26 April 2023: Clinical Research
Factors Associated with Sarcopenia in Patients with Chronic Kidney Disease: A Cross-Sectional Single-Center Study
Sibel Gulcicek1ABCDEFG*, Nurhan Seyahi2ACEFGDOI: 10.12659/MSM.939457
Med Sci Monit 2023; 29:e939457
Abstract
BACKGROUND: Sarcopenia is a recognized complication of chronic kidney disease (CKD) and increases risk of increased morbidity from cardiovascular events and mortality. This single-center cross-sectional study aimed to determine the prevalence and factors associated with sarcopenia in CKD patients.
MATERIAL AND METHODS: Patients with non-dialysis-dependent (NDD)-CKD were examined for sarcopenia by handgrip strength testing, bioelectrical impedance analysis (BIA), and 4-minute gait speed test. We divided 220 patients into 2 groups – No Probable Sarcopenia (NPS; n=120) and Probable Sarcopenia (PS; n=100) – according to muscle strength defined by handgrip strength, then into another 2 groups – No Sarcopenia (NS; n=189) and Confirmed Sarcopenia (CS; n=31) – according to muscle mass defined by BIA.
RESULTS: Mean age and prevalence of coronary heart disease were significantly higher and mean body mass index (BMI) was lower in the PS and CS groups than that of NPS and NS groups (P<0.05). Male predominance was observed in the PS group (P=0.0124). Median urea, creatinine levels, urine protein/creatinine ratio, and percent of anemic patients in the PS group were significantly higher and estimated glomerular filtration rate (eGFR), albumin, hemoglobin and hematocrit levels were lower than in the NPS group (P<0.05). Uric acid, leukocyte, and platelet concentrations were significantly lower in the CS group compared to the NS group (P<0.05). Increasing age significantly amplified risk of PS by 1.12 and of CS by 1.15 (P<0.0001) but not eGFR.
CONCLUSIONS: In patients with NDD-CKD, sarcopenia is common and is associated with risk factors of age, male gender, BMI, comorbidities, proteinuria, and anemia. Age is an associated risk factor for both PS and CS.
Keywords: muscle strength, Renal Insufficiency, Chronic, sarcopenia, Risk Factors, Humans, Cross-Sectional Studies, Hand Strength, Prevalence
Background
Chronic kidney disease (CKD) is a catabolic state associated with protein wasting and with multiple metabolic imbalances due to uremia [1]. CKD is a critical public health issue that has become an epidemic worldwide [1]. The prevalence of CKD was reported as 15.7% in Turkey, regardless of the disease stage, meaning that 1 of every 6–7 adults have kidney disease [2]. The incidence of CKD with an estimated glomerular filtration rate (eGFR) <60 ml/min increases with age [3]. Generally, CKD is associated with several adverse clinical outcomes, including cardiovascular events, kidney failure requiring renal replacement therapy, mortality, and poor quality of life for survivors [3].
Patients with CKD are at risk of loss of muscle mass due to several metabolic alterations developed by the uremic syndrome, even among non-dialysis-dependent (NDD) patients [4]. The progressive and generalized loss of muscle mass and strength results in a sarcopenia syndrome characterized by a risk of adverse outcomes, including physical disability, poor quality of life, and death [5]. Sarcopenia is determined by loss of muscle strength, muscle mass, and motor function and is associated with physiological aging, including reduced muscle mass and strength [6]. Sarcopenia is also recognized as a complication of CKD and increases the risk of increased morbidity from cardiovascular events and mortality [7]. In the European Working Group on Sarcopenia in Older People 2 (EWGSOP2) criteria, probable sarcopenia is defined as reduced muscle strength only (normal muscle mass) [5]. A sarcopenia diagnosis is confirmed by the presence of low muscle quantity or quality [5]. Severe sarcopenia is determined by the presence of low muscle strength, muscle mass, and muscle performance [5].
Studies have reported the prevalence of sarcopenia at between 4% to 63%, changing according to the samples size, the stage of CKD, and age of patients [8–12]. The prevalence of sarcopenia has been reported to be 5.9% to 15.4% in NDD-CKD patients at stages 3–5 [9]. Clinical studies indicated that sarcopenia is associated with reduction in eGFR, higher albuminuria, and stage of CKD, all of which cause poor clinical outcomes, poor prognosis, and higher mortality rates [10–12]. The etiology of CKD-related sarcopenia, which can occur early in adult life and may progress rapidly, includes the multiple metabolic and nutritional abnormalities associated with increased muscle degradation and impaired muscle regeneration, accumulation of uremic toxins, deficiency of vitamin D [13], polypharmacy, inflammation, depression, and cognitive dysfunction [14]. To the best of our knowledge, there are limited data on sarcopenia in patients with NDD-CKD and on the association of unfavorable prognosis with CKD-related sarcopenia [15]. Therefore, the present cross-sectional study from a single center aimed to investigate the prevalence of sarcopenia and evaluate factors associated with the sarcopenia in 31 patients with NDD-CKD, focusing on clinical outcomes and laboratory findings.
Material and Methods
ETHICS STATEMENT:
The study was conducted according to the principles of the Declaration of Helsinki. The study protocol was approved by the Istanbul Training and Research Hospital Clinical Research Ethics Committee (Number: 2015–626, Date: 04/03/2015), in accordance with the Committee on Publication Ethics (COPE) guidelines. Informed consent to participate in the study was obtained from all volunteer patients prior to their participation in the study.
PATIENTS SELECTION:
This single-center cross-sectional study recruited patients over the age of 18 years with a diagnosis of CKD, who were followed up regularly at least for 3-months in the nephrology clinic of Istanbul Training and Research Hospital and who were not yet on dialysis. We enrolled 220 patients in whom sarcopenia could be assessed by 3 components: muscle strength, muscle mass, and physical performance). The exclusion criteria were having hemodialysis, a diagnosis of cancer or cerebrovascular disease, or having an amputation or a heart pacemaker or bone marrow and solid organ transplantations.
EVALUATION OF KIDNEY FUNCTION:
Data on serum protein, creatinine, and urine protein/creatinine (P/C) ratio, which were measured at local level by standard methods, were obtained from patients’ records. The eGFR was calculated according to the equations determined by the Chronic Kidney Disease Epidemiological Collaboration (CKD-EPI) [16,17].
