25 November 2020: Database Analysis
Bioinformatics for The Prognostic Value and Function of Cubilin (CUBN) in Colorectal Cancer
Yibin Wu1BCDEF, Ye Xu1ACG*DOI: 10.12659/MSM.922447
Med Sci Monit 2020; 26:e922447
Abstract
BACKGROUND: Cubilin (CUBN) gene was reported to be a novel risk variant for colorectal cancer (CRC). Previous studies have shown that germline variants in known cancer driver genes are predictive of patient outcome, but no study has systematically analyzed CRC to identify CUBN that can predict patient outcome and function by using bioinformatics.
MATERIAL AND METHODS: The association in expression, clinicopathological parameters, and survival were analyzed by using Oncomine, UNCLA, and GEPIA, while CUBN alterations and related functional networks were identified using cBioPortal. The Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes pathways (KEGG) of CUBN in CRC were explored by using LinkOmics. Gene set enrichment analysis (GSEA) examined target networks of kinases, miRNAs, and transcription factors.
RESULTS: We found that CUBN was overexpressed in CRC. Patients who were in advanced TNM stage tended to express higher CUBN mRNA levels, while those who received radiotherapy tended to express relatively lower CUBN mRNA levels. Higher expression of CUBN was found to be associated with shorter overall survival (OS) and disease-free survival (DFS). Moreover, functional networks analysis suggested that CUBN can regulate mismatch repair, terpenoid backbone biosynthesis, base excision repair, and proteasome via vitamin digestion and absorption pathway to influence CRC occurrence.
CONCLUSIONS: These findings suggested that CUBN could serve as a prognostic and therapeutic biomarker of CRC in the future.
Keywords: Colorectal Neoplasms, HMGA1a Protein, Computational Biology, Gene Regulatory Networks, Genome, Human, Protein Interaction Maps, RNA, Messenger, Receptors, Cell Surface, Transcription Factors
Background
Colorectal cancer is the third most common cancer and the second leading cause of cancer-related death worldwide [1]. The pathogenesis of CRC is a complex multistage process, involving sequential mutational events occurring along with progression of the cancer [2]. Recent studies have shown that genes mutated in cancer-related pathways, including Wnt, Notch, EGFR/MAPK, TGF-β, and PI3K signaling pathways, are responsible for increased proliferation, progression, invasion, and apoptosis in CRC cells [3,4]. Despite the limited drugs currently being developed, certain patients with advanced CRC are responsive to the treatments based on cancer-related genes. Some patients benefit from drugs such as Bevacizumab, Cetuximab, and PD-1 [5–7]. Thus, identifying novel variants for CRC is essential to optimizing current therapeutic strategies.
Culibin – known as intestinal intrinsic factor, intrinsic factor-cobalamin receptor, or intrinsic factor-vitamin B12 receptor – acts as a co-transporter in many absorptive epithelia, including intestine, renal proximal tubules, and embryonic yolk sac, and helps in the uptake of lipoprotein, vitamin, and iron [8]. In addition, CUBN was reported to be linked with the development and progression of cancers. Pontén et al. found that CUBN expression is highly specific to renal cell carcinoma (RCC), and lower expression of CUBN is significantly and independently correlated with poor prognosis [9,10]. Li et al. identified CUBN as a vitamin B12 metabolism gene in gastric carcinogenesis, in which CUBN haplotypes in the vitamin B12 metabolism pathway increase GC risk by regulating the balance of vitamin B12 metabolism [11]. High-throughput genotyping analysis revealed that by somatic mutation of CUBN, isoleucine changed into valine in osteosarcoma, which may stratify patients at risk of recurrence based on molecular signatures [12]. Genome-wide association studies (GWAS) are widely used to identify risk variants related to the occurrence of cancers. Our literature search revealed that a novel significant association at 10p13 with rs10904849 intronic to cubilin was correlated with the risk of CRC occurrence [13]. To date, no studies have reported the prognostic value and function of CUBN in CRC.
Thus, we investigated the expression, clinicopathological parameters, and survival data on CUBN in CRC by using Oncomine, UNCLA, and GEPIA, and we identified the somatic mutations of CUBN in CRC patients with independent sequencing data from the cBioPortal database. We also examined the pathway and predicted the target networks of kinase, mRNA, and transfer factor of CUBN in CRC.
Material and Methods
ONCOMINE DATABASE:
The Oncomine database (www.oncomine.org) is an integrated data mining platform for DNA or RNA sequences analysis, which includes 715 datasets of gene expression and data from 86,733 cancer and the normal tissues [14]. In this study, 2 datasets (GSE9689 and GSE20842) from Oncomine were selected to analyze the expression mRNA of CUBN in CRC tissues compared with normal tissues. Students’ t-test was used to compare the difference in transcriptional expression. P value <0.05 means the data was statistically significant. * p<0.05, ** p<0.01, *** p<0.001.
GEPIA ANALYSIS:
GEPIA (
CBIOPORTAL ANALYSIS:
cBioPortal (www.cbioportal.org), currently containing 225 cancer studies, is an online open-access website resource for exploring, visualizing, and analyzing multidimensional cancer genomics data [15]. In this study, we used cBioPortal to analyze the genomic profiles of CUBN in the TCGA CRC samples, which contained the alteration frequency of gene and the network of CUBN neighboring genes. Then, neighboring genes were used to explore GO and KEGG pathway enrichment analyses by DAVID. The GO annotation contained 3 parts: cellular component (CC), biological process (BP), and molecular function (MF).
LINKEDOMICS ANALYSIS:
LinkedOmics is a publicly available portal that contains data from all 32 TCGA cancer types, and has 3 analytical modules: LinkFinder, LinkInterpreter, and LinkCompare [16]. In this study, the LinkFinder module was used to explore the correlation between CUBN expression and clinicopathologic features, including T stage, clinical stage, metastasis, lymph node involvement, and radiotherapy. The LinkFinder can generate statistical plots for individual genes. The results are graphically shown in volcano plots and heat maps. We used LinkedOmics to explore the functional network of differentially expressed genes. Data from the LinkFinder results were signed and ranked, and GSEA was also performed for the analyses of GO, KEGG pathways, kinase, miRNA, and transcription factor-target enrichment. The rank criterion was a P<0.05, and 500 simulations were used.
GENEMANIA ANALYSIS:
GeneMANIA (http://genemania.org/) is a publicly available portal that can predict the function of genes and gene sets. The website features some bioinformatics methods such as physical interaction, gene co-expression, gene co-location, gene enrichment analysis, and website prediction [17]. We used GeneMANIA to reveal the gene networks and predict function of genes that were identified by GSEA as being enriched in CRC: kinase_PIK3CD and transcription factor FOXO4.
Results
OVEREXPRESSION OF CUBN MRNA IN PATIENTS WITH CRC:
To explore the role of CUBN in CRC, we used 2 datasets (GSE9689 and GSE20842) downloaded from the Oncomine database to analyze the mRNA expression of CUBN in CRC. As shown in Figure 1A, 1B, the expression mRNA of CUBN was significantly upregulated in primary CRC tissues compared to normal tissues in the 2 datasets (* p<0.05, *** p<0.001), which suggested that CUBN is an oncogene in CRC.
