17 June 2020: Database Analysis
Coexpression Analysis of the EZH2 Gene Using The Cancer Genome Atlas and Oncomine Databases Identifies Coexpressed Genes Involved in Biological Networks in Breast Cancer, Glioblastoma, and Prostate Cancer
Jin Zhu1AG, Lu Jin1CEF*, Aili Zhang2BCD, Peng Gao1DF, Guangcheng Dai1BF, Ming Xu1CF, Lijun Xu1AD, Dongrong Yang1AEGDOI: 10.12659/MSM.922346
Med Sci Monit 2020; 26:e922346
Abstract
BACKGROUND: This study aimed to perform coexpression analysis of the EZH2 gene using The Cancer Genome Atlas (TCGA) and the Oncomine databases to identify coexpressed genes involved in biological networks in breast cancer, glioblastoma, and prostate cancer, with functional analysis of the EZH2 gene in the C4-2 human prostate cancer cell line in vitro.
MATERIAL AND METHODS: Data from TCGA and Oncomine databases were analyzed to determine the expression of EZH2 and the top five coexpressed genes in breast cancer, glioblastoma, and prostate cancer and the clinical significance the coexpressed genes. Gene Ontology (GO) analysis was performed to predict the functions and pathways of EZH2 using pathway annotation. The role of EZH2 in the C4-2 human prostate cancer cell line was studied in vitro.
RESULTS: Analysis of 16 micro-arrays identified 185 genes that were coexpressed with EZH2. The top five coexpressed genes were MCM4, KIAA0101, MKI67, RRM2, and CDC25a. Increased expression of these genes and EZH2 were associated with reduced survival. Coexpressed genes were involved in biological networks associated with the cell cycle, mitosis, and DNA damage. The effects of EZH2 on prostate cancer cell was validated in vitro as knockdown of EZH2 resulted in a G2/M cell cycle arrest, increased DNA damage, and reduced colony number.
CONCLUSIONS: Coexpression analysis of EZH2 identified its role in the cell cycle, mitosis, and DNA repair. The molecular mechanisms involved in EZH2 gene expression in the cell response to DNA damage requires further study to determine whether EZH2 is a potential human cancer biomarker.
Keywords: DNA Damage, DNA Repair, gene ontology, Tumor Markers, Biological, Breast Neoplasms, Databases, Genetic, G2 Phase Cell Cycle Checkpoints, In Vitro Techniques, Ki-67 Antigen, Minichromosome Maintenance Complex Component 4, Prostatic Neoplasms, Ribonucleoside Diphosphate Reductase, cdc25 Phosphatases
Background
Several types of cancer initially respond to treatment due to improved surgical techniques and advances in medical oncology, but tumor relapse, progression, and resistance to chemotherapy and radiation therapy may occur [1]. For example, many patients with newly diagnosed prostate cancer present with advanced stage malignancy and initially respond to androgen suppression therapy [2,3]. However, within months to years, patients no longer respond to androgen suppression therapy, and the tumor progresses and metastasizes and becomes refractory to all forms of treatment, including chemotherapy and radiotherapy [4,5]. The main processes involved in survival and resistance to treatment by malignant tumors involve molecular mechanisms that increase cell proliferation, reduce apoptosis, and enhance DNA repair.
Previously published studies have shown that epigenetic mechanisms play key roles in cancer biology and that aberrant epigenetic control of DNA methylation and histone modification may result in the deregulation of tumor suppressor genes [6,7]. The enhancer of zeste homolog 2 (EZH2) gene is a known epigenetic tumor suppressor. EZH2 is a histone methyltransferase that functions by forming polycomb repressive complex 2 (PRC2) that consists of EED, SUZ12, and RBBP4 [8,9]. There is increasing evidence that EZH2 is overexpressed several types of cancer and promotes cell proliferation, invasion, and resistance to chemotherapy and radiation therapy [10,11]. Knockdown of EZH2 expression has been shown to inhibit cell proliferation, reverse drug resistance, and induce radiation sensitivity in some cancers [12,13]. The role of EZH2 in the promotion of tumor progression remains to be studied, but it is generally accepted that EZH2 serves as a transcription repressor of tumor-suppressive genes through histone H3 on lysine 27 (H3K27) methylation.
Several studies have shown that rather than repress, EZH2 can activate transcription of genes in a polycomb-independent manner and do not involve H3K27 methylation [14,15]. Knockdown EZH2 expression by RNA interference (RNAi) led to a significant decrease in G1/S-expressed cyclins. EZH2 activates transcription of c-Myc and cyclin D1 in breast cancer cells [16]. However, the question of which genes are commonly activated in cancer by EZH2 has not yet been answered.
Therefore, this study aimed to perform coexpression analysis of the EZH2 gene using The Cancer Genome Atlas (TCGA) and the Oncomine databases to identify coexpressed genes involved in biological networks in breast cancer, glioblastoma, and prostate cancer, with functional analysis of the EZH2 gene in the C4-2 human prostate cancer cell line
Material and Methods
ONCOMINE ANALYSIS AND EXTRACTION OF EZH2 COEXPRESSED GENES:
The study protocol and data analysis are shown in Figure 1. Retrieval of EZH2 coexpressed genes was performed from the published literature. The Oncomine database (http://oncomine.org) was searched using the parameters of the gene, EZH2, analysis type, and coexpression analysis. The statistical threshold was set as P-value <1E-4, fold change ≥2, and gene rank=top 1%. In each selected dataset, the first 200 coexpressed genes were recorded, with repetitive gene symbols removed. Coexpressed genes that appeared in six (37.5% out of 16) or more lists were considered as significant EZH2 coexpressed genes.
GENE ONTOLOGY (GO) AND PATHWAY ANALYSIS:
The Database for Annotation, Visualization and Integrated Discovery (DAVID) (
DETERMINATION OF THE REGULATIVE RELATIONSHIP BETWEEN EZH2 AND COEXPRESSED GENES:
The National Center for Biotechnology Information (NCBI) Gene Expression Omnibus (GEO) dataset, GDS2445, analyzed the gene expression profile of human embryonic fibroblasts depleted of the polycomb group proteins EZH2, EED, SUZ12, or BMI-1. We queried each of the EZH2 coexpressed genes in GDS2445 to acquire the expression difference between EZH2 depletion versus control human embryonic fibroblasts (
EXTRACTION OF EZH2 COEXPRESSED GENES FROM TCGA DATASETS:
The open-source software platform cBioPortal for Cancer Genomics (
KAPLAN-MEIER ANALYSIS OF PROGNOSIS:
The Kaplan-Meier plotter (
CELL CULTURE:
The C4-2 human prostate cancer cell line was purchased from American Type Culture Collection (ATCC) (Manassas, VA, USA) and maintained in Dulbecco’s modified Eagle’s medium (DMEM) (Gibco, Thermo Fisher Scientific, Waltham, MA, USA) with 10% fetal bovine serum (FBS) (Gibco, Thermo Fisher Scientific, Waltham, MA, USA), 1% streptomycin-penicillin (Invitrogen, Carlsbad, CA, USA), and 1% L-glutamine (Invitrogen, Carlsbad, CA, USA) at 37°C in a humidified atmosphere with 5% carbon dioxide.
TRANSFECTION OF SIRNA OR MIRNA MIMIC:
For transient transfection, cells at 50% confluence were transfected with chemically synthesized siRNA or miRNA mimic (Qiagen, Hilden, Germany) using Lipofectamine 3000 (Invitrogen, Carlsbad, CA, USA), according to the manufacturer’s instructions.
QUANTITATIVE REAL-TIME POLYMERASE CHAIN REACTION (QRT-PCR):
Total RNA was isolated in TRIzol reagent (Invitrogen, Carlsbad, CA, USA), and 1 μg of total RNA underwent reverse transcription using Superscript III transcriptase (Invitrogen, Carlsbad, CA, USA). Quantitative real-time polymerase chain reaction (qRT-PCR) was performed using a CFX96 system (Bio-Rad, Hercules, CA, USA) with SYBR green to determine the mRNA expression of specific genes. Expression levels were normalized against the expression of GAPDH. All experiments were performed in triplicate.
CELL CYCLE ANALYSIS:
The C4-2 human prostate cancer cells were fixed in 70% ethanol overnight at −20°C. The cells were treated with DNA staining solution containing 3.4 mM Tris-Cl (pH 7.4), propidium iodide (PI), 0.1% Triton X-100 buffer, and 100 mg/ml of RNase A. The cell cycle was then analyzed using a BD FACSCalibur flow cytometer (BD Biosciences, Franklin Lakes, NJ, USA)
SINGLE-CELL AGAROSE GEL ELECTROPHORESIS:
For agarose gel electrophoresis, C4-2 human prostate cancer cells were transfected with shEZH2 or control and cultured for 48 h. The procedure used was as previously described [17].
