01 September 2020: Lab/In Vitro Research
Atorvastatin Enhances Effects of Radiotherapy on Prostate Cancer Cells and Xenograft Tumor Mice Through Triggering Interaction Between Bcl-2 and MSH2
Zhenhua He1ABCDEFG*, Jingmin Yuan1BCDEF, Fuhui Shen2BCDF, Fangang Zeng2BCDF, Ping Qi3BCD, Zhiping Wang4BCF, Zhenxing Zhai4BFDOI: 10.12659/MSM.923560
Med Sci Monit 2020; 26:e923560
Abstract
BACKGROUND: Prostate cancer (PCa) is considered to be the 4th most common cancer in males in the world. This study aimed to explore effects of atorvastatin on colony formation of PCa cells and radio-resistance of xenograft tumor models.
MATERIAL AND METHODS: PCa cell lines, including PC3, DU145, and Lncap, were treated with irradiation (4 Gy) and/or atorvastatin (6 μg/mL). Cells were divided into tumor cell group, irradiation treatment group (IR group) and irradiation+atorvastatin treatment group (IR-AS group). Xenograft tumor mouse model was established. Plate clone formation assay (multi-target/single-hit model) was conducted to evaluate colony formation. Flow cytometry analysis was employed to detect apoptosis. Interaction between Bcl-2 and MSH2 was evaluated with immuno-fluorescence assay.
RESULTS: According to the plate colony formation assay and multi-target/single-hit model, IR-treatment significantly suppressed colony formation in PCa cells (including PC3, DU145, and Lncap cells) compared to no-IR treated cells (P<0.05). Atorvastatin remarkably enhanced inhibitive effects of irradiation on colony formation of PCa cells (P<0.05), however, the IR+AS group demonstrated no effects on apoptosis, comparing to IR group (P>0.05). Atorvastatin administration (IR+AS group) significantly reduced tumor size of IR-treated PCa cells-induced xenograft tumor mice (P<0.05). Bcl-2 interacted with MSH2 both in tumor tissues of xenograft tumor mice.
CONCLUSIONS: Atorvastatin administration inhibited colony formation in PCa cells and enhanced effects of radiotherapy on tumor growth of xenograft tumor mice, which might be associated with interaction between Bcl-2 and MSH2 molecule.
Keywords: beta-MSH, Drug Resistance, Prostatic Neoplasms, Radiotherapy, atorvastatin, Hydroxymethylglutaryl-CoA Reductase Inhibitors, MutS Homolog 2 Protein, Protein Interaction Maps, Proto-Oncogene Proteins c-bcl-2
Background
Clinically, prostate cancer (PCa) is considered to be the 4th most common cancer in males and the leading non-cutaneous tumor in males worldwide [1,2]. Meanwhile, PCa is also considered as the 6th most prevalent cause for cancer-associated death [3]. In the USA, appropriately 190 000 new PCa cases were diagnosed, causing about 26 000 death, according to the report of American Cancer Society [4]. Although the current therapeutic strategies, such as radiotherapy and chemotherapy, are effective and available for most patients; all of these therapeutic strategies always induce radio- or chemo-resistance and impact life-quality of PCa patients [5,6]. PCa radio-resistance usually causes relapse of PCa in a clinical setting, which might be associated with the inhibition of tumor cell apoptosis and modulation of tumor cell cycles [7,8]. Recent studies [9,10] reported that effects and clinical outcomes of radiotherapy (or radio-resistance of PCa) are closely associated with DNA repair processes of tumor cells [9,10]. Moreover, the apoptosis in tumor cells has also been proven to be correlated with DNA repair related molecules [11]. Therefore, we attempted to clarify the roles of DNA repair and Bcl-2 molecule in the PCa cell proliferation and tumor growth.
Statins, as a class of lipid-lowering drugs, could decrease the cancer-associated death of plenty cancers, such as PCa [12,13]. Among these statins, atorvastatin is one of the most commonly used statins for treating cancers clinically [14,15]. He et al. [16] proved that atorvastatin could promote the radio-sensitivity and trigger the tumor cell apoptosis, however, the effects-associated mechanism is also unclear. Previous studies [16] have reported that irradiation causes radio-resistance of cancer cells by suppressing apoptosis and activating autophagy. However, the other signaling pathways might also participate in the anti-tumor effects of atorvastatin and radio-resistance. Therefore, we speculated that atorvastatin might suppress PCa cells proliferation and inhibit tumor growth by modulating apoptosis-associated or DNA repair-associated mechanisms.
Material and Methods
CELL CULTURE, TREATMENT, AND TRIAL GROUPING:
In this study, human PCa cell lines, PC-3, DU145, and Lncap (Shanghai Cell Bank of China Academia Sinica, Shanghai, China), were utilized in the following experiments. DU145 cells were cultured in Dulbecco’s modified eagle medium (DMEM, Gibco BRL., Co., Ltd., Grand Island, NY, USA), while PC-3 and Lncap cells were cultured in Roswell park memorial institute 1640 (RPMI-1640, Gibco BRL., Co., Ltd.). All cells were cultured in aforementioned mediums supplementing with 1% penicillin-streptomycin (Beyotime Biotech., Shanghai, China) and containing 10% fetal bovine serum (FBS, Gibco BRL. Co. Ltd.) at 37°C in 5% CO2. All PC-3, DU145, and Lncap cells were treated with different dose of irradiation as followings: 2 Gy/min, 4 Gy/min, and 8 Gy/min.
For each cell line, cells were divided into 3 groups, including PCa cells without any treatment (PC3 group, DU145 group, Lncap group), irradiation administrated PCa cells (PC3+IR group, DU145+IR group, Lncap+IR group) and irradiation combining atorvastatin administrated PCa cells (PC3+IR+AS group, DU145+IR+AS group, Lncap+IR+AS group). Atorvastatin used in the aforementioned groups was purchased from Shanghai Yuanye Bio-Technology Co., Ltd. (Shanghai, China).
QUANTITATIVE REAL-TIME POLYMERASE CHAIN REACTION (QRT-PCR) ASSAY:
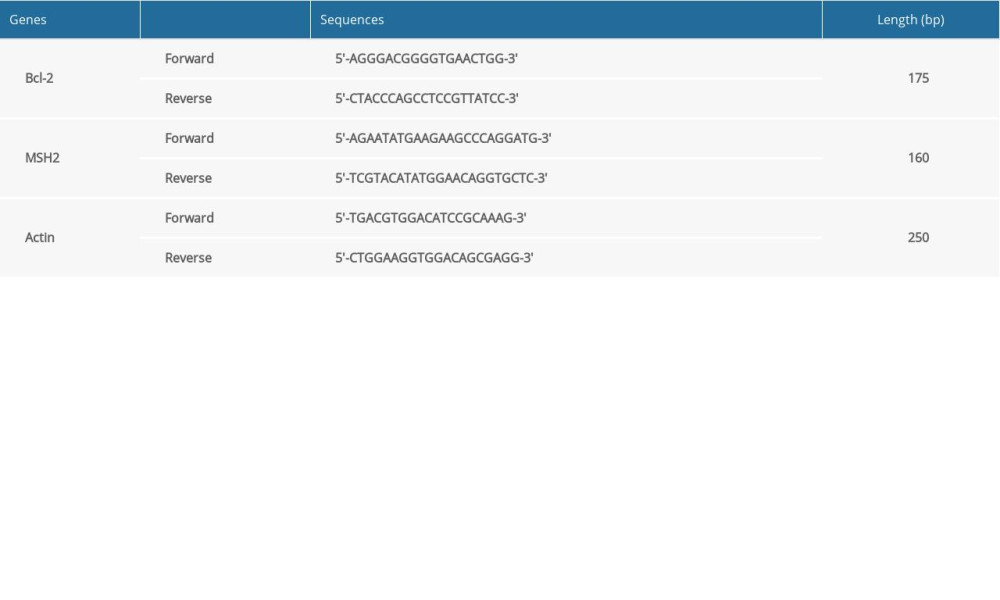
Total RNAs of tumor cells or tumor tissues were extracted using TRIzol and associated regents (Beyotime Biotech., Co., Ltd., Shanghai, China). Take the extracted RNAs as template, the complementary DNAs (cDNAs) were synthesized using Takara PrimeScript II 1st strand cDNA Synthesis Kit (Cat. No. 6210A/B, Takara, Dalian, China) according to the instruction of manufacturer. Also, take the synthesized cDNAs as template, mRNA levels of both Bcl-2 and MSH2 were determined using SYBR Green I PCR kit (Western Biotech.) with the specific primers (Table 1). The amplified genes were captured and analyzed using a professional gel scanning system (Mode: GDS8000, UVP, Sacramento, CA, USA) due to instruction of the equipment. The final levels of Bcl-2 and MSH2 mRNAs were calculated and analyzed using the 2−ΔΔct method [17].