EXAMINATION FOR SARCOPENIA:
Firstly, the muscle strength of all patients was measured with the handgrip test [18,19], using a hydraulic dynamometer (JAMAR Hydraulic Hand, Model J00105, Lafayette Instrument Company, USA). Participants were encouraged to squeeze as hard as they could 3 times for each hand on the handgrip, alternating sides, with the elbow supported in a sitting position, and the maximum measurement was recorded. According to the latest revised EWGSOP2 criteria, the cut-off points for low muscle strength were determined as <27 kg for men and <16 kg for women [5]. Probable sarcopenia was defined as reduced muscle strength determined by handgrip strength testing, and all patients were divided into 2 groups – No Probable Sarcopenia (NPS) and Probable Sarcopenia (PS)—according to muscle strength. The NPS group comprised 120 patients with normal handgrip strength and the PS group comprised100 patients with low handgrip strength.
Secondly, the body composition of probable sarcopenic patients in terms of fat and fat-free mass was determined using a bioimpedance analysis. BIA indices were measured with a bioimpedance device (BIA 450 bioimpedance analyzer, Biodynamics Corp., Shoreline, WA, USA), using current with a constant frequency of 50 kHz. Measurements were made on the patient in a supine position with arms and legs slightly abducted. In the upper extremity, the proximal electrode was placed on the dorsal aspect of the wrist and the distal electrode was placed on the dorsal aspect of the third metacarpal bone. Resistance and reactance values were measured directly by placing the proximal electrode on the anterior aspect of the ankle and the distal electrode on the dorsal surface of the third metatarsal bone in the lower extremity. The appendicular skeletal muscle mass (ASM) was measured with the Sergi equation, which is a cross-validated equation for standardization specifically derived from older European populations, as recommended by the EWGSOP2 consensus [20]. According to low muscle quantity defined by EWGSOP, the cut-off point of ASM for men was <20 kg and those for women was <15 kg. The diagnosis of sarcopenia was confirmed with reduced amount of muscle mass according to the findings of BIA in 31 patients with probable sarcopenia. Then, all patients were divided into another 2 groups – No Sarcopenia (NS) and Confirmed Sarcopenia (CS) – according to the amount of muscle mass. The NS group comprised 189 patients with normal muscle mass and the CS group comprised 31 patients with low grip strength and low muscle mass.
Finally, physical performance was measured with the standard 4-minute gait speed test [5]. Physical performance was evaluated by calculating walking speed over a distance of 4 meters at a speed of <0.8 m/s.
DATA COLLECTION:
The demographic, clinical, and laboratory data of all patients were collected from the hospital recording system. The demographics included age, gender, anthropometrics (height, weight, BMI, obesity), comorbidities, smoking status, and alcohol usage. The laboratory findings included the amount of urea, creatinine, uric acid, albumin, total protein, calcium (Ca+2), phosphor (P), parathyroid hormone (PTH), 25 hydroxy vitamin D (25(OH)D)), bicarbonate (HCO3−), hemoglobin, hematocrit, platelets (PLT), white blood cells (WBC), ferritin, C-reactive protein (CRP), glucose, hemoglobin A1c (HbA1c), and ratio of eGFR and P/C ratio. Acidosis, anemia, and proteinuria were also assessed. Hypertension and diabetes were diagnosed at the time of enrollment. Hypertension was defined as systolic blood pressure ≥140 mmHg, diastolic blood pressure ≥90 mmHg, or previous treatment with antihypertensive drugs [21]. Diabetes was defined as fasting plasma glucose ≥126 mg/dl [22] or taking hypoglycemic drugs or receiving parenteral insulin therapy. The demographics, laboratory data, and kidney function test results related to chronic renal failure were compared between groups, which were determined according to the muscle strength, muscle mass, and physical performance.
STATISTICAL ANALYSIS:
The sample size was estimated by G*Power Version 3.1.9.6 considering the confirmed sarcopenia outcomes compared to the no sarcopenic patients. A statistical significance of 5% (α=0.05), an effect size (d) of 0.5, and a power test of 80% (β=0.95) were used for the confirmed sarcopenic outcome, which indicated a minimum sample of 88 patients per group, for a total of 176 patients. Therefore, a sample of 220 patients was considered for this study.
All statistical analyses were performed using GraphPad Instat. The Kolmogorov-Smirnov test was used to test the normality of quantitative variables. Normally distributed quantitative variables are expressed as mean±standard deviation (SD) and non-normally distributed quantitative variables are expressed as median (interquartile range: IQR). The continuous variables of 2 groups were analyzed by the unpaired
Results
PATIENT DEMOGRAPHICS:
General demographics of the 220 patients diagnosed with CKD are presented in Table 1 and laboratory findings are presented in Table 2. The mean age was 65.4±11.7 years and 33.6% of all patients were older than 65 years. There were 117 (53.2%) male patients and 103 female patients (46.8%). Regarding the anthropometrics, the mean body mass index (BMI) was 31.0±5.5 kg/m2, 81 patients (36.8%) were overweight (25≤ BMI <30 kg/m2) and 117 patients (53.2%) were obese (BMI ≥30 kg/m2). Current smoking was declared by 18.2% of patients, 17.7% were ex-smokers, and 4.1% were alcohol users. The most frequent comorbidities were hypertension, diabetes mellitus, and coronary heart disease (95.9%, 55%, and 20%, respectively). Other comorbidities included cerebrovascular accident (3.2%), chronic respiratory disease (2.3%), and peripheral vascular disease (1.8%). The most frequent medications used currently were calcium channel blockers (51.8%), diuretics (46.4%), and oral antidiabetics (42.3%). The median number of medications was 4 (range: 0–9) and the number of patients using multiple medications (more than 4) was 129 (Supplementary Table 1).
FINDINGS FOR SARCOPENIA:
The median muscle strength of all patients was 22 kg (range: 8–48 kg) and 100 (46%) patients showed low muscle strength according to the latest cut-off values revised by EWGSOP2 criteria, and these were assigned to the PS group and the remaining patients were assigned to the NPS group (Table 3). The mean age of patients with PS was significantly older than in the NPS group (P<0.0001); 87% of patients with PS were older than 65 years (P<0.0001) and 62% were male (P=0.0124). Mean weight and BMI of PS group were significantly lower than that of NPS patients (P=0.0021 and 0.0332, respectively). Coronary heart disease was present in35% of PS patients, while 1.7% of NPS patients had this comorbidity (P<0.0001). Use of a statin drug was reported by 31% of PS patients, while 18.3% of NPS patients used a statin (P=0.0424) (Table 3).