CELL COLONY FORMATION ASSAYS:
The C4-2 human prostate cancer cells were transfected with siEZH2 or control. They were then suspended and then plated in six-well plates at 500 cells per well. The cells were maintained in a humidified atmosphere with 5% CO2 at 37°C for 14 days. After fixation, the cells were stained using Giemsa. The colonies that were 50 μm in diameter or more were counted and photographed.
STATISTICAL ANALYSIS:
GraphPad Prism version 5.0 software (GraphPad Software, La Jolla, CA, USA) was used for data analysis. Data were presented as the mean±standard deviation (SD). Statistical analysis was performed using Student’s t-test. A P-value <0.05 was considered to be statistically significant.
Results
EZH2 COEXPRESSED GENES WERE ENRICHED IN MULTIPLE ONCOMINE HUMAN CANCER MICROARRAYS:
A total of 16 different microarrays were screened (Table 1), including 11 different human cancer tissue arrays, one mixed human cancer tissue array, and four arrays of different cancer cell lines. These indicated that EZH2 might play important roles in several types of cancer.
META-ANALYSIS IDENTIFIED 185 GENES TO BE STRONGLY COEXPRESSED WITH EZH2:
To identify the most frequently coexpressed genes of EZH2, the top 200 EZH2 coexpressed genes were identified from each of the 16 microarrays. Genes that were included in six or more arrays were considered to be significant. A total of 185 genes were found to be strongly coexpressed with EZH2 (Figure 2). Most of these genes were cell cycle-related genes, which may indicate the role of EZH2 in cancer cell proliferation.
INTEGRATIVE ANALYSIS SHOWED THAT EZH2 COEXPRESSED GENES WERE INVOLVED IN BIOLOGICAL NETWORKS ASSOCIATED WITH THE CELL CYCLE, MITOSIS, AND DNA DAMAGE:
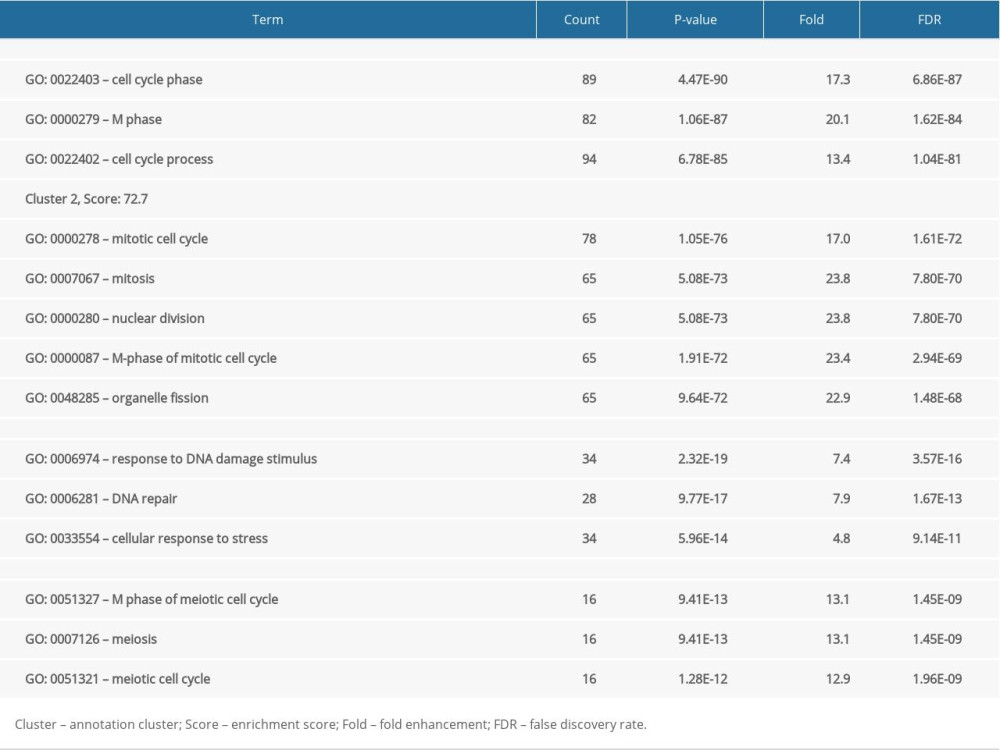
To further investigate the biological function of EZH2 and its coexpressed genes, we then performed functional enrichment analysis of the 185 genes using the DAVID functional annotation tool (GOTERM_BP_FAT) with high stringency conditions of P<0.0001, count ≥10, and fold enrichment >1.5. A total of four annotation clusters were identified that were associated with the cell cycle, mitosis, and DNA damage responses (Figure 3, Table 2). These networks were involved in cancer development, progression, and response to treatments that included chemotherapy and radiation therapy.
COEXPRESSION OF THE 185 GENES AND EZH2 COEXPRESSED GENES EXTRACTED FROM THE CANCER GENOME ATLAS (TCGA) DATASETS:
Because the 185 EZH2 coexpressed genes were all extracted from the single Oncomine database, to avoid bias, TCGA database was used to verify the coexpressed genes. From TCGA database, three cancers were randomly chosen for validation, including breast cancer, glioblastoma, and prostate cancer.
The TCGA breast cancer dataset included 65 EZH2 coexpressed genes with the criteria of a Pearson’ s correlation ≥0.60, and the majority (n=53; 81.54%) overlapped with the 185 Oncomine EZH2 coexpressed genes (Figure 4A). Similarly, 300 EZH2 coexpressed genes were identified from TCGA glioblastoma dataset with Pearson’s correlation ≥0.60 and n≤300. Further analysis showed that there were 131 overlapping genes in the 185 genes extracted from Oncomine and TCGA glioblastoma derived 300 genes, which accounted for 70.81% of the 185 genes (Figure 4B). In TCGA prostate cancer dataset, there were 166 EZH2 coexpressed genes, 109 (65.66%) of which overlapped with the 185 genes (Figure 4C).
According to the two databases based on different datasets, more than 60% of coexpressed genes overlapped. The findings shown in Figure 4A–4C indicated that the 185 genes extracted from the Oncomine database were genuinely EZH2 coexpressed genes. The results also indicated that in addition to breast cancer, ovarian cancer, and colorectal cancer, these genes also contributed to other cancers, and EZH2 and coexpressed genes might play important roles in most human cancers through this novel functional network.
THE IMPACT OF EZH2 AND COEXPRESSED GENES ON PATIENT OVERALL SURVIVAL (OS):
The Kaplan-Meier plotter database included survival data of 1,402 breast cancer patients, 1,657 ovarian cancer patients, 882 gastric cancer patients, and 1,928 lung cancer patients. The prognostic value of EZH2 and the five most frequent genes were analyzed, including MCM4, KIAA0101, MKI67, RRM2, and CDC25A, which occurred 16 times in the array, and three acknowledged tumor suppressor genes, including PTEN, RB, and TSLC1, which were used as negative controls.
The cutoff value was set as autoselect by the database for each gene. As shown in Figure 5, increased expression of EZH2, MCM4, KIAA0101, MKI67, RRM2, and CDC25A predicted worse overall survival in all the four tumors. Also, increased expression of genes that appeared 15 times in the array was associated with poor lung cancer survival. Three tumor suppressor genes, including PTEN, RB, and TSLC1 that were used as negative controls, showed an increased expression that predicted better overall survival. Therefore, EZH2 and these coexpressed genes were associated with reduced overall survival in multiple tumors. Further studies are required that use larger sample sizes to determine whether these genes may be used as prognostic biomarkers in cancer.
PROBABLE RELATIONSHIP BETWEEN EZH2 AND THE COEXPRESSED GENES:
The STRING database was used to investigate the coexpression of EZH2 with these 185 genes as the protein-protein interaction (PPI) networks were searched, as shown in Figure 6A. Based on the description of the predicted function of these genes in the STRING database and the text mining results, five possible mechanisms were identified between EZH2 and these genes (Figure 6B). In the first group, these coexpressed genes were regulated by EZH2, including CCNB2, KIAA0101, MKI67, and TOP2A. The C4-2 human prostate cancer cells were transfected with siEZH2, and expression of the genes were detected with quantitative real-time polymerase chain reaction (qRT-PCR). The results are shown in Figure 6C. The expression levels of CCNB2, MKI67, KIF20A, CCNA2, and CDC20 were significantly down-regulated after knockdown of EZH2 (EZH2, P=0.003; CCNB2, P=0.007; MKI67, P=0.003; KIF20A, P=0.002; CCNA2, P=0.005; CDC20, P=0.032).