PLATE CLONE FORMATION ASSAY:
PC-3, DU145, and Lncap cells were cultured and seeded onto 6-well plates (Costar-Corning, Corning, NY, USA) at final density of 1000 cells per well. When the aforementioned cells were adherent, cells were treated with HCl contained solvent control. When cells in 6-well plates growth to the density of 50% per cluster, which were washed with the phosphate-buffered saline (PBS) and fixed with 4% paraformaldehyde (Sigma-Aldrich, St. Louis, MO, USA). The cells were stained using 0.1% crystal violet (Sigma-Aldrich) and washed with PBS again. Eventually, the formed clones were observed using naked eye from the randomly selected fields (at least 6 fields) under the microscopy (100× magnification, Mode: CFI60, Nikon, Japan).
COLONY FORMATION ASSAY ON MULTI-TARGET/SINGLE-HIT MODEL:
In this study, the colony formation assay was also conducted on the multi-target/single-hit model. The atorvastatin (at final concentration 6 μg/mL) with mild cytotoxicity was selected for the following tests or experiments. The single cell suspensions for the PC3, DU145, and Lncap cells in logarithmic-phase were harvested and cultured in the 24-well cell culture plates at density of 200 cells/well. Each PCa cell (PC3, DU145, and Lncap) included 2 groups: IR group and IR+AS group. At 24 hours post the treatment, PCa cells were irradiated using irradiation (at dose of 0 Gy, 4 Gy and 8 Gy) for the following 10-day culture, fixed using the ethyl alcohol (100%) for 15 minutes and stained using the 0.1% crystal-violet for another 20 minutes. The numbers for the clones equal or more than 50 were counted using the inverted microscope to analyze cloning efficacy (CE). Based on multi-target/single-hit model (surviving fraction (SF)=1−(1−e−Dq/D0)N), the cell-survival curve was drawn using the Sigma-Plot 2001 Demo software. In the aforementioned formula, Dq was designed as quasi-threshold dose, D0 was designed as mean lethal dose. Meanwhile, the sensitization enhancement ratio (SER) was also calculated with the following formula: SER=D0 in control group/D0 in experimental group. The aforementioned experiments were conducted at least for 6 times.
FLOW CYTOMETRY ANALYSIS:
About 24 hours post treatment of irradiation and/or atorvastatin, PC-3, DU145, and Lncap cells were stained with the propidium iodide (PI) and fluorescein isothiocyanate (FITC)-conjugated Annexin V (Apoptosis Detection Kit, Beyotime Biotech. Shanghai, China), according to the manufacturer’s instruction. The apoptosis of PCa cells was captured and analyzed with FACScan flow cytometer (BD Biosciences, San Jose, CA, USA). The apoptotic rate in this study was represented as the late apoptotic rate plus the early apoptotic rate.
IMMUNO-FLUORESCENCE ASSAY:
PCa cells or the tumor tissues were fixed using 10% paraformaldehyde (Sangon Biotech.) and treated with 3% H2O2 (Beyotime Biotech.) to inactivate the endogenous peroxidase. Briefly, the PCa cells were incubated with the rabbit anti-human Bcl-2 polyclonal antibody (1: 1000, Cat. No. ab32124) and mouse anti-human MSH2 monoclonal antibody (Cat. No. ab52266; 1: 200) at 4°C overnight. Then, the PCa cells were incubated with Alexa Fluor FITC-conjugated goat anti-rabbit IgG (Cat. No. ab6718; 1: 500) to detect Bcl-2 signals or treated with Alexa Fluor 647-conjugated goat anti-mouse IgG (Cat. No. ab150115; 1: 500) to examine MSH2 signals. Finally, the merged co-immunofluorescence images were captured and combined using the laser scanning confocal microscopy (Mode: GCS-SP5, Leica Inc., Ltd., Germany), according to the instruction of the equipment.
ESTABLISHMENT OF XENOGRAFT TUMOR ANIMAL MODEL AND TUMOR VOLUME ANALYSIS:
BCLB/C nude mice (Chongqing Tengxin Biotech., Co., Ltd., Chongqing, China), male (6 weeks old), weighting about 20 g, were applied in this study to establish xenograft tumor animal model. In brief, PC-3, DU145 or Lncap cells were injected into mice flanks subcutaneously at 1st week and ending at the 4th week. Then, from the 1st week to 4th week post the PCa cells injection, the sizes of tumor (using the formula: (length×width2)/2) and body weight of mice in each group were measured and calculated, according to the previous study described [19]. Meanwhile, the tumor tissues were also extracted and employed to evaluate expressions of Bcl-2 using western blotting assay.
All procedures followed the Guidance of the National institute of Health Guide for Care and Use of Laboratory Animals. Also, the animal experiments were approved by Ethics Committee of Lanzhou University Second Hospital, Lanzhou, China.
STATISTICAL ANALYSIS:
Data in this study were represented as the mean±standard deviation (SD) and analyzed using professional SPSS software (version: 20.0, SPSS Inc., Chicago, lL, USA). The ANOVA validated by Tukey’s post-hoc test was used to compare differences among multiple groups. Student’s
Results
IRRADIATION ADMINISTRATION SUPPRESSED THE COLONY FORMATION OF PCA CELLS:
In this study, the PCa cells, including PC3, DU145, and Lncap, were treated with irradiation at dose of 0 Gy, 4 Gy, and 8 Gy, respectively (Figure 1A). The results indicated that irradiation administration (both for 4 Gy and 8 Gy IR) significantly decreased colony formation of PC-3/DU145/Lncap cells compared to that of tumor cells un-treated with irradiation (Figure 1B, P<0.05). Also, 4 Gy irradiation treatment significantly reduced the colony formation of PCa cells compared to that without irradiation treated cells (Figure 1B, P<0.05).
ATORVASTATIN ENHANCED INHIBITIVE EFFECTS OF IRRADIATION ON COLONY FORMATION OF PCA CELLS:
The results illustrated that the atorvastatin administration significantly reduced the percentages of colony formation in irradiation-treated PC3 cells (Figure 2A, P<0.05), DU145 cells (Figure 2B, P<0.05) and Lncap cells (Figure 2C, P<0.05). These findings suggest that atorvastatin strengthened the inhibitive effects of irradiation on colony formation in PC3 cells, DU145 cells, and Lncap cells.
Moreover, colony formation assay for the PCa cells was also conducted on the multi-target/single-hit model. Together with the administration of atorvastatin for 24 hours, PCa cells were irradiated using the distinguish doses of the irradiation (0 Gy, 4 Gy and 8 Gy). The results demonstrated that the SF value was significantly lower in the AS+IR group compared with that in the AS group at 4 Gy and 8 Gy in all PC3 (Figure 3A, P<0.05), DU145 (Figure 3B, P<0.05) and Lncap cells (Figure 3C, P<0.05). The SER values for IR+AS group in PC3 cells, DU145 cells and Lncap cells were 1.47, 1.28, and 1.19, respectively.