The median ASM of all patients as derived from BIA was 19.7 kg (range: 13.3–28.5 kg) and 31 patients (14.1%) had low ASM according to the criteria recommended by EWGSOP, and these were assigned to the CS group. According to the physical performance measured with 4-minute gait speed, 11 patients (5%) with CS showed low physical performance, which was defined as severe sarcopenia. The mean age of the CS group was significantly older than in the NS group (P<0.0001) (Table 4), and 90.3% of the CS group was older than 65 years (P=0.0045). A male predominance was also observed in the CS group, without any significant difference compared to NS patients. The height, weight, and BMI values were significantly lower in the CS group compared to the NS group (P<0.05). However, no significant difference was found in the prevalence of overweight and obese patients between the CS and NS groups (P>0.05). Coronary heart disease was present in 41.9% of CS patients, while 16.4% of NS patients had this comorbidity (P=0.0023). There were no statistically significant differences between the CS and NS groups in smoking, alcohol use, other comorbidities, and multiple medications currently used (Table 4).
LABORATORY FINDINGS:
The laboratory findings of patients compared according to the sarcopenia categories are given in Tables 3 and 4. The median urea and creatinine levels of PS group was significantly higher than those of the NPS group (P=0.0002 and 0.0006, respectively), while the median albumin concentration of PS patients was significantly lower than those of the NPS group (P<0.0001 and 0.0026, respectively). The mean eGFR was significantly lower in PS patients compared to the NPS group (P<0.0001) but this difference was not observed in CS patients compared to NS patients (P=0.201). Total protein levels did not significantly differ among the PS group, the CS group, and patients without sarcopenia. The median urine P/C ratio and PTH concentration were significantly higher in the PS group compared to the NPS group (P=0.0085 and 0.003, respectively), and this was again not detected in CS patients. The mean hemoglobin concentration and hematocrit ratio were lower in PS patients compared to NPS patients (P=0.0007 and 0.0033, respectively) and 51% of patients in the PS group were anemic (P=0.0083), but this was not observed for CS patients. However, the mean uric acid levels and median numbers of PLT and WBC were significantly lower in CS patients compared to NS patients (P=0.049, 0.037, and 0.0083, respectively). The Ca+2 and 25(OH)D concentrations of CS patients were significantly higher than those of the NS group (P=0.035 and 0.048, respectively). The proteinuria levels were higher in the PS and CS patients than those of NPS and NS patients, but the differences were not statistically significant (Tables 3 and 4).
RISK FACTORS FOR SARCOPENIA:
In the multivariate logistic regression model (Table 5), after adjusting for gender and age (≥65 years) and eGFR, probable sarcopenia was still significantly correlated with gender (odds ratio [OR]: 2.30; 95% confidence interval [CI]: 1.22–4.338; p=0.010) and age (OR: 1.124; 95% CI: 1.079–1.17; P=0.000). In particular, the risk of probable sarcopenia among males was 2.3 times higher than for females. Older age significantly increased the risk of probable sarcopenia by 1.12 but eGFR levels did not. The confirmed sarcopenia was still significantly correlated with age (OR: 1.153; 95% CI: 1.082–1.227; P= 0.000), but not with gender and eGFR level (P>0.05). In particular, older age significantly increased the risk of confirmed sarcopenia by 1.15 times (Table 5).
Discussion
The present study showed that older age, male gender, and lower BMI were associated risk factors for sarcopenia in NDD-CKD patients. Laboratory findings, including indicators for anemia and microalbuminuria, varied according to the diagnosis of sarcopenia. As eGFR decreased, the prevalence of probable sarcopenia increased. The presence of comorbidities, including coronary heart disease, and use of medications, especially statins, may affect the course of disease and the diagnosis of sarcopenia may alter the laboratory parameters, including renal function tests and hematologic parameters.
Sarcopenia has a variety of causes, most of which are related to the complex and poorly understood natural aging process [23]. A number of chronic illnesses, including CKD, that have a negative impact on the musculoskeletal system and physical activity are linked to and may even contribute to sarcopenia [15,23]. In a single-center cross-sectional study by Umakanthan et al, the prevalence of sarcopenia was 18% in maintenance hemodialysis patients from an Australian cohort, with low serum albumin and phosphate as significant risk factors [24]. There are also other reports showing a very high prevalence of sarcopenia in elderly patients with CKD [11,25], and we found that increasing age was correlated with the increasing risk of sarcopenia. Considering that most CKD patients are elderly, the evaluation and surveillance of sarcopenia should be considered in all patients.
EWGSOP2 is an algorithm that presents as follows: “find-assess-confirm-severity”. Identification of PS includes the screening questionnaire for strength and assessing muscle strength with the handgrip test of the chair stand test. The sarcopenia is confirmed by dual-energy X-ray absorptiometry (DXA) or magnetic resonance imaging (MRI) and computed tomography (CT). Lastly, the severity of sarcopenia can be identified by gait speed test, the Short physical performance battery, Timed-up and go test, and the 100-meter walk test [26]. The EWGSOP updated the original definition of sarcopenia [5], considering muscle strength as a central determinant of sarcopenia, suggesting specific tools and cut-off points for each variable defining sarcopenia, and also proposing a clinical algorithm for the identification, diagnosis, and severity assessment of sarcopenia in a stepwise manner [5]. Thus, in the present study, sarcopenia was considered probable in patients with low muscle strength, which was detected in 46% of a total 220 patients with NDD-CKD. Sarcopenia was considered to be confirmed when low muscle mass was verified (31 patients, 14.1% of all patients), and severe when low physical performance was also present (11 patients, 5% of all patients).
BIA is a widely used noninvasive and cost-effective method for estimation of ASM quantity. BIA prediction models are most applicable to the populations in which they have been originated, and the Sergi equation is based on older European populations [20]. The present study also used the BIA prediction model including Sergi equation to estimate ASM quantity in a Turkish population. Age, ethnicity, and other related differences between European populations should be considered in the clinical use of this equation. In addition, BIA measurements can also be influenced by hydration status of the patient, which was not measured in our study. However, BIA-based devices are more affordable and practical due to use of defined cut-off points for low ASM than the other techniques estimating or measuring muscle quantity including, DXA or MRI and CT [19]. However, more studies comparing these devices are required to validate prediction equations for the Turkish population.
