04 July 2020: Clinical Research
Implementation of Thromboelastometry for Coagulation Management in Isolated Traumatic Brain Injury Patients Undergoing Craniotomy
Marius Rimaitis1ABCDEFG*, Diana Bilskienė1ABE, Tomas Tamošuitis2BE, Rimantas Vilcinis2BDE, Kęstutis Rimaitis1DE, Andrius Macas1ADEGDOI: 10.12659/MSM.922879
Med Sci Monit 2020; 26:e922879
Abstract
BACKGROUND: Coagulopathy (CP) is a modifiable factor linked with secondary brain damage and poor outcome of traumatic brain injury (TBI). A shift towards goal-directed coagulation management has been observed recently. We investigated whether rotational thromboelastometry (ROTEM) based management could be successfully implemented in TBI patients and improve outcomes.
MATERIAL AND METHODS: A prospective, case-control study was performed. Adult patients with isolated TBI requiring craniotomy were included in this study. All patients underwent standard coagulation tests (SCT). Patients were identified as either in control group or in case group. Patients in the case group were additionally tested with ROTEM to specify their coagulation status. Management of the patients in the control group was based on SCT, whereas management of patients in the case group was guided by ROTEM. Outcome measures were as follows: CP rate, protocol adhesion, blood loss, transfusions, progressive hemorrhagic injury (PHI), re-intervention, Glasgow coma score (GCS) and Glasgow outcome score (GOS) at discharge, and in-hospital mortality.
RESULTS: There were 134 patients enrolled (65 patients in the control group and 69 patients in the case group). Twenty-six patients in the control group (40%) were found to be coagulopathic (control-CP subgroup) and 34 patients in the case group (49.3%) were found to be coagulopathic (case-CP subgroup). Twenty-five case-CP patients had ROTEM abnormalities triggering protocolized intervention, and 24 of them were treated. Overall ROTEM-based protocol adhesion rate was 85.3%. Postoperative ROTEM parameters of case-CP patients significantly improved, and the number of coagulopathic patients decreased. The incidence of PHI (control versus case group) and neurosurgical re-intervention (control-CP versus case-CP subgroup) was in favor of ROTEM guidance (P<0.05). Mortality and GCS and GOS at discharge did not differ significantly between groups.
CONCLUSIONS: ROTEM led to consistent coagulation management, improved clot quality, and decreased incidence of PHI and neurosurgical re-intervention. Further studies are needed to confirm benefits of ROTEM in cases of TBI.
Keywords: Blood Coagulation, Brain Injuries, Intracranial Hemorrhage, Traumatic, Thrombelastography, Blood Coagulation Disorders, Blood Coagulation Tests, Blood Transfusion, Brain Injuries, Traumatic, Case-Control Studies, Craniotomy, Hemorrhage, Prospective Studies
Background
Traumatic brain injury (TBI) remains a major cause of preventable death and disability [1]. Despite relatively low blood loss, coagulopathy (CP) is a common finding in patients with blunt isolated TBI. Its association with secondary brain damage and poor clinical outcome has been reported [2–5]. Mechanisms responsible for the development of CP include increased tissue factor release, disseminated intravascular coagulation, quantitative or qualitative platelet deficits, and activation of protein C pathways [2,6]. Traditionally, standard coagulation tests (SCT) are used to identify CP. However, they often provide insufficient information on global hemostasis. Viscoelastic tests, such as rotational thromboelastometry (ROTEM), are becoming increasingly implemented in clinical practice [7,8]. They reflect functional coagulation status of whole blood in a real-time manner from initiation to fibrinolysis, and they provide the possibility of goal-directed therapy. Evidence from clinical studies in trauma and cardiac surgery highlight decreased perioperative bleeding and blood product use in patients managed based on a goal-directed approach [9–11]. The interest in whether such an approach could be applicable to neurosurgical patients is increasing [12–14]. Theoretically, targeted optimization of coagulation could lead to improved outcome. However, viscoelastic tests have not yet been tested in controlled studies to confirm their possible benefit for targeted hemostatic therapy in neurosurgical patients with TBI.
The aim of our study was to determine whether ROTEM guidance could be reasonably implemented in perioperative coagulation management of patients with isolated TBI undergoing craniotomy, and lead to decreased incidence of progressive hemorrhagic injury, lower need for re-intervention, and better clinical outcome.
Material and Methods
COAGULATION TESTING IN THE CONTROL GROUP AND THE CASE GROUP:
Coagulation testing of the control group was limited to standard coagulation tests (SCT): prothrombin time index (PTI), international normalized ratio (INR), partial thromboplastin time (APTT), platelet count (PLT), and fibrinogen concentration (FIB). Management of the case group was run by anesthesiology staff familiar with ROTEM. These patients were additionally tested with basic ROTEM assays (extrinsic coagulation pathway thromboelastometry [EXTEM], intrinsic coagulation pathway thromboelastometry [INTEM], fibrin polymerization thromboelastometry [FIBTEM]) (ROTEM® delta, TEM International GmbH, Munich, Germany). EXTEM with antifibrinolytic (APTEM) was performed in presence of abnormal basic ROTEM assays. The first venous blood sample for SCT (control and case groups) and ROTEM (case group only) was taken into citrated blood collection tubes preoperatively, before any blood product transfusions, following the decision to perform urgent craniotomy, and sent to the laboratory for analysis. Coagulopathy was defined as any abnormality on SCT (PTI <70%, INR >1.2, APTT >38 seconds, FIB <2.0 g/L, PLT <100×109/L) and/or ROTEM (EXTEM: clotting time [CT] >80 seconds, clot formation time [CFT] >159 seconds, clot amplitude 10 minutes after CT [A10] <45 mm, maximum clot firmness [MCF] <55 mm; INTEM: CT >240 seconds, CFT >110 seconds, A10 <45 mm, MCF <55 mm; FIBTEM: A10 <7 mm, MCF <9 mm; signs of hyperfibrinolysis). Hyperfibrinolysis was defined as maximum lysis [ML] ≥15% on EXTEM, INTEM or FIBTEM, and/or better APTEM parameters in presence of pathologic EXTEM readings. Subsequently, control and case groups were divided into subgroups: control-CP (abnormal SCT) and control-N (normal SCT), as well as case-CP (abnormal SCT and/or ROTEM) and case-N (normal SCT and ROTEM).