IRRADIATION ADMINISTRATION DEMONSTRATED NO EFFECTS ON APOPTOSIS:
Due to the reduction of colony formation caused by atorvastatin, we investigated the effects of irradiation administration on apoptosis of PCa cells. The results showed that there were no significant differences for apoptosis in all of PC3 cells (Figure 4A, P>0.05), DU145 cells (Figure 4B, P>0.05), and Lncap cells (Figure 4C, P>0.05), however, only with a slight inhibition of apoptosis in all PCa cells. These results suggest that atorvastatin caused reduced colony formation of PCa cells might be associated with the other cell death associated pathways, but not associated with apoptosis.
ATORVASTATIN ADMINISTRATION TRIGGERED BCL-2 EXPRESSION IN PCA CELLS:
Although no significant effects of atorvastatin on apoptosis were observed, we speculated that atorvastatin caused slight inhibition of apoptosis might be related to expression of Bcl-2, which is a biomarker for tumor cell proliferation [20]. Therefore, we evaluated Bcl-2 mRNA and protein expression in atorvastatin treated PCa cells using real-time polymerase chain reaction (RT-PCR) assay. The results indicated that irradiation treatment significantly increased Bcl-2 mRNA in PC3 (Figure 5A), DU145 (Figure 5B), and Lncap cells (Figure 5C) (P<0.05).
ATORVASTATIN ADMINISTRATION REDUCED MSH2 EXPRESSION IN PCA CELLS:
Due to the former description for association between Bcl-2 and DNA repair of tumor cells, the MSH2 as a DNA repair biomarker, was evaluated [21]. Our findings showed that irradiation administration significantly reduced MSH2 mRNA expression in PC-3 (Figure 6A), DU145 (Figure 6B), and Lncap cells (Figure 6C) compared to that in un-treated tumor cells (P<0.05). These results hint that atorvastatin modulate the tumor cell colony formation might through regulating MSH2 molecule expression.
ATORVASTATIN ADMINISTRATION REDUCED TUMOR SIZE OF IRRADIATION TREATED PCA CELLS DERIVED XENOGRAFT TUMOR MICE:
According to the findings for xenograft tumor model (Figure 7A), we found that irradiation treated PCa cells (including PC-3, DU145, and Lncap cells) transplantation remarkably decreased the tumor volume of xenograft mice at 2nd week and 4th week compared to that at 1st week (Figure 7B, P<0.05). Interestingly, atorvastatin administration significantly strengthened the reductive effects of irradiation treated PCa cells (including PC-3, DU145, and Lncap cells) transplantation on tumor volume (including PC-3, DU145, and Lncap cells) at 2nd week and 4th week compared to that at 1st week (Figure 7B, P<0.05). Moreover, there were no significant differences for the body weight among all 3 groups (Figure 7C, P>0.05).
BCL-2 INTERACTED WITH MSH2 IN ATORVASTATIN ADMINISTRATED XENOGRAFT TUMOR MODEL:
According to the changes of Bcl-2 and MSH2 levels in the PCa cells, we determined the interaction between Bcl-2 and MSH2. The immuno-fluorescence demonstrated that Bcl-2 could also directly interact with MSH2 in the tumor tissues of PC3-transplanted (Figure 8A), DU145-transplanted (Figure 8B), and Lncap-transplanted (Figure 8C) xenograft tumor mice.
ATORVASTATIN ADMINISTRATION INCREASED BCL-2 AND DECREASED MSH2 EXPRESSION IN L2 TUMOR TISSUES OF XENOGRAFT TUMOR MODEL:
The results showed that atorvastatin administration remarkably increased Bcl-2 expression and decreased MSH2 protein expression in PC3-transplanted (Figure 8A), DU145-transplanted (Figure 8B), and Lncap-transplanted (Figure 8C) and irradiation-administrated xenograft tumor mice compared to cells without atorvastatin treatment (P<0.05). These results also suggest that atorvastatin regulates tumor growth through modulating and Bcl-2 and MSH2 molecule expression.
Discussion
In the present study, we proved that atorvastatin administration could inhibit colony formation of irradiation-treated PCa cell lines and suppress tumor volume of the xenograft tumor mouse models. Our results suggest that atorvastatin could promote the radio-sensitivity of PCa cells and inhibit tumor growth of PCa mouse models through modulating Bcl-2 and MSH2 expressions.
Actually, the previous studies [13,22,23] have demonstrated the anti-tumor effects of atorvastatin on many cancers, however, our findings proved the effects of atorvastatin combining irradiation on tumor growth for the first time. In this study, we applied 4 Gy of irradiation and 6 μg/mL of atorvastatin in PCa cells, for both
In the former studies [26,27], the anti-tumor effects are usually played by inducing apoptosis of tumor cells, however, the atorvastatin triggered inhibition of apoptosis in this study. Moreover, atorvastatin administration also increased Bcl-2 expression in PCa cells, which seems to be controversial to the anti-tumor effects comparing with previous study [28]. Our results at least proved that atorvastatin induced colony formation information of PCa cells is not mediated by the apoptosis-associated signaling pathway. Therefore, there must be the other signaling pathways that mediated the anti-tumor effects of atorvastatin in proliferation of PCa cells and tumor growth. We speculated that the signaling pathways, such as cell death-associated autophagy [29] and cell necrosis [30], might be involved in the anti-tumor effects of atorvastatin.
The former study reported that inhibition of DNA repair processes in tumor cells usually causes the radio-resistance [31]. Actually, MSH2 molecule examined in this study is a kind of DNA repair-associated molecule [32]. In this study, we speculated that the downregulation of MSH2 in PCa cells might be correlated with the decreased tumor growth of xenograft tumor mice and the reduced colony formation of PCa cells. Therefore, the changes of MSH2 in our study are consistent with the previous study [33]. Moreover, we also speculated that the bioactivity of enhanced Bcl-2 expression might be blocked by MSH2 expression in PCa cells through triggering the intracellular interaction. This point needs to be proven in the future studies. Therefore, MSH2 might be a key biomarker for mediating atorvastatin triggered anti-tumor effects in PCa cells.
This study also illustrated a few limitations: Firstly, reasons of atorvastatin caused colony formation inhibition in PCa cells have not been clarified. In the following study, we would determine the autophagy in atorvastatin treated PCa cells for clarifying mechanism of atorvastatin-caused apoptosis. Secondarily, this study has not inhibited or silenced Bcl-2 and MSH2 gene expression to further confirm effects of atorvastatin on cell proliferation in PCa cells. In future, we would confirm the roles of Bcl-2 and MSH2 by silencing these 2 genes expression. Thirdly, a simple anti-neoplastic additive effect or a true radio-sensitization with super-additive effect deriving from combination of radiation and drug was not systematically clarified in this study. Fourthly, although the Bcl-2 and MSH2 expressions in tumor tissues were determined using immunofluorescence assay, however, the western blot assay is more appropriate to reflect the quantitative analysis of proteins. In future studies, we would conduct the western blot assay for determining Bcl-2 and MSH2 expression in tumor tissues.
The present research demonstrated that atorvastatin could enhance inhibitive effects of the irradiation on colony formation, apoptosis, Bcl-2/MSH2 expression and tumor growth, comparing with single-irradiation group. Therefore, treatment with the atorvastatin might demonstrate potential prospects for enhancing effects of radiotherapy on the PCa clinically. Our findings would also provide a potential strategy to resolve the problems of radio-resistance and would be benefit to the treatment of prostate cancer.
Conclusions
Atorvastatin administration could inhibit colony formation in PCa cells and promote effect of radiotherapy on tumor growth of xenograft tumor mice, which might be associated with interaction between Bcl-2 and MSH2 molecule.