As body composition is suggested as a biomarker for monitoring the prognosis of CKD, its measurement needs to be part of routine CKD care, although it is often indefinite in clinical settings [26]. The correlation between body composition and eGFR has previously been reported in patients with CKD [27,28]. However, eGFR is dependent on estimates using plasma creatinine, which is based on muscle mass, which carries a risk of inaccuracy when relating eGFR to muscle mass. In a previous study by Zhou et al, loss of lean body mass, especially ASM, in patients with CKD stages 3–5 was reported to be significantly related to GFR decline, and males were more prone to sarcopenia than females during kidney function decline [29]. In a cohort of 260 patients with NDD-CKD reported by Ishikawa et al, 25.0% of subjects had sarcopenia, and they found that an increased risk of sarcopenia was significantly associated with age, male gender, BMI, diabetes mellitus, and loop diuretics, which are commonly used in patients with CKD [14]. The present study found that the mean age of all sarcopenic groups was significantly older than those of the No Sarcopenia group. BMI was reported to be another risk factor for the high prevalence of sarcopenia. BMI was shown to have a positive significant relationship with GFR [29]. The mean BMI of confirmed sarcopenic patients was significantly lower than in sarcopenic patients, and BMI significantly increased the risk of confirmed sarcopenia by 1.26 times. In addition, a male predominance was observed among probable sarcopenic patients, suggesting that body composition of CKD patients was associated with sarcopenia, especially among older patients, male patients, and patients with lower BMI.
The comorbidities of NDD-CKD patients become more evident in more advanced stages of CKD [4,30]. As a consequence of the negative energy-protein balance due to deficient food intake combined with elevated protein catabolism in CKD patients exposed to the uremic milieu, a progression may be enhanced by catabolic effects of the comorbidities, including chronic diseases. Moreover, multiple medications for these diseases (polypharmacy) are common among patients with CKD [31]. Patients with CKD are frequently treated with a multi-drug regimen, usually including antihyperglycemics, antihypertensive agents, most commonly diuretics, renin–angiotensin–aldosterone system inhibitors, antihyperuricemics, statins, and vitamin D analogs [14]. In the present study, the ratio of patients with coronary heart disease increased with the diagnosis of sarcopenia. On the other hand, the most frequent currently used medications were calcium channel blockers, diuretics, and oral antidiabetics, and more than half of all patients (58.6%) have used multiple medications (more than 4). Statin drugs were previously reported to have myopathic effects, which can be potentiated by several drug interactions [31], especially among elderly people [32]. Consistent with the literature, the number of probable sarcopenic patients using statins was higher than those of patients without sarcopenic. This effect of statins on the muscle function suggests the sarcopenic effects of these drugs. Both findings suggest that the comorbidities and polypharmacy in CKD patients may be related to the increased risk of sarcopenia.
Sarcopenia is more common among patients in the most advanced stages of CKD and is significantly associated with GFR decline [29]. Reduced eGFR was reported to be correlated with insulin resistance, endothelial dysfunction, oxidative stress, and inflammation [33–36], which are also risk factors for sarcopenia [34], Studies reported a positive association between sarcopenia and lower eGFR [29,35]. One of these studies, on 148 patients at CKD stages 3–5, showed that every 1 ml/min/1.73 m2 reduction of eGFR was associated with 0.15±0.07 kg reduction in lean mass and a 0.12±0.03 kg reduction in appendicular skeletal muscle mass [30]. A systematic review and meta-analysis in patients with diabetes reported that sarcopenia was significantly associated with urinary albumin level, urinary protein level, and decreased eGFR, probably because urinary albumin level and decreased eGFR are related to insulin resistance, inflammation, oxidative stress, and vascular endothelial dysfunction [35]. In the present study, we also found a significantly lower eGFR level in probable sarcopenic patients compared to non-sarcopenic ones, suggesting that low muscle mass is associated with increased progression of disease. Detection of sarcopenia in CKD is crucial, and more longitudinal studies are needed to assess skeletal muscle mass as a predictor of eGFR decline and albuminuria in CKD patients.
An observational cohort study by An et al showed that the level, anemia, and total amount of carbon dioxide (CO2) were important related factors for sarcopenia in NDD-CKD patients [25]. In patients with moderately reduced renal function with CKD stage 3B or higher, low hemoglobin and total CO2 levels are common, but the effect of decreased renal function on sarcopenia may be greater than that of anemia and total CO2 levels. Moreover, there is an association between serum albumin levels and sarcopenia based on eGFR levels. However, a recent study has shown that proteinuria was not significantly correlated with either body composition or sarcopenia [25]. In the present study, the mean hemoglobin level of the probable sarcopenic group was significantly lower than those of patients without sarcopenic, and 51% of probable and 45.2% of confirmed sarcopenic patients were anemic, while 22.2% of patients without confirmed sarcopenic were anemic. The mean eGFR and median albumin levels of probable sarcopenic patients were significantly lower than in those without sarcopenia, but the total protein concentrations and proteinuria levels did not differ among groups. Therefore, in sarcopenic patients with CKD, the serum hemoglobin and albumin levels need to be closely and routinely monitored since the anemia and microalbuminuria may be correlated with the ASM quantity in these patients.
Sarcopenia can be prevented and managed by engaging in physical exercise, especially resistance training, which successfully slows muscle loss and increases strength [37,36], and by increasing total protein intake through food sources or dietary supplements. It is particularly suggested to consume 20–35 grams of protein per meal, since this provides an adequate amount of amino acids to enhance muscle protein synthesis and reduce age-related muscle loss [39], and also to consume 1.0 to 1.2 g/kg (body weight) of protein each day [40]. Additionally, it appears that combining resistance exercise with high-protein diets will have the greatest advantages because they have a synergistic impact [41]. Future work should concentrate on conducting more extensive research examining whether interventions like intensive dietetic education and exercise physiotherapy prescription could lower the incidence of sarcopenia in CKD patients [24].
The limitations of our single-center study are the limited number of sarcopenic patients and not comparing patients at all stages of CKD. However, many advanced patients with NDD-CKD were included, suggesting a considerable strength as a cohort study. We used an indirect measurement of muscle mass by using BIA, which is accepted as a practical method to reflect muscle mass. However, there is a paucity of data to demonstrate a good correlation with the criterion standard measurement of skeletal muscle mass by DXA. Other limitations are the lack of nutritional status and current exercise status, which affect BMI directly and may affect ASM indirectly. Moreover, Ca+2 and vitamin D concentrations of confirmed sarcopenic patients were higher than in those without sarcopenia, which may be considered as a controversial finding in the literature, but the use of supplementations including Ca+2 and vitamin D were not evaluated. Therefore, this finding may not be clinically important.