THE CORRELATIONS BETWEEN CUBN EXPRESSION WITH CLINICOPATHOLOGICAL PARAMETERS IN CRC:
We further analyzed the correlations between CUBN expression with clinicopathological parameters, including T stage, metastasis, lymph nodes, clinical stage, and radiotherapy, by using the LinkedOmics database. The results showed that patients who were in advanced T stage tended to express higher CUBN mRNA, which suggested CUBN was positively correlated with T stage (Figure 2A). Patients in stage IV or N2 or M1 stage expressed higher CUBN mRNA levels than those in stage I/II/III or N0/1 or M0 (Figure 2B–2D, 2F), while patients who accepted radiotherapy tended to have lower mRNA levels of CUBN expression. However, there was no significant association between CUBN expression with MSI-H and MSS status (Figure 2E). In short, our study demonstrated that CUBN expression was significantly associated with clinicopathological parameters in CRC patients, and CUBN might serve as a biomarker for poor prognosis in CRC. * p<0.05, ** p<0.01, *** p<0.001.
PROGNOSTIC VALUE OF CUBN EXPRESSION IN CRC:
To evaluate the prognostic value of CUBN expression in CRC, we next used GEPIA to analyze the survival data of patients with low CUBN expression compared to those with high CUBN expression. We found that higher CUBN mRNA expression was significantly associated with shorter OS (Figure 3A, HR=1.9, Log rank p=0.011) and DFS (Figure 3B, HR=1.7, log rank p=0.048). In conclusion, our study demonstrated that CUBN was an indicator of poor prognosis in patients with CRC.
GENOMIC ALTERATIONS OF CUBN IN CRC:
Genomic instability involves a high frequency of mutations within the genome, including changes in chromosomal rearrangements, nucleic acid sequences, or aneuploidy. Genomic instability is a consequence of carcinogenesis. To analyze the effect of CUBN alteration in CRC, we used cBioPortal database to explore the genomic instability of CUBN in a TCGA CRC database with 212 cases. We found that 12% (25 of 212 cases) of CUBN alteration was observed in CRC patients, which suggested that CUBN mutation increases the malignancy of CRC (Figure 4A). We identified 4 neighbored genes of CUBN interacting with CUBN that were correlated with CRC occurrence, such as LRP2, LGMN, GC, and APOA1 (Figure 4B). Then, we used CUBN combined with its neighbored genes to explore the functional networks in CRC by DAVID. As shown in Table 1, biological processes such as GO: 0042359 (vitamin D metabolic process), GO: 0051180 (vitamin transport), GO: 0042157 (lipoprotein metabolic process), GO: 0008203 (cholesterol metabolic process), and GO: 0006898 (receptor-mediated endocytosis) were remarkably regulated by the CUBN mutation in CRC. Cellular components, including GO: 0030139 (endocytic vesicle), GO: 0072562 (blood microparticle), GO: 0034366 (spherical high-density lipoprotein particle), and GO: 0042627 (chylomicron) were significantly correlated with the CUBN alterations. Also, CUBN mutation prominently affected the molecular functions, such as GO: 0070653 (high-density lipoprotein particle receptor binding), GO: 0008035 (high-density lipoprotein particle binding), GO: 0060228 (phosphatidylcholine-sterol O-acyltransferase activator activity), GO: 0017127 (cholesterol transporter activity), and GO: 0031210 (phosphatidylcholine binding). In KEGG analysis, hsa04977 (vitamin digestion and absorption) (Figure 5) was found to be associated with CUBN mutation in CRC, which suggested that CUBN was involved in metabolism activity.
ENRICHMENT ANALYSIS OF CUBN FUNCTIONAL NETWORKS IN CRC:
To further study the function of CUBN in CRC, we used LinkedOmics to analyze mRNA sequencing data in a 379-case TCGA CRC dataset. As shown in the volcano plot (Figure 6A), 2,681 genes (dark green dots) was significant negatively associated with CUBN, while 3,891 genes (dark red dots) were significant positively associated with CUBN. The heat map presents the 50 most significant positively or negatively co-expression genes of CUBN (Figure 6B, 6C). Significant GO term analysis by GSEA found that genes differentially expressed in relation to CUBN were located in extracellular matrix, platelet alpha granule, and protein complex involved in cell adhesion and collage trimer (Figure 7A), where they participated in tricarboxylic acid metabolic process, cytochrome complex assembly, base excision repair, and mitochondrial RNA metabolic process (Figure 7B). They served as extracellular matrix structural constituents, cytokine binding, collagen binding, and immunoglobulin binding (Figure 7C). KEGG pathway analysis showed the enrichment in mismatch repair, DNA replication, pyruvate metabolism, and proteasome (Figure 7D).
CUBN NETWORKS OF KINASE, MIRNA, OR TRANSCRIPTION FACTOR TARGETS IN CRC:
To further predict the targets of CUBN in CRC, we analyzed the kinase, miRNA, and transcription factor (TF) targets generated by Gene Set Enrichment Analysis (GSEA). As shown in Table 2, the top 5 significant target networks were kinase-target network, mainly related to PIK3CD, PRKG1, BLK, TEC, and CSNK1G3. The top 5 significant target networks were the mRNA-target network related to MIR-517B (ATGCACG), MIR-208 (CGTCTTA), MIR-452 (TGCAAAC), MIR-19A (TTTGCAC), and MIR-493 (ATGTACA). The top 5 significant target networks were TF-target network mainly related to FOXO4_01, MEF2_Q6_01, FOXJ2_01, MEF2_01, and PBX1_02. The protein-protein interaction networks generated by GeneMANIA predicted the association among genes for the kinases PIK3CD and TF_FOXO4. As the results show in Figure 8A, the gene set enriched for kinase_PIK3CD was involved in phosphatidylinositol 3-kinase activity, phosphatidylinositol metabolic process, and T cell receptor signaling pathway. The gene set enriched for FOXO4_01 included response to insulin, cellular response to insulin stimulus, DNA binding, and negative regulation of phosphorylation (Figure 8B).
Discussion
The heterogeneity of CRC makes it difficult to determine which patients need further therapy beyond surgical resection and which patients have a poor prognosis [18]. Although the majority of CRC patients initially are response to chemotherapy, some patients eventually experience drug resistance, which results in recurrence and metastasis [19,20]. A better prognostic biomarker for outcome prediction and therapy assignment is urgently needed.
In this study, we first used bioinformatics methods to demonstrate that CUBN was upregulated in CRC and its overexpression was associated with advanced TNM stage. We also validated the prediction power of CUBN expression in prognosis and revealed that higher CUBN expression was correlated with shorter OS and DFS. Furthermore, we found somatic mutation of CUBN occurred in CRC patients and CUBN can participate in metabolism activities to regulate the development and progression of CRC.
