09 March 2023: Clinical Research
A Study of 57 Patients with Prostate Cancer to Compare Outcomes of Estimated Blood Loss and Postoperative Pain Between Robot-Assisted Laparoscopic Radical Prostatectomy and Standard Laparoscopic Radical Prostatectomy
Dowon Lee12F, Boo-young Hwang12A*, Jae-young Kwon2E, Ohyun Kim2B, Jeong-min Hong2D, Hyae Jin Kim2EDOI: 10.12659/MSM.939584
Med Sci Monit 2023; 29:e939584
Abstract
BACKGROUND: Robot-assisted radical prostatectomy (RARP) is becoming an increasingly common method for treatment of prostate cancer. This study aimed to compare outcomes of estimated blood loss and postoperative pain, determined by patient-controlled analgesia (PCA), between RARP and standard laparoscopic radical prostatectomy (LRP).
MATERIAL AND METHODS: We enrolled 57 patients who had localized prostate cancer (28 patients in RARP, 29 patients in LRP). Primary outcomes were estimated blood loss (EBL) measured by gravimetric method for gauze and visual estimation for suction bottle, and PCA bolus count that the bolus doses were injected at the 1st, the 6th, the 24th, and the 48th hour after the operation. We recorded anesthesia and operation time, pneumoperitoneum duration, vital signs, fluid volume, and remifentanil use. Using the numeric rating scale (NRS), adverse effects were checked at the 1st, the 6th, the 24th, and the 48th hour and patient satisfaction was assessed at the 48th hour after the operation.
RESULTS: Anesthesia time, operation time, and gas insufflation time were longer (P=0.001, P=0.003, P=0.021), and patient-controlled analgesia (PCA) bolus counts at the 1st hour after the operation and volumes of administered crystalloid and remifentanil were higher in the RARP group than in the LRP group (P=0.013, P=0.011, P=0.031). There were no significant differences in EBL.
CONCLUSIONS: The RARP group required longer anesthetic time and more analgesics during the acute postoperative period compared to the LRP group. Regarding anesthesia, LRP is as good a surgical procedure as RARP until the operation time and the number of ports are reduced.
Keywords: Analgesia, Patient-Controlled, Laparoscopy, Pain, Postoperative, Prostatectomy, Robotic Surgical Procedures, Male, Humans, Robotics, remifentanil, Treatment Outcome, Prostatic Neoplasms
Background
Prostate cancer has become one of the most commonly diagnosed cancers since the prostate-specific antigen (PSA) test method became common in the 1990s. Prostate cancer accounts for nearly 1 in 5 new cancer diagnoses. Early detection and surgery have resulted in a gradually increasing survival rate for prostate cancer patients [1]. Radical prostatectomy is the stage-matched therapeutic strategy for low- and intermediate-risk localized prostate cancer [2]. Surgical approaches to improve surgical outcomes and to reduce the rate of transfusion and estimated blood loss (EBL) have led to laparoscopic radical prostatectomy (LRP) and robot-assisted radical prostatectomy (RARP) compared to open radical prostatectomy [3,4]. Both techniques have shown fewer postoperative complications and better outcomes than traditional open radical prostatectomy [5]. RARP accounts for 70% of all radical prostatectomy in the United States, and is known to produce better functional outcomes compared to conventional radical prostatectomy [6].
There is a controversy about which operation is better between RARP and LRP. Considering the oncologic outcome, Silberstein et al have reported that the specific surgical technique is not an independent predictor of positive surgical margin (PSM) [7]. However, unlike previous results, the results of a meta-analysis assessing oncologic outcomes demonstrated a significantly lower PSM ratio for RARP compared to LRP [8]. The rate of biochemical recurrence-free survival was also significantly higher in RARP than in LRP [9]. They concluded that RARP is superior to LRP in terms of recurrence and recovery.
Although the most important value of radical prostatectomy is related to the removal of tumors associated with survival, the long-term maintenance of quality of life (QOL) is also an important issue. Urinary incontinence and erectile dysfunction have been the most common factors affecting QOL after radical prostatectomy [10]. Damages and postoperative fibrosis induced incontinence after radical prostatectomy [11]. RARP showed better outcomes in continence and sexuality compared to LRP [12]. However, urinary incontinence and erectile dysfunction are complex multifactorial conditions that require further study. Huynh et al showed that RARP has a similar result in continence as open radical prostatectomy [13].
