11 August 2020: Review Articles
Current Status of Research on the Role of Circular RNAs in Hepatocellular Carcinoma and Clinical Implications
Ren-Chao Zou1E, Ling-Lin Li23CDE, Hong-Ling Yuan2A*DOI: 10.12659/MSM.923832
Med Sci Monit 2020; 26:e923832
Abstract
ABSTRACT: The latest statistics show that rates of morbidity and mortality for hepatocellular carcinoma are gradually increasing over time. Accumulating evidence indicates that circular RNAs (circRNAs) participate in the regulation of gene transcription and translation and exert a crucial role in endogenous RNA network. circRNAs are implicated in the pathogenesis of numerous tumors including hepatocellular carcinoma (HCC), gastric carcinoma and bladder cancer. Of note, the effect of circRNAs in HCC has drawn increasing public attention. Previous studies revealed that the function of circRNAs mainly consists of sponges of miRNA and RNA-binding proteins, alternative splicing of pre-mRNAs, transcriptional and translational regulators, and potential to encode proteins. In addition, recent research data indicate that the expression level of circRNAs is closely correlated with metastasis, invasion, and occurrence of HCC in patients. These findings imply that circRNAs may be useful as biomarkers for diagnosis and prediction of prognosis of HCC. In this review, we have systemically summarized current viewpoints regarding the role of circRNAs expression in HCC to provide an important reference illustrating the underlying mechanism of HCC.
Keywords: Biological Markers, Carcinoma, Hepatocellular, Biomarkers, Tumor, Liver Neoplasms, RNA, Circular, RNA-Binding Proteins
Background
Hepatocellular carcinoma (HCC) is a common disease that seriously threatens human health worldwide. Prevalence of the disease and deaths due to it are a continuously increasing with development of the economy and the environment [1–3]. The prognosis for HCC patients remains extremely poor although significant progress has been achieved. Strategies for early diagnosis are urgently needed because the majority of patients with HCC are diagnosed in very late stages. However, the molecular mechanism of HCC has not been clearly defined. Circular RNAs (circRNAs) are a new class of RNA molecules that have functions as regulators of parental gene transcription, in alternative splicing, and as miRNA sponges [4]. Through use of RNA deep sequencing gtechnology, numerous circRNAs have been identified as the predominant regulatory elements in diseases [5]. Moreover, accumulating evidence shows that circRNAs play pivotal roles in many diseases. In particular, abnormally expressed circRNAs are involved in tumor progression including cell proliferation, migration, and invasion [6–8]. In addition, some research indicates that circRNAs level are closely correlated wit specific phenotypes and tumorigenesis in HCC [9–11]. Nevertheless, the research concerning circRNAs is frankly in its infancy, which greatly hinders the application of circRNAs as biomarkers for diagnosis of HCC in clinics.
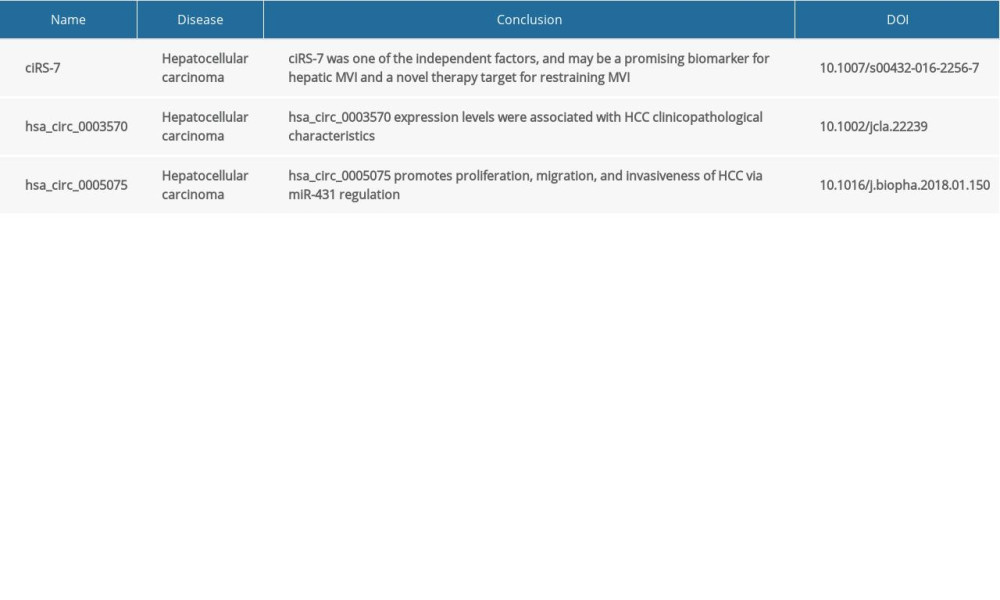
Related research shows that circRNAs possess great potential to be used for diagnosis of HCC [12,13]. Recent studies have found that hsa_circ_0067934 plays oncogenic roles by accelerating cell proliferation and metastasis in glioblastoma (GBM) [14,15]. circ-SMARCA5 was significantly elevated, and thereby suppressed cell apoptosis and arrested cell cycle in prostate cancer [16]. In addition, previous studies have shown that downregulation of hsa_circ_0005986 facilitated cell proliferation by promoting the G0/G1 to S phase transition in HCC [17]. Similarly, alteration in expression of circRNAs correlated with development and metastasis of malignant tumors [18]. These data suggest that circRNAs may be of greater benefit in clinical diagnosis of HCC. However, reliable circRNA biomarkers for HCC are still lacking. Therefore, this review synthetically integrates available data on the role of circRNA in HCC progression, and attempts to provide crucial clues for investigating the molecular mechanism regarding HCC.