Studies on CUBN have widely reported that CUBN is correlated with the occurrence of cancers such as RCC, GC, and osteosarcoma [9,11,12]. GWAS has identified CUBN as a novel risk variant for CRC [13,21], but no experimental data have ever verified the prognostic value and function of CUBN in CRC. Thus, to study the role of CUBN in CRC might produce a novel prognostic biomarker. In our study, we demonstrated that the expression of CUBN mRNA was higher in cancer tissues than in normal tissues, which suggested that CUBN may be an oncogene of CRC. Our analysis of correlations between CUBN with clinicopathological features showed that patients with more advanced clinical stages and T/N/M stage tended to express higher CUBN levels, which predicted CUBN to be an oncogene unfavorable to patient prognosis. We also found that patients who accepted radiation therapy tended to express lower CUBN levels, and our data suggest that CUBN could be used to predict therapeutic efficacy of radiotherapy. In addition, patients with higher CUBN expression had a shorter OS and DFS than those with lower CUBN expression. Previous studies demonstrated that CUBN overexpression was significantly associated with poor prognosis in RCC and osteosarcoma, which is consistent with our results [9,12]. Thus, it is plausible that CUBN is an oncogene in human CRC progression and represents a promising clinical prognostic biomarker and therapeutic target for this disease.
Genetic factors play a significant role in the etiology of both familial and sporadic CRC. As reported, altered related-gene expression and its dysfunction in CRC may result from alterations in chromosomal structure [22]. The next step in our study was to explore the alteration frequency of CUBN in CRC. Based on cBioPortal database analysis, we learned that the copy number of CUBN was increased in CRC, and mutation was the major type of CUBN alteration. We speculated that CUBN mutation can interact with its neighbored genes to promote CRC formation by regulating related pathways. Indeed, neighbored gene networks close to CUBN also showed different degrees of alterations in CRC. Related functional networks of these altered genes were involved in vitamin digestion and absorption. Consistent with these results, Arem et al. reported that CUBN participates in the vitamin D metabolic pathway to increase pancreatic cancer risk [23]. Another study also had identified CUBN as a significant variant for gastric cancer by regulating vitamin B12 metabolism [11]. Thus, we speculated that CUBN alterations may be involved in the core node of metabolic activity to increase cancer risk, including CRC.
Enrichment analysis of target gene sets using GSEA can help explore significant networks of target kinases, miRNA, and transcription factors [24]. Our data showed that the functional networks of CUBN in CRC are involved in mismatch repair, DNA replication, pyruvate metabolism, and proteasome. Thus, we concluded that alteration of CUBN induced abnormal transcription to cause major dysfunction and even cause cancers such as CRC. Genomic instability and mutagenesis are the fundamental features of cancer cells, and kinases and their related signaling pathways help stabilize and repair genomic DNA [25,26]. Therefore, we further analyzed the association between CUBN and the network targets of kinases, miRNAs, and transcription factors. Our data showed that the most significant targets, including kinase, miRNA and TF, were PIK3CD, miRNA-517B, and FOXO4. In fact, PIK3CD plays an important role in mediating immune responses, activating signaling cascades involved in cell growth, survival, proliferation, motility, and morphology [27,28]. In CRC, CUBN regulates metabolic activities via PIK3CD pathways.
Our study also identified several transcription factors that are correlated with CUBN. The particular TFs in our study are involved in regulation of the insulin signaling pathway and cell cycle. In fact, studies have shown that overexpression of FOXO4 is correlated with poor prognosis in CRC patients, and suggests FOXO4 as a diagnostic and prognostic marker of CRC [29,30]. Therefore, our findings suggest that FOXO4 is a vital target of CUBN, and that CUBN acts through this factor to regulate the cell cycle and proliferation capacity of CRC. Further studies are needed test this hypothesis.
Conclusions
This study provides multi-level evidence to assess the clinical value and the function network of CUBN in CRC. Our results suggest that CUBN overexpression in CRC has far-reaching effects on poor prognosis and at multiple steps of gene expression (e.g., mismatch repair, DNA replication, pyruvate metabolism, and proteasome) and of the cell cycle. CUBN is significantly associated with several tumor-related kinases (e.g., PIK3CD), miRNAs (e.g., miRNA-517B), and transcription factors (e.g., FOXD4).
Figures
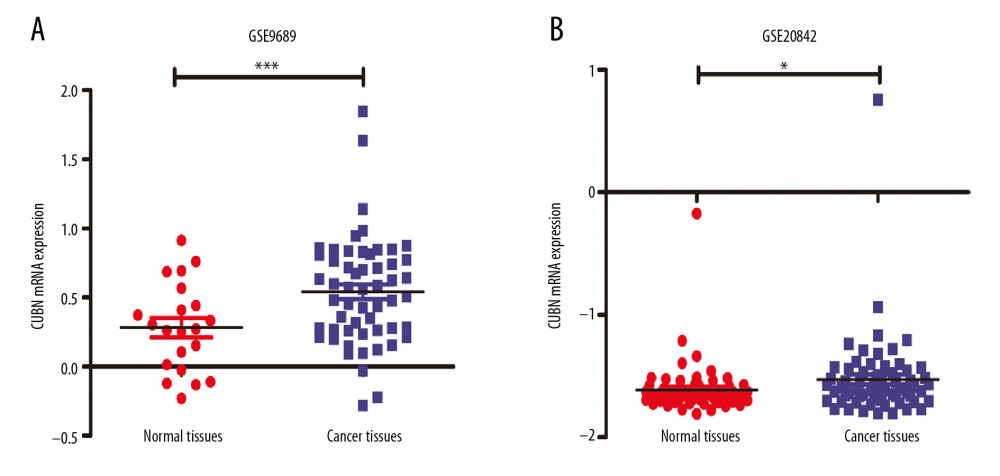 Figure 1. Expression of CUBN mRNA in CRC (ONCOMINE). (A, B) Levels of CUBN mRNA expression in GSE9689 and GSE20842 were both significantly higher in CRC tissues than in the normal tissues.
Figure 1. Expression of CUBN mRNA in CRC (ONCOMINE). (A, B) Levels of CUBN mRNA expression in GSE9689 and GSE20842 were both significantly higher in CRC tissues than in the normal tissues. 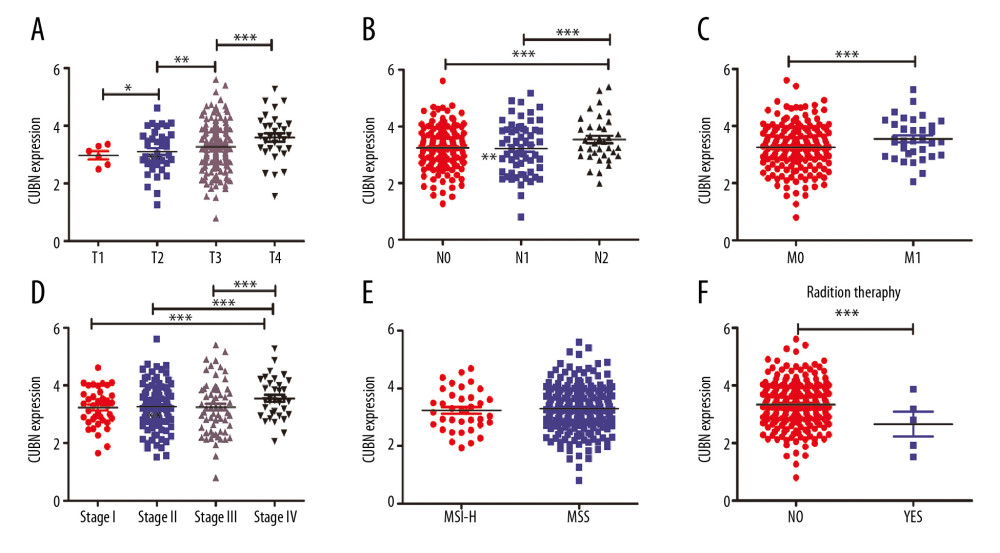 Figure 2. Associations of CUBN mRNA expression with clinicopathological features in CRC. (A) CUBN mRNA expression was significantly associated with T stage, and as T stage increased, the mRNA expression of CUBN tended to be higher. (B–D) Expression of CUBN mRNA in clinical stage IV or N2 or M1 was remarkably higher than that in stage I/II/III or N0/1 or M0. (F) Patients who accepted radiotherapy tended to express lower CUBN than those who did not. (E) There was no significant association between CUBN expression in MSI-H and MSS. * p<0.05, ** p<0.01, *** p<0.001.