While there are many studies about recurrence and QOL after RARP and LRP, data are lacking regarding EBL, intraoperative anesthetic factors, and postoperative analgesia. Therefore, this study of 57 patients with prostate cancer aimed to compare outcomes of EBL and postoperative pain, determined by PCA, between RARP and LRP.
Material and Methods
ETHICS STATEMENT:
This prospective randomized study was in progress between May and December 2018, at the Department of Anesthesia and Pain Medicine at Pusan National University Hospital, Korea. The study protocol was approved by the Ethics Committee of Pusan National University Hospital (PNUH IRB Approval No. 1704-001-064, CRIS registration No. KCT0002970). This study was performed in accordance with the Declaration of Helsinki. After receiving IRB certification, patients were recruited through public notice. There were no significant deviations from the treatment the patient would normally receive or unethical matters due to the experiment. Patients were informed about all the possible adverse effect of surgery, and medication and were told that they could stop whenever they wanted. All participants signed an informed consent form prior to the surgery.
PATIENT SELECTION:
This study included 60 patients with localized prostate cancer, who had American Society of Anesthesiologists (ASA) physical status grades I or II, aged 45–75 years old, and scheduled to undergo prostatectomy surgery in Pusan National University Hospital.
INCLUSION AND EXCLUSION CRITERIA:
We excluded individuals with adverse effects from morphine, nefopam, ropivacaine, oxycodone, and ramosetron. Individuals who are not able to use the numeric rating scale (NRS) or with psychological disorders, pain disorders, hepatic failure, renal failure, or preoperative administration of opioids were excluded. All patients received explanations about both surgeries, and they were divided into RARP or LRP according to their choice.
TREATMENT FOR PATIENTS IN LRP GROUP:
All patients were administered glycopyrrolate (0.004 mg/kg) by intramuscular injection, 30 min prior to the induction. All patients were prepared in a supine position, with hands and arms tucked at their side. The intravascular and arterial lines were then extended.
Anesthesia was induced with propofol (2 mg/kg) and rocuronium (0.8 mg/kg). Desflurane was titrated to maintain anesthesia with bispectral index (BIS™) monitoring (Medtronic, Boulder, CO, USA). A tidal volume was set at 7 mL per kilogram of body weight, with the respiratory rate (RR) adjusted to maintain an end-tidal CO2 (et CO2) of 35–40 mmHg, with adjustment after being placed in the Trendelenburg position. Four laparoscopic ports were inserted, and we used CO2 for pneumoperitoneum insufflation by Endoflator® (Karl Storz, Tuttlingen, Germany). Remifentanil was titrated to maintain the basal vital signs within 20%. Rocuronium (0.1 mg/kg) was injected if train of 4 (TOF) exceeded 2. Ramosetron (0.3 mg) and loading dose (5 mg oxycodone and 20 mg nefopam) were administered 30 min before the end of surgery.
TREATMENT FOR PATIENTS IN RARP GROUP:
All other preparations before surgery and anesthesia method were the same as for the patients in the LRP group. The Da Vinci Si surgical system (Intuitive Surgical®, Sunnyvale, CA, USA) was used for the operation. Especially in the RARP group, the hip was flexed to prevent femoral and obturator nerve injury, and an extra pad was added on the patient’s shoulder, as patients are prone to slipping off the operating table while in the steep Trendelenburg position [13]. We used CO2 for pneumoperitoneum insufflation with maintaining the intra-abdominal pressure (IAP) at 12–15 mmHg. Five laparoscopic ports were inserted in the abdomen. The IAP, tidal volume, and Trendelenburg angle were decreased and the RR was increased as needed in patients with severe cardiovascular and respiratory complications such as hypotension and hypercarbia.