Overview of circRNA
CircRNAs are a category of single-stranded, closed-circle molecules, which take part in multifaceted biological regulation [19]. Recently, research has verified that the majority of circRNAs are synthesized by back-spliced exons, and that others are formed from intron, intergenic, and untranslated regions (UTR). Therefore, biogenesis of circRNAs can be divided into EIciRNAs (exon-intron circRNAs), ecircRNAs (circular exonic RNAs), and ciRNAs (circular intronic RNAs) [20]. Meanwhile, over 20,000 circRNAs have been identified [21], and this type of transcript has been considered a new form of gene expression [22]. Generally, the structure of the transcription is inverted, and the order of genomic exons is altered and these exons are spliced. Over time, the biological functions of circRNAs gradually have been recognized, including roles in embryonic development, maintainenance of homeostasis, and promotion of tumor progression (Figure 1) [23].
Properties of circRNAs
circRNAs recently have attracted great attention related to their pathological role in disease development. Compared with linear RNAs, circRNAs have special properties, including biological roles and clinical use. circRNAs are mainly enriched in certain body fluids comprising blood, saliva, and urine [24,25]. They are covalently closed loop structures [26]. Degradation of most RNA is highly dependent on RNA exonuclease or RNase. Hence, circRNAs remain highly stable based on their high resistance to enzyme degradation [27]. Moreover, studies have shown that expression of circRNAs is tissue-specific and correlated with different phases of development [28,29], and they exhibit different expression patterns at different developmental stages [21,30].
Roles of circRNAs
Accumulating evidence shows that circRNAs play a crucial role in the pathogenesis of diseases as a result of their complex biological functions. Generally, the molecular functions of circRNAs mainly include being sponges of miRNA, acting as RNA-binding proteins, performing alternative splicing of pre-mRNAs, regulating transcription and translation, and potentially encoding proteins. These properties are described in detail below.
Sponges of miRNA
The different types of circRNAs have different miRNA binding sites. Some circRNAs negatively regulate miRNAs by absorbing and specifically binding to miRNAs, then decreasing miRNA activity and elevating expression of miRNA-related target genes [31,32]. Researchers have shown that ciRS-7 inhibits miR-7 function, and positively mediates miR-7 target genes, acting as a molecular sponge [33]. In addition, functional analyses have indicated that circRNAs constitute an entire molecular regulatory network which specifically regulates degradation of miRNAs as miRNA sponges [34].
CircRNAs-binding proteins
RNA binding proteins (RBPs) are a broad class of proteins involved in gene transcription, translation, and interaction. Studies suggest that distribution of RBPs is widespread, in many tissue types. Furthermore, RBPs participate in development of disorders by regulating post-transcriptional regulation of RNAs. RBPs assemble ribonucleoprotein complexes to bind RNA sequences, thereby affecting the function of the target RNAs [35]. Previous research has shown that circRNAs serve as protein decoys to harbor binding sites of specific proteins and block protein activity. Circ-Foxo3 induces cell cycle arrest, resulting in defective CDK2 gene function by binding to p21 and CDK2 [36]. Moreover, circRNA cia-cGAS binds to cGAS protein and suppresses enzymatic activity of cGAS, thereby preventing cGAS from recognizing self-DNA [37].
circRNAs Regulate Alternative Splicing, Transcription and Translation
Cellular localization of most circRNAs is cytoplasmic, which is the basis for the biological function of miRNA and protein decoys. Several studies have suggested that circRNAs participate in RNA splicing, assembly, and biosynthesis. Recently, research has shown that circRNAs may play pivotal roles in regulating alternative splicing, transcription, and translation. In addition, the exon of the splicing factor may form a circRNA by affecting formation of linear RNA. EIciRNAs interact with the U1 small nuclear ribonucleoprotein(snRNP), thereby regulating parental gene transcription by binding to RNA polymerase II [38].
Interestingly, translation of circRNAs is mediated by IRES and N6-methyladenosine (m6A), and translation efficiency of circRNA is regulated by the level of m6A modification [39]. Moreover, circFBXW7 effectively inhibits glioma proliferation and cell cycle progression by antagonizing USP28-induced c-Myc stabilization [40].
Potential to Encode Proteins
circRNAs are implicated in numerous physiological processes and pathogenesis of diseases. Strong evidence indicates that circRNAs can encode proteins by mimicking DNA rolling circle amplification [41]. Related studies indicate that circRNA circPPP1R12A plays a key molecular role by encoding a functional protein (circPPP1R12A-73aa), which promotes proliferation, migration, and invasion of colon cancer [42]. circANRIL interacts with pescadillo zebrafish homolog 1 (PES1) to mediate ribosome biogenesis and pre-rRNA processing in vascular macrophages and smooth muscle cells [43]. These studies have significantly increased the knowledge base about the biological functions of circRNAs.