Figure 2. Associations of CUBN mRNA expression with clinicopathological features in CRC. (A) CUBN mRNA expression was significantly associated with T stage, and as T stage increased, the mRNA expression of CUBN tended to be higher. (B–D) Expression of CUBN mRNA in clinical stage IV or N2 or M1 was remarkably higher than that in stage I/II/III or N0/1 or M0. (F) Patients who accepted radiotherapy tended to express lower CUBN than those who did not. (E) There was no significant association between CUBN expression in MSI-H and MSS. * p<0.05, ** p<0.01, *** p<0.001. 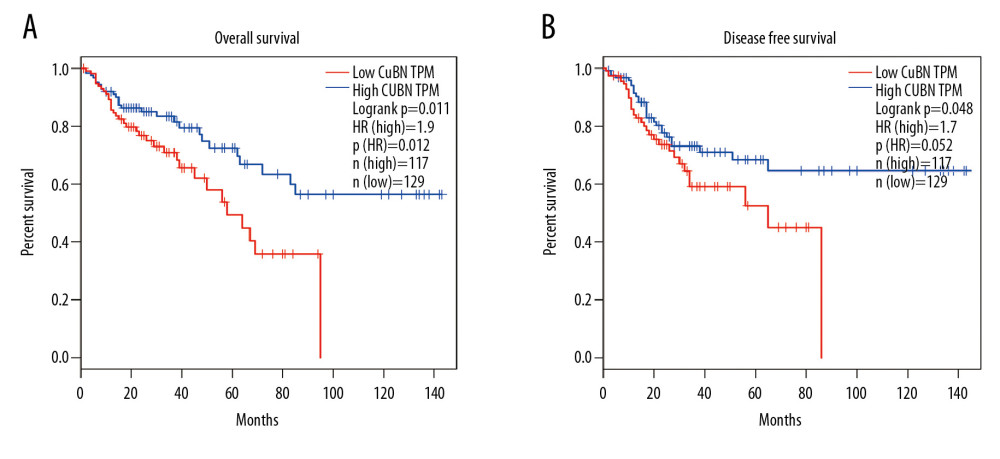 Figure 3. Prognostic value of CUBN mRNA expression in CRC patients. (A, B) Patients with higher CUBN expression tend to be have a poor prognosis in OS (p=0.011) and DFS (p=0.048) at 10-year follow-up visit.
Figure 3. Prognostic value of CUBN mRNA expression in CRC patients. (A, B) Patients with higher CUBN expression tend to be have a poor prognosis in OS (p=0.011) and DFS (p=0.048) at 10-year follow-up visit. 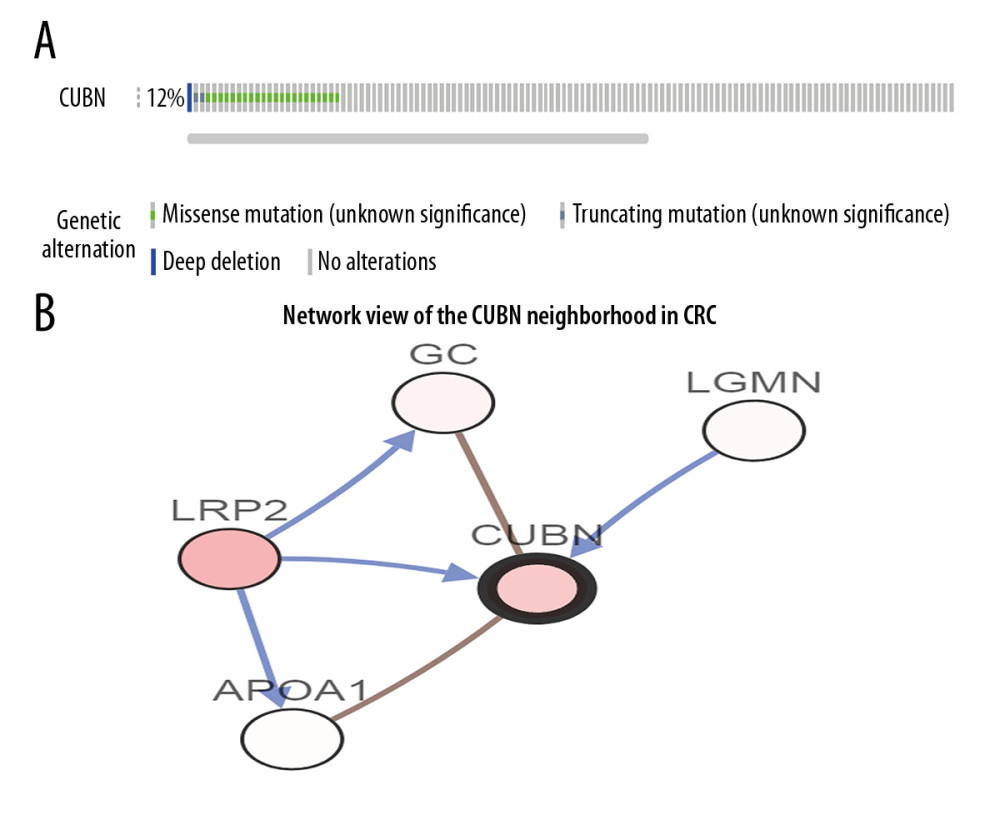 Figure 4. Visual summary of CUBN alterations and biological interaction network in CRC. (A) OncoPrint of CUBN alterations in CRC. The OncoPrint provided an overview of CUBN alteration in CRC from the TCGA CRC data. Different colors were used to highlight the different types of genetic alterations. (B) Networks of the neighboring genes of CUBN in CRC. CUBN was the seed gene, and all other genes are automatically identified as altered in CRC. Darker red indicates elevated frequency of alteration in CRC. The blue connection indicates that the first protein controls a reaction that changes the state of the second protein; the red connection suggests that the proteins were members of the same complex.
