized controlled double-blind study: J Int Neuropsychol Soc, 2020; 26(1); 7-18
139. Cirillo G, Di Pino G, Capone F, Neurobiological after-effects of non-invasive brain stimulation: Brain Stimul, 2017; 10(1); 1-18
140. Morrison RA, Danaphongse TT, Pruitt DT, A limited range of vagus nerve stimulation intensities produce motor cortex reorganization when delivered during training: Behav Brain Res, 2020; 391; 112705
141. Morrison RA, Hays SA, Kilgard MP, Vagus nerve stimulation as a potential adjuvant to rehabilitation for post-stroke motor speech disorders: Front Neurosci, 2021; 15; 715928
142. Park JW, Oh JC, Lee JW, The effect of 5Hz high-frequency rTMS over contralesional pharyngeal motor cortex in post-stroke oropharyngeal dysphagia: A randomized controlled study: Neurogastroenterol Motil, 2013; 25(4); 324-e250
143. Du J, Yang F, Liu L, Repetitive transcranial magnetic stimulation for rehabilitation of poststroke dysphagia: A randomized, double-blind clinical trial: Clin Neurophysiol, 2016; 127(3); 1907-13
144. Tarameshlu M, Ansari NN, Ghelichi L, Jalaei S, The effect of repetitive transcranial magnetic stimulation combined with traditional dysphagia therapy on poststroke dysphagia: A pilot double-blinded randomized-controlled trial: Int J Emerg Med, 2019; 42(2); 133-38
145. Du Y, Wei L, Lu Y, Gao H, The effects of different frequencies of repetitive transcranial magnetic stimulation (rTMS) on patients with swallowing disorders after cerebral infarction: Neurorehabilitation, 2022; 50(1); 115-22
146. Cheng IKY, Chan KMK, Wong CS, Neuronavigated high-frequency repetitive transcranial magnetic stimulation for chronic post-stroke dysphagia: A randomized controlled study: J Rehabil Med, 2017; 49(6); 475-81
147. Ünlüer N, Temuçin Ç M, Demir N, Effects of low-frequency repetitive transcranial magnetic stimulation on swallowing function and quality of life of post-stroke patients: Dysphagia, 2019; 34(3); 360-71
148. Park E, Kim MS, Chang WH, Effects of bilateral repetitive transcranial magnetic stimulation on post-stroke dysphagia: Brain Stimul, 2017; 10(1); 75-82
149. Kumar S, Wagner CW, Frayne C, Noninvasive brain stimulation may improve stroke-related dysphagia: A pilot study: Stroke, 2011; 42(4); 1035-40
150. Yang EJ, Baek SR, Shin J, Effects of transcranial direct current stimulation (tDCS) on post-stroke dysphagia: Restor Neurol Neurosci, 2012; 30(4); 303-11
151. Shigematsu T, Fujishima I, Ohno K, Transcranial direct current stimulation improves swallowing function in stroke patients: Neurorehabil Neural Repair, 2013; 27(4); 363-69
152. Ahn YH, Sohn HJ, Park JS, Effect of bihemispheric anodal transcranial direct current stimulation for dysphagia in chronic stroke patients: A randomized clinical trial: J Rehabil Med, 2017; 49(1); 30-35
153. Suntrup-Krueger S, Ringmaier C, Muhle P, Randomized trial of transcranial direct current stimulation for poststroke dysphagia: Ann Neurol, 2018; 83(2); 328-40
154. Kumar S, Marchina S, Langmore S, Fostering eating after stroke (FEASt) trial for improving post-stroke dysphagia with non-invasive brain stimulation: Sci Rep, 2022; 12(1); 9607
155. Gu SY, Chang MC, The effects of 10-Hz repetitive transcranial magnetic stimulation on depression in chronic stroke patients: Brain Stimul, 2017; 10(2); 270-74
156. Shen X, Liu M, Cheng Y, Repetitive transcranial magnetic stimulation for the treatment of post-stroke depression: A systematic review and meta-analysis of randomized controlled clinical trials: J Affect Disord, 2017; 211; 65-74
157. Li Y, Li K, Feng R, Mechanisms of repetitive transcranial magnetic stimulation on post-stroke depression: A resting-state functional magnetic resonance imaging study: Brain Topogr, 2022; 35(3); 363-74
158. Valiengo LC, Goulart AC, de Oliveira JF, Transcranial direct current stimulation for the treatment of post-stroke depression: Results from a randomised, sham-controlled, double-blinded trial: J Neurol Neurosurg Psychiatry, 2017; 88(2); 170-75
159. Liu Y, Zhang L, Zhang X, Effect of combined vagus nerve stimulation on recovery of upper extremity function in patients with stroke: A systematic review and meta-analysis: J Stroke Cerebrovasc Dis, 2022; 31(6); 106390
160. Brighina F, Bisiach E, Oliveri M, 1 Hz repetitive transcranial magnetic stimulation of the unaffected hemisphere ameliorates contralesional visuospatial neglect in humans: Neurosci Lett, 2003; 336(2); 131-33