INTRAOPERATIVE PERIOD:
All patients underwent balanced general endotracheal anesthesia according to the American Society of Anesthesiologists (ASA) standards. Invasive hemodynamic monitoring was used and any hypotension (systolic blood pressure <90 mmHg) was treated promptly with fluids and/or vasopressors. Blood loss was estimated by clinical observation of the patient, surgical field, gauzes used, and blood volume collected in the suction canister. Point-of-care hemoglobin checks were performed to guide red blood cell transfusions. Red blood cells were transfused in the presence of hemoglobin <90 g/L. Coagulation management of control group patients was not protocolized and was based on SCT results and clinical judgement. Coagulation management of case group patients was guided by thromboelastometry according to a predefined protocol (Figure 1) [6,9,10,19–21] and clinical judgement. The use of intravenous fluids, blood products, coagulation factor concentrates, antifibrinolytics, and estimated blood loss were registered.
POSTOPERATIVE PERIOD:
Following surgery, all patients were transferred to the specialized neurosurgical intensive care unit and treated according to contemporary Brain Trauma Foundation guidelines [22]. Postoperative blood sampling was performed following the completion of surgery. Postoperative head computed tomography (CT) scans were performed within 6 hours following craniotomy and evaluated by a qualified radiologist unaware of the study. Further head CT scans were performed according to patient‘s neurological condition. Progressive hemorrhagic, ischemic, and edematous changes were registered. Description of any new, increased, or recurrent hemorrhagic lesions was considered as progressive hemorrhagic injury (PHI). Postoperative neurological outcome was assessed by Glasgow coma score (GCS) on postoperative days 3, 7, and at discharge, and Glasgow outcome score (GOS) at discharge [23,24]. GOS of more than 3 was considered as favorable outcome.
STUDY OUTCOMES:
Primary outcomes were GCS and GOS at discharge, and in-hospital mortality. Secondary outcome measures included the prevalence of coagulopathy, ROTEM protocol adhesion, blood loss, transfusions, the incidence of progressive hemorrhagic injury, and re-intervention.
STATISTICAL ANALYSIS:
The normality of quantitative data was assessed using the Kolmogorov-Smirnov test. Normally distributed variables were compared using Student’s
RESULTS:
From 2018 until 2020, 1293 patients were admitted to the Brain Trauma Department, and 256 craniotomies were performed to treat TBI. During the recruitment period, a total of 134 patients with severe isolated TBI needing craniotomy and matching the inclusion criteria, were enrolled (65 patients in the control group and 69 patients in the case group). Patient demographic and trauma characteristics were comparable between the control and case group as well as subgroups of normal and coagulopathic patients (control-N versus case-N, and control-CP versus case-CP), and are presented in Table 1.
THE PREVALENCE OF PREOPERATIVE COAGULOPATHY:
Overall coagulation profile of study patients according to SCT did not differ significantly between groups. The prevalence of SCT abnormalities was comparable between the control group and the case group (40%, n=26 versus 36.2%, n=25). Coagulopathic controls formed the control-CP subgroup (n=26). The most common SCT abnormality was decreased PTI. Selective analysis of separate SCT abnormalities revealed that deviations from test reference values were mostly mild to moderate (Table 2).
ROTEM abnormalities were present in 25 out of 69 patients (36.2%) in the case group. Nine patients were coagulopathic according to SCT, but not ROTEM. Another 9 patients had normal SCT despite abnormal ROTEM. Subsequently, any abnormality on preoperative coagulation tests (SCT and/or ROTEM) was found in 49.3% of the case group patients and formed the case-CP subgroup (n=34). Coagulation profile agreement (positive or negative) between SCT and ROTEM was found in 51 patients (73.9%) in the case group.
COAGULOPATHY MANAGEMENT:
Out of 26 patients in the control-CP subgroup, 17 patients (65.4%) received procoagulant intervention either with tranexamic acid (TXA), blood products, or coagulation factor concentrates. Nine patients (34.6%) in the control-CP subgroup did not receive any procoagulant intervention despite observed abnormalities in standard coagulation tests as the treating anesthesiologist considered them clinically insignificant.
Management of case-CP patients (n=34) was guided by our ROTEM-based protocol. Nine patients (26.5%) did not need intervention according to ROTEM. Another 25 patients (73.5%) in the case-CP subgroup had ROTEM abnormalities triggering protocolized treatment. All but one patient (24 out of 25 patients, 96%) received procoagulant intervention either with TXA, blood products, or coagulation factor concentrates. Five out of 7 patients with ROTEM signs of hypofibrinogenemia received cryoprecipitate, and 2 were treated with fresh frozen plasma (which was not first-line choice according to protocol). One patient received platelets due to low platelet count despite normal ROTEM traces. Two patients did not receive plasma despite prolonged clotting times on ROTEM as there was no significant intraoperative bleeding. TXA was given to 18 patients (52.9%) in the case-CP subgroup. All patients corresponding with our definition of hyperfibrinolysis (n=9), received TXA. Administration of TXA in patients without evidence of hyperfibrinolysis was not considered as protocol violation. Complete adherence to our ROTEM-based protocol was found in 85.3% (29 out of 34 patients).