EVALUATION OF EBL: EBL was measured by gravimetric method for blood absorbed by gauze and by visual estimation for blood collected in the suction bottle. We calculated the amount of blood absorbed by weighing the gauze after surgery and subtracting the gauze weight before surgery. In addition, after surgery, the scale of the suction bottle was checked and the amount of blood collected was calculated by subtracting the volume of the solution used during surgery [14].
EVALUATION OF POSTOPERATIVE PAIN: A patient-controlled analgesia (PCA) device (GemStar® Infusion System, Hospira, IL, USA) was used for pain control. Patient-controlled intravenous analgesia (PCIA) was set to administer a bolus of 1 mL (oxycodone 1 mg and nefopam 1 mg), lockout interval of 6 min, and 4-hour limit of 40 mL according to PCIA protocol [15]. After the operation, a mixture of pyridostigmine 10 mg and glycopyrrolate 0.4 mg was administered intravascularly in all patients, and they were transferred to the post-anesthesia care unit (PACU). Patients with Aldrete scores of 9 to 10 can be discharged from the PACU [16]. If the NRS was 5 or more, 50 μg of fentanyl was administered. We checked NRS, PCA bolus count, and adjuvant drugs at 1, 6, 24, and 48 hours after the operation. The PCA count was checked by the actual numbers of times a bolus dose was injected, not the number of times the device was pressed.
ASSESSMENT FOR SECONDARY OUTCOMES:
Vital signs were monitored every 5 minutes upon their arrival in the operating room. A BIS (XP version 4.1; Aspect Medical Systems, Newton, MA, USA) and TOF monitoring were used during the operation. Desflurane was titrated to target a BIS level of 40–60. We injected rocuronium bolus (0.1 mg/kg body weight) when 2 to 3 responses to TOF were present. We checked gas insufflation pressure, peak airway pressure (Ppeak), end-tidal CO2 (ETCO2), RR, and vital signs at intubation time (T1), incision time (T2), gas insufflation time (T3), anastomosis time (T4), and gas deflation time (T5). We recorded pneumoperitoneum duration, fluid volume, ephedrine use, administered remifentanil dose, and rocuronium bolus injection during the operation. Postoperative transfusion, length of PACU stay, and adverse effects were recorded at 1, 6, 24, and 48 hours, and patients were asked to rank their satisfaction at 48 hours after the operation according to the following scale: 1=very unsatisfied, 2=unsatisfied, 3=neutral, 4=satisfied, and 5=very satisfied. The duration of hospital stay was recorded before discharge.
STATISTICAL ANALYSIS:
The estimated sample size needed was 27 patients per group with 80% power to detect a difference in EBL (480 mL vs 380 mL) after the operation at 0.05 alpha level, with a standard deviation (SD) of 130.0 for each group in the preliminary test. We enrolled 60, assuming a 10% exclusion rate. Normally distributed data were analyzed using independent
Results
PARTICIPANTS AND DEMOGRAPHIC DATA:
Two patients declined to participate in the study and one patient in the LRP group discontinued treatment due to severe nausea, so there were 29 patients in the RARP group and 28 patients in the LRP. The groups showed no differences in demographic data, including weight, height, adjustable body weight (ABW), age, history of motion sickness or PONV, and smoking history (Table 1).
ANESTHETIC OUTCOMES:
The anesthesia time and operation time of the RARP group were 157.76±20.20 min, 131.38±20.31, and those of the LRP group were 138.75±17.14 min, 115.40±16.39 min (Table 2. P=0.001, P=0.003). The pneumoperitoneum duration was 102.10±18.25 min in the RARP group, and 90.57±15.62 min in the LRP group (Table 2, P=0.021). The volume of administered crystalloid was 1263.79±401.98 ml in the RARP group and 982.14±417.27 ml in the LRP group (Table 2, P=0.011). The administered dose of remifentanil was 531.43±155.61 mcg in the RARP group, and 420.00±182.62 in the LRP group (Table 2, P=0.031). EBL was not different between the 2 groups (P=0.169). There were no significant differences in gas insufflation pressure, administered colloid volume, ephedrine use, rocuronium bolus count, incidence of postoperative transfusion, length of stay in the PACU, or duration of hospital stay between groups (Table 2). There was no significant difference in Ppeak, RR, heart rate, diastolic blood pressure, mean blood pressure, saturation, BIS, or TOF between the groups. The systolic blood pressure at T4 and T5 in the RARP group were lower than those in the LRP group (P<0.05).