In the second group, EZH2 and the coexpressed genes were regulated by the same gene or transcription factor (E2F1, ANCCA, and FOXM1). For example, E2F1 could regulate EZH2, MCM4, RRM2, and CDC6. ANCCA regulated EZH2, TOP2A, KIF11, and KIF15. Expression levels of related genes were detected after transfection of siE2F1, siANCCA, or siFOXM1 to C4-2 cells (Figure 6D) as follows: EZH2, P=0.008; MCM4, P=0.026; RRM2, P=0.002; DHFR, P=0.005; CCNE2, P=0.035; CDC25A, P=0.002. Figure 6E, EZH2, P=0.006; TOP2A, P=0.032; MYBL2, P<0.001; BUB1, P=0.003; CCNA2, P=0.026; CDC6, P=0.001. Figure 6F, EZH2, P=0.003; ARUKA, P<0.001; MAD2L1, P=0.006; ZNF367, P=0.005; KPNA2, P=0.002; and TPX2, P=0.005.
In the third group, EZH2 interacted with other genes and then regulated the coexpressed genes, such as HOTAIR. The expression of Hotair in C4-2 cells was knocked down with the transfection of siHOTAIR. The expression of CCNA2, CHEK1, HMMR, NUSAP1, FANCI, and NCAPG were also down-regulated (Figure 6G) as follows: CCNA2, P=0.002; CHEK1, P=0.003; HMMR, P=0.008; NUSAP1, P=0.006; FANCI, P=0.006; and NCAPG, P=0.007.
In the fourth group, EZH2 and the coexpressed genes were regulated by the same miRNA or miRNA associated network such as miR-221 and miR-101. After transfection of the miR-221 mimic, the expression of EZH2 and CDC20 were significantly upregulated (Figure 6H) as follows: EZH2, P=0.009; CDC20, P=0.002. There may have been a miRNA regulated network between miR-221 and EZH2. When cells were transfected with miR-101 mimic, EZH2, STMN, PLK1, and BUB1B were down-regulated (Figure 6I) as follows: EZH2, P=0.028; STMN, P=0.037; PLK1, P=0.006; and BUB1B, P=0.005.
THE FUNCTION OF EZH2 IN C4-2 HUMAN PROSTATE CANCER CELLS:
The cell cycle in C4-2 human prostate cancer cell was analyzed with flow cytometry after the transfection of shEZH2 or the negative control (Figure 7A, 7B). Knockdown of EZH2 resulted in G2/M cell cycle arrest (23.98% for the control vs. 28.12% for shEZH2; P=0.026) and reduction in G1/S (76.02% for the control vs. 71.88% for shEZH2). To investigate the level of DNA damage, the single-cell agarose gel electrophoresis study was performed. As shown in Figure 7C, the comet tail length of the cell transfected with shEZH2 was significantly greater than the cell in the control group (Figure 7D) (9.98±1.51 vs. 16.31±2.62; P=0.007).
The colony formation assay was also performed to assess cell proliferation and cell resistance to radiation. Cell colony number was reduced in cells transfected with shEZH2, and the colony number was further reduced when treated with radiation, which showed that shEZH2 enhanced radiotherapy sensitivity (Figure 7E, 7F) (P=0.025 for 2 Gy and P=0.005 for 6 Gy). Also, the relationship between the expression of the six genes and cancer recurrence or progression after radiation was evaluated, which showed that these genes could mediate cancer cell proliferation or DNA damage repair (Figure 7G).
Discussion
EZH2 is an oncogene that is overexpressed in several human cancers [18–20]. EZH2 acts as a transcriptional repressor, inhibiting tumor suppressor genes, such as E-CADHERIN and DAB2IP [21,22]. However, recent studies have shown that EZH2 may also function as a transcriptional activator in some cancers. For example, EZH2 is a transcription activator in castration-resistant prostate cancer and is polycomb-independent [15,23]. EZH2 can promote the expression of CCND3, CCNE2, CDK4, and CDK6 genes in nasopharyngeal carcinoma [24]. EZH2 has been shown to enhance the transactivation of c-MYC and cyclin D1 promoters in breast cancer cell lines [25]. Ectopic over-expression of EZH2 is associated with the upregulation of β-Catenin, CCND1, and EGFR in human hepatocellular carcinoma [26].
EZH2 may play dual roles in gene regulation, but the mechanisms involved in the activation of these genes by EZH2 remain unclear. EZH2 has been shown to bind to p38, and overexpression of EZH2 leads to phosphorylation and activation of p38. In castration-resistant prostate cancer, EZH2 is involved in the PI3K-Akt pathway and acts as a transcriptional coactivator of the androgen receptor. EZH2 also allows constitutive Wnt/β-catenin signaling in hepatocellular carcinoma (HCC) cells [26]. These findings prompted the investigation of genes that are positively regulated by EZH2, including in EZH2 knockout human fibroblasts [27]. However, previous studies have been cancer-specific or cell-specific, and functional studies have not been previously performed.
In this study, we used a meta-analysis method to extract the common outlier EZH2 coexpressed genes from various cancer microarrays. The majority (81%) of these genes were shown to be reduced in EZH2-knockout human fibroblasts, suggesting that EZH2 might be the driver gene in the gene coexpression network. The integrative analysis showed that the 185 EZH2 coexpressed genes were involved in biological networks related to the cell cycle, mitosis, the DNA damage response, and the p53 signaling pathway. The study findings also showed that knockdown of EZH2 in prostate cancer cells resulted in reduced colony number, G2/M cell cycle arrest, and an increased rate of DNA damage.
Previous studies showed the importance of EZH2 in cancer cell proliferation, but only a few cell cycle-related genes were identified. In this study, the majority of EZH2 coexpressed genes were cell cycle-related genes, which further emphasize the role of EZH2 in cell cycle and cell division. Therefore, EZH2 may be involved in complicated gene networks to regulate the cell cycle and cell proliferation in a controlled manner.
There have been few studies on the role of EZH2 in the cancer cell response to DNA damage, and the results are controversial. Wu et al. reported that the major response to DNA damage of EZH2-inhibited cells was enhanced apoptosis, indicating that EZH2 may play a protective role during cell stress [27]. Conversely, Zeidler et al. reported that overexpression of EZH2 impaired DNA repair in breast epithelial cells and reduced clonogenic capacity following DNA damage induced by etoposide or ionizing radiation [28]. In the current study, more than 30 DNA damage and repair related genes are coexpressed with EZH2, including the previously reported CHEK1 and RAD51 [29,30]. The findings from the present study showed that EZH2 might play critical roles in DNA damage and repair. Further studies are required to investigate the mechanisms involved.
The present study investigated the common outlier EZH2 coexpressed genes from different cancer types. The EZH2 coexpressed genes were not cell-specific or cancer-specific; they were common in several cancer types as the biologically conserved functional partner network of EZH2. Among the 185 genes, five genes, MCM4, KIAA0101, CDC25A, MKI67, and RRM2, were the most frequently coexpressed genes that appeared in 15 out of 16 microarrays. To determine the prognostic value of the five genes and EZH2, overall survival (OS) analysis was studied in different tumors (breast cancer, gastric cancer, liver cancer, and lung cancer). The results showed that increased expression of the six genes was associated with reduced survival, which indicated the genes might have a potential role as prognostic biomarkers in human malignancy.
Minichromosome maintenance-deficient 4 homolog (MCM4) encodes a subunit of the MCM2-7 complex, which is the replication licensing factor and helicase. MCM2-MCM7 were included in the 185 gene list [31]. High MCM4 expression was correlated with high EZH2 expression and acted as a prognostic factor in ovarian cancer, esophageal carcinoma, and cervical cancer [32–34].
The PCNA-associated factor of 15 KDa, p15 PAF (KIAA0101), is involved in cell proliferation. Park and colleagues showed that KIAA0101 enhances Wnt target gene transactivation by binding to β-catenin in colon cancer cells and recruiting EZH2 to the coactivator complex [35]. Cell division cycle 25A (CDC25A) is required for progression from G1 to the S phase of the cell cycle [36]. CDC25A acts as an oncogene by preventing cells with damaged DNA from cell division [37], but the relationship between CDC25A and EZH2 has not been reported. Marker of proliferation Ki-67 (Mki67) is associated with multiple cancer cell proliferation, and its overexpression is well correlated with increased EZH2 in cancer [38–40]. Ribonucleotide reductase M2 (RRM2) overexpression is found to exist in different cancers and promote cancer progression [41,42]. EZH2 and RRM2 might be putative ZBTB4 downstream target genes [43].