Conclusions
Our present results suggest that the prevalence sarcopenia is very high in CKD patients, and that age, male gender, BMI, comorbidities, proteinuria, and anemia are associated with diagnosis sarcopenia among NDD-CKD patients. Only age is an associated risk factor for both PS and CS. Careful consideration of the risk of sarcopenia may be clinically crucial among NDD-CKD patients. To elucidate these effects and understand the etiology of NDD-CKD-related sarcopenia, more data from observational and clinical studies are required.
Tables
Table 1. Demographics of all patients.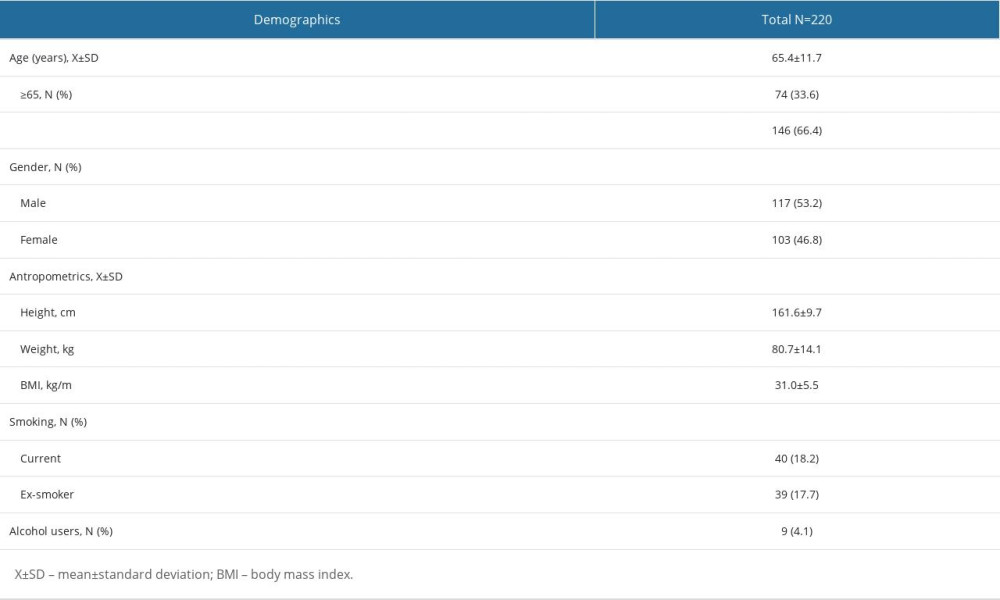 Table 2. Laboratory findings of all patients (n=220).
Table 2. Laboratory findings of all patients (n=220).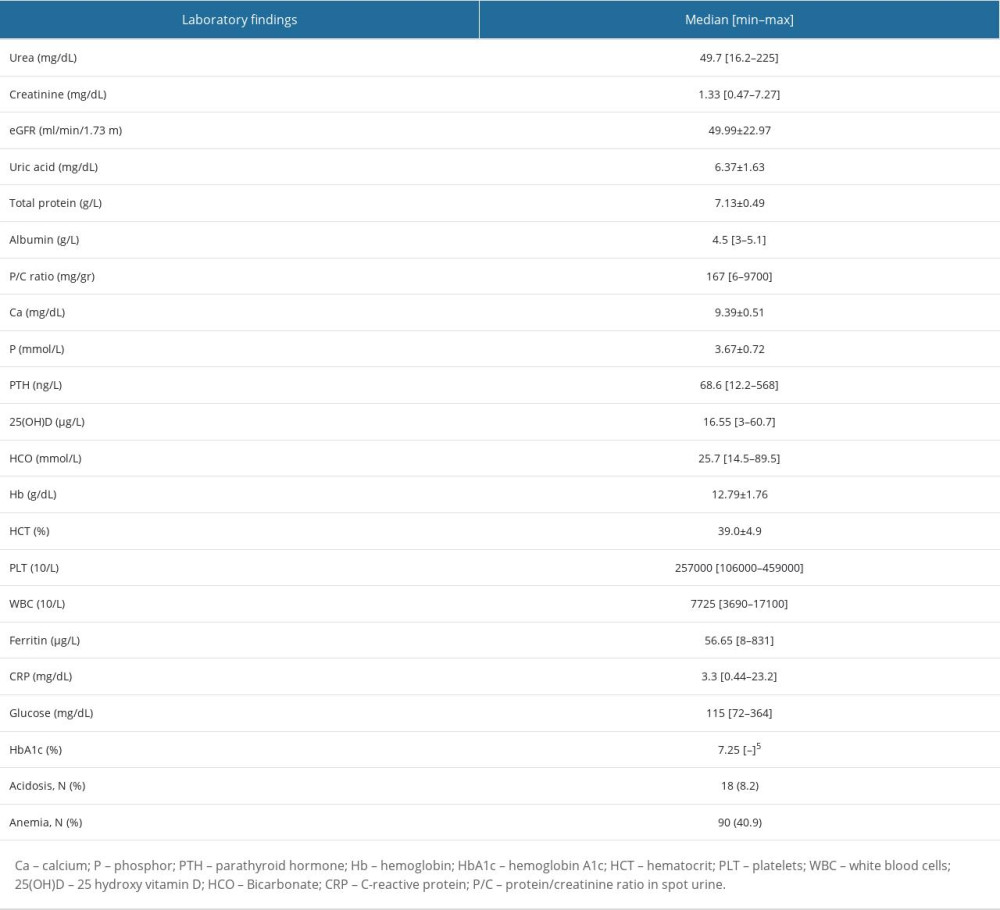 Table 3. Comparison of demographics of the patients by the presence of probable sarcopenia by handgrip strength testing.