Estimated blood loss, transfusions, and procoagulant interventions in the study groups and subgroups are summarized in Table 3. As expected, coagulopathic patients received more blood products as compared to patients with normal coagulation profiles within both the control group and the case group. No significant between-group differences were found with regard to transfusions.
PREOPERATIVE VERSUS POSTOPERATIVE COAGULATION PARAMETERS:
Summarized preoperative and postoperative hematologic parameters of the control group and the case group are presented in Tables 4 and 5. Clear negative trends were observed in postoperative versus preoperative SCT in both the control group and the case group. However, significant improvements in postoperative ROTEM parameters (INTEM CT and CFT, EXTEM CT, CFT, A10 and MCF, FIBTEM A10 and ML) were observed among the case-CP subgroup patients. The number of patients with abnormal ROTEM, significantly decreased. In contrast to the control group and the case-N subgroup in which postoperative fibrinogen levels decreased as compared with preoperative measures, we observed an increase in fibrinogen levels and significantly improved FIBTEM A10 in the case-CP subgroup, suggesting better recognition and treatment of fibrinogen deficiency.
There were no significant differences in terms of preoperative or postoperative SCT results between control and case subgroups with and without coagulopathy (control-N versus case-N, and control-CP versus case-CP).
OUTCOME MEASURES:
The incidence of progressive hemorrhagic injury was significantly lower when all the patients in the case group were compared to all the patients in the control group (47.7% versus 30.4%, P=0.04). Lower need for neurosurgical re-interventions was found in coagulopathic case subgroup (case-CP), as compared to coagulopathic control subgroup (control-CP), P=0.020. The chance of favorable outcome (according to GOS) was higher (but not statistically significant) among the case-CP subgroup as compared to control-CP subgroup (38.2% versus 15.4%, P=0.052). There were no significant differences regarding other outcome parameters (Table 6).
Binary logistic regression analysis identified age, higher ISS, presence of brain parenchyma lesion, pupil abnormalities, midline shift >5 mm, higher Rotterdam score, lower admission GCS, higher ASA class, and presence of progressive hemorrhagic or ischemic injury as factors significantly associated with poor GOS in the control group and the case group. Aforementioned variables did not differ between the groups significantly (Table 1). Interestingly, the presence of coagulopathy on standard coagulation tests was found to be associated with poor outcome in the control group only (odds ratio [OR] 7.02 [95% confidence interval [CI] 1.63–30.24], P=0.009) whereas in the case group, there was no such association (OR 1.66 [95% CI 0.56–4.95], P=0.193). Subsequent multivariate logistic regression analysis confirmed coagulopathy on SCT as an independent risk factor for poor GOS in the control group, but not in the case group.
Discussion
For many years, observational studies have linked brain trauma with coagulation abnormalities and poor clinical outcome [2–4]. The lack of guidance to the clinician on the management of these changes in the perioperative setting is widely recognized, but there are no controlled trials to address this issue.
Our study is the first prospective controlled clinical trial that aimed to determine possible benefits of a protocolized ROTEM-based approach to coagulation management of brain trauma patients during craniotomy. We chose a specific cohort of isolated traumatic brain injury (TBI) patients who needed craniotomies to evacuate hematomas and/or to decompress brain to prevent herniation. We considered them at high perioperative bleeding risk and very high predisposition to possible secondary brain damage. Aiming to specifically address coagulation abnormalities directly associated with brain trauma, we did not include patients with polytrauma, known use of anticoagulants or antiplatelet use, or significant hematologic or liver diseases.
The overall coagulation profile of our TBI patients was good, and this finding has been reported by other authors [3,25]. The prevalence of coagulopathy on preoperative coagulation tests, depending on testing approach and definition, varied from 36.2% to 49.3%, and was similar to the numbers reported by other studies and systematic reviews [2,5,26]. The fact that standard coagulation test result deviations from normal values in selected patients with coagulopathy were mostly mild to moderate, underlines the complexity of clinical decision-making in the neurosurgical setting associated with hazards of bleeding into a closed compartment.
Our modified coagulation management protocol was based on published algorithms and guidelines [6,9,10,19–21]. However, as they were not specifically developed and tested with neurosurgical patients, we chose a higher target for MCF (55 mm). It should be noted that optimal cutoff values for intervention in neurosurgical setting have not yet been defined neither for SCT nor ROTEM except for recommendations to target test normalization and platelet count higher than 80–100×109/L, which is also debatable.
A pilot study by Gratz et al. [12] aiming to evaluate ROTEM-based algorithm implementation success in TBI patients found high protocol adherence rates of 88% to 91%. However, that study did not include any clinical outcomes. Our ROTEM-based protocol was also followed successfully (adherence rate: 85.3%) and resulted in significantly improved clot quality as confirmed by postoperative thromboelastometric results among the patients in the case-CP subgroup.
The fact that 96% of patients with ROTEM abnormalities triggering protocolized intervention received treatment as compared with 65% of patients treated in the control-CP subgroup, confirms that clear guidance to the clinician results in more consistent treatment. We believe that the ROTEM-based protocol enabled us to objectively differentiate between clinical significance of SCT abnormalities, as well as identify patients which would have been overlooked by conventional testing. Furthermore, it enabled detection of hyperfibrinolysis and targeted antifibrinolytic therapy.