circRNAs in Diseases
circRNAs are involved in processes that lead to development of various disorders, such as neuronal and cardiovascular diseases, and cancers. circRNAs participate in regulating gene transcription and protein expression, and are indirectly and directly associated with time- and region-specific variations [30,44]. As mentioned previously, abnormal expression of circRNAs is implicated in neurological disorders [28,45,46]. Atherosclerosis and ribosomal RNA maturation reportedly are regulated by circANRIL [43]. Simultaneously, some studies have suggested that circRNAs upregulation significantly affects sprouting and proliferation of vascular endothelial cells, and elicits vascular dysfunction [47,48]. Recently, several experiments have implicated circRNAs in pathogenesis of cancer via activation of a series of cascade reactions. However, the underlying mechanism for the effect of circRNAs in initiation and progression of tumors has not been fully clarified [49]. To date, related studies have revealed that certain circRNAs are highly expressed in tumor tissues, and overexpression of circRNAs promotes tumor proliferation and deterioration. An investigation revealed that Hsa_circ_002059 was downregulated in gastric cancer [14], while hsa_circ_0004018 was upregulated in HCC [50]. Meanwhile, tumor-specific circRNAs candidates were screened in lung adenocarcinoma tissue by microarrays and 59 circRNAs were identified (20 down-regulated and 39 up-regulated) [18]. Of the 59 circRNAs, hsa_circ_0013958 clearly was positive correlated with lymph node metastasis and TNM stage. These findings indicate that circRNAs have important roles in tumor progression, and may have potential for broad applicatoins in medicine science.
Overview of HCC
HCC is one of the most prevalent tumors worldwide, with 21 000 diagnoses and approximately 700 000 deaths annually [51,52]. Epidemiological survey data indicate that morbidity and mortality from HCC are gradually increasing [51]. Risk factors for HCC include diabetes mellitus, obesity, smoking, alcohol consumption, older age, male sex, chronic HBV, liver cirrhosis, and chronic hepatitis C virus (HCV). The primary risk factors include liver cirrhosis, viral hepatitis, alcohol intake, and obesity [53]. Worldwide, approximately 50% HCC patients are infected with hepatitis B virus (HBV) [54,55] or HCV [56,57]. In addition, alcohol abuse is a crucial factor for onset of HCC [58–60]. Obesity, hypertension, and diabetes are closely linked with development of HCC, but specific correlations remain unknown [61]. Moreover, regular screening has been widely applied for early detection and to ensure effective treatment of HCC. Most commonly, good results have been achieved with regular screening with ultrasonography, blood alpha-fetoprotein content testing, MRI, and CT [62,63].
Generally, surgical resection and chemotherapy are mainstays of therapy in patients with HCC. Yet, some tumors cannot be fully removed, which results in tumor growth, invasion, and metastasis. Local and systemic metastases are the main reasons for the unsatisfactory prognosis in patients with HCC. Therefore, more effective therapeutic approaches need to be developed.
Roles of circRNAs in HCC
Numerous studies have documented the important role that circRNAs play in tumorigenesis, metastasis, and invasion. Research has shown that circRNAs are localized in the nucleus and interfere with transcription and promote alternative splicing. circPVT1 is overexpressed in gastric cancer tissues compared with non-tumor tissues, and circPVT1 acts as an oncogene to mediate expression of miR-497-5p [64]. However, studies concerning the role of circRNAs in development and progression of HCC remain in their infancy.
Tumor Inhibition
Currently, circRNAs are considered promising diagnostic biomarkers and ideal therapeutic targets for HCC [65,66]. Studies have revealed that circ-ITCH inhibits tumor proliferation by suppressing the Wnt/beta-Catenin pathway. Expression of circ-ITCH has been positively correlated with good survival outcome in patients with HCC [67]. Analysis of the circRNAs expression profile in human HCC tissues showed that circMTO1 was markedly decreased in HCC tissues and that expression of circMTO1 was positively correlated with survival rate. circMTO1 reportedly inhibits HCC progress by sponging miR-9 and thereby increasing p21 expression [68]. Meanwhile, overexpression of hsa_circ_0001649 negatively affects invasion and proliferation and promotes apoptosis of HCC cells [69]. Downregulation of ZKSCAN1 and circZKSCAN1 enhances cell proliferation and promotes progression of HCC [70].
Tumor Promotion
In patients with HCC, Cdr1was more abundant in tumor specimens than in adjacent normal tissues. Cdr1as effectively suppresses the invasion and proliferation of HCC cells by targeting miR-7 [71,72]. Some reports have shown that hsa_circ_0000673 is significantly upregulated in HCC tissues and hsa_circ_0000673 downregulation markedly inhibits proliferation and invasion of HCC cells
Biomarker
Previous studies have shown that circRNAs are closely related to development of tumors. Clinicopathological features in patients with HCC are correlated to with levels of expression of ciRS-7 and its targeted mRNAs [71]. Global circRNA expression profile analysis showed that hsa_circ_0005075 exhibited significant differences in tumor tissue versus adjacent tissues in patients with HCC. Expression of hsa_circ_0005075 also was related to tumor proliferation and metastasis [76]. Therefore, an increasing number of circRNAs have been identified as diagnostic markers, as summarized in Table 1 [77].
Conclusions
Given the high incidence and mortality fo HCC worldwide, it is one of the most serious diseases threatening human health. Increasing attention is being paid due to this serious situation [51]. Evidence is increasing to support the close association between circRNAs progression of HCC. circRNAs may play an important role in the occurrence and development of tumors. However, the molecular mechanism underlying the relationship between circRNAs and HCC has not been fully elucidated. Therefore, in-depth research is needed on the potential regulatory relationships, and to uncover regulatory patterns between circRNAs and HCC so that new diagnostic markers for HCC can be developed.