Conclusions
This study aimed to perform coexpression analysis of the EZH2 gene using The Cancer Genome Atlas (TCGA) and the Oncomine databases to identify coexpressed genes involved in biological networks in breast cancer, glioblastoma, and prostate cancer, with functional analysis of the EZH2 gene in the C4-2 human prostate cancer cell line
Figures
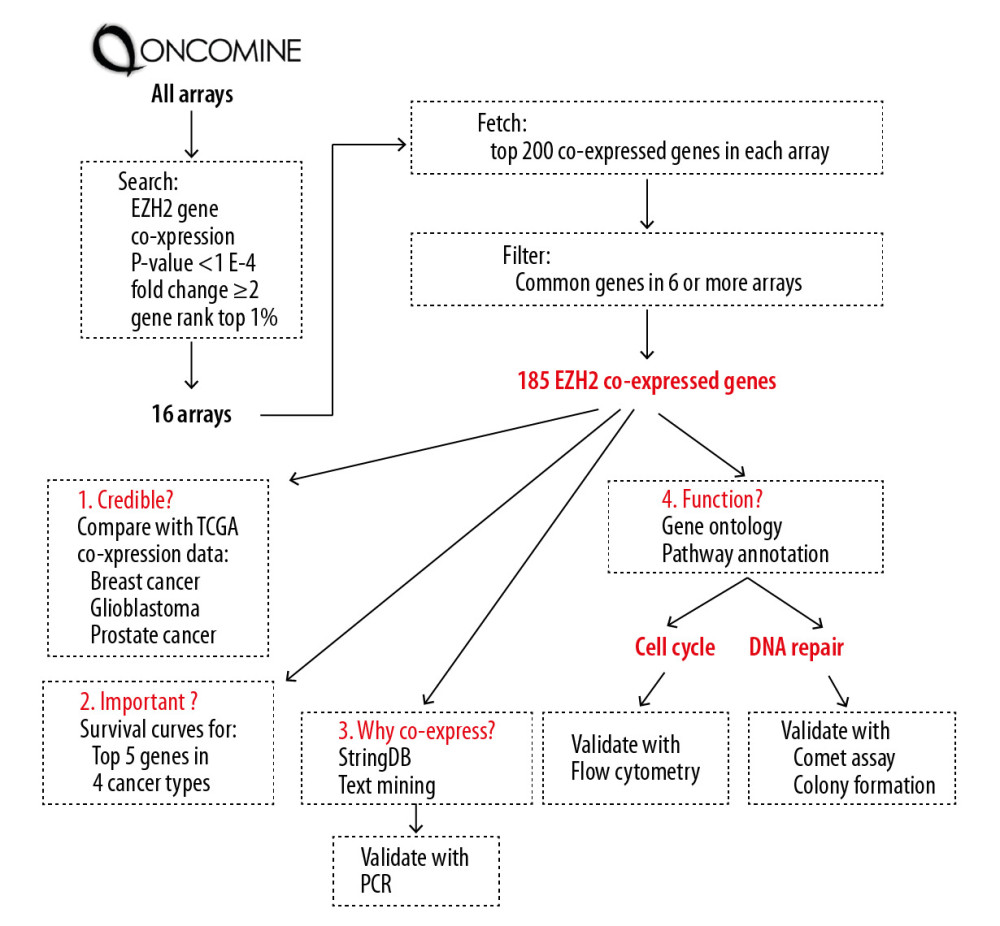 Figure 1. Flowchart of the study design.
Figure 1. Flowchart of the study design. 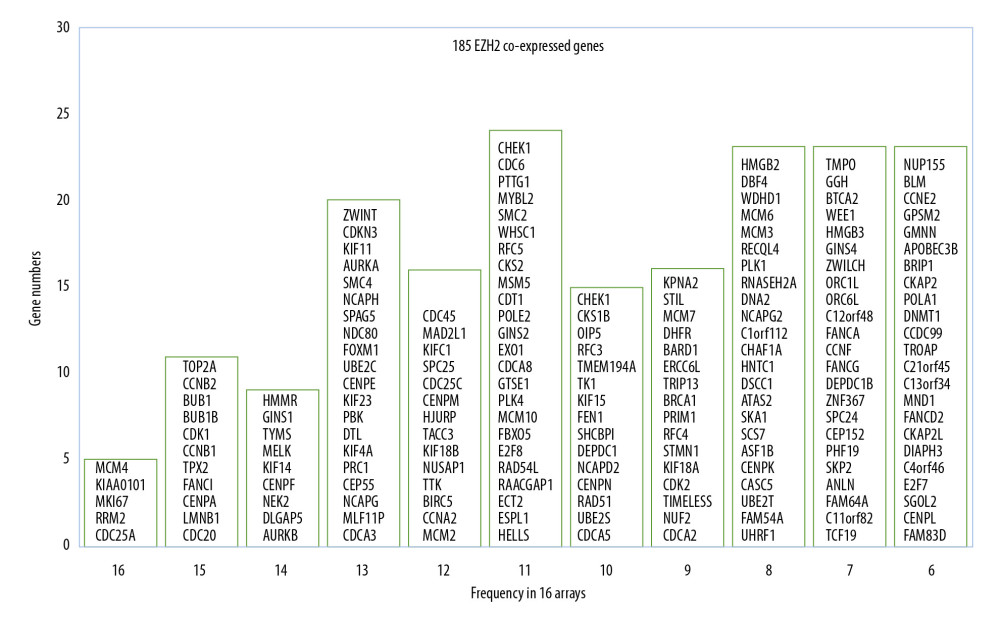 Figure 2. The 185 coexpressed genes with EZH2 in 16 arrays. The X-axis represents the frequency of the genes expressed in 16 arrays. The Y-axis represents the gene numbers.
Figure 2. The 185 coexpressed genes with EZH2 in 16 arrays. The X-axis represents the frequency of the genes expressed in 16 arrays. The Y-axis represents the gene numbers. 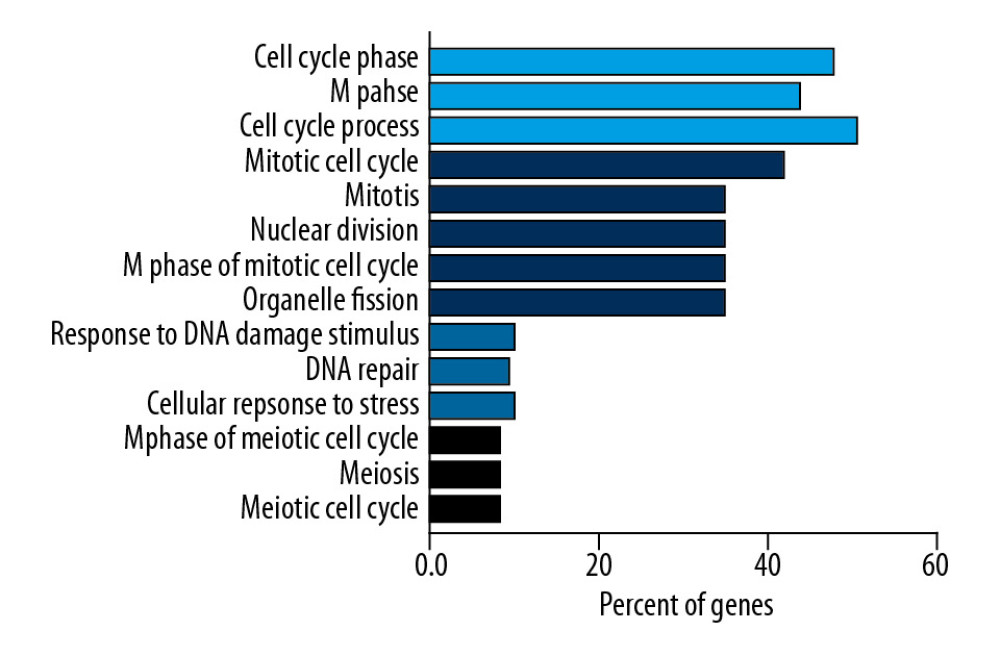 Figure 3. Functional enrichment analysis based on the DAVID functional annotation tool. The coexpressed genes are enriched in pathways related to the cell cycle, mitosis, and DNA damage.