ANALGESIC OUTCOMES:
There were no significant differences in NRS scores between the groups (Table 3). The PCA bolus count at 1 hour after the operation was 3.66±2.21 times in the RARP group and 2.29±1.65 times in the LRP groups (P=0.013). There was no difference in PCA bolus count between the both groups from 6 to 48 hours (Table 3). There were not significantly differences in adverse effects, including nausea, vomiting, headache, shivering, pruritus, delirium, and respiratory depression, during the administration of PCA (Table 4). There was also no significant difference in patients’ satisfaction between groups (Table 5).
Discussion
In this study, we found that there were no significant differences in EBL and analgesic factors except pain in the 1st hour after the operation. When we designed this experiment, robotic surgery was expected to have a lower EBL and faster recovery. However, we observed no advantage in EBL, PACU stay, and hospital stay. Rather, the anesthesia and operation times were prolonged and the gas insufflation time increased, resulting in increased administered fluid volume and remifentanil dose.
Many studies suggested better surgical and functional outcomes for RARP compared to LRP. Lee et al suggested that RARP showed better outcomes in terms of functional, oncologic and perioperative complications [17]. Some studies concluded that RARP induced better functional outcomes and less transfusion, but no difference in oncologic outcome [18–20]. These studies assessed and compared lymph node involvement, cancer size, degree, and life pattern-related economic conditions, all of which are factors that may affect continence and recurrence.
Most studies have focused on functional and oncologic outcomes after the operation. When investigating intraoperative information, RARP is known to induce less EBL and has a lower transfusion rate [17–19]. Our experimental outcomes differed from these previous experimental results. There was also no case of massive bleeding requiring blood transfusion in either group in this study.
However, Rozet et al demonstrated that the surgical outcome, including EBL, recovery, and oncologic outcome of LRP, were equivalent to those of RARP when performed by skilled urological surgeons [21]. A study showed that there was no significant difference in functional outcome and patients’ QOL between RARP and LRP [22]. These studies emphasized that RARP requires the surgeon’s expertise, experience, and ability to achieve overwhelmingly good results compared to LRP. RARP is known to have a short learning curve, but it seems that it will take some time for surgeons to get used to it [13]. In our experiment, LRP and RARP also did not show a difference in EBL, and the operation time of RARP was longer.
Carbonara et al found that RARP showed better functional and oncologic outcomes up to 12 months after the operation, but high cost remained a concern [23]. There is also an opinion that the cost-effectiveness of the surgical outcomes after RARP should be judged more strictly [24]. The total cost of RARP was 2-fold higher than that of LRP in South Korea. However, upon closer examination of the costs, the surgical cost of RARP was 14,253 (USD) and LRP was 4,073, a 3-fold difference [25]. In our institution (Pusan National University Hospital, South Korea), RARP costs 8 to 12 million won after surgery, compared to 2,374,320 won for LRP. Thus, RARP is actually 4–5 times more expensive than LRP. RARP is more expensive to perform than LRP; thus, its use is limited to certain patients. Since this difference in economic ability is directly related to the living environment or health care after surgery, the type of surgery can bring about a bias that affects the postoperative progress.
After the operation, while we observed no increase in adverse effects in the RARP group, these patients required more analgesics to maintain similar effects. In our medical center, RARP requires 5 ports for robotic arms compared to 4 ports for LRP. Most patients reported having pain after the surgical procedure, primarily incisional pain, which likely contributed to the need for more analgesics in the RARP group. Postoperative pain and incision size are important factors to consider when choosing a surgical method. This study evaluating the incidence of postoperative pain is meaningful in inducing the evolution of surgical methods.
Single-port RARP has been used for pain reduction and cosmesis more than multi-port surgery. A surgeon who is proficient and familiar with da Vinci Si can perform surgery using the single-port da Vinci SP without significant change in EBL and operation time [26,27].