Figures
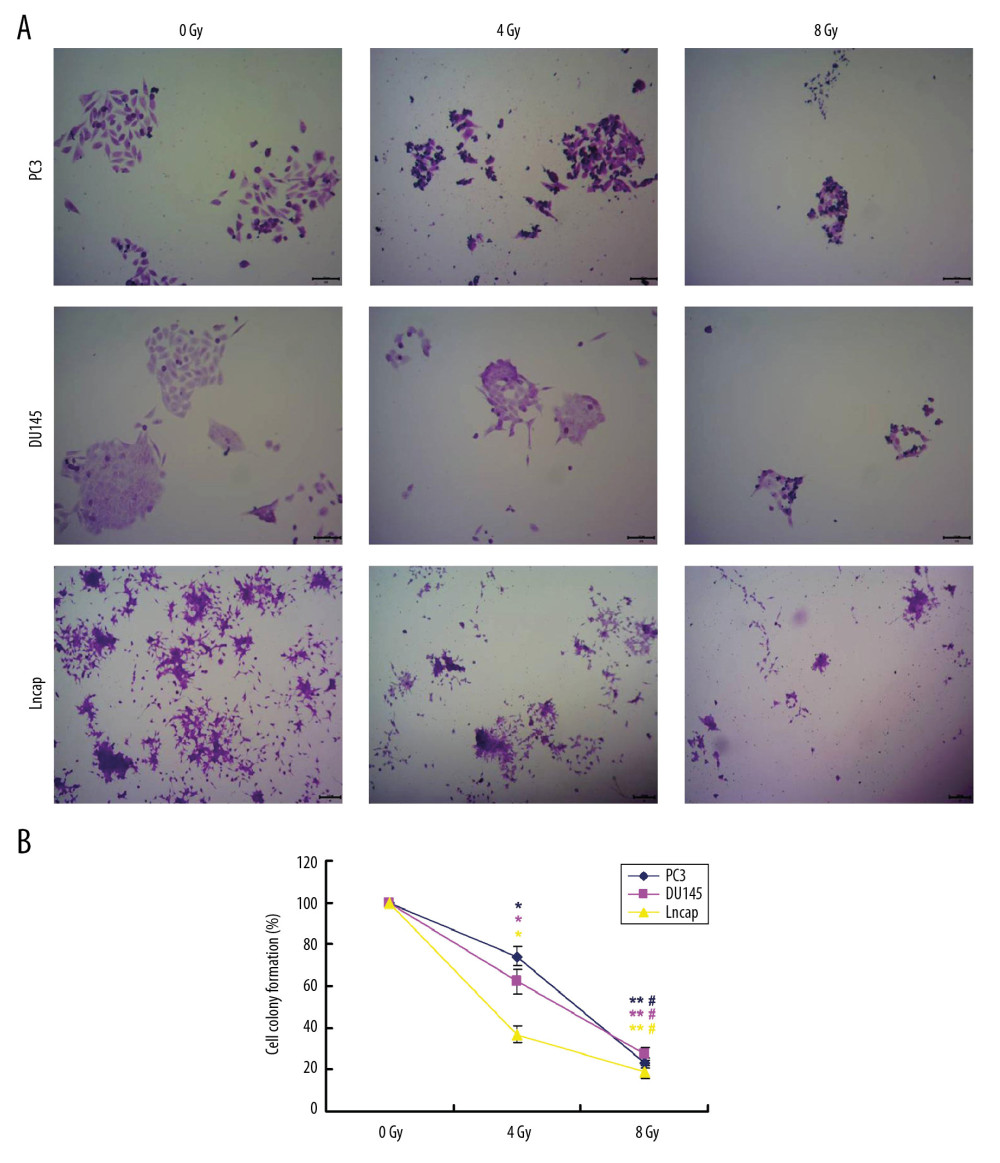 Figure 1. The irradiation administration significantly reduced colony formation of PC-3, DU145 and Lncap cells. (A) Colony formation images for PC-3, DU145, and Lncap cells. (B) Statistical analysis for colony formation of PC-3, DU145, and Lncap cells undergoing irradiation treatment. * P<0.05, ** P<0.01 versus 0 Gy group, # P<0.05 versus 4 Gy group.
Figure 1. The irradiation administration significantly reduced colony formation of PC-3, DU145 and Lncap cells. (A) Colony formation images for PC-3, DU145, and Lncap cells. (B) Statistical analysis for colony formation of PC-3, DU145, and Lncap cells undergoing irradiation treatment. * P<0.05, ** P<0.01 versus 0 Gy group, # P<0.05 versus 4 Gy group. 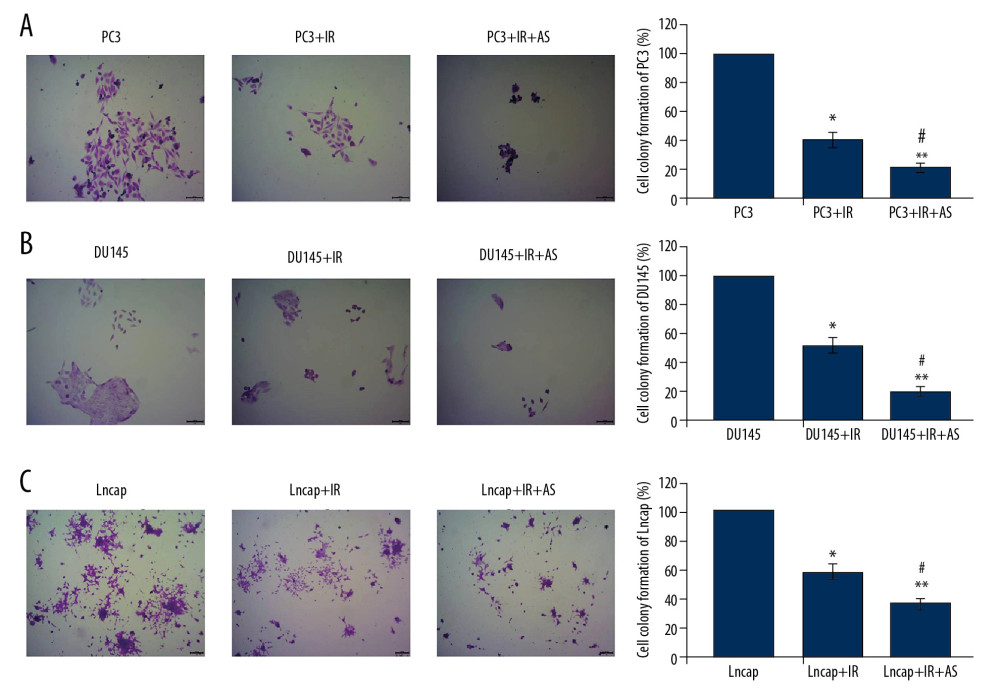 Figure 2. Assessment for colony formation of PC3, DU145, and Lncap cells undergoing irradiation (IR) and/or atorvastatin administration. (A) Colony formations of PC3 cells undergoing atorvastatin (AS) and/or IR administration. (B) Colony formations of DU145 cells undergoing AS and/or IR administration. (C) Colony formations of Lncap cells undergoing AS and/or IR administration. * P<0.05, ** P<0.01 versus PC3 or DU145 or Lncap group. # P<0.05 versus PC3+IR or DU145+IR or Lncap+IR group.