Figures
![Biogenesis of circular RNAs [23]. (A) Lariat-driven circularization. The 3′ hydroxyl of the upstream exon reacts with the 5′ phosphate of the downstream exon to form a covalent linkage, then producing a lariat including exons and introns. The 2′ hydroxyl of the 5′ intron interacts with the 5′ phosphate of the 3′-intron to form an ecircRNA following an interaction between the 3′ hydroxyl of the 3′ exon and the 5′ phosphate of the 5′ exon. (B) RNA-binding protein (RBP)-driven circularization. RBPs accelerate interaction of the downstream intron and upstream intron, thereby promoting formation of ecircRNA. (C) Base-pairing-driven circularization. The downstream introns and upstream introns are paired depends on inverse-repeating/complementary sequences. Formation of ecircRNA/EIciRNA was derived from the introns are removed/retained. (D) Biosynthesis of ciRNA. Formation of ciRNAs mainly based on a 7-nt GU-rich element and an 11-nt C-rich element to escape debranching and exonucleolytic degradation. (E) Formation of tricRNA. tRNA splicing enzymes divide pre-tRNA into two parts: TricRNAs are generated by a 3′–5′ phosphodiester bond and the other part generates tRNAs.](https://jours.isi-science.com/imageXml.php?i=medscimonit-26-e923832-g001.jpg&idArt=923832&w=1000) Figure 1. Biogenesis of circular RNAs [23]. (A) Lariat-driven circularization. The 3′ hydroxyl of the upstream exon reacts with the 5′ phosphate of the downstream exon to form a covalent linkage, then producing a lariat including exons and introns. The 2′ hydroxyl of the 5′ intron interacts with the 5′ phosphate of the 3′-intron to form an ecircRNA following an interaction between the 3′ hydroxyl of the 3′ exon and the 5′ phosphate of the 5′ exon. (B) RNA-binding protein (RBP)-driven circularization. RBPs accelerate interaction of the downstream intron and upstream intron, thereby promoting formation of ecircRNA. (C) Base-pairing-driven circularization. The downstream introns and upstream introns are paired depends on inverse-repeating/complementary sequences. Formation of ecircRNA/EIciRNA was derived from the introns are removed/retained. (D) Biosynthesis of ciRNA. Formation of ciRNAs mainly based on a 7-nt GU-rich element and an 11-nt C-rich element to escape debranching and exonucleolytic degradation. (E) Formation of tricRNA. tRNA splicing enzymes divide pre-tRNA into two parts: TricRNAs are generated by a 3′–5′ phosphodiester bond and the other part generates tRNAs.
Figure 1. Biogenesis of circular RNAs [23]. (A) Lariat-driven circularization. The 3′ hydroxyl of the upstream exon reacts with the 5′ phosphate of the downstream exon to form a covalent linkage, then producing a lariat including exons and introns. The 2′ hydroxyl of the 5′ intron interacts with the 5′ phosphate of the 3′-intron to form an ecircRNA following an interaction between the 3′ hydroxyl of the 3′ exon and the 5′ phosphate of the 5′ exon. (B) RNA-binding protein (RBP)-driven circularization. RBPs accelerate interaction of the downstream intron and upstream intron, thereby promoting formation of ecircRNA. (C) Base-pairing-driven circularization. The downstream introns and upstream introns are paired depends on inverse-repeating/complementary sequences. Formation of ecircRNA/EIciRNA was derived from the introns are removed/retained. (D) Biosynthesis of ciRNA. Formation of ciRNAs mainly based on a 7-nt GU-rich element and an 11-nt C-rich element to escape debranching and exonucleolytic degradation. (E) Formation of tricRNA. tRNA splicing enzymes divide pre-tRNA into two parts: TricRNAs are generated by a 3′–5′ phosphodiester bond and the other part generates tRNAs. 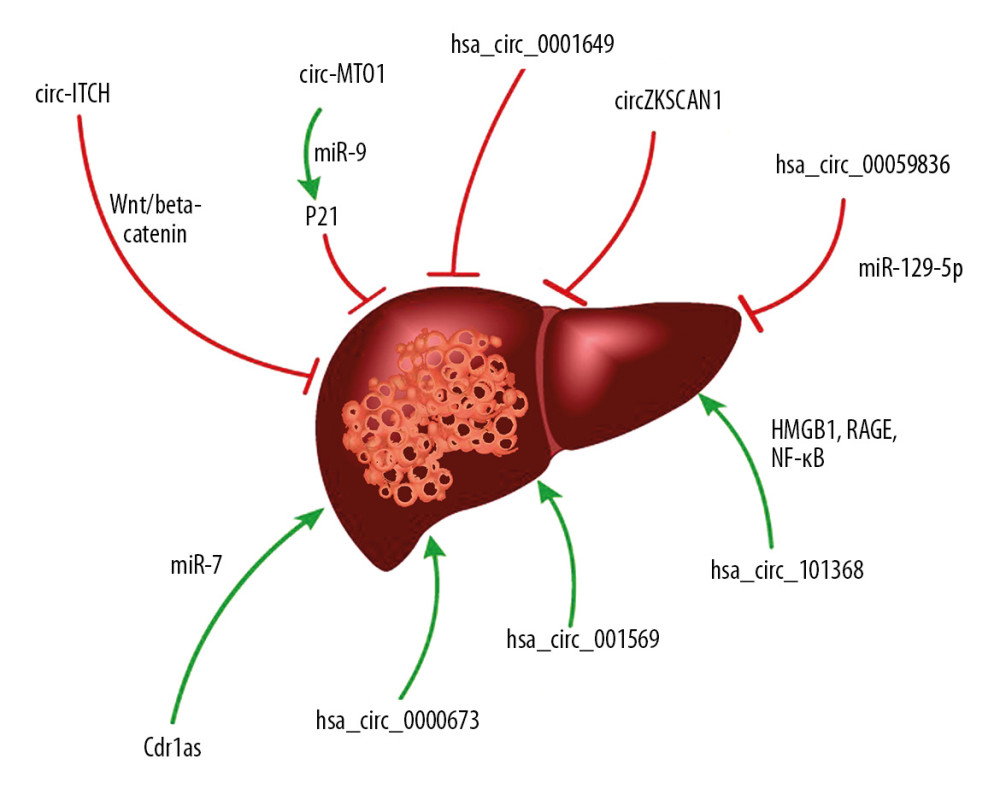 Figure 2. The function of circRNAs in HCC carcinogenesis. This graph demonstrates the role of circRNAs in HCC carcinogenesis, including positive and negative effects, respectively.