Figure 4. Visual summary of CUBN alterations and biological interaction network in CRC. (A) OncoPrint of CUBN alterations in CRC. The OncoPrint provided an overview of CUBN alteration in CRC from the TCGA CRC data. Different colors were used to highlight the different types of genetic alterations. (B) Networks of the neighboring genes of CUBN in CRC. CUBN was the seed gene, and all other genes are automatically identified as altered in CRC. Darker red indicates elevated frequency of alteration in CRC. The blue connection indicates that the first protein controls a reaction that changes the state of the second protein; the red connection suggests that the proteins were members of the same complex. 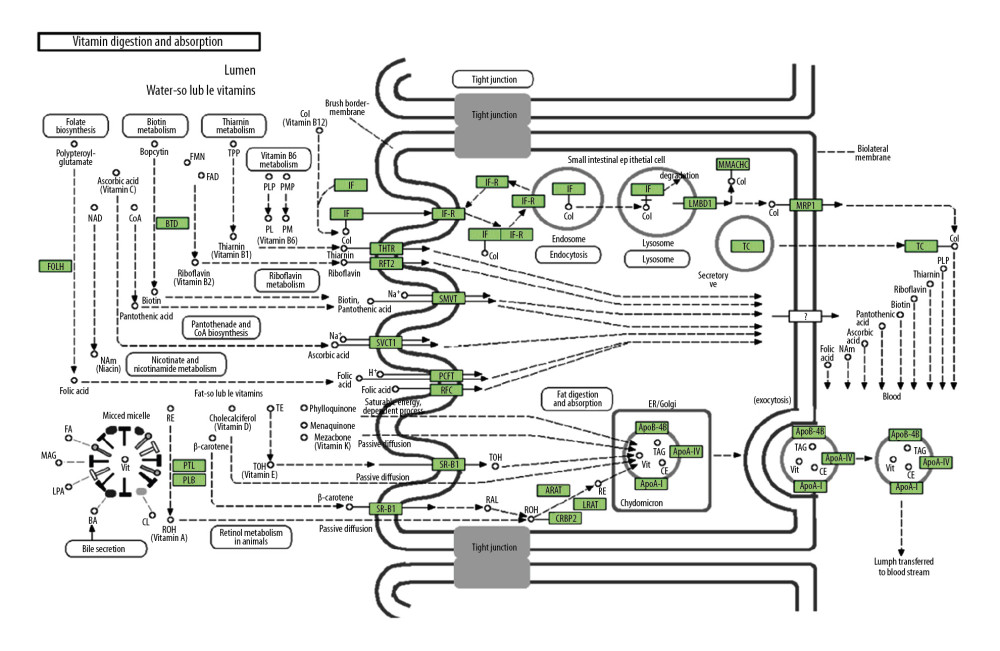 Figure 5. Visualization of KEGG pathway of the genes altered in the CUBN neighborhood in CRC. Vitamin digestion and absorption.
Figure 5. Visualization of KEGG pathway of the genes altered in the CUBN neighborhood in CRC. Vitamin digestion and absorption. 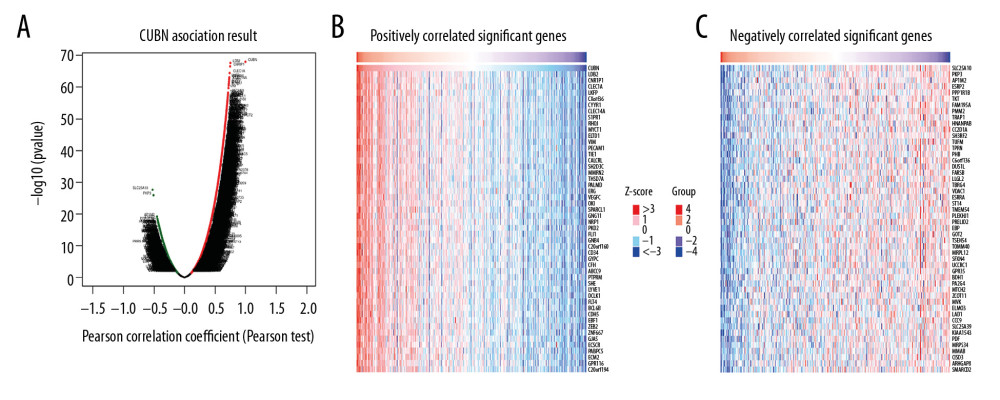 Figure 6. Differentially-expressed genes associated with CUBN in CRC. (A) Volcano plot graphically presents the associations between CUBN and genes differentially expressed in CRC. (B, C) Genes positively and negatively correlated with CUBN in CRC, shown by heat maps (Top 50). Red suggests positively correlated genes and green suggests negatively correlated genes.
Figure 6. Differentially-expressed genes associated with CUBN in CRC. (A) Volcano plot graphically presents the associations between CUBN and genes differentially expressed in CRC. (B, C) Genes positively and negatively correlated with CUBN in CRC, shown by heat maps (Top 50). Red suggests positively correlated genes and green suggests negatively correlated genes. 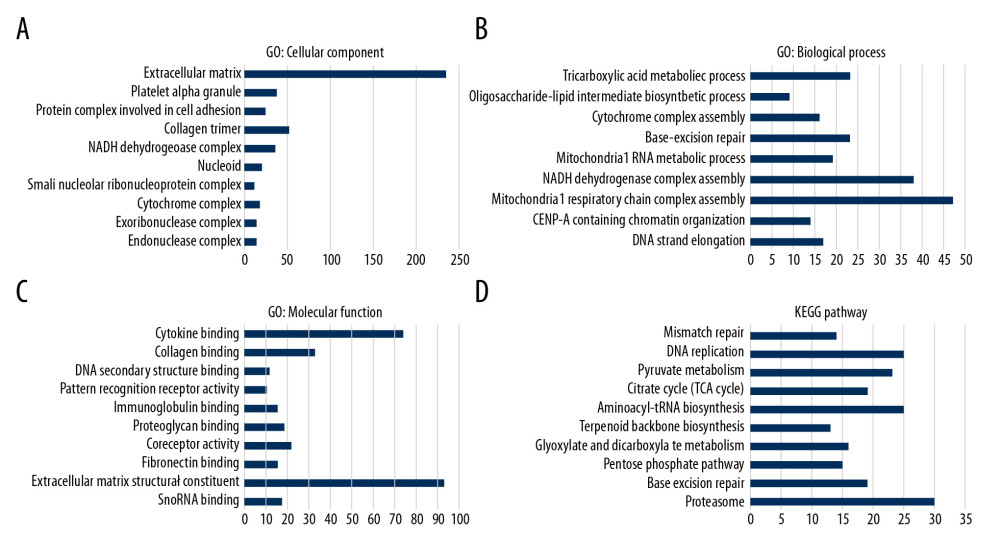 Figure 7. Significantly enriched GO annotations and KEGG pathways of CUBN in CRC. GSEA was used to analyze the significantly enriched GO annotations and KEGG pathways of CUBN co-expression genes in CRC. The top 10 significant networks were chosen. (A) CC; (B) BP; (C) MF; (D) KEGG pathway analysis.