Figure 2. Assessment for colony formation of PC3, DU145, and Lncap cells undergoing irradiation (IR) and/or atorvastatin administration. (A) Colony formations of PC3 cells undergoing atorvastatin (AS) and/or IR administration. (B) Colony formations of DU145 cells undergoing AS and/or IR administration. (C) Colony formations of Lncap cells undergoing AS and/or IR administration. * P<0.05, ** P<0.01 versus PC3 or DU145 or Lncap group. # P<0.05 versus PC3+IR or DU145+IR or Lncap+IR group. 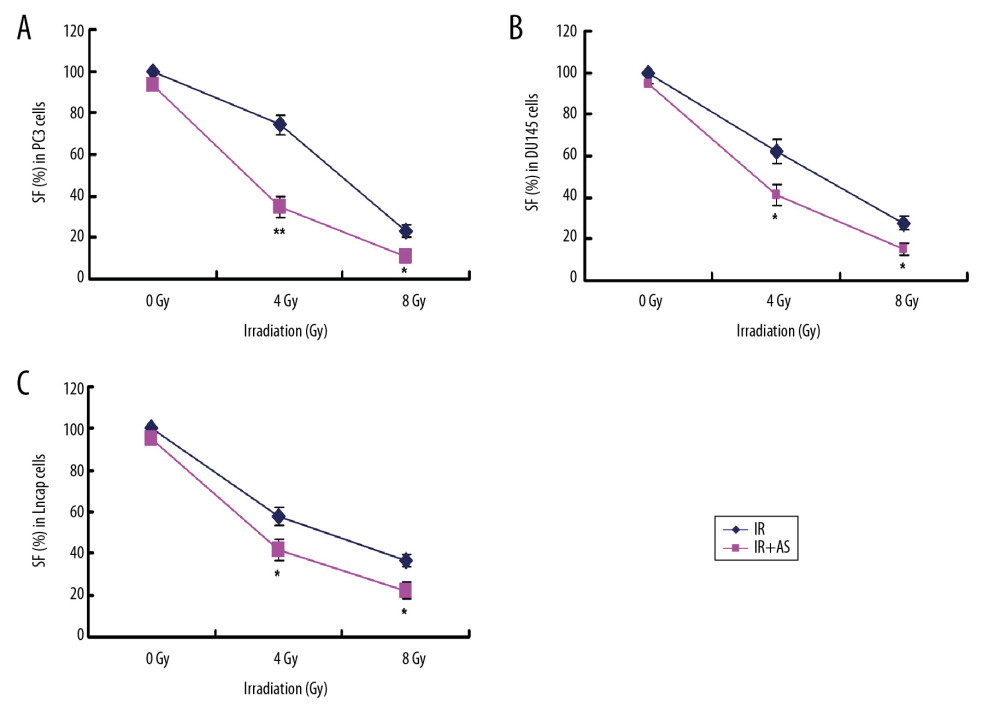 Figure 3. The cell survival curve dependent on clone formation on multi-target/single-hit model. (A) Clone formation for PC3 cells. (B) Clone formation for DU145 cells. (C) Clone formation for Lncap cells. * P<0.05, ** P<0.01 versus PCa cells in PC3 or DU145 or Lncap group without AS treatment.
Figure 3. The cell survival curve dependent on clone formation on multi-target/single-hit model. (A) Clone formation for PC3 cells. (B) Clone formation for DU145 cells. (C) Clone formation for Lncap cells. * P<0.05, ** P<0.01 versus PCa cells in PC3 or DU145 or Lncap group without AS treatment. 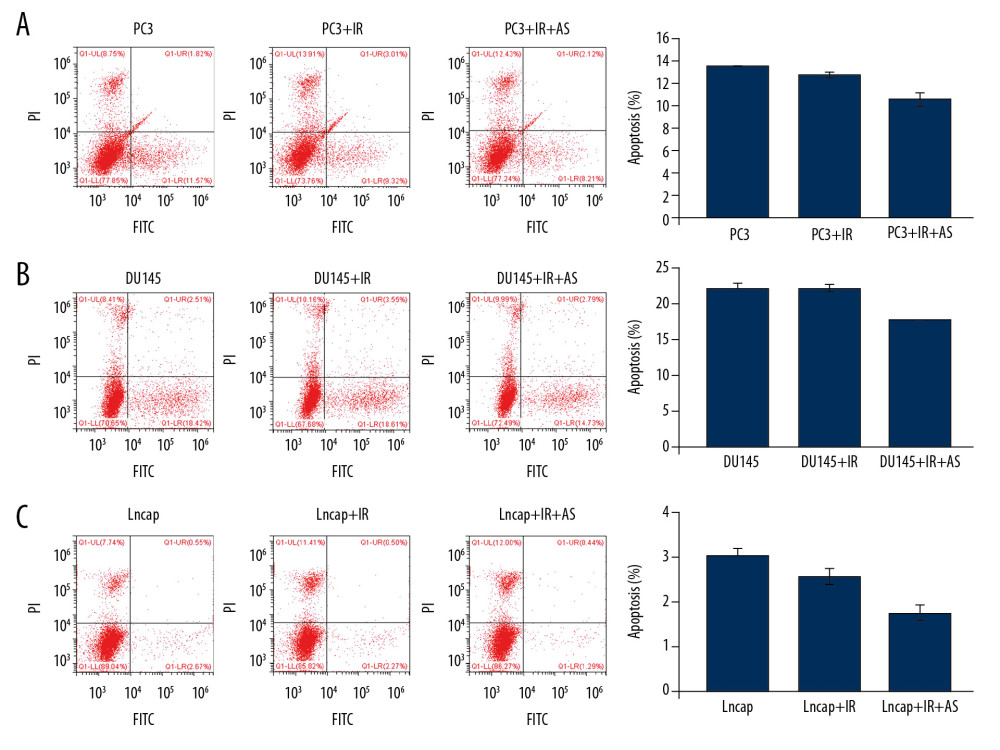 Figure 4. Evaluation for apoptosis of PC3, DU145 and Lncap cells undergoing IR and/or AS administration. (A) Flow cytometry images and statistical analysis for apoptosis of PC3 cells. (B) Flow cytometry images and statistical analysis for apoptosis of DU145 cells. (C) Flow cytometry images and statistical analysis for apoptosis of Lncap cells.
Figure 4. Evaluation for apoptosis of PC3, DU145 and Lncap cells undergoing IR and/or AS administration. (A) Flow cytometry images and statistical analysis for apoptosis of PC3 cells. (B) Flow cytometry images and statistical analysis for apoptosis of DU145 cells. (C) Flow cytometry images and statistical analysis for apoptosis of Lncap cells. 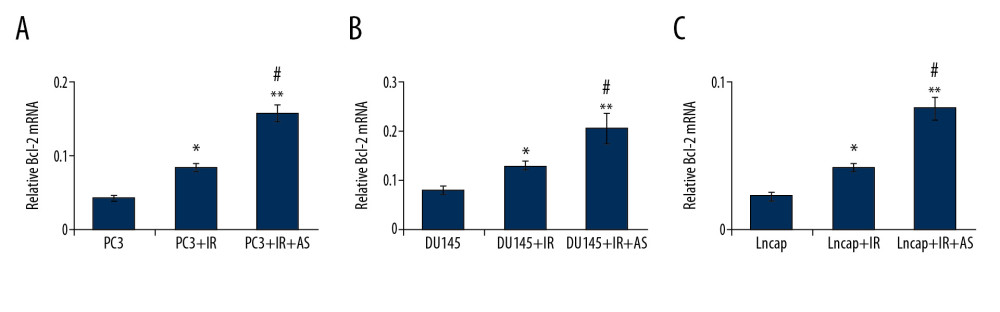 Figure 5. Effects of irradiation (IR) and/or atorvastatin (AS) administration on Bcl-2 mRNA expression in PCa cells. (A) Statistical analysis for Bcl-2 mRNA expression in PC3 cells. (B) Statistical analysis for Bcl-2 mRNA expression in DU145 cells. (C) Statistical analysis for Bcl-2 mRNA expression in Lncap cells. * P<0.05, ** P<0.01 versus PC3 or DU145 or Lncap group. # P<0.05 versus PC3+IR or DU145+IR or Lncap+IR group.