Table 3. Comparison of demographics of the patients by the presence of probable sarcopenia by handgrip strength testing.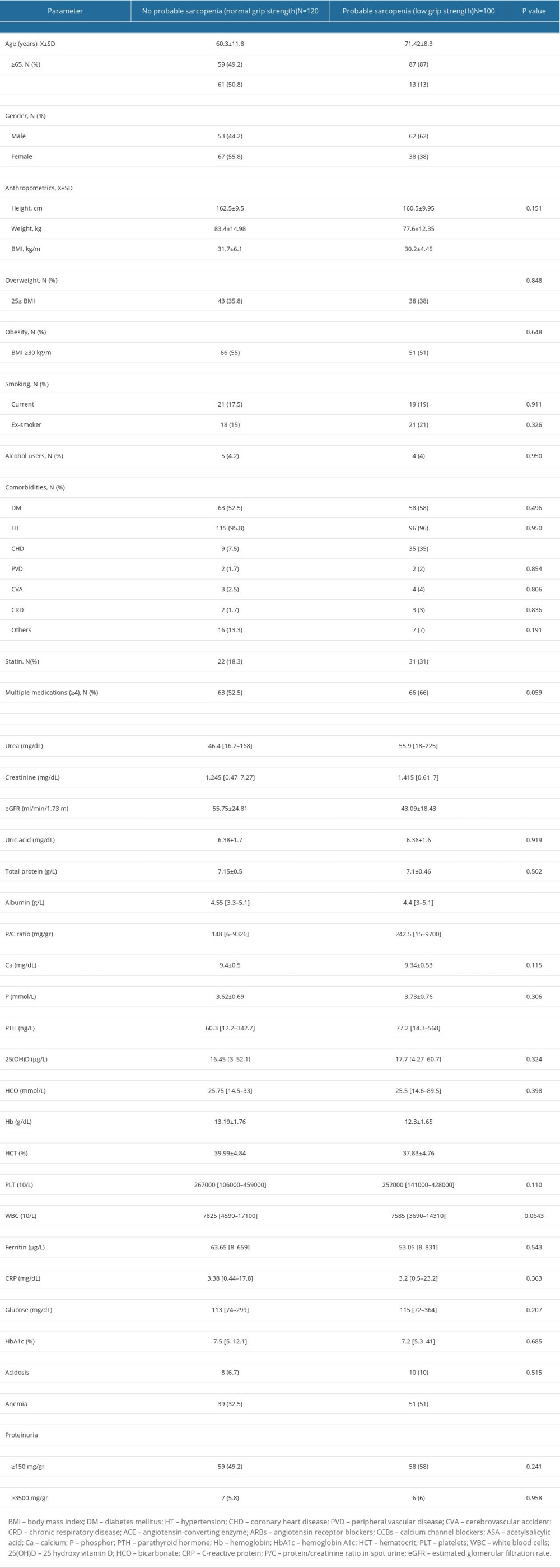 Table 4. Comparison of demographics of the patients by confirmation of sarcopenia determined by bioelectrical impedance analysis (BIA).
Table 4. Comparison of demographics of the patients by confirmation of sarcopenia determined by bioelectrical impedance analysis (BIA).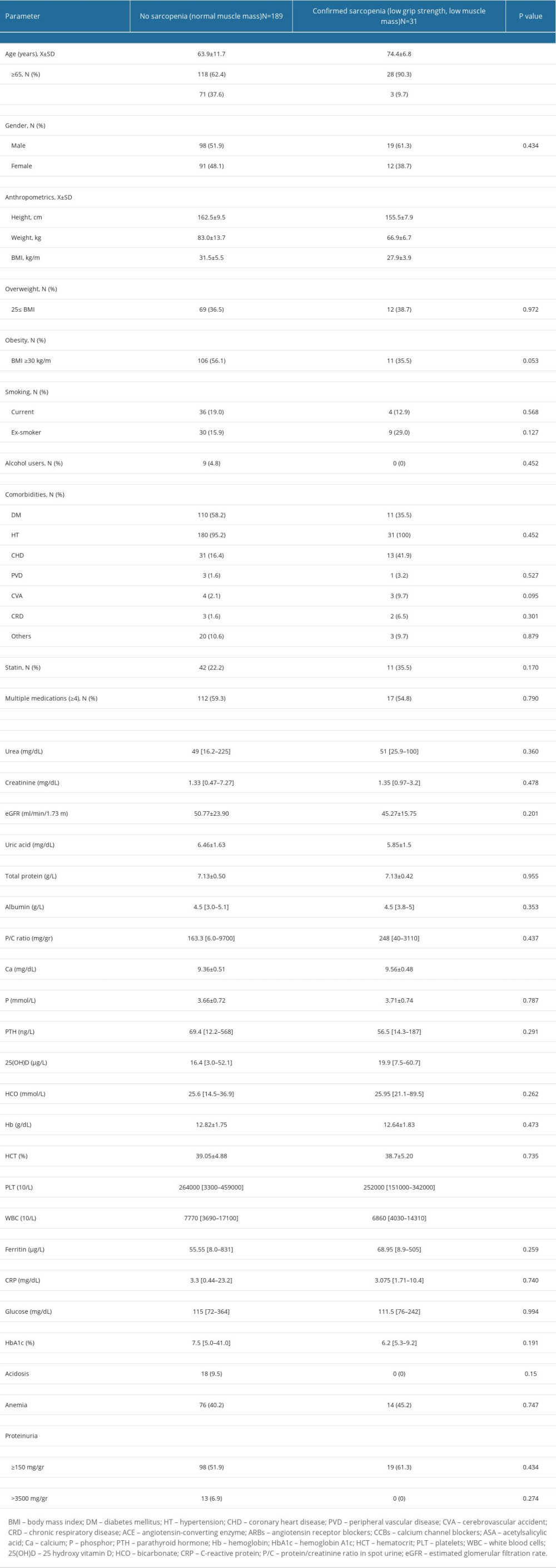 Table 5. Multivariate logistic regression analysis of associated risk factors for the sarcopenia categories.
Table 5. Multivariate logistic regression analysis of associated risk factors for the sarcopenia categories.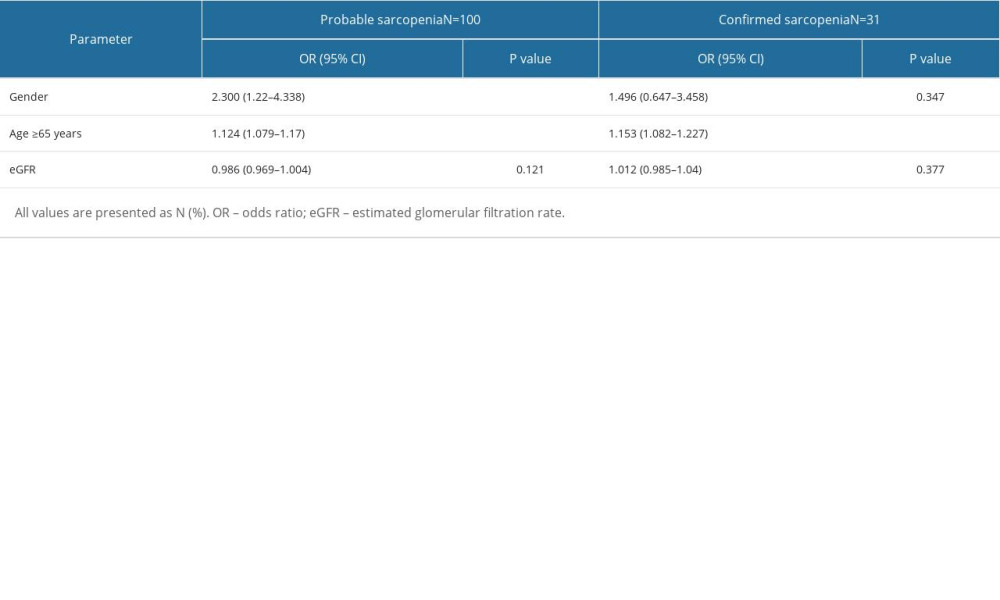 Supplementary Table 1. Distribution of current medications used by the patients.