Figure 3. Functional enrichment analysis based on the DAVID functional annotation tool. The coexpressed genes are enriched in pathways related to the cell cycle, mitosis, and DNA damage. 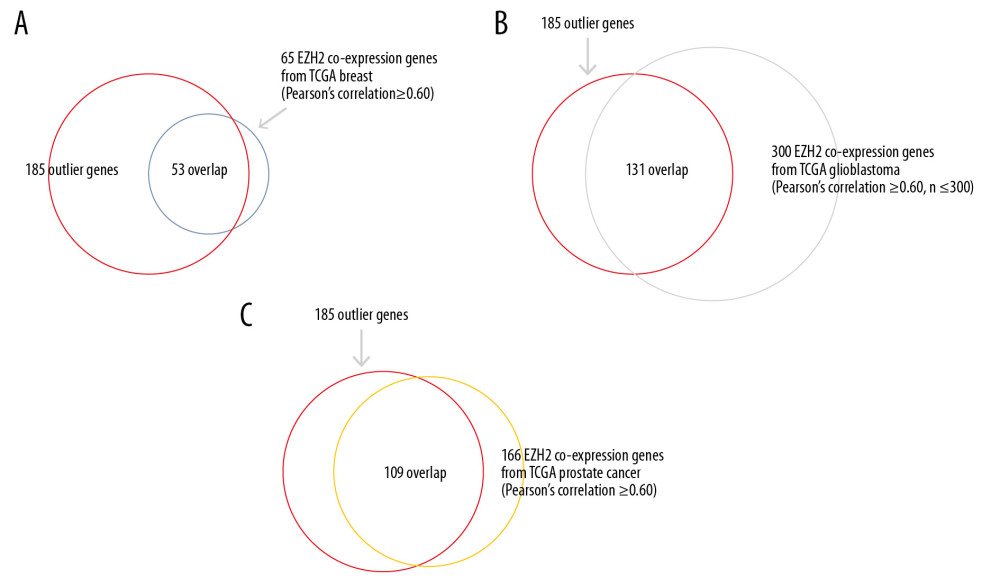 Figure 4. Coexpressed genes verified from The Cancer Genome Atlas (TCGA) database. (A) Based on TCGA database, in breast cancer, 65 EZH2 coexpressed gene were identified, and the majority (n=53, 81.54%) overlapped with the 185 Oncomine EZH2 coexpressed genes. (B) TCGA glioblastoma dataset showed 300 EZH2 coexpressed genes and there were 131 overlapping genes (43.67%). (C) In TCGA prostate cancer dataset, there were 166 EZH2 coexpressed genes, 109 (65.66%) of which overlapped with the 185 genes.
Figure 4. Coexpressed genes verified from The Cancer Genome Atlas (TCGA) database. (A) Based on TCGA database, in breast cancer, 65 EZH2 coexpressed gene were identified, and the majority (n=53, 81.54%) overlapped with the 185 Oncomine EZH2 coexpressed genes. (B) TCGA glioblastoma dataset showed 300 EZH2 coexpressed genes and there were 131 overlapping genes (43.67%). (C) In TCGA prostate cancer dataset, there were 166 EZH2 coexpressed genes, 109 (65.66%) of which overlapped with the 185 genes. 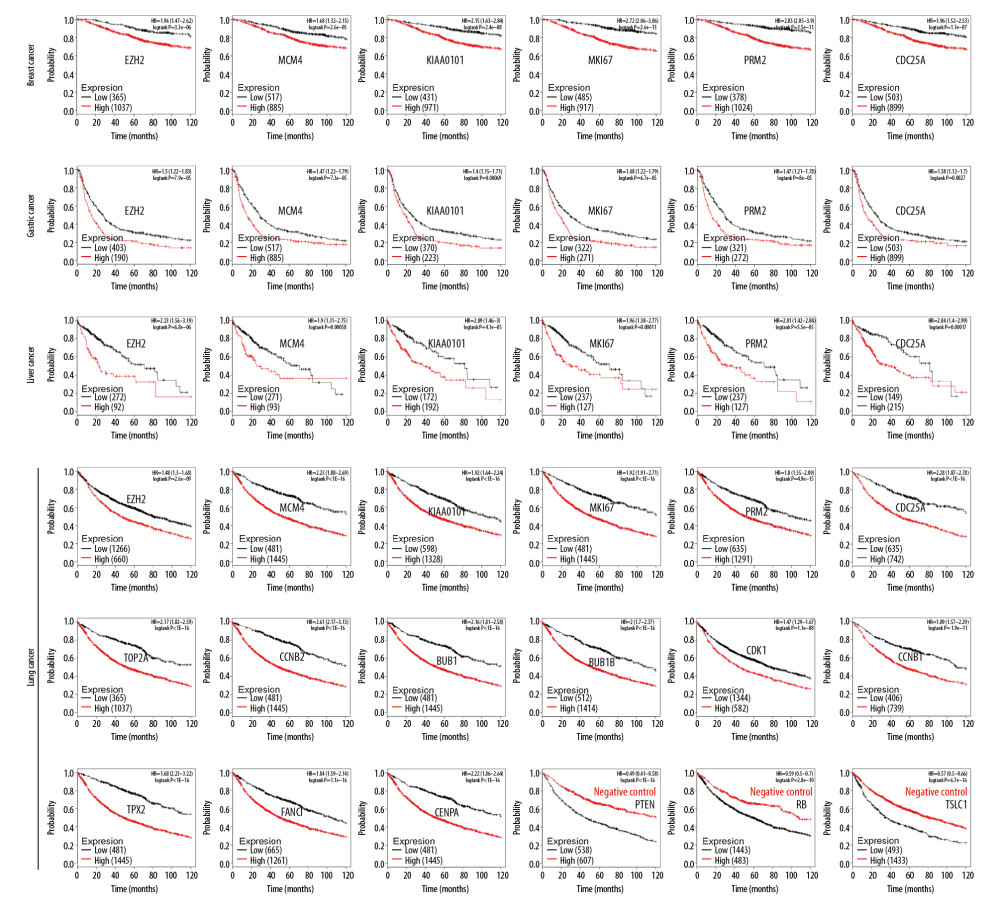 Figure 5. The impact of EZH2 and coexpressed genes on the overall survival (OS) of patients with cancer. Increased expression of EZH2 and the five most frequently expressed genes (MCM4, KIAA0101, MKI67, RRM2, and CDC25A) are associated with a lower survival rate in breast cancer, gastric cancer, liver cancer, and lung cancer. In lung cancer, the 15 most commonly expressed genes are also analyzed. Increased expression of nine genes (TOP2A, CCNB2, BUB1, BUB1B, CDK1, CCNB1, TPX2, FANCI, and CENPA) predict a lower overall survival. Tumor suppressor genes, PTEN, RB, and TSLC1, are used as negative controls.
Figure 5. The impact of EZH2 and coexpressed genes on the overall survival (OS) of patients with cancer. Increased expression of EZH2 and the five most frequently expressed genes (MCM4, KIAA0101, MKI67, RRM2, and CDC25A) are associated with a lower survival rate in breast cancer, gastric cancer, liver cancer, and lung cancer. In lung cancer, the 15 most commonly expressed genes are also analyzed. Increased expression of nine genes (TOP2A, CCNB2, BUB1, BUB1B, CDK1, CCNB1, TPX2, FANCI, and CENPA) predict a lower overall survival. Tumor suppressor genes, PTEN, RB, and TSLC1, are used as negative controls. 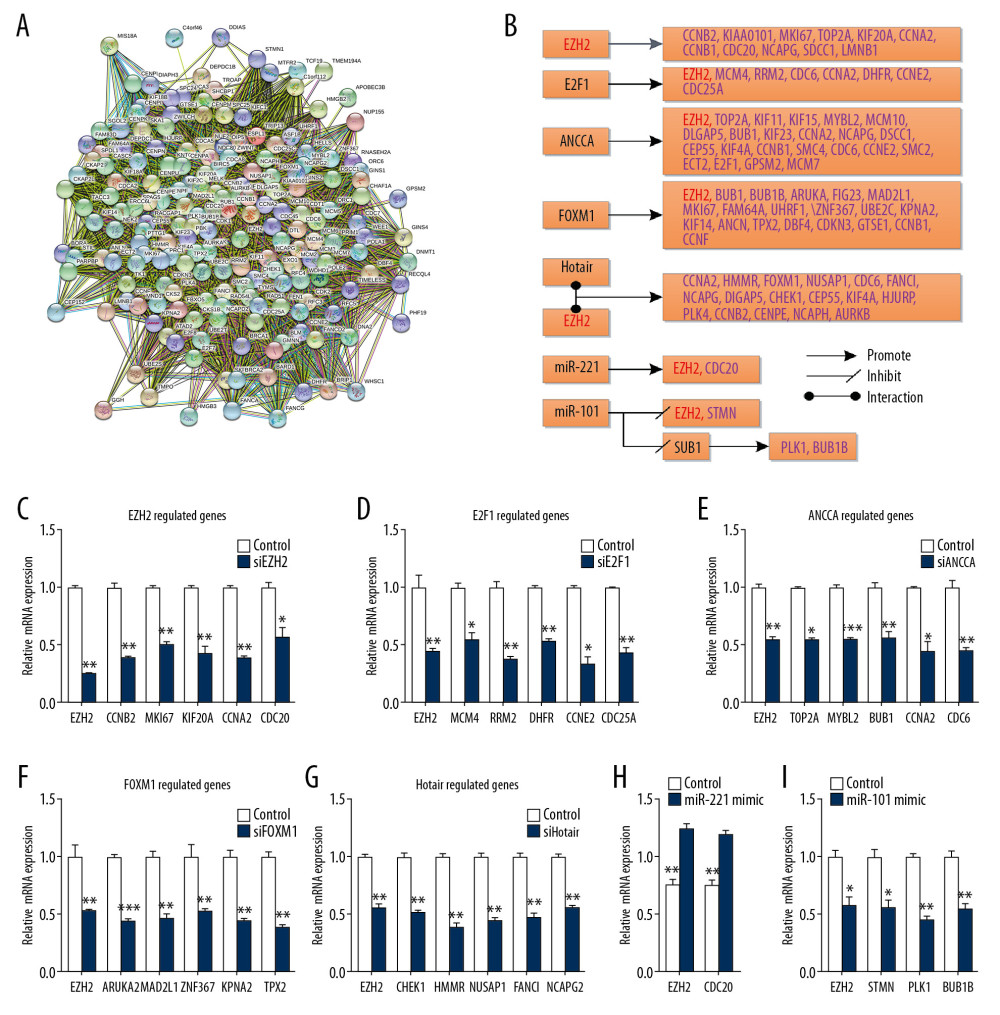 Figure 6. The relationship between EZH2 and the 185 coexpressed genes. (A) The network constructed using the STRING database. (B) The relationship between EZH2 and the 185 coexpressed genes. (C–I) Quantitative real-time polymerase chain reaction (qRT-PCR) was performed to verify the associations. (C) The quantitative real-time polymerase chain reaction (qRT-PCR) findings show that knockdown of EZH2 with si-EZH2 inhibited expression of CCNB2, MKI67, KIF20A, CCNA2, and CDC20. (D) The knockdown of E2F1 inhibited the expression of EZH2, MCM4, RRM2, DHFR, CCNE2, and CDC25A. These coexpressed genes and EZH2 are regulated by the same gene, E2F1. (E) EZH2 and the coexpressed genes (TOP2A, MYBL2, BUB1, CCNA2, and CDC6) were down-regulated when ANNCA was knocked down. (F) EZH2 and the coexpressed genes, ARUKA, MAD2L1, ZNF367, KPNA2, and TPX2 are regulated by FOXM1, and knockdown of FOXM1 deregulated these genes. (G) Knockdown of HOTAIR deregulated CCNA2, CHEK1, HMMR, NUSAP1, FANCI, and NCAPG. (H) Upregulation of miR-221 promoted the expression of EZH2 and CDC20. (I) Upregulation of miR-101 inhibited the expression of EZH2, STMN, PLK1, and BUB1B. (* P<0.05; ** P<0.01; *** P<0.001).