Figure 5. Effects of irradiation (IR) and/or atorvastatin (AS) administration on Bcl-2 mRNA expression in PCa cells. (A) Statistical analysis for Bcl-2 mRNA expression in PC3 cells. (B) Statistical analysis for Bcl-2 mRNA expression in DU145 cells. (C) Statistical analysis for Bcl-2 mRNA expression in Lncap cells. * P<0.05, ** P<0.01 versus PC3 or DU145 or Lncap group. # P<0.05 versus PC3+IR or DU145+IR or Lncap+IR group. 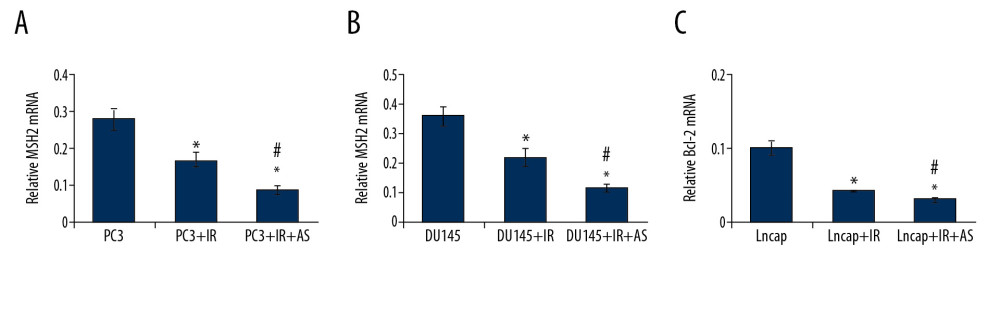 Figure 6. Determination for the effects of irradiation (IR) and/or atorvastatin (AS) administration on MSH2 mRNA expression in PCa cells. (A) Statistical analysis for MSH2 mRNA expression in PC3 cells. (B) Statistical analysis for MSH2 mRNA expression in DU145 cells. (C) Statistical analysis for MSH2 mRNA expression in Lncap cells. * P<0.05 versus PC3 or DU145 or Lncap group. #P < 0.05 versus PC3+IR or DU145+IR or Lncap+IR group.
Figure 6. Determination for the effects of irradiation (IR) and/or atorvastatin (AS) administration on MSH2 mRNA expression in PCa cells. (A) Statistical analysis for MSH2 mRNA expression in PC3 cells. (B) Statistical analysis for MSH2 mRNA expression in DU145 cells. (C) Statistical analysis for MSH2 mRNA expression in Lncap cells. * P<0.05 versus PC3 or DU145 or Lncap group. #P < 0.05 versus PC3+IR or DU145+IR or Lncap+IR group. 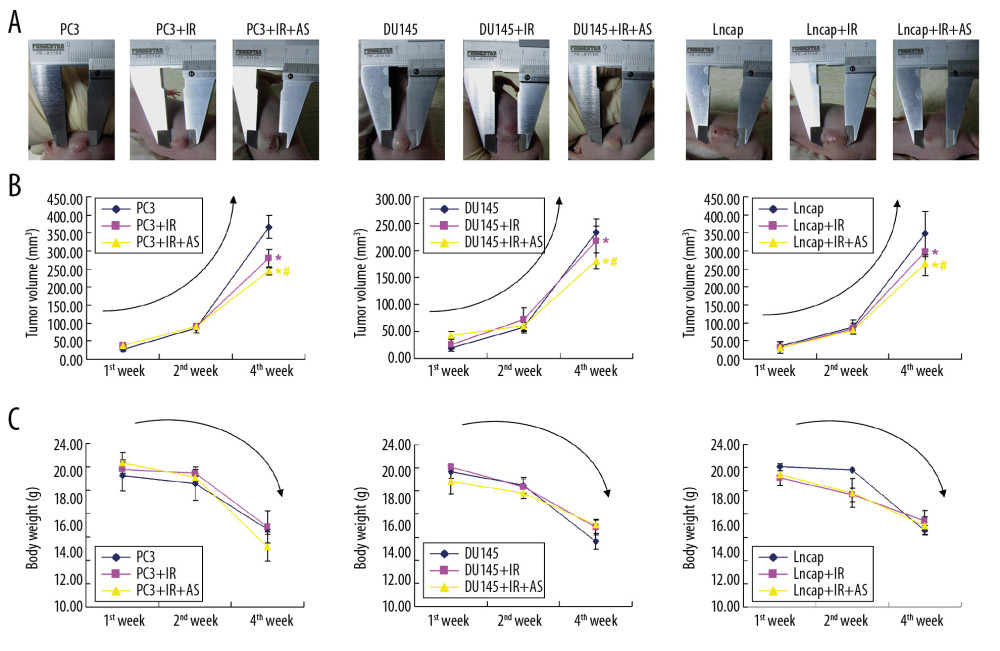 Figure 7. Effects of irradiation (IR) and/or atorvastatin (AS) administration on tumor size and tumor weight of xenograft tumor mice. (A) Representative images for the subcutaneous tumor xenografts. (B) Statistical analysis for the tumor size in PC-3, DU145 and Lncap cells transplanted xenograft tumor mice models. (C) Statistical analysis for tumor weight in PC-3, DU145, and Lncap cells transplanted xenograft tumor mice models. * P<0.05 versus PC3 or DU145 or Lncap group at the same time point. # P<0.05 versus PC3+IR or DU145+IR or Lncap+IR group at the same time point.
Figure 7. Effects of irradiation (IR) and/or atorvastatin (AS) administration on tumor size and tumor weight of xenograft tumor mice. (A) Representative images for the subcutaneous tumor xenografts. (B) Statistical analysis for the tumor size in PC-3, DU145 and Lncap cells transplanted xenograft tumor mice models. (C) Statistical analysis for tumor weight in PC-3, DU145, and Lncap cells transplanted xenograft tumor mice models. * P<0.05 versus PC3 or DU145 or Lncap group at the same time point. # P<0.05 versus PC3+IR or DU145+IR or Lncap+IR group at the same time point. 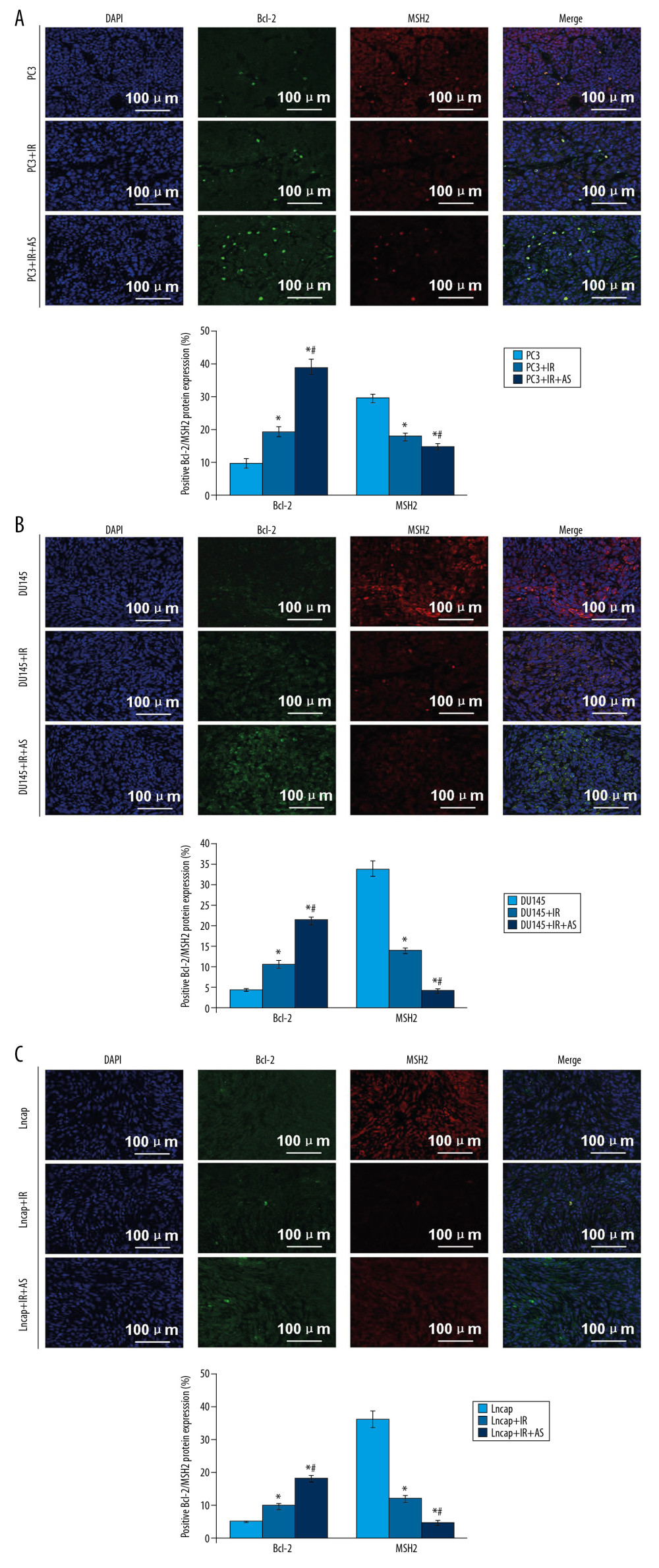 Figure 8. Observation for the interaction between Bcl-2 and MSH2 in tumor tissues of xenograft tumor mice using immuno-fluorescence assay. (A) Expression of Bcl-2 and MSH2 in tumor tissues of PC3-transplanted xenograft tumor mice. (B) Expression of Bcl-2 and MSH2 in tumor tissues of DU145-transplanted xenograft tumor mice. (C) Expression of Bcl-2 and MSH2 in tumor tissues of Lncap-transplanted xenograft tumor mice. * P<0.05 versus PC3 or DU145 or Lncap group. # P<0.05 versus PC3+IR or DU145+IR or Lncap+IR group. The scale bars were illustrated in images.