Supplementary Table 1. Distribution of current medications used by the patients.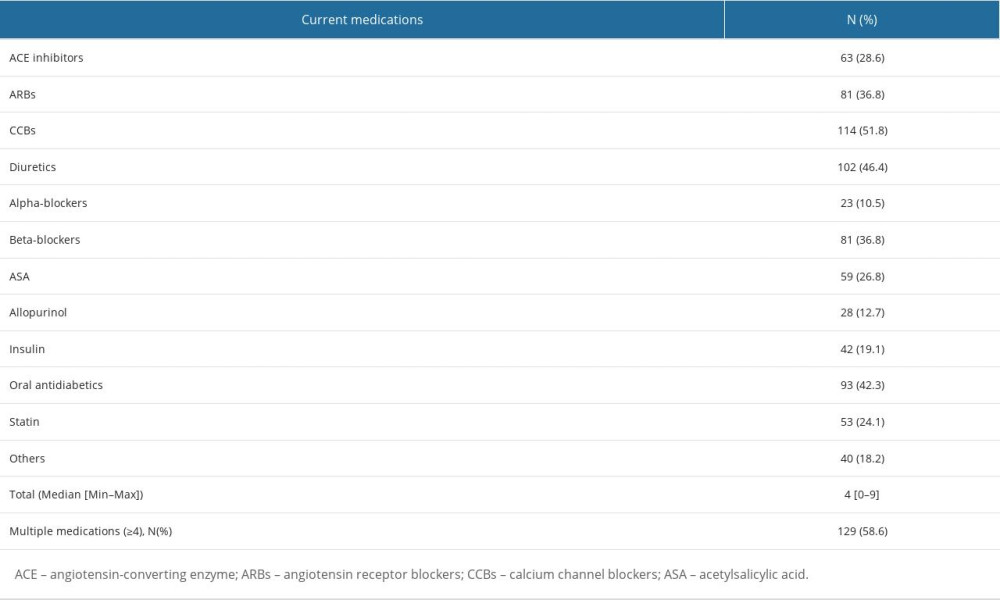
References
1. Wang XH, Mitch WE, Mechanisms of muscle wasting in chronic kidney disease: Nat Rev Nephrol, 2014; 10; 504-16
2. Süleymanlar G, Utaş C, Arinsoy T, A population-based survey of chronic renal disease in Turkey, the CREDIT study: Nephrol Dial Transplant, 2011; 26(6); 1862-71
3. Bello AK, Alrukhaimi M, Ashuntantang GE, Complications of chronic kidney disease: Current state, knowledge gaps, and strategy for action: Kidney Int Suppl, 2017; 7(2); 122-29
4. Gamboa JL, Roshanravan B, Towse T, Skeletal muscle mitochondrial dysfunction is present in patients with CKD before initiation of maintenance hemodialysis: Clin J Am Soc Nephrol, 2020; 15; 926-36
5. Cruz-Jentoft AJ, Bahat G, Bauer JWriting Group for the European Working Group on Sarcopenia in Older People 2 (EWGSOP2), and the Extended Group for EWGSOP2, Sarcopenia: revised European consensus on definition and diagnosis: Age Ageing, 2019; 48; 16-31
6. Tsekoura M, Billis E, Matzaroglou C, Prevalence of probable sarcopenia in community-dwelling older Greek people: J Frailty Sarcopenia Falls, 2021; 6(4); 204-8
7. Wathanavasin W, Banjongjit A, Avihingsanon Y, Prevalence of sarcopenia and its impact on cardiovascular events and mortality among dialysis patients: A systematic review and meta-analysis: Nutrients, 2022; 14(19); 4077
8. Lamarca F, Carrero JJ, Rodrigues JC, Prevalence of sarcopenia in elderly maintenance hemodialysis patients: the impact of different diagnostic criteria: J Nutr Health Aging, 2014; 18(7); 710-17
9. Pereira R, Cordeiro A, Avesani C, Sarcopenia in chronic kidney disease on conservative therapy: prevalence and association with mortality: Nephrol Dial Transplant, 2015; 30(10); 1718-25
10. Celik G, Oc B, Kara I, Comparison of nutritional parameters among adult and elderly hemodialysis patients: Int J Med Sci, 2011; 8(7); 628-34
11. D’Alessandro C, Piccoli GB, Barsotti M, Prevalence and correlates of sarcopenia among elderly CKD outpatients on tertiary care: Nutrients, 2018; 10(12); 1951
12. Moreno-Gonzalez R, Corbella X, Mattace-Raso FSCOPE investigators, Prevalence of sarcopenia in community-dwelling older adults using the updated EWGSOP2 definition according to kidney function and albuminuria: The Screening for CKD among Older People across Europe (SCOPE) study: BMC Geriatr, 2020; 20(Suppl 1); 327
13. Avin KG, Moorthi RN, Bone is not alone: The effects of skeletal muscle dysfunction in chronic kidney disease: Curr Osteoporos Rep, 2015; 13; 173-79
14. Ishikawa S, Naito S, Iimori S, Loop diuretics are associated with greater risk of sarcopenia in patients with non-dialysis-dependent chronic kidney disease: PLoS One, 2018; 13(2); e0192990
15. Souza VA, Oliveira D, Barbosa SR, Sarcopenia in patients with chronic kidney disease not yet on dialysis: Analysis of the prevalence and associated factors: PLoS One, 2017; 12(4); e0176230
16. Levey AS, Stevens LA, Schmid CH, CKD-EPI (chronic kidney disease epidemiology collaboration). A new equation to estimate glomerular filtration rate: Ann Intern Med, 2009; 150; 604-12
17. Stevens PE, Levin AKidney Disease: Improving Global Outcomes Chronic Kidney Disease Guideline Development Work Group Members, Evaluation and management of chronic kidney disease: Synopsis of the kidney disease: Improving global outcomes 2012 clinical practice guideline: Ann Intern Med, 2013; 158(11); 825-30
18. Roberts HC, Denison HJ, Martin HJ, A review of the measurement of grip strength in clinical and epidemiological studies: Towards a standardized approach: Age Ageing, 2011; 40; 423-29
19. Sousa-Santos AR, Barros D, Montanha TL, Which is the best alternative to estimate muscle mass for sarcopenia diagnosis when DXA is unavailable?: Arch Gerontol Geriatr, 2021; 97; 104517
20. Sergi G, De Rui M, Veronese N, Assessing appendicular skeletal muscle mass with bioelectrical impedance analysis in free-living Caucasian older adults: Clin Nutr, 2015; 34; 667-73
21. Gorostidi M, Gijón-Conde T, de la Sierra A2022 Practice guidelines for the management of arterial hypertension of the Spanish Society of Hypertension: Spanish Hipertens Riesgo Vasc, 2022; 39(4); 174-94 [in Spanish]
22. American Diabetes Association, Diagnosis and classification of diabetes mellitus: Diabetes Care, 2013; 6(Suppl1); S67-74