Figure 7. Significantly enriched GO annotations and KEGG pathways of CUBN in CRC. GSEA was used to analyze the significantly enriched GO annotations and KEGG pathways of CUBN co-expression genes in CRC. The top 10 significant networks were chosen. (A) CC; (B) BP; (C) MF; (D) KEGG pathway analysis. 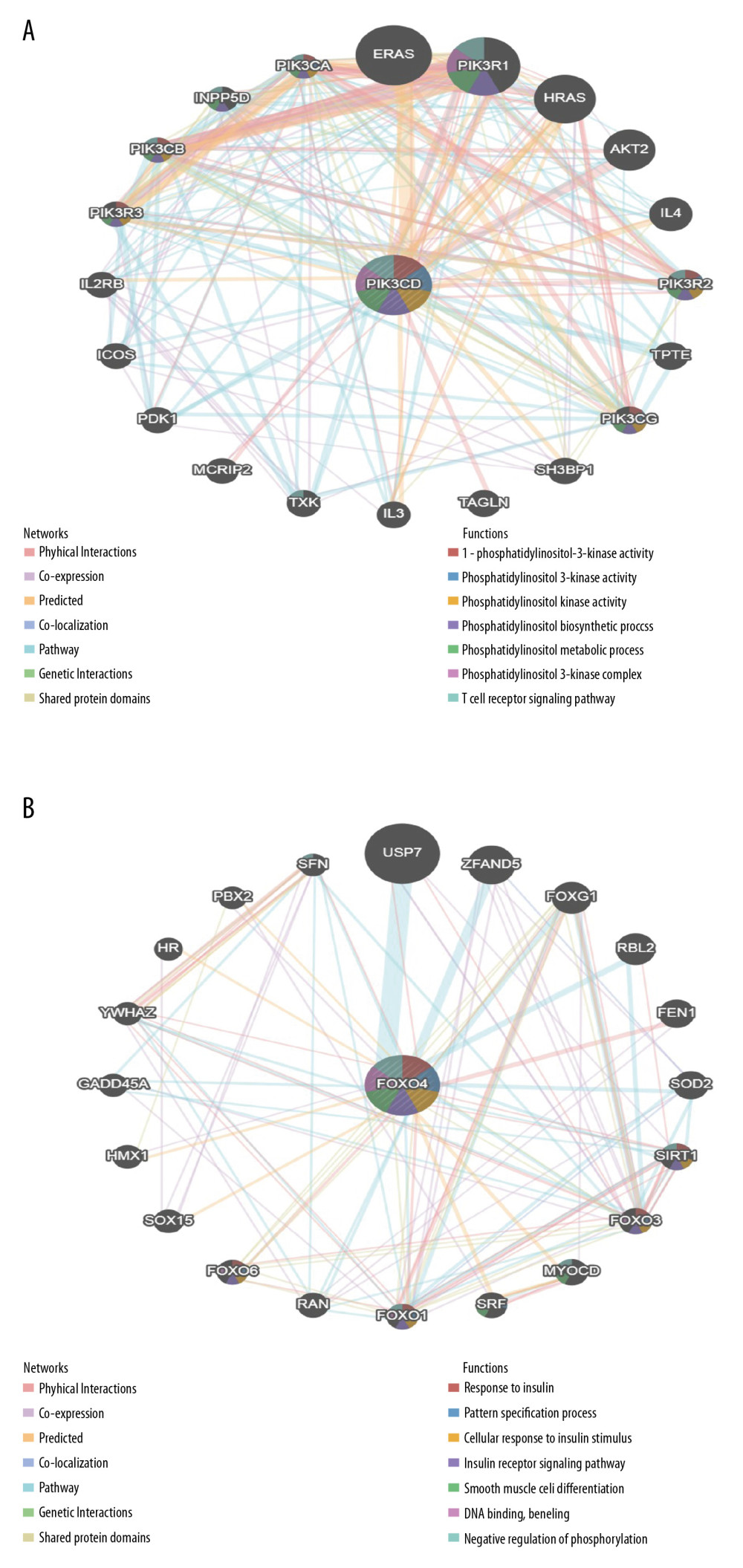 Figure 8. Protein-protein interaction networks of kinase PIK3CD and transcription factor FOXD4_01 (GeneMANIA). (A, B) PPI network and functional analysis suggested the gene set enriched in the target networks of kinase_PIK3CD and TF_ FOXD4.
Figure 8. Protein-protein interaction networks of kinase PIK3CD and transcription factor FOXD4_01 (GeneMANIA). (A, B) PPI network and functional analysis suggested the gene set enriched in the target networks of kinase_PIK3CD and TF_ FOXD4. References
1. Siegel RL, Miller KD, Fedewa SA, Colorectal cancer statistics, 2017: Cancer J Clin, 2017; 67(3); 177-93
2. Ng C, Li H, Wu WKK, Genomics and metagenomics of colorectal cancer: J Gastrointest Oncol, 2019; 10(6); 1164-70
3. Farooqi AA, de la Roche M, Djamgoz MBA, Overview of the oncogenic signaling pathways in colorectal cancer: Mechanistic insights: Semin Cancer Biol, 2019; 58; 65-79
4. Wu WKK, Wang XJ, Cheng ASL, Dysregulation and crosstalk of cellular signaling pathways in colon carcinogenesis: Crit Rev Oncol Hematol, 2013; 86(3); 251-77
5. Ducreux M, Chamseddine A, Laurent-Puig P, Molecular targeted therapy of BRAF-mutant colorectal cancer: Ther Adv Med Oncol, 2019; 11; 432495762
6. Yaghoubi N, Soltani A, Ghazvini K, PD-1/PD-L1 blockade as a novel treatment for colorectal cancer: Biomed Pharmacother, 2019; 110; 312-18
7. Martinelli E, Ciardiello D, Martini G, Implementing anti-epidermal growth factor receptor (EGFR) therapy in metastatic colorectal cancer: challenges and future perspectives: Ann Oncol, 2020; 31(1); 30-40
8. Shaik AP, Alsaeed AH, Kiranmayee S, Phylogenetic analysis of cubilin (CUBN) gene: Bioinformation, 2013; 9(1); 29-36
9. Gremel G, Djureinovic D, Niinivirta M, A systematic search strategy identifies cubilin as independent prognostic marker for renal cell carcinoma: BMC Cancer, 2017; 17(1); 9
10. Ma J, Guan M, Bowden DW, Association analysis of the cubilin (CUBN) and megalin (LRP2) genes with ESRD in African Americans: Clin J Am Soc Nephrol, 2016; 11(6); 1034-43
11. Zhao L, Wei Y, Song A, Association study between genome-wide significant variants of vitamin B12 metabolism and gastric cancer in a Han Chinese population: IUBMB Life, 2016; 68(4); 303-10
12. Choy E, Hornicek F, MacConaill L, High-throughput genotyping in osteosarcoma identifies multiple mutations in phosphoinositide-3-kinase and other oncogenes: Cancer, 2012; 118(11); 2905-14
13. Al-Tassan NA, Whiffin N, Hosking FJ, A new GWAS and meta-analysis with 1000Genomes imputation identifies novel risk variants for colorectal cancer: Sci Rep, 2015; 5(1); 10442
14. Rhodes DR, Yu J, Shanker K, ONCOMINE: A cancer microarray database and integrated data-mining platform1: Neoplasia (New York, NY), 2004; 6(1); 1-6
15. Gao J, Aksoy BA, Dogrusoz U, Integrative analysis of complex cancer genomics and clinical profiles using the cBioPortal: Sci Signal, 2013; 6(269); l1
16. Vasaikar SV, Straub P, Wang J, LinkedOmics: Analyzing multi-omics data within and across 32 cancer types: Nucleic Acids Res, 2018; 46(D1); D956-63
17. Franz M, Rodriguez H, Lopes C, GeneMANIA update 2018: Nucleic Acids Res, 2018; 46(W1); W60-64
18. Molinari C, Marisi G, Passardi A, Heterogeneity in colorectal cancer: A challenge for personalized medicine?: Int J Mol Sci, 2018; 19(12); 3733
19. Jeught KVD, Xu H, Li Y, Drug resistance and new therapies in colorectal cancer: World J Gastroenterol, 2018; 24(34); 3834-48
20. Bardelli A, Siena S, Molecular mechanisms of resistance to cetuximab and panitumumab in colorectal cancer: J Clin Oncol, 2010; 28(7); 1254-61
21. Zinck JW, de Groh M, MacFarlane AJ, Genetic modifiers of folate, vitamin B-12, and homocysteine status in a cross-sectional study of the Canadian population: Am J Clin Nutr, 2015; 101(6); 1295-304
22. Seaman L, Chen H, Brown M, Nucleome analysis reveals structure-function relationships for colon cancer: Mol Cancer Res, 2017; 15(7); 821-30
23. Arem H, Yu K, Xiong X, Vitamin D metabolic pathway genes and pancreatic cancer risk: PLoS One, 2015; 10(3); e117574
24. Wu X, Hasan MA, Chen JY, Pathway and network analysis in proteomics: J Theor Biol, 2014; 362; 44-52
25. Karimian A, Ahmadi Y, Yousefi B, Multiple functions of p21 in cell cycle, apoptosis and transcriptional regulation after DNA damage: DNA Repair, 2016; 42; 63-71
26. Yogosawa S, Yoshida K, Tumor suppressive role for kinases phosphorylating p53 in DNA damage-induced apoptosis: Cancer Sci, 2018; 109(11); 3376-82