Figure 8. Observation for the interaction between Bcl-2 and MSH2 in tumor tissues of xenograft tumor mice using immuno-fluorescence assay. (A) Expression of Bcl-2 and MSH2 in tumor tissues of PC3-transplanted xenograft tumor mice. (B) Expression of Bcl-2 and MSH2 in tumor tissues of DU145-transplanted xenograft tumor mice. (C) Expression of Bcl-2 and MSH2 in tumor tissues of Lncap-transplanted xenograft tumor mice. * P<0.05 versus PC3 or DU145 or Lncap group. # P<0.05 versus PC3+IR or DU145+IR or Lncap+IR group. The scale bars were illustrated in images. References
1. Siegel RL, Miller KD, Jemal A, Cancer statistics, 2017: Cancer J Clin, 2017; 67; 7-30
2. Rebbeck TR, Prostate cancer genetics: Variation by race, ethnicity, and geography: Semin Radiat Oncol, 2017; 27; 3-10
3. Jemal A, Bray F, Center MM, Global cancer statistics: Cancer J Clin, 2011; 6; 69-90
4. Gheewala T, Skwor T, Munirathinam G, Photosensitizers in prostate cancer therapy: Oncotarget, 2017; 8; 30524-38
5. Adamska A, Elaskalani O, Emmanouilidi A, Molecular and cellular mechanisms of chemoresistance in pancreatic cancer: Adv Biol Regul, 2018; 68; 77-87
6. Shrivastava S, Mansure J, Almajed W, The role of HMGB1 in radioresistance of bladder cancer: Mol Cancer Ther, 2016; 15; 471-79
7. Yuan M, Gao Y, Li L, Phospholipase C (PLC) promotes androgen receptor antagonist resistance via the bone morphogenetic protein (BMP)-6/SMAD axis in a castration-resistant prostate cancer cell line: Med Sci Monit, 2019; 25; 4438-49
8. Shaikh T, Handorf EA, Meyer JE, Mismatch repair deficiency testing in patients with colorectal cancer and nonadherence to testing guidance in young adults: JAMA Oncol, 2018; 8; e173580
9. Yu S, Wang M, Ding X, Testicular orphan nuclear receptor 4 is associated with the radio-sensitivity of prostate cancer: Prostate, 2015; 75; 1632-42
10. Pajic M, Froio D, Daly S, miR-139-5p modulates radiotherapy resistance in breast cancer by repressing multiple gene networks of DNA repair and ROS defense: Cancer Res, 2018; 78; 501-15
11. Batar B, Mutlu T, Bostanci M, DNA repair and apoptosis: roles in radiotherapy related acute reactions in breast cancer patients: Cell Mol Biol (Noisy-le-grand), 2018; 64; 64-70
12. Jeong JH, Yum KS, Chang JY, Dose-specific effects of simvastatin on hypoxia-induced HIF-alpha BACE expression Alzheimer’s disease cybrid cells: BMC Neurol, 2015; 15; 127
13. Chen B, Zhang M, Xing D, Atorvastatin enhances radiosensitivity in hypoxia-induced prostate cancer cells related with HIF-1 alpha inhibition: Biosci Rep, 2017; 37 BSR20170340
14. He Y, Huang H, Farischon C, Combined effects of atorvastatin and aspirin on growth and apoptosis in human prostate cancer cells: Oncol Rep, 2017; 37; 953-60
15. Li X, Dardik A, Guo R, Atorvastatin regulates pericardial patch healing via the microRNA140-ADAM10-ephrinB2: Am J Transl Res, 2018; 10; 4054-64
16. He Q, Li J, Dong F, LKB1 promotes radioresistance in esophageal cancer cells exposed to radiation by suppression of apoptosis and activation of autophagy via the AMPK pathway: Mol Med Rep, 2017; 16; 2205-10
17. Peng WX, Wan YY, Gong AH, Egr-1 regulates irradiation-induced autophagy through Atg4B radioresistance in hepatocellular carcinoma cells: Oncogenesis, 2017; 6; e292
18. Zhao Y, Chen R, Wang Y, Alpha-pinene inhibits human prostate cancer growth in a mouse Xenograft model: Chemotherapy, 2018; 63; 1-7
19. Wang Y, Yao B, Li H, Assessment of tumor stiffness with shear ware elastography in h human prostate cancer Xenograft implication model: J Ultrasound Med, 2017; 36; 955-63
20. Chen X, Liu Y, Wu J, Mechanistic study of inhibitory effects of atorvastatin and docetaxel in combination on prostate cancer: Cancer Genomics Proteomics, 2016; 13; 151-60
21. Giarnieri E, Mancini R, Pisani T: Clin Cancer Res, 2000; 6; 3600-6
22. Hannan R, Tumati V, Xie XJ, Stereotactic body radiation therapy for low and intermediate risk prostate cancer-results from a multi-institutional clinical trial: Eur J Cancer, 2016; 59; 142-51
23. Gobel A, Thiele S, Browne AJ, Combined inhibition of the mevalonate pathway with statins and zoledronic acid potentiates their anti-tumor effects in human breast cancer cells: Cancer Lett, 2006; 375; 162-71
24. Wang ZS, Huang HR, Zhang LY: Biol Pharm Bull, 2017; 40; 1247-54
25. He Z, Yuan J, Qi P: Mol Med Rep, 2015; 11; 4403-8
26. Xiang M, Su H, Shu G, Amplexicaule A exerts anti-tumor effects by inducing apoptosis in human breast cancer: Oncotarget, 2016; 7; 18521-30
27. Xu J, Wang Y, Zhang Y, Astemizole promotes the anti-tumor effect of vitamin D through inhibiting miR-125a-5p-mediated regulation of VDR in HCC: Biomed Pharmacother, 2018; 107; 1682-91
28. Huang Y, Cai Y, Huang R, Rosmarinic acid combined with adriamycin induces apoptosis by triggering mitochondria-mediated signaling pathway in HepG2 and Bel-7402 cells: Med Sci Monit, 2018; 24; 7898-908
29. Dong Y, Wu Y, Zhao GL, Inhibition of autophagy by 3-MA promotes hypoxia-induced apoptosis in human colorectal cancer cells: Eur Rev Med Pharmacol Sci, 2019; 23; 1047-54
30. Vanden Berghe T, Linkermann A, Jouan-Lanhouet S, Regulated necrosis: The expanding network of non-apoptotic cell death pathways: Nat Rev Mol Cell Biol, 2014; 15; 135-47
31. Schulz A, Meyer F, Dubrovska A, Cancer stem cells and radioresistance: DNA repair and beyond: Cancers (Basel), 2019; 11; E862
32. Guedes LB, Antonarakis ES, Schweizer MT, MSH2 loss in primary prostate cancer: Clin Cancer Res, 2017; 23; 6863-74
33. Srivastava T, Chattopadhyay P, Mahapatra AK, Increased hMSH2 protein expression in glioblastoma multiforme: J Neurooncol, 2004; 66; 51-57
Figures
 Figure 1. The irradiation administration significantly reduced colony formation of PC-3, DU145 and Lncap cells. (A) Colony formation images for PC-3, DU145, and Lncap cells. (B) Statistical analysis for colony formation of PC-3, DU145, and Lncap cells undergoing irradiation treatment. * P<0.05, ** P<0.01 versus 0 Gy group, # P<0.05 versus 4 Gy group.