Figure 2. The function of circRNAs in HCC carcinogenesis. This graph demonstrates the role of circRNAs in HCC carcinogenesis, including positive and negative effects, respectively. References
1. Bray F, Ferlay J, Soerjomataram I, Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries: Cancer J Clin, 2018; 68(6); 394-424
2. Feng RM, Zong YN, Cao SM, Xu RH, Current cancer situation in China: Good or bad news from the 2018 Global Cancer Statistics?: Cancer Commun (Lond), 2019; 39(1); 22
3. Jemal A, Bray F, Center MM, Global cancer statistics: Cancer J Clin, 2011; 61(2); 69-90
4. Li R, Jiang J, Shi H, CircRNA: A rising star in gastric cancer: Cell Mol Life Sci, 2020; 77(9); 1661-80
5. Salzman J, Gawad C, Wang PL, Circular RNAs are the predominant transcript isoform from hundreds of human genes in diverse cell types: PLoS One, 2012; 7(2); e30733
6. Lukiw WJ, Circular RNA (circRNA) in Alzheimer’s disease (AD): Front Genet, 2013; 4; 307
7. Liu Y, Yang Y, Wang Z, Insights into the regulatory role of circRNA in angiogenesis and clinical implications: Atherosclerosis, 2020; 298; 14-26
8. Zhao Y, Alexandrov PN, Jaber V, Lukiw WJ, Deficiency in the ubiquitin conjugating enzyme UBE2A in Alzheimer’s disease (AD) is linked to deficits in a natural circular miRNA-7 sponge (circRNA; ciRS-7): Genes (Basel), 2016; 7(12); 12
9. Shen F, Liu P, Xu Z, CircRNA_001569 promotes cell proliferation through absorbing miR–145 in gastric cancer: J Biochem, 2019; 165(1); 27-36
10. Song T, Xu A, Zhang Z, CircRNA hsa_circRNA_101996 increases cervical cancer proliferation and invasion through activating TPX2 expression by restraining miR-8075: J Cell Physiol, 2019; 234(8); 14296-305
11. Min L, Wang H, Zeng Y, CircRNA_104916 regulates migration, apoptosis and epithelial–mesenchymal transition in colon cancer cells: Front Biosci (Landmark Ed), 2019; 24; 819-32
12. Verduci L, Strano S, Yarden Y, Blandino G, The circRNA-microRNA code: Emerging implications for cancer diagnosis and treatment: Mol Oncol, 2019; 13(4); 669-80
13. Wei J, Wei W, Xu H, Circular RNA hsa_circRNA_102958 may serve as a diagnostic marker for gastric cancer: Cancer Biomark, 2020; 27(2); 139-45
14. Li P, Chen S, Chen H, Using circular RNA as a novel type of biomarker in the screening of gastric cancer: Clin Chim Acta, 2015; 444; 132-36
15. Xin J, Zhang XY, Sun DK, Up-regulated circular RNA hsa_circ_0067934 contributes to glioblastoma progression through activating PI3K-AKT pathway: Eur Rev Med Pharmacol Sci, 2019; 23(8); 3447-54
16. Kong Z, Wan X, Zhang Y, Androgen-responsive circular RNA circSMARCA5 is up-regulated and promotes cell proliferation in prostate cancer: Biochem Biophys Res Commun, 2017; 493(3); 1217-23
17. Fu L, Chen Q, Yao T, Hsa_circ_0005986 inhibits carcinogenesis by acting as a miR-129-5p sponge and is used as a novel biomarker for hepatocellular carcinoma: Oncotarget, 2017; 8(27); 43878-88
18. Zhu X, Wang X, Wei S, hsa_circ_0013958: A circular RNA and potential novel biomarker for lung adenocarcinoma: FEBS J, 2017; 284(14); 2170-82
19. Zhang Q, Wang W, Zhou Q, Roles of circRNAs in the tumour microenvironment: Mol Cancer, 2020; 19(1); 14
20. Qu Z, Jiang C, Wu J, Ding Y, Exosomes as potent regulators of HCC malignancy and potential bio-tools in clinical application: Int J Clin Exp Med, 2015; 8(10); 17088-95
21. Memczak S, Jens M, Elefsinioti A, Circular RNAs are a large class of animal RNAs with regulatory potency: Nature, 2013; 495(7441); 333-38
22. Cocquerelle C, Mascrez B, Hetuin D, Bailleul B, Mis-splicing yields circular RNA molecules: FASEB J, 1993; 7(1); 155-60
23. Zhao X, Cai Y, Xu J, Circular RNAs: Biogenesis, mechanism, and function in human cancers: Int J Mol Sci, 2019; 20(16); 3926
24. Qu S, Yang X, Li X, Circular RNA: A new star of noncoding RNAs: Cancer Lett, 2015; 365(2); 141-48