This study has limitations. First, since we compared only the data of a single surgeon and a single medical center, we think that the surgeon’s proficiency or surgical experience may have influenced the results. Therefore, it is necessary to conduct a multicenter study so that more diverse surgeons and patient groups can be investigated. And we have to compare both groups according to the surgeon’s proficiency and experience and the patients’ financial resources. Second, our follow-up period was short because we focused on perioperative information and postoperative analgesia. It would be better to study the chronic pain or complications with longer follow-up periods.
Conclusions
The superiority of RARP over LRP is controversial. Some authors suggest the need for more data, outcomes, and experiences of urologists. Moreover, rather than surgical method alone, robust surgeon-level outcome measures are needed. Although RARP has been known to have many advantages in terms of functional and oncologic outcomes, it takes longer than conventional surgery and increases the pain in the acute stage because it requires more incisions for ports. Reducing the operation time with simple docking and manipulation might compensate for these results, and cut costs as well, and RARP may eventually completely replace LRP. Until then, LRP might be also a good method for prostate cancer management.
Tables
Table 1. Demographic data.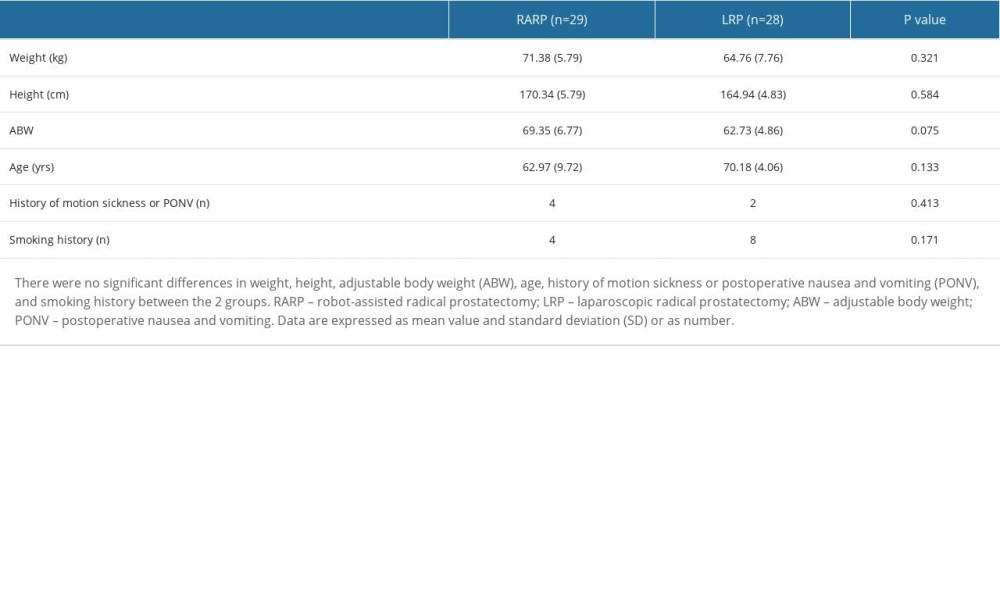 Table 2. Anesthetic data.