27. Burke JE, Structural basis for regulation of phosphoinositide kinases and their involvement in human disease: Mol Cell, 2018; 71(5); 653-73
28. Nunes-Santos CJ, Uzel G, Rosenzweig SD, PI3K pathway defects leading to immunodeficiency and immune dysregulation: J Allergy Clin Immun, 2019; 143(5); 1676-87
29. Kwon IK, Wang R, Thangaraju M, PKG inhibits TCF signaling in colon cancer cells by blocking beta-catenin expression and activating FOXO4: Oncogene, 2010; 29(23); 3423-34
30. Liu X, Zhang Z, Sun L, microRNA-499-5p promotes cellular invasion and tumor metastasis in colorectal cancer by targeting FOXO4 and PDCD4: Carcinogenesis, 2011; 32(12); 1798-805
Figures
 Figure 1. Expression of CUBN mRNA in CRC (ONCOMINE). (A, B) Levels of CUBN mRNA expression in GSE9689 and GSE20842 were both significantly higher in CRC tissues than in the normal tissues.
Figure 1. Expression of CUBN mRNA in CRC (ONCOMINE). (A, B) Levels of CUBN mRNA expression in GSE9689 and GSE20842 were both significantly higher in CRC tissues than in the normal tissues. Figure 2. Associations of CUBN mRNA expression with clinicopathological features in CRC. (A) CUBN mRNA expression was significantly associated with T stage, and as T stage increased, the mRNA expression of CUBN tended to be higher. (B–D) Expression of CUBN mRNA in clinical stage IV or N2 or M1 was remarkably higher than that in stage I/II/III or N0/1 or M0. (F) Patients who accepted radiotherapy tended to express lower CUBN than those who did not. (E) There was no significant association between CUBN expression in MSI-H and MSS. * p<0.05, ** p<0.01, *** p<0.001.
Figure 2. Associations of CUBN mRNA expression with clinicopathological features in CRC. (A) CUBN mRNA expression was significantly associated with T stage, and as T stage increased, the mRNA expression of CUBN tended to be higher. (B–D) Expression of CUBN mRNA in clinical stage IV or N2 or M1 was remarkably higher than that in stage I/II/III or N0/1 or M0. (F) Patients who accepted radiotherapy tended to express lower CUBN than those who did not. (E) There was no significant association between CUBN expression in MSI-H and MSS. * p<0.05, ** p<0.01, *** p<0.001. Figure 3. Prognostic value of CUBN mRNA expression in CRC patients. (A, B) Patients with higher CUBN expression tend to be have a poor prognosis in OS (p=0.011) and DFS (p=0.048) at 10-year follow-up visit.
Figure 3. Prognostic value of CUBN mRNA expression in CRC patients. (A, B) Patients with higher CUBN expression tend to be have a poor prognosis in OS (p=0.011) and DFS (p=0.048) at 10-year follow-up visit. Figure 4. Visual summary of CUBN alterations and biological interaction network in CRC. (A) OncoPrint of CUBN alterations in CRC. The OncoPrint provided an overview of CUBN alteration in CRC from the TCGA CRC data. Different colors were used to highlight the different types of genetic alterations. (B) Networks of the neighboring genes of CUBN in CRC. CUBN was the seed gene, and all other genes are automatically identified as altered in CRC. Darker red indicates elevated frequency of alteration in CRC. The blue connection indicates that the first protein controls a reaction that changes the state of the second protein; the red connection suggests that the proteins were members of the same complex.
Figure 4. Visual summary of CUBN alterations and biological interaction network in CRC. (A) OncoPrint of CUBN alterations in CRC. The OncoPrint provided an overview of CUBN alteration in CRC from the TCGA CRC data. Different colors were used to highlight the different types of genetic alterations. (B) Networks of the neighboring genes of CUBN in CRC. CUBN was the seed gene, and all other genes are automatically identified as altered in CRC. Darker red indicates elevated frequency of alteration in CRC. The blue connection indicates that the first protein controls a reaction that changes the state of the second protein; the red connection suggests that the proteins were members of the same complex. Figure 5. Visualization of KEGG pathway of the genes altered in the CUBN neighborhood in CRC. Vitamin digestion and absorption.
Figure 5. Visualization of KEGG pathway of the genes altered in the CUBN neighborhood in CRC. Vitamin digestion and absorption. Figure 6. Differentially-expressed genes associated with CUBN in CRC. (A) Volcano plot graphically presents the associations between CUBN and genes differentially expressed in CRC. (B, C) Genes positively and negatively correlated with CUBN in CRC, shown by heat maps (Top 50). Red suggests positively correlated genes and green suggests negatively correlated genes.