A negative trend in standard coagulation tests despite improvements of ROTEM results reflects that the whole blood viscoelastometric tests represent patient coagulation status from a different perspective and the overall clot quality in some individuals may be adequate or even improved despite abnormalities found in standard coagulation tests. Clinical relevance of thromboelastometric versus standard coagulation test changes at different points in the perioperative course of patients with isolated TBI is a complicated issue and requires further investigations to identify which SCT and ROTEM alterations, and to what extent, require prompt intervention, and which can be tolerated.
Lower incidence of progressive hemorrhagic injury and lower need for neurosurgical re-intervention are promising findings. However, in our study, statistically significant differences were found among certain patient groups only. Primary outcomes (GCS, GOS, and mortality), despite being in favor of the case group, did not differ significantly.
Interestingly, logistic regression analysis revealed that the presence of coagulopathy according to SCT increased the odds of poor GOS by 7 times in the control group whereas in the case group, there was no such association, suggesting that goal-directed coagulation management could affect (or even eliminate) coagulopathy as a factor leading to poor outcome of TBI patients.
We have to admit that our clinical study was not randomized, and the risk of bias could not be excluded. Epidural hematoma, as predominant injury, although not significantly, was more frequent in the case group, and could have influenced the results. Study groups were recruited irrespective of the initial coagulation status and therefore, a fraction of patients required no therapeutic interventions. Preliminary screening for standard coagulation test abnormalities before inclusion might have purified the study sample even further. However, as anticipated, we found a considerable proportion of patients who were not coagulopathic according to SCT, but who were coagulopathic on ROTEM, and vice versa. Universal screening with ROTEM would have resulted in loss of the control group, as the investigators considered withholding of available and potentially beneficial information unethical. We can only guess how ROTEM could have influenced the management of patients in the control group. Ethical issues regarding the consent to additional blood sampling for ROTEM also existed. Our institution was only equipped with thromboelastometer in the central laboratory, therefore thromboelastometry could not be performed at bedside. The investigators were able to connect to the laboratory and observe real-time development of thromboelastograms, but the need to send the sample to the laboratory could have eliminated the possible benefits of faster turnaround times as compared to standard laboratory tests. On the other hand, the procedure handling was performed by certified laboratory staff unaware of the study. As aforementioned, the control group patients were not analyzed with ROTEM, therefore between-group comparative analysis of thromboelastograms was not available. During the recruitment period of the study, fibrinogen concentrate was not available in our institution and prothrombin complex concentrate was reserved mainly for vitamin-K antagonist reversal, therefore cryoprecipitate was considered the first-line choice for fibrinogen replacement, whereas fresh frozen plasma was used when clotting initiation was delayed.
Conclusions
Implementation of a ROTEM-based protocol led to consistent coagulation management, improved clot quality, and decreased incidence of PHI and neurosurgical re-intervention in patients with isolated TBI. However, our study failed to demonstrate significant differences between the control group and the case group in terms of GCS, GOS, and mortality. Further, well-organized studies are needed to confirm benefits of ROTEM in the setting of brain trauma.
Tables
Table 1. Patient demographic and injury characteristics.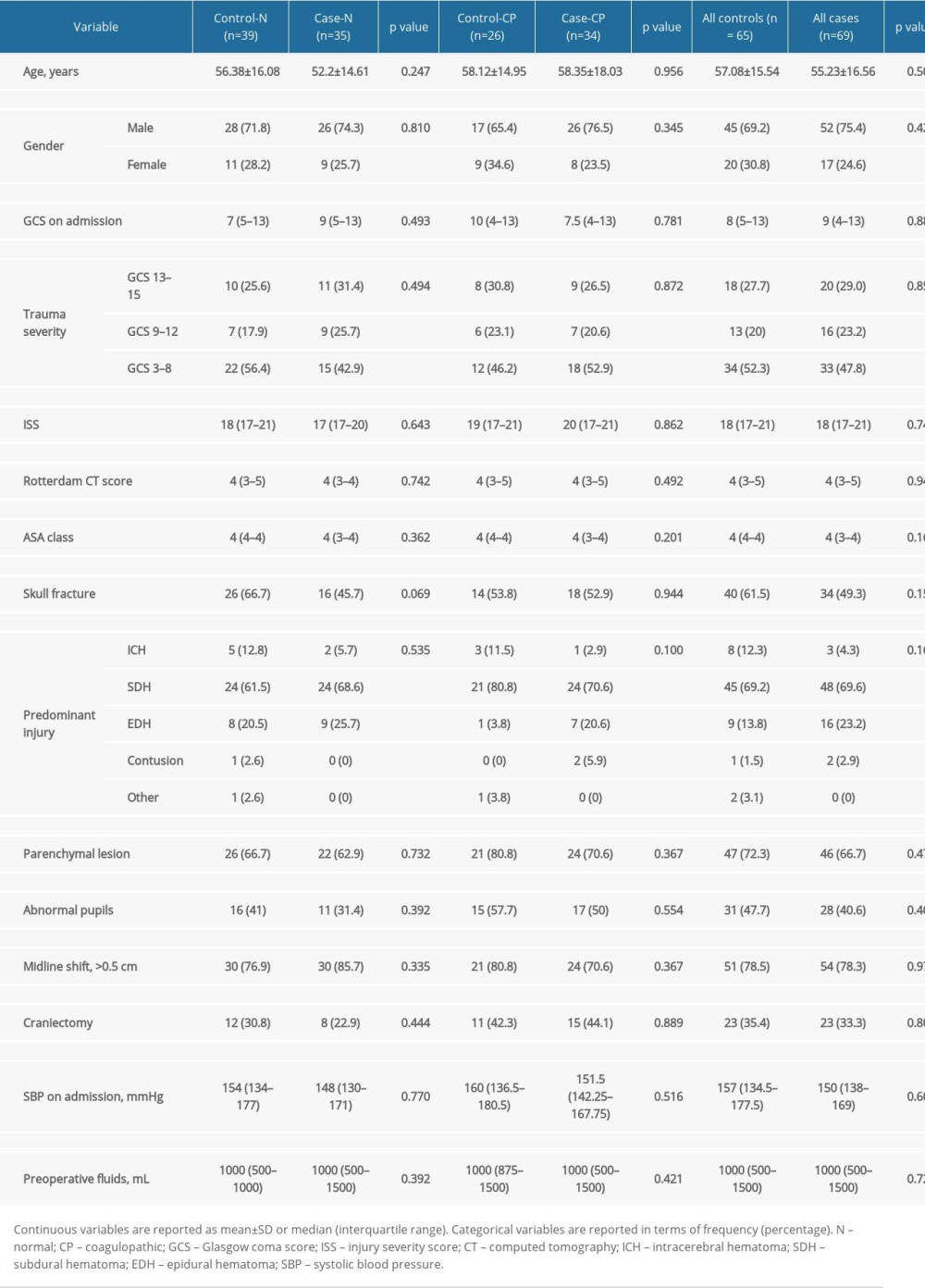 Table 2. Patients with abnormal preoperative SCT and the extent of test abnormalities.