Figure 6. The relationship between EZH2 and the 185 coexpressed genes. (A) The network constructed using the STRING database. (B) The relationship between EZH2 and the 185 coexpressed genes. (C–I) Quantitative real-time polymerase chain reaction (qRT-PCR) was performed to verify the associations. (C) The quantitative real-time polymerase chain reaction (qRT-PCR) findings show that knockdown of EZH2 with si-EZH2 inhibited expression of CCNB2, MKI67, KIF20A, CCNA2, and CDC20. (D) The knockdown of E2F1 inhibited the expression of EZH2, MCM4, RRM2, DHFR, CCNE2, and CDC25A. These coexpressed genes and EZH2 are regulated by the same gene, E2F1. (E) EZH2 and the coexpressed genes (TOP2A, MYBL2, BUB1, CCNA2, and CDC6) were down-regulated when ANNCA was knocked down. (F) EZH2 and the coexpressed genes, ARUKA, MAD2L1, ZNF367, KPNA2, and TPX2 are regulated by FOXM1, and knockdown of FOXM1 deregulated these genes. (G) Knockdown of HOTAIR deregulated CCNA2, CHEK1, HMMR, NUSAP1, FANCI, and NCAPG. (H) Upregulation of miR-221 promoted the expression of EZH2 and CDC20. (I) Upregulation of miR-101 inhibited the expression of EZH2, STMN, PLK1, and BUB1B. (* P<0.05; ** P<0.01; *** P<0.001). 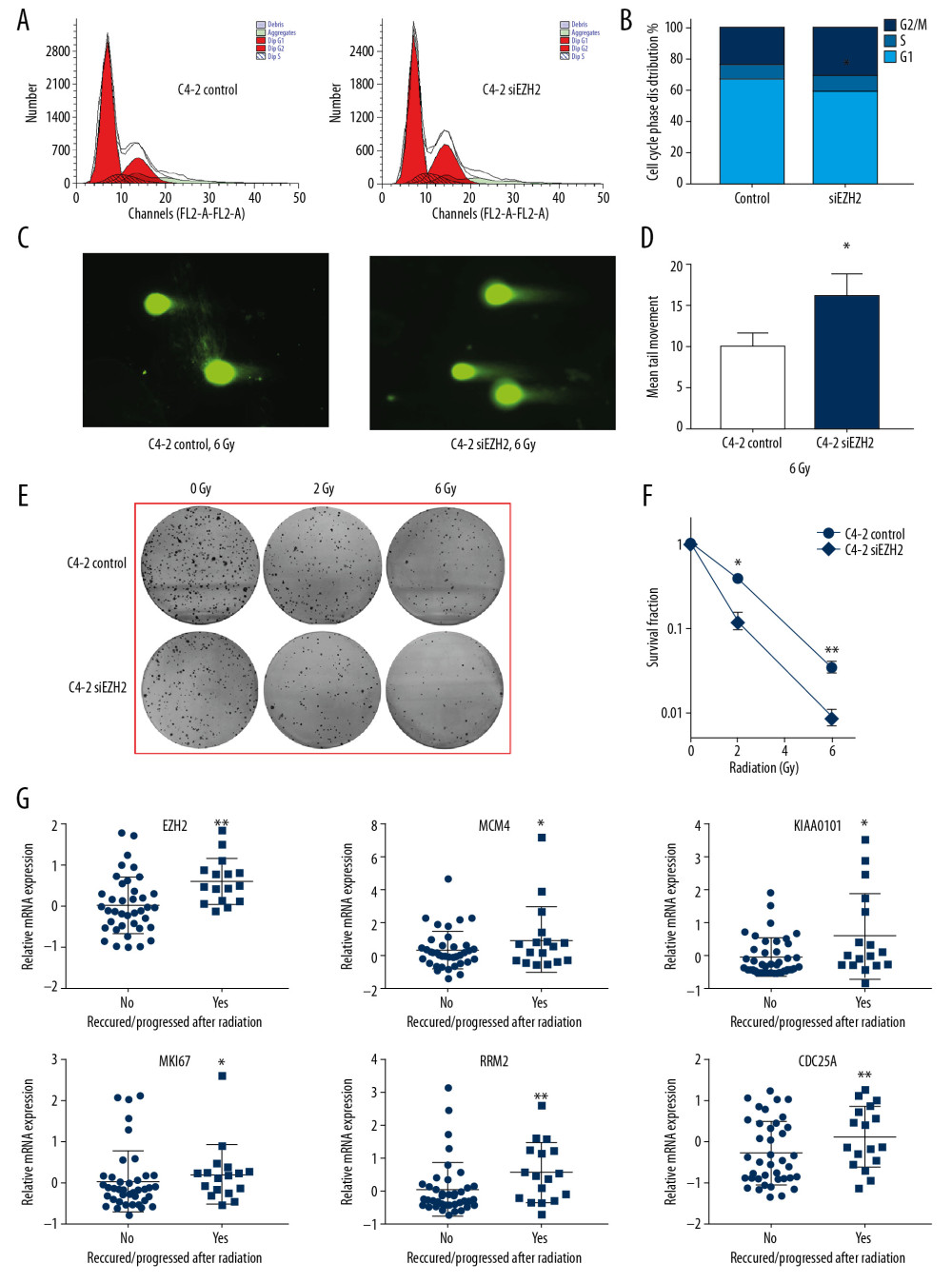 Figure 7. The effects of siEZH2 in the C4-2 human prostate cancer cell line. (A) The results of cell cycle analysis showed that knockdown of EZH2 resulted in G2/M arrest (B) 23.98% for the control vs. 28.12% for shEZH2 (P<0.05). (C) Single-cell agarose gel electrophoresis for the cell transfected with control or siEZH2. (D) The results showed the tail length of cells transfected with shEZH2 was significantly longer than the cell in the control group (9.9 8±1.51 for the control group vs. 16.31±2.6 for the siEZH2 group). (E) Colony formation assay shows that knockdown of EZH2 inhibited cell proliferation when irradiated, and the results are shown in panel F. (G) Based on The Cancer Genome Atlas (TCGA) prostate dataset, the increased expression of EZH2, MCM4, KIAA0101, MKI67, RRM2, and CDC25A were associated with recurrence or progression after radiation. (* P<0.05; ** P<0.01)
Figure 7. The effects of siEZH2 in the C4-2 human prostate cancer cell line. (A) The results of cell cycle analysis showed that knockdown of EZH2 resulted in G2/M arrest (B) 23.98% for the control vs. 28.12% for shEZH2 (P<0.05). (C) Single-cell agarose gel electrophoresis for the cell transfected with control or siEZH2. (D) The results showed the tail length of cells transfected with shEZH2 was significantly longer than the cell in the control group (9.9 8±1.51 for the control group vs. 16.31±2.6 for the siEZH2 group). (E) Colony formation assay shows that knockdown of EZH2 inhibited cell proliferation when irradiated, and the results are shown in panel F. (G) Based on The Cancer Genome Atlas (TCGA) prostate dataset, the increased expression of EZH2, MCM4, KIAA0101, MKI67, RRM2, and CDC25A were associated with recurrence or progression after radiation. (* P<0.05; ** P<0.01) References
1. De Angelis ML, Francescangeli F, La Torre F, Zeuner A, Stem cell plasticity and dormancy in the development of cancer therapy resistance: Front Oncol, 2019; 9; 626
2. Ventimiglia E, Seisen T, Abdollah F, A systematic review of the role of definitive local treatment in patients with clinically lymph node-positive prostate cancer: Eur Urol Oncol, 2019; 2; 294-301
3. Pagliarulo V, Bracarda S, Eisenberger MA, Contemporary role of androgen deprivation therapy for prostate cancer: Eur Urol, 2012; 61; 11-25
4. Khullar K, Parikh RR, The role of radiotherapy in metastatic prostate cancer: Am J Clin Exp Urol, 2019; 7; 92-97
5. Heidenreich A, Bastian PJ, Bellmunt J, EAU guidelines on prostate cancer. Part II: Treatment of advanced, relapsing, and castration-resistant prostate cancer: Eur Urol, 2014; 65; 467-79
6. Yen CY, Huang HW, Shu CW, DNA methylation, histone acetylation and methylation of epigenetic modifications as a therapeutic approach for cancers: Cancer Lett, 2016; 373; 185-92
7. Bhol CS, Panigrahi DP, Praharaj PP, Epigenetic modifications of autophagy in cancer and cancer therapeutics: Semin Cancer Biol; 2019 [Epub ahead of print]
8. Wang X, Brea LT, Yu J, Immune modulatory functions of EZH2 in the tumor microenvironment: Implications in cancer immunotherapy: Am J Clin Exp Urol, 2019; 7; 85-91
9. Tremblay-LeMay R, Rastgoo N, Pourabdollah M, Chang H, EZH2 as a therapeutic target for multiple myeloma and other haematological malignancies: Biomark Res, 2018; 6; 34
10. Zhao Y, Wang XX, Wu W, EZH2 regulates PD-L1 expression via HIF-1alpha in non-small cell lung cancer cells: Biochem Biophys Res Commun, 2019; 517(2); 201-9
11. Yan KS, Lin CY, Liao TW, EZH2 in cancer progression and potential application in cancer therapy: A friend or foe?: Int J Mol Sci, 2017; 18(6) pii: E1172
12. Li Z, Hou P, Fan D, The degradation of EZH2 mediated by lncRNA ANCR attenuated the invasion and metastasis of breast cancer: Cell Death Differ, 2017; 24; 59-71