25. Bahn JH, Zhang Q, Li F, The landscape of microRNA, Piwi-interacting RNA, and circular RNA in human saliva: Clin Chem, 2015; 61(1); 221-30
26. Hsu MT, Coca-Prados M, Electron microscopic evidence for the circular form of RNA in the cytoplasm of eukaryotic cells: Nature, 1979; 280(5720); 339-40
27. Yu X, Odenthal M, Fries JW, Exosomes as miRNA carriers: Formation-function-future: Int J Mol Sci, 2016; 17(12); 2028
28. Hanan M, Soreq H, Kadener S, CircRNAs in the brain: RNA Biol, 2017; 14(8); 1028-34
29. Constantin L, Circular RNAs and neuronal development: Adv Exp Med Biol, 2018; 1087; 205-13
30. van Rossum D, Verheijen BM, Pasterkamp RJ, Circular RNAs: Novel regulators of neuronal development: Front Mol Neurosci, 2016; 9; 74
31. Ebert MS, Sharp PA, MicroRNA sponges: Progress and possibilities: RNA, 2010; 16(11); 2043-50
32. Ebert MS, Neilson JR, Sharp PA, MicroRNA sponges: Competitive inhibitors of small RNAs in mammalian cells: Nat Methods, 2007; 4(9); 721-6
33. Hansen TB, Jensen TI, Clausen BH, Natural RNA circles function as efficient microRNA sponges: Nature, 2013; 495(7441); 384-88
34. Hsiao KY, Lin YC, Gupta SK, Noncoding effects of circular RNA CCDC66 promote colon cancer growth and metastasis: Cancer Res, 2017; 77(9); 2339-50
35. Janga SC, Mittal N, Construction, structure and dynamics of post-transcriptional regulatory network directed by RNA-binding proteins: Adv Exp Med Biol, 2011; 722; 103-17
36. Du WW, Yang W, Liu E, Foxo3 circular RNA retards cell cycle progression via forming ternary complexes with p21 and CDK2: Nucleic Acids Res, 2016; 44(6); 2846-58
37. Xia P, Wang S, Ye B, A Circular RNA protects dormant hematopoietic stem cells from DNA sensor cGAS-mediated exhaustion: Immunity, 2018; 48(4); 688-701.e7
38. Li Z, Huang C, Bao C, Exon-intron circular RNAs regulate transcription in the nucleus: Nat Struct Mol Biol, 2015; 22(3); 256-64
39. Yang Y, Fan X, Mao M, Extensive translation of circular RNAs driven by N(6)-methyladenosine: Cell Res, 2017; 27(5); 626-41
40. Yang Y, Gao X, Zhang M, Novel role of FBXW7 circular RNA in repressing glioma tumorigenesis: J Natl Cancer Inst, 2018; 110(3); 304-15
41. Abe N, Hiroshima M, Maruyama H, Rolling circle amplification in a prokaryotic translation system using small circular RNA: Angew Chem Int Ed Engl, 2013; 52(27); 7004-8
42. Zheng X, Chen L, Zhou Y, A novel protein encoded by a circular RNA circPPP1R12A promotes tumor pathogenesis and metastasis of colon cancer via Hippo–YAP signaling: Mol Cancer, 2019; 18(1); 47
43. Holdt LM, Stahringer A, Sass K, Circular non-coding RNA ANRIL modulates ribosomal RNA maturation and atherosclerosis in humans: Nat Commun, 2016; 7; 12429
44. Gokul S, Rajanikant GK, Circular RNAs in brain physiology and disease: Adv Exp Med Biol, 2018; 1087; 231-37
45. Idda ML, Munk R, Abdelmohsen K, Gorospe M, Noncoding RNAs in Alzheimer’s disease: Wiley Interdiscip Rev RNA, 2018; 9(2); 10
46. Luo Q, Chen Y, Long noncoding RNAs and Alzheimer’s disease: Clin Interv Aging, 2016; 11; 867-72
47. Li CY, Ma L, Yu B, Circular RNA hsa_circ_0003575 regulates oxLDL induced vascular endothelial cells proliferation and angiogenesis: Biomed Pharmacother, 2017; 95; 1514-19
48. Chen J, Cui L, Yuan J, Circular RNA WDR77 target FGF-2 to regulate vascular smooth muscle cells proliferation and migration by sponging miR-124: Biochem Biophys Res Commun, 2017; 494(1–2); 126-32
49. Kristensen LS, Hansen TB, Veno MT, Kjems J, Circular RNAs in cancer: Opportunities and challenges in the field: Oncogene, 2018; 37(5); 555-65
50. Fu L, Yao T, Chen Q, Screening differential circular RNA expression profiles reveals hsa_circ_0004018 is associated with hepatocellular carcinoma: Oncotarget, 2017; 8(35); 58405-16
51. Massarweh NN, El-Serag HB, Epidemiology of hepatocellular carcinoma and intrahepatic cholangiocarcinoma: Cancer Control, 2017; 24(3) 1073274817729245