Table 2. Patients with abnormal preoperative SCT and the extent of test abnormalities.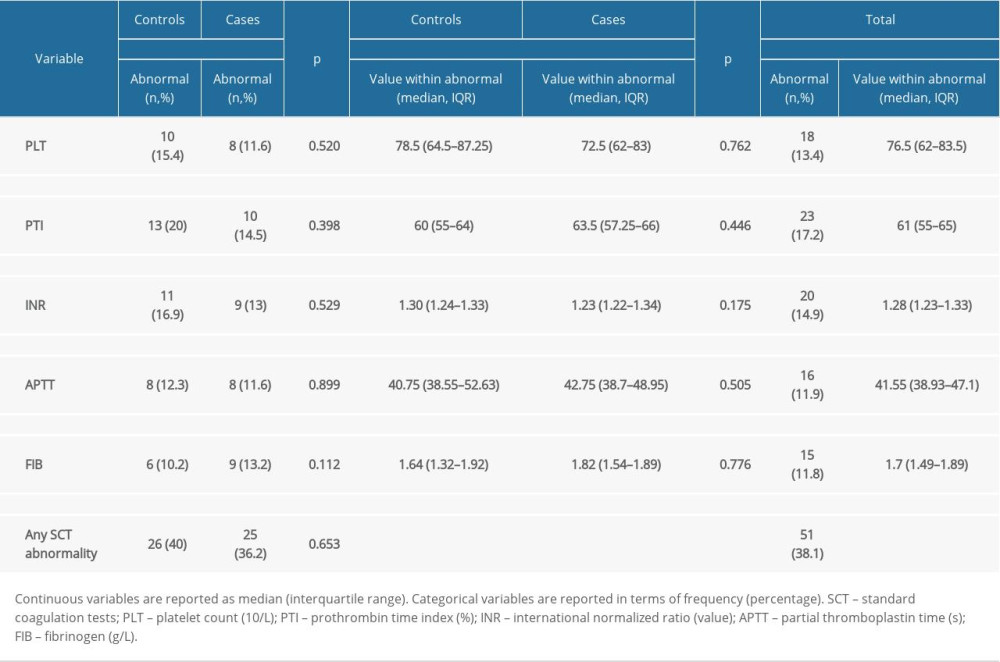 Table 3. Blood loss, blood product use and procoagulant interventions among the control and case group patients.
Table 3. Blood loss, blood product use and procoagulant interventions among the control and case group patients.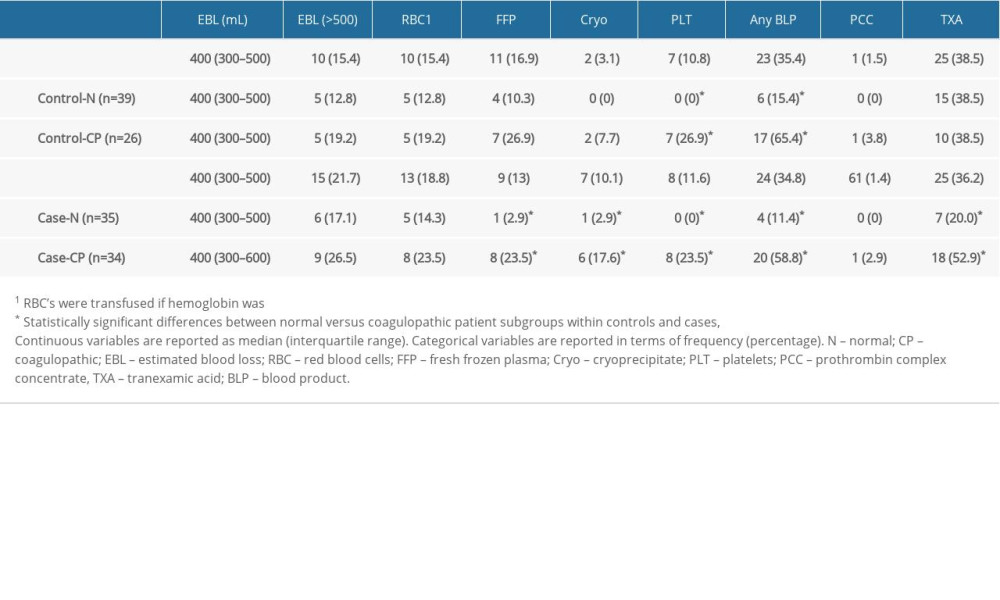 Table 4. Summarized hematologic parameters of the control group patients.