23. Fielding RA, Vellas B, Evans WJ, Sarcopenia: An undiagnosed condition in older adults. Current consensus definition: Prevalence, etiology, and consequences. International Working Group on Sarcopenia: J Am Med Dir Assoc, 2011; 12(4); 249-56
24. Umakanthan M, Li JW, Sud K, Prevalence and factors associated with sarcopenia in patients on maintenance dialysis in Australia – a single-centre, cross-sectional study: Nutrients, 2021; 13(9); 3284
25. An JN, Kim JK, Lee HS, Late stage 3 chronic kidney disease is an independent risk factor for sarcopenia, but not proteinuria: Sci Rep, 2021; 11; 18472
26. Ardeljan AD, Hurezeanu R, Sarcopenia [Updated 2022 Jul 4]: StatPearls [Internet], 2022, Treasure Island (FL), StatPearls Publishing Available from: https://www.ncbi.nlm.nih.gov/books/NBK560813/
27. Bellafronte NT, Vega-Piris L, Cuadrado GB, Performance of bioelectrical impedance and anthropometric predictive equations for estimation of muscle mass in chronic kidney disease patients: Front Nutr, 2021; 8; 683393
28. Sharma D, Hawkins M, Abramowitz MK, Association of sarcopenia with eGFR and misclassification of obesity in adults with CKD in the United States: Clin J Am Soc Nephrol, 2014; 9; 2079-88
29. Chew-Harris JS, Florkowski CM, Elmslie JL, Lean mass modulates glomerular filtration rate in males of normal and extreme body composition: Intern Med J, 2014; 44; 749-56
30. Zhou Y, Hellberg M, Svensson P, Sarcopenia and relationships between muscle mass, measured glomerular filtration rate and physical function in patients with chronic kidney disease stages 3–5: Nephrol Dial Transplant, 2018; 33; 342-48
31. Shahzadi A, Sonmez I, Kose C, The prevalence of potential drug-drug interactions in CKD – a retrospective observational study of Cerrahpasa Nephrology Unit: Medicina, 2022; 58(2); 183
32. Gupta R, Alcantara R, Popli T, Myopathy associated with statins and SGLT2 – a review of literature: Curr Probl Cardiol, 2021; 46(4); 100765
33. Zhu Y, Chiang CW, Wang L, A multistate transition model for statin-induced myopathy and statin discontinuation: CPT Pharmacometrics Syst Pharmacol, 2021; 10(10); 1236-44
34. Foley RN, Wang C, Ishani A, Kidney function and sarcopenia in the United States general population: NHANES III: Am J Nephrol, 2007; 27(3); 279-86
35. Ida S, Kaneko R, Imataka K, Association between sarcopenia and renal function in patients with diabetes: A systematic review and meta-analysis: J Diabetes Res, 2019; 2019; 1365189
36. Yang R, Zhang Y, Shen X, Sarcopenia associated with renal function in the patients with type 2 diabetes: Diabetes Res Clin Pract, 2016; 118; 121-29
37. Dent E, Morley JE, Cruz-Jentoft AJ, International Clinical Practice Guidelines for Sarcopenia (ICFSR): Screening, diagnosis and management: J Nutr Health Aging, 2018; 22(10); 1148-61
38. Beckwée D, Delaere A, Aelbrecht S, Exercise interventions for the prevention and treatment of sarcopenia. A systematic umbrella review: J Nutr Health Aging, 2019; 23(6); 494-502
39. Paddon-Jones D, Rasmussen BB, Dietary protein recommendations and the prevention of sarcopenia: Curr Opin Clin Nutr Metab Care, 2009; 12(1); 86-90
40. Yanai H, Nutrition for sarcopenia: J Clin Med Res, 2015; 7(12); 926-31
41. Damanti S, Azzolino D, Roncaglione C, Efficacy of nutritional interventions as stand-alone or synergistic treatments with exercise for the management of sarcopenia: Nutrients, 2019; 11(9); 1991
Tables
 Table 1. Demographics of all patients.
Table 1. Demographics of all patients. Table 2. Laboratory findings of all patients (n=220).
Table 2. Laboratory findings of all patients (n=220). Table 3. Comparison of demographics of the patients by the presence of probable sarcopenia by handgrip strength testing.
Table 3. Comparison of demographics of the patients by the presence of probable sarcopenia by handgrip strength testing. Table 4. Comparison of demographics of the patients by confirmation of sarcopenia determined by bioelectrical impedance analysis (BIA).
Table 4. Comparison of demographics of the patients by confirmation of sarcopenia determined by bioelectrical impedance analysis (BIA). Table 5. Multivariate logistic regression analysis of associated risk factors for the sarcopenia categories.
Table 5. Multivariate logistic regression analysis of associated risk factors for the sarcopenia categories. Supplementary Table 1. Distribution of current medications used by the patients.
Supplementary Table 1. Distribution of current medications used by the patients. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