Table 2. Anesthetic data.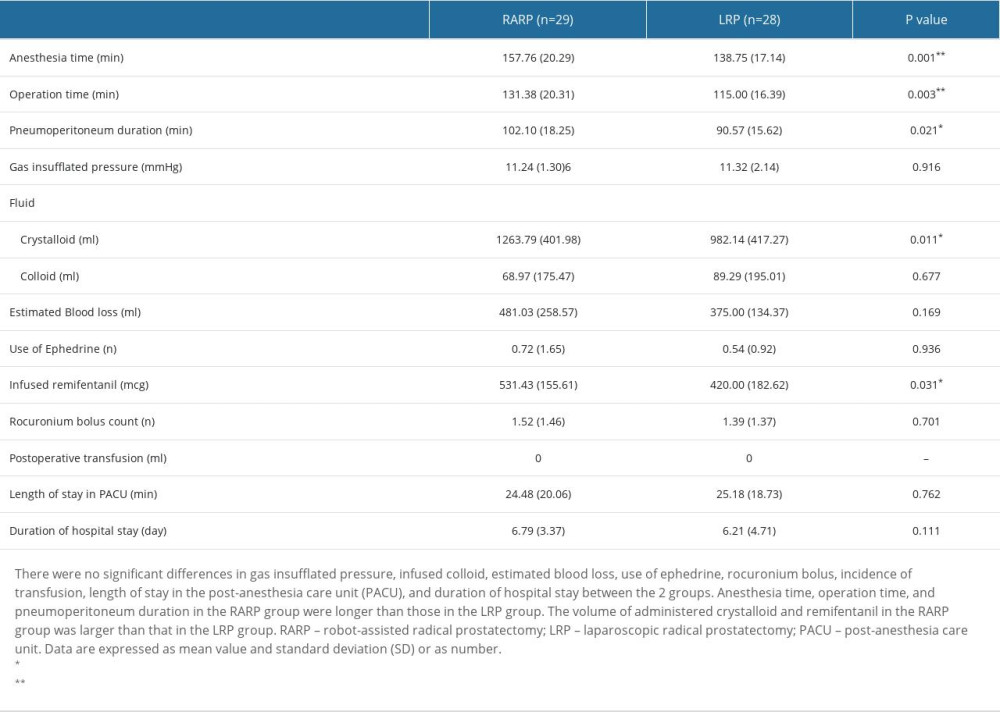 Table 3. Numeric rating scale of pain intensity and patient-controlled analgesia bolus counts at the 1st, the 6th, the 24th, and the 48th hour after the end of the operation.
Table 3. Numeric rating scale of pain intensity and patient-controlled analgesia bolus counts at the 1st, the 6th, the 24th, and the 48th hour after the end of the operation. Table 4. Adverse effects of patient-controlled analgesia.
Table 4. Adverse effects of patient-controlled analgesia.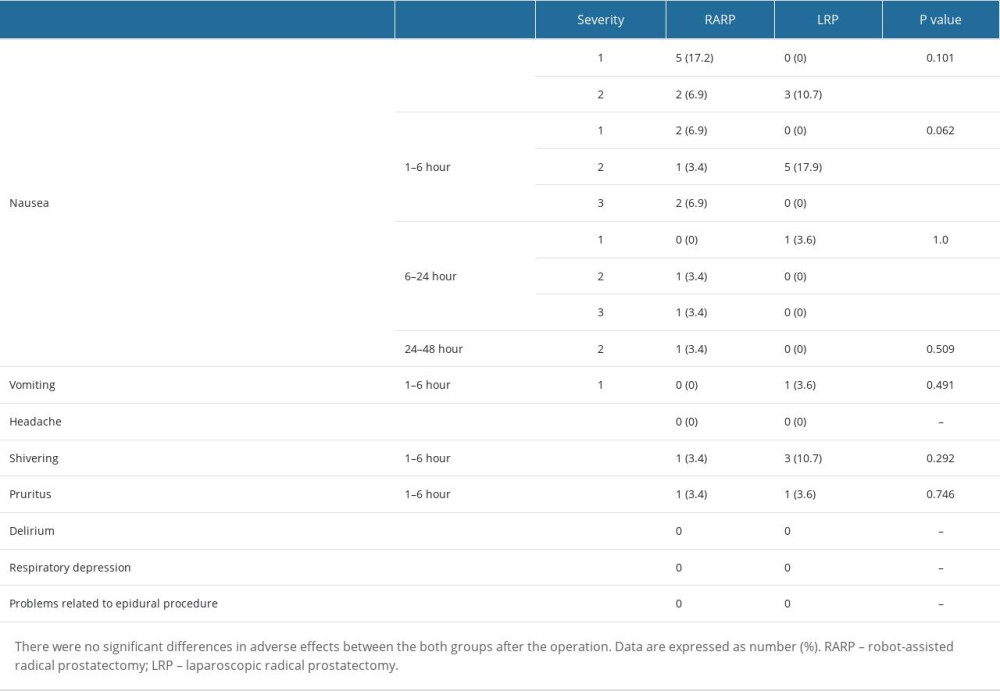 Table 5. Patient satisfaction with patient-controlled analgesia.