13. Hernando H, Gelato KA, Lesche R, EZH2 inhibition blocks multiple myeloma cell growth through upregulation of epithelial tumor suppressor genes: Mol Cancer Ther, 2016; 15; 287-98
14. Xu K, Wu ZJ, Groner AC, EZH2 oncogenic activity in castration-resistant prostate cancer cells is Polycomb-independent: Science, 2012; 338; 1465-69
15. Yang YA, Yu J, EZH2, an epigenetic driver of prostate cancer: Protein Cell, 2013; 4; 331-41
16. Lee YC, Chang WW, Chen YY, Hsp90α mediates BMI1 expression in breast cancer stem/progenitor cells through facilitating nuclear translocation of c-Myc and EZH2: Int J Mol Sci, 2017; 18(9) pii: E1986
17. Tuluce Y, Lak PTA, Koyuncu I, The apoptotic, cytotoxic and genotoxic effect of novel binuclear boron-fluoride complex on endometrial cancer: Biometals, 2017; 30; 933-44
18. Wang A, Dai H, Gong Y, ANLN-induced EZH2 upregulation promotes pancreatic cancer progression by mediating miR-218-5p/LASP1 signaling axis: J Exp Clin Cancer Res, 2019; 38; 347
19. Ma J, Zhang J, Weng YC, Wang JC, EZH2-mediated microRNA-139-5p regulates epithelial-mesenchymal transition and lymph node metastasis of pancreatic cancer: Mol Cells, 2018; 41; 868-80
20. Vantaku V, Putluri V, Bader DA, Epigenetic loss of AOX1 expression via EZH2 leads to metabolic deregulations and promotes bladder cancer progression: Oncogene, 2019 [Epub ahead of print]
21. Sellers WR, Loda M, The EZH2 polycomb transcriptional repressor – a marker or mover of metastatic prostate cancer?: Cancer Cell, 2002; 2; 349-50
22. Lund K, Adams PD, Copland M, EZH2 in normal and malignant hematopoiesis: Leukemia, 2014; 28; 44-49
23. Deb G, Thakur VS, Gupta S, Multifaceted role of EZH2 in breast and prostate tumorigenesis: epigenetics and beyond: Epigenetics, 2013; 8; 464-76
24. Lu J, He ML, Wang L, MiR-26a inhibits cell growth and tumorigenesis of nasopharyngeal carcinoma through repression of EZH2: Cancer Res, 2011; 71; 225-33
25. Kumari K, Das B, Adhya A, Nicotine associated breast cancer in smokers is mediated through high level of EZH2 expression which can be reversed by methyltransferase inhibitor DZNepA: Cell Death Dis, 2018; 9; 152
26. Cheng AS, Lau SS, Chen Y, EZH2-mediated concordant repression of Wnt antagonists promotes beta-catenin-dependent hepatocarcinogenesis: Cancer Res, 2011; 71; 4028-39
27. Wu Z, Lee ST, Qiao Y, Polycomb protein EZH2 regulates cancer cell fate decision in response to DNA damage: Cell Death Differ, 2011; 18; 1771-79
28. Zeidler M, Varambally S, Cao Q, The Polycomb group protein EZH2 impairs DNA repair in breast epithelial cells: Neoplasia, 2005; 7; 1011-19
29. Abe H, Alavattam KG, Kato Y, CHEK1 coordinates DNA damage signaling and meiotic progression in the male germline of mice: Hum Mol Genet, 2018; 27; 1136-49
30. Pastushok L, Fu Y, Lin L, A novel cell-penetrating antibody fragment inhibits the DNA repair protein RAD51: Sci Rep, 2019; 9; 11227
31. Wang X, Ishimi Y, Function of the amino-terminal region of human MCM4 in helicase activity: J Biochem, 2018; 164; 449-60
32. Xie L, Li T, Yang LH, E2F2 induces MCM4, CCNE2 and WHSC1 upregulation in ovarian cancer and predicts poor overall survival: Eur Rev Med Pharmacol Sci, 2017; 21; 2150-56
33. Choy B, LaLonde A, Que J, MCM4 and MCM7, potential novel proliferation markers, significantly correlated with Ki-67, Bmi1, and cyclin E expression in esophageal adenocarcinoma, squamous cell carcinoma, and precancerous lesions: Hum Pathol, 2016; 57; 126-35
34. Gan N, Du Y, Zhang W, Zhou J, Increase of Mcm3 and Mcm4 expression in cervical squamous cell carcinomas: Eur J Gynaecol Oncol, 2010; 31; 291-94
35. Jung HY, Jun S, Lee M, PAF and EZH2 induce Wnt/beta-catenin signaling hyperactivation: Mol Cell, 2013; 52; 193-205
36. Zhang X, Neganova I, Przyborski S, A role for NANOG in G1 to S transition in human embryonic stem cells through direct binding of CDK6 and CDC25A: J Cell Biol, 2009; 184; 67-82
37. Sadeghi H, Golalipour M, Yamchi A, CDC25A pathway toward tumorigenesis: Molecular targets of CDC25A in cell-cycle regulation: J Cell Biochem, 2019; 120; 2919-28
38. Dowsett M, Nielsen TO, A’Hern R, Assessment of Ki67 in breast cancer: Recommendations from the International Ki67 in Breast Cancer working group: J Natl Cancer Inst, 2011; 103; 1656-64
39. Liu B, Pang B, Wang Q, EZH2 upregulation correlates with tumor invasiveness, proliferation, and angiogenesis in human pituitary adenomas: Hum Pathol, 2017; 66; 101-7
40. Ahmed S, Rashed H, Hegazy A, Prognostic value of ALDH1, EZH2 and Ki-67 in astrocytic gliomas: Turk Patoloji Derg, 2016; 32; 70-81
41. Chen WX, Yang LG, Xu LY, Bioinformatics analysis revealing prognostic significance of RRM2 gene in breast cancer: Biosci Rep, 2019; 39(4) pii: BSR20182062
42. Zheng S, Wang X, Weng YH, siRNA knockdown of RRM2 effectively suppressed pancreatic tumor growth alone or synergistically with doxorubicin: Mol Ther Nucleic Acids, 2018; 12; 805-16
43. Yang WS, Chadalapaka G, Cho SG, The transcriptional repressor ZBTB4 regulates EZH2 through a microRNA-ZBTB4-specificity protein signaling axis: Neoplasia, 2014; 16; 1059-69
Figures
 Figure 1. Flowchart of the study design.