52. Salem R, Gilbertsen M, Butt Z, Increased quality of life among hepatocellular carcinoma patients treated with radioembolization, compared with chemoembolization: Clin Gastroenterol Hepatol, 2013; 11(10); 1358-65.e1
53. Ozer ED, Suna N, Boyacioglu AS, Management of hepatocellular carcinoma: Prevention, surveillance, diagnosis, and staging: Exp Clin Transplant, 2017; 15(Suppl 2); 31-35
54. Lou W, Liu J, Ding B, Identification of potential miRNA-mRNA regulatory network contributing to pathogenesis of HBV-related HCC: J Transl Med, 2019; 17(1); 7
55. Yang T, Hu LY, Li ZL, Liver resection for hepatocellular carcinoma in non-alcoholic fatty liver disease: A multicenter propensity matching analysis with HBV-HCC: J Gastrointest Surg, 2020; 24(2); 320-29
56. Nishibatake KM, Minami T, Tateishi R, Impact of direct-acting antivirals on early recurrence of HCV-related HCC: Comparison with interferon-based therapy: J Hepatol, 2019; 70(1); 78-86
57. Toyoda H, Kumada T, Tada T, The impact of HCV eradication by direct-acting antivirals on the transition of precancerous hepatic nodules to HCC: A prospective observational study: Liver Int, 2019; 39(3); 448-54
58. Zhao J, O’Neil M, Vittal A, PRMT1-dependent macrophage IL-6 production is required for alcohol-induced HCC progression: Gene Expr, 2019; 19(2); 137-50
59. Vandenbulcke H, Moreno C, Colle I, Alcohol intake increases the risk of HCC in hepatitis C virus-related compensated cirrhosis: A prospective study: J Hepatol, 2016; 65(3); 543-51
60. Fabris C, Toniutto P, Falleti E, MTHFR C677T polymorphism and risk of HCC in patients with liver cirrhosis: Role of male gender and alcohol consumption: Alcohol Clin Exp Res, 2009; 33(1); 102-7
61. Vernon G, Baranova A, Younossi ZM, Systematic review: The epidemiology and natural history of non-alcoholic fatty liver disease and non-alcoholic steatohepatitis in adults: Aliment Pharmacol Ther, 2011; 34(3); 274-85
62. Bruix J, Reig M, Sherman M, Evidence-based diagnosis, staging, and treatment of patients with hepatocellular carcinoma: Gastroenterology, 2016; 150(4); 835-53
63. Zhang BH, Yang BH, Tang ZY, Randomized controlled trial of screening for hepatocellular carcinoma: J Cancer Res Clin Oncol, 2004; 130(7); 417-22
64. Verduci L, Ferraiuolo M, Sacconi A, The oncogenic role of circPVT1 in head and neck squamous cell carcinoma is mediated through the mutant p53/YAP/TEAD transcription-competent complex: Genome Biol, 2017; 18(1); 237
65. Yu J, Xu QG, Wang ZG, Circular RNA cSMARCA5 inhibits growth and metastasis in hepatocellular carcinoma: J Hepatol, 2018; 68(6); 1214-27
66. Wang M, Yu F, Li P, Circular RNAs: Characteristics, function and clinical significance in hepatocellular carcinoma: Cancers (Basel), 2018; 10(8); 258
67. Guo W, Zhang J, Zhang D, Polymorphisms and expression pattern of circular RNA circ-ITCH contributes to the carcinogenesis of hepatocellular carcinoma: Oncotarget, 2017; 8(29); 48169-77
68. Han D, Li J, Wang H, Circular RNA circMTO1 acts as the sponge of microRNA-9 to suppress hepatocellular carcinoma progression: Hepatology, 2017; 66(4); 1151-64
69. Qin M, Liu G, Huo X, Hsa_circ_0001649: A circular RNA and potential novel biomarker for hepatocellular carcinoma: Cancer Biomark, 2016; 16(1); 161-69
70. Yao Z, Luo J, Hu K, ZKSCAN1 gene and its related circular RNA (circZKSCAN1) both inhibit hepatocellular carcinoma cell growth, migration, and invasion but through different signaling pathways: Mol Oncol, 2017; 11(4); 422-37
71. Xu L, Zhang M, Zheng X, The circular RNA ciRS-7 (Cdr1as) acts as a risk factor of hepatic microvascular invasion in hepatocellular carcinoma: J Cancer Res Clin Oncol, 2017; 143(1); 17-27
72. Yu L, Gong X, Sun L, The circular RNA Cdr1as act as an oncogene in hepatocellular carcinoma through targeting miR-7 expression: PLoS One, 2016; 11(7); e0158347