Figure 6. Differentially-expressed genes associated with CUBN in CRC. (A) Volcano plot graphically presents the associations between CUBN and genes differentially expressed in CRC. (B, C) Genes positively and negatively correlated with CUBN in CRC, shown by heat maps (Top 50). Red suggests positively correlated genes and green suggests negatively correlated genes. Figure 7. Significantly enriched GO annotations and KEGG pathways of CUBN in CRC. GSEA was used to analyze the significantly enriched GO annotations and KEGG pathways of CUBN co-expression genes in CRC. The top 10 significant networks were chosen. (A) CC; (B) BP; (C) MF; (D) KEGG pathway analysis.
Figure 7. Significantly enriched GO annotations and KEGG pathways of CUBN in CRC. GSEA was used to analyze the significantly enriched GO annotations and KEGG pathways of CUBN co-expression genes in CRC. The top 10 significant networks were chosen. (A) CC; (B) BP; (C) MF; (D) KEGG pathway analysis. Figure 8. Protein-protein interaction networks of kinase PIK3CD and transcription factor FOXD4_01 (GeneMANIA). (A, B) PPI network and functional analysis suggested the gene set enriched in the target networks of kinase_PIK3CD and TF_ FOXD4.
Figure 8. Protein-protein interaction networks of kinase PIK3CD and transcription factor FOXD4_01 (GeneMANIA). (A, B) PPI network and functional analysis suggested the gene set enriched in the target networks of kinase_PIK3CD and TF_ FOXD4. Tables
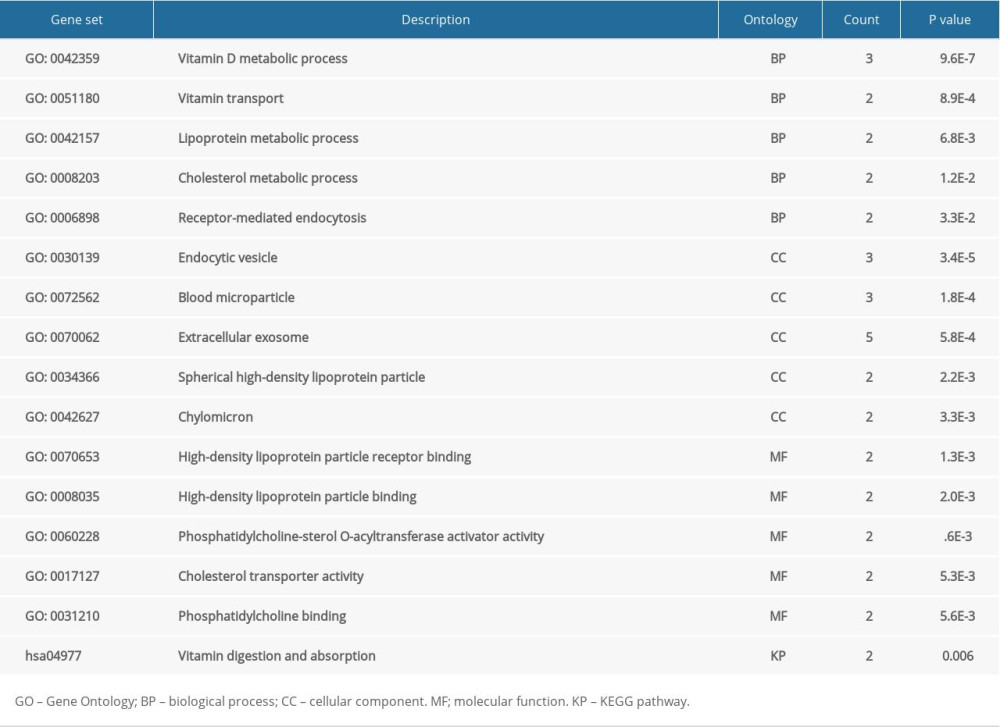 Table 1. Enrichment analysis of CUBN and its significant neighborhood genes altered in CRC as determined using DAVID.
Table 1. Enrichment analysis of CUBN and its significant neighborhood genes altered in CRC as determined using DAVID.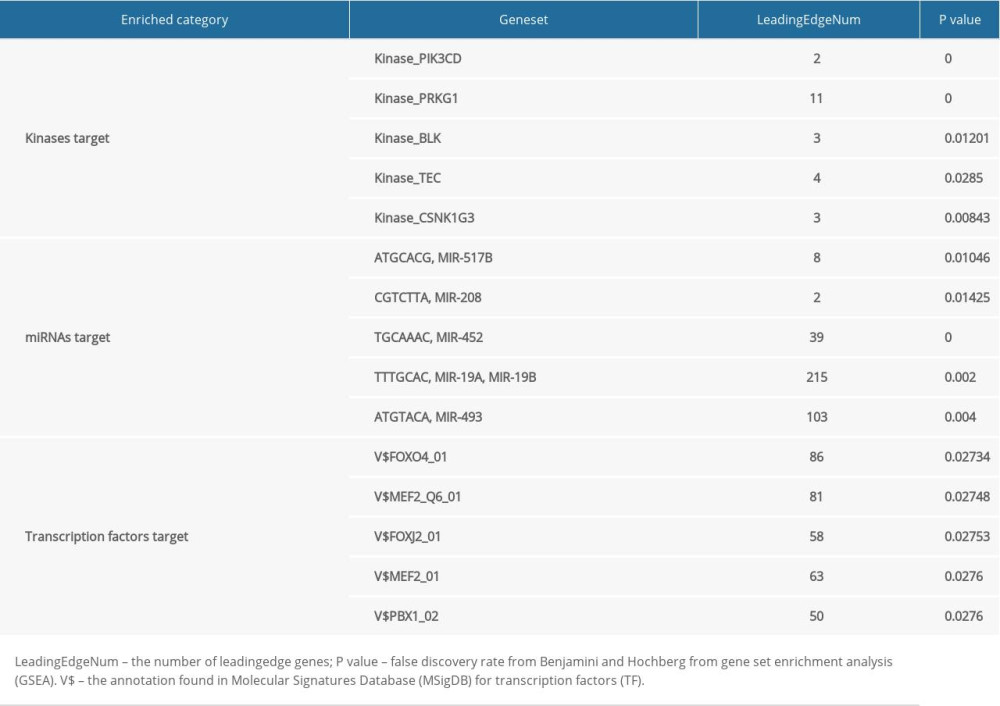 Table 2. The kinase, miRNA, and transcription factor-target networks of CUBN in CRC (LinkedOmics).
Table 2. The kinase, miRNA, and transcription factor-target networks of CUBN in CRC (LinkedOmics). Table 1. Enrichment analysis of CUBN and its significant neighborhood genes altered in CRC as determined using DAVID.
Table 1. Enrichment analysis of CUBN and its significant neighborhood genes altered in CRC as determined using DAVID. Table 2. The kinase, miRNA, and transcription factor-target networks of CUBN in CRC (LinkedOmics).
Table 2. The kinase, miRNA, and transcription factor-target networks of CUBN in CRC (LinkedOmics). In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