161. Kim BR, Chun MH, Kim DY, Lee SJ, Effect of high- and low-frequency repetitive transcranial magnetic stimulation on visuospatial neglect in patients with acute stroke: A double-blind, sham-controlled trial: Arch Phys Med Rehabil, 2013; 94(5); 803-7
162. Zhang RG, Liu SX, Wang FYTreatment of unilateral neglect using repetitive transcranial magnetic stimulation (rTMS) and sensory cueing (SC) in stroke patients: Sichuan Da Xue Xue Bao Yi Xue Ban, 2017; 48(2); 309-13 [in Chinese]
163. Iwański S, Leśniak M, Polanowska K, Neuronavigated 1 Hz rTMS of the left angular gyrus combined with visuospatial therapy in post-stroke neglect: Neurorehabilitation, 2020; 46(1); 83-93
164. Ko MH, Han SH, Park SH, Improvement of visual scanning after DC brain polarization of parietal cortex in stroke patients with spatial neglect: Neurosci Lett, 2008; 448(2); 171-74
165. Sparing R, Thimm M, Hesse MD, Bidirectional alterations of interhemispheric parietal balance by non-invasive cortical stimulation: Brain, 2009; 132(Pt 11); 3011-20
166. Sunwoo H, Kim YH, Chang WH, Effects of dual transcranial direct current stimulation on post-stroke unilateral visuospatial neglect: Neurosci Lett, 2013; 554; 94-98
167. Turgut N, Miranda M, Kastrup A, tDCS combined with optokinetic drift reduces egocentric neglect in severely impaired post-acute patients: Neuropsychol Rehabil, 2018; 28(4); 515-26
168. Learmonth G, Benwell CSY, Märker G, Non-invasive brain stimulation in stroke patients (NIBS): A prospective randomized open blinded end-point (PROBE) feasibility trial using transcranial direct current stimulation (tDCS) in post-stroke hemispatial neglect: Neuropsychol Rehabil, 2021; 31(8); 1163-89
169. Kim BR, Kim DY, Chun MH, Effect of repetitive transcranial magnetic stimulation on cognition and mood in stroke patients: A double-blind, sham-controlled trial: Am J Phys Med Rehabil, 2010; 89(5); 362-68
170. Lu H, Zhang T, Wen M, Sun L, Impact of repetitive transcranial magnetic stimulation on post-stroke dysmnesia and the role of BDNF Val66Met SNP: Med Sci Monit, 2015; 21; 761-68
171. Yin M, Liu Y, Zhang L, Effects of rTMS treatment on cognitive impairment and resting-state brain activity in stroke patients: A randomized clinical trial: Front Neural Circuits, 2020; 14; 563777
172. Tsai PY, Lin WS, Tsai KT, High-frequency versus theta burst transcranial magnetic stimulation for the treatment of poststroke cognitive impairment in humans: J Psychiatry Neurosci, 2020; 45(4); 262-70
173. Li H, Ma J, Zhang J, Repetitive transcranial magnetic stimulation (rTMS) modulates thyroid hormones level and cognition in the recovery stage of stroke patients with cognitive dysfunction: Med Sci Monit, 2021; 27; e931914
174. Jo JM, Kim YH, Ko MH, Enhancing the working memory of stroke patients using tDCS: Am J Phys Med Rehabil, 2009; 88(5); 404-9
175. Kang EK, Baek MJ, Kim S, Paik NJ, Non-invasive cortical stimulation improves post-stroke attention decline: Restor Neurol Neurosci, 2009; 27(6); 645-50
176. Park SH, Koh EJ, Choi HY, Ko MH, A double-blind, sham-controlled, pilot study to assess the effects of the concomitant use of transcranial direct current stimulation with the computer assisted cognitive rehabilitation to the prefrontal cortex on cognitive functions in patients with stroke: J Korean Neurosurg Soc, 2013; 54(6); 484-88
177. Shaker HA, Sawan SAE, Fahmy EM, Effect of transcranial direct current stimulation on cognitive function in stroke patients: Egypt J Neurol Psychiatr Neurosurg, 2018; 54(1); 32
178. Wang Y, Li F, He MJ, Chen SJ, The effects and mechanisms of transcranial ultrasound stimulation combined with cognitive rehabilitation on post-stroke cognitive impairment: Neurol Sci, 2022; 43(7); 4315-23
179. Liu AF, Zhao FB, Wang J, Effects of vagus nerve stimulation on cognitive functioning in rats with cerebral ischemia reperfusion: J Transl Med, 2016; 14; 101
180. Kakuda W, Abo M, Sasanuma J, Combination protocol of low-frequency rTMS and intensive occupational therapy for post-stroke upper limb hemiparesis: A 6-year experience of more than 1700 Japanese patients: Transl Stroke Res, 2016; 7(3); 172-79
181. Chou YH, Ton That V, Chen AY, TMS-induced seizure cases stratified by population, stimulation protocol, and stimulation site: A systematic literature search: Clin Neurophysiol, 2020; 131(5); 1019-20
182. Tendler A, Roth Y, Zangen A, Rate of inadvertently induced seizures with deep repetitive transcranial magnetic stimulation: Brain Stimul, 2018; 11(6); 1410-14