Table 5. Patient satisfaction with patient-controlled analgesia.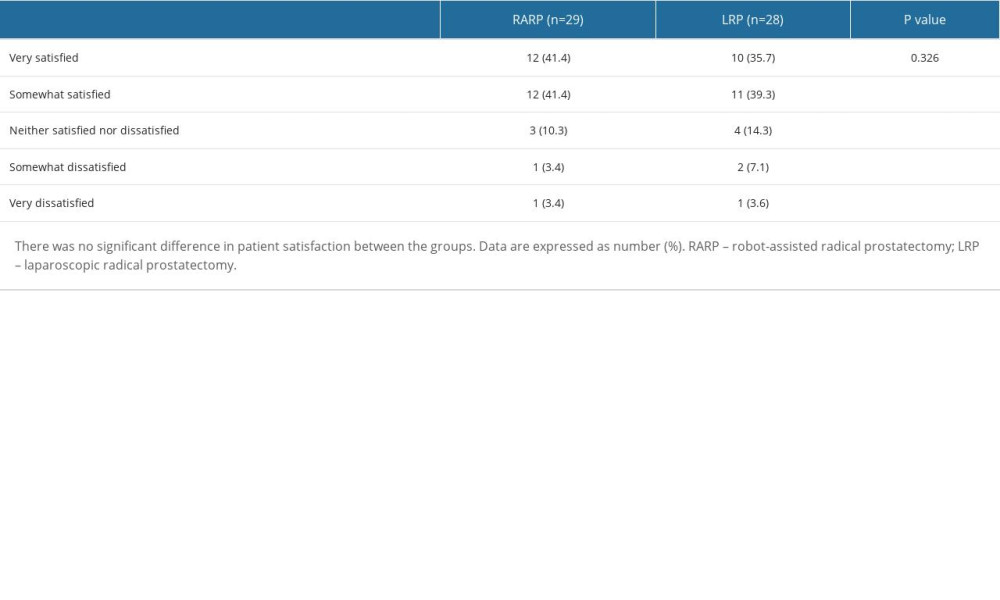
References
1. Siegel RL, Miller KD, Jemal A, Cancer statistics, 2019: Cancer J Clin, 2019; 69; 7-34
2. Parker C, Castro E, Fizazi K, Prostate cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up: Ann Oncol, 2020; 31; 1119-34
3. Cao L, Yang Z, Qi L, Robot-assisted and laparoscopic vs open radical prostatectomy in clinically localized prostate cancer: Perioperative, functional, and oncological outcomes: A Systematic review and meta-analysis: Medicine (Baltimore), 2019; 98; e15770
4. Ilic D, Evans SM, Allan CA, Laparoscopic and robotic-assisted versus open radical prostatectomy for the treatment of localised prostate cancer: Cochrane Database Syst Rev, 2017; 9; CD009625
5. Sood A, Jeong W, Peabody JO, Robot-assisted radical prostatectomy: Inching toward gold standard: Urol Clin North Am, 2014; 41; 473-84
6. Gandaglia G, Montorsi F, Karakiewicz PI, Robot-assisted radical prostatectomy in prostate cancer: Future Oncol, 2015; 11; 2767-73
7. Silberstein JL, Su D, Glickman L, A case-mix-adjusted comparison of early oncological outcomes of open and robotic prostatectomy performed by experienced high volume surgeons: BJU Int, 2013; 111; 206-12
8. Du Y, Long Q, Guan B, Robot-assisted radical prostatectomy is more beneficial for prostate cancer patients: A system review and meta-analysis: Med Sci Monit, 2018; 24; 272-87
9. Okegawa T, Omura S, Samejima M, Laparoscopic radical prostatectomy versus robot-assisted radical prostatectomy: Comparison of oncological outcomes at a single center: Prostate Int, 2020; 8; 16-21
10. Popiolek A, Brzoszczyk B, Jarzemski P, Quality of life of prostate cancer patients undergoing prostatectomy and affective temperament: Cancer Manag Res, 2022; 14; 1743-55
11. Heesakkers J, Farag F, Bauer RM, Pathophysiology and contributing factors in postprostatectomy incontinence: A review: Eur Urol, 2017; 71; 936-44
12. Long JA, Poinas G, Fiard G, Robot assisted radical prostatectomy: What are the evidences at the time of a specific funding?: Prog Urol, 2017; 27; 146-57
13. Huynh LM, Ahlering TE, Robot-assisted radical prostatectomy: A step-by-step guide: J Endourol, 2018; 32; S28-S32
14. Tran A, Heuser J, Ramsay T, Techniques for blood loss estimation in major non-cardiac surgery: A systematic review and meta-analysis: Can J Anaesth, 2021; 68; 245-55
15. Hwang BY, Kwon JY, Lee DW, A randomized clinical trial of nefopam versus ketorolac combined with oxycodone in patient-controlled analgesia after gynecologic surgery: Int J Med Sci, 2015; 12; 644-49
16. Robert C, Soulier A, Sciard D, Cognitive status of patients judged fit for discharge from the post-anaesthesia care unit after general anaesthesia: A randomized comparison between desflurane and propofol: BMC Anesthesiol, 2021; 21; 76