Table 4. Summarized hematologic parameters of the control group patients.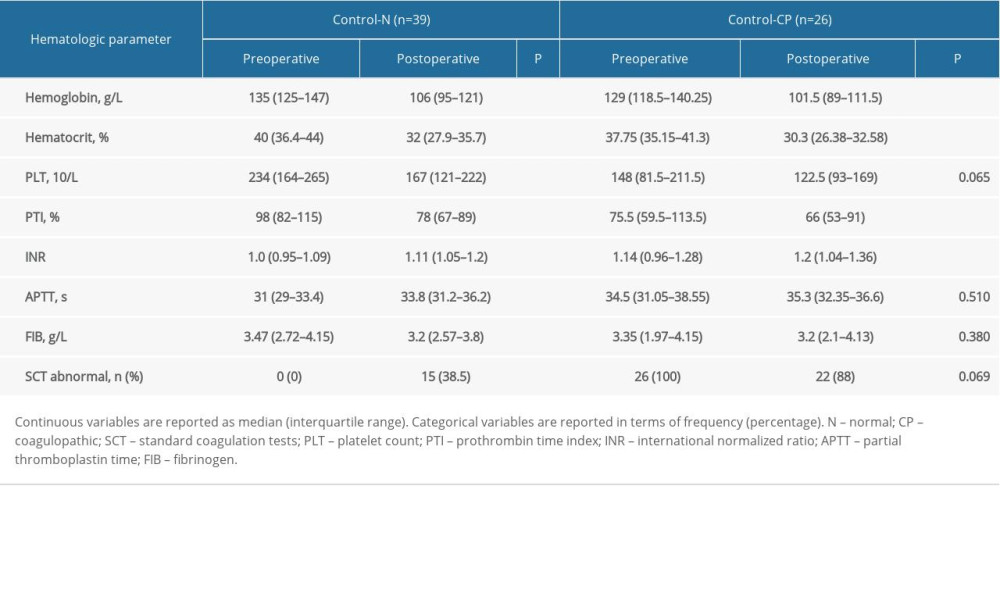 Table 5. Summarized hematologic parameters of the case group patients.
Table 5. Summarized hematologic parameters of the case group patients.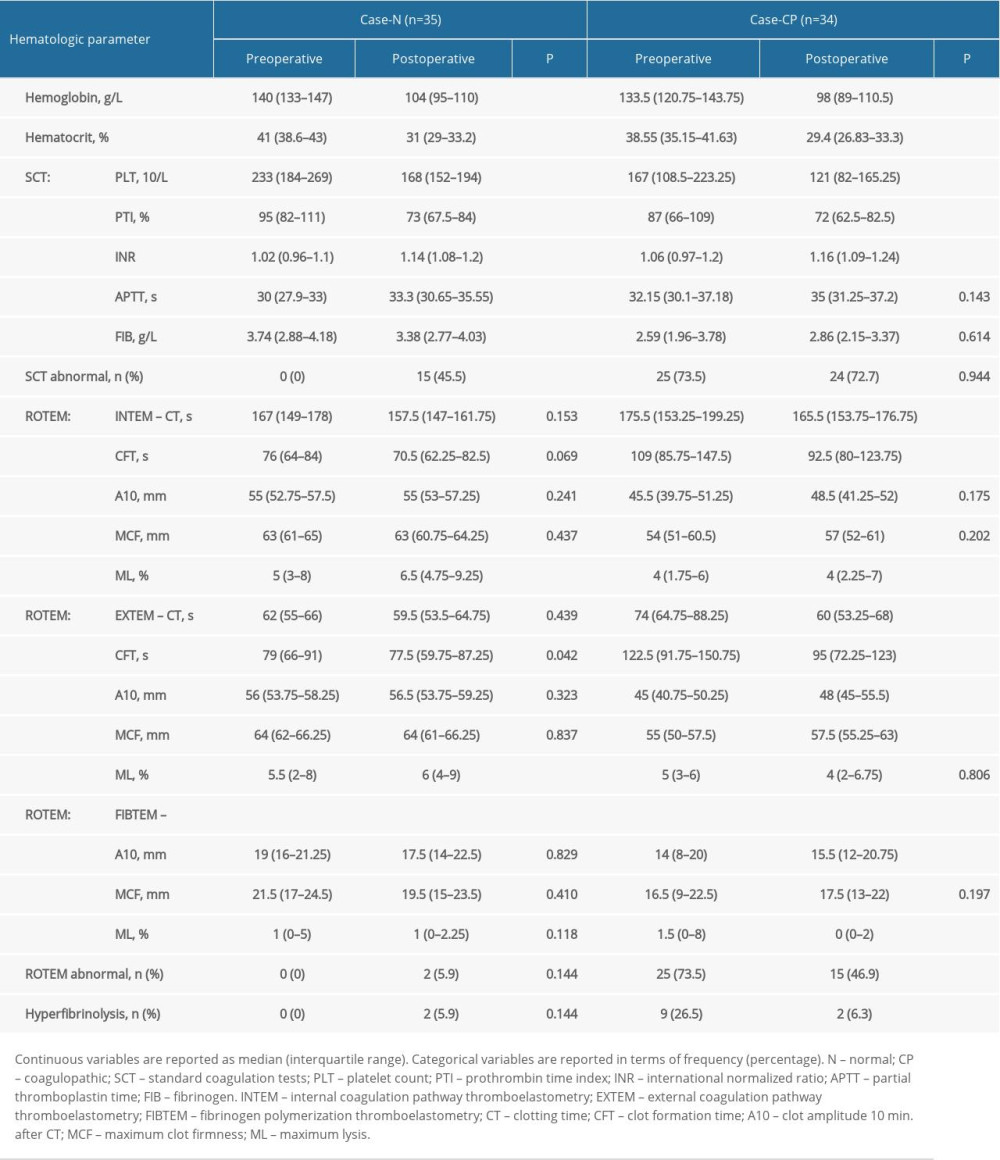 Table 6. Outcome parameters of the control and case group patients.