Figure 1. Flowchart of the study design. Figure 2. The 185 coexpressed genes with EZH2 in 16 arrays. The X-axis represents the frequency of the genes expressed in 16 arrays. The Y-axis represents the gene numbers.
Figure 2. The 185 coexpressed genes with EZH2 in 16 arrays. The X-axis represents the frequency of the genes expressed in 16 arrays. The Y-axis represents the gene numbers. Figure 3. Functional enrichment analysis based on the DAVID functional annotation tool. The coexpressed genes are enriched in pathways related to the cell cycle, mitosis, and DNA damage.
Figure 3. Functional enrichment analysis based on the DAVID functional annotation tool. The coexpressed genes are enriched in pathways related to the cell cycle, mitosis, and DNA damage. Figure 4. Coexpressed genes verified from The Cancer Genome Atlas (TCGA) database. (A) Based on TCGA database, in breast cancer, 65 EZH2 coexpressed gene were identified, and the majority (n=53, 81.54%) overlapped with the 185 Oncomine EZH2 coexpressed genes. (B) TCGA glioblastoma dataset showed 300 EZH2 coexpressed genes and there were 131 overlapping genes (43.67%). (C) In TCGA prostate cancer dataset, there were 166 EZH2 coexpressed genes, 109 (65.66%) of which overlapped with the 185 genes.
Figure 4. Coexpressed genes verified from The Cancer Genome Atlas (TCGA) database. (A) Based on TCGA database, in breast cancer, 65 EZH2 coexpressed gene were identified, and the majority (n=53, 81.54%) overlapped with the 185 Oncomine EZH2 coexpressed genes. (B) TCGA glioblastoma dataset showed 300 EZH2 coexpressed genes and there were 131 overlapping genes (43.67%). (C) In TCGA prostate cancer dataset, there were 166 EZH2 coexpressed genes, 109 (65.66%) of which overlapped with the 185 genes. Figure 5. The impact of EZH2 and coexpressed genes on the overall survival (OS) of patients with cancer. Increased expression of EZH2 and the five most frequently expressed genes (MCM4, KIAA0101, MKI67, RRM2, and CDC25A) are associated with a lower survival rate in breast cancer, gastric cancer, liver cancer, and lung cancer. In lung cancer, the 15 most commonly expressed genes are also analyzed. Increased expression of nine genes (TOP2A, CCNB2, BUB1, BUB1B, CDK1, CCNB1, TPX2, FANCI, and CENPA) predict a lower overall survival. Tumor suppressor genes, PTEN, RB, and TSLC1, are used as negative controls.
Figure 5. The impact of EZH2 and coexpressed genes on the overall survival (OS) of patients with cancer. Increased expression of EZH2 and the five most frequently expressed genes (MCM4, KIAA0101, MKI67, RRM2, and CDC25A) are associated with a lower survival rate in breast cancer, gastric cancer, liver cancer, and lung cancer. In lung cancer, the 15 most commonly expressed genes are also analyzed. Increased expression of nine genes (TOP2A, CCNB2, BUB1, BUB1B, CDK1, CCNB1, TPX2, FANCI, and CENPA) predict a lower overall survival. Tumor suppressor genes, PTEN, RB, and TSLC1, are used as negative controls. Figure 6. The relationship between EZH2 and the 185 coexpressed genes. (A) The network constructed using the STRING database. (B) The relationship between EZH2 and the 185 coexpressed genes. (C–I) Quantitative real-time polymerase chain reaction (qRT-PCR) was performed to verify the associations. (C) The quantitative real-time polymerase chain reaction (qRT-PCR) findings show that knockdown of EZH2 with si-EZH2 inhibited expression of CCNB2, MKI67, KIF20A, CCNA2, and CDC20. (D) The knockdown of E2F1 inhibited the expression of EZH2, MCM4, RRM2, DHFR, CCNE2, and CDC25A. These coexpressed genes and EZH2 are regulated by the same gene, E2F1. (E) EZH2 and the coexpressed genes (TOP2A, MYBL2, BUB1, CCNA2, and CDC6) were down-regulated when ANNCA was knocked down. (F) EZH2 and the coexpressed genes, ARUKA, MAD2L1, ZNF367, KPNA2, and TPX2 are regulated by FOXM1, and knockdown of FOXM1 deregulated these genes. (G) Knockdown of HOTAIR deregulated CCNA2, CHEK1, HMMR, NUSAP1, FANCI, and NCAPG. (H) Upregulation of miR-221 promoted the expression of EZH2 and CDC20. (I) Upregulation of miR-101 inhibited the expression of EZH2, STMN, PLK1, and BUB1B. (* P<0.05; ** P<0.01; *** P<0.001).
Figure 6. The relationship between EZH2 and the 185 coexpressed genes. (A) The network constructed using the STRING database. (B) The relationship between EZH2 and the 185 coexpressed genes. (C–I) Quantitative real-time polymerase chain reaction (qRT-PCR) was performed to verify the associations. (C) The quantitative real-time polymerase chain reaction (qRT-PCR) findings show that knockdown of EZH2 with si-EZH2 inhibited expression of CCNB2, MKI67, KIF20A, CCNA2, and CDC20. (D) The knockdown of E2F1 inhibited the expression of EZH2, MCM4, RRM2, DHFR, CCNE2, and CDC25A. These coexpressed genes and EZH2 are regulated by the same gene, E2F1. (E) EZH2 and the coexpressed genes (TOP2A, MYBL2, BUB1, CCNA2, and CDC6) were down-regulated when ANNCA was knocked down. (F) EZH2 and the coexpressed genes, ARUKA, MAD2L1, ZNF367, KPNA2, and TPX2 are regulated by FOXM1, and knockdown of FOXM1 deregulated these genes. (G) Knockdown of HOTAIR deregulated CCNA2, CHEK1, HMMR, NUSAP1, FANCI, and NCAPG. (H) Upregulation of miR-221 promoted the expression of EZH2 and CDC20. (I) Upregulation of miR-101 inhibited the expression of EZH2, STMN, PLK1, and BUB1B. (* P<0.05; ** P<0.01; *** P<0.001). Figure 7. The effects of siEZH2 in the C4-2 human prostate cancer cell line. (A) The results of cell cycle analysis showed that knockdown of EZH2 resulted in G2/M arrest (B) 23.98% for the control vs. 28.12% for shEZH2 (P<0.05). (C) Single-cell agarose gel electrophoresis for the cell transfected with control or siEZH2. (D) The results showed the tail length of cells transfected with shEZH2 was significantly longer than the cell in the control group (9.9 8±1.51 for the control group vs. 16.31±2.6 for the siEZH2 group). (E) Colony formation assay shows that knockdown of EZH2 inhibited cell proliferation when irradiated, and the results are shown in panel F. (G) Based on The Cancer Genome Atlas (TCGA) prostate dataset, the increased expression of EZH2, MCM4, KIAA0101, MKI67, RRM2, and CDC25A were associated with recurrence or progression after radiation. (* P<0.05; ** P<0.01)
Figure 7. The effects of siEZH2 in the C4-2 human prostate cancer cell line. (A) The results of cell cycle analysis showed that knockdown of EZH2 resulted in G2/M arrest (B) 23.98% for the control vs. 28.12% for shEZH2 (P<0.05). (C) Single-cell agarose gel electrophoresis for the cell transfected with control or siEZH2. (D) The results showed the tail length of cells transfected with shEZH2 was significantly longer than the cell in the control group (9.9 8±1.51 for the control group vs. 16.31±2.6 for the siEZH2 group). (E) Colony formation assay shows that knockdown of EZH2 inhibited cell proliferation when irradiated, and the results are shown in panel F. (G) Based on The Cancer Genome Atlas (TCGA) prostate dataset, the increased expression of EZH2, MCM4, KIAA0101, MKI67, RRM2, and CDC25A were associated with recurrence or progression after radiation. (* P<0.05; ** P<0.01) In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387