73. Jiang W, Wen D, Gong L, Circular RNA hsa_circ_0000673 promotes hepatocellular carcinoma malignance by decreasing miR-767-3p targeting SET: Biochem Biophys Res Commun, 2018; 500(2); 211-16
74. Liu H, Xue L, Song C, Overexpression of circular RNA circ_001569 indicates poor prognosis in hepatocellular carcinoma and promotes cell growth and metastasis by sponging miR-411-5p and miR-432-5p: Biochem Biophys Res Commun, 2018; 503(4); 2659-65
75. Li S, Gu H, Huang Y, Circular RNA 101368/miR-200a axis modulates the migration of hepatocellular carcinoma through HMGB1/RAGE signaling: Cell Cycle, 2018; 17(19–20); 2349-59
76. Shang X, Li G, Liu H, Comprehensive circular RNA profiling reveals that hsa_circ_0005075, a new circular RNA biomarker, is involved in hepatocellular carcinoma development: Medicine (Baltimore), 2016; 95(22); e3811
77. Yao T, Chen Q, Shao Z, Circular RNA 0068669 as a new biomarker for hepatocellular carcinoma metastasis: J Clin Lab Anal, 2018; 32(8); e22572
Figures
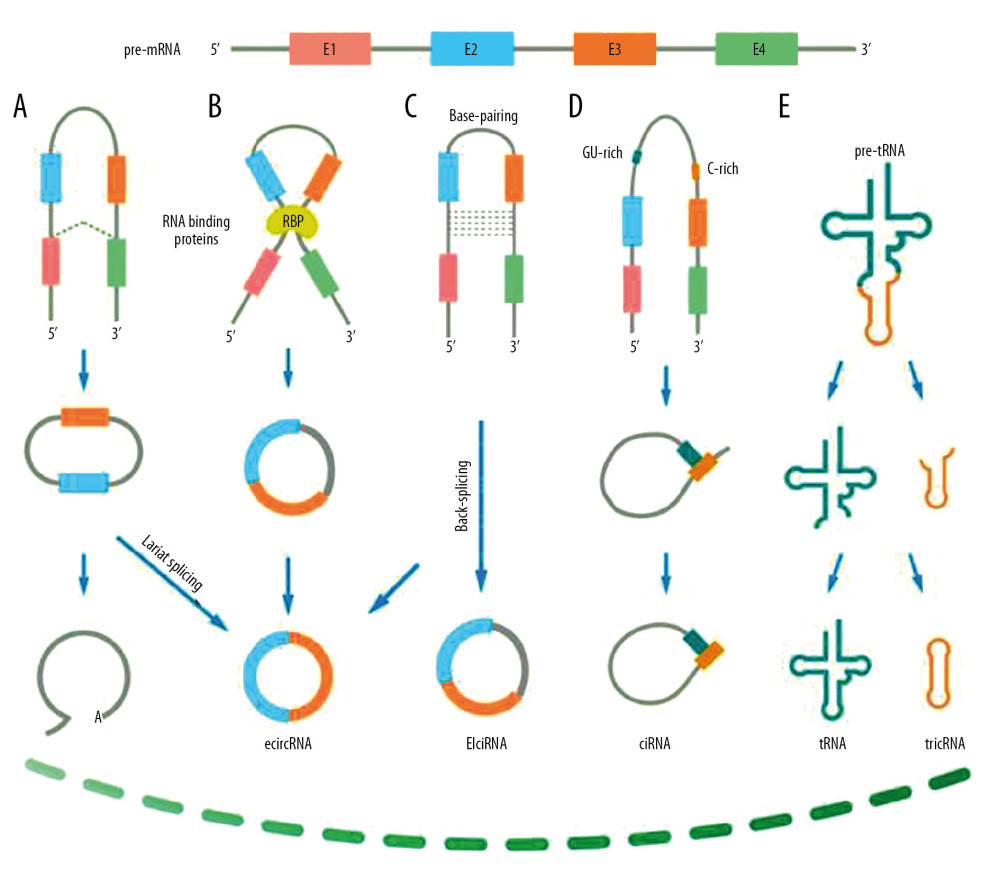 Figure 1. Biogenesis of circular RNAs [23]. (A) Lariat-driven circularization. The 3′ hydroxyl of the upstream exon reacts with the 5′ phosphate of the downstream exon to form a covalent linkage, then producing a lariat including exons and introns. The 2′ hydroxyl of the 5′ intron interacts with the 5′ phosphate of the 3′-intron to form an ecircRNA following an interaction between the 3′ hydroxyl of the 3′ exon and the 5′ phosphate of the 5′ exon. (B) RNA-binding protein (RBP)-driven circularization. RBPs accelerate interaction of the downstream intron and upstream intron, thereby promoting formation of ecircRNA. (C) Base-pairing-driven circularization. The downstream introns and upstream introns are paired depends on inverse-repeating/complementary sequences. Formation of ecircRNA/EIciRNA was derived from the introns are removed/retained. (D) Biosynthesis of ciRNA. Formation of ciRNAs mainly based on a 7-nt GU-rich element and an 11-nt C-rich element to escape debranching and exonucleolytic degradation. (E) Formation of tricRNA. tRNA splicing enzymes divide pre-tRNA into two parts: TricRNAs are generated by a 3′–5′ phosphodiester bond and the other part generates tRNAs.
Figure 1. Biogenesis of circular RNAs [23]. (A) Lariat-driven circularization. The 3′ hydroxyl of the upstream exon reacts with the 5′ phosphate of the downstream exon to form a covalent linkage, then producing a lariat including exons and introns. The 2′ hydroxyl of the 5′ intron interacts with the 5′ phosphate of the 3′-intron to form an ecircRNA following an interaction between the 3′ hydroxyl of the 3′ exon and the 5′ phosphate of the 5′ exon. (B) RNA-binding protein (RBP)-driven circularization. RBPs accelerate interaction of the downstream intron and upstream intron, thereby promoting formation of ecircRNA. (C) Base-pairing-driven circularization. The downstream introns and upstream introns are paired depends on inverse-repeating/complementary sequences. Formation of ecircRNA/EIciRNA was derived from the introns are removed/retained. (D) Biosynthesis of ciRNA. Formation of ciRNAs mainly based on a 7-nt GU-rich element and an 11-nt C-rich element to escape debranching and exonucleolytic degradation. (E) Formation of tricRNA. tRNA splicing enzymes divide pre-tRNA into two parts: TricRNAs are generated by a 3′–5′ phosphodiester bond and the other part generates tRNAs. Figure 2. The function of circRNAs in HCC carcinogenesis. This graph demonstrates the role of circRNAs in HCC carcinogenesis, including positive and negative effects, respectively.
Figure 2. The function of circRNAs in HCC carcinogenesis. This graph demonstrates the role of circRNAs in HCC carcinogenesis, including positive and negative effects, respectively. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387