Table 6. Outcome parameters of the control and case group patients.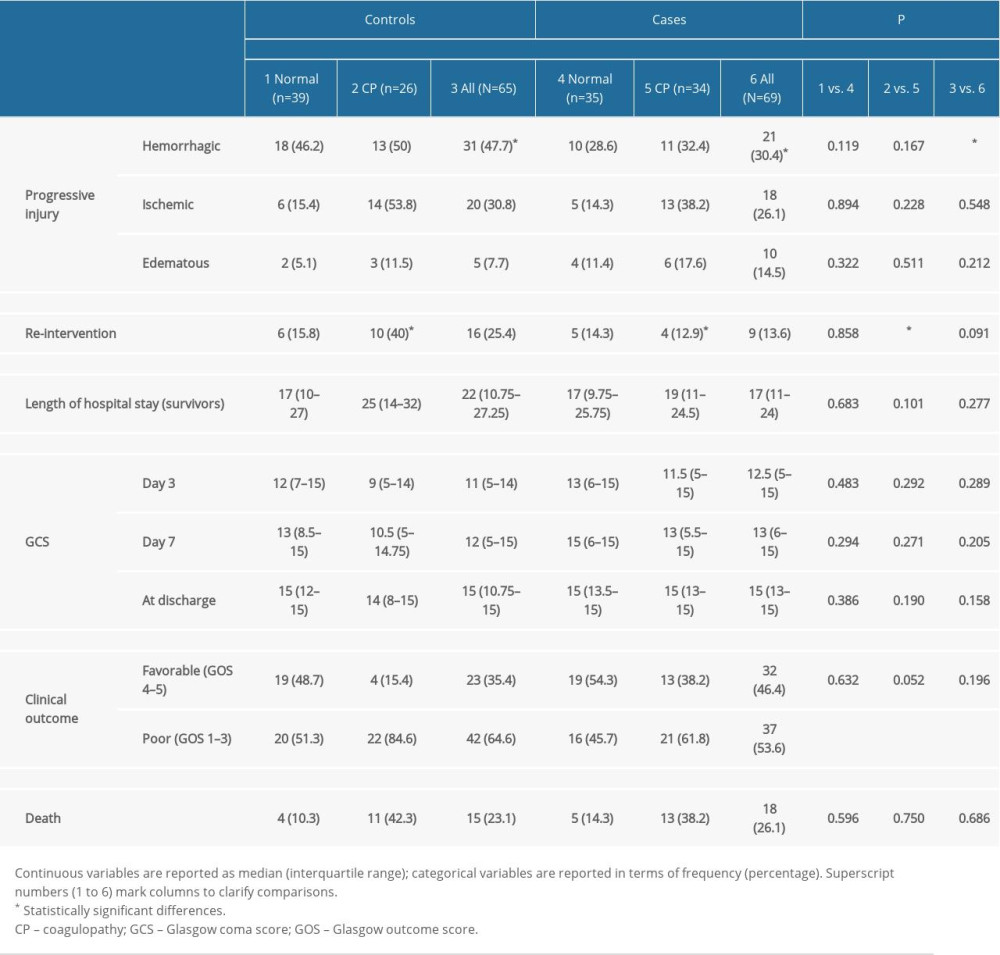
References
1. Faul M, Xu L, Wald MM, Coronado V, Traumatic brain injury in the United States: Emergency Department visits, hospitalizations and deaths: Inj Prev, 2010; 16(Suppl 1); A268
2. Harhangi BS, Kompanje EJ, Leebeeck FW, Maas AIR, Coagulation disorders after traumatic brain injury: Acta Neurochir, 2008; 150; 165-75
3. De Oliveira Manoel AL, Neto AC, Veigas PV, Rizoli S, Traumatic brain injury associated coagulopathy: Neurocrit Care, 2015; 22; 34-44
4. Kunio NR, Differding JA, Watson KM, Thrombelastography-identified coagulopathy is associated with increased morbidity and mortality after traumatic brain injury: Am J Surg, 2012; 203; 584-88
5. Schochl H, Solomon C, Traintinger S, Thromboelastometric (ROTEM) findings in patients suffering from isolated severe traumatic brain injury: J Neurotrauma, 2011; 28; 2033-41
6. Maegele M, Schochl H, Menovsky T, Coagulopathy and haemorrhagic progression in traumatic brain injury: Advances in mechanisms, diagnosis, and management: Lancet Neurol, 2017; 16; 630-47
7. Rossaint R, Bouillon B, Cerny V, The European guideline on management of major bleeding and coagulopathy following trauma: Fourth edition: Crit Care, 2016; 20; 100
8. Gonzalez E, Moore EE, Moore HB, Management of trauma induced coagulopathy with thrombelastography: Crit Care Clin, 2017; 33; 119-34
9. Wikkelsoe A, Wetterslev J, Moller AM, Afshari A, Thromboelastography (TEG) or thromboelastometry (ROTEM) to monitor haemostatic treatment versus usual care in adults or children with bleeding (review): Cochrane Database Syst Rev, 2016; 8; CD007871
10. Schochl H, Nienaber U, Hofer G, Goal-directed coagulation management of major trauma patients using thromboelastometry (ROTEM)-guided administration of fibrinogen concentrate and prothrombin complex concentrate: Crit Care, 2010; 14; R55
11. Fahrendorff M, Oliveri RS, Johansson PI, The use of viscoelastic haemostatic assays in goal-directing treatment with allogenic blood products – A systematic review and meta-analysis: Scand J Trauma Resusc Emerg Med, 2017; 25(1); 39
12. Gratz J, Guting H, Thorn S, Protocolised thromboelastometric-guided haemostatic management in patients with traumatic brain injury: A pilot study: Anaesthesia, 2019; 74; 883-90
13. Ellenberger C, Garofano N, Barcelos G, Assessment of Haemostasis in patients undergoing emergent neurosurgery by rotational Elastometry and standard coagulation tests: A prospective observational study: BMC Anesthesiology, 2017; 17; 146
14. Benyon C, Unterberg AW, Sakowitzv OW, Point of care coagulation testing in neurosurgery: J Clin Neurosci, 2015; 22; 252-57
15. Schwarze J: Grundlagen der Statistik 2, 1993, Herne/Berlin, Neue Wirtschaftsbriefe [in German]
16. Gennarelli TA, Wodzin E, AIS 2005: A contemporary injury scale: Injury, 2006; 37(12); 1083-91
17. Copes WS, Champion HR, Sacco WJ, The injury severity score revisited: J Trauma, 1988; 28(1); 69-77
18. Maas AI, Hukkelhoven CW, Marshall LF, Steyerberg EW, Prediction of outcome in traumatic brain injury with computed tomographic characteristics: A comparison between the computed tomographic classification and combinations of computed tomographic predictors: Neurosurgery, 2006; 57(6); 1173-82
19. Lier H, Vorweg M, Hanke A, Gorlinger K, Thromboelastometry guided therapy of severe bleeding. Essener Runde algorithm: Hamostaseologie, 2013; 33(1); 51-61