183. Rossi S, Antal A, Bestmann S, Safety and recommendations for TMS use in healthy subjects and patient populations, with updates on training, ethical and regulatory issues: Expert Guidelines: Clin Neurophysiol, 2021; 132(1); 269-306
184. Gomez LJ, Yücel AC, Hernandez-Garcia L, Uncertainty quantification in transcranial magnetic stimulation via high-dimensional model representation: IEEE Trans Biomed Eng, 2015; 62(1); 361-72
185. Lu S, Jiang H, Li C, Hong B, Genetic algorithm for TMS coil position optimization in stroke treatment: Front Public Health, 2021; 9; 794167
186. Nikolin S, Huggins C, Martin D, Safety of repeated sessions of transcranial direct current stimulation: A systematic review: Brain Stimul, 2018; 11(2); 278-88
187. Chhatbar PY, Chen R, Deardorff R, Safety and tolerability of transcranial direct current stimulation to stroke patients – a phase I current escalation study: Brain Stimul, 2017; 10(3); 553-59
188. Chhatbar PY, Ramakrishnan V, Kautz S, Transcranial direct current stimulation post-stroke upper extremity motor recovery studies exhibit a dose-response relationship: Brain Stimul, 2016; 9(1); 16-26
189. Legon W, Adams S, Bansal P, A retrospective qualitative report of symptoms and safety from transcranial focused ultrasound for neuromodulation in humans: Sci Rep, 2020; 10(1); 5573
190. Legon W, Bansal P, Tyshynsky R, Transcranial focused ultrasound neuromodulation of the human primary motor cortex: Sci Rep, 2018; 8(1); 10007
191. Lee W, Lee SD, Park MY, Image-guided focused ultrasound-mediated regional brain stimulation in sheep: Ultrasound Med Biol, 2016; 42(2); 459-70
192. di Biase L, Falato E, Di Lazzaro V, Transcranial focused ultrasound (tFUS) and transcranial unfocused ultrasound (tUS) neuromodulation: From theoretical principles to stimulation practices: Front Neurol, 2019; 10; 549
193. Grazzi L, Egeo G, Calhoun AH, Non-invasive vagus nerve stimulation (nVNS) as mini-prophylaxis for menstrual/menstrually related migraine: An open-label study: Journal Headache Pain, 2016; 17(1); 91
194. Silberstein SD, Mechtler LL, Kudrow DB, Non-invasive vagus nerve stimulation for the acute treatment of cluster headache: Findings from the randomized, double-blind, sham-controlled ACT1 study: Headache, 2016; 56(8); 1317-32
195. Steyn E, Mohamed Z, Husselman C, Non-invasive vagus nerve stimulation for the treatment of acute asthma exacerbations-results from an initial case series: Int J Emer Med, 2013; 6(1); 7
196. Laqua R, Leutzow B, Wendt M, Usichenko T, Transcutaneous vagal nerve stimulation may elicit anti- and pro-nociceptive effects under experimentally-induced pain – a crossover placebo-controlled investigation: Auton Neurosci, 2014; 185; 120-22
197. Muffel T, Shih PC, Kalloch B, Differential effects of anodal and dual tDCS on sensorimotor functions in chronic hemiparetic stroke patients: Brain Stimul, 2022; 15(2); 509-22
198. Wei-Wei Z, Clinical effect of transcranial ultrasound combined with acupoint application of Traditional Chinese herb in treating acute cerebral infarction complicated with limb dysfunction: Journal of Appropriate Clinical Medication, 2017; 10(27); 5-6
199. Huihui W, Yana L, Ying X, Effects of motor imagery combined with transcranial ultrasound neuromuscular electrical stimulation on upper limb and daily living ability in patients with stroke: Modern Medicine & Health, 2020; 36(11); 1642-1644
200. Pu B, Shen J, Fang L, Clinical effect of motor imagery combined with transcranial ultrasonic neuromuscular stimulation on improving upper limb function and daily living ability of stroke patients: Guizhou Medical Journal, 2021; 45(05); 719-20
Tables
 Table 1. Summary of clinical studies on post-stroke patients using rTMS.
Table 1. Summary of clinical studies on post-stroke patients using rTMS. Table 2. Summary of clinical studies on post-stroke patients using tDCS.
Table 2. Summary of clinical studies on post-stroke patients using tDCS. Table 3. Summary of clinical studies on post-stroke patients using tACS.
Table 3. Summary of clinical studies on post-stroke patients using tACS. Table 4. Summary of clinical studies on post-stroke patients using tFUS.
Table 4. Summary of clinical studies on post-stroke patients using tFUS. Table 5. Summary of clinical studies on post-stroke patients using tVNS.
Table 5. Summary of clinical studies on post-stroke patients using tVNS. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