17. Lee SH, Seo HJ, Lee NR, Robot-assisted radical prostatectomy has lower biochemical recurrence than laparoscopic radical prostatectomy: Systematic review and meta-analysis: Investig Clin Urol, 2017; 58; 152-63
18. Huang X, Wang L, Zheng X, Comparison of perioperative, functional, and oncologic outcomes between standard laparoscopic and robotic-assisted radical prostatectomy: A systemic review and meta-analysis: Surg Endosc, 2017; 31; 1045-60
19. Yun JE, Lee NR, Kwak C, Clinical outcomes and costs of robotic surgery in prostate cancer: A multiinstitutional study in Korea: Prostate Int, 2019; 7; 19-24
20. Porpiglia F, Fiori C, Bertolo R, Five-year outcomes for a prospective randomised controlled trial comparing laparoscopic and robot-assisted radical prostatectomy: Eur Urol Focus, 2018; 4; 80-86
21. Rozet F, Jaffe J, Braud G, A direct comparison of robotic assisted versus pure laparoscopic radical prostatectomy: A single institution experience: J Urol, 2007; 178; 478-82
22. Nossiter J, Sujenthiran A, Charman SC, Robot-assisted radical prostatectomy vs laparoscopic and open retropubic radical prostatectomy: Functional outcomes 18 months after diagnosis from a national cohort study in England: Br J Cancer, 2018; 118; 489-94
23. Carbonara U, Srinath M, Crocerossa F, Robot-assisted radical prostatectomy versus standard laparoscopic radical prostatectomy: An evidence-based analysis of comparative outcomes: World J Urol, 2021; 39; 3721-32
24. Laviana AA, Williams SB, King ED, Robot assisted radical prostatectomy: The new standard?: Minerva Urol Nefrol, 2015; 67; 47-53
25. Yun JE, Lee NR, Kwak C, Clinical outcomes and costs of robotic surgery in prostate cancer: A multiinstitutional study in Korea: Prostate Int, 2019; 7; 19-24
26. Agarwal DK, Sharma V, Toussi A, Initial experience with da vinci single-port robot-assisted radical prostatectomies: Eur Urol, 2020; 77; 373-79
27. Rassweiler JJ, Autorino R, Klein J, Future of robotic surgery in urology: BJU Int, 2017; 120; 822-41
Tables
 Table 1. Demographic data.
Table 1. Demographic data. Table 2. Anesthetic data.
Table 2. Anesthetic data. Table 3. Numeric rating scale of pain intensity and patient-controlled analgesia bolus counts at the 1st, the 6th, the 24th, and the 48th hour after the end of the operation.
Table 3. Numeric rating scale of pain intensity and patient-controlled analgesia bolus counts at the 1st, the 6th, the 24th, and the 48th hour after the end of the operation. Table 4. Adverse effects of patient-controlled analgesia.
Table 4. Adverse effects of patient-controlled analgesia. Table 5. Patient satisfaction with patient-controlled analgesia.
Table 5. Patient satisfaction with patient-controlled analgesia. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