20. Schochl H, Maegele M, Solomon C, Early and individualized goal-directed therapy for trauma-induced coagulopathy: Scand J Trauma Resusc Emerg Med, 2012; 20; 15
21. Inaba K, Rizoli S, Veigas PV, 2014 Consensus conference on viscoelastic test-based transfusion guidelines for early trauma resuscitation: Report of the panel: J Trauma Acute Care Surg, 2015; 78; 1220-29
22. Carney N, Totten AM, O’Reilly C, Guidelines for the management of severe traumatic brain injury, fourth edition: Neurosurgery, 2017; 80(1); 6-15
23. Teasdale G, Jennett B, Assessment of coma and impaired consciousness. A practical scale: Lancet, 1974; 2(7872); 81-84
24. Jennett B, Bond M, Assessment of outcome after severe brain damage: Lancet, 1975; 1(7905); 480-84
25. Lee TH, Hampton DA, Diggs BS, Traumatic brain injury is not associated with coagulopathy out of proportion to injury in other body regions: Trauma Acute Care Surg, 2014; 77; 67-72
26. Yuan Q, Yu J, Wu X, Prognostic value of coagulation tests for in-hospital mortality in patients with traumatic brain injury: Scand J Trauma Resusc Emerg Med, 2018; 26; 3
Tables
 Table 1. Patient demographic and injury characteristics.
Table 1. Patient demographic and injury characteristics. Table 2. Patients with abnormal preoperative SCT and the extent of test abnormalities.
Table 2. Patients with abnormal preoperative SCT and the extent of test abnormalities. Table 3. Blood loss, blood product use and procoagulant interventions among the control and case group patients.
Table 3. Blood loss, blood product use and procoagulant interventions among the control and case group patients. Table 4. Summarized hematologic parameters of the control group patients.
Table 4. Summarized hematologic parameters of the control group patients. Table 5. Summarized hematologic parameters of the case group patients.
Table 5. Summarized hematologic parameters of the case group patients. Table 6. Outcome parameters of the control and case group patients.
Table 6. Outcome parameters of the control and case group patients. Table 1. Patient demographic and injury characteristics.
Table 1. Patient demographic and injury characteristics. Table 2. Patients with abnormal preoperative SCT and the extent of test abnormalities.
Table 2. Patients with abnormal preoperative SCT and the extent of test abnormalities. Table 3. Blood loss, blood product use and procoagulant interventions among the control and case group patients.
Table 3. Blood loss, blood product use and procoagulant interventions among the control and case group patients. Table 4. Summarized hematologic parameters of the control group patients.
Table 4. Summarized hematologic parameters of the control group patients. Table 5. Summarized hematologic parameters of the case group patients.
Table 5. Summarized hematologic parameters of the case group patients. Table 6. Outcome parameters of the control and case group patients.
Table 6. Outcome parameters of the control and case group patients. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387









![ROTEM-based algorithm for goal-directed coagulation management in traumatic brain injury [6,9,10,19–21]. CT – clotting time; A10 – clot amplitude 10 min after CT; ML – maximum lysis; MCF – maximum clot firmness; PLT – platelets; TXA – tranaxemic acid; FC – fibrinogen concentrate; Cryo – cryoprecipitate; PCC – prothrombin complex concentrate; FPP – fresh frozen plasma. * Depending on the availability of coagulation factor concentrates.](https://jours.isi-science.com/imageXml.php?i=medscimonit-26-e922879-g001.jpg&idArt=922879&w=1000)