Figure 1. The irradiation administration significantly reduced colony formation of PC-3, DU145 and Lncap cells. (A) Colony formation images for PC-3, DU145, and Lncap cells. (B) Statistical analysis for colony formation of PC-3, DU145, and Lncap cells undergoing irradiation treatment. * P<0.05, ** P<0.01 versus 0 Gy group, # P<0.05 versus 4 Gy group. Figure 2. Assessment for colony formation of PC3, DU145, and Lncap cells undergoing irradiation (IR) and/or atorvastatin administration. (A) Colony formations of PC3 cells undergoing atorvastatin (AS) and/or IR administration. (B) Colony formations of DU145 cells undergoing AS and/or IR administration. (C) Colony formations of Lncap cells undergoing AS and/or IR administration. * P<0.05, ** P<0.01 versus PC3 or DU145 or Lncap group. # P<0.05 versus PC3+IR or DU145+IR or Lncap+IR group.
Figure 2. Assessment for colony formation of PC3, DU145, and Lncap cells undergoing irradiation (IR) and/or atorvastatin administration. (A) Colony formations of PC3 cells undergoing atorvastatin (AS) and/or IR administration. (B) Colony formations of DU145 cells undergoing AS and/or IR administration. (C) Colony formations of Lncap cells undergoing AS and/or IR administration. * P<0.05, ** P<0.01 versus PC3 or DU145 or Lncap group. # P<0.05 versus PC3+IR or DU145+IR or Lncap+IR group. Figure 3. The cell survival curve dependent on clone formation on multi-target/single-hit model. (A) Clone formation for PC3 cells. (B) Clone formation for DU145 cells. (C) Clone formation for Lncap cells. * P<0.05, ** P<0.01 versus PCa cells in PC3 or DU145 or Lncap group without AS treatment.
Figure 3. The cell survival curve dependent on clone formation on multi-target/single-hit model. (A) Clone formation for PC3 cells. (B) Clone formation for DU145 cells. (C) Clone formation for Lncap cells. * P<0.05, ** P<0.01 versus PCa cells in PC3 or DU145 or Lncap group without AS treatment. Figure 4. Evaluation for apoptosis of PC3, DU145 and Lncap cells undergoing IR and/or AS administration. (A) Flow cytometry images and statistical analysis for apoptosis of PC3 cells. (B) Flow cytometry images and statistical analysis for apoptosis of DU145 cells. (C) Flow cytometry images and statistical analysis for apoptosis of Lncap cells.
Figure 4. Evaluation for apoptosis of PC3, DU145 and Lncap cells undergoing IR and/or AS administration. (A) Flow cytometry images and statistical analysis for apoptosis of PC3 cells. (B) Flow cytometry images and statistical analysis for apoptosis of DU145 cells. (C) Flow cytometry images and statistical analysis for apoptosis of Lncap cells. Figure 5. Effects of irradiation (IR) and/or atorvastatin (AS) administration on Bcl-2 mRNA expression in PCa cells. (A) Statistical analysis for Bcl-2 mRNA expression in PC3 cells. (B) Statistical analysis for Bcl-2 mRNA expression in DU145 cells. (C) Statistical analysis for Bcl-2 mRNA expression in Lncap cells. * P<0.05, ** P<0.01 versus PC3 or DU145 or Lncap group. # P<0.05 versus PC3+IR or DU145+IR or Lncap+IR group.
Figure 5. Effects of irradiation (IR) and/or atorvastatin (AS) administration on Bcl-2 mRNA expression in PCa cells. (A) Statistical analysis for Bcl-2 mRNA expression in PC3 cells. (B) Statistical analysis for Bcl-2 mRNA expression in DU145 cells. (C) Statistical analysis for Bcl-2 mRNA expression in Lncap cells. * P<0.05, ** P<0.01 versus PC3 or DU145 or Lncap group. # P<0.05 versus PC3+IR or DU145+IR or Lncap+IR group. Figure 6. Determination for the effects of irradiation (IR) and/or atorvastatin (AS) administration on MSH2 mRNA expression in PCa cells. (A) Statistical analysis for MSH2 mRNA expression in PC3 cells. (B) Statistical analysis for MSH2 mRNA expression in DU145 cells. (C) Statistical analysis for MSH2 mRNA expression in Lncap cells. * P<0.05 versus PC3 or DU145 or Lncap group. #P < 0.05 versus PC3+IR or DU145+IR or Lncap+IR group.
Figure 6. Determination for the effects of irradiation (IR) and/or atorvastatin (AS) administration on MSH2 mRNA expression in PCa cells. (A) Statistical analysis for MSH2 mRNA expression in PC3 cells. (B) Statistical analysis for MSH2 mRNA expression in DU145 cells. (C) Statistical analysis for MSH2 mRNA expression in Lncap cells. * P<0.05 versus PC3 or DU145 or Lncap group. #P < 0.05 versus PC3+IR or DU145+IR or Lncap+IR group. Figure 7. Effects of irradiation (IR) and/or atorvastatin (AS) administration on tumor size and tumor weight of xenograft tumor mice. (A) Representative images for the subcutaneous tumor xenografts. (B) Statistical analysis for the tumor size in PC-3, DU145 and Lncap cells transplanted xenograft tumor mice models. (C) Statistical analysis for tumor weight in PC-3, DU145, and Lncap cells transplanted xenograft tumor mice models. * P<0.05 versus PC3 or DU145 or Lncap group at the same time point. # P<0.05 versus PC3+IR or DU145+IR or Lncap+IR group at the same time point.
Figure 7. Effects of irradiation (IR) and/or atorvastatin (AS) administration on tumor size and tumor weight of xenograft tumor mice. (A) Representative images for the subcutaneous tumor xenografts. (B) Statistical analysis for the tumor size in PC-3, DU145 and Lncap cells transplanted xenograft tumor mice models. (C) Statistical analysis for tumor weight in PC-3, DU145, and Lncap cells transplanted xenograft tumor mice models. * P<0.05 versus PC3 or DU145 or Lncap group at the same time point. # P<0.05 versus PC3+IR or DU145+IR or Lncap+IR group at the same time point. Figure 8. Observation for the interaction between Bcl-2 and MSH2 in tumor tissues of xenograft tumor mice using immuno-fluorescence assay. (A) Expression of Bcl-2 and MSH2 in tumor tissues of PC3-transplanted xenograft tumor mice. (B) Expression of Bcl-2 and MSH2 in tumor tissues of DU145-transplanted xenograft tumor mice. (C) Expression of Bcl-2 and MSH2 in tumor tissues of Lncap-transplanted xenograft tumor mice. * P<0.05 versus PC3 or DU145 or Lncap group. # P<0.05 versus PC3+IR or DU145+IR or Lncap+IR group. The scale bars were illustrated in images.
Figure 8. Observation for the interaction between Bcl-2 and MSH2 in tumor tissues of xenograft tumor mice using immuno-fluorescence assay. (A) Expression of Bcl-2 and MSH2 in tumor tissues of PC3-transplanted xenograft tumor mice. (B) Expression of Bcl-2 and MSH2 in tumor tissues of DU145-transplanted xenograft tumor mice. (C) Expression of Bcl-2 and MSH2 in tumor tissues of Lncap-transplanted xenograft tumor mice. * P<0.05 versus PC3 or DU145 or Lncap group. # P<0.05 versus PC3+IR or DU145+IR or Lncap+IR group. The scale bars were illustrated in images